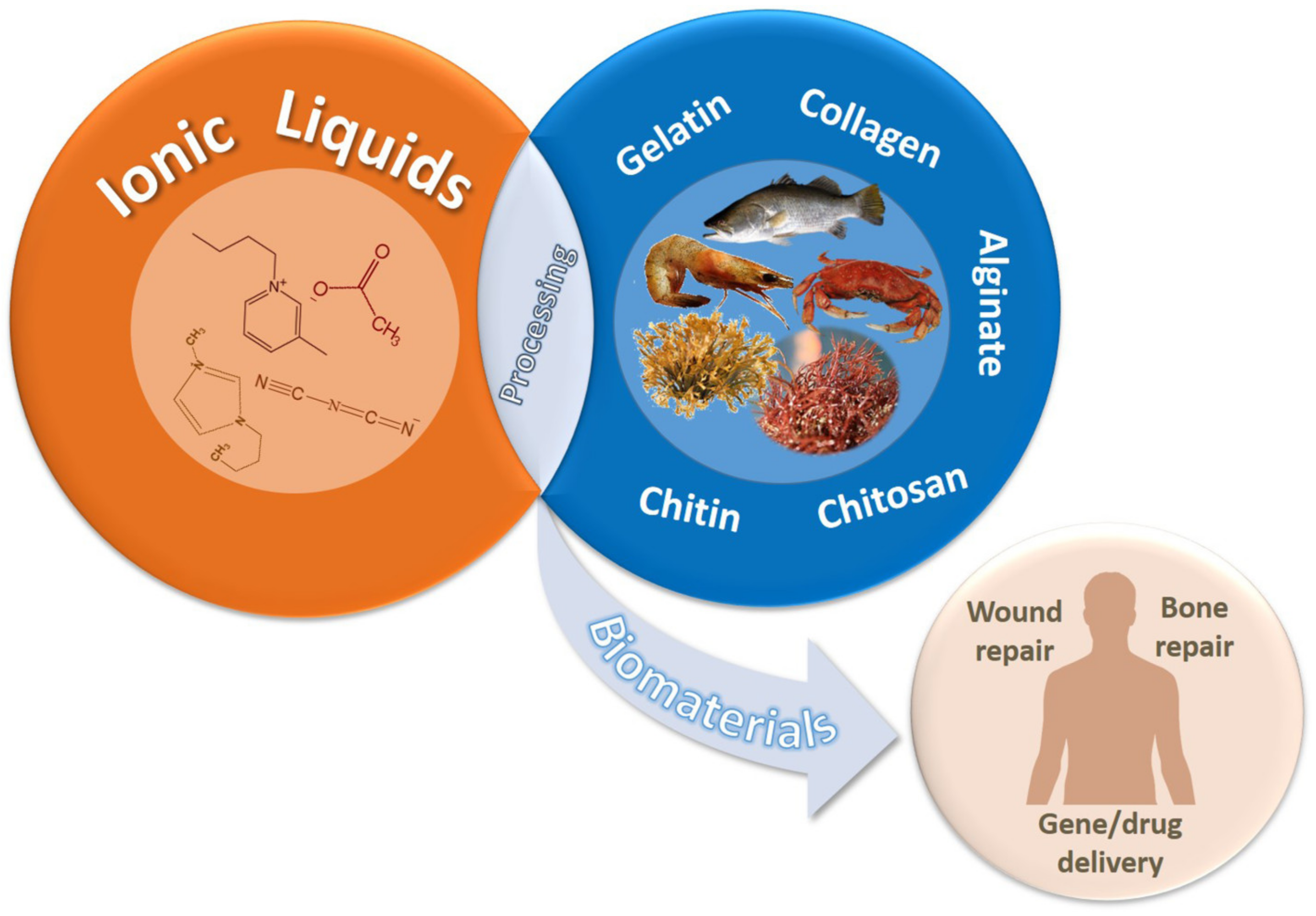
Marine-Derived Polymers in Ionic Liquids: Architectures Development and Biomedical Applications
Abstract
1. Introduction
2. Dissolution of Marine-Derived Polymers Using Ionic Liquids
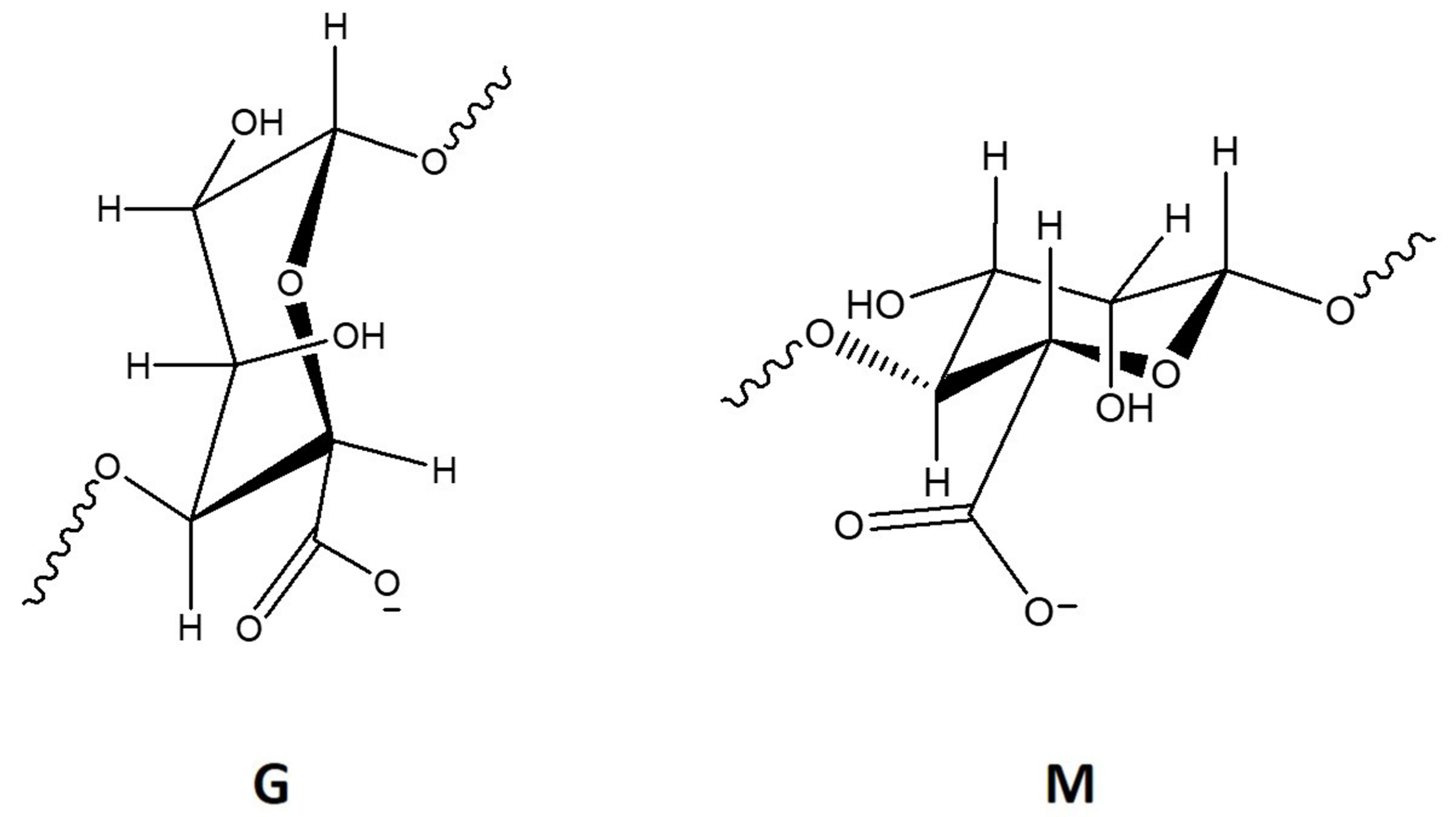
2.1. Alginate
2.2. Chitin
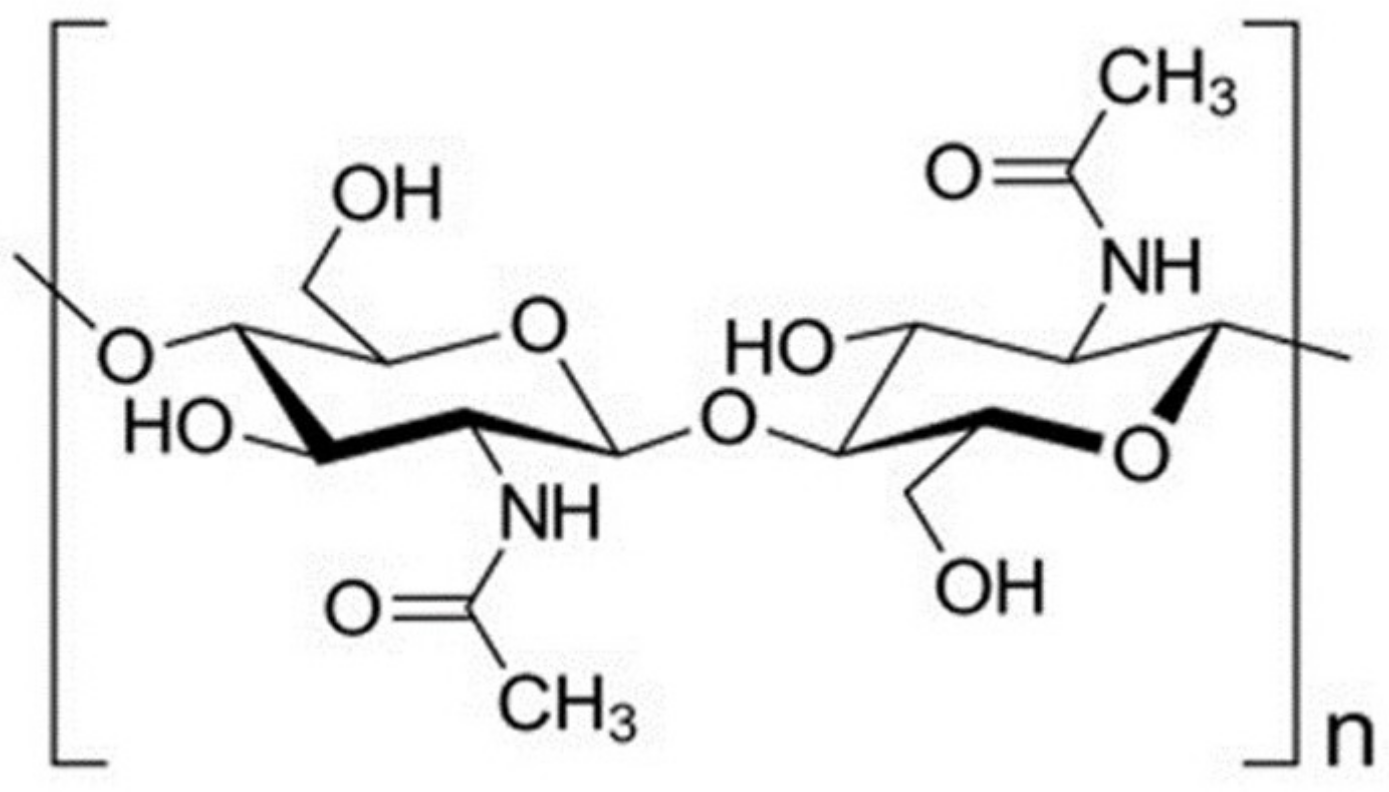
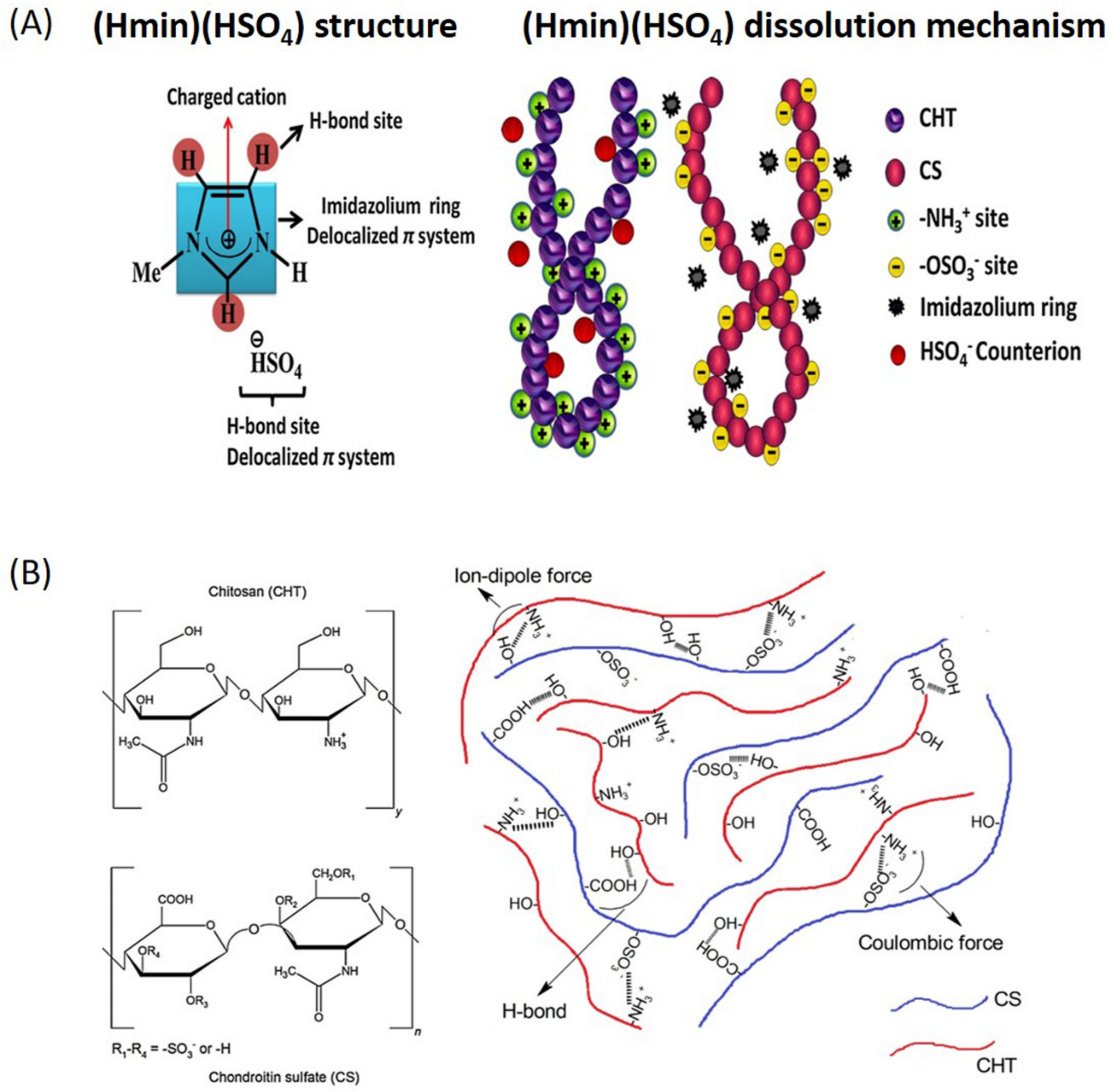
2.3. Chitosan
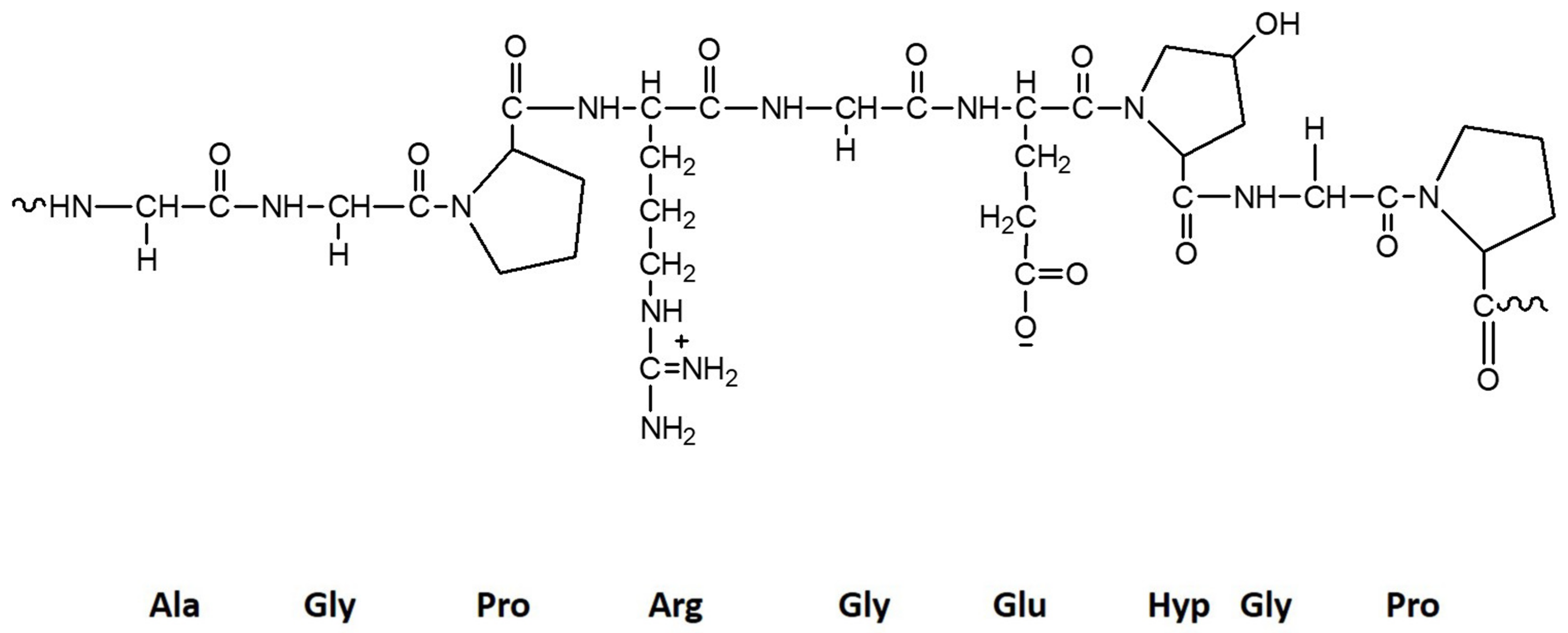
2.4. Collagen
2.5. Gelatin
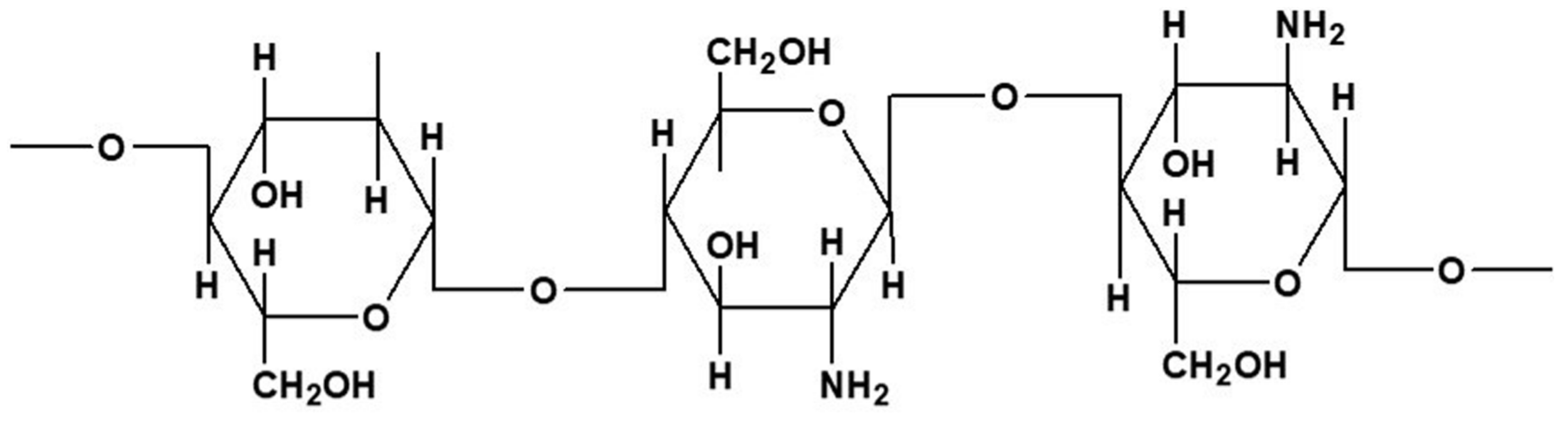
2.6. Other Marine-Derived Polymers
3. Development of Marine-Polymeric Architectures via Ionic Liquids
3.1. Films and Hydrogels
3.2. Nanomicrofibers and Nanomicroparticles
3.3. Scaffolds, Sponges, and Beads
4. Environmental and Biological Impact of ILs Used in the Development of Marine Polymer-Based Architectures
4.1. IL’s Recycling and Reuse
4.2. Biocompatibility
| Polymer/Matrix | Ionic Liquid/other Reagents | Process | Improved Properties | Potential Applications | References |
|---|---|---|---|---|---|
| Chitin | (Emim)(OAc) | Extraction/dissolution/ electrospinning |
| Not defined | [23,24,102] |
| Chitosan | (Gly·H)Cl and (Bmim)Cl | Dissolution/dry-wet spinning |
| Not defined | [103] |
| (Bmim)(EtSO4) or (Omim)(Cl) | Ionic crosslinking |
| Not defined | [116] | |
| Collagen | (Emim)(Br)/ (Emim)(Cl)/ (Emim)(OAc) | Self-assembling |
| Not defined | [106] |
| Collagen-based hydrogels | (Emim)(OAc) | Sol-gel transition |
| Tissue engineering and cancer therapy. | [128] |
| Gelatin Microcapsules | (Bmim)(BF4) | Microemulsion |
| MR imaging-guided MW thermotherapy. | [29] |
| Gelatin Ion Gels | (Emim)(EtSO4) | Dissolution/Photoreduction |
| Not defined | [31] |
| (Emim)(Cl) | Gelation |
| biomedical engineering | [92] | |
| (Emim)(EtSO4) | Dissolution/Gelation |
| colorimetric glucose detection | [95] | |
| Gelatin films | (Emim)(OAc) | Doping |
| smart windows and other ECD-based devices | [129] |
| Gelatin hydrogels | Omim·PF6 | Ultrasonication |
| immobilization of enzymes and fabrication of biosensors | [96] |
| Polymer Blends | Ionic Liquid/other Reagents | Process | Improved Properties | Potential Applications | References |
|---|---|---|---|---|---|
| Chitin/ Antheraea pernyi silk fibroin based sponges | (Bmim))(OAc)) | Co-dissolution/Freeze-drying |
| Cartilage regeneration | [13] |
| Chitin-sodium alginate film | BPPF6 | Solutions mixing |
| biosensor | [26] |
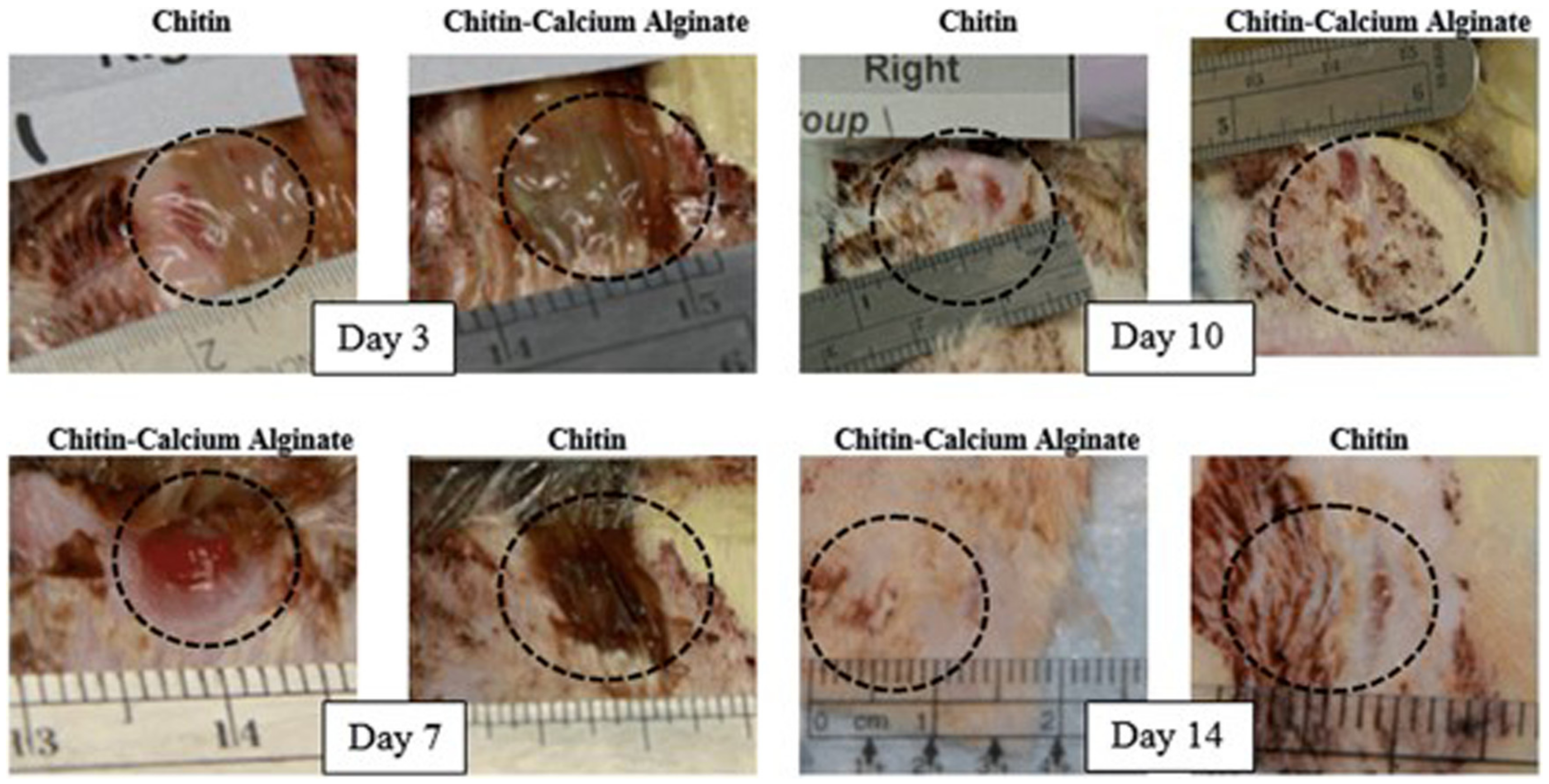
| Chitin-calcium alginate fibers | (Emim)(OAc) | Microwave IL-assisted extraction/dissolution/electrospinning |
| wound care dressings | [25] |
| Chitin–Cellulose Nanofibers | (Emim)(OAc) | Electrospinning |
| Not defined | [24] |
| Chitin and hydroxyapatite | (Bmim)(OAc) | Dissolution |
| bone tissue engineering | [19,118] |
| Chitin–poly(lactic acid) Fibers | (Emim)(OAc) | Co-dissolution/wet-jet spun |
| Not defined | [130] |
| Chitin/SAIB scaffolds | (Bmim)(OAc) | Co-dissolution/freeze-drying |
| tissue engineering scaffolding | [131] |
| Agarose/chitosan ionogels | (Bmim)(Cl) | Blending/Gelation |
| quasisolid dye sensitized solar cells, actuators, sensors or electrochromic displays | [30] |
| Carrageenan/cellulose gels | (Bmim)(Cl) | Co-dissolution |
| [98] | |
| Chitosan/cellulose | (Gly·H)(Cl) and (Bmim)(Cl) | Dissolution/dry-wet spinning |
| [104] | |
| (Emim)(OAc) | Dissolution/electrospinning |
| Wound treatment | [105] | |
| (Bmim)(Cl) | Co-dissolution/cast into substrate |
| Adsorption of microcystin LR, produced by cyanobacteria present in drinking waterWound dressings | [126,132] | |
| Chitosan/cellulose/hydroxyapatite | (Bmim)(Cl) | Dissolution |
| bone tissue engineering | [27] |
| Chitosan/cellulose/keratin | (Bmim)(Cl) | Co-dissolution/cast into substrate |
| treatment of chronic and ulcerous wounds | [133] |
| Chitosan/chondroitin sulfate hydrogels | (Hmim)(HSO4) | Blending/gelation |
| treatment of water and wastewater | [28] |
| Chitosan/silk fibroin hydrogels | (Bmim)(OAc) | Blending/Gelation |
| skin tissue engineering approaches | [14] |
| Collagen-alginate-hydroxyapatite beads | (TEA)(OAc) | CaCl2-based crosslinking |
| bone regeneration | [122] |
| Collagen/ Hydroxyapatite/ Alginate | (TEA)(OAc) | Dissolution |
| treatment of deep intraosseous defects | [134] |
| Collagen/PVA hydrogels | (Bmim)(OAc) | Blending |
| osteochondral patches | [135] |
| Gelatin/Poly(Vinyl Alcohol) films | Lidocaine–Diclofenac IL | Freeze-thawing |
| transdermal patches | [97] |
5. Biomedical Applications of Marine-derived Polymers in Ionic Liquids
5.1. Wound Repair
5.2. Bone Regeneration
5.3. Drug and Gene Delivery
6. Conclusions and Future Trends
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AgNO3 | silver nitrate |
| (Amim)(Br) | 1-allyl-3-methylimidazolium bromide |
| Amim | 1-allyl-3-methylimidazolium |
| Ala | alanine |
| APIs | active pharmaceutical ingredients |
| APIs-IL | active pharmaceutical ingredients in ionic liquids systems |
| Arg | arginine |
| Bio-ILs | biocompatible IL |
| BPPF6 | N-butylpyridinium hexafluorophosphate |
| (Bmim) | 1-butyl-3-methylimidazolium |
| (Bmim)(OAc) | 1-butyl-3-methylimidazolium acetate |
| (Bmmim)(OAc) | 1-butyl-2,3-dimethylimidazolium acetate |
| (Bmim)(BF4) | 1-butyl-3-methylimidazolium tetrafluoroborate |
| (Bmim)](C2OSO3) | 1-ethyl-3-methylimidazolium ethylsulfate |
| (Bmim)(Cl) | 1-butyl-3-methylimidazolium chloride |
| (Ch)(DHP) | Choline dihydrogen phosphate |
| (Dmim)(Cl) | 1-decyl-3-methylimidazolium chloride |
| (Emim)(Cl) | 1-ethyl-3-methylimidazolium chloride |
| (Emim)(OAc) | 1-ethyl-3-methylimidazolium acetate |
| (Emim)(Br) | 1-ethyl-3-methylimidazolium bromide |
| (Dmim) | 1,3-dimethylimidazolium |
| (Emim) (EtSO4) | 1-ethyl-3-methyl-imidazolium ethyl sulfate |
| Gly | glycine |
| (Gly·H)Cl | glycine hydrochloride |
| Glu | glutamate |
| GOD | glucose oxidase |
| HA | hydroxyapatite |
| H2O2 | Hydrogen peroxide |
| HT29 | epithelial colorectal adenocarcinoma cells |
| (Hmim)/ OAc) | 1-hydrogen-3-methylimidazolium acetate |
| (Hmim)(HSO4) | 1-hydrogen-3-methylimidazolium hydrogen sulfate |
| HRP | horseradish peroxidase |
| Hyp | hydroxyproline |
| IGs | ion gels |
| IL | ionic liquid; |
| (Mim)(OAc) | 1-methylimidazolium acetate |
| MCC | Microcrystalline cellulose |
| MW | microwave |
| MR | magnetic resonance |
| (mim) | 1-hydrogen-3-methylimidazolium |
| NPs | nanoparticles |
| (Omim)(OAc) | 1-octyl-3-methylimidazolium acetate |
| (Omim)(Cl) | 1-octyl-3-methylimidazolium chloride |
| OMIM·PF6 | 1-Octyl-3- methylimidazolium hexafluorophsohate |
| PBS | phosphate-buffered saline |
| PEI | polyethylenimine |
| PE | polymer electrolyte |
| Pro | proline |
| PLA | poly(lactic acid) |
| PVA | poly(vinyl alcohol) |
| (TEA)(Ac) | triethanolamine acetate |
| VERO | healthy kidney epithelial cells originated from African green monkey |
| 3D | three-dimensional |
| Κ | kappa |
| λ | lambda |
References
- Silva, T.; Duarte, A.; Moreira-Silva, J.; Mano, J.F.; Reis, R.L. Biomaterials from Marine-Origin Biopolymers; Mano, J.F., Ed.; Wiley-VCH Verlag: Weinheim, Germany, 2012; pp. 3–24. [Google Scholar]
- Silva, T.H.; Alves, A.; Ferreira, B.M.; Oliveira, J.M.; Reys, L.L.; Ferreira, R.J.F.; Sousa, R.A.; Silva, S.S.; Mano, J.F.; Reis, R.L. Materials of marine origin: A review on polymers and ceramics of biomedical interest. Int. Mater. Rev. 2012, 57, 276–306. [Google Scholar] [CrossRef]
- Hsiang-Jung, T.; Tai-Li, T.; Hsian-Jenn, W.; Shan-hui, H. Characterization of chitosan–gelatin scaffolds for dermal tissue engineering. J. Tissue Eng. Regen. Med. 2013, 7, 20–31. [Google Scholar]
- Anthony, J.; Brennecke, J.; Holbrey, J.; Maginn, E.; Mantz, R.; Trulove, P.; Visser, A.; Welton, T. Ionic Liquids in Synthesis; Wasserscheid, P., Welton, T., Eds.; Wiley-VCH: Weinheim, Germany, 2002; pp. 41–55. [Google Scholar]
- Qin, Y.; Lu, X.; Sun, N.; Rogers, R.D. Dissolution or extraction of crustacean shells using ionic liquids to obtain high molecular weight purified chitin and direct production of chitin films and fibers. Green Chem. 2010, 12, 968–971. [Google Scholar] [CrossRef]
- Singh, T.; Trivedi, T.J.; Kumar, A. Dissolution, regeneration and ion-gel formation of agarose in room-temperature ionic liquids. Green Chem. 2010, 12, 1029–1035. [Google Scholar] [CrossRef]
- Trivedi, T.; Kumar, A. Efficient extraction of agarose from red algae using ionic liquids. Green Sustain. Chem. 2014, 4, 190–201. [Google Scholar] [CrossRef]
- Tolesa, L.D.; Gupta, B.S.; Lee, M.J. Chitin and chitosan production from shrimp shells using ammonium-based ionic liquids. Int. J. Biol. Macromol. 2019, 130, 818–826. [Google Scholar] [CrossRef]
- Shi, R.; Wang, Y. Dual ionic and organic nature of ionic liquids. Sci. Rep. 2016, 6, 19644. [Google Scholar] [CrossRef]
- Hulsbosch, J.; De Vos, D.E.; Binnemans, K.; Ameloot, R. Biobased ionic liquids: Solvents for a green processing industry? ACS Sustain. Chem. Eng. 2016, 4, 2917–2931. [Google Scholar] [CrossRef]
- Silva, S.S.; Reis, R.L. CHAPTER 11 Ionic liquids as tools in the production of smart polymeric hydrogels. In Polymerized Ionic Liquids; The Royal Society of Chemistry: Burlington House : London, UK, 2018; pp. 304–318. [Google Scholar]
- Silva, S.S.; Popa, E.G.; Gomes, M.E.; Oliveira, M.B.; Nayak, S.; Subia, B.; Mano, J.F.; Kundu, S.C.; Reis, R.L. Silk hydrogels from non-mulberry and mulberry silkworm cocoons processed with ionic liquids. Acta Biomater. 2013, 9, 8972–8982. [Google Scholar] [CrossRef]
- Silva, S.S.; Gomes, J.; Vale, A.; Lu, S.; Reis, R.L.; Kundu, S. Green pathway for processing non-mulberry antheraea pernyi silk fibroin/chitin-based sponges: Biophysical and biochemical characterization. Front. Mater. 2020, 7, 1–9. [Google Scholar] [CrossRef]
- Silva, S.S.; Santos, T.C.; Cerqueira, M.T.; Marques, A.P.; Reys, L.L.; Silva, T.H.; Caridade, S.G.; Mano, J.F.; Reis, R.L. The use of ionic liquids in the processing of chitosan/silk hydrogels for biomedical applications. Green Chem. 2012, 14, 1463–1470. [Google Scholar] [CrossRef]
- Takegawa, A.; Murakami, M.; Kaneko, Y.; Kadokawa, J. Preparation of chitin/cellulose composite gels and films with ionic liquids. Carbohyd. Polym. 2010, 79, 85–90. [Google Scholar] [CrossRef]
- Zhang, J.; Wu, J.; Yu, J.; Zhang, X.; He, J.; Zhang, J. Application of ionic liquids for dissolving cellulose and fabricating cellulose-based materials: State of the art and future trends. Mater. Chem. Front. 2017, 1, 1273–1290. [Google Scholar] [CrossRef]
- Wu, Y.; Sasaki, T.; Irie, S.; Sakurai, K. A novel biomass-ionic liquid platform for the utilization of native chitin. Polymer 2008, 49, 2321–2327. [Google Scholar] [CrossRef]
- Swatloski, R.P.; Spear, S.K.; Holbrey, J.D.; Rogers, R.D. Dissolution of cellulose with ionic liquids. J. Am. Chem. Soc. 2002, 124, 4974–4975. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.S.; Mano, J.F.; Reis, R.L. Ionic liquids in the processing and chemical modification of chitin and chitosan for biomedical applications. Green Chem. 2017, 19, 1208–1220. [Google Scholar] [CrossRef]
- Singh, S.K. Solubility of lignin and chitin in ionic liquids and their biomedical applications. Int. J. Biol. Macromol. 2019, 132, 265–277. [Google Scholar] [CrossRef]
- Mallik, A.K.; Shahruzzaman, M.; Zaman, A.; Biswas, S.; Ahmed, T.; Sakib, M.N.; Haque, P.; Rahman, M.M. 4-Fabrication of polysaccharide-based materials using ionic liquids and scope for biomedical use. In Functional Polysaccharides for Biomedical Applications; Maiti, S., Jana, S., Eds.; Woodhead Publishing: Duxford, UK, 2019; pp. 131–171. [Google Scholar]
- Mahmood, H.; Moniruzzaman, M. Recent advances of using ionic liquids for biopolymer extraction and processing. Biotechnol. J. 2019, 14, e1900072. [Google Scholar] [CrossRef]
- Barber, P.S.; Griggs, C.S.; Bonner, J.R.; Rogers, R.D. Electrospinning of chitin nanofibers directly from an ionic liquid extract of shrimp shells. Green Chem. 2013, 15, 601–607. [Google Scholar] [CrossRef]
- Shamshina, J.L.; Zavgorodnya, O.; Choudhary, H.; Frye, B.; Newbury, N.; Rogers, R.D. In search of stronger/cheaper chitin nanofibers through electrospinning of chitin–cellulose composites using an ionic liquid platform. ACS Sustain. Chem. Eng. 2018, 6, 14713–14722. [Google Scholar] [CrossRef]
- Shamshina, J.L.; Gurau, G.; Block, L.E.; Hansen, L.K.; Dingee, C.; Walters, A.; Rogers, R.D. Chitin–calcium alginate composite fibers for wound care dressings spun from ionic liquid solution. J. Mater. Chem. B 2014, 2, 3924–3936. [Google Scholar] [CrossRef] [PubMed]
- Ding, C.; Zhang, M.; Zhao, F.; Zhang, S. Disposable biosensor and biocatalysis of horseradish peroxidase based on sodium alginate film and room temperature ionic liquid. Anal. Biochem. 2008, 378, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Mututuvari, T.M.; Harkins, A.L.; Tran, C.D. Facile synthesis, characterization, and antimicrobial activity of cellulose-chitosan-hydroxyapatite composite material: A potential material for bone tissue engineering. J. Biomed. Mater. Res. A 2013, 101, 3266–3277. [Google Scholar] [CrossRef] [PubMed]
- Nunes, C.S.; Rufato, K.B.; Souza, P.R.; de Almeida, E.A.M.S.; da Silva, M.J.V.; Scariot, D.B.; Nakamura, C.V.; Rosa, F.A.; Martins, A.F.; Muniz, E.C. Chitosan/chondroitin sulfate hydrogels prepared in [Hmim][HSO4] ionic liquid. Carbohyd. Polym. 2017, 170, 99–106. [Google Scholar] [CrossRef]
- Du, Q.; Ma, T.; Fu, C.; Liu, T.; Huang, Z.; Ren, J.; Shao, H.; Xu, K.; Tang, F.; Meng, X. Encapsulating ionic liquid and Fe3O4 nanoparticles in gelatin microcapsules as microwave susceptible agent for MR imaging-guided tumor thermotherapy. ACS Appl. Mater. Interfaces 2015, 7, 13612–13619. [Google Scholar] [CrossRef]
- Trivedi, T.J.; Rao, K.S.; Kumar, A. Facile preparation of agarose-chitosan hybrid materials and nanocomposite ionogels using an ionic liquid via dissolution, regeneration and sol-gel transition. Green Chem. 2014, 16, 320–330. [Google Scholar] [CrossRef]
- Singh, G.; Singh, G.; Damarla, K.; Sharma, P.K.; Kumar, A.; Kang, T.S. Gelatin-based highly stretchable, self-healing, conducting, multiadhesive, and antimicrobial ionogels embedded with Ag2O nanoparticles. ACS Sustain. Chem. Eng. 2017, 5, 6568–6577. [Google Scholar] [CrossRef]
- Łabowska, M.B.; Michalak, I.; Detyna, J. Methods of extraction, physicochemical properties of alginates and their applications in biomedical field—A review. Open Chem. 2019, 17, 738. [Google Scholar] [CrossRef]
- Gereniu, C.R.N.; Saravana, P.S.; Chun, B.-S. Recovery of carrageenan from Solomon Islands red seaweed using ionic liquid-assisted subcritical water extraction. Sep. Purif. Technol. 2018, 196, 309–317. [Google Scholar] [CrossRef]
- Mehta, M.; Bharmoria, P.; Bhayani, K.; Kumar, A. Gelatin solubility and processing in ionic liquids: An approach towards waste to utilization. ChemistrySelect 2017, 2, 9895–9900. [Google Scholar] [CrossRef]
- Tarannum, A.; Muvva, C.; Mehta, A.; Rao, J.R.; Fathima, N.N. Phosphonium based ionic liquids-stabilizing or destabilizing agents for collagen? RSC Adv. 2016, 6, 4022–4033. [Google Scholar] [CrossRef]
- Tarannum, A.; Muvva, C.; Mehta, A.; Raghava Rao, J.; Fathima, N.N. Role of preferential ions of ammonium ionic liquid in destabilization of collagen. J. Phys. Chem. B 2016, 120, 6515–6524. [Google Scholar] [CrossRef] [PubMed]
- Tarannum, A.; Rao, J.R.; Fathima, N.N. Choline-based Amino Acid ILs–collagen interaction: Enunciating its role in stabilization/destabilization phenomena. J. Phys. Chem. B 2018, 122, 1145–1151. [Google Scholar] [CrossRef] [PubMed]
- Tarannum, A.; Adams, A.; Blümich, B.; Fathima, N.N. Impact of ionic liquids on the structure and dynamics of collagen. J. Phys. Chem. B 2018, 122, 1060–1065. [Google Scholar] [CrossRef]
- Schindl, A.; Hagen, M.L.; Muzammal, S.; Gunasekera, H.A.D.; Croft, A.K. Proteins in ionic liquids: Reactions, applications, and futures. Front. Chem. 2019, 7, 347. [Google Scholar] [CrossRef]
- Mehta, A.; Rao, J.R.; Fathima, N.N. Effect of ionic liquids on the different hierarchical order of type I collagen. Colloids Surf. B Biointerfaces 2014, 117, 376–382. [Google Scholar] [CrossRef]
- Tarannum, A.; Jonnalagadda, R.R.; Nishter, N.F. Stability of collagen in ionic liquids: Ion specific Hofmeister series effect. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2019, 212, 343–348. [Google Scholar] [CrossRef]
- Gomes, J.M.; Silva, S.S.; Reis, R.L. Biocompatible ionic liquids: Fundamental behaviours and applications. Chem. Soc. Rev. 2019, 48, 4317–4335. [Google Scholar] [CrossRef]
- Silva, S.S.; Fernandes, E.M.; Pina, S.; Silva-Correia, J.; Vieira, S.; Oliveira, J.M.; Reis, R.L. Natural-origin materials for tissue engineering and regenerative medicine. In Comprehensive Biomaterials II; Ducheyne, P., Healy, K.E., Hutmacher, D.E., Grainger, D.W., Kirkpatrick, C.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Siew, C.K.; Williams, P.A.; Young, N.W.G. New Insights into the mechanism of gelation of alginate and pectin: Charge annihilation and reversal mechanism. Biomacromolecules 2005, 6, 963–969. [Google Scholar] [CrossRef]
- Drury, J.L.; Dennis, R.G.; Mooney, D.J. The tensile properties of alginate hydrogels. Biomaterials 2004, 25, 3187–3199. [Google Scholar] [CrossRef]
- Kumar, M.N.V.R. A review of chitin and chitosan applications. React. Funct. Polym. 2000, 46, 1–27. [Google Scholar] [CrossRef]
- Muzzarelli, R.A.A. Chitins and chitosans for the repair of wounded skin, nerve, cartilage and bone. Carbohyd. Polym. 2009, 76, 167–182. [Google Scholar] [CrossRef]
- Younes, I.; Rinaudo, M. Chitin and chitosan preparation from marine sources. Structure, properties and applications. Mar. Drugs 2015, 13, 1133–1174. [Google Scholar] [CrossRef] [PubMed]
- Rinaudo, M. Chitin and chitosan: Properties and applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Pillai, C.K.S.; Paul, W.; Sharma, C.P. Chitin and chitosan polymers: Chemistry, solubility and fiber formation. Prog. Polym. Sci. 2009, 34, 641–678. [Google Scholar] [CrossRef]
- Jang, M.-K.; Kong, B.-G.; Jeong, Y.-I.; Lee, C.H.; Nah, J.-W. Physicochemical characterization of α-chitin, β-chitin, and γ-chitin separated from natural resources. J. Polym. Sci. Part A Polym. Chem. 2004, 42, 3423–3432. [Google Scholar] [CrossRef]
- Setoguchi, T.; Kato, T.; Yamamoto, K.; Kadokawa, J.-I. Facile production of chitin from crab shells using ionic liquid and citric acid. Int. J. Biol. Macromol. 2012, 50, 861–864. [Google Scholar] [CrossRef]
- Wang, W.T.; Zhu, J.; Wang, X.L.; Huang, Y.; Wang, Y.Z. Dissolution behavior of chitin in ionic liquids. J. Macromol. Sci. B 2010, 49, 528–541. [Google Scholar] [CrossRef]
- Li, J.; Huang, W.-C.; Gao, L.; Sun, J.; Liu, Z.; Mao, X. Efficient enzymatic hydrolysis of ionic liquid pretreated chitin and its dissolution mechanism. Carbohyd. Polym. 2019, 211, 329–335. [Google Scholar] [CrossRef]
- Uto, T.; Idenoue, S.; Yamamoto, K.; Kadokawa, J.I. Understanding dissolution process of chitin crystal in ionic liquids: Theoretical study. Phys. Chem. Chem. Phys. PCCP 2018, 20, 20669–20677. [Google Scholar] [CrossRef]
- Tajiri, R.; Setoguchi, T.; Wakizono, S.; Yamamoto, K.; Kadokawa, J.-I. Preparation of self-assembled chitin nanofibers by regeneration from ion gels using calcium halide · dihydrate/methanol solutions. J. Biobased Mater. Bioenergy 2013, 7, 655–659. [Google Scholar] [CrossRef]
- Ifuku, S.; Saimoto, H. Chitin nanofibers: Preparations, modifications, and applications. Nanoscale 2012, 4, 3308–3318. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.S.; Duarte, A.R.C.; Mano, J.F.; Reis, R.L. Design and functionalization of chitin-based microsphere scaffolds. Green Chem. 2013, 15, 3252–3258. [Google Scholar] [CrossRef]
- Jayakumar, R.; Prabaharan, M.; Nair, S.V.; Tamura, H. Novel chitin and chitosan nanofibers in biomedical applications. Biotechnol. Adv. 2010, 28, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Brondani, D.; Dupont, J.; Spinelli, A.; Vieira, I.C. Development of biosensor based on ionic liquid and corn peroxidase immobilized on chemically crosslinked chitin. Sens. Actuators B Chem. 2009, 138, 236–243. [Google Scholar] [CrossRef]
- Xie, H.; Zhang, S.; Li, S. Chitin and chitosan dissolved in ionic liquids as reversible sorbents of CO2. Green Chem. 2006, 8, 630–633. [Google Scholar] [CrossRef]
- Sun, X.F.; Tian, Q.Q.; Xue, Z.M.; Zhang, Y.W.; Mu, T.C. The dissolution behaviour of chitosan in acetate-based ionic liquids and their interactions: From experimental evidence to density functional theory analysis. Rsc. Adv. 2014, 4, 30282–30291. [Google Scholar] [CrossRef]
- Chen, J.; Xie, F.; Li, X.; Chen, L. Ionic liquids for the preparation of biopolymer materials for drug/gene delivery: A review. Green Chem. 2018, 20, 4169–4200. [Google Scholar] [CrossRef]
- Chen, Q.; Xu, A.; Li, Z.; Wang, J.; Zhang, S. Influence of anionic structure on the dissolution of chitosan in 1-butyl-3-methylimidazolium-based ionic liquids. Green Chem. 2011, 13, 3446–3452. [Google Scholar] [CrossRef]
- Feng, J.X.; Zang, H.J.; Yan, Q.; Li, M.G.; Jiang, X.Q.; Cheng, B.W. Dissolution and utilization of chitosan in a 1-carboxymethyl-3-methylimidazolium hydrochloride ionic salt aqueous solution. J. Appl. Polym. Sci. 2015, 132, 41965. [Google Scholar] [CrossRef]
- Wei, S.; Ching, Y.C.; Chuah, C.H. Synthesis of chitosan aerogels as promising carriers for drug delivery: A review. Carbohyd. Polym. 2020, 231, 115744. [Google Scholar] [CrossRef] [PubMed]
- Bozec, L.; de Groot, J.; Odlyha, M.; Nicholls, B.; Nesbitt, S.; Flanagan, A.; Horton, M. Atomic force microscopy of collagen structure in bone and dentine revealed by osteoclastic resorption. Ultramicroscopy 2005, 105, 79–89. [Google Scholar] [CrossRef]
- Bailey, A.J.; Macmillan, J.; Shrewry, P.R.; Tatham, A.S.; Puxkandl, R.; Zizak, I.; Paris, O.; Keckes, J.; Tesch, W.; Bernstorff, S.; et al. Viscoelastic properties of collagen: Synchrotron radiation investigations and structural model. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2002, 357, 191–197. [Google Scholar]
- Bhattacharjee, A.; Bansal, M. Collagen structure: The madras triple helix and the current scenario. IUBMB Life 2005, 57, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Eyre, D. Articular cartilage and changes in Arthritis: Collagen of articular cartilage. Arthritis Res. Ther. 2001, 3, 107. [Google Scholar]
- Meng, Z.; Zheng, X.; Tang, K.; Liu, J.; Ma, Z.; Zhao, Q. Dissolution and regeneration of collagen fibers using ionic liquid. Int. J. Biol. Macromol. 2012, 51, 440–448. [Google Scholar] [CrossRef]
- Luca, S.; Nunzia, G.; Lucia, N.M.; Lorena, C.; Paola, L.; Marta, M.; Stella, B.F.; Angelo, C.; Loredana, C.; Alessandro, S. Marine collagen and its derivatives: Versatile and sustainable bio-resources for healthcare. Mater. Sci. Eng. C 2020, 113, 110963. [Google Scholar]
- Silvipriya, K.; Kumar, K.; Bhat, A.; Kumar, B.D.; John, A.; Lakshmanan, P. Collagen: Animal sources and biomedical application. J. Appl. Pharm. Sci. 2015, 5, 123–127. [Google Scholar] [CrossRef]
- Liu, D.; Zhang, X.; Li, T.; Yang, H.; Zhang, H.; Regenstein, J.M.; Zhou, P. Extraction and characterization of acid- and pepsin-soluble collagens from the scales, skins and swim-bladders of grass carp (Ctenopharyngodon idella). Food Biosci. 2015, 9, 68–74. [Google Scholar] [CrossRef]
- Silva, T.H.; Moreira-Silva, J.; Marques, A.L.P.; Domingues, A.; Bayon, Y.; Reis, R.L. Marine origin collagens and its potential applications. Mar. Drugs 2014, 12, 5881–5901. [Google Scholar] [CrossRef]
- Yadavalli, V.K.; Svintradze, D.V.; Pidaparti, R.M. Nanoscale measurements of the assembly of collagen to fibrils. Int. J. Biol. Macromol. 2010, 46, 458–464. [Google Scholar] [CrossRef] [PubMed]
- Ideia, P.; Pinto, J.; Ferreira, R.; Figueiredo, L.; Spínola, V.; Castilho, P.C. Fish processing industry residues: A review of valuable products extraction and characterization methods. Waste Biomass Valorization 2019. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, C.; Qiao, C.; Mu, X.; Li, T.; Xu, J.; Shi, L.; Zhang, D. A simple and convenient method to synthesize N-[(2-hydroxyl)-propyl-3-trimethylammonium] chitosan chloride in an ionic liquid. Carbohydr. Polym. 2015, 130, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Guillén, M.C.; Giménez, B.; López-Caballero, M.E.; Montero, M.P. Functional and bioactive properties of collagen and gelatin from alternative sources: A review. Food Hydrocoll. 2011, 25, 1813–1827. [Google Scholar] [CrossRef]
- Faria-Silva, C.; Ascenso, A.; Costa, A.M.; Marto, J.; Carvalheiro, M.; Ribeiro, H.M.; Simões, S. Feeding the skin: A new trend in food and cosmetics convergence. Trends Food Sci. Technol. 2020, 95, 21–32. [Google Scholar] [CrossRef]
- Farage, M.A.; Miller, K.W.; Elsner, P.; Maibach, H.I. Intrinsic and extrinsic factors in skin ageing: A review. Int. J. Cosmet. Sci. 2008, 30, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, M.I.A.; Barroso, L.G.R.; Sánchez, M.L. Collagen: A review on its sources and potential cosmetic applications. J. Cosmet. Dermatol. 2018, 17, 20–26. [Google Scholar] [CrossRef]
- Sionkowska, A.; Kozłowska, J. Characterization of collagen/hydroxyapatite composite sponges as a potential bone substitute. Int. J. Biol. Macromol. 2010, 47, 483–487. [Google Scholar] [CrossRef]
- Mandal, A.; Panigrahi, S.; Zhang, C. Collagen as biomaterial for medical application—Drug delivery and scaffolds for tissue regeneration: A review. Biol. Eng. Trans. 2010, 2, 63–88. [Google Scholar] [CrossRef]
- Mandal, A.; Meda, V.; Zhang, W.J.; Farhan, K.M.; Gnanamani, A. Synthesis, characterization and comparison of antimicrobial activity of PEG/TritonX-100 capped silver nanoparticles on collagen scaffold. Colloids Surf. B Biointerfaces 2012, 90, 191–196. [Google Scholar] [CrossRef]
- Iqbal, B.; Muhammad, N.; Jamal, A.; Ahmad, P.; Khan, Z.U.H.; Rahim, A.; Khan, A.S.; Gonfa, G.; Iqbal, J.; Rehman, I.U. An application of ionic liquid for preparation of homogeneous collagen and alginate hydrogels for skin dressing. J. Mol. Liq. 2017, 243, 720–725. [Google Scholar] [CrossRef]
- Bella, J. Collagen structure: New tricks from a very old dog. Biochem. J. 2016, 473, 1001–1025. [Google Scholar] [CrossRef] [PubMed]
- Friess, W. Collagen–biomaterial for drug delivery1Dedicated to Professor Dr. Eberhard Nürnberg, Friedrich-Alexander-Universität Erlangen-Nürnberg, on the occasion of his 70th birthday.1. Eur. J. Pharm. Biopharm. 1998, 45, 113–136. [Google Scholar] [CrossRef]
- Poluboyarov, V.; Voloskova, E.; Yankovaya, V.; Guryanova, T. Intensification of collagen dissolution process with the help of mechanochemical treatment. Chem. Sustain. Dev. 2009, 17, 177–183. [Google Scholar]
- Mehta, A.; Raghava Rao, J.; Fathima, N.N. Electrostatic forces mediated by choline dihydrogen phosphate stabilize collagen. J. Phys. Chem. B 2015, 119, 12816–12827. [Google Scholar] [CrossRef]
- Vijayaraghavan, R.; Thompson, B.C.; MacFarlane, D.R.; Kumar, R.; Surianarayanan, M.; Aishwarya, S.; Sehgal, P.K. Biocompatibility of choline salts as crosslinking agents for collagen based biomaterials. Chem. Commun. 2010, 46, 294–296. [Google Scholar] [CrossRef]
- Sharma, A.; Rawat, K.; Solanki, P.R.; Bohidar, H.B. Self-healing gelatin ionogels. Int. J. Biol. Macromol. 2017, 95, 603–607. [Google Scholar] [CrossRef]
- Lourenço, N.; Nunes, A.; Duarte, C.; Vidinha, P. Ionic liquids gelation with polymeric materials: The ion jelly approach. In Applications of Ionic Liquids in Science and Technology; Handy, P.S., Ed.; Scott Handy, IntechOpen: London, UK, 2011; pp. 155–172. [Google Scholar]
- Carvalho, T.; Augusto, V.; Rocha, Â.; Lourenço, N.M.T.; Correia, N.T.; Barreiros, S.; Vidinha, P.; Cabrita, E.J.; Dionísio, M. Ion jelly conductive properties using dicyanamide-based ionic liquids. J. Phys. Chem. B 2014, 118, 9445–9459. [Google Scholar] [CrossRef]
- Lourenço, N.M.T.; Österreicher, J.; Vidinha, P.; Barreiros, S.; Afonso, C.A.M.; Cabral, J.M.S.; Fonseca, L.P. Effect of gelatin–ionic liquid functional polymers on glucose oxidase and horseradish peroxidase kinetics. React. Funct. Polym. 2011, 71, 489–495. [Google Scholar] [CrossRef]
- Yan, R.; Zhao, F.; Li, J.; Xiao, F.; Fan, S.; Zeng, B. Direct electrochemistry of horseradish peroxidase in gelatin-hydrophobic ionic liquid gel films. Electrochim. Acta 2007, 52, 7425–7431. [Google Scholar] [CrossRef]
- Maneewattanapinyo, P.; Yeesamun, A.; Watthana, F.; Panrat, K.; Pichayakorn, W.; Suksaeree, J. Controlled release of lidocaine–diclofenac ionic liquid drug from freeze-thawed Gelatin/Poly(Vinyl Alcohol) transdermal patches. AAPS PharmSciTech 2019, 20, 322. [Google Scholar] [CrossRef] [PubMed]
- Prasad, K.; Kaneko, Y.; Kadokawa, J.-I. Novel gelling systems of κ-, ι- and λ-Carrageenans and their composite gels with cellulose using ionic liquid. Macromol. Biosci. 2009, 9, 376–382. [Google Scholar] [CrossRef] [PubMed]
- King, C.; Shamshina, J.L.; Gurau, G.; Berton, P.; Khan, N.F.A.F.; Rogers, R.D. A platform for more sustainable chitin films from an ionic liquid process. Green Chem. 2017, 19, 117–126. [Google Scholar] [CrossRef]
- Dias, A.M.A.; Cortez, A.R.; Barsan, M.M.; Santos, J.B.; Brett, C.M.A.; de Sousa, H.C. Development of greener multi-responsive chitosan biomaterials doped with biocompatible ammonium ionic liquids. Acs Sustain. Chem. Eng. 2013, 1, 1480–1492. [Google Scholar] [CrossRef]
- Jia, X.; Yang, Y.; Wang, C.; Zhao, C.; Vijayaraghavan, R.; MacFarlane, D.R.; Forsyth, M.; Wallace, G.G. Biocompatible ionic liquid–biopolymer electrolyte-enabled thin and compact magnesium–air batteries. ACS Appl. Mater. Interfaces 2014, 6, 21110–21117. [Google Scholar] [CrossRef] [PubMed]
- Shamshina, J.L. Chitin in ionic liquids: Historical insights into the polymer’s dissolution and isolation. A review. Green Chem. 2019, 21, 3974–3993. [Google Scholar] [CrossRef]
- Ma, B.; Qin, A.; Li, X.; He, C. High tenacity regenerated chitosan fibers prepared by using the binary ionic liquid solvent (Gly·HCl)-[Bmim]Cl. Carbohyd. Polym. 2013, 97, 300–305. [Google Scholar] [CrossRef]
- Ma, B.; Zhang, M.; He, C.; Sun, J. New binary ionic liquid system for the preparation of chitosan/cellulose composite fibers. Carbohyd. Polym. 2012, 88, 347–351. [Google Scholar] [CrossRef]
- Park, T.-J.; Jung, Y.J.; Choi, S.-W.; Park, H.; Kim, H.; Kim, E.; Lee, S.H.; Kim, J.H. Native chitosan/cellulose composite fibers from an ionic liquid via electrospinning. Macromol. Res. 2011, 19, 213–215. [Google Scholar] [CrossRef]
- Zhai, Z.; Wang, H.; Wei, B.; Yu, P.; Xu, C.; He, L.; Zhang, J.; Xu, Y. Effect of ionic liquids on the fibril-formation and gel properties of grass carp (Ctenopharyngodon idellus) skin collagen. Macromol. Res. 2018, 26, 609–615. [Google Scholar] [CrossRef]
- Shi, D.; Mi, G.; Bhattacharya, S.; Nayar, S.; Webster, T.J. Optimizing superparamagnetic iron oxide nanoparticles as drug carriers using an in vitro blood-brain barrier model. Int. J. Nanomed. 2016, 11, 5371–5379. [Google Scholar] [CrossRef] [PubMed]
- Sehgal, P.K.; Srinivasan, A. Collagen-coated microparticles in drug delivery. Expert Opin. Drug Deliv. 2009, 6, 687–695. [Google Scholar] [CrossRef] [PubMed]
- David, G. Chapter 35—Collagen-based 3D structures—Versatile, efficient materials for biomedical applications. In Biopolymer-Based Formulations; Pal, K., Banerjee, I., Sarkar, P., Kim, D., Deng, W.-P., Dubey, N.K., Majumder, K., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 881–906. [Google Scholar]
- Maeda, Y.; Wei, Z.; Matsui, H. Biomimetic assembly of proteins into microcapsules on oil-in-water droplets with structural reinforcement via biomolecular-recognition-based cross-linking of surface peptides. Small 2012, 8, 1341–1344. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, M.-a.; Takano, A.; Tao, S.; Kimizuka, N. Biopolymer-Encapsulated Protein Microcapsules Spontaneously Formed at the Ionic Liquid–Water Interface. Biomacromolecules 2012, 13, 4075–4080. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Alexandridis, P. Nanoparticles in ionic liquids: Interactions and organization. Phys. Chem. Chem. Phys. 2015, 17, 18238–18261. [Google Scholar] [CrossRef]
- Montalbán, M.G.; Carissimi, G.; Lozano-Pérez, A.A.; Cenis, J.L.; Coburn, J.M.; Kaplan, D.L.; Víllora, G. Biopolymeric Nanoparticle Synthesis in Ionic Liquids; IntechOpen: London, UK, 2018; pp. 3–26. [Google Scholar]
- Chan, O.C.M.; So, K.F.; Chan, B.P. Fabrication of nano-fibrous collagen microspheres for protein delivery and effects of photochemical crosslinking on release kinetics. J. Control. Release 2008, 129, 135–143. [Google Scholar] [CrossRef]
- Solanki, P. Gelatin nanoparticles as a delivery system for proteins. J. Nanomed. Res. 2015, 2, 1–3. [Google Scholar] [CrossRef]
- Bharmoria, P.; Singh, T.; Kumar, A. Complexation of chitosan with surfactant like ionic liquids: Molecular interactions and preparation of chitosan nanoparticles. J. Colloid Interface Sci. 2013, 407, 361–369. [Google Scholar] [CrossRef]
- Silva, S.S.; Duarte, A.R.; Mano, J.F.; Reis, R.L. Development of a supercritical assisted particle-agglomeration method for the preparation of bioactive chitin-based matrices. In Proceedings of the 10th International Symposium on Supercritical Fluids San Franciscorices, San Francisco, CA, USA., 13–16 May 2012. [Google Scholar]
- Silva, S.S.; Duarte, A.R.C.; Oliveira, J.M.; Mano, J.F.; Reis, R.L. Alternative methodology for chitin–Hydroxyapatite composites using ionic liquids and supercritical fluid technology. J. Bioact. Compat. Polym. 2013, 28, 481–491. [Google Scholar] [CrossRef]
- Kuzmina, O. Chapter 5-methods of IL recovery and destruction. In Application, Purification, and Recovery of Ionic Liquids; Kuzmina, O., Hallett, J.P., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 205–248. [Google Scholar]
- Shamshina, J.L.; Berton, P. Use of ionic liquids in chitin biorefinery: A systematic review. Front. Bioeng. Biotechnol. 2020, 8, 11. [Google Scholar] [CrossRef]
- Barber, P.S.; Griggs, C.S.; Gurau, G.; Liu, Z.; Li, S.; Li, Z.; Lu, X.; Zhang, S.; Rogers, R.D. Coagulation of chitin and cellulose from 1-ethyl-3-methylimidazolium acetate ionic-liquid solutions using carbon dioxide. Angew. Chem. 2013, 52, 12350–12353. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, B.; Sarfaraz, Z.; Muhammad, N.; Ahmad, P.; Iqbal, J.; Khan, Z.U.H.; Gonfa, G.; Iqbal, F.; Jamal, A.; Rahim, A. Ionic liquid as a potential solvent for preparation of collagen-alginate-hydroxyapatite beads as bone filler. J. Biomater. Sci. Polym. Ed. 2018, 29, 1168–1184. [Google Scholar] [CrossRef] [PubMed]
- Tran, C.D.; Duri, S.; Harkins, A.L. Recyclable synthesis, characterization, and antimicrobial activity of chitosan-based polysaccharide composite materials. J. Biomed. Mater. Res. Part A 2013, 101, 2248–2257. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Gao, Y.; Wang, L.; Chen, H.; Yan, N. Effect of treatment methods on chitin structure and its transformation into nitrogen-containing chemicals. ChemPlusChem 2015, 80, 1565–1572. [Google Scholar] [CrossRef]
- Earl, J.M.; Seddon, K.R. Preparation of imidazole carbene and the use thereof for the synthesis of ionic liquids. WO 2001, 1, 20. [Google Scholar]
- Tran, C.D.; Duri, S.; Delneri, A.; Franko, M. Chitosan-cellulose composite materials: Preparation, Characterization and application for removal of microcystin. J. Hazard. Mater. 2013, 252, 355–366. [Google Scholar] [CrossRef]
- Singh, N.; Koziol, K.K.K.; Chen, J.; Patil, A.J.; Gilman, J.W.; Trulove, P.C.; Kafienah, W.; Rahatekar, S.S. Ionic liquids-based processing of electrically conducting chitin nanocomposite scaffolds for stem cell growth. Green Chem. 2013, 15, 1192–1202. [Google Scholar] [CrossRef]
- Li, X.; Fan, D. Smart collagen hydrogels based on 1-Ethyl-3-methylimidazolium acetate and microbial transglutaminase for potential applications in tissue engineering and cancer therapy. ACS Biomater. Sci. Eng. 2019, 5, 3523–3536. [Google Scholar] [CrossRef]
- Leones, R.; Sentanin, F.; Rodrigues, L.C.; Ferreira, R.A.S.; Marrucho, I.M.; Esperança, J.M.S.S.; Pawlicka, A.; Carlos, L.D.; Silva, M.M. Novel polymer electrolytes based on gelatin and ionic liquids. Opt. Mater. 2012, 35, 187–195. [Google Scholar] [CrossRef]
- Shamshina, J.L.; Zavgorodnya, O.; Berton, P.; Chhotaray, P.K.; Choudhary, H.; Rogers, R.D. Ionic liquid platform for spinning composite Chitin–Poly(lactic acid) fibers. ACS Sustain. Chem. Eng. 2018, 6, 10241–10251. [Google Scholar] [CrossRef]
- Gonçalves, C.; Silva, S.S.; Gomes, J.M.; Oliveira, I.M.; Canadas, R.F.; Maia, F.R.; Radhouani, H.; Reis, R.L.; Oliveira, J.M. Ionic liquid-mediated processing of SAIB-Chitin scaffolds. ACS Sustain. Chem. Eng. 2020, 8, 3986–3994. [Google Scholar] [CrossRef]
- Harkins, A.L.; Duri, S.; Kloth, L.C.; Tran, C.D. Chitosan-cellulose composite for wound dressing material. Part 2. Antimicrobial activity, blood absorption ability, and biocompatibility. J. Biomed. Mater. Res. Part B Appl. Biomater. 2014, 102, 1199–1206. [Google Scholar] [CrossRef] [PubMed]
- Tran, C.D.; Mututuvari, T.M. Cellulose, chitosan, and keratin composite materials. controlled drug release. Langmuir 2015, 31, 1516–1526. [Google Scholar] [CrossRef] [PubMed]
- Scabbia, A.; Trombelli, L. A comparative study on the use of a HA/collagen/chondroitin sulphate biomaterial (Biostite®) and a bovine-derived HA xenograft (Bio-Oss®) in the treatment of deep intra-osseous defects. J. Clin. Periodontol. 2004, 31, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, B.; Muhammad, N.; Rahim, A.; Iqbal, F.; Sharif, F.; Safi, S.Z.; Khan, A.S.; Gonfa, G.; Uroos, M.; Rehman, I.U. Development of collagen/PVA composites patches for osteochondral defects using a green processing of ionic liquid. Int. J. Polym. Mater. Polym. Biomater. 2019, 68, 590–596. [Google Scholar] [CrossRef]
- Ion, R.-M.; Poinescu, A.; Doncea, S. Novel three-component composite materials (Hydroxyapatite/Polymer mixtures) for bone regeneration. Key Eng. Mater. 2014, 587, 197–204. [Google Scholar] [CrossRef]
- Wahl, D.; Sachlos, E.; Liu, C.; Czernuszka, J. Controlling the processing of collagen-hydroxyapatite scaffolds for bone tissue engineering. J. Mater. Sci. Mater. Med. 2007, 18, 201–209. [Google Scholar] [CrossRef]
- Ryan, A.J.; Gleeson, J.P.; Matsiko, A.; Thompson, E.M.; O’Brien, F.J. Effect of different hydroxyapatite incorporation methods on the structural and biological properties of porous collagen scaffolds for bone repair. J. Anat. 2015, 227, 732–745. [Google Scholar] [CrossRef]
- Hua, D.; Jiang, J.; Kuang, L.; Jiang, J.; Zheng, W.; Liang, H. Smart chitosan-based stimuli-responsive nanocarriers for the controlled delivery of hydrophobic pharmaceuticals. Macromolecules 2011, 44, 1298–1302. [Google Scholar] [CrossRef]
- Chen, H.; Cui, S.; Zhao, Y.; Wang, B.; Zhang, S.; Peng, X. O-Alkylation of chitosan for gene delivery by using ionic liquid in an in- situ reactor. Engineering 2012, 4, 114–117. [Google Scholar] [CrossRef]
- Chen, H.; Cui, S.; Zhao, Y.; Zhang, C.; Zhang, S.; Peng, X. Grafting chitosan with polyethylenimine in an ionic liquid for efficient gene delivery. PLoS ONE 2015, 10, e0121817. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, S.S.; Gomes, J.M.; Rodrigues, L.C.; Reis, R.L. Marine-Derived Polymers in Ionic Liquids: Architectures Development and Biomedical Applications. Mar. Drugs 2020, 18, 346. https://doi.org/10.3390/md18070346
Silva SS, Gomes JM, Rodrigues LC, Reis RL. Marine-Derived Polymers in Ionic Liquids: Architectures Development and Biomedical Applications. Marine Drugs. 2020; 18(7):346. https://doi.org/10.3390/md18070346
Chicago/Turabian StyleSilva, Simone S., Joana M. Gomes, Luísa C. Rodrigues, and Rui L. Reis. 2020. "Marine-Derived Polymers in Ionic Liquids: Architectures Development and Biomedical Applications" Marine Drugs 18, no. 7: 346. https://doi.org/10.3390/md18070346
APA StyleSilva, S. S., Gomes, J. M., Rodrigues, L. C., & Reis, R. L. (2020). Marine-Derived Polymers in Ionic Liquids: Architectures Development and Biomedical Applications. Marine Drugs, 18(7), 346. https://doi.org/10.3390/md18070346








