Immunomodulatory and Antitumoral Activity of Gold Nanoparticles Synthesized by Red Algae Aqueous Extracts
Abstract
:1. Introduction
2. Results and Discussion
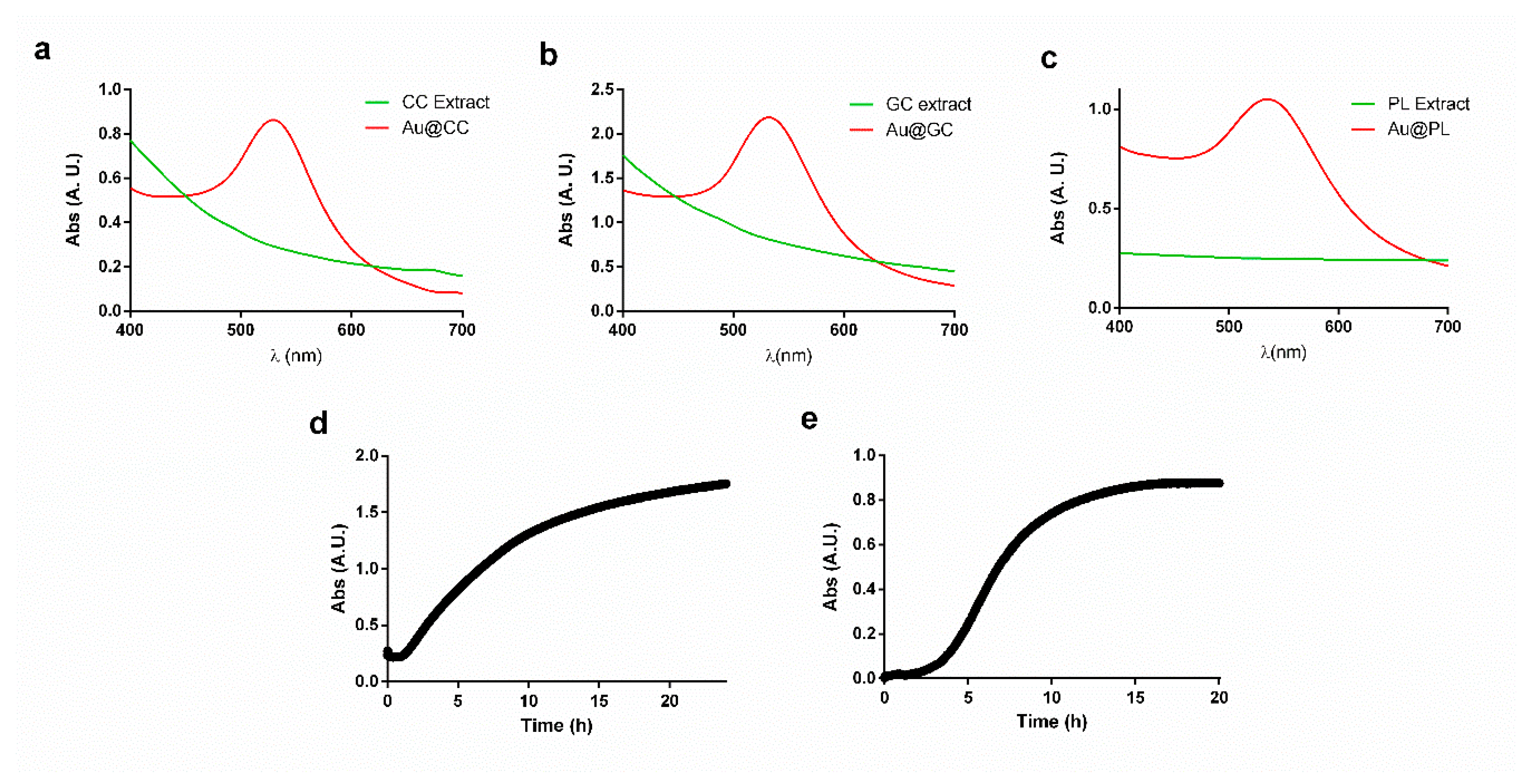
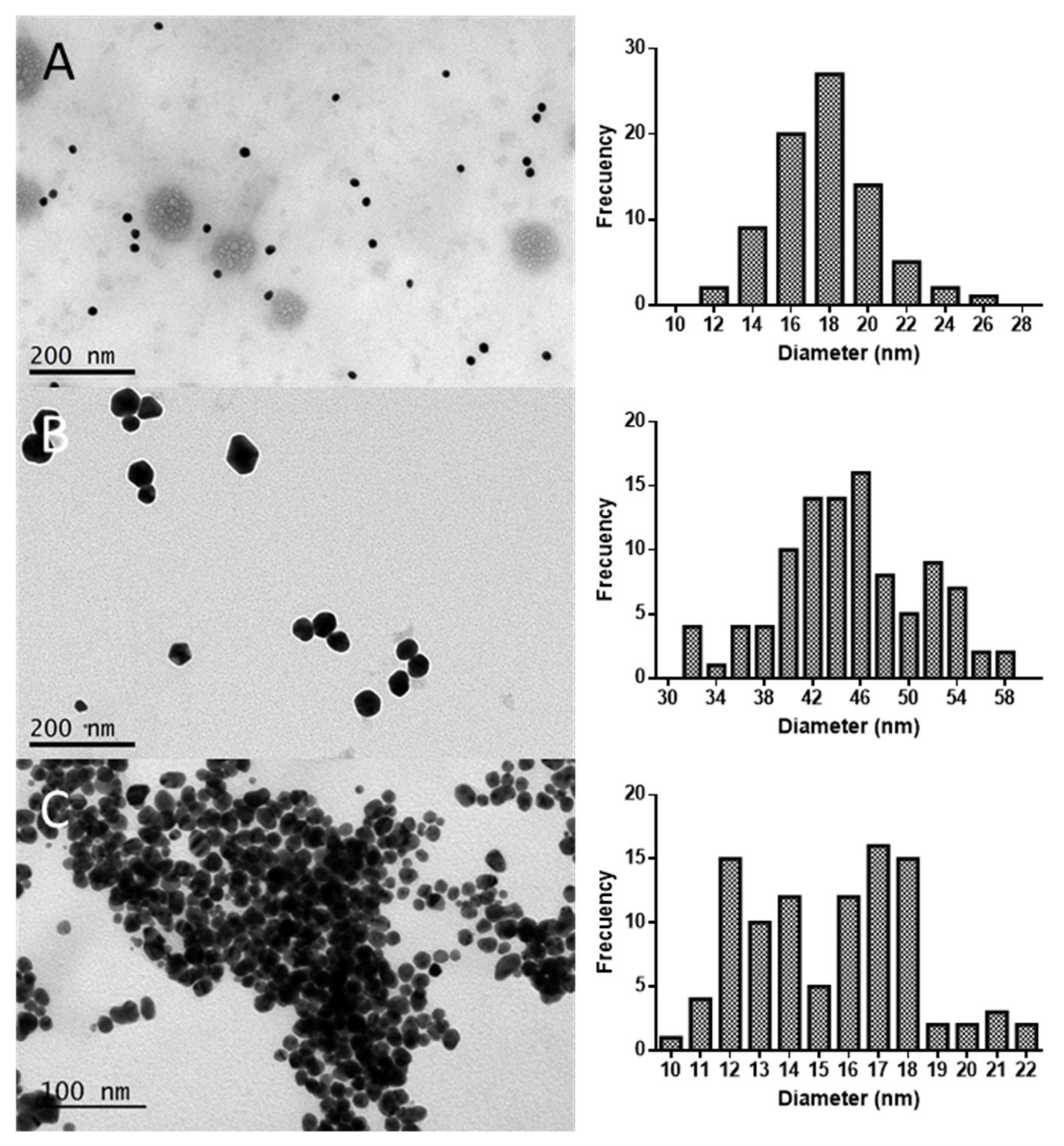
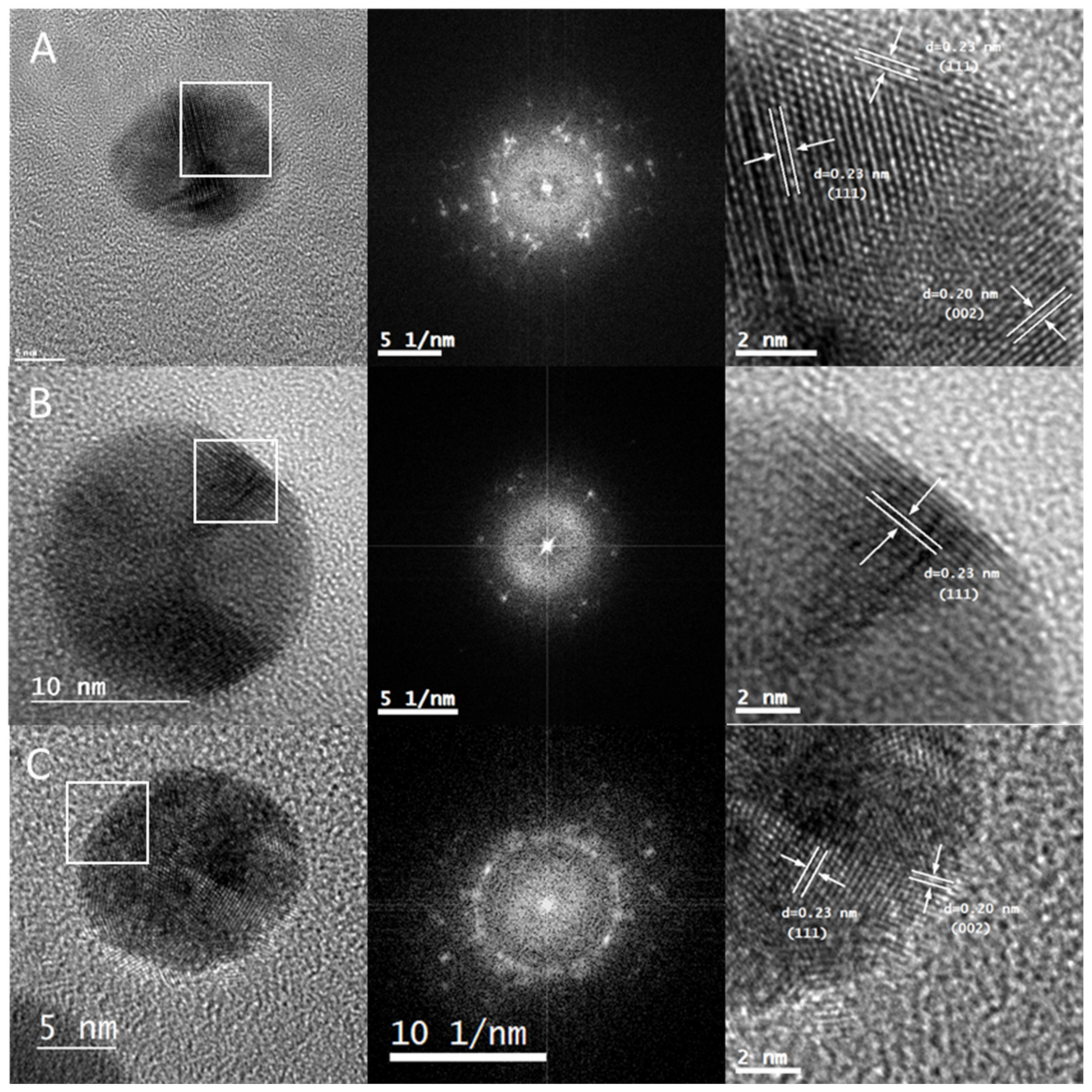
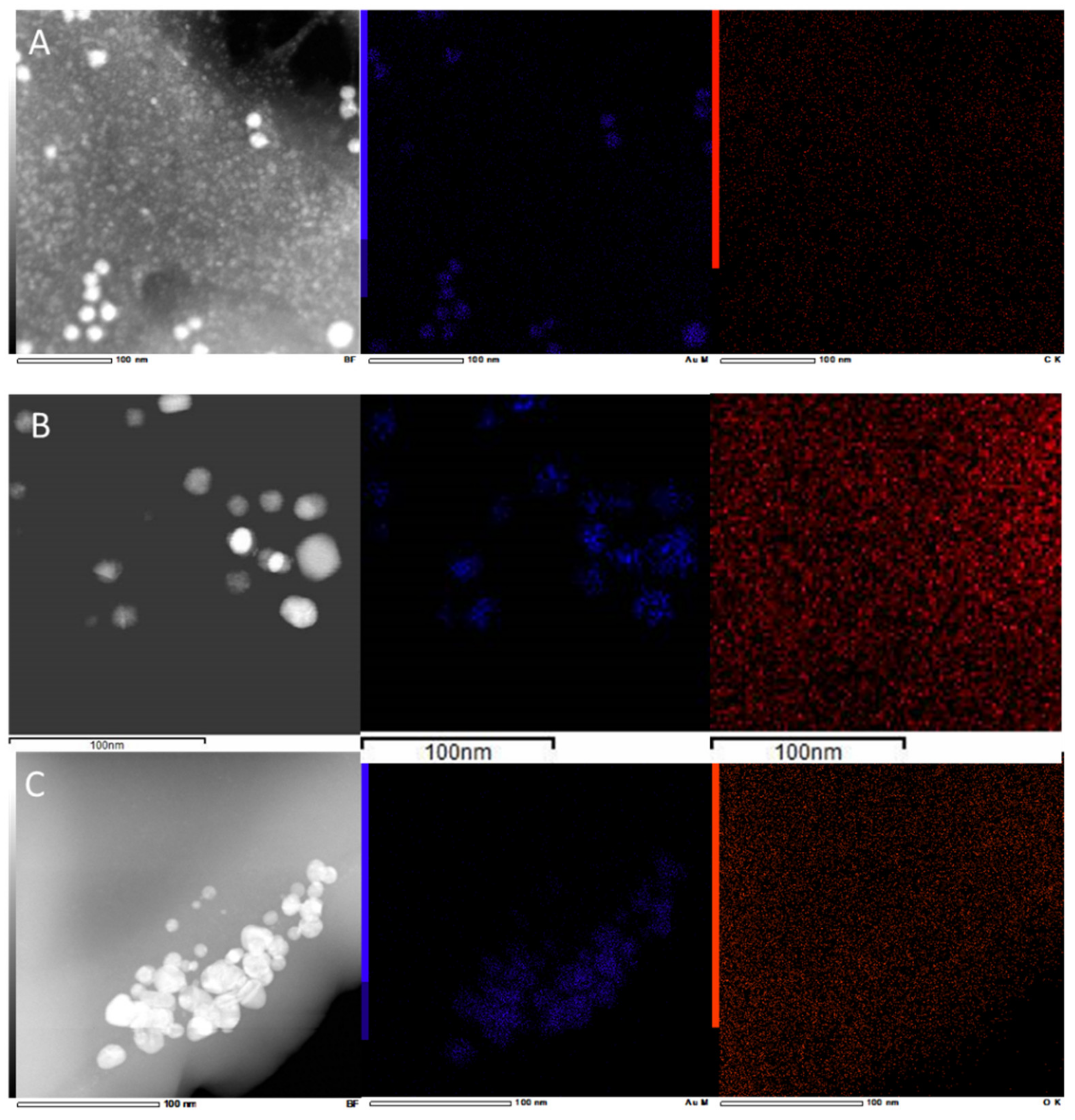
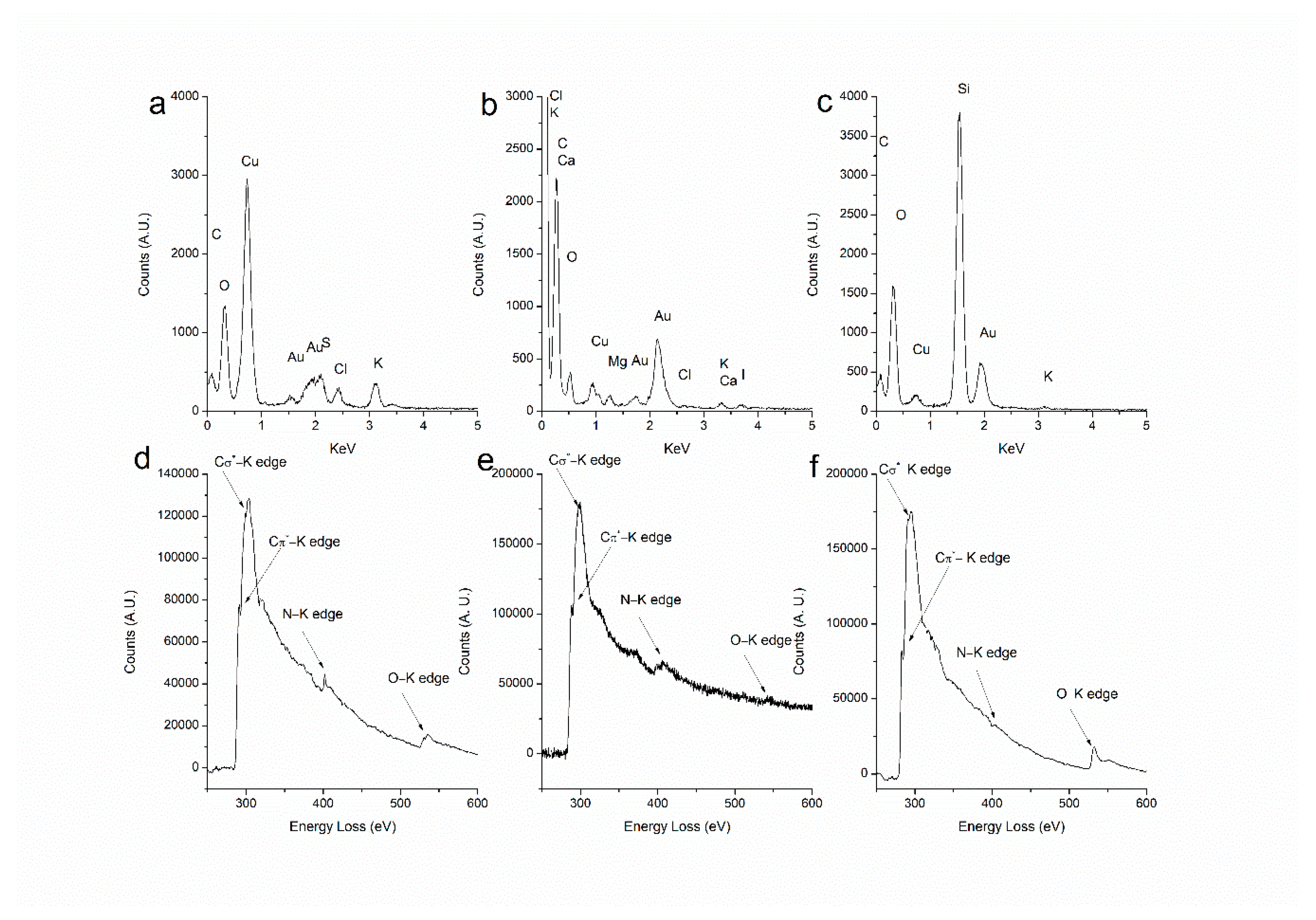
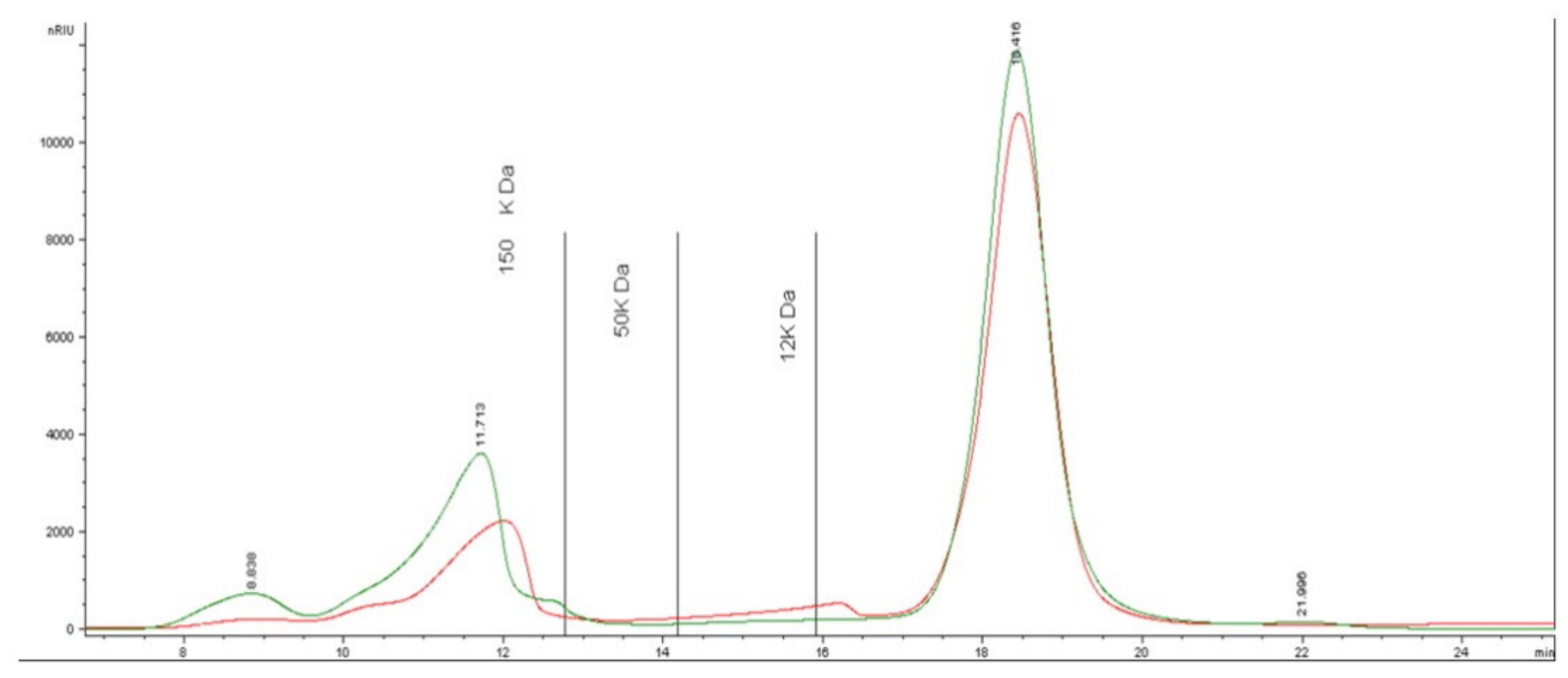
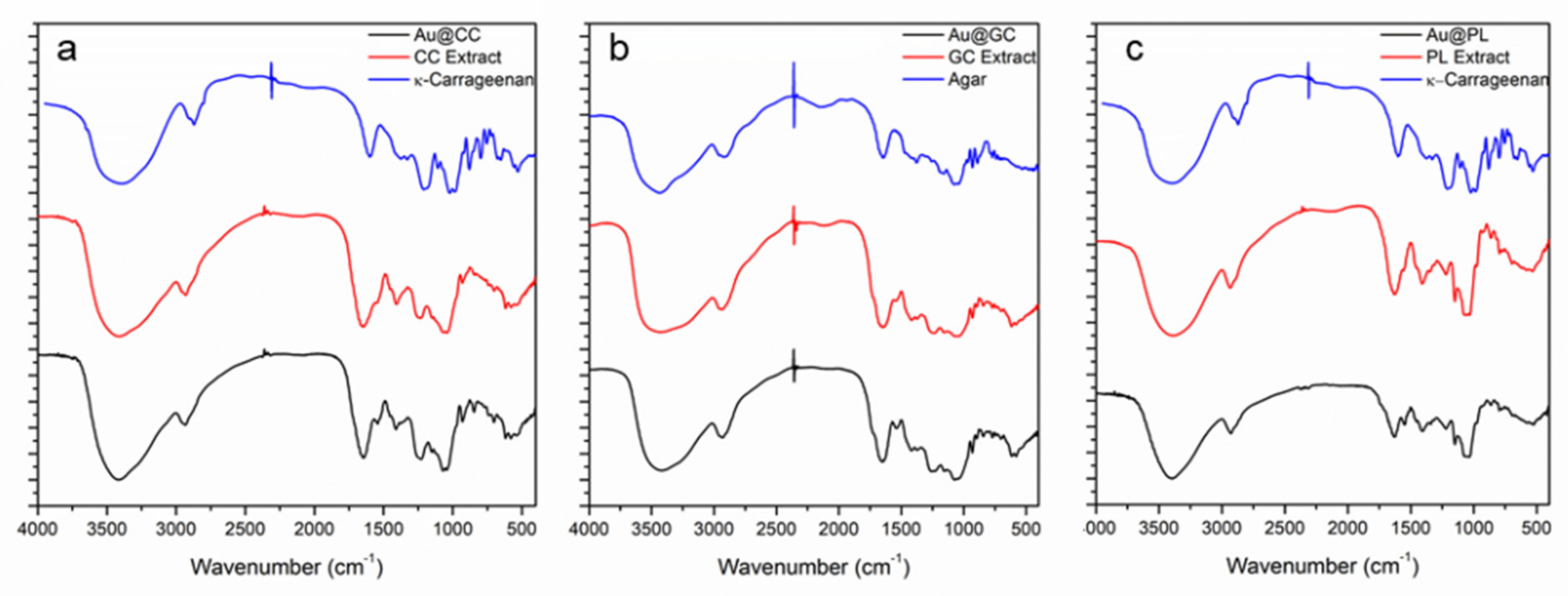
2.1. Synthesis and Characterization of AuNPs
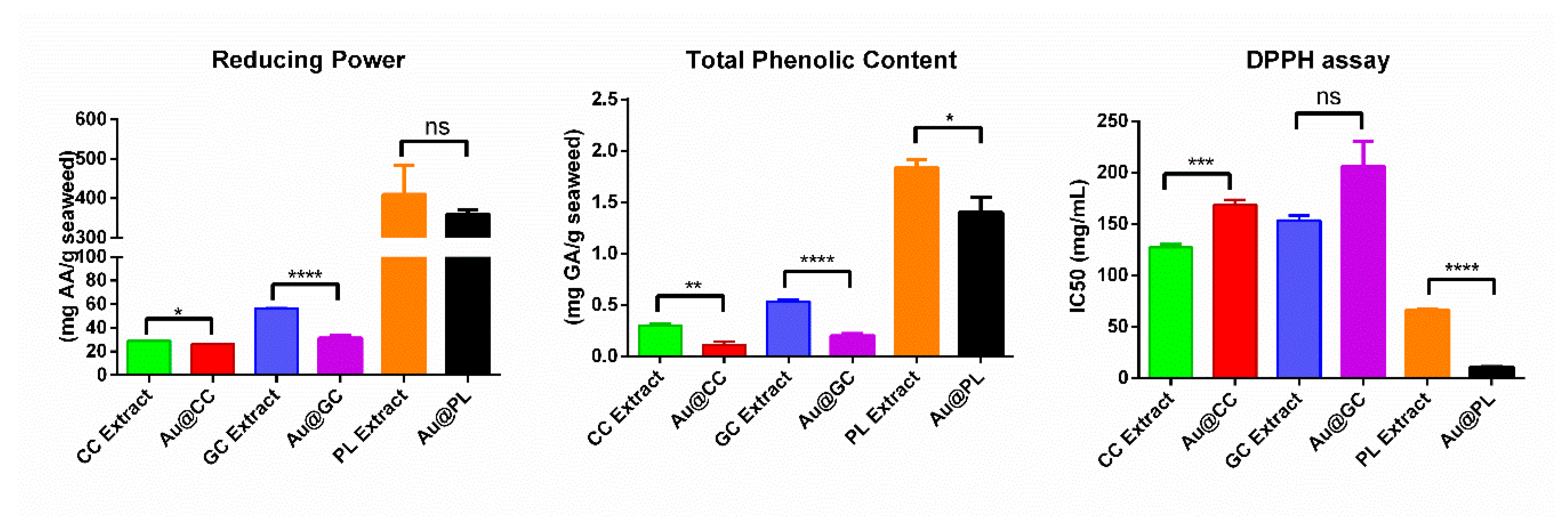
2.2. Antioxidant Activity
2.3. Evaluation of the Biological Activity In Vitro
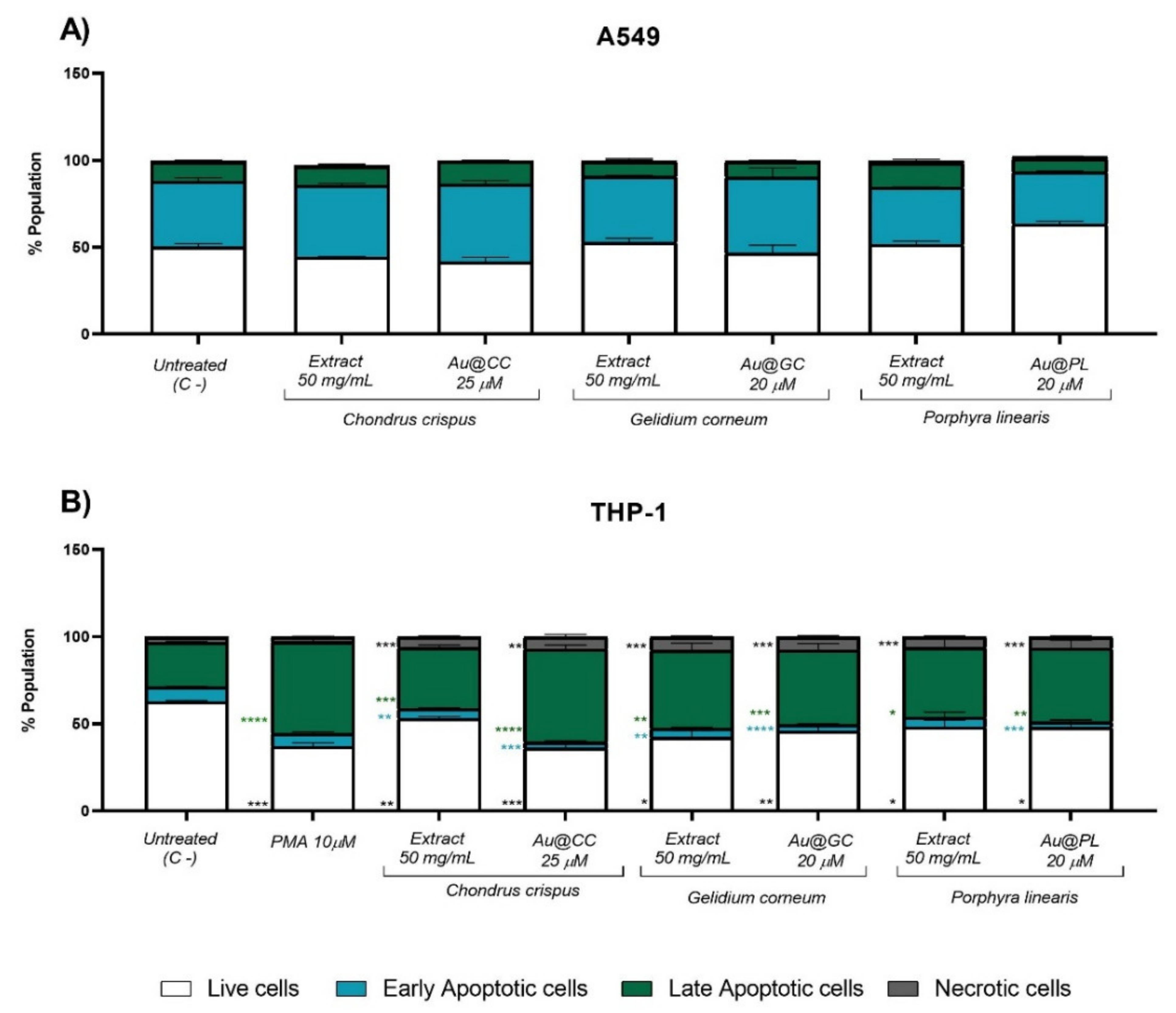
2.3.1. Antitumoral Activity in a Human Lung Epithelial and a Monocytic Cell Line
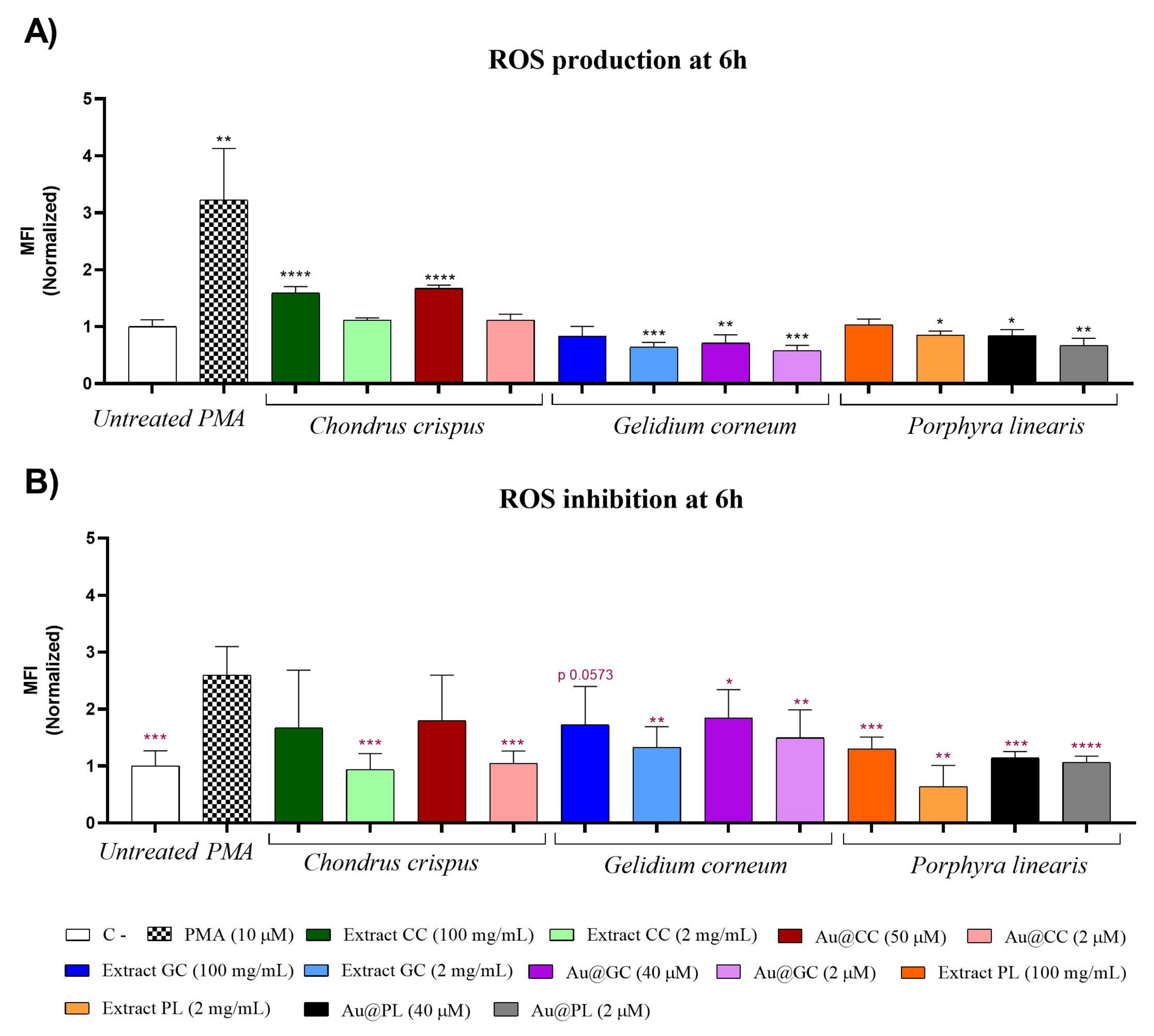
2.3.2. Antioxidant Activity in Human Promyelocytic Cells
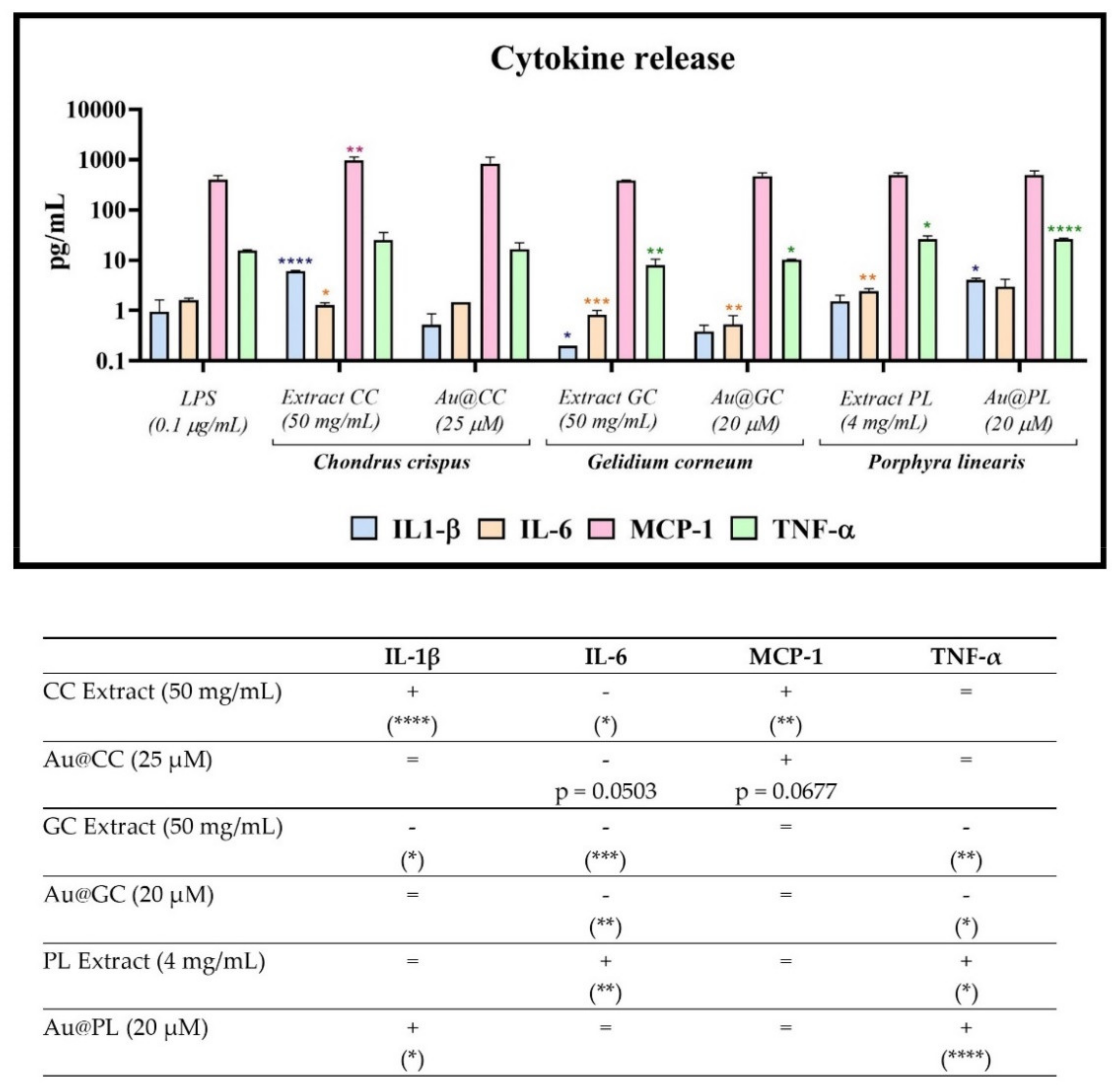
2.3.3. Immunomodulatory Activity in a Cell Model of Inflammation
3. Materials and Methods
3.1. Preparation and Characterization of Algae Extracts
3.2. Analysis of Carbohydrates
3.3. Synthesis of AuNPs
3.4. Characterization of AuNPs
3.5. Evaluation of the Antioxidant Activity
3.6. Evaluation of the Biological Activity In Vitro
3.6.1. Apoptotic Assay
3.6.2. Evaluation of the Antioxidant Activity in Cells
3.6.3. Cell Model of Inflammation and Cytokine Release
3.7. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Paidari, S.; Ibrahim, S.A. Potential Application of Gold Nanoparticles in Food Packaging: A Mini Review. Gold Bull. (World Gold Counc.) 2021, 54, 31–36. [Google Scholar] [CrossRef]
- Fu, Y.; Yin, Z.; Qin, L.; Huang, D.; Yi, H.; Liu, X.; Liu, S.; Zhang, M.; Li, B.; Li, L.; et al. Recent Progress of Noble Metals with Tailored Features in Catalytic Oxidation for Organic Pollutants Degradation. J. Hazard. Mater. 2022, 422, 126950. [Google Scholar] [CrossRef] [PubMed]
- Nejati, K.; Dadashpour, M.; Gharibi, T.; Mellatyar, H.; Akbarzadeh, A. Biomedical Applications of Functionalized Gold Nanoparticles: A Review. J. Clust. Sci. 2022, 33, 1–16. [Google Scholar] [CrossRef]
- D’Acunto, M.; Cioni, P.; Gabellieri, E.; Presciuttini, G. Exploiting Gold Nanoparticles for Diagnosis and Cancer Treatments. Nanotechnology 2021, 32, 192001. [Google Scholar] [CrossRef]
- Okkeh, M.; Bloise, N.; Restivo, E.; De Vita, L.; Pallavicini, P.; Visai, L. Gold Nanoparticles: Can they be the Next Magic Bullet for Multidrug-Resistant Bacteria? Nanomaterials 2021, 11, 312. [Google Scholar] [CrossRef]
- Agarwal, H.; Nakara, A.; Shanmugam, V.K. Anti-Inflammatory Mechanism of various Metal and Metal Oxide Nanoparticles Synthesized using Plant Extracts: A Review. Biomed. Pharmacother. 2019, 109, 2561–2572. [Google Scholar] [CrossRef]
- Brusini, R.; Varna, M.; Couvreur, P. Advanced Nanomedicines for the Treatment of Inflammatory Diseases. Adv. Drug Deliv. Rev. 2020, 157, 161–178. [Google Scholar] [CrossRef]
- Dinarello, C. Historical Insights into Cytokines. Eur. J. Immunol. 2021, 37, S34–S45. [Google Scholar] [CrossRef] [Green Version]
- Berraondo, P.; Sanmamed, M.F.; Ochoa, M.C.; Etxeberria, I.; Aznar, M.A.; Pérez-Gracia, J.L.; Rodríguez-Ruiz, M.E.; Ponz-Sarvise, M.; Castañón, E.; Melero, I. Cytokines in Clinical Cancer Immunotherapy. Br. J. Cancer 2019, 120, 6–15. [Google Scholar] [CrossRef] [Green Version]
- Wraith, D.C. The Future of Immunotherapy: A 20-Year Perspective. Front. Immunol. 2017, 8, 1668. [Google Scholar] [CrossRef] [Green Version]
- Pulendran, B.; Arunachalam, P.S.; O’Hagan, D.T. Emerging Concepts in the Science of Vaccine Adjuvants. Nat. Rev. Drug Discov. 2021, 20, 454–475. [Google Scholar] [CrossRef] [PubMed]
- Kotsafti, A.; Scarpa, M.; Castagliuolo, I.; Scarpa, M. Reactive Oxygen Species and Antitumor Immunity—From Surveillance to Evasion. Cancers 2020, 12, 1748. [Google Scholar] [CrossRef] [PubMed]
- Ferdous, U.T.; Yusof, Z.N.B. Medicinal Prospects of Antioxidants from Algal Sources in Cancer Therapy. Front. Pharmacol. 2021, 12, 157. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, M.S. Improving Cancer Immunotherapy through Nanotechnology. Nat. Rev. Cancer 2019, 19, 587–602. [Google Scholar] [CrossRef] [PubMed]
- Cai, F.; Li, S.; Huang, H.H.; Iqbal, J.J.; Wang, C.; Jiang, X. Green Synthesis of Gold Nanoparticles for Immune Response Regulation: Mechanisms, Applications, and Perspectives. J. Biomed. Mater. Res. 2021, 110, 424. [Google Scholar] [CrossRef]
- Madani, M.; Hosny, S.; Alshangiti, D.M.; Nady, N.; Alkhursani, S.A.; Alkhaldi, H.; Al-Gahtany, S.A.; Ghobashy, M.M.; Gaber, G.A. Green Synthesis of Nanoparticles for Varied Applications: Green Renewable Resources and Energy-Efficient Synthetic Routes. Nanotechnol. Rev. 2022, 11, 731–759. [Google Scholar] [CrossRef]
- Ahmed, S.F.; Mofijur, M.; Rafa, N.; Chowdhury, A.T.; Chowdhury, S.; Nahrin, M.; Islam, A.B.M.S.; Ong, H.C. Green Approaches in Synthesising Nanomaterials for Environmental Nanobioremediation: Technological Advancements, Applications, Benefits and Challenges. Environ. Res. 2022, 204, 111967. [Google Scholar] [CrossRef]
- Pushparaj, K.; Liu, W.; Meyyazhagan, A.; Orlacchio, A.; Pappusamy, M.; Vadivalagan, C.; Robert, A.A.; Arumugam, V.A.; Kamyab, H.; Klemeš, J.J.; et al. Nano- from Nature to Nurture: A Comprehensive Review on Facets, Trends, Perspectives and Sustainability of Nanotechnology in the Food Sector. Energy 2022, 240, 122732. [Google Scholar] [CrossRef]
- González-Ballesteros, N.; Prado-López, S.; Rodríguez-González, J.B.; Lastra, M.; Rodríguez-Argüelles, M.C. Green Synthesis of Gold Nanoparticles using Brown Algae Cystoseira baccata: Its Activity in Colon Cancer Cells. Colloids Surf. B Biointerfaces 2017, 153, 190–198. [Google Scholar] [CrossRef]
- González-Ballesteros, N.; Diego-González, L.; Lastra-Valdor, M.; Grimaldi, M.; Cavazza, A.; Bigi, F.; Rodríguez-Argüelles, M.C.; Simón-Vázquez, R. Saccorhiza polyschides used to Synthesize Gold and Silver Nanoparticles with Enhanced Antiproliferative and Immunostimulant Activity. Mater. Sci. Eng. C 2021, 123, 111960. [Google Scholar] [CrossRef]
- González-Ballesteros, N.; Diego-González, L.; Lastra-Valdor, M.; Rodríguez-Argüelles, M.C.; Grimaldi, M.; Cavazza, A.; Bigi, F.; Simón-Vázquez, R. Immunostimulant and Biocompatible Gold and Silver Nanoparticles Synthesized by the Ulva intestinalis L. Aqueous Extract. J. Mater. Chem. B 2019, 7, 4677–4691. [Google Scholar] [CrossRef] [PubMed]
- González-Ballesteros, N.; Rodríguez-Argüelles, M.C.; Prado-López, S.; Lastra, M.; Grimaldi, M.; Cavazza, A.; Nasi, L.; Salviati, G.; Bigi, F. Macroalgae to Nanoparticles: Study of Ulva lactuca Role in Biosynthesis of Gold and Silver Nanoparticles and of their Cytotoxicity on Colon Cancer Cell Lines. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 97, 498–509. [Google Scholar] [CrossRef] [PubMed]
- Aziz, E.; Batool, R.; Khan, M.U.; Rauf, A.; Akhtar, W.; Heydari, M.; Rehman, S.; Shahzad, T.; Malik, A.; Mosavat, S.H.; et al. An Overview on Red Algae Bioactive Compounds and their Pharmaceutical Applications. J. Complement. Integr. Med. 2020, 17, 20190203. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A.G.; Fraga-Corral, M.; Garcia-Oliveira, P.; Lourenço-Lopes, C.; Carpena, M.; Prieto, M.A.; Simal-Gandara, J. The use of Invasive Algae Species as a Source of Secondary Metabolites and Biological Activities: Spain as Case-Study. Mar. Drugs 2021, 19, 178. [Google Scholar] [CrossRef]
- Lee, H.; Selvaraj, B.; Lee, J.W. Anticancer Effects of Seaweed-Derived Bioactive Compounds. Appl. Sci. 2021, 11, 11261. [Google Scholar] [CrossRef]
- Nag, M.; Lahiri, D.; Dey, A.; Sarkar, T.; Joshi, S.; Ray, R.R. Evaluation of Algal Active Compounds as Potent Antibiofilm Agent. J. Basic Microbiol. 2021. [Google Scholar] [CrossRef]
- Cotas, J.; Leandro, A.; Pacheco, D.; Gonçalves, A.M.M.; Pereira, L. A Comprehensive Review of the Nutraceutical and Therapeutic Applications of Red Seaweeds (Rhodophyta). Life 2020, 10, 19. [Google Scholar] [CrossRef] [Green Version]
- Ismail, M.M.; Alotaibi, B.S.; EL-Sheekh, M. Therapeutic Uses of Red Macroalgae. Molecules 2020, 25, 4411. [Google Scholar] [CrossRef]
- Hwang, J.; Yadav, D.; Lee, P.C.; Jin, J.O. Immunomodulatory Effects of Polysaccharides from Marine Algae for Treating Cancer, Infectious Disease, and Inflammation. Phytother. Res. 2021, 36, 761–777. [Google Scholar] [CrossRef]
- Collén, J.; Cornish, M.L.; Craigie, J.; Ficko-Blean, E.; Hervé, C.; Krueger-Hadfield, S.A.; Leblanc, C.; Michel, G.; Potin, P.; Tonon, T.; et al. Chondrus crispus—A Present and Historical Model Organism for Red Seaweeds. Adv. Bot. Res. 2014, 71, 53–89. [Google Scholar]
- Sangha, J.S.; Wally, O.; Banskota, A.H.; Stefanova, R.; Hafting, J.T.; Critchley, A.T.; Prithiviraj, B. A Cultivated Form of a Red Seaweed (Chondrus crispus), Suppresses Β-Amyloid-Induced Paralysis in Caenorhabditis elegans. Mar. Drugs 2015, 13, 6407–6424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rudtanatip, T.; Lynchc, S.A.; Wongprasert, K.; Culloty, S.C. Assessment of the Effects of Sulfated Polysaccharides Extracted from the Red Seaweed Irish Moss Chondrus crispus on the Immune-Stimulant Activity in Mussels Mytilus spp. Fish Shellfish Immunol. 2018, 75, 284–290. [Google Scholar] [CrossRef] [PubMed]
- Torres, M.D.; Flórez-Fernández, N.; Domínguez, H. Chondrus crispus Treated with Ultrasound as a Polysaccharides Source with Improved Antitumoral Potential. Carbohydr. Polym. 2021, 273, 118588. [Google Scholar] [CrossRef] [PubMed]
- Alkhalaf, M.I. Chemical Composition, Antioxidant, Anti-Inflammatory and Cytotoxic Effects of Chondrus crispus Species of Red Algae Collected from the Red Sea Along the Shores of Jeddah City. J. King Saud University Sci. 2021, 33, 101210. [Google Scholar] [CrossRef]
- Quintano, E.; Díez, I.; Muguerza, N.; Santolaria, A.; Gorostiaga, J.M. Epiphytic Flora on Gelidium corneum (Rhodophyta: Gelidiales) in Relation to Wave Exposure and Depth. Sci. Mar. 2015, 79, 479–486. [Google Scholar] [CrossRef]
- Yoon, M.H.; Lee, Y.W.; Lee, H.C.; Seo, Y.B. Simultaneous Production of Bio-Ethanol and Bleached Pulp from Red Algae. Bioresour. Technol. 2012, 126, 198–201. [Google Scholar] [CrossRef]
- Lim, G.O.; Jang, S.A.; Song, K.B. Physical and Antimicrobial Properties of Gelidium corneum/Nano-Clay Composite Film Containing Grapefruit Seed Extract or Thymol. J. Food Eng. 2010, 98, 415–420. [Google Scholar] [CrossRef]
- Jang, S.; Lim, G.; Song, K.B. Use of Nano-Clay Improves Tensile Strength and Vapour Permeability in Agar Rich Red Algae (Gelidium corneum)-Gelatin Composite Films. Int. J. Food Sci. Technol. 2010, 45, 1883. [Google Scholar] [CrossRef]
- Abdala Díaz, R.T.; Casas Arrojo, V.; Arrojo Agudo, M.A.; Cárdenas, C.C.; Dobretsov, S.S.; Figueroa, F.L. Immunomodulatory and Antioxidant Activities of Sulfated Polysaccharides from Laminaria ochroleuca, Porphyra umbilicalis, and Gelidium corneum. Mar. Biotechnol. 2019, 21, 577–587. [Google Scholar] [CrossRef]
- Varela-Álvarez, E.; Stengel, D.B.; Guiry, M.D. Seasonal Growth and Phenotypic Variation in Porphyra linearis (Rhodophyta) Populations on the West Coast of Ireland. J. Phycol. 2007, 43, 90–100. [Google Scholar] [CrossRef]
- Sanchez, N.; Vergés, A.; Peteiro, C.; Sutherland, J.E.; Brodie, J. Diversity of Bladed Bangiales (Rhodophyta) in Western Mediterranean: Recognition of the Genus Themis and Descriptions of T. ballesterosii Sp. Nov., T. iberica Sp. Nov., and Pyropia parva Sp. Nov. J. Phycol. 2014, 50, 908–929. [Google Scholar] [CrossRef] [PubMed]
- Varela-Álvarez, E.; Paulino, C.; Serrão, E.A. Development and Characterization of Twelve Microsatellite Markers for Porphyra linearis Greville. Genetica 2017, 145, 127–130. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Xue, Y. Optimization of Microwave Assisted Extraction, Chemical Characterization and Antitumor Activities of Polysaccharides from Porphyra haitanensis. Carbohydr. Polym. 2019, 206, 179–186. [Google Scholar] [CrossRef]
- Castro, L.; Blázquez, M.L.; Muñoz, J.A.; González, F.; Ballester, A. Biological Synthesis of Metallic Nanoparticles using Algae. IET Nanobiotechnol. 2013, 7, 109–116. [Google Scholar] [CrossRef]
- Murphin Kumar, P.S.; MubarakAlic, D.; Saratalee, R.G.; Saratale, G.D.; Pugazhendhig, A.; Gopalakrishnanh, K.; Thajuddind, N. Synthesis of Nano-Cuboidal Gold Particles for Effective Antimicrobial Property Against Clinical Human Pathogens. Microb. Pathog. 2017, 113, 68–73. [Google Scholar] [CrossRef]
- Tibbetts, S.M.; Milley, J.E.; Lall, S.P. Nutritional Quality of some Wild and Cultivated Seaweeds: Nutrient Composition, Total Phenolic Content and In Vitro Digestibility. J. Appl. Phycol. 2016, 28, 3575–3585. [Google Scholar] [CrossRef]
- Din, N.G.S.; El-Sherif, Z.M. Nutritional Value of some Algae from the North-Western Mediterranean Coast of Egypt. J. Appl. Phycol. 2012, 24, 613–626. [Google Scholar] [CrossRef]
- Cian, R.E.; Fajardo, M.A.; Alaiz, M.; Vioque, J.; González, R.J.; Drago, S.R. Chemical Composition, Nutritional and Antioxidant Properties of the Red Edible Seaweed Porphyra columbina. Int. J. Food. Sci. Nutr. 2014, 65, 299–305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gómez-Ordóñez, E.; Jiménez-Escrig, A.; Rupérez, P. Molecular Weight Distribution of Polysaccharides from Edible Seaweeds by High-Performance Size-Exclusion Chromatography (HPSEC). Talanta 2012, 93, 153–159. [Google Scholar] [CrossRef] [Green Version]
- Usov, A.I. Chapter 4—Polysaccharides of the red algae. In Advances in Carbohydrate Chemistry and Biochemistry; Horton, D., Ed.; Academic Press: Cambridge, MA, USA, 2011; Volume 65, pp. 115–217. [Google Scholar]
- Stengel, D.B.; Connan, S.; Popper, Z.A. Algal Chemodiversity and Bioactivity: Sources of Natural Variability and Implications for Commercial Application. Biotechnol. Adv. 2011, 29, 483–501. [Google Scholar] [CrossRef]
- Guerrero, P.; Etxabide, A.; Leceta, I.; Peñalba, M.; De la Caba, K. Extraction of Agar from Gelidium sesquipedale (Rodhopyta) and Surface Characterization of Agar Based Films. Carbohydr. Polym. 2014, 99, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Pereira, L.; Gheda, S.F.; Ribeiro-Claro, P.J.A. Analysis by Vibrational Spectroscopy of Seaweed Polysaccharides with Potential use in Food, Pharmaceutical, and Cosmetic Industries. Int. J. Carbohydr. Chem. 2013, 2013, 537202. [Google Scholar] [CrossRef]
- Dong, M.; Xue, Z.; Liu, J.; Yan, M.; Xia, Y.; Wang, B. Preparation of Carrageenan Fibers with Extraction of Chondrus Via Wet Spinning Process. Carbohydr. Polym. 2018, 194, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Pereira, L.; Amado, A.M.; Critchley, A.T.; Velde, F.V.; Ribeiro-Claro, P. Identification of Selected Seaweed Polysaccharides (Phycocolloids) by Vibrational Spectroscopy (FTIR-ATR and FT-Raman). Food Hydrocoll. 2009, 23, 1903–1909. [Google Scholar] [CrossRef] [Green Version]
- Pereira, L. Population Studies and Carrageenan Properties in Eight Gigartinales (Rhodophyta) from Western Coast of Portugal. Sci. World J. 2013, 2013, 939830. [Google Scholar] [CrossRef] [Green Version]
- Chew, K.W.; Show, P.L.; Yap, Y.J.; Juan, J.C.; Phang, S.M.; Ling, T.C.; Chang, J.S. Sonication and Grinding Pre-Treatments on Gelidium amansii Seaweed for the Extraction and Characterization of Agarose. Front. Environ. Sci. Eng. 2018, 12, 2. [Google Scholar] [CrossRef]
- Rajasulochana, N.; Gunasekaran, S. Fourier Transform Infrared Spectral Analysis of Agars of Gelidium micropterum and Gracilaria fergusonii. Asian J. Chem. 2009, 21, 4543–4546. [Google Scholar]
- Pugazhendhi, A.; Prabakar, D.; Jacob, J.M.; Karuppusamy, I.; Saratale, R.G. Synthesis and Characterization of Silver Nanoparticles using Gelidium amansii and its Antimicrobial Property Against various Pathogenic Bacteria. Microb. Pathog. 2018, 114, 41–45. [Google Scholar] [CrossRef]
- Bhatiaa, S.; Ratheea, P.; Sharmab, K.; Chaugulec, B.B.; Kard, N.; Berad, T. Immuno-Modulation Effect of Sulphated Polysaccharide (Porphyran) from Porphyra vietnamensis. Int. J. Biol. Macromol. 2013, 57, 50–56. [Google Scholar] [CrossRef]
- Zhang, Q.; Qi, H.; Zhao, T.; Deslandes, E.; Ismaeli, N.M.; Molloyd, F.; Critchley, A.T. Chemical Characteristics of a Polysaccharide from Porphyra capensis (Rhodophyta). Carbohydr. Res. 2005, 340, 2447–2450. [Google Scholar] [CrossRef]
- González-Ballesteros, N.; Rodríguez-Argüelles, M.C.; Lastra-Valdor, M. Evaluation of the Antioxidant Capacities of Antarctic Macroalgae and their use for Nanoparticles Production. Molecules 2021, 26, 1182. [Google Scholar] [CrossRef] [PubMed]
- González-Ballesteros, N.; Torres, M.D.; Flórez-Fernández, N.; Diego-González, L.; Simón-Vázquez, R.; Rodríguez-Argüelles, M.C.; Domínguez, H. Eco-Friendly Extraction of Mastocarpus stellatus Carrageenan for the Synthesis of Gold Nanoparticles with Improved Biological Activity. Int. J. Biol. Macromol. 2021, 183, 1436–1449. [Google Scholar] [CrossRef] [PubMed]
- Varshosaz, J.; Zaki, M.R.; Minaiyan, M.; Banoozadeh, J. Preparation, Optimization, and Screening of the Effect of Processing Variables on Agar Nanospheres Loaded with Bupropion HCl by a D-Optimal Design. BioMed Res. Int. 2015, 2015, 571816. [Google Scholar] [CrossRef]
- González Ballesteros, N.; Rodríguez-Argüelles, M.C.; Lastra-Valdor, M.; González-Mediero, G.; Rey-Cao, S.; Grimaldi, M.; Cavazza, A.; Bigi, F. Synthesis of Silver and Gold Nanoparticles by Sargassum muticum Biomolecules and Evaluation of their Antioxidant Activity and Antibacterial Properties. J. Nanostruct. Chem. 2020, 10, 317–330. [Google Scholar] [CrossRef]
- Takei, M.; Kuda, T.; Eda, M.; Shikano, A.; Takahashi, H.; Kimura, B. Antioxidant and Fermentation Properties of Aqueous Solutions of Dried Algal Products from the Boso Peninsula, Japan. Food Biosci. 2017, 19, 85–91. [Google Scholar] [CrossRef]
- Farvin, K.H.S.; Jacobsen, C. Phenolic Compounds and Antioxidant Activities of Selected Species of Seaweeds from Danish Coast. Food Chem. 2013, 138, 1670–1681. [Google Scholar] [CrossRef]
- Lee, J.H.; Kim, G.H. Evaluation of Antioxidant Activity of Marine Algae-Extracts from Korea. J. Aquat. Food Prod. Technol. 2015, 24, 227–240. [Google Scholar] [CrossRef]
- Zhang, W.W.; Duan, X.J.; Huang, H.L.; Zhang, Y.; Wang, B.G. Evaluation of 28 Marine Algae from the Qingdao Coast for Antioxidative Capacity and Determination of Antioxidant Efficiency and Total Phenolic Content of Fractions and Subfractions Derived from Symphyocladia latiuscula (Rhodomelaceae). J. Appl. Phycol. 2007, 19, 97–108. [Google Scholar] [CrossRef]
- Nunes, N.; Ferraz, S.; Valente, S.; Barreto, M.C.; Pinheiro, D.C. Biochemical Composition, Nutritional Value, and Antioxidant Properties of Seven Seaweed Species from the Madeira Archipelago. J. Appl. Phycol. 2017, 29, 2427–2437. [Google Scholar] [CrossRef]
- Cho, S.H.; Kang, S.E.; Cho, J.Y.; Kim, A.R.; Park, S.M.; Hong, Y.K.; Ahn, D.H. The Antioxidant Properties of Brown Seaweed (Sargassum siliquastrum) Extracts. J. Med. Food 2007, 10, 479–485. [Google Scholar] [CrossRef]
- Jiménez-Escrig, A.; Jiménez-Jiménez, I.; Pulido, R.; Saura-Calixto, F. Antioxidant Activity of Fresh and Processed Edible Seaweeds. J. Sci. Food Agric. 2001, 534, 530–534. [Google Scholar] [CrossRef]
- Lee, O.H.; Yoon, K.Y.; Kim, K.J.; You, S.G.; Lee, B.Y. Seaweed Extracts as a Potential Tool for the Attenuation of Oxidative Damage in Obesity-Related Pathologies. J. Phycol. 2011, 47, 548–556. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Jónsdóttir, R.; Ólafsdóttir, G. Total Phenolic Compounds, Radical Scavenging and Metal Chelation of Extracts from Icelandic Seaweeds. Food Chem. 2009, 116, 240–248. [Google Scholar] [CrossRef]
- Pinteus, S.; Silva, J.; Alves, C.; Horta, A.; Fino, N.; Rodrigues, A.I.; Mendes, S.; Pedrosa, R. Cytoprotective Effect of Seaweeds with High Antioxidant Activity from the Peniche Coast (Portugal). Food Chem. 2017, 218, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Jan, R.; Chaudhry, G.-E.-S. Understanding Apoptosis and Apoptotic Pathways Targeted Cancer Therapeutics. Adv. Pharm. Bull. 2019, 9, 205–218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Babu, B.; Palanisamy, S.; Vinosha, M.; Anjali, R.; Kumar, P.; Pandi, B.; Tabarsa, M.; You, S.; Prabhu, N.M. Bioengineered Gold Nanoparticles from Marine Seaweed Acanthophora spicifera for Pharmaceutical Uses: Antioxidant, Antibacterial, and Anticancer Activities. Bioprocess Biosyst. Eng. 2020, 43, 2231–2242. [Google Scholar] [CrossRef]
- El-Kassas, H.Y.; El-Sheekh, M.M. Cytotoxic Activity of Biosynthesized Gold Nanoparticles with an Extract of the Red Seaweed Corallina Officinalis on the MCF-7 Human Breast Cancer Cell Line. Asian Pac. J. Cancer Prev. 2014, 15, 4311–4317. [Google Scholar] [CrossRef] [Green Version]
- Sies, H.; Jones, D.P. Reactive Oxygen Species (ROS) as Pleiotropic Physiological Signalling Agents. Nat. Rev. Mol. Cell Biol. 2020, 21, 363–383. [Google Scholar] [CrossRef]
- Aghamirzaei, M.; Khiabani, M.S.; Hamishehkar, H.; Mokaram, R.R.; Amjadi, M. Antioxidant, Antimicrobial and Cytotoxic Activities of Biosynthesized Gold Nanoparticles (AuNPs) from Chinese Lettuce (CL) Leave Extract (Brassica rapa Var. Pekinensis). Mater. Today Commun. 2021, 29, 102831. [Google Scholar] [CrossRef]
- Pu, S.; Li, J.; Sun, L.; Zhong, L.; Ma, Q. An in Vitro Comparison of the Antioxidant Activities of Chitosan and Green Synthesized Gold Nanoparticles. Carbohydr. Polym. 2019, 211, 161–172. [Google Scholar] [CrossRef]
- Choy, E.H.; De Benedetti, F.; Takeuchi, T.; Hashizume, M.; John, M.R.; Kishimoto, T. Translating IL-6 Biology into Effective Treatments. Nat. Rev. Rheumatol. 2020, 16, 335–345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Monaco, C.; Nanchahal, J.; Taylor, P.; Feldmann, M. Anti-TNF Therapy: Past, Present and Future. Int. Immunol. 2015, 27, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Campbell, L.; Chen, C.; Bhagat, S.S.; Parker, R.A.; Östör, A.J.K. Risk of Adverse Events Including Serious Infections in Rheumatoid Arthritis Patients Treated with Tocilizumab: A Systematic Literature Review and Meta-Analysis of Randomized Controlled Trials. Rheumatol. 2011, 50, 552–562. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernández-Ruiz, M.; Aguado, J.M. Risk of Infection Associated with Anti-TNF-A Therapy. Expert Rev. Anti-Infect. Ther. 2018, 16, 939–956. [Google Scholar] [CrossRef] [PubMed]
- Trier, N.H.; Güven, E.; Skogstrand, K.; Ciplys, E.; Slibinskas, R.; Houen, G. Comparison of Immunological Adjuvants. APMIS 2021, 127, 635–641. [Google Scholar] [CrossRef]
- Yamashita, M.; Passegué, E. TNF-A Coordinates Hematopoietic Stem Cell Survival and Myeloid Regeneration. Cell Stem Cell 2019, 25, 357–372.e7. [Google Scholar] [CrossRef] [Green Version]
- Chang, S.N.; Dey, D.K.; Oh, S.T.; Kong, W.H.; Cho, K.H.; Al-Olayan, E.M.; Hwang, B.S.; Kang, S.C.; Park, J.G. Phorbol 12-Myristate 13-Acetate Induced Toxicity Study and the Role of Tangeretin in Abrogating HIF-1α-NF-κB Crosstalk in Vitro and in Vivo. Int. J. Mol. Sci. 2020, 21, 9261. [Google Scholar] [CrossRef]
- Chen, C.; Kao, C.; Liu, C. The Cancer Prevention, Anti-Inflammatory and Anti-Oxidation of Bioactive Phytochemicals Targeting the TLR4 Signaling Pathway. Int. J. Mol. Sci. 2018, 19, 2729. [Google Scholar] [CrossRef] [Green Version]
- González-Ballesteros, N.; Rodríguez-González, J.B.; Lastra-Valdor, M.; Rodríguez-Argüelles, M.C. New Application of Two Antarctic Macroalgae Palmaria decipiens and Desmarestia menziesii in the Synthesis of Gold and Silver Nanoparticles. Polar Sci. 2018, 15, 49–54. [Google Scholar] [CrossRef]
- Diego-González, L.; Crecente-Campo, J.; Paul, M.J.; Singh, M.; Reljic, R.; Alonso, M.J.; González-Fernández, Á.; Simón-Vázquez, R. Design of Polymeric Nanocapsules for Intranasal Vaccination Against Mycobacterium Tuberculosis: Influence of the Polymeric Shell and Antigen Positioning. Pharmaceutics 2020, 12, 489. [Google Scholar] [CrossRef]
| Algae | [Extract] (g/mL) | [Au] (mM) | T (°C) | t (h) | Code |
|---|---|---|---|---|---|
| C. crispus | 1 | 0.5 | 30 | 48 | Au@CC |
| G. corneum | 1 | 0.4 | 30 | 24 | Au@GC |
| P. linearis | 0.08 | 0.4 | 30 | 24 | Au@PL |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Ballesteros, N.; Diego-González, L.; Lastra-Valdor, M.; Grimaldi, M.; Cavazza, A.; Bigi, F.; Rodríguez-Argüelles, M.C.; Simón-Vázquez, R. Immunomodulatory and Antitumoral Activity of Gold Nanoparticles Synthesized by Red Algae Aqueous Extracts. Mar. Drugs 2022, 20, 182. https://doi.org/10.3390/md20030182
González-Ballesteros N, Diego-González L, Lastra-Valdor M, Grimaldi M, Cavazza A, Bigi F, Rodríguez-Argüelles MC, Simón-Vázquez R. Immunomodulatory and Antitumoral Activity of Gold Nanoparticles Synthesized by Red Algae Aqueous Extracts. Marine Drugs. 2022; 20(3):182. https://doi.org/10.3390/md20030182
Chicago/Turabian StyleGonzález-Ballesteros, Noelia, Lara Diego-González, Mariano Lastra-Valdor, Maria Grimaldi, Antonella Cavazza, Franca Bigi, M. Carmen Rodríguez-Argüelles, and Rosana Simón-Vázquez. 2022. "Immunomodulatory and Antitumoral Activity of Gold Nanoparticles Synthesized by Red Algae Aqueous Extracts" Marine Drugs 20, no. 3: 182. https://doi.org/10.3390/md20030182
APA StyleGonzález-Ballesteros, N., Diego-González, L., Lastra-Valdor, M., Grimaldi, M., Cavazza, A., Bigi, F., Rodríguez-Argüelles, M. C., & Simón-Vázquez, R. (2022). Immunomodulatory and Antitumoral Activity of Gold Nanoparticles Synthesized by Red Algae Aqueous Extracts. Marine Drugs, 20(3), 182. https://doi.org/10.3390/md20030182








