Preparation, Supramolecular Aggregation and Immunological Activity of the Bona Fide Vaccine Adjuvant Sulfavant S
Abstract
1. Introduction
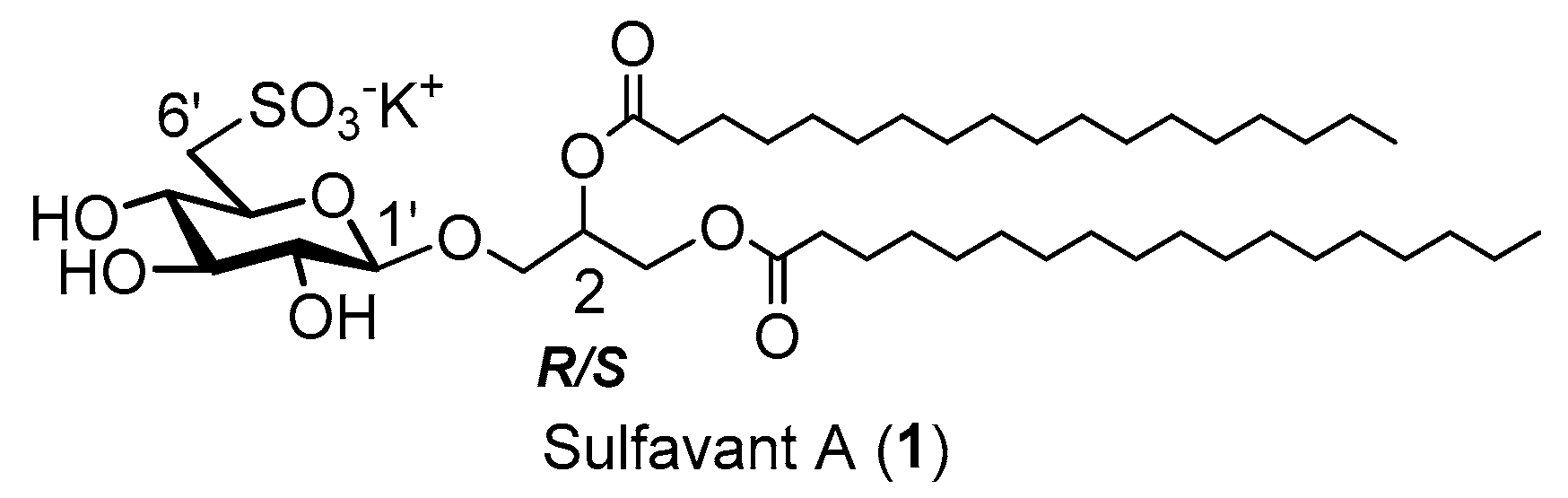

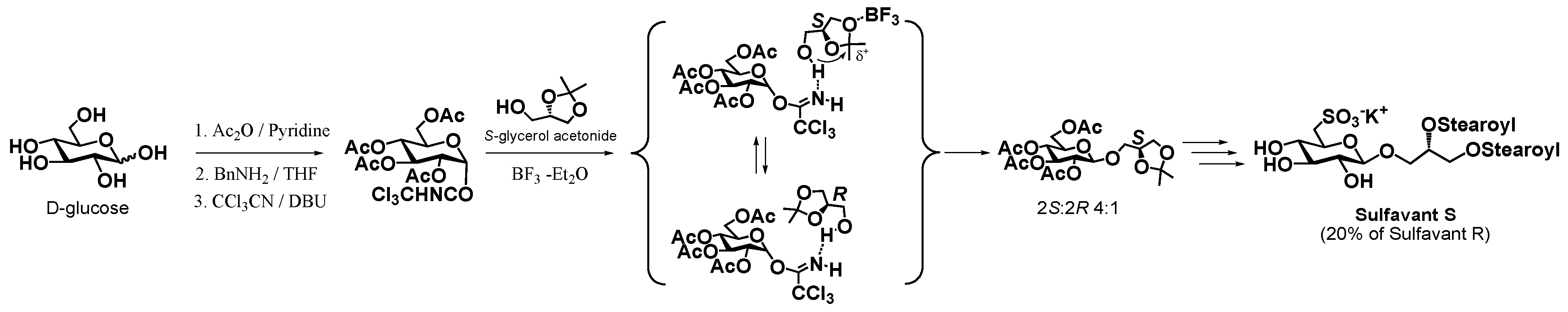
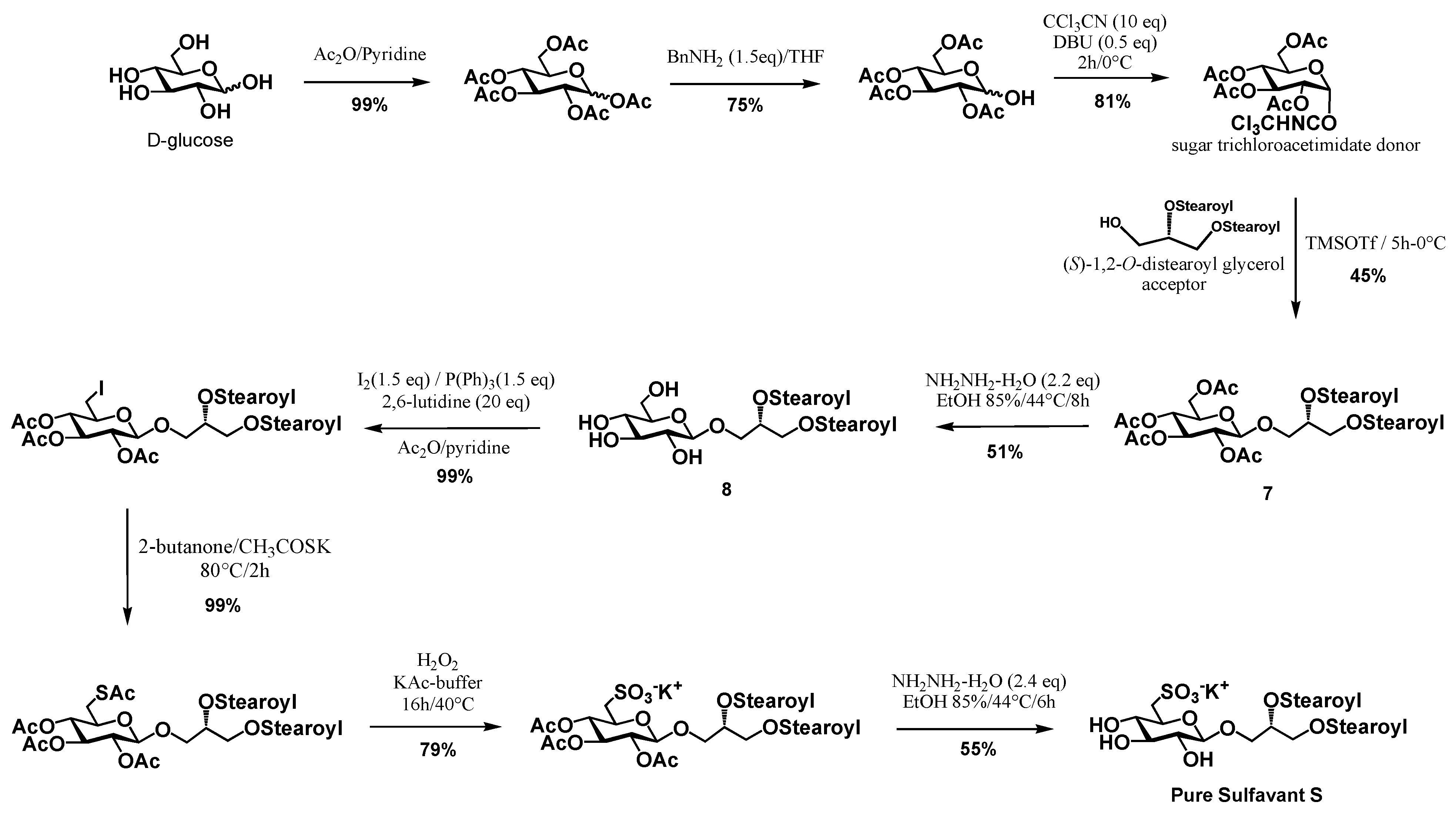
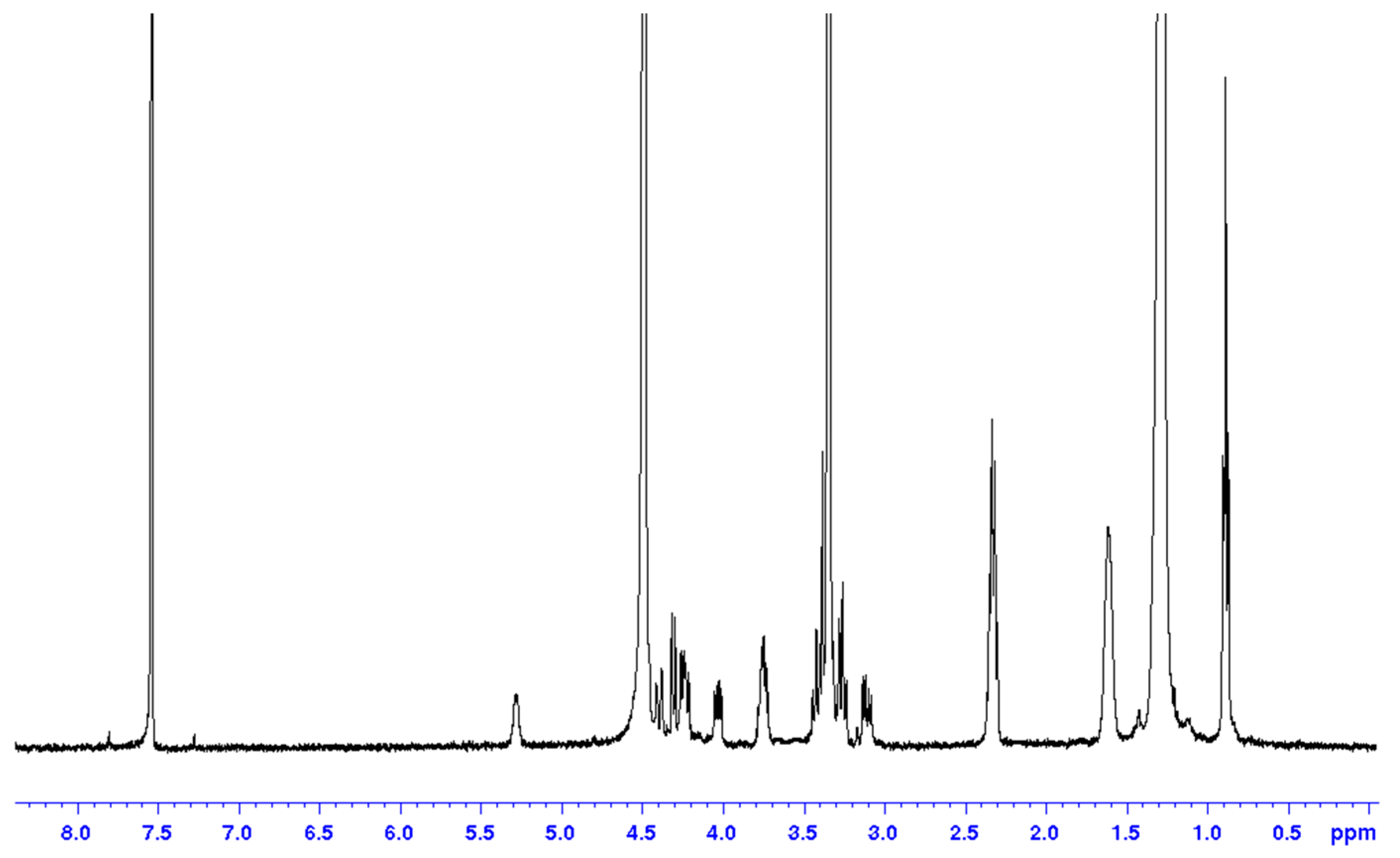
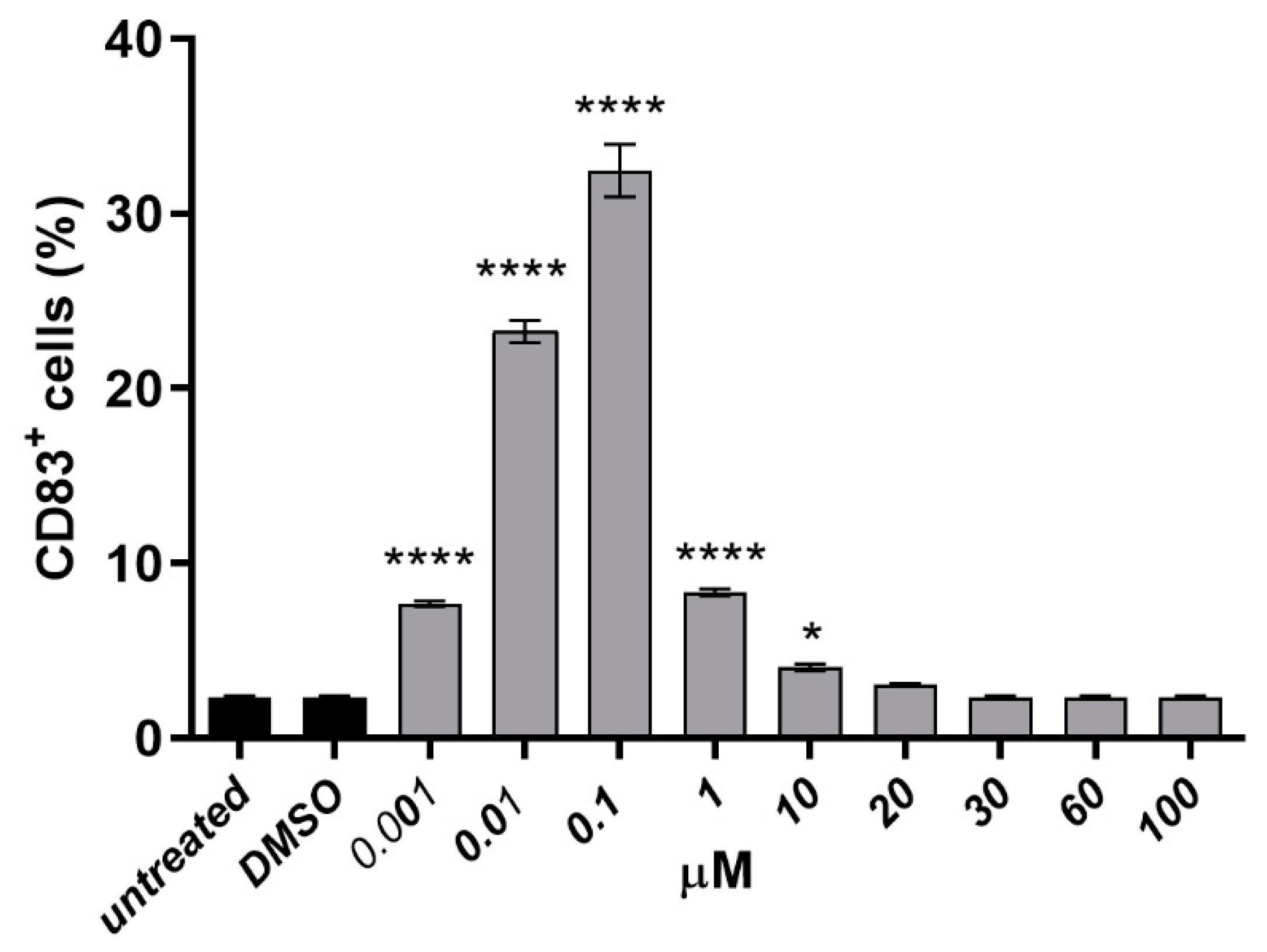
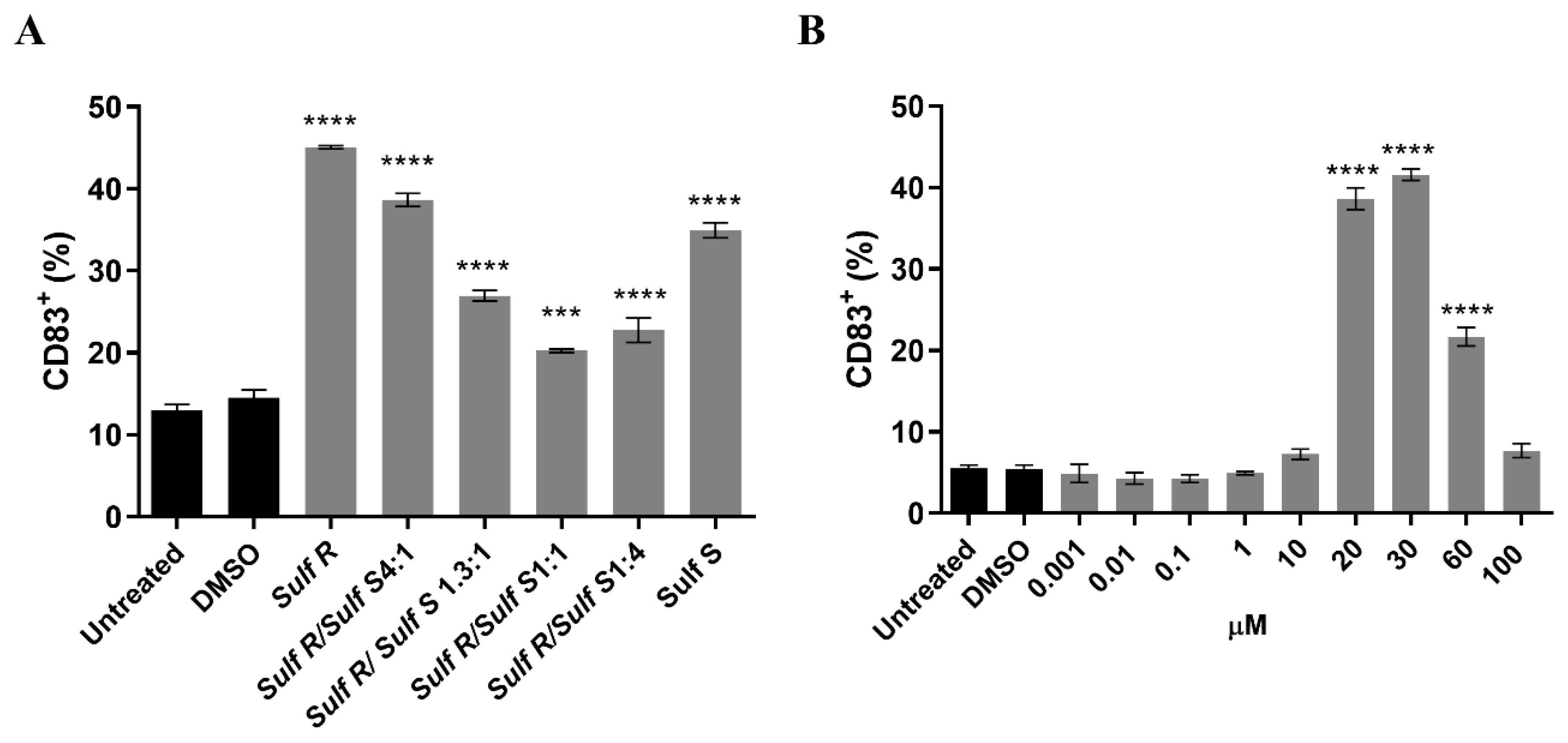
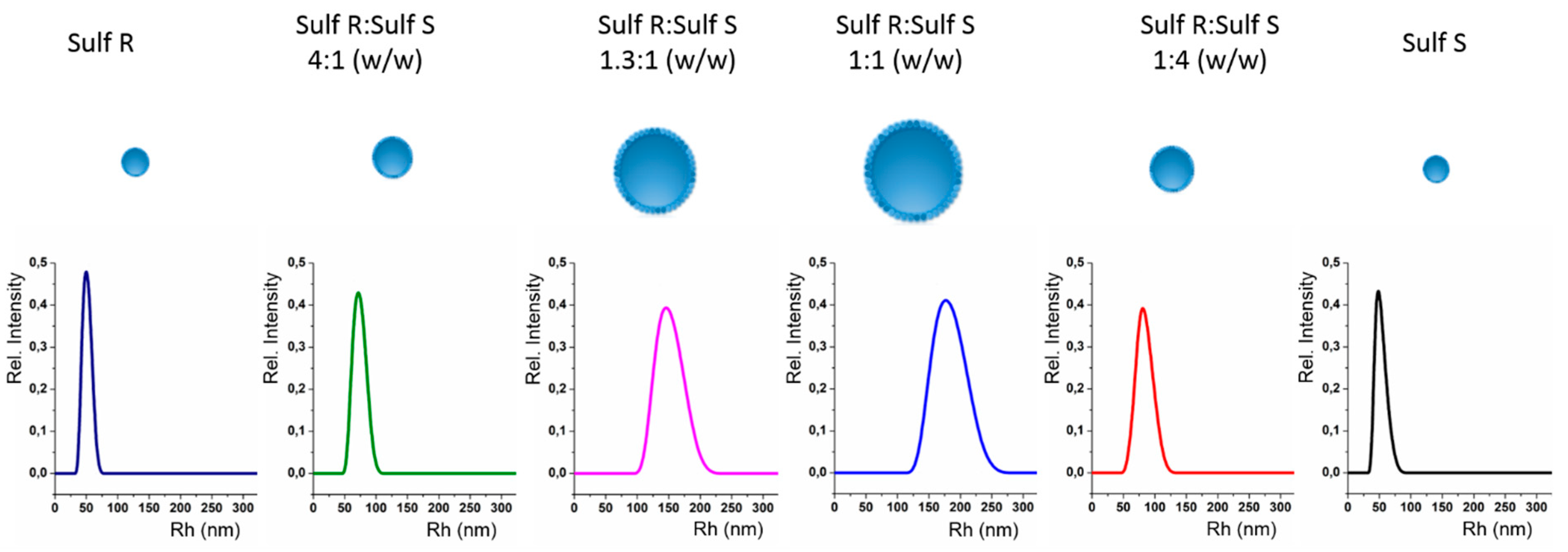
2. Results and Discussion
3. Materials and Methods
3.1. Dynamic Light Scattering (DLS)
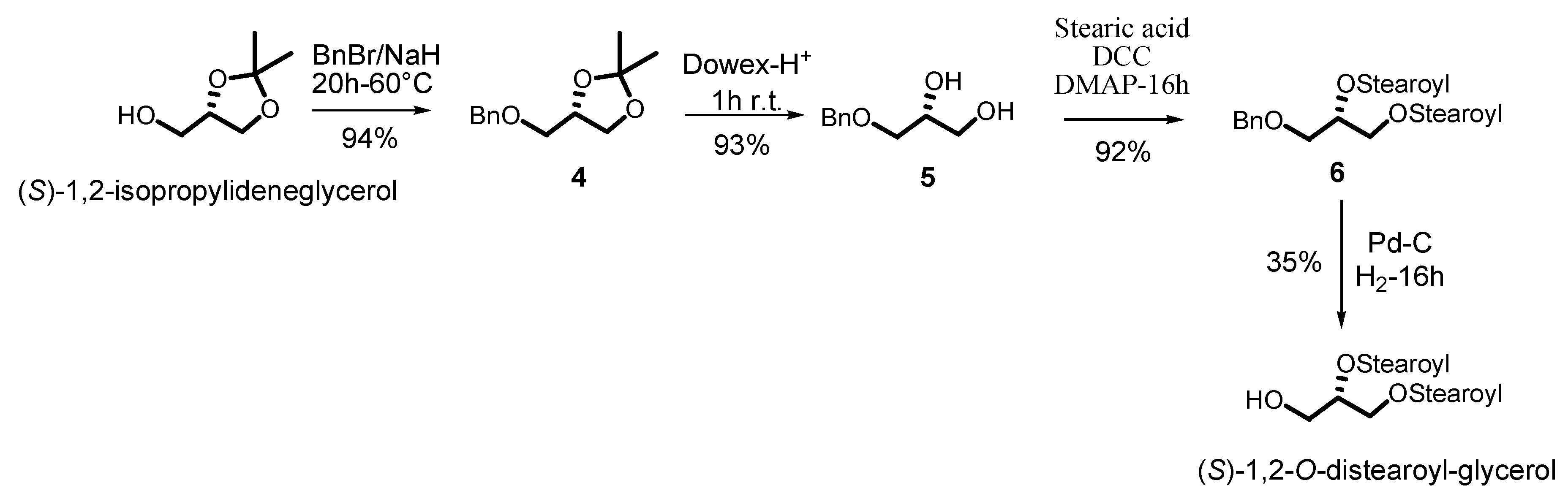
3.2. Synthetic Procedures and Characterization of Intermediates 4–7, (S)-1,2-O-Distearoyl Glycerol and Pure Sulfavant S (3)
3.3. Human Monocyte-Dendritic Cell Differentation
3.4. Cells Staining and Stimulation
3.5. Statistical Analysis
3.6. Characterization of Colloid Nanoparticles
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lehn, J.M. Toward self-organization and complex matter. Science 2002, 295, 2400–2403. [Google Scholar] [CrossRef] [PubMed]
- Waring, M.J. Lipophilicity in drug discovery. Expert Opin. Drug Discov. 2010, 5, 235–248. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, J.C.; Rodríguez, E.G. Vaccine adjuvants revisited. Vaccine 2007, 25, 3752–3762. [Google Scholar] [CrossRef]
- Xu, F.; Valiante, N.M.; Ulmer, J.B. Small molecule immunopotentiators as vaccine adjuvants. In Vaccine Adjuvants and Delivery Systems; Singh, M., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2007; pp. 175–189. [Google Scholar]
- Egli, A.; Santer, D.M.; Barakat, K.; Zand, M.; Levin, A.; Vollmer, M.; Weisser, M.; Khanna, N.; Kumar, D.; Tyrrell, D.L.; et al. Vaccine adjuvants—Understanding molecular mechanisms to improve vaccines. Swiss Med. Wkly. 2014, 144, w13940. [Google Scholar] [CrossRef] [PubMed]
- De Gregorio, E.; D’Oro, U.; Wack, A. Immunology of TLR-independent vaccine adjuvants. Curr. Opin. Immunol. 2009, 21, 339–345. [Google Scholar] [CrossRef]
- Wu, T.Y.-H.; Singh, M.; Miller, A.T.; De Gregorio, E.; Doro, F.; D’Oro, U.; Skibinski, D.A.G.; Mbow, M.L.; Bufali, S.; Herman, A.E.; et al. Rational design of small molecules as vaccine adjuvants. Sci. Transl. Med. 2014, 6, 263ra160. [Google Scholar] [CrossRef]
- De Gregorio, E.; Rappuoli, R. From empiricism to rational design: A personal perspective of the evolution of vaccine development. Nat. Rev. Immunol. 2014, 14, 505–514. [Google Scholar] [CrossRef]
- Johnson, D.A.; Baldridge, J.R. TLR4 Agonists as vaccine adjuvants. In Vaccine Adjuvants and Delivery Systems; Singh, M., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2007; pp. 131–156. [Google Scholar]
- Reed, S.G.; Orr, M.T.; Fox, C.B. Key roles of adjuvants in modern vaccine. Nat. Med. 2013, 19, 1597–1608. [Google Scholar] [CrossRef]
- Garcon, N.; Di Pasquale, A. From discovery to licensure, the Adjuvant System story. Human Vaccines Immunother. 2017, 13, 19–33. [Google Scholar] [CrossRef]
- Manzo, E.; Gallo, C.; Fioretto, L.; Nuzzo, G.; Barra, G.; Pagano, D.; Russo Krauss, I.; Paduano, L.; Ziaco, M.; DellaGreca, M.; et al. Diasteroselective colloidal self-assembly affects the immunological response of the molecular adjuvant Sulfavant. ACS Omega 2019, 4, 7807–7814. [Google Scholar] [CrossRef]
- Manzo, E.; Cutignano, A.; Pagano, D.; Gallo, C.; Barra, G.; Nuzzo, G.; Sansone, C.; Ianora, A.; Urbanek, K.; Fenoglio, D.; et al. A new marine-derived sulfoglycolipid triggers dendritic cell activation and immune adjuvant response. Sci. Rep. 2017, 7, 6286. [Google Scholar] [CrossRef] [PubMed]
- Manzo, E.; Fioretto, L.; Pagano, D.; Nuzzo, G.; Gallo, C.; De Palma, R.; Fontana, A. Chemical synthesis of marine-derived sulfoglycolipids, a new class of molecular adjuvants. Mar. Drugs 2017, 15, 288. [Google Scholar] [CrossRef] [PubMed]
- Manzo, E.; Ciavatta, M.L.; Pagano, D.; Fontana, A. An efficient and versatile chemical synthesis of bioactive glycoglycerolipids. Tetrahedron Lett. 2012, 53, 879–881. [Google Scholar] [CrossRef]
- Mellman, I. Dendritic cells: Master regulators of the immune response. Cancer Immunol. Res. 2013, 1, 145–149. [Google Scholar] [CrossRef] [PubMed]
- Pearce, E.J.; Everts, B. Dendritic cell metabolism. Nat. Rev. Immunol. 2015, 15, 18–29. [Google Scholar] [CrossRef]
- Sallusto, F.; Lanzavecchia, A. Efficient Presentation of Soluble Antigen by Cultured Human Dendritic Cells Is Maintained by Granulocyte/Macrophage Colony-stimulating Factor Plus Iuterleukin 4 and Downregulated by Tumor Necrosis Factor α. J. Exp. Med. 1994, 179, 1109–1118. [Google Scholar] [CrossRef] [PubMed]
- Rossi, M.; Young, J.W. Human Dendritic Cells: Potent Antigen-Presenting Cells at the Crossroads of Innate and Adaptive Immunity. J. Immunol. 2005, 175, 1373–1381. [Google Scholar] [CrossRef]
- Hart, D.N.J. Dendritic Cells: Unique Leukocyte Populations Which Control the Primary Immune Response. J. Am. Soc. Hematol 1997, 90, 3245–3287. [Google Scholar] [CrossRef]
- Guermonprez, P.; Valladeau, J.; Zitvogel, L.; Théry, C.; Amigorena, S. Antigen presentation and T Cell stimulation by Dendritic Cells. Annu. Rev. Immunol. 2002, 20, 621–677. [Google Scholar] [CrossRef]
- Lanzavecchia, A.; Sallusto, F. Regulation of T cell immunity by Dendritic cells. Cell 2001, 106, 263–266. [Google Scholar] [CrossRef]
- Steinman, R.M.; Hemmi, H. Dendritic cells: Translating innate to adaptive immunity. Curr. Top. Microbiol. Immunol. 2006, 311, 17–58. [Google Scholar] [PubMed]
- Steinman, R.M.; Pope, M. Exploiting dendritic cells to improve vaccine efficacy. J. Clin. Invest. 2002, 109, 1519–1526. [Google Scholar] [CrossRef] [PubMed]
- Kastenmüller, W.; Kastenmüller, K.; Kurts, C.; Seder, R.A. Dendritic cell-targeted vaccines—Hope or hype? Nat. Rev. Immunol. 2014, 14, 705–711. [Google Scholar] [CrossRef] [PubMed]
- Palucka, K.; Banchereau, J. Cancer immunotherapy via dendritic cells. Nat. Rev. Cancer 2012, 12, 265–277. [Google Scholar] [CrossRef]
- Zhou, L.J.; Tedder, T.F. Human blood dendritic cells selectively express CD83, a member of the immunoglobulin superfamily. J. Immunol. 1995, 154, 3821–3835. [Google Scholar]
- Owen, S.C.; Doak, A.K.; Ganesh, A.N.; Nedyalkova, L.; McLaughlin, C.K.; Shoichet, B.K.; Shoichet, M.S. Colloidal drug formulations can explain “bell-shaped” concentration-response curves. ACS Chem. Biol. 2014, 9, 777–784. [Google Scholar] [CrossRef]
- Matsumoto, K.; Sakai, H.; Takeuchi, R.; Tsuchiya, K.; Ohta, K.; Sugawara, F.; Abe, M.; Sakaguchi, K. Effective form of sulfoquinovosyldiacyglycerol (SQDG) vesicles for DNA polymerase inhibition. Colloids Surfaces B Biointerfaces 2005, 46, 175–181. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Sahara, H.; Takenouchi, M.; Matsumoto, Y.; Imai, A.; Fujita, T.; Tamura, Y.; Takahashi, N.; Gasa, S.; Matsumoto, K.; et al. Inhibition of CD62L+T-cell response in vitro via a novel sulfo-glycolipid, β-SQAG9 liposome that binds to CD62L molecule on the cell surface. Cell. Immunol. 2004, 232, 105–115. [Google Scholar] [CrossRef]
- Aoki, S.; Ohta, K.; Matsumoto, K.; Sakai, H.; Abe, M.; Miura, M.; Sugawara, F.; Sakaguchi, K. An emulsion of sulfoquinovosylacylglycerol with long-chain alkanes increases its permeability to tumor cells. J. Membr. Biol. 2006, 213, 11–18. [Google Scholar] [CrossRef]
- Matsumoto, K.; Takenouchi, M.; Ohta, K.; Ohta, Y.; Imura, T.; Oshige, M.; Yamamoto, Y.; Sahara, H.; Sakai, H.; Abe, M.; et al. Design of vesicles of 1,2-di-O-acyl-3-O-(β-D-sulfoquinovosyl)-glyceride bearing two stearic acids (β-SQDG-C18), a novel immunosuppressive drug. Biochem. Pharmacol. 2004, 68, 2379–2386. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manzo, E.; Fioretto, L.; Gallo, C.; Ziaco, M.; Nuzzo, G.; D’Ippolito, G.; Borzacchiello, A.; Fabozzi, A.; De Palma, R.; Fontana, A. Preparation, Supramolecular Aggregation and Immunological Activity of the Bona Fide Vaccine Adjuvant Sulfavant S. Mar. Drugs 2020, 18, 451. https://doi.org/10.3390/md18090451
Manzo E, Fioretto L, Gallo C, Ziaco M, Nuzzo G, D’Ippolito G, Borzacchiello A, Fabozzi A, De Palma R, Fontana A. Preparation, Supramolecular Aggregation and Immunological Activity of the Bona Fide Vaccine Adjuvant Sulfavant S. Marine Drugs. 2020; 18(9):451. https://doi.org/10.3390/md18090451
Chicago/Turabian StyleManzo, Emiliano, Laura Fioretto, Carmela Gallo, Marcello Ziaco, Genoveffa Nuzzo, Giuliana D’Ippolito, Assunta Borzacchiello, Antonio Fabozzi, Raffaele De Palma, and Angelo Fontana. 2020. "Preparation, Supramolecular Aggregation and Immunological Activity of the Bona Fide Vaccine Adjuvant Sulfavant S" Marine Drugs 18, no. 9: 451. https://doi.org/10.3390/md18090451
APA StyleManzo, E., Fioretto, L., Gallo, C., Ziaco, M., Nuzzo, G., D’Ippolito, G., Borzacchiello, A., Fabozzi, A., De Palma, R., & Fontana, A. (2020). Preparation, Supramolecular Aggregation and Immunological Activity of the Bona Fide Vaccine Adjuvant Sulfavant S. Marine Drugs, 18(9), 451. https://doi.org/10.3390/md18090451










