Releasing Bioactive Compounds from Brown Seaweed with Novel Cold-Adapted Alginate Lyase and Alcalase
Abstract
1. Introduction
2. Results and Discussion
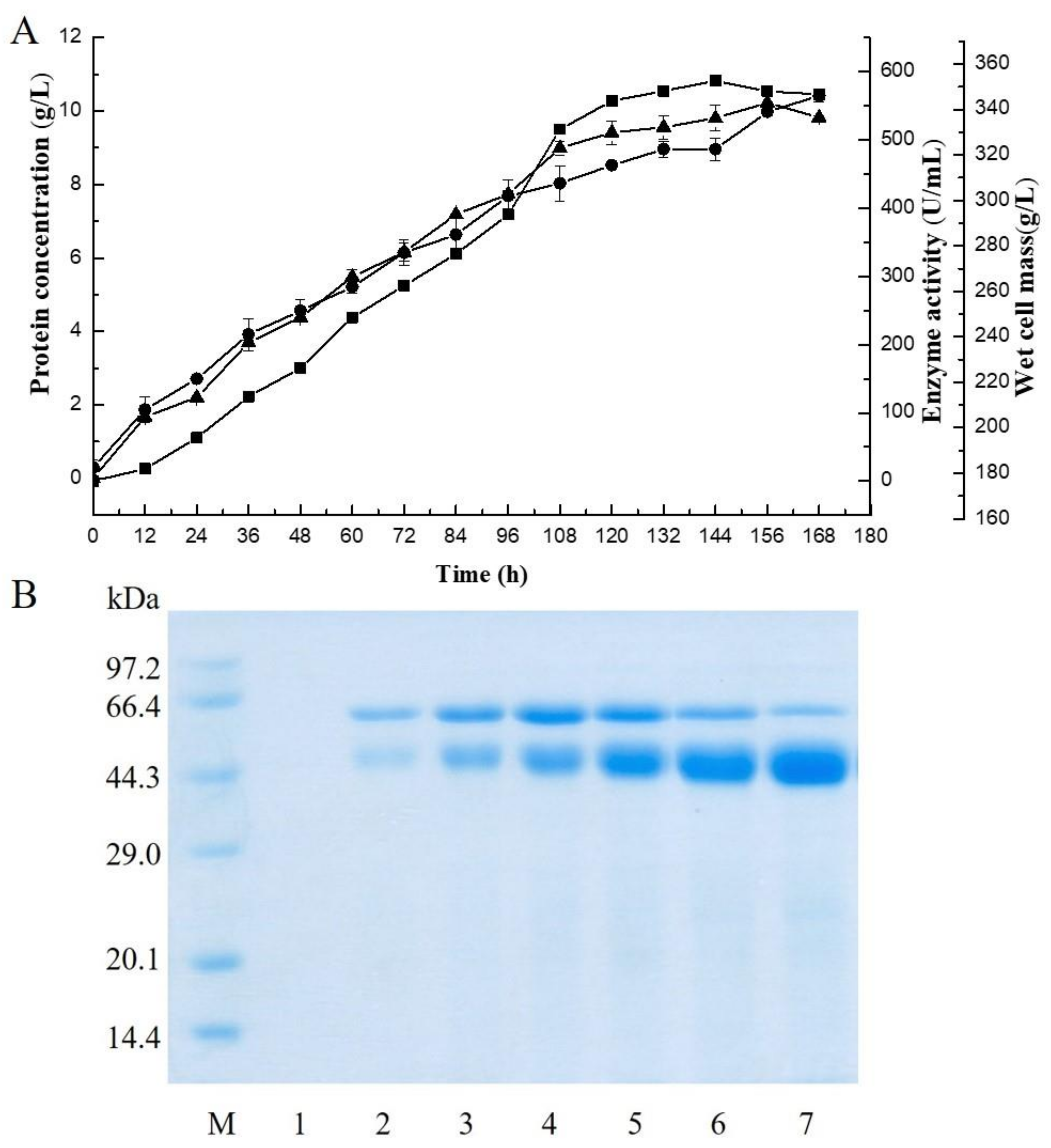
2.1. Expression of the Alginate Lyase Gene in P. pastoris
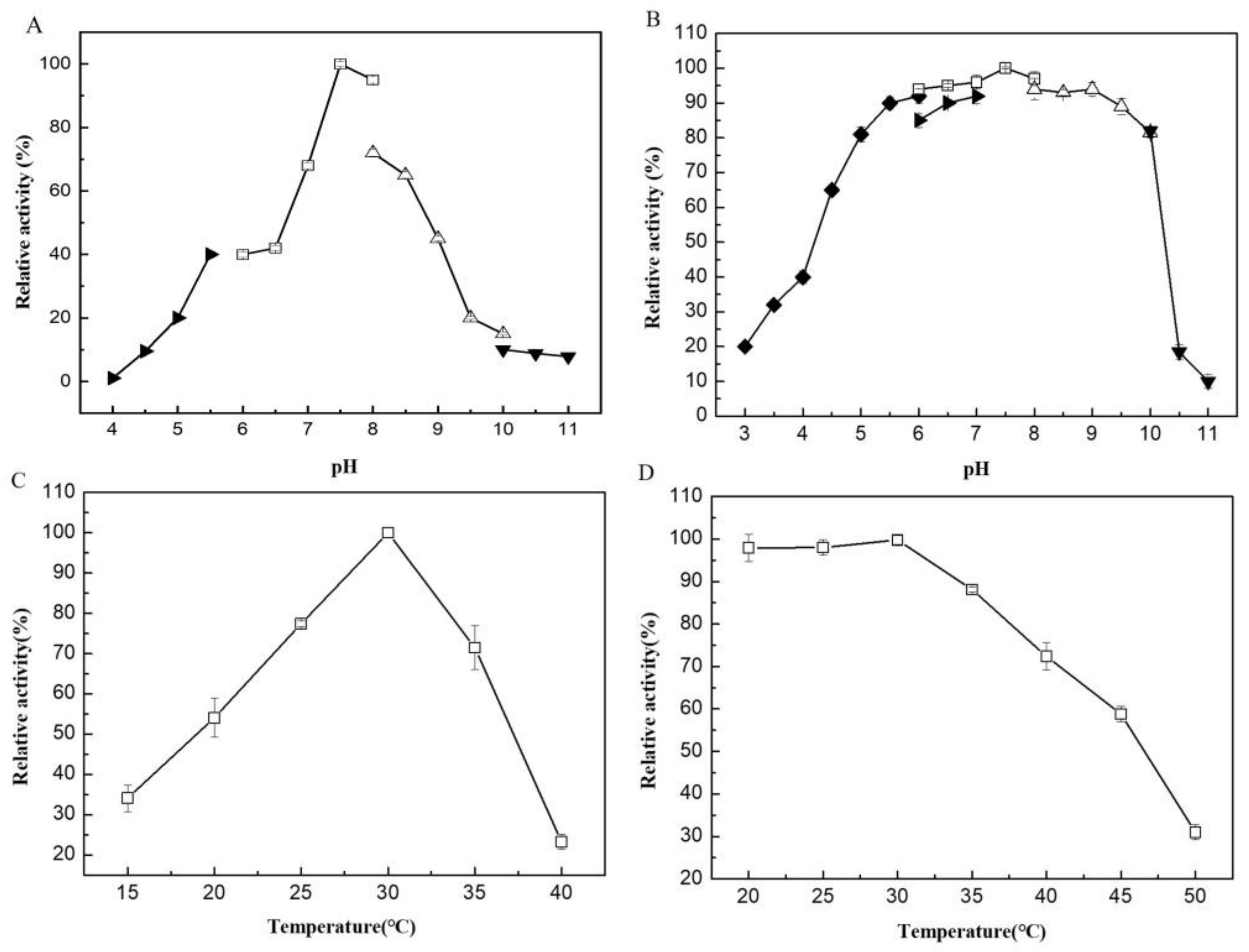
2.2. Biochemical Characterization of VfAly7
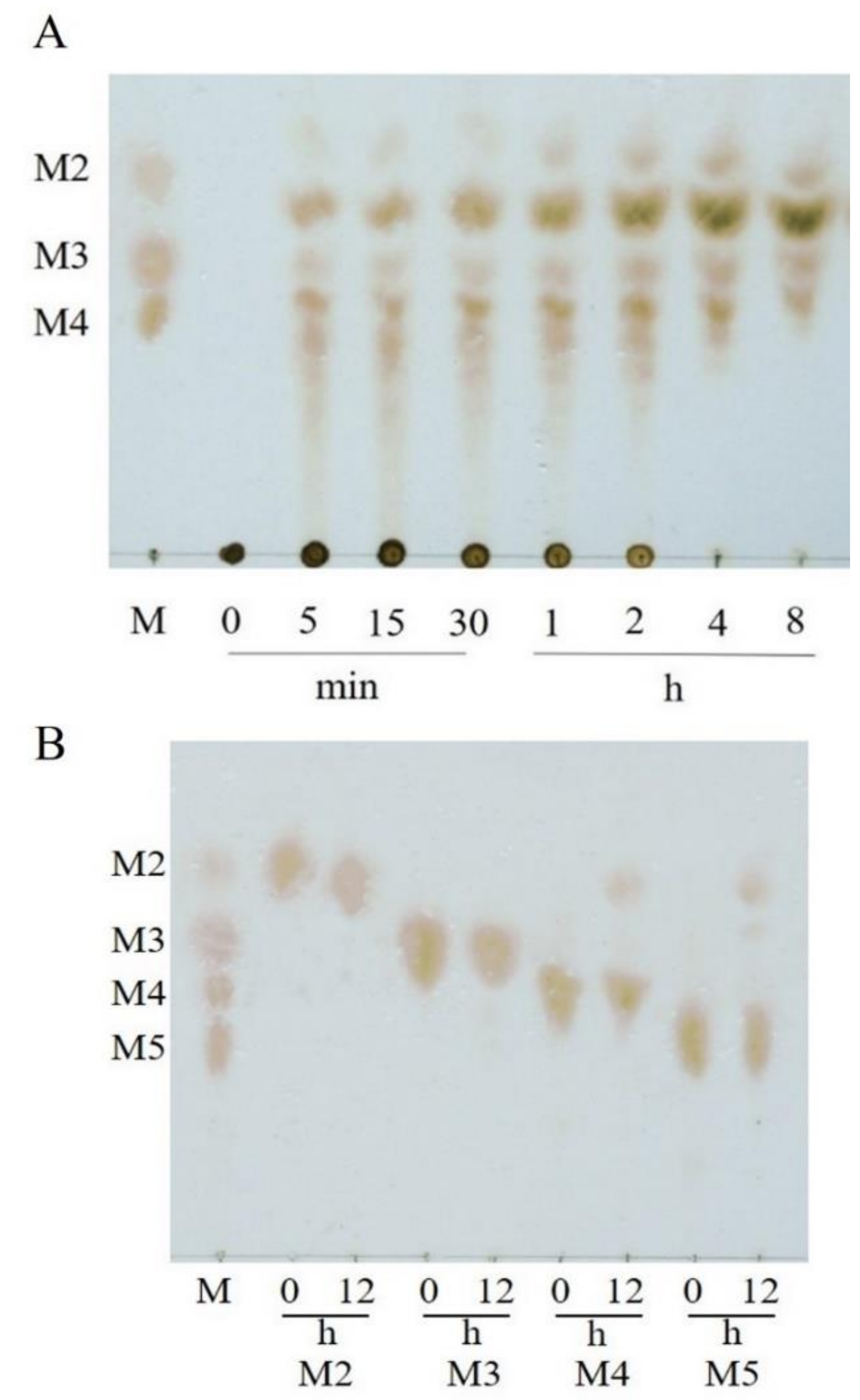
2.3. Substrate Specificity and Hydrolysis Property of VfAly7
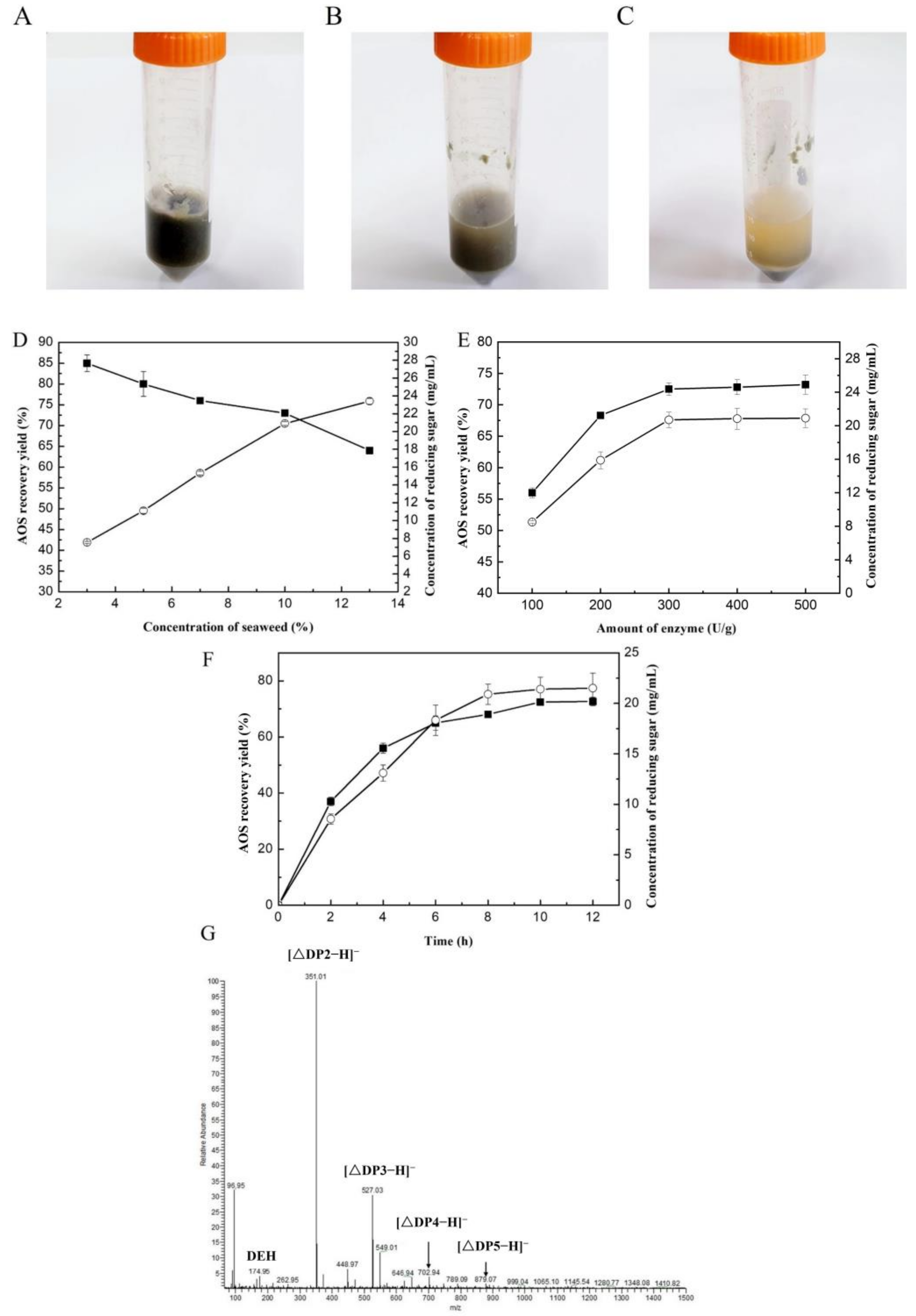
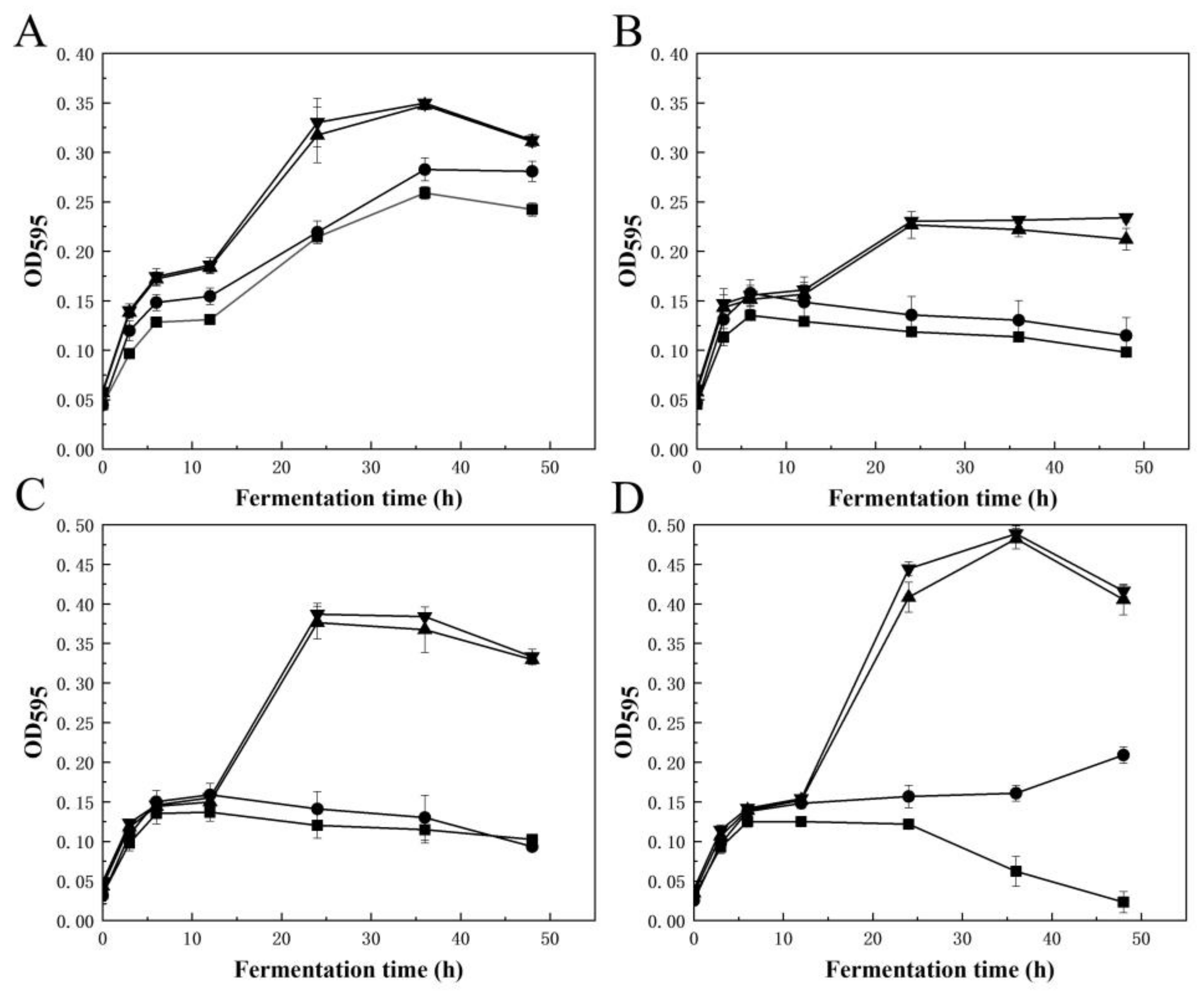
2.4. Production of AOSs and Their Prebiotic Activity
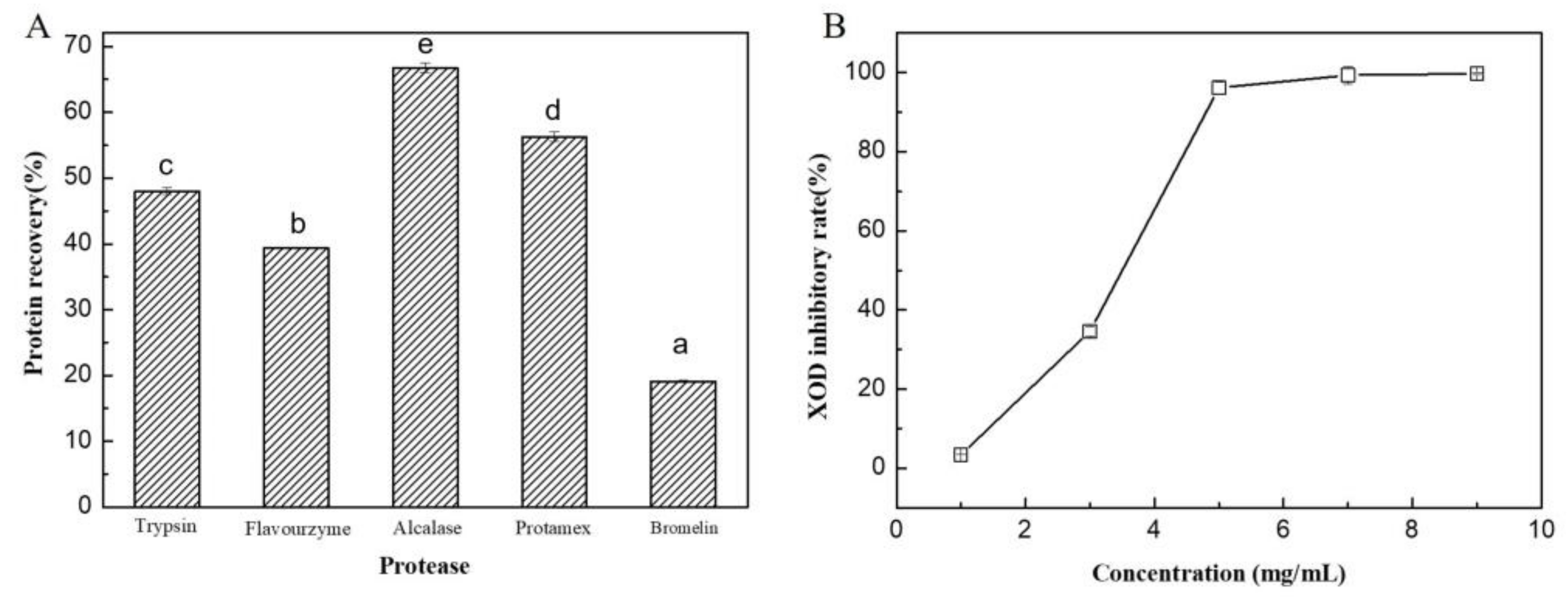
2.5. Production of Algal Protein Hydrolysate and Its XOD Inhibitory Activity
2.6. Enzymatic Approach for the Production of Bioactive Compounds from Brown Seaweed
3. Materials and Methods
3.1. Materials
3.2. Gene Cloning and Expression
3.3. Enzyme Activity Determination
3.4. Purification and Biochemical Characterization of VfAly7
3.5. Substrate Specificity and Hydrolysis Properties of VfAly7
3.6. Degradation of Brown Seaweed by VfAly7
3.7. Recovery of AOSs from Brown Seaweed by VfAly7
3.8. Prebiotic Activity Assay of AOSs
3.9. Recovery of Protein Hydrolysates with XOD Inhibitory Activity
3.10. XOD Inhibitory Activity Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dave, N.; Selvaraj, R.; Varadavenkatesan, T.; Vinayagam, R. A critical review on production of bioethanol from macroalgal biomass. Algal Res. 2019, 42, 101606. [Google Scholar] [CrossRef]
- Vasudevan, U.M.; Lee, O.K.; Lee, E.Y. Alginate derived functional oligosaccharides: Recent developments, barriers, and future outlooks. Carbohydr. Polym. 2021, 267, 118158. [Google Scholar] [CrossRef] [PubMed]
- Ferdouse, F.; Holdt, S.L.; Smith, R.; Murúa, P.; Yang, Z. The Global Satus of Seaweed Production, Trade and Utilization; FAO: Rome, Italy, 2018; Volume 124. [Google Scholar]
- Nadeeshani, H.; Hassouna, A.; Lu, J. Proteins extracted from seaweed Undaria pinnatifida and their potential uses as foods and nutraceuticals. Crit. Rev. Food Sci. Nutr. 2021, 62, 6187–6203. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.S.; Luan, F.; Hu, J.W.; Liu, Y.; Zhang, X.M.; Qin, T.T.; Zhang, X.; Liu, R.; Zeng, N. Recent research advances in polysaccharides from Undaria pinnatifida: Isolation, structures, bioactivities, and applications. Int. J. Biol. Macromol. 2022, 206, 325–354. [Google Scholar] [CrossRef]
- Kerton, F.M.; Liu, Y.; Omari, K.W.; Hawboldt, K. Green chemistry and the ocean-based biorefinery. Green Chem. 2013, 15, 860–871. [Google Scholar] [CrossRef]
- Sharma, S.; Horn, S.J. Enzymatic saccharification of brown seaweed for production of fermentable sugars. Bioresour. Technol. 2016, 213, 155–161. [Google Scholar] [CrossRef]
- Liu, J.; Yang, S.Q.; Li, X.T.; Yan, Q.J.; Reaney, M.J.T.; Jiang, Z.Q. Alginate oligosaccharides: Production, biological activities, and potential applications. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1859–1881. [Google Scholar] [CrossRef]
- Jonathan, M.; Souza da Silva, C.; Bosch, G.; Schols, H.; Gruppen, H. In vivo degradation of alginate in the presence and in the absence of resistant starch. Food Chem. 2015, 172, 117–120. [Google Scholar] [CrossRef]
- Falkeborg, M.; Cheong, L.Z.; Gianfico, C.; Sztukiel, K.M.; Kristensen, K.; Glasius, M.; Guo, Z. Alginate oligosaccharides: Enzymatic preparation and antioxidant property evaluation. Food Chem. 2014, 164, 185–194. [Google Scholar] [CrossRef]
- Gao, J.; Lin, L.Z.; Sun, B.Z.; Zhao, M.M. Comparison study on polysaccharide fractions from Laminaria japonica: Structural characterization and bile acid binding capacity. J. Agric. Food Chem. 2017, 65, 9790–9798. [Google Scholar] [CrossRef]
- Admassu, H.; Gasmalla, M.A.A.; Yang, R.J.; Zhao, W. Bioactive peptides derived from seaweed protein and their health benefits: Antihypertensive, antioxidant, and antidiabetic properties. J. Food Sci. 2018, 83, 6–16. [Google Scholar] [CrossRef]
- Sapatinha, M.; Oliveira, A.; Costa, S.; Pedro, S.; Goncalves, A.; Mendes, R.; Bandarra, N.M.; Pires, C. Red and brown seaweeds extracts: A source of biologically active compounds. Food Chem. 2022, 393, 133453. [Google Scholar] [CrossRef]
- Zhong, H.; Abdullah; Zhang, Y.P.; Deng, L.L.; Zhao, M.J.; Tang, J.; Zhang, H.; Feng, F.Q.; Wang, J. Exploring the potential of novel xanthine oxidase inhibitory peptide (ACECD) derived from Skipjack tuna hydrolysates using affinity-ultrafiltration coupled with HPLC-MALDI-TOF/TOF-MS. Food Chem. 2021, 347, 129068. [Google Scholar] [CrossRef]
- Barzkar, N.; Sheng, R.L.; Sohail, M.; Jahromi, S.T.; Babich, O.; Sukhikh, S.; Nahavandi, R. Alginate lyases from marine bacteria: An enzyme ocean for sustainable future. Molecules 2022, 27, 3375. [Google Scholar] [CrossRef]
- Gao, S.; Zhang, Z.L.; Li, S.Y.; Su, H.; Tang, L.Y.; Tan, Y.L.; Yu, W.G.; Han, F. Characterization of a new endo-type polysaccharide lyase (PL) family 6 alginate lyase with cold-adapted and metal ions-resisted property. Int. J. Biol. Macromol. 2018, 120, 729–735. [Google Scholar] [CrossRef]
- Uchimura, K.; Miyazaki, M.; Nogi, Y.; Kobayashi, T.; Horikoshi, K. Cloning and sequencing of alginate lyase genes from deep-sea strains of Vibrio and Agarivorans and characterization of a new Vibrio enzyme. Mar. Biotechnol. 2010, 12, 526–533. [Google Scholar] [CrossRef]
- Zhou, H.X.; Xu, S.S.; Yin, X.J.; Wang, F.L.; Li, Y. Characterization of a new bifunctional and cold-adapted polysaccharide lyase (PL) family 7 alginate lyase from Flavobacterium sp. Mar. Drugs 2020, 18, 388. [Google Scholar] [CrossRef]
- Tang, L.Y.; Wang, Y.; Gao, S.; Wu, H.; Wang, D.N.; Yu, W.G.; Han, F. Biochemical characteristics and molecular mechanism of an exo-type alginate lyase VxAly7D and its use for the preparation of unsaturated monosaccharides. Biotechnol. Biofuels 2020, 13, 99. [Google Scholar] [CrossRef]
- Zhang, Z.L.; Tang, L.Y.; Bao, M.M.; Liu, Z.G.; Yu, W.G.; Han, F. Functional characterization of carbohydrate-binding modules in a new alginate lyase, TsAly7B, from Thalassomonas sp. LD5. Mar. Drugs 2020, 18, 25. [Google Scholar] [CrossRef]
- Zhu, B.W.; Li, K.K.; Wang, W.X.; Ning, L.M.; Tan, H.D.; Zhao, X.M.; Yin, H. Preparation of trisaccharides from alginate by a novel alginate lyase Alg7A from marine bacterium Vibrio sp. W13. Int. J. Biol. Macromol. 2019, 139, 879–885. [Google Scholar] [CrossRef]
- Li, S.Y.; Wang, Z.P.; Wang, L.N.; Peng, J.X.; Wang, Y.N.; Han, Y.T.; Zhao, S.F. Combined enzymatic hydrolysis and selective fermentation for green production of alginate oligosaccharides from Laminaria japonica. Bioresour. Technol. 2019, 281, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Yue, M.M.; Gong, W.W.; Qiao, Y.; Ding, H.B. A method for efficient expression of Pseudomonas aeruginosa alginate lyase in Pichia pastoris. Prep. Biochem. Biotechnol. 2016, 46, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.X.; Liu, Z.M.; Fu, X.D.; Zhu, C.L.; Kong, Q.; Yang, M.; Mou, H.J. Expression and characterization of an alginate lyase and its thermostable mutant in Pichia pastoris. Mar. Drugs 2020, 18, 305. [Google Scholar] [CrossRef] [PubMed]
- Li, H.F.; Wang, S.L.; Zhang, Y.Y.; Chen, L.H. High-level expression of a thermally stable alginate lyase using pichia pastoris, characterization and application in producing brown alginate oligosaccharide. Mar. Drugs 2018, 16, 158. [Google Scholar] [CrossRef]
- Kim, D.E.; Lee, E.Y.; Kim, H.S. Cloning and characterization of alginate lyase from a marine bacterium Streptomyces sp. ALG-5. Mar. Biotechnol. 2009, 11, 10–16. [Google Scholar] [CrossRef]
- Chen, P.; Zhu, Y.M.; Men, Y.; Zeng, Y.; Sun, Y.X. Purification and characterization of a novel alginate lyase from the marine bacterium Bacillus sp. Alg07. Mar. Drugs 2018, 16, 86. [Google Scholar] [CrossRef]
- Deng, J.J.; Mao, H.H.; Fang, W.; Li, Z.Q.; Shi, D.; Li, Z.W.; Luo, X.C. Enzymatic conversion and recovery of protein, chitin, and astaxanthin from shrimp shell waste. J. Clean. Prod. 2020, 271, 122655. [Google Scholar] [CrossRef]
- Sun, C.X.; Zhou, J.L.; Duan, G.L.; Yu, X.B. Hydrolyzing Laminaria japonica with a combination of microbial alginate lyase and cellulase. Bioresour. Technol. 2020, 311, 123548. [Google Scholar] [CrossRef]
- Wang, Y.; Han, F.; Hu, B.; Li, J.B.; Yu, W.G. In vivo prebiotic properties of alginate oligosaccharides prepared through enzymatic hydrolysis of alginate. Nutr. Res. 2006, 26, 597–603. [Google Scholar] [CrossRef]
- Seghetta, M.; Romeo, D.; D’Este, M.; Alvarado-Morales, M.; Angelidaki, I.; Bastianoni, S.; Thomsen, M. Seaweed as innovative feedstock for energy and feed-Evaluating the impacts through a Life Cycle Assessment. J. Clean. Prod. 2017, 150, 1–15. [Google Scholar] [CrossRef]
- Fleurence, J. The enzymatic degradation of algal cell walls: A useful approach for improving protein accessibility. J. Appl. Phycol. 1999, 11, 313–314. [Google Scholar] [CrossRef]
- Li, Q.Y.; Kang, X.Y.; Shi, C.C.; Li, Y.J.; Majumder, K.; Ning, Z.X.; Ren, J.Y. Moderation of hyperuricemia in rats via consuming walnut protein hydrolysate diet and identification of new antihyperuricemic peptides. Food Funct. 2018, 9, 107–116. [Google Scholar] [CrossRef]
- Nongonierma, A.B.; Fitzgerald, R.J. Tryptophan-containing milk protein-derived dipeptides inhibit xanthine oxidase. Peptides 2012, 37, 263–272. [Google Scholar] [CrossRef]
- He, W.W.; Su, G.W.; Sun-Waterhouse, D.X.; Waterhouse, G.I.N.; Zhao, M.M.; Liu, Y. In vivo anti-hyperuricemic and xanthine oxidase inhibitory properties of tuna protein hydrolysates and its isolated fractions. Food Chem. 2019, 272, 453–461. [Google Scholar] [CrossRef]
- Sudhakar, M.P.; Kumar, B.R.; Mathimani, T.; Arunkumar, K. A review on bioenergy and bioactive compounds from microalgae and macroalgae-sustainable energy perspective. J. Clean. Prod. 2019, 228, 1320–1333. [Google Scholar] [CrossRef]
- Haug, A.; Larsen, B.; Smidsrod, O. Studies on the sequence of uronic acid residues in alginic acid. Acta Chem. Scand. 1967, 21, 691–704. [Google Scholar] [CrossRef]
- Meng, Q.; Zhou, L.C.; Hassanin, H.A.M.; Jiang, B.; Liu, Y.C.; Chen, J.J.; Zhang, T. A new role of family 32 carbohydrate binding module in alginate lyase from Vibrio natriegens SK42.001 in altering its catalytic activity, thermostability and product distribution. Food Biosci. 2021, 42, 101112. [Google Scholar] [CrossRef]
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Blumenkr, N.; Asboehan, G. New method for quantitative determination of uronic acids. Anal. Biochem. 1973, 54, 484–489. [Google Scholar] [CrossRef]
- Liu, Y.H.; Ma, J.W.; Shi, R.; Li, T.; Yan, Q.J.; Jiang, Z.Q.; Yang, S.Q. Biochemical characterization of a β-N-acetylhexosaminidase from Catenibacterium mitsuokai suitable for the synthesis of lacto-N-triose II. Process Biochem. 2021, 102, 360–368. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, J.; Jiang, Z.; Yan, Q.; Han, S.; Yang, S. Releasing Bioactive Compounds from Brown Seaweed with Novel Cold-Adapted Alginate Lyase and Alcalase. Mar. Drugs 2023, 21, 208. https://doi.org/10.3390/md21040208
Jiang J, Jiang Z, Yan Q, Han S, Yang S. Releasing Bioactive Compounds from Brown Seaweed with Novel Cold-Adapted Alginate Lyase and Alcalase. Marine Drugs. 2023; 21(4):208. https://doi.org/10.3390/md21040208
Chicago/Turabian StyleJiang, Jun, Zhengqiang Jiang, Qiaojuan Yan, Susu Han, and Shaoqing Yang. 2023. "Releasing Bioactive Compounds from Brown Seaweed with Novel Cold-Adapted Alginate Lyase and Alcalase" Marine Drugs 21, no. 4: 208. https://doi.org/10.3390/md21040208
APA StyleJiang, J., Jiang, Z., Yan, Q., Han, S., & Yang, S. (2023). Releasing Bioactive Compounds from Brown Seaweed with Novel Cold-Adapted Alginate Lyase and Alcalase. Marine Drugs, 21(4), 208. https://doi.org/10.3390/md21040208






