Anti-Proliferation Activity of a Decapeptide from Perinereies aibuhitensis toward Human Lung Cancer H1299 Cells
Abstract
1. Introduction
2. Results and Discussion
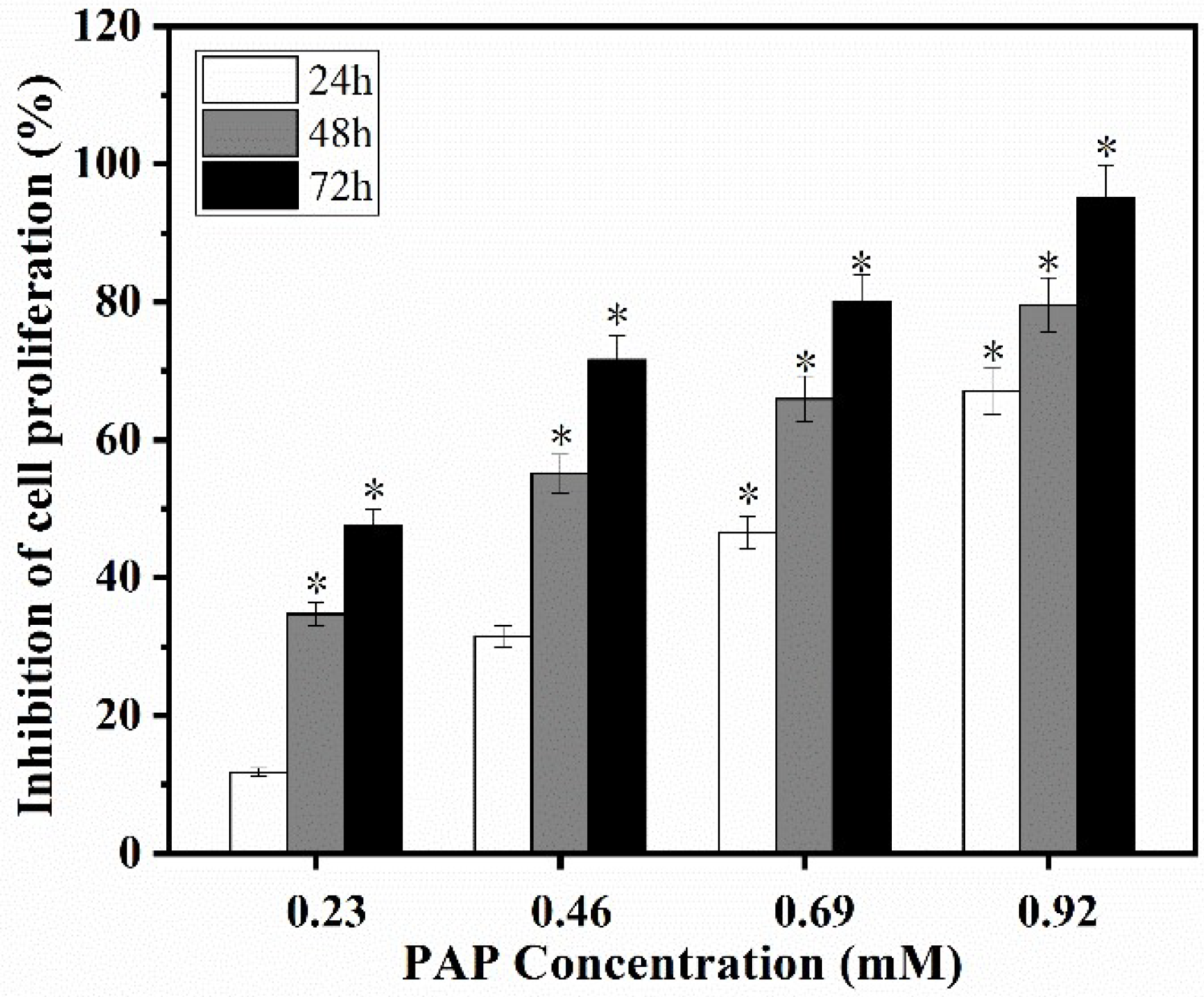
2.1. Effects of PAP on the Proliferation of H1299 Cells
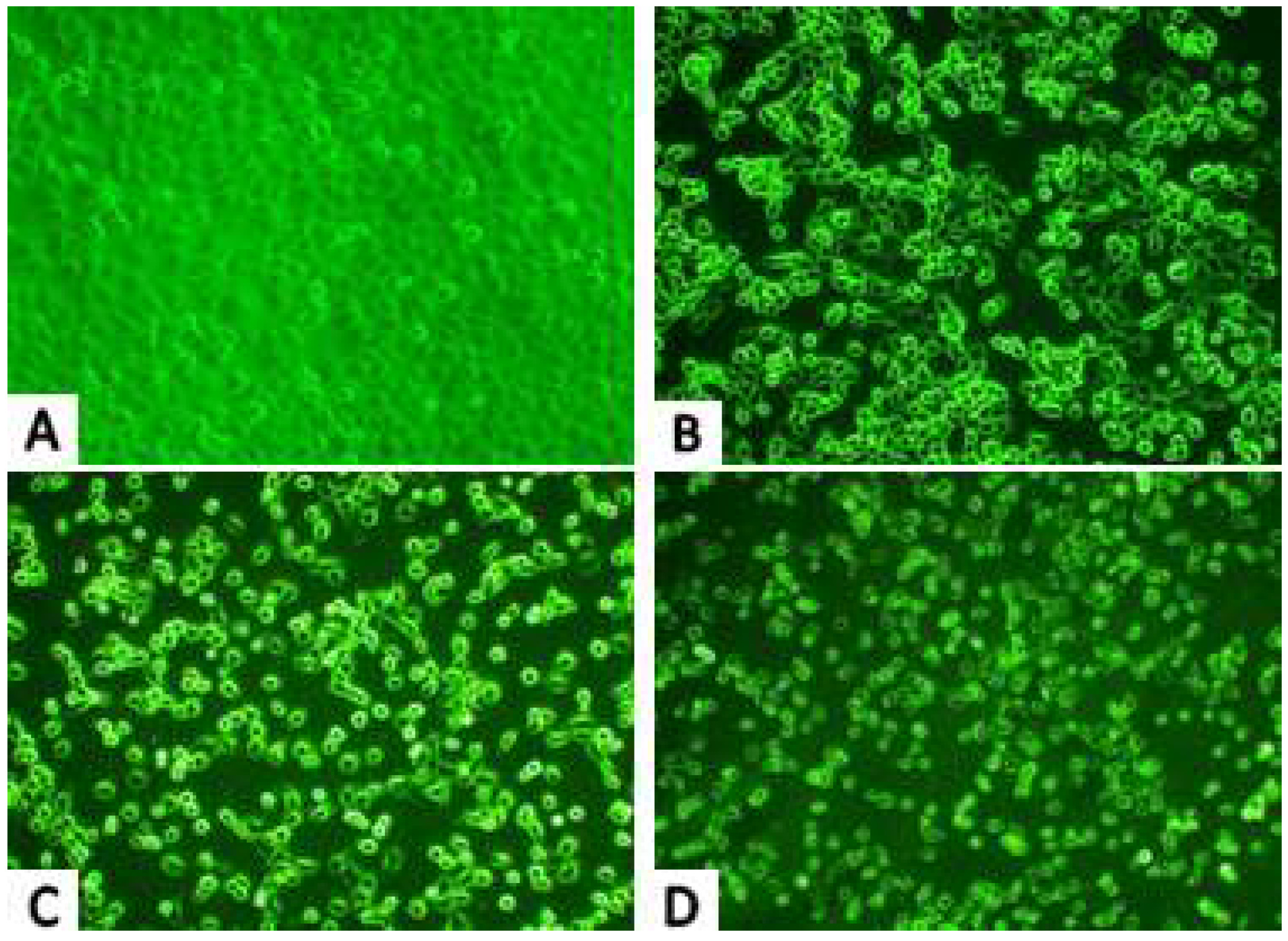
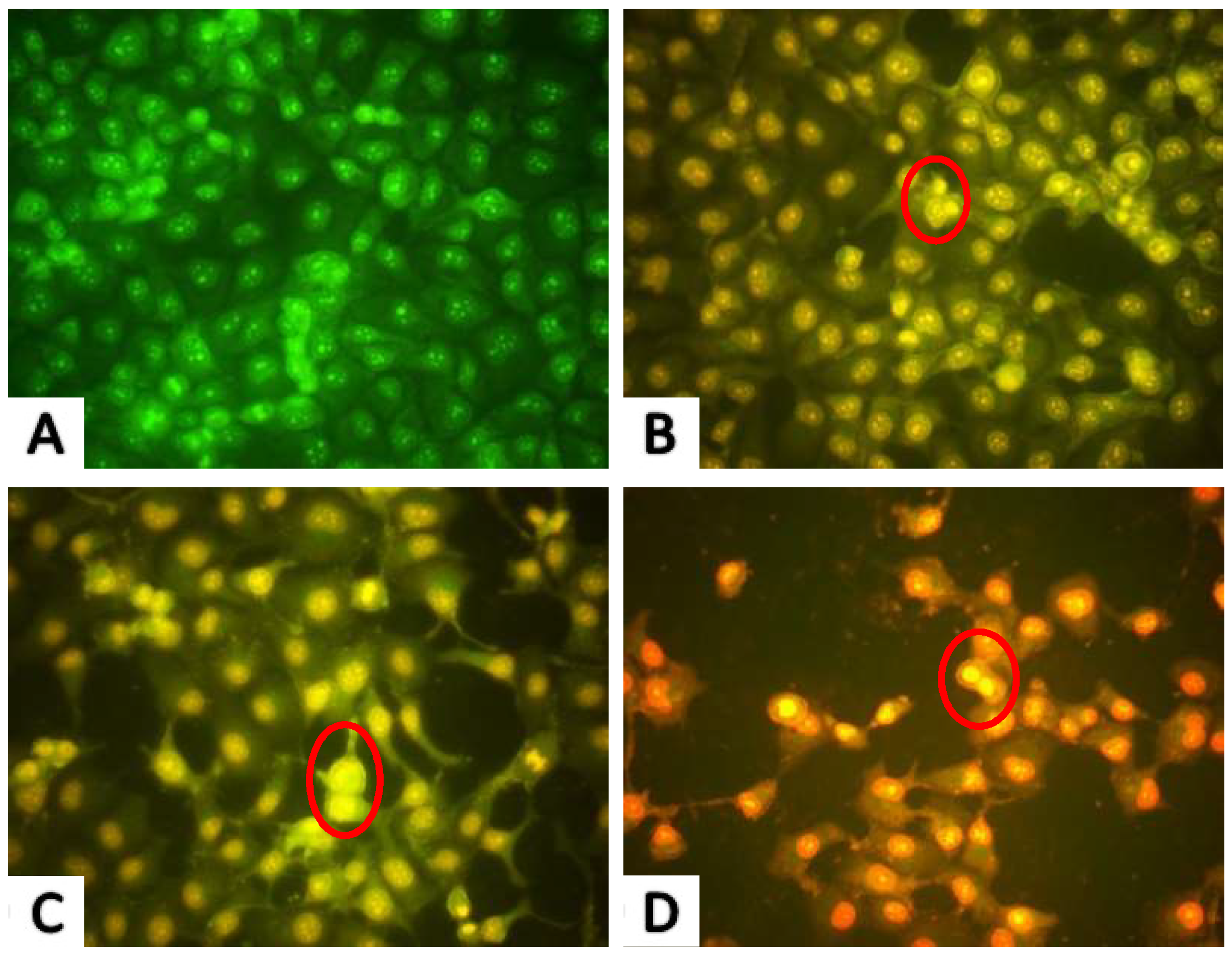
2.2. Morphological Observations
2.2.1. Inverted Microscope Observations
2.2.2. AO/EB Fluorescence Staining Results
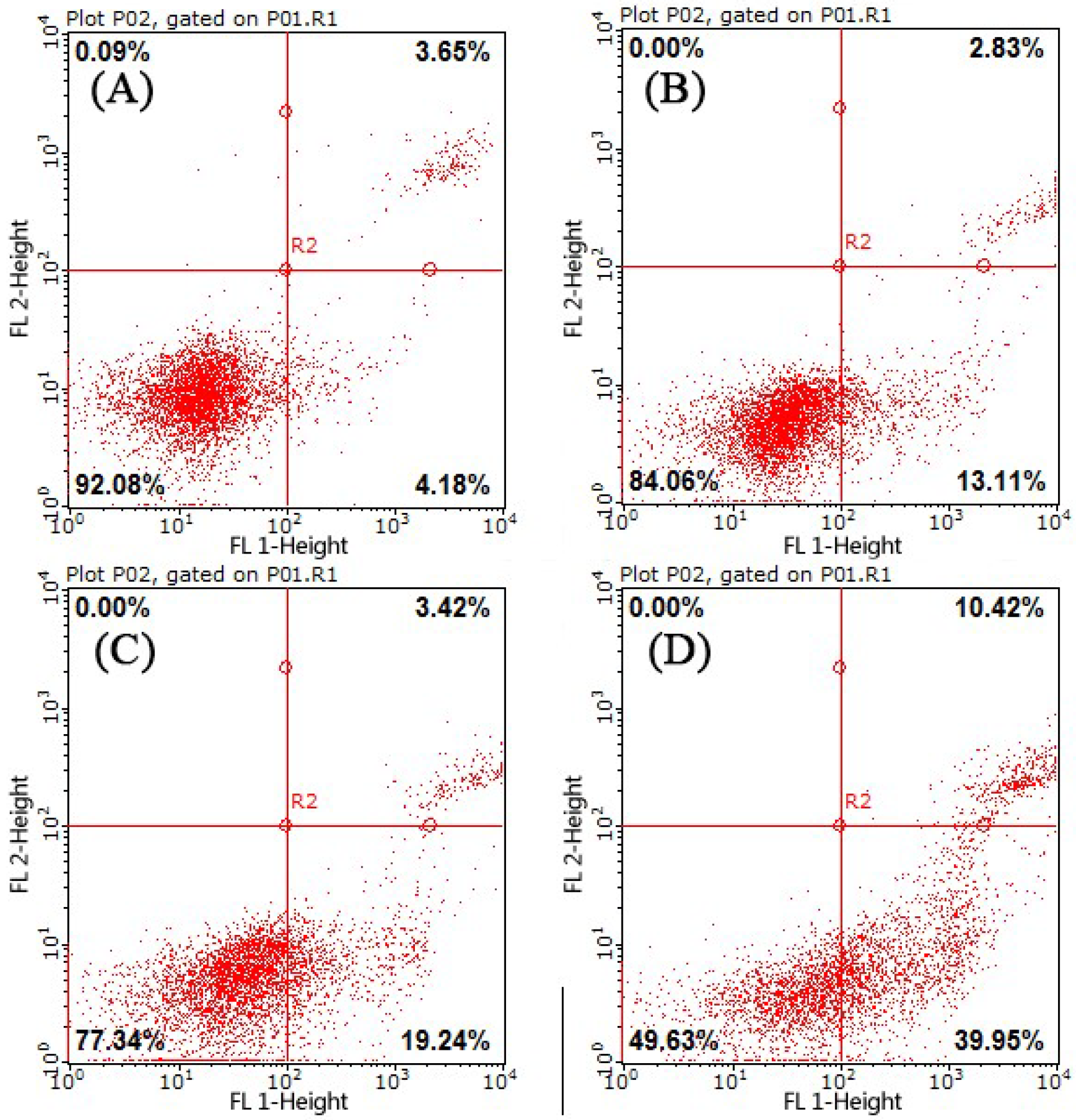
2.3. Cell Apoptosis Analysis
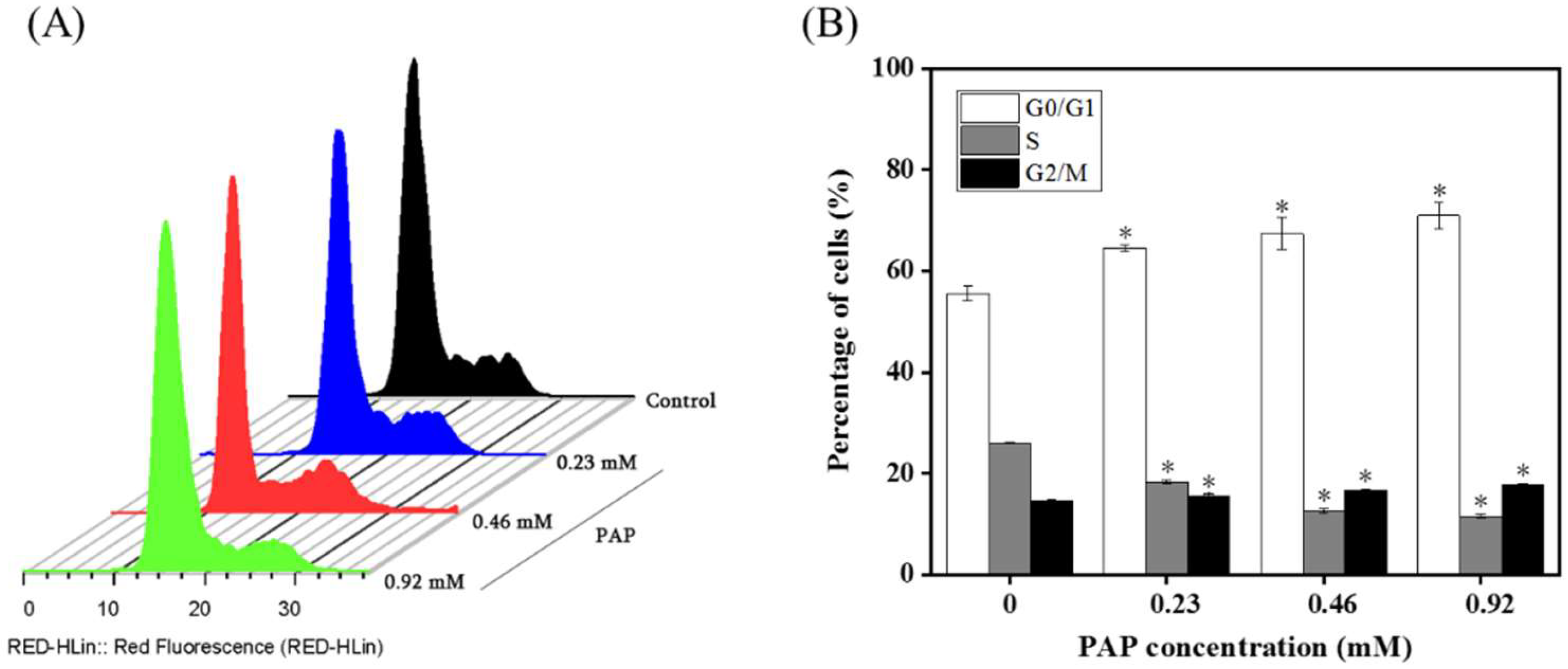
2.4. Effects of PAP on the Cell Cycle Distribution of H1299 Cells
2.5. Western Blotting Results
3. Materials and Methods
3.1. Materials and Reagents
3.2. Detection of Anti-Proliferation Activity Using CCK-8
3.3. Cell Morphology Observation Using an Inverted Microscope
3.4. Cell Morphological Analysis by AO/EB Staining
3.5. Cell Apoptosis Analysis Using Annexin V FITC/PI
3.6. Cell Cycle Analysis by Propidium Iodide Staining
3.7. Detection of Protein Expression by Western Blotting
3.8. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Teh, E.; Belcher, E. Lung cancer: Diagnosis, staging and treatment. Surgery (Oxford) 2014, 32, 242–248. [Google Scholar] [CrossRef]
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Dyba, T.; Randi, G.; Bettio, M.; Gavin, A.; Visser, O.; Bray, F. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries and 25 major cancers in 2018. Eur. J. Cancer 2018, 103, 356–387. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Chirieac, L.R.; Attanoos, R.L. 26—Usual Lung Cancers. In Pulmonary Pathology, 2nd ed.; Zander, D.S., Farver, C.F., Eds.; Content Repository Only: Philadelphia, PA, USA, 2018; pp. 534–551. [Google Scholar]
- Semreen, M.H.; El-Gamal, M.I.; Abdin, S.; Alkhazraji, H.; Kamal, L.; Hammad, S.; El-Awady, F.; Waleed, D.; Kourbaj, L. Recent updates of marine antimicrobial peptides. Saudi Pharm. J. 2018, 26, 396–409. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Nong, X.-H.; Ren, Z.; Wang, J.; Liang, X.; Wang, L.; Qi, S.-H. Antiviral peptides from marine gorgonian-derived fungus Aspergillus sp. SCSIO 41501. Tetrahedron Lett. 2017, 58, 1151–1155. [Google Scholar] [CrossRef]
- Lee, Y.; Phat, C.; Hong, S.-C. Structural diversity of marine cyclic peptides and their molecular mechanisms for anticancer, antibacterial, antifungal, and other clinical applications. Peptides 2017, 95, 94–105. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Hur, S.J. Antihypertensive peptides from animal products, marine organisms, and plants. Food Chem. 2017, 228, 506–517. [Google Scholar] [CrossRef] [PubMed]
- Gogineni, V.; Hamann, M.T. Marine natural product peptides with therapeutic potential: Chemistry, biosynthesis, and pharmacology. BBA-Gen Subj. 2018, 1862, 81–196. [Google Scholar] [CrossRef]
- Sila, A.; Bougatef, A. Antioxidant peptides from marine by-products: Isolation, identification and application in food systems. A review. J. Funct. Foods 2016, 21, 10–26. [Google Scholar] [CrossRef]
- Yu, F.; Zhang, Y.; Ye, L.; Tang, Y.; Ding, G.; Zhang, X.; Yang, Z. A novel antiproliferative pentapeptide (ILYMP) isolated from Cyclina sinensis protein hydrolysate induces apoptosis of DU145 prostate cancer cells. Mol. Med. Rep. 2018, 18, 771–778. [Google Scholar]
- Wu, Z.Z.; Ding, G.F.; Huang, F.F.; Yang, Z.S.; Yu, F.M.; Tang, Y.P.; Jia, Y.L.; Zheng, Y.Y.; Chen, R. Anticancer Activity of Anthopleura anjunae Oligopeptides in Prostate Cancer DU-145 Cells. Mar. Drugs 2018, 16, 125. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, X. Isolation and identification of anti-proliferative peptides from Spirulina platensis using three-step hydrolysis. J. Sci. Food Agric. 2016, 97, 918–922. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Zhao, Y.-Q.; Hu, F.-Y.; Chi, C.-F.; Wang, B. Anticancer Activity of a Hexapeptide from Skate (Raja porosa) Cartilage Protein Hydrolysate in HeLa Cells. Mar. Drugs 2016, 14, 153. [Google Scholar] [CrossRef] [PubMed]
- Sun, R.P. Fauna Sinica Invertebrata (Vol.33) Annelida Polychaeta II Nereidida; Science Press: Beijing, China, 2004. [Google Scholar]
- Samidurai, K.; Saravanakumar, A. Mosquitocidal properties of nereistoxin against Anopheles stephensi, Aedes aegypti and Culex quinquefasciatus (Diptera: Culicidae). Parasitol. Res. 2011, 109, 1107–1112. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, J.; Liu, T.; Wang, Y.; Zhou, Z.; Cheng, F.; Feng, C.; Cheng, X.; Liu, H.; Chen, X. Preparation and antithrombotic activity identification of Perinereis aibuhitensis extract: A high temperature and wide pH range stable biological agent. Food Funct. 2017, 8, 3533–3541. [Google Scholar] [CrossRef]
- Chen, L.; Wang, S. Preparation of an ACE-inhibitory peptide from Perinereis aibuhitensis protein. Biotechnol. Biotechnol. Equip. 2017, 31, 1231–1236. [Google Scholar] [CrossRef]
- Pan, W.; Liu, X.; Ge, F.; Han, J.; Zheng, T. Perinerin, a novel antimicrobial peptide purified from the clamworm Perinereis aibuhitensis grube and its partial characterization. J. Biochem. 2004, 135, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Ding, G.; Yang, Z.; Yu, F.; Zheng, Y.; Wu, Z.; Rui, C. Anticancer Activity of a Novel Peptide Derived from Hydrolysates of Perinereies aibuhitensis against Lung Cancer A549 Cells. Food Sci. 2017, 38, 27–35. [Google Scholar]
- Moody, T.W.; Pradhan, T.; Mantey, S.A.; Jensen, R.T.; Dyba, M.; Moody, D.; Tarasova, N.I.; Michejda, C.J. Bombesin marine toxin conjugates inhibit the growth of lung cancer cells. Life Sci. 2008, 82, 855–861. [Google Scholar] [CrossRef]
- Huang, F.; Yang, Z.; Yu, D.; Wang, J.; Li, R.; Ding, G. Sepia ink oligopeptide induces apoptosis in prostate cancer cell lines via caspase-3 activation and elevation of Bax/Bcl-2 ratio. Mar. Drugs 2012, 10, 2153–2165. [Google Scholar] [CrossRef]
- Yang, Z.; Zhao, Y.; Yan, H.; Xu, L.; Ding, G.; Yu, D.; Sun, Y. Isolation and purification of oligopeptides from Ruditapes philippinarum and its inhibition on the growth of DU145 cells in vitro. Mol. Med. Rep. 2015, 11, 1063–1068. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.Z.; Dai, X.L.; Liu, H.Y. Molecular mechanisms underlying the α-tomatine-directed apoptosis in human malignant glioblastoma cell lines A172 and U-118 MG. Exp. Ther. Med. 2017, 14, 6183–6192. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Farkas, R.; Pozsgai, E.; Bellyei, S.; Cseke, L.; Szigeti, A.; Vereczkei, A.; Marton, S.; Mangel, L.; Horvath, O.P.; Papp, A. Correlation between tumor-associated proteins and response to neoadjuvant treatment in patients with advanced squamous-cell esophageal cancer. Anticancer Res. 2011, 31, 1769–1775. [Google Scholar] [PubMed]
- Callagy, G.M.; Pharoah, P.D.; Pinder, S.E.; Hsu, F.D.; Nielsen, T.O.; Ragaz, J.; Ellis, I.O.; Huntsman, D.; Caldas, C. Bcl-2 is a prognostic marker in breast cancer independently of the Nottingham Prognostic Index. Clin. Cancer Res. 2006, 12, 2468–2475. [Google Scholar] [CrossRef] [PubMed]
- Gross, A.; Jockel, J.; Wei, M.C.; Korsmeyer, S.J. Enforced dimerization of BAX results in its translocation, mitochondrial dysfunction and apoptosis. Embo J. 1998, 17, 3878–3885. [Google Scholar] [CrossRef]
- Wan, L.; Zhang, D.; Zhang, J.; Ren, L. TT-1, an analog of melittin, triggers apoptosis in human thyroid cancer TT cells via regulating caspase, Bcl-2 and Bax. Oncol. Lett. 2018, 15, 1271–1278. [Google Scholar] [CrossRef]
- Fuentes-Prior, P.; Salvesen, G.S. The protein structures that shape caspase activity, specificity, activation and inhibition. Biochem. J. 2004, 384, 201–232. [Google Scholar] [CrossRef]
- Che, G.; Zhou, Q.; Wang, Y.; Liu, L.; Qin, Y.; Sun, Z.; Sun, Z.; Chen, X. Effect of nm23-H1 on reversing malignant phenotype on human lung cancer cell line L9981. J. Biomed. Eng. 2005, 22, 530–534. [Google Scholar]
- Marino, N.; Marshall, J.-C.; Steeg, P.S. Protein-protein interactions: A mechanism regulating the anti-metastatic properties of Nm23-H1. N-S Arch. Pharmacol. 2011, 384, 351–362. [Google Scholar] [CrossRef]
- O’Byrne, K.J.; Koukourakis, M.I.; Giatromanolaki, A.; Cox, G.; Turley, H.; Steward, W.P.; Gatter, K.; Harris, A.L. Vascular endothelial growth factor, platelet-derived endothelial cell growth factor and angiogenesis in non-small-cell lung cancer. Br. J. Cancer 2000, 82, 1427–1432. [Google Scholar] [CrossRef]
- Xu, T.; Li, D.; Wang, H.; Zheng, T.; Wang, G.; Xin, Y. MUC1 downregulation inhibits non-small cell lung cancer progression in human cell lines. Exp. Ther. Med. 2017, 14, 4443–4447. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Hu, X.; Liu, G.; Liu, H.; Hu, J.; Feng, Z.; Tang, B.; Qian, J.; Wang, Q.; Zhang, Y. A potential anticancer agent 1,2-di (quinazolin-4-yl) diselane induces apoptosis in non-small-cell lung cancer A549 cells. Med. Chem. Res. 2015, 24, 2085–2096. [Google Scholar] [CrossRef]
- Yoon, H.-E.; Kim, S.-A.; Choi, H.-S.; Ahn, M.-Y.; Yoon, J.-H.; Ahn, S.-G. Inhibition of Plk1 and Pin1 by 5′-nitro-indirubinoxime suppresses human lung cancer cells. Cancer Lett. 2012, 316, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Tang, Y.; Yu, F.; Sun, Y.; Huang, F.; Chen, Y.; Yang, Z.; Ding, G. Inhibition of Prostate Cancer DU-145 Cells Proliferation by Anthopleura anjunae Oligopeptide (YVPGP) via PI3K/AKT/mTOR Signaling Pathway. Mar. Drugs 2018, 16, 325. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Fu, Z.-Z.; Guo, C.-S.; Zhang, Y.-X.; Di, Y.; Jiang, B.; Li, Q.-W. Effects and Mechanism of Baicalin on Apoptosis of Cervical Cancer HeLa Cells in vitro. Iran J. Pharm. Res. 2015, 14, 251–261. [Google Scholar] [PubMed]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, S.; Jia, Y.; Tang, Y.; Zheng, D.; Han, X.; Yu, F.; Chen, Y.; Huang, F.; Yang, Z.; Ding, G. Anti-Proliferation Activity of a Decapeptide from Perinereies aibuhitensis toward Human Lung Cancer H1299 Cells. Mar. Drugs 2019, 17, 122. https://doi.org/10.3390/md17020122
Jiang S, Jia Y, Tang Y, Zheng D, Han X, Yu F, Chen Y, Huang F, Yang Z, Ding G. Anti-Proliferation Activity of a Decapeptide from Perinereies aibuhitensis toward Human Lung Cancer H1299 Cells. Marine Drugs. 2019; 17(2):122. https://doi.org/10.3390/md17020122
Chicago/Turabian StyleJiang, Shuoqi, Yinglu Jia, Yunping Tang, Die Zheng, Xingbiao Han, Fangmiao Yu, Yan Chen, Fangfang Huang, Zuisu Yang, and Guofang Ding. 2019. "Anti-Proliferation Activity of a Decapeptide from Perinereies aibuhitensis toward Human Lung Cancer H1299 Cells" Marine Drugs 17, no. 2: 122. https://doi.org/10.3390/md17020122
APA StyleJiang, S., Jia, Y., Tang, Y., Zheng, D., Han, X., Yu, F., Chen, Y., Huang, F., Yang, Z., & Ding, G. (2019). Anti-Proliferation Activity of a Decapeptide from Perinereies aibuhitensis toward Human Lung Cancer H1299 Cells. Marine Drugs, 17(2), 122. https://doi.org/10.3390/md17020122





