Antibacterial Indole Diketopiperazine Alkaloids from the Deep-Sea Cold Seep-Derived Fungus Aspergillus chevalieri
Abstract
1. Introduction
2. Results and Discussion
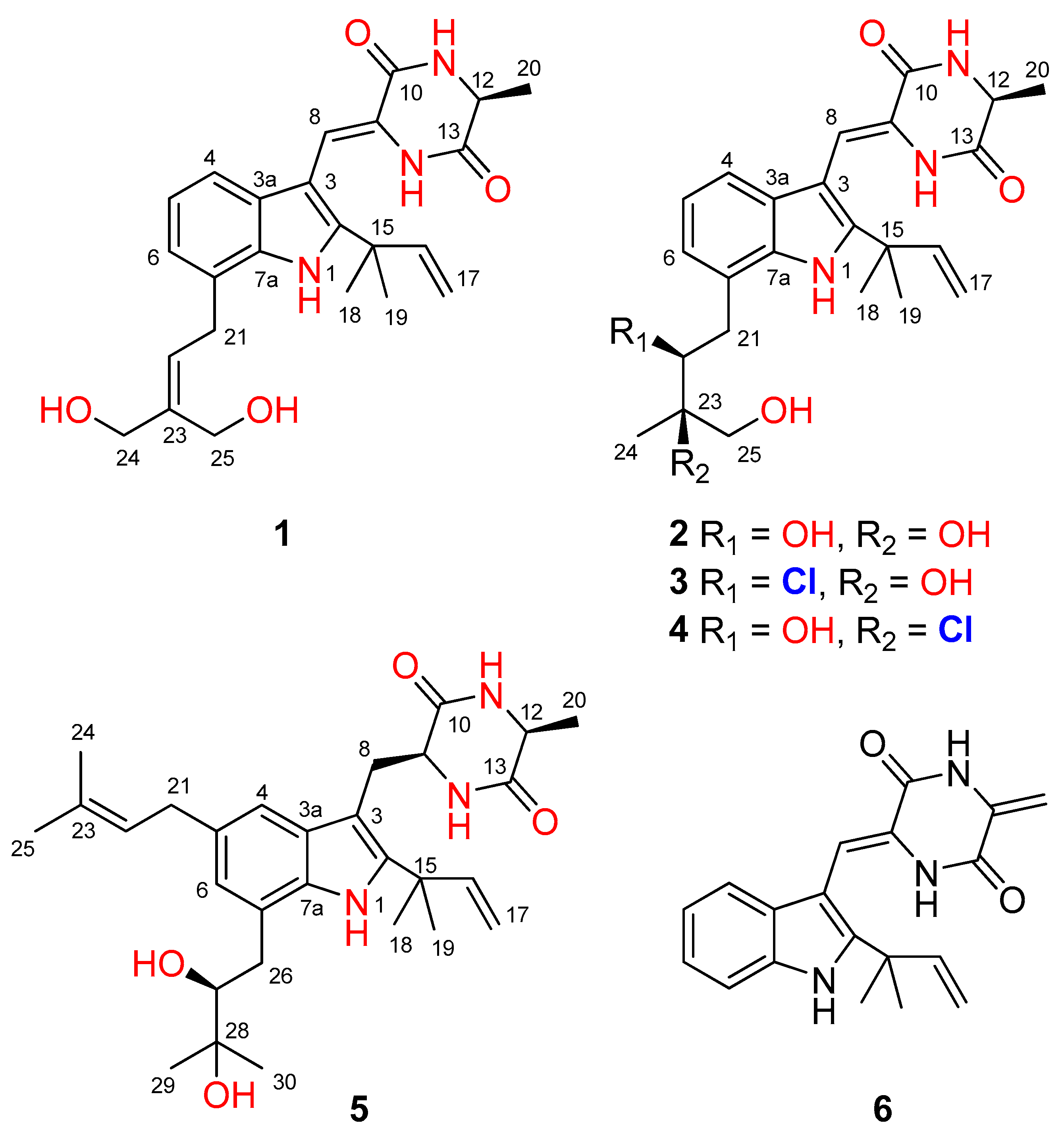
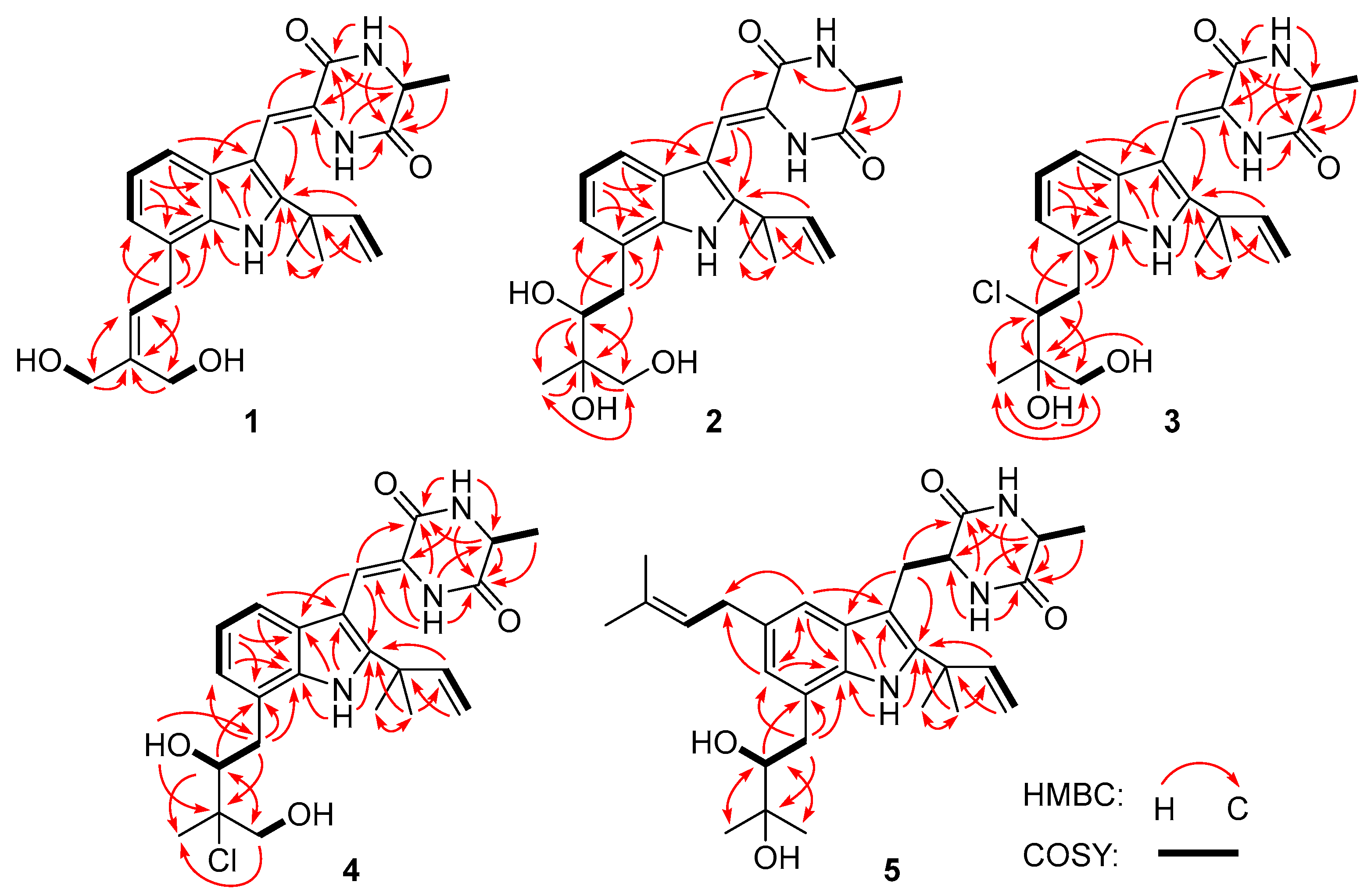
2.1. Structure Elucidation
2.2. Antimicrobial Activity
3. Experimental Section
3.1. General Experimental Procedures
3.2. Fungal Material
3.3. Fermentation, Extraction, and Isolation
- 24,25-Dihydroxyvariecolorin G (1): colorless amorphous powder; [α]25 D –17.4 (c =0.23, MeOH); UV (MeOH) λmax (log ε) 226 (3.26) nm, 252 (3.11) nm, 279 (2.90) nm, 335 (2.96) nm; ECD (0.35 mM, MeOH) λmax (Δε) 214 (–5.52), 240 (+2.22), 342 (–1.46) nm; 1H and 13C NMR data, Table 1 and Table 2; HRESIMS m/z 424.2225 [M+H]+ (calcd for C24H30N3O4, 424.2231).
- 25-Hydroxyrubrumazine B (2): colorless amorphous powder; [α]25 D –23.8 (c =0.21, MeOH); UV (MeOH) λmax (log ε) 225 (3.66) nm, 255 (3.28) nm, 282 (3.13) nm, 339 (3.22) nm; ECD (0.57 mM, MeOH) λmax (Δε) 221 (–11.88), 248 (+0.52), 333 (–1.97) nm; 1H and 13C NMR data, Table 1 and Table 2; HRESIMS m/z 442.2330 [M+H]+ (calcd for C24H32N3O5, 442.2336).
- 22-Chloro-25-hydroxyrubrumazine B (3): colorless amorphous powder; [α]25 D –25.0 (c =0.16, MeOH); UV (MeOH) λmax (log ε) 224 (3.56) nm, 257 (3.15) nm, 278 (2.98) nm, 335 (3.06) nm; ECD (0.35 mM, MeOH) λmax (Δε) 206 (–9.54), 234 (+7.56), 328 (–3.46) nm; 1H and 13C NMR data, Table 1 and Table 2; HRESIMS m/z 460.1987 [M+H]+ (calcd for C24H30ClN3O4, 460.1998).
- 25-Hydroxyvariecolorin F (4): colorless amorphous powder; [α]25 D –53.8 (c =0.26, MeOH); UV (MeOH) λmax (log ε) 226 (3.70) nm, 255 (3.30) nm, 279 (3.13) nm, 334 (3.22) nm; ECD (0.28 mM, MeOH) λmax (Δε) 204 (–12.69), 240 (+2.38), 334 (–2.71) nm; 1H and 13C NMR data, Table 1 and Table 2; HRESIMS m/z 460.1992 [M+H]+ (calcd for C24H30ClN3O4, 460.1998).
- 27-epi-Aspechinulin D (5): colorless amorphous powder; [α]25 D –23.1 (c =0.13, MeOH); UV (MeOH) λmax (log ε) 231 (3.61) nm, 280 (2.99) nm; ECD (0.26 mM, MeOH) λmax (Δε) 226 (–9.63), 271 (+1.03) nm; 1H and 13C NMR data, Table 1 and Table 2; HRESIMS m/z 496.3157 [M+H]+ (calcd for C29H42N3O4, 496.3170).
3.4. Computational NMR Chemical Shift Calculation and DP4+ Analysis
3.5. Acidic Hydrolysis of Compounds 1–5
3.6. 2,2-Dimethoxypropane Derivatization of Compound 4
3.7. Antibacterial Assay
3.8. Scanning Electron Microscopy (SEM)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Yan, L.-H.; Li, X.-M.; Chi, L.-P.; Li, X.; Wang, B.-G. Six new antimicrobial metabolites from the deep-sea sediment-derived fungus Aspergillus fumigatus SD-406. Mar. Drugs 2022, 20, 4. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-H.; Li, X.-M.; Li, X.; Yang, S.-Q.; Shi, X.-S.; Li, H.-L.; Wang, B.-G. Antibacterial alkaloids and polyketide derivatives from the deep sea-derived fungus Penicillium cyclopium SD-413. Mar. Drugs 2020, 18, 553. [Google Scholar] [CrossRef] [PubMed]
- Shang, Z.; Salim, A.A.; Khalil, Z.; Quezada, M.; Bernhardt, P.V.; Capon, R.J. Viridicatumtoxins: Expanding on a rare tetracycline antibiotic scaffold. J. Org. Chem. 2015, 80, 12501–12508. [Google Scholar] [CrossRef]
- Cui, H.; Su, X.; Chen, F.; Holland, M.; Yang, S.; Liang, J.; Su, P.; Dong, H.; Hou, W. Microbial diversity of two cold seep systems in gas hydrate-bearing sediments in the South China Sea. Mar. Environ. Res. 2019, 44, 230–239. [Google Scholar] [CrossRef]
- Hu, X.-Y.; Li, X.-M.; Yang, S.-Q.; Li, X.; Wang, B.-G.; Meng, L.-H. New cytochalasin derivatives from deep-sea cold seep-derived endozoic fungus Curvularia verruculosa CS-129. Chem. Biodivers. 2022, 19, e202200550. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.-Y.; Wang, C.-Y.; Li, X.-M.; Yang, S.-Q.; Li, X.; Wang, B.-G.; Si, S.-Y.; Meng, L.-H. Cytochalasin derivatives from the endozoic Curvularia verruculosa CS-129, a fungus isolated from the deep-sea squat lobster Shinkaia crosnieri living in the cold seep environment. J. Nat. Prod. 2021, 84, 3122–3130. [Google Scholar] [CrossRef]
- Song, Q.; Yang, S.-Q.; Li, X.-M.; Hu, X.-Y.; Li, X.; Wang, B.-G. Aromatic polyketides from the deep-Sea cold-seep mussel associated endozoic fungus Talaromyces minioluteus CS-138. Mar. Drugs 2022, 20, 529. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.-Y.; Li, X.; Yang, S.-Q.; Li, X.-M.; Wang, B.-G.; Meng, L.-H. Vercytochalasins A and B: Two unprecedented biosynthetically related cytochalasins from the deep-sea-sourced endozoic fungus Curvularia verruculosa. Chin. Chem. Lett. 2023, 34, 107516. [Google Scholar] [CrossRef]
- Yan, L.-H.; Li, P.-H.; Li, X.-M.; Yang, S.-Q.; Liu, K.-C.; Wang, B.-G.; Li, X. Chevalinulins A and B, proangiogenic alkaloids with a spiro[bicyclo[2.2.2]octane-diketopiperazine] skeleton from deep-sea cold-seep-derived fungus Aspergillus chevalieri CS-122. Org. Lett. 2022, 24, 2684–2688. [Google Scholar] [CrossRef]
- Wang, W.-L.; Lu, Z.-Y.; Tao, H.-W.; Zhu, T.-J.; Fang, Y.-C.; Gu, Q.-Q.; Zhu, W.-M. Isoechinulin-type alkaloids, variecolorins A–L, from halotolerant Aspergillus variecolor. J. Nat. Prod. 2007, 70, 1558–1564. [Google Scholar] [CrossRef]
- Marchelli, R.; Dossena, A.; Pochini, A.; Dradi, E. The structures of five new didehydropeptides related to neoechinulin, isolated from Aspergillus amstelodami. J. Chem. Soc. Perkin Trans. 1977, 1, 713–717. [Google Scholar] [CrossRef]
- Meng, L.-H.; Du, F.-Y.; Li, X.-M.; Pedpradab, P.; Xu, G.-M.; Wang, B.-G. Rubrumazines A–C, indolediketopiperazines of the isoechinulin class from Eurotium rubrum MA-150, a fungus obtained from marine mangrove-derived rhizospheric soil. J. Nat. Prod. 2015, 78, 909–913. [Google Scholar] [CrossRef] [PubMed]
- Du, F.-Y.; Li, X.; Li, X.-M.; Zhu, L.-W.; Wang, B.G. Indolediketopiperazine alkaloids from Eurotium cristatum EN-220, an endophytic fungus isolated from the marine alga Sargassum thunbergia. Mar. Drugs 2017, 15, 24. [Google Scholar] [CrossRef]
- Wei, X.; Feng, C.; Wang, S.-Y.; Zhang, D.-M.; Li, X.-H.; Zhang, C.-X. New indole diketopiperazine alkaloids from soft coral-associated epiphytic fungus Aspergillus sp. EGF 15-0-3. Chem. Biodivers. 2020, 17, e2000106. [Google Scholar] [CrossRef]
- Li, Y.-F.; Wu, X.-B.; Niaz, S.-I.; Zhang, L.-H.; Huang, Z.-J.; Lin, Y.-C.; Li, J.; Liu, L. Effect of culture conditions on metabolites produced by the crinoid-derived fungus Aspergillus ruber 1017. Nat. Prod. Res. 2017, 31, 1299–1304. [Google Scholar] [CrossRef] [PubMed]
- Marcarino, M.O.; Cicetti, S.; Zanardi, M.M.; Sarotti, A.M. A critical review on the use of DP4+ in the structural elucidation of natural products: The good, the bad and the ugly. A practical guide. Nat. Prod. Rep. 2022, 39, 58–76. [Google Scholar] [CrossRef]
- Ryu, M.-J.; Hillman, P.-F.; Lee, J.; Hwang, S.; Lee, E.-Y.; Cha, S.-S.; Yang, I.; Oh, D.-C.; Nam, S.-J.; Fenical, W. Antibacterial meroterpenoids, merochlorins G-J from the marine bacterium Streptomyces sp. Mar. Drugs 2021, 19, 618. [Google Scholar] [CrossRef] [PubMed]
- Zu, W.-Y.; Tang, J.-W.; Hu, K.; Zhou, Y.-F.; Gou, L.-L.; Su, X.-Z.; Lei, X.; Sun, H.-D.; Puno, P.-T. Chaetolactam A, an Azaphilone Derivative from the Endophytic Fungus Chaetomium sp. g1. J. Org. Chem. 2021, 86, 475–483. [Google Scholar] [CrossRef]
- Rychnovsky, C.D.; Rogers, B.N.; Richardson, T.I. Configurational assignment of polyene macrolide antibiotics using the [13C] acetonide analysis. Acc. Chem. Res. 1998, 31, 9–17. [Google Scholar] [CrossRef]
- Liu, Z.; Chen, Y.; Li, S.; Hu, C.; Liu, H.; Zhang, W. Indole diketopiperazine alkaloids from the deep-sea-derived fungus Aspergillus sp. FS445. Nat. Prod. Res. 2021, 36, 5213–5221. [Google Scholar] [CrossRef]
- Glass, N.L.; Donaldson, G.C. Development of primer sets designed for use with the PCR to amplify conserved genes from filamentous ascomycetes. Appl. Environ. Microbiol. 1995, 61, 1323–1330. [Google Scholar] [CrossRef] [PubMed]
- Grimblat, N.; Zanardi, M.M.; Sarotti, A.M. Beyond DP4: An improved probability for the stereochemical assignment of isomeric compounds using quantum chemical calculations of NMR shifts. J. Org. Chem. 2015, 80, 12526–12534. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, M.; Morganti, S.; Trombini, C. 3-Bromopropenyl esters in organic synthesis: Indium- and zinc-mediated entries to alk-1-ene-3,4-diols. J. Org. Chem. 2003, 68, 997–1006. [Google Scholar] [CrossRef]
- Yang, H.; Lu, B.; Zhou, D.; Zhao, L.; Song, W.; Wang, L. Identification of the first cathelicidin gene from skin of Chinese giant salamanders Andrias davidianus with its potent antimicrobial activity. Dev. Comp. Immunol. 2017, 77, 141–149. [Google Scholar] [CrossRef] [PubMed]


| No. | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|
| 1-NH | 10.58, s | 10.46, s | 10.13, s | 10.10, s | 9.89, s |
| 4 | 7.04, dd, (7.4, 1.7) | 7.05, m | 7.09, m | 7.08, dd, (7.3, 1.7) | 7.07, s |
| 5 | 6.96, t, (7.4) | 6.94, overlap | 6.99, overlap | 6.98, overlap | |
| 6 | 6.93, dd, (7.4, 1.7) | 6.94, overlap | 7.00, overlap | 7.00, overlap | 6.71, s |
| 8 | 6.88, s | 6.90, s | 6.88, s | 6.89, s | Ha, 3.01, dd, (14.5, 9.6) Hb, 3.34, m |
| 9 | 3.96, m | ||||
| 11-NH | 8.31, s | 8.31, s | 8.33, s | 8.34, s | 8.18, s |
| 12 | 4.15, qd, (6.9, 1.6) | 4.16, q, (6.9) | 4.14, qd, (6.9, 1.4) | 4.17, qd, (6.9, 1.9) | 3.82, qd, (6.9, 2.3) |
| 14-NH | 8.62, s | 8.56, s | 8.74, s | 8.57, s | 7.41, s |
| 16 | 6.10, dd, (17.6, 10.3) | 6.09, dd, (17.3, 10.6) | 6.11, dd, (17.2, 10.6) | 6.10, dd, (17.3, 10.6) | 6.16, dd, (17.4, 10.6) |
| 17 | Ha, 5.03, dd, (17.6, 1.3) Hb, 5.03, dd, (10.3, 1.3) | Ha, 5.05, dd, (17.3, 1.2) Hb, 5.06, dd, (10.6, 1.2) | Ha, 5.04, d, (17.2) Hb, 5.05, d, (10.6) | Ha, 5.06, dd, (17.3, 1.2) Hb, 5.07, dd, (10.6, 1.2) | Ha, 5.08, d, (17.4) Hb, 5.04, d, (10.6) |
| 18 | 1.48, s | 1.49, s | 1.50, s | 1.50, s | 1.48, s |
| 19 | 1.48, s | 1.49, s | 1.50, s | 1.50, s | 1.49, s |
| 20 | 1.37, d, (6.9) | 1.37, d, (6.9) | 1.39, d, (6.9) | 1.38, d, (6.9) | 1.33, d, (6.9) |
| 21 | 3.76, d, (7.8) | Ha, 2.76, dd, (14.6, 8.8) Hb, 3.25, d, (14.6) | Ha, 3.15, dd, (15.5, 10.6) Hb, 3.67, d, (15.5) | Ha, 2.93, dd, (14.6, 8.8) Hb, 3.31, d, (14.6) | 3.31, d, (7.2) |
| 22 | 5.59, t, (7.8) | 3.70, d, (8.8) | 4.37, d, (10.6) | 3.97, t, (8.8) | 5.32, t, (7.2) |
| 22-OH | 5.31, d, (7.2) | ||||
| 23-OH | 5.14, s | ||||
| 24 | 3.96, d, (4.2) | 1.12, s | 1.28, s | 1.56, s | 1.71, s |
| 24-OH | 4.70, t, (4.2) | ||||
| 25 | 4.19, d, (3.8) | Ha, 3.46, d, (10.9) Hb, 3.40, d, (10.9) | 3.57, m | 3.75, m | 1.70, s |
| 25-OH | 5.32, t, (3.8) | 5.26, t, (5.5) | 5.54, t, (6.1) | ||
| 26 | Ha, 2.65, dd, (14.5, 8.7) Hb, 3,13, d, (14.5) | ||||
| 27 | 3.42, m | ||||
| 27-OH | 4.99, d, (5.7) | ||||
| 28-OH | 4.69, s | ||||
| 29 | 1.16, s | ||||
| 30 | 1.17, s |
| No. | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|
| 2 | 143.8, C | 143.5, C | 143.6, C | 143.5, C | 141.0, C |
| 3 | 103.9, C | 103.8, C | 104.2, C | 104.0, C | 104.9, C |
| 3a | 126.0, C | 125.9, C | 126.1, C | 126.0, C | 129.1, C |
| 4 | 117.0, CH | 116.8, CH | 117.3, CH | 117.0, CH | 114.8, CH |
| 5 | 121.1, CH | 119.6, CH | 119.5, CH | 119.7, CH | 131.4, C |
| 6 | 119.7, CH | 121.7, CH | 121.3, CH | 121.9, CH | 122.2, CH |
| 7 | 123.7, C | 125.2, C | 122.6, C | 123.8, C | 124.1, C |
| 7a | 133.9, C | 134.3, C | 133.9, C | 134.2, C | 132.7, C |
| 8 | 110.3, CH | 110.2, CH | 110.1, CH | 110.0, CH | 31.3, CH2 |
| 9 | 125.1, C | 124.8, C | 125.2, C | 125.1, C | 55.6, CH |
| 10 | 159.9, C | 159.9, C | 159.8, C | 159.8, C | 167.4, C |
| 12 | 50.5, CH | 50.5, CH | 50.6, CH | 50.5, CH | 50.3, CH |
| 13 | 166.4, C | 166.3, C | 166.4, C | 166.4, C | 167.9, C |
| 15 | 39.1, C | 38.9, C | 39.0, C | 38.9, C | 38.7, C |
| 16 | 145.4, CH | 145.1, CH | 145.2, CH | 145.1, CH | 146.5, CH |
| 17 | 111.4, CH2 | 111.7, CH2 | 111.7, CH2 | 111.8, CH2 | 111.1, CH2 |
| 18 | 27.6, CH3 | 27.4, CH3 | 27.6, CH3 | 27.5, CH3 | 27.9, CH3 |
| 19 | 27.6, CH3 | 27.4, CH3 | 27.5, CH3 | 27.4, CH3 | 27.9, CH3 |
| 20 | 19.6, CH3 | 19.6, CH3 | 19.8, CH3 | 19.6, CH3 | 20.7, CH3 |
| 21 | 29.6, CH2 | 34.0, CH2 | 33.8, CH2 | 34.2, CH2 | 34.2, CH2 |
| 22 | 124.7, CH | 74.8, CH | 66.9, CH | 77.7, CH | 124.9, CH |
| 23 | 139.2, C | 74.2, C | 74.2, C | 74.0, C | 130.2, C |
| 24 | 63.5, CH2 | 19.1, CH3 | 21.0, CH3 | 22.8, CH3 | 17.7, CH3 |
| 25 | 57.1, CH2 | 67.6, CH2 | 67.5, CH2 | 68.1, CH2 | 25.6, CH3 |
| 26 | 34.5, CH2 | ||||
| 27 | 79.0, CH | ||||
| 28 | 72.2, C | ||||
| 29 | 23.6, CH3 | ||||
| 30 | 27.0, CH3 |
| Strain | Compound | ||||||
|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | Chloramphenicol b | |
| Vibrio harveyi | 16 | 32 | 8 | 32 | 16 | - | 2 |
| Edwardsiella tarda | -a | 16 | - | - | 32 | - | 8 |
| Aeromonas hydrophila | - | 32 | - | - | 32 | 4 | 2 |
| Escherichia coli | 4 | 16 | 32 | 32 | 32 | 8 | 2 |
| Micrococcus luteus | - | 32 | - | - | 16 | - | 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, L.-H.; Du, F.-Y.; Li, X.-M.; Yang, S.-Q.; Wang, B.-G.; Li, X. Antibacterial Indole Diketopiperazine Alkaloids from the Deep-Sea Cold Seep-Derived Fungus Aspergillus chevalieri. Mar. Drugs 2023, 21, 195. https://doi.org/10.3390/md21030195
Yan L-H, Du F-Y, Li X-M, Yang S-Q, Wang B-G, Li X. Antibacterial Indole Diketopiperazine Alkaloids from the Deep-Sea Cold Seep-Derived Fungus Aspergillus chevalieri. Marine Drugs. 2023; 21(3):195. https://doi.org/10.3390/md21030195
Chicago/Turabian StyleYan, Li-Hong, Feng-Yu Du, Xiao-Ming Li, Sui-Qun Yang, Bin-Gui Wang, and Xin Li. 2023. "Antibacterial Indole Diketopiperazine Alkaloids from the Deep-Sea Cold Seep-Derived Fungus Aspergillus chevalieri" Marine Drugs 21, no. 3: 195. https://doi.org/10.3390/md21030195
APA StyleYan, L.-H., Du, F.-Y., Li, X.-M., Yang, S.-Q., Wang, B.-G., & Li, X. (2023). Antibacterial Indole Diketopiperazine Alkaloids from the Deep-Sea Cold Seep-Derived Fungus Aspergillus chevalieri. Marine Drugs, 21(3), 195. https://doi.org/10.3390/md21030195







