Optimization of Extraction of Phlorotannins from the Arctic Fucus vesiculosus Using Natural Deep Eutectic Solvents and Their HPLC Profiling with Tandem High-Resolution Mass Spectrometry
Abstract
1. Introduction
2. Results and Discussion
2.1. The Ultrasound-Assisted Extraction of Phlorotannins from F. vesiculosus with NADES
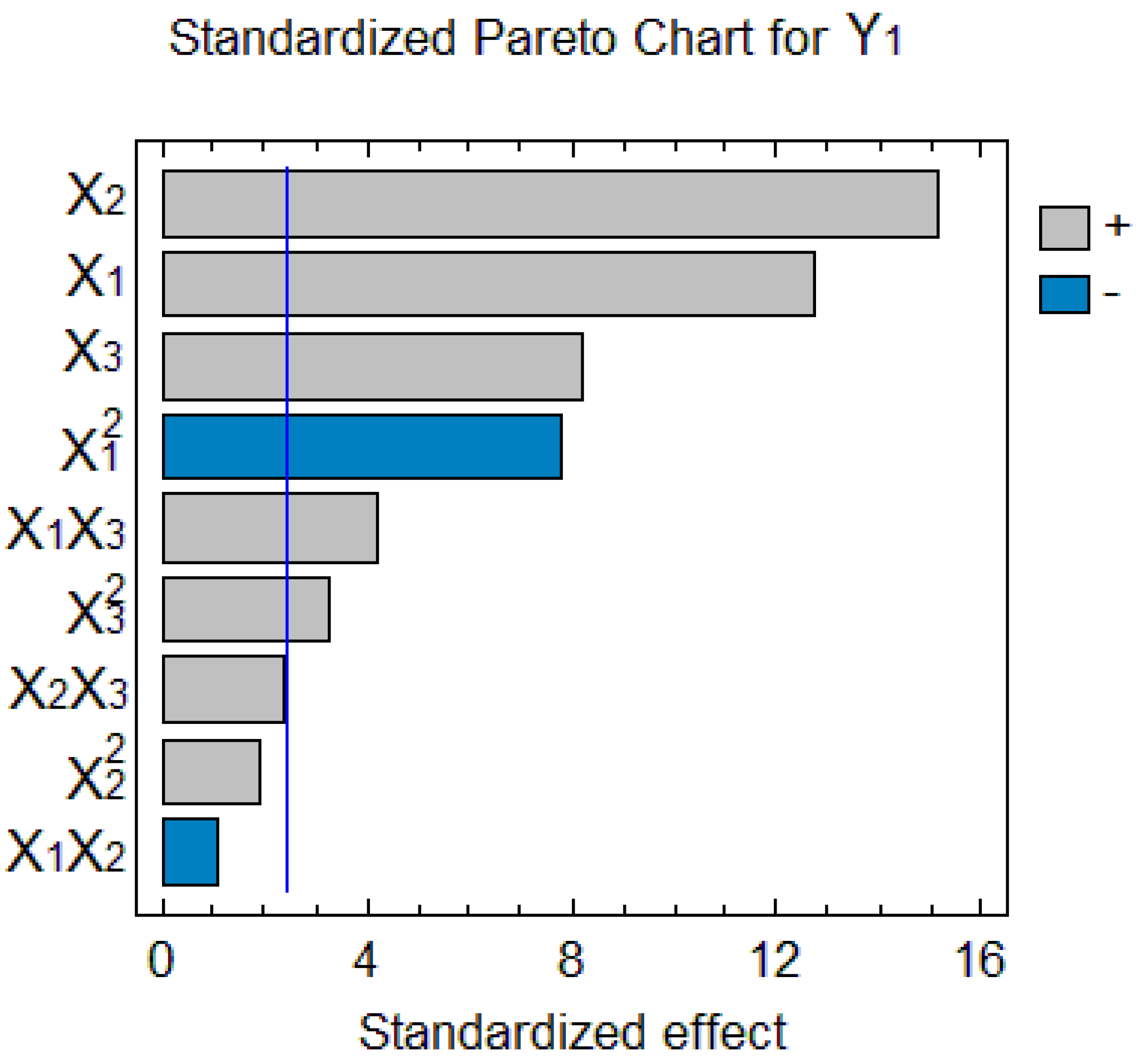
2.2. Model Adjustment and Analysis of Variance
2.3. Response Surface Methodology and Optimization
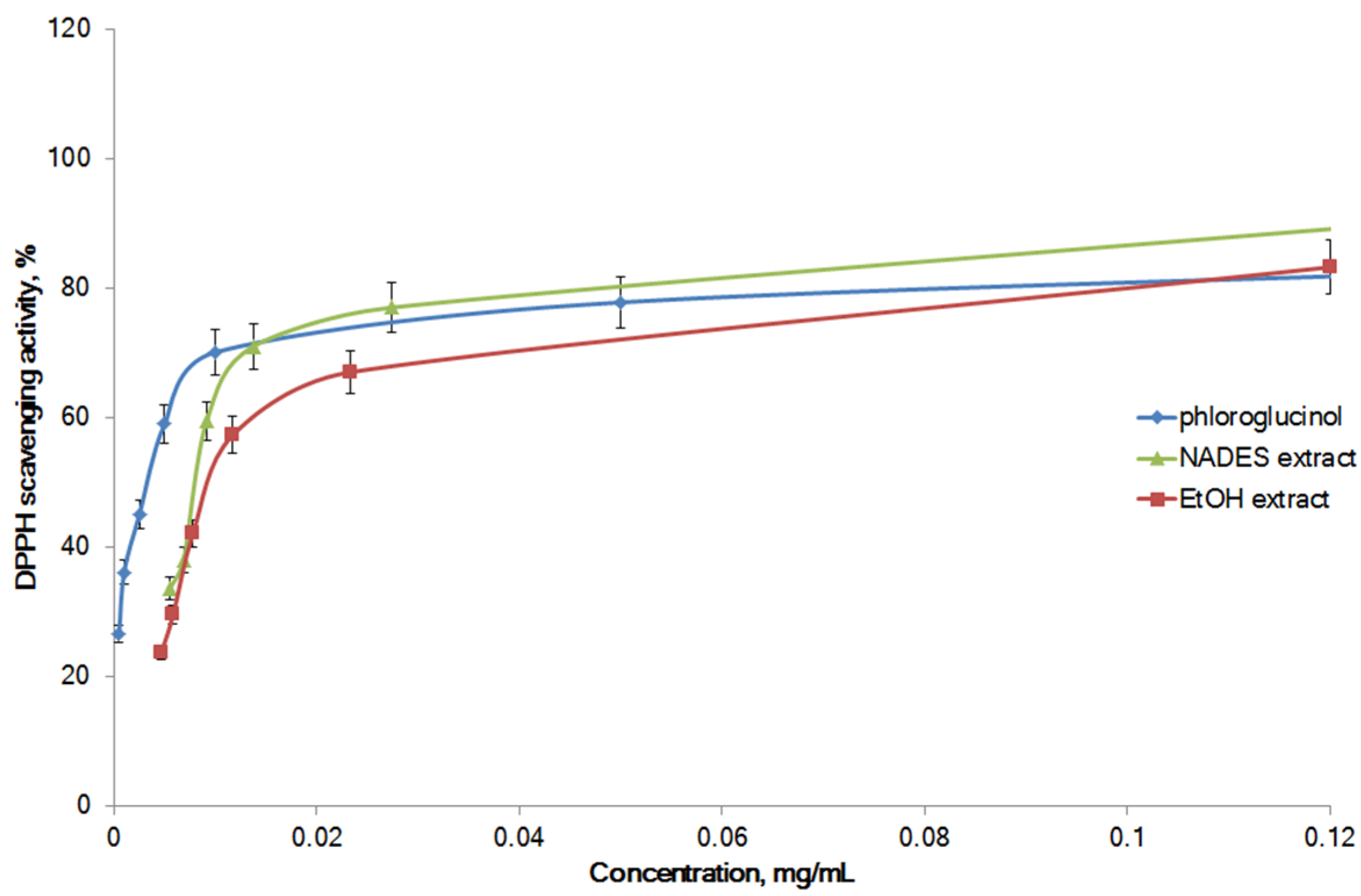
2.4. Antioxidant Activity of Optimized Extract
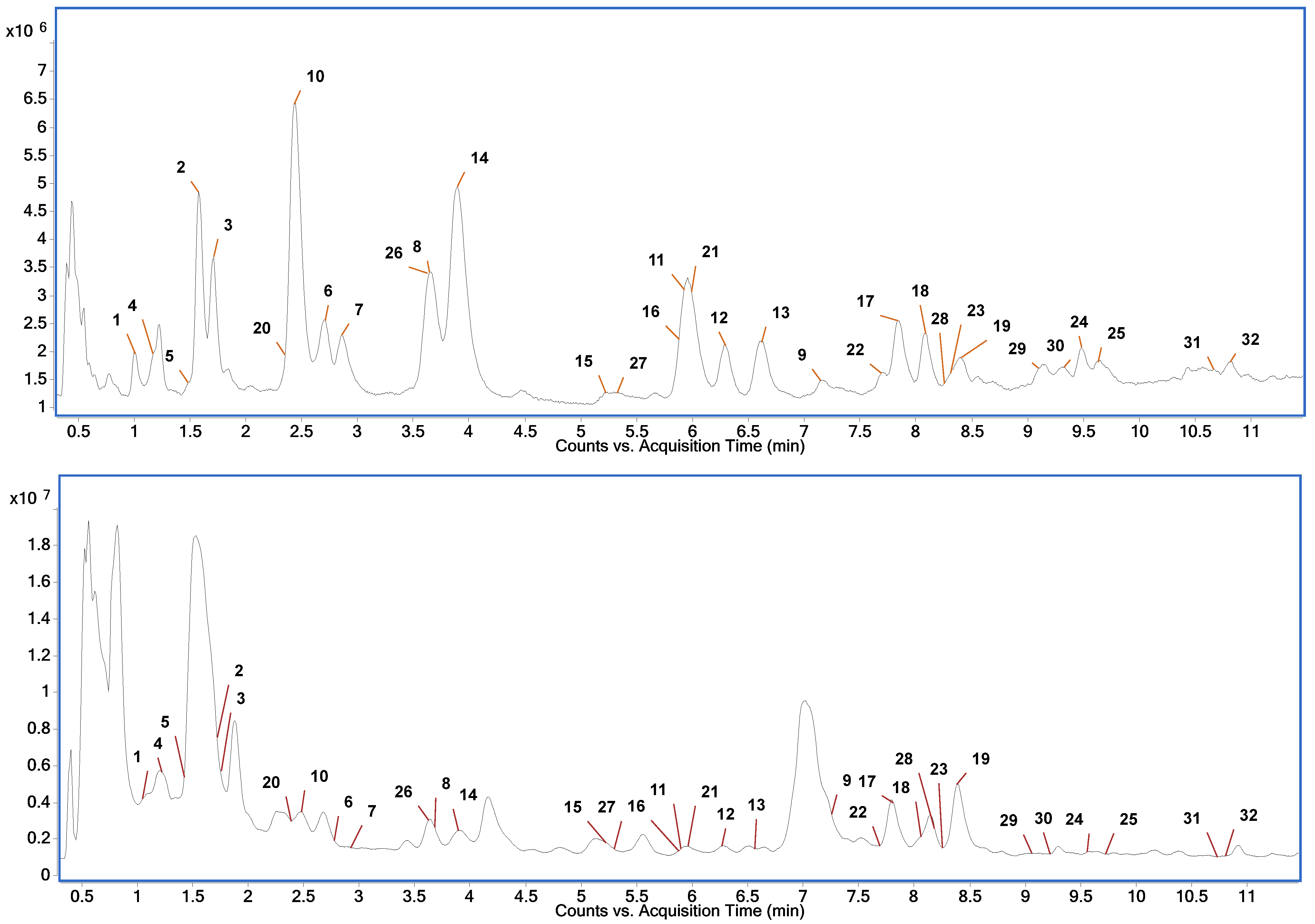
2.5. HPLC-HRMS Profiling of Phlorotannins in Extracts of F. vesiculosus
3. Materials and Methods
3.1. Materials
3.2. NADES Preparation
3.3. Extraction of Phlorotannins with NADES and EtOH
3.4. Experimental Design and Statistical Analysis
3.5. The Phlorotannin Content
3.6. The Antioxidant Activity
3.7. HPLC-HRMS Analysis
3.7.1. Sample Preparation
3.7.2. HPLC-HRMS and MS/MS Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Obluchinskaya, E.D. Biologically active substances of brown algae (Phaeophyta): Composition and pharmacological properties. Pharmacy 2014, 4, 49–51. [Google Scholar]
- Generalić Mekinić, I.; Skroza, D.; Šimat, V.; Hamed, I.; Čagalj, M.; Popović Perković, Z. Phenolic content of brown algae (Pheophyceae) species: Extraction, identification, and quantification. Biomolecules 2019, 9, 244. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Lopez, C.; Pereira, A.G.; Lourenço-Lopes, C.; Garcia-Oliveira, P.; Cassani, L.; Fraga-Corral, M.; Prieto, M.A.; Simal-Gandara, J. Main bioactive phenolic compounds in marine algae and their mechanisms of action supporting potential health benefits. Food Chem. 2021, 341, 128262. [Google Scholar] [CrossRef] [PubMed]
- Meng, W.; Mu, T.; Sun, H.; Garcia-Vaquero, M. Phlorotannins: A review of extraction methods, structural characteristics, bioactivities, bioavailability, and future trends. Algal Res. 2021, 60, 102484. [Google Scholar] [CrossRef]
- Khan, F.; Jeong, G.-J.; Khan, M.S.A.; Tabassum, N.; Kim, Y.-M. Seaweed-Derived phlorotannins: A review of multiple biological roles and action mechanisms. Mar. Drugs 2022, 20, 384. [Google Scholar] [CrossRef]
- Fernando, I.P.S.; Lee, W.; Ahn, G. Marine algal flavonoids and phlorotannins; an intriguing frontier of biofunctional secondary metabolites. Crit. Rev. Biotechnol. 2022, 42, 23–45. [Google Scholar] [CrossRef]
- Maheswari, V.; Babu, P.A. Phlorotannin and its derivatives, a potential antiviral molecule from brown seaweeds, an overview. Russ. J. Mar. Biol. 2022, 48, 309–324. [Google Scholar] [CrossRef]
- Pradhan, B.; Nayak, R.; Bhuyan, P.P.; Patra, S.; Behera, C.; Sahoo, S.; Ki, J.-S.; Quarta, A.; Ragusa, A.; Jena, M. Algal phlorotannins as novel antibacterial agents with reference to the antioxidant modulation: Current advances and future directions. Mar. Drugs 2022, 20, 403. [Google Scholar] [CrossRef]
- Wang, T.; Jónsdóttir, R.; Liu, H.; Gu, L.; Kristinsson, H.G.; Raghavan, S.; Ólafsdóttir, G. Antioxidant capacities of phlorotannins extracted from the brown algae Fucus vesiculosus. J. Agric. Food Chem. 2012, 60, 5874–5883. [Google Scholar] [CrossRef]
- Wang, C.; Li, X.; Jin, L.; Zhao, Y.; Zhu, G.; Shen, W. Dieckol inhibits non-small–cell lung cancer cell proliferation and migration by regulating the PI3K/AKT signaling pathway. J. Biochem. Mol. Toxicol. 2019, 33, e22346. [Google Scholar] [CrossRef]
- Kim, A.R.; Shin, T.S.; Lee, M.S.; Park, J.Y.; Park, K.E.; Yoon, N.Y.; Kim, J.S.; Choi, J.S.; Jang, B.C.; Byun, D.S.; et al. Isolation and identification of phlorotannins from Ecklonia stolonifera with antioxidant and anti-inflammatory properties. J. Agric. Food Chem. 2009, 57, 3483–3489. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Amarsanaa, K.; Lee, J.H.; Rhim, J.K.; Kwon, J.M.; Kim, S.H.; Park, J.M.; Jung, S.C.; Eun, S.Y. Neuroprotective mechanisms of dieckol against glutamate toxicity through reactive oxygen species scavenging and nuclear factor-like 2/heme oxygenase-1 pathway. Korean J. Physiol. Pharmacol. 2019, 23, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Je, J.-G.; Yang, H.-W.; Jeon, Y.-J.; Lee, S. Dieckol, an algae-derived phenolic compound, suppresses UVB-induced skin damage in human dermal fibroblasts and its underlying mechanisms. Antioxidants 2021, 10, 352. [Google Scholar] [CrossRef] [PubMed]
- Soleimani, S.; Yousefzadi, M.; Babaei Mahani Nezhad, S.; Pozharitskaya, O.N.; Shikov, A.N. Potential of the ethyl acetate fraction of Padina boergesenii as a natural UV filter in sunscreen cream formulation. Life 2023, 13, 239. [Google Scholar] [CrossRef] [PubMed]
- Hermund, D.B.; Torsteinsen, H.; Vega, J.; Figueroa, F.L.; Jacobsen, C. Screening for new cosmeceuticals from brown algae Fucus vesiculosus with antioxidant and photo-protecting properties. Mar. Drugs 2022, 20, 687. [Google Scholar] [CrossRef] [PubMed]
- Catarino, M.D.; Pires, S.M.G.; Silva, S.; Costa, F.; Braga, S.S.; Pinto, D.C.G.A.; Silva, A.M.S.; Cardoso, S.M. Overview of phlorotannins’ constituents in Fucales. Mar. Drugs 2022, 20, 754. [Google Scholar] [CrossRef]
- Zayed, A.; Ulber, R. Fucoidans: Downstream processes and recent applications. Mar. Drugs 2020, 18, 170. [Google Scholar] [CrossRef]
- Zheng, H.; Zhao, Y.; Guo, L. A Bioactive substance derived from brown seaweeds: Phlorotannins. Mar. Drugs 2022, 20, 742. [Google Scholar] [CrossRef]
- Martinez, J.H.; Torres Castañeda, H.G. Preparation and chromatographic analysis of phlorotannins. J. Chromatogr. Sci. 2013, 51, 825–838. [Google Scholar] [CrossRef]
- Cunha, S.C.; Fernandes, J.O. Extraction techniques with deep eutectic solvents. TrAC Trends Anal. Chem. 2018, 105, 225–239. [Google Scholar] [CrossRef]
- Li, Y.; Fu, X.; Duan, D.; Liu, X.; Xu, J.; Gao, X. Extraction and identification of phlorotannins from the brown alga, Sargassum fusiforme (Harvey) Setchell. Mar. Drugs 2017, 15, 49. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Yuen, A.K.L.; Magnusson, M.; Wright, J.T.; de Nys, R.; Masters, A.F.; Maschmeyer, T. A comparative assessment of the activity and structure of phlorotannins from the brown seaweed Carpophyllum flexuosum. Algal Res. 2018, 29, 130–141. [Google Scholar] [CrossRef]
- Heavisides, E.; Rouger, C.; Reichel, A.F.; Ulrich, C.; Wenzel-Storjohann, A.; Sebens, S.; Tasdemir, D. Seasonal variations in the metabolome and bioactivity profile of Fucus vesiculosus extracted by an optimized, pressurized liquid extraction protocol. Mar. Drugs 2018, 16, 503. [Google Scholar] [CrossRef] [PubMed]
- Saravana, P.S.; Getachew, A.T.; Cho, Y.J.; Choi, J.H.; Park, Y.B.; Woo, H.C.; Chun, B.S. Influence of co-solvents on fucoxanthin and phlorotannin recovery from brown seaweed using supercritical CO2. J. Supercrit. Fluid. 2017, 120, 295–303. [Google Scholar] [CrossRef]
- Ummat, V.; Tiwari, B.K.; Jaiswal, A.K.; Condon, K.; Garcia-Vaquero, M.; O’Doherty, J.; O’Donnell, C.; Rajauria, G. Optimisation of ultrasound frequency, extraction time and solvent for the recovery of polyphenols, phlorotannins and associated antioxidant activity from brown seaweeds. Mar. Drugs 2020, 18, 250. [Google Scholar] [CrossRef]
- Chemat, F.; Abert-Vian, M.; Cravotto, G. Green extraction of natural products: Concept and principles. Int. J. Mol. Sci. 2012, 13, 8615–8627. [Google Scholar] [CrossRef]
- Chemat, F.; Rombaut, N.; Sicaire, A.G.; Meullemiestre, A.; Fabiano-Tixier, A.S.; Abert-Vian, M. Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications. A review. Ultrason. Sonochem. 2017, 34, 540–560. [Google Scholar] [CrossRef]
- Ojha, K.S.; Aznar, R.; O’Donnell, C.; Tiwari, B.K. Ultrasound technology for the extraction of biologically active molecules from plant, animal and marine sources. TrAC Trends Anal. Chem. 2020, 122, 115663. [Google Scholar] [CrossRef]
- Kumar, K.; Srivastav, S.; Sharanagat, V.S. Ultrasound assisted extraction (UAE) of bioactive compounds from fruit and vegetable processing by-products: A review. Ultrason. Sonochem. 2021, 70, 105325. [Google Scholar] [CrossRef]
- Dang, T.T.; Van Vuong, Q.; Schreider, M.J.; Bowyer, M.C.; Van Altena, I.A.; Scarlett, C.J. Optimisation of ultrasound-assisted extraction conditions for phenolic content and antioxidant activities of the alga Hormosira banksii using response surface methodology. J. Appl. Phycol. 2017, 29, 3161–3173. [Google Scholar] [CrossRef]
- Hassan, I.H.; Pham, H.N.T.; Nguyen, T.H. Optimization of ultrasound-assisted extraction conditions for phenolics, antioxidant, and tyrosinase inhibitory activities of Vietnamese brown seaweed (Padina australis). J. Food Process. Preserv. 2021, 45, e15386. [Google Scholar] [CrossRef]
- Vázquez-Rodríguez, B.; Gutiérrez-Uribe, J.A.; Antunes-Ricardo, M.; Santos-Zea, L.; Cruz-Suárez, L.E. Ultrasound-assisted extraction of phlorotannins and polysaccharides from Silvetia compressa (Phaeophyceae). J. Appl. Phycol. 2020, 32, 1441–1453. [Google Scholar] [CrossRef]
- Kadam, S.U.; Tiwari, B.K.; Smyth, T.J.; O’Donnell, C.P. Optimization of ultrasound assisted extraction of bioactive components from brown seaweed Ascophyllum nodosum using response surface methodology. Ultrason. Sonochem. 2015, 23, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Obluchinsksya, E.D.; Makarova, M.N.; Pozharitskaya, O.N.; Shikov, A.N. Effects of ultrasound treatment on the chemical composition and anticoagulant properties of dry fucus extract. Pharm. Chem. J. 2015, 49, 183–186. [Google Scholar] [CrossRef]
- Santana-Mayor, Á.; Rodríguez-Ramos, R.; Herrera-Herrera, A.V.; Socas-Rodríguez, B.; Rodríguez-Delgado, M.Á. Deep eutectic solvents. The new generation of green solvents in analytical chemistry. TrAC Trends Anal. Chem. 2021, 134, 116108. [Google Scholar] [CrossRef]
- Li, X.; Row, K.H. Development of deep eutectic solvents applied in extraction and separation. J. Sep. Sci. 2016, 39, 3505–3520. [Google Scholar] [CrossRef]
- Vanda, H.; Dai, Y.; Wilson, E.G.; Verpoorte, R.; Choi, Y.H. Green solvents from ionic liquids and deep eutectic solvents to natural deep eutectic solvents. Comptes Rendus Chim. 2018, 21, 628–638. [Google Scholar] [CrossRef]
- Pavlić, B.; Mrkonjić, Ž.; Teslić, N.; Kljakić, A.C.; Pojić, M.; Mandić, A.; Stupar, A.; Santos, F.; Duarte, A.R.C.; Mišan, A. Natural Deep Eutectic Solvent (NADES) extraction improves polyphenol yield and antioxidant activity of wild thyme (Thymus serpyllum L.) extracts. Molecules 2022, 27, 1508. [Google Scholar] [CrossRef]
- Rebocho, S.; Mano, F.; Cassel, E.; Anacleto, B.; do Rosário Bronze, M.; Paiva, A.; Duarte, A.R.C. Fractionated extraction of polyphenols from mate tea leaves using a combination of hydrophobic/hydrophilic NADES. Curr. Res. Nutr. Food Sci. 2022, 5, 571–580. [Google Scholar] [CrossRef]
- Wu, L.; Li, L.; Chen, S.; Wang, L.; Lin, X. Deep eutectic solvent-based ultrasonic-assisted extraction of phenolic compounds from Moringa oleifera L. leaves: Optimization, comparison and antioxidant activity. Sep. Purif. Technol. 2020, 247, 117014. [Google Scholar] [CrossRef]
- Alasalvar, H.; Yildirim, Z. Ultrasound-assisted extraction of antioxidant phenolic compounds from Lavandula angustifolia flowers using natural deep eutectic solvents: An experimental design approach. Sustain. Chem. Pharm. 2021, 22, 100492. [Google Scholar] [CrossRef]
- Wen, C.; Zhang, J.; Zhang, H.; Dzah, C.S.; Zandile, M.; Duan, Y.; Luo, X. Advances in ultrasound assisted extraction of bioactive compounds from cash crops—A review. Ultrason. Sonochem. 2018, 48, 538–549. [Google Scholar] [CrossRef] [PubMed]
- Myers, R.H.; Montgomery, D.C.; Anderson-Cook, C.M. (Eds.) Response Surface Methodology: Process and Product Optimization Using Designed Experiments, 4th ed.; Wiley Series in Probability and Statistics; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2016; 856p. [Google Scholar]
- Obluchinskaya, E.D.; Daurtseva, A.V.; Pozharitskaya, O.N.; Flisyuk, E.V.; Shikov, A.N. Natural Deep Eutectic Solvents as alternatives for extracting phlorotannins from brown algae. Pharm. Chem. J. 2019, 53, 243–247. [Google Scholar] [CrossRef]
- Wojnowski, W.; Tobiszewski, M.; Pena-Pereira, F.; Psillakis, E. AGREEprep–Analytical greenness metric for sample preparation. TrAC Trends Anal. Chem. 2022, 149, 116553. [Google Scholar] [CrossRef]
- Shikov, A.N.; Obluchinskaya, E.D.; Flisyuk, E.V.; Terninko, I.I.; Generalova, Y.E.; Pozharitskaya, O.N. The impact of natural deep eutectic solvents and extraction method on the co-extraction of trace metals from Fucus vesiculosus. Mar. Drugs 2022, 20, 324. [Google Scholar] [CrossRef] [PubMed]
- Obluchinskaya, E.D.; Pozharitskaya, O.N.; Zakharova, L.V.; Daurtseva, A.V.; Flisyuk, E.V.; Shikov, A.N. Efficacy of natural deep eutectic solvents for extraction of hydrophilic and lipophilic compounds from Fucus vesiculosus. Molecules 2021, 26, 4198. [Google Scholar] [CrossRef]
- De Mello, B.T.F.; dos Santos Garcia, V.A.; da Silva, C. Ultrasound-Assisted extraction of oil from chia (Salvia hispânica L.) seeds: Optimization extraction and fatty acid profile. J. Food Process Eng. 2015, 40, e12298. [Google Scholar] [CrossRef]
- Dai, Y.; Van Spronsen, J.; Witkamp, G.J.; Verpoorte, R.; Choi, Y.H. Natural deep eutectic solvents as new potential media for green technology. Anal. Chim. Acta 2013, 766, 61–68. [Google Scholar] [CrossRef]
- Vieira, V.; Prieto, M.A.; Barros, L.; Coutinho, J.A.; Ferreira, I.C.; Ferreira, O. Enhanced extraction of phenolic compounds using choline chloride based deep eutectic solvents from Juglans regia L. Ind. Crops Prod. 2018, 115, 261–271. [Google Scholar] [CrossRef]
- Sumampouw, G.A.; Jacobsen, C.; Getachew, A.T. Optimization of phenolic antioxidants extraction from Fucus vesiculosus by pressurized liquid extraction. J. Appl. Phycol. 2021, 33, 1195–1207. [Google Scholar] [CrossRef]
- Catarino, M.D.; Silva, A.M.; Mateus, N.; Cardoso, S.M. Optimization of phlorotannins extraction from Fucus vesiculosus and evaluation of their potential to prevent metabolic disorders. Mar. Drugs 2019, 17, 162. [Google Scholar] [CrossRef] [PubMed]
- Toan, T.Q.; Phong, T.D.; Tien, D.D.; Linh, N.M.; Mai Anh, N.T.; Hong Minh, P.T.; Duy, L.X.; Nghi, D.H.; Pham Thi, H.H.; Tri Nhut, P.; et al. Optimization of microwave-assisted extraction of phlorotannin from Sargassum swartzii (Turn.) C. Ag. with ethanol/water. Nat. Prod. Commun. 2021, 16, 1934578X21996184. [Google Scholar] [CrossRef]
- Wang, T.; Jónsdóttir, R.; Ólafsdóttir, G. Total phenolic compounds, radical scavenging and metal chelation of extracts from icelandic seaweeds. Food Chem. 2009, 116, 240–248. [Google Scholar] [CrossRef]
- Ahn, G.N.; Kim, K.N.; Cha, S.H.; Song, C.B.; Lee, J.; Heo, M.S.; Yeo, I.K.; Lee, N.H.; Jee, Y.H.; Kim, J.S.; et al. Antioxidant activities of phlorotannins purified from Ecklonia cava on free radical scavenging using ESR and H2O2-mediated DNA damage. Eur. Food Res. Technol. 2007, 226, 71–79. [Google Scholar] [CrossRef]
- Prieto, P.; Pineda, M.; Aguilar, M. Spectrophotometric quantitation of antioxidant capacity through the formation of a phos-phomolybdenum complex: Specific application to the determination of vitamin E. Anal. Biochem. 1999, 269, 337–341. [Google Scholar] [CrossRef]
- Rukavina, I.; Rodrigues, M.J.; Pereira, C.G.; Mansinhos, I.; Romano, A.; Ślusarczyk, S.; Matkowski, A.; Custódio, L. Greener is better: First approach for the use of natural deep eutectic solvents (NADES) to extract antioxidants from the medicinal halophyte Polygonum maritimum L. Molecules 2021, 26, 6136. [Google Scholar] [CrossRef] [PubMed]
- Pal, C.B.T.; Jadeja, G.C. Deep eutectic solvent-based extraction of polyphenolic antioxidants from onion (Allium cepa L.) peel. J. Sci. Food Agric. 2019, 99, 1969–1979. [Google Scholar] [CrossRef]
- Vissers, A.M.; Caligiani, A.; Sforza, S.; Vincken, J.P.; Gruppen, H. Phlorotannin composition of Laminaria digitata. Phytochem. Anal. 2017, 28, 487–495. [Google Scholar] [CrossRef]
- Lopes, G.; Barbosa, M.; Vallejo, F.; Gil-Izquierdo, Á.; Andrade, P.B.; Valentão, P.; Pereira, D.M.; Ferreres, F. Profiling phlorotannins from Fucus spp. of the Northern Portuguese coastline: Chemical approach by HPLC-DAD-ESI/MS and UPLC-ESI-QTOF/MS. Algal Res. 2018, 29, 113–120. [Google Scholar] [CrossRef]
- Hermund, D.B.; Plaza, M.; Turner, C.; Jónsdóttir, R.; Kristinsson, H.G.; Jacobsen, C.; Nielsen, K.F. Structure dependent antioxidant capacity of phlorotannins from Icelandic Fucus vesiculosus by UHPLC-DAD-ECD-QTOFMS. Food Chem. 2018, 240, 904–909. [Google Scholar] [CrossRef]
- Erpel, F.; Camilo, C.; Mateos, R.; Pérez-Correa, J.R. A macroporous resin purification process to obtain food-grade phlorotannin-rich extracts with α-glucosidase inhibitory activity from Chilean brown seaweeds: An UHPLC-MS profiling. Food Chem. 2023, 402, 134472. [Google Scholar] [CrossRef]
- Montero, L.; Sánchez-Camargo, A.P.; García-Cañas, V.; Tanniou, A.; Stiger-Pouvreau, V.; Russo, M.; Rastrelli, L.; Cifuentes, A.; Herrero, M.; Ibáñez, E. Anti-proliferative activity and chemical characterization by comprehensive two-dimensional liquid chromatography coupled to mass spectrometry of phlorotannins from the brown macroalga Sargassum muticum collected on North-Atlantic coasts. J. Chromatogr. A 2016, 1428, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Ovchinnikov, D.V.; Bogolitsyn, K.G.; Druzhinina, A.S.; Kaplitsin, P.A.; Parshina, A.E.; Pikovskoi, I.I.; Khoroshev, O.Y.; Turova, P.N.; Stavrianidi, A.N.; Shpigun, O.A. Study of polyphenol components in extracts of arctic brown algae of Fucus vesiculosus type by liquid chromatography and mass-spectrometry. J. Anal. Chem. 2020, 75, 633–639. [Google Scholar] [CrossRef]
- Che Zain, M.S.; Yeoh, J.X.; Lee, S.Y.; Afzan, A.; Shaari, K. Integration of choline chloride-based natural deep eutectic solvents and macroporous resin for green production of enriched oil palm flavonoids as natural wound healing agents. Antioxidants 2021, 10, 1802. [Google Scholar] [CrossRef] [PubMed]
- Ford, L.; Theodoridou, K.; Sheldrake, G.N.; Walsh, P.J. A critical review of analytical methods used for the chemical characterisation and quantification of phlorotannin compounds in brown seaweeds. Phytochem. Anal. 2019, 30, 587–599. [Google Scholar] [CrossRef] [PubMed]
- Rajauria, G.; Foley, B.; Abu-Ghannam, N. Identification and characterization of phenolic antioxidant compounds from brown Irish seaweed Himanthalia elongata using LC-DAD–ESI-MS/MS. Innov. Food Sci. Emerg. Technol. 2016, 37, 261–268. [Google Scholar] [CrossRef]



| Factors | Levels | ||
|---|---|---|---|
| −1 | 0 | +1 | |
| Time (min), X1 | 10 | 20 | 30 |
| Water concentration in NADES (%), X2 | 30 | 40 | 50 |
| Sample to solvent ratio (g/g), X3 | 7.5 | 10.0 | 12.5 |
| Run | Independent Variables (Factors) | Total Phlorotannin Content (mg PhE/g DW) | |||
|---|---|---|---|---|---|
| X1 | X2 | X3 | Experimental | Predicted | |
| 1 | 1 | −1 | 0 | 54.2 ± 0.6 | 50.8 |
| 2 | 0 | 0 | 0 | 60.3 ± 2.9 | 60.2 |
| 3 | −1 | −1 | 0 | 22.0 ± 0.3 | 20.9 |
| 4 | 0 | −1 | −1 | 44.7 ± 2.8 | 47.1 |
| 5 | 1 | 0 | 1 | 80.4 ± 2.0 | 81.6 |
| 6 | 0 | 1 | −1 | 73.8 ± 4.6 | 71.7 |
| 7 | 0 | 0 | 0 | 60.4 ± 0.1 | 60.2 |
| 8 | −1 | 1 | 0 | 52.3 ± 2.8 | 55.8 |
| 9 | 1 | 0 | −1 | 51.1 ± 1.1 | 52.1 |
| 10 | −1 | 0 | 1 | 43.7 ± 3.2 | 42.6 |
| 11 | 1 | 1 | 0 | 78.1 ± 1.7 | 79.2 |
| 12 | 0 | 0 | 0 | 60.0 ± 3.6 | 60.2 |
| 13 | −1 | 0 | −1 | 39.2 ± 1.4 | 37.9 |
| 14 | 0 | 1 | 1 | 98.2 ± 2.9 | 95.8 |
| 15 | 0 | −1 | 1 | 55.0 ± 2.0 | 57.1 |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value |
|---|---|---|---|---|---|
| Model | 4864.01 | 9 | 540.446 | 51.86 | 0.0002 |
| X1 | 1416.98 | 1 | 1416.98 | 135.98 | 0.0001 |
| X2 | 1999.02 | 1 | 1999.02 | 191.84 | 0.0000 |
| X3 | 585.675 | 1 | 585.675 | 56.20 | 0.0007 |
| X1X2 | 10.3684 | 1 | 10.3684 | 1.00 | 0.3643 |
| X1X3 | 152.646 | 1 | 152.646 | 14.65 | 0.0123 |
| X2X3 | 50.1264 | 1 | 50.1264 | 4.81 | 0.0798 |
| 486.01 | 1 | 486.01 | 46.64 | 0.0010 | |
| 31.0434 | 1 | 31.0434 | 2.98 | 0.1449 | |
| 85.6774 | 1 | 85.6774 | 8.22 | 0.0351 |
| Optimal Parameters | Predicted Results (mg PhE/g DW) | Experimental Results (mg PhE/g DW) | ||
|---|---|---|---|---|
| Extraction Time (min) | Water Concentration (%) | Sample to Solvent Ratio (m/m) | ||
| 22.8 | 30.0 | 1.0:12.0 | 140.3 | 137.3 ± 2.9 |
| DP | Molecular Formula | No. | tR (min) | MS/MS (CID) Spectrum * | Tentative Identification |
|---|---|---|---|---|---|
| Trimer | C18H14O9 | 1 | 1.02 | 355.0455 (-H2O), 329.0659, 305.0663, 287.0560, 261.0770, 247.0229 (-1PGU), 243.0662, 231.0294 (-1PGU, -H2O, +2), 216.0057, 205.0500, 189.0553, 165.0191, 149.0240, 141.0189, 124.0163 | Fucophlorethol |
| Tetramer | C24H18O12 | 2 | 1.57 | 479.0613 (-H2O), 453.0817, 435.0708, 371.0401 (-1PGU), 353.0297 (-1PGU, -H2O), 339.0504, 335.0191 (-1PGU, -2H2O), 309.0397, 247.0240 (-2PGU, +2), 229.0145 (-2PGU, -H2O, +2), 219.0299, 139.0032, 125.0240 | Fucodiphlorethol |
| 3 | 1.71 | 479.0606 (-H2O), 453.0813, 371.0399 (-1PGU), 353.0298 (-1PGU, -H2O), 339.0504, 335.0195 (-1PGU, -2H2O), 313.0711, 309.0399, 265.0357, 247.0238 (-2PGU, +2), 229.0131 (-2PGU, -H2O, +2), 139.0030, 125.0232 | Fucodiphlorethol | ||
| Pentamer | C30H22O15 | 4 | 1.20 | 603.0763 (-H2O), 585.0653 (-2H2O), 577.0957, 559.0865, 495.0561 (-1PGU), 477.0453 (-1PGU, -H2O), 455.0612, 433.0554, 331.0452, 311.0196, 289.0346, 267.0299, 247.0241, 229.0136, 207.0291, 165.0190, 139.0030, 125.024 | Trifucophlorethol |
| 5 | 1.49 | 603.0769 (-H2O), 585.0662 (-2H2O), 577.0974, 559.0868, 535.0865, 495.0556 (-1PGU), 477.0455 (-1PGU, -H2O), 455.0618, 433.0545, 413.0497, 329.0294, 311.0185, 289.0358, 267.0293, 245.0092, 229.0130, 207.0301, 165.0190, 139.0032, 125.0234 | Trifucophlorethol | ||
| 6 | 2.74 | 603.0769 (-H2O), 585.0666 (-2H2O), 559.0870, 495.0558 (-1PGU), 477.0456 (-1PGU,-H2O), 461.0508, 371.0405 (-2PGU, +2), 353.0297 (-2PGU, -16/-2PGU,-H2O, +2), 335.0194 (-2PGU, -2H2O, +2), 309.0397, 249.0400, 229.0137 (-3PGU, -H2O, +4), 139.0033, 125.0240 | Fucotriphlorethol | ||
| 7 | 2.89 | 603.0777 (-H2O), 585.0641 (-2H2O), 559.0869, 495.0562 (-1PGU), 477.0454 (-1PGU, -H2O), 371.0397 (-2PGU, +2), 353.0297 (-2PGU, -16/-2PGU,-H2O, +2), 335.0190 (-2PGU, -2H2O, +2), 309.0399, 249.0397, 229.0138 (-3PGU, -H2O, +4), 139.0030, 125.0240 | Fucotriphlorethol | ||
| 8 | 3.68 | 603.0770 (-H2O), 585.0660 (-2H2O), 559.0867, 495.0565 (-1PGU), 477.0457 (-1PGU,-H2O), 371.0409 (-2PGU, +2), 353.0298 (-2PGU, -16/-2PGU, -H2O, +2), 339.0507, 337.0350, 335.0191 (-2PGU, -2H2O, +2), 245.0088 (-3PGU, +2), 229.0139 (-3PGU, -H2O, +4), 139.0032, 125.0241 | Fucotriphlorethol | ||
| 9 | 7.16 | 603.0753 (-H2O), 495.0552 (-1PGU), 477.0444 (-1PGU, -H2O), 459.0354 (-1PGU, -2H2O), 389.0505, 371.0403 (-2PGU, +2), 353.0300 (-2PGU, -16/-2PGU, -H2O, +2), 337.0337, 263.0190, 247.0240, 245.0087 (-3PGU, +2), 229.0140 (-3PGU, -H2O, +4), 139.0029, 125.0240 | Fucotriphlorethol | ||
| Hexamer | C36H26O18 | 10 | 2.45 | 727.0931 (-H2O), 709.0823 (-2H2O), 683.1031, 585.0669, 579.0768, 455.0615, 437.0511, 411.0714, 289.0348, 165.0189, 139.0032 | Hexafucol |
| 11 | 5.96 | 727.0933 (-H2O), 709.0825 (-2H2O), 619.0745 (-1PGU), 601.0626 (-1PGU, -H2O), 583.0513, 477.0456 (-2PGU, -16/-2PGU, -H2O, +2), 461.0522, 373.0557, 353.0295, 335.0189, 245.0086, 229.0134, 139.0034 | Fucotetraphlorethol | ||
| 12 | 6.31 | 727.0925 (-H2O), 709.0803 (-2H2O), 619.0707 (-1PGU), 477.0451 (-2PGU, -16/-2PGU, -H2O, +2), 371.0401, 353.0298, 337.0343, 247.0238, 229.0139, 139.0033 | Fucotetraphlorethol | ||
| 13 | 6.61 | 727.0924 (-H2O), 709.0797 (-2H2O), 601.0605 (-1PGU, -H2O), 583.0512, 477.0461 (-2PGU, -16/-2PGU, -H2O, +2), 337.0345, 245.0091, 229.0138, 139.0030 | Fucotetraphlorethol | ||
| Heptamer | C42H30O21 | 14 | 3.92 | 851.1090 (-H2O), 833.0983 (-2H2O), 708.0750, 703.0936, 579.0773, 455.0614, 437.0516, 413.0511, 289.0351, 269.0088, 165.0191, 139.0033, 125.0242 | Heptafucol |
| 15 | 5.26 | 851.1085 (-H2O), 833.0976 (-2H2O), 703.0940, 579.0783, 455.0601, 413.0508, 289.0356, 139.0030 | Heptafucol | ||
| 16 | 5.90 | 851.1086 (-H2O), 833.1003 (-2H2O), 579.0775, 289.0349, 139.0030 | Heptafucol | ||
| 17 | 7.86 | 851.1089 (-H2O), 727.0934, 725.0766 (-1PGU, -H2O), 707.0670 (-1PGU, -2H2O), 619.0771 (-2PGU, +2), 601.0614 (-2PGU, -16/-2PGU, -H2O, +2), 583.0501, 497.0720, 479.0626, 461.0512, 371.0404, 353.0300, 335.0192, 247.0235, 229.0138, 139.0033 | Fucophlorethol | ||
| 18 | 8.09 | 851.1090 (-H2O), 725.0770 (-1PGU, -H2O), 707.0676 (-1PGU, -2H2O), 601.0609 (-2PGU, -16/-2PGU, -H2O, +2), 461.0515, 371.0399, 353.0299, 335.0198, 229.0137 | Fucophlorethol | ||
| 19 | 8.41 | 851.1081 (-H2O), 725.0769 (-1PGU, -H2O), 707.0659 (-1PGU, -2H2O), 477.0452, 461.0515, 371.0403, 353.0301, 337.0349, 229.0139 | Fucophlorethol | ||
| Octamer | C48H34O24 | 20 | 2.36 | 975.1219 (-H2O), 957.1110 (-2H2O), 939.1012, 745.1023, 727.0914, 708.0727, 455.0608, 289.0341, 247.0238, 229.0140 | Octafucol |
| 21 | 5.99 | 975.1245 (-H2O), 957.1143 (-2H2O), 867.1024 (-1PGU), 849.0925 (-1PGU, -H2O), 832.0905 707.0670 692.0798 579.0777 454.0540 413.0514 287.0198 269.0089 165.0192 125.0243 | Fucophlorethol | ||
| 22 | 7.70 | 975.1239 (-H2O), 957.1132 (-2H2O), 832.0891, 703.0926, 579.0769, 413.0509, 289.0350, 287.0196, 269.0092, 139.0030, 125.0243 | Octafucol | ||
| 23 | 8.31 | 975.1227 (-H2O), 850.1005 727.0926 603.0744, 477.0458, 461.0503, 372.0482, 353.0287, 338.0435, 247.0248, 229.0141, 139.0039 | Octafucol | ||
| 24 | 9.48 | 975.1236 (-H2O), 849.0921 (-1PGU, -H2O), 831.0824 (-1PGU, -2H2O), 601.0619, 583.0514, 495.0571, 477.0454, 461.0514, 353.0305, 335.0195, 229.0141, 139.0035 | Fucophlorethol | ||
| 25 | 9.64 | 975.1237 (-H2O), 849.0922 (-1PGU, -H2O), 831.0825 (-1PGU, -2H2O), 725.0769, 495.0568, 477.0463, 461.0508, 371.0408, 353.0305, 335.0195, 245.0088, 229.0141, 139.0034 | Fucophlorethol | ||
| Nonamer | C54H38O27 | 26 | 3.64 | 1099.1391 (-H2O), 1081.1257 (-2H2O), 1063.1158, 869.1167, 851.1078, 832.0883, 579.0775, 519.0556, 413.0492, 287.0180, 247.0240, 229.0130 | Nonafucol |
| 27 | 5.33 | 1099.1422 (-H2O), 1081.1278 (-2H2O), 1063.1163, 956.1099, 938.0925, 809.0996, 745.1041, 725.0770, 708.0751, 643.0702, 555.0580, 455.0609, 353.0298, 289.0365, 229.0149 | Nonafucol | ||
| 28 | 8.25 | 1099.1397 (-H2O), 1081.1279 (-2H2O), 955.0955 (-1PGU, -2H2O), 851.1070, 727.0926, 603.0767, 477.0472, 371.0412, 353.0302, 247.0247, 229.0142 | Fucophlorethol | ||
| 29 | 9.11 | 1099.1403 (-H2O), 1081.1309 (-2H2O), 956.1074, 827.1070, 703.0928, 455.0607, 413.0512, 287.0194, 269.0090, 247.0247, 229.0144, 165.0181, 139.0031 | Nonafucol | ||
| 30 | 9.34 | 1099.1389 (-H2O), 1081.1278 (-2H2O), 973.1061 (-1PGU, -H2O), 956.1036, 827.1058, 725.0738, 708.0724, 455.0610, 373.0562, 289.0340, 269.0090, 247.0238, 229.0133, 165.0186, 139.0032 | Fucophlorethol | ||
| 31 | 10.67 | 1099.1405 (-H2O), 973.1087 (-1PGU, -H2O), 849.0923 (-2PGU, -16/-2PGU, -H2O, +2), 835.1120, 727.0931, 709.0806, 601.0609, 461.0519, 353.0299, 229.0142, 139.0028 | Fucophlorethol | ||
| 32 | 10.82 | 1099.1398 (-H2O), 973.1063 (-1PGU, -H2O), 955.0959 (-1PGU, -2H2O), 849.0919 (-2PGU, -16/-2PGU, -H2O, +2), 745.1022, 725.0747, 601.0600, 495.0559, 477.0463, 353.0303, 229.0142, 139.0035 | Fucophlorethol |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Obluchinskaya, E.D.; Pozharitskaya, O.N.; Shevyrin, V.A.; Kovaleva, E.G.; Flisyuk, E.V.; Shikov, A.N. Optimization of Extraction of Phlorotannins from the Arctic Fucus vesiculosus Using Natural Deep Eutectic Solvents and Their HPLC Profiling with Tandem High-Resolution Mass Spectrometry. Mar. Drugs 2023, 21, 263. https://doi.org/10.3390/md21050263
Obluchinskaya ED, Pozharitskaya ON, Shevyrin VA, Kovaleva EG, Flisyuk EV, Shikov AN. Optimization of Extraction of Phlorotannins from the Arctic Fucus vesiculosus Using Natural Deep Eutectic Solvents and Their HPLC Profiling with Tandem High-Resolution Mass Spectrometry. Marine Drugs. 2023; 21(5):263. https://doi.org/10.3390/md21050263
Chicago/Turabian StyleObluchinskaya, Ekaterina D., Olga N. Pozharitskaya, Vadim A. Shevyrin, Elena G. Kovaleva, Elena V. Flisyuk, and Alexander N. Shikov. 2023. "Optimization of Extraction of Phlorotannins from the Arctic Fucus vesiculosus Using Natural Deep Eutectic Solvents and Their HPLC Profiling with Tandem High-Resolution Mass Spectrometry" Marine Drugs 21, no. 5: 263. https://doi.org/10.3390/md21050263
APA StyleObluchinskaya, E. D., Pozharitskaya, O. N., Shevyrin, V. A., Kovaleva, E. G., Flisyuk, E. V., & Shikov, A. N. (2023). Optimization of Extraction of Phlorotannins from the Arctic Fucus vesiculosus Using Natural Deep Eutectic Solvents and Their HPLC Profiling with Tandem High-Resolution Mass Spectrometry. Marine Drugs, 21(5), 263. https://doi.org/10.3390/md21050263






