Bioactive Compounds from Marine Organisms: Potential for Bone Growth and Healing
Abstract
1. Introduction
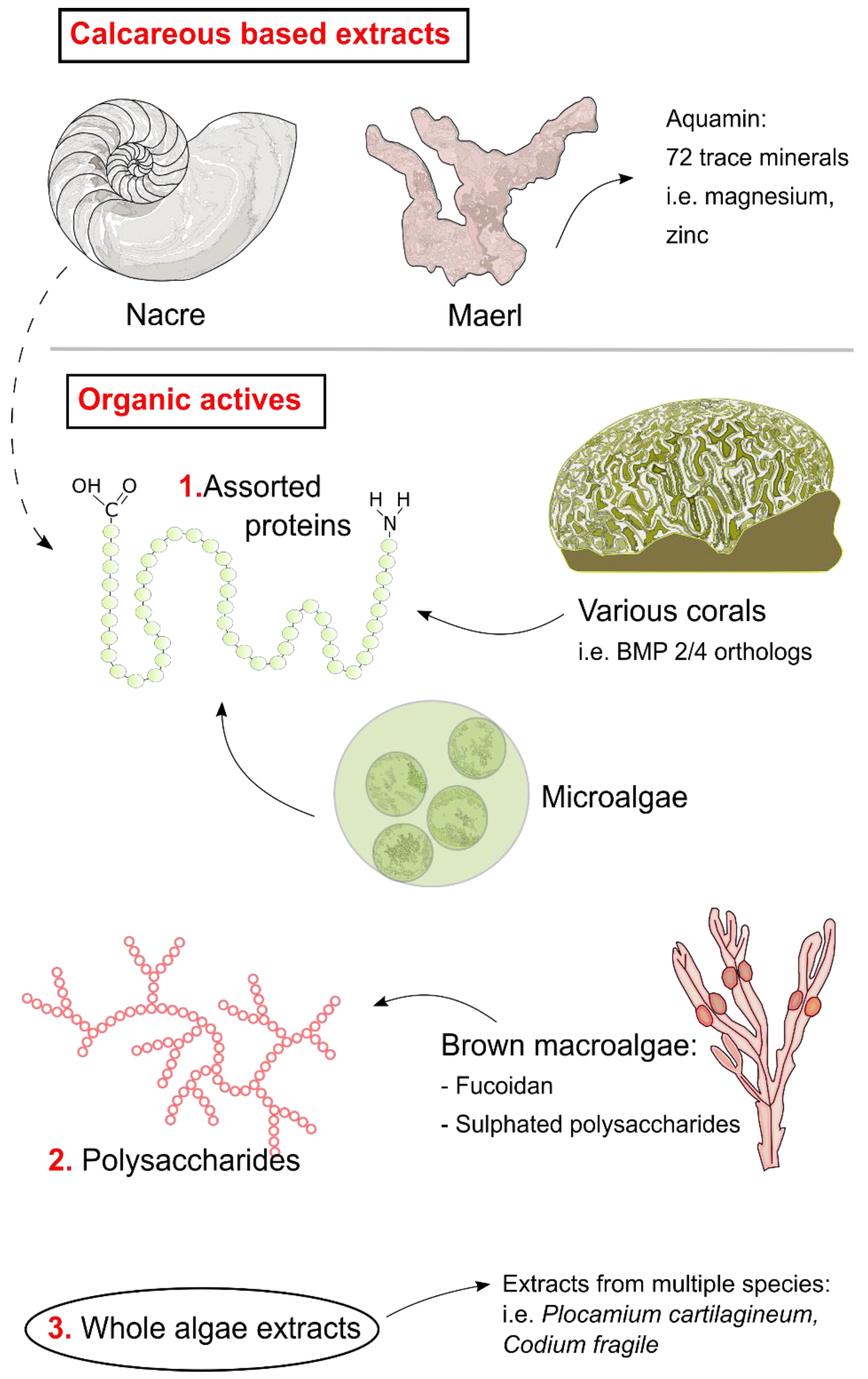
2. Mollusca
Nacre
3. Algae and Seaweed
3.1. Aquamin
3.2. Fucoidan
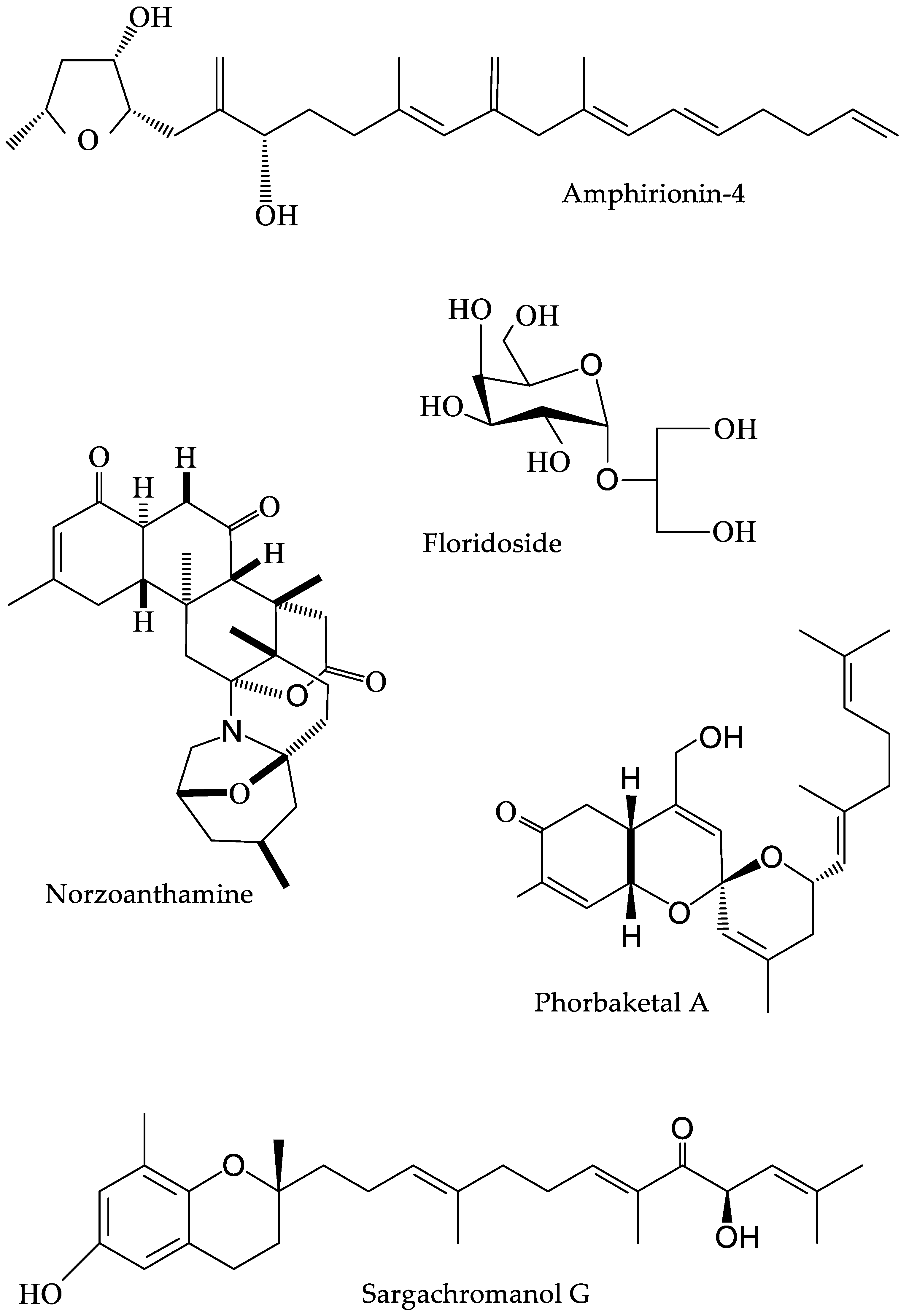
3.3. Other Algal Bioactives
4. Assorted Taxa
4.1. Microalgae
4.2. Corals
5. Challenges to Using Marine Bioactives
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Johnell, O.; Kanis, J.A. An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos. Int. 2006, 17, 1726–1733. [Google Scholar] [CrossRef] [PubMed]
- Kanis, J.A. Assessment of fracture risk and its application to screening for postmenopausal osteoporosis: Synopsis of a WHO report. Osteoporos. Int. 1994, 4, 368–381. [Google Scholar] [CrossRef] [PubMed]
- Kanis, J.A.; Johnell, O.; Oden, A.; Johansson, H.; McCloskey, E. FRAXTM and the assessment of fracture probability in men and women from the UK. Osteoporos. Int. 2008, 19, 385–397. [Google Scholar] [CrossRef] [PubMed]
- Riggs, B.L.; Khosla, S.; Melton, L.J. A unitary model for involutional osteoporosis: Estrogen deficiency causes both type I and type II osteoporosis in postmenopausal women and contributes to bone loss in aging men. J. Bone Miner. Res. 1998, 13, 763–773. [Google Scholar] [CrossRef] [PubMed]
- Lips, P.; Bouillon, R.; Van Schoor, N.M.; Vanderschueren, D.; Verschueren, S.; Kuchuk, N.; Milisen, K.; Boonen, S. Reducing fracture risk with calcium and vitamin D. Clin. Endocrinol. 2010, 73, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Hernlund, E.; Svedbom, A.; Ivergård, M.; Compston, J.; Cooper, C.; Stenmark, J.; McCloskey, E.V.; Jönsson, B.; Kanis, J.A. Osteoporosis in the European Union: Medical management, epidemiology and economic burden: A report prepared in collaboration with the International Osteoporosis Foundation (IOF) and the European Federation of Pharmaceutical Industry Associations (EFPIA). Arch. Osteoporos. 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Adler, R.A.; El-Hajj Fuleihan, G.; Bauer, D.C.; Camacho, P.M.; Clarke, B.L.; Clines, G.A.; Compston, J.E.; Drake, M.T.; Edwards, B.J.; Favus, M.J.; et al. Managing Osteoporosis in Patients on Long-Term Bisphosphonate Treatment: Report of a Task Force of the American Society for Bone and Mineral Research. J. Bone Miner. Res. 2016, 31, 16–35. [Google Scholar] [CrossRef] [PubMed]
- Bilezikian, J.P. Efficacy of Bisphosphonates in Reducing Fracture Risk in Postmenopausal Osteoporosis. Am. J. Med. 2009, 122, S14–S21. [Google Scholar] [CrossRef] [PubMed]
- Ensrud, K.E.; Barrett-Connor, E.L.; Schwartz, A.; Santora, A.C.; Bauer, D.C.; Suryawanshi, S.; Feldstein, A.; Haskell, W.L.; Hochberg, M.C.; Torner, J.C.; et al. Randomized Trial of Effect of Alendronate Continuation Versus Discontinuation in Women With Low BMD: Results From the Fracture Intervention Trial Long-Term Extension. J. Bone Miner. Res. 2004, 19, 1259–1269. [Google Scholar] [CrossRef] [PubMed]
- Black, D.M.; Rosen, C.J. Postmenopausal Osteoporosis. N. Engl. J. Med. 2016, 374, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Canalis, E.; Giustina, A.; Bilezikian, J.P. Mechanisms of anabolic therapies for osteoporosis. N. Engl. J. Med. 2007, 357, 905–916. [Google Scholar] [CrossRef] [PubMed]
- Charissoux, J.-L.; Vergnenegre, G.; Pelissier, M.; Fabre, T.; Mansat, P. Epidemiology of distal humerus fractures in the elderly. Orthop. Traumatol. Surg. Res. 2013, 99, 765–769. [Google Scholar] [CrossRef] [PubMed]
- Ringe, J.; Kaps, C.; Burmester, G.-R.; Sittinger, M. Stem cells for regenerative medicine: Advances in the engineering of tissues and organs. Naturwissenschaften 2002, 89, 338–351. [Google Scholar] [CrossRef] [PubMed]
- Clarke, S.A.; Walsh, P.; Maggs, C.A.; Buchanan, F. Designs from the deep: Marine organisms for bone tissue engineering. Biotechnol. Adv. 2011, 29, 610–617. [Google Scholar] [CrossRef] [PubMed]
- Granito, R.N.; Custódio, M.R.; Rennó, A.C.M. Natural marine sponges for bone tissue engineering: The state of art and future perspectives. J. Biomed. Mater. Res. Part B Appl. Biomater. 2017, 105, 1717–1727. [Google Scholar] [CrossRef] [PubMed]
- Lordan, S.; Ross, R.P.; Stanton, C. Marine bioactives as functional food ingredients: Potential to reduce the incidence of chronic diseases. Mar. Drugs 2011, 9, 1056–1100. [Google Scholar] [CrossRef] [PubMed]
- Smith, V.J.; Desbois, A.P.; Dyrynda, E.A. Conventional and Unconventional Antimicrobials from Fish, Marine Invertebrates and Micro-algae. Mar. Drugs 2010, 8, 1213–1262. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, R.S.; Morrissey, M.T. Marine Biotechnology for Production of Food Ingredients. Adv. Food Nutr. Res. 2007, 52, 237–292. [Google Scholar] [CrossRef] [PubMed]
- Jha, R.K.; Zi-rong, X. Biomedical Compounds from Marine organisms. Mar. Drugs 2004, 2, 123–146. [Google Scholar] [CrossRef]
- Senthilkumar, K.; Venkatesan, J.; Kim, S.-K. Marine derived natural products for osteoporosis. Biomed. Prev. Nutr. 2014, 4, 1–7. [Google Scholar] [CrossRef]
- Kim, B.-S.; Yang, S.-S.; You, H.-K.; Shin, H.-I.; Lee, J. Fucoidan-induced osteogenic differentiation promotes angiogenesis by inducing vascular endothelial growth factor secretion and accelerates bone repair. J. Tissue Eng. Regen. Med. 2018, 12, e1311–e1324. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.; Zhu, L.; Li, X.; Jia, J.; Zhang, Y.; Sun, X.; Ma, J.; Liu, Z.; Ma, X. Low-molecular weight fucoidan inhibits the differentiation of osteoclasts and reduces osteoporosis in ovariectomized rats. Mol. Med. Rep. 2017, 15, 890–898. [Google Scholar] [CrossRef] [PubMed]
- Kose, O.; Arabaci, T.; Yemenoglu, H.; Kara, A.; Ozkanlar, S.; Kayis, S.; Duymus, Z. Influences of Fucoxanthin on Alveolar Bone Resorption in Induced Periodontitis in Rat Molars. Mar. Drugs 2016, 14, 70. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M.; Matsumoto, T. Marine Algae Sargassum Horneri Bioactive Factor Stimulates Osteoblastogenesis and Suppresses Osteoclastogenesis in Vitro. OA Biotechnol. 2012, 1, 3. [Google Scholar] [CrossRef]
- Yoon, W.-J.; Kim, K.-N.; Heo, S.-J.; Han, S.-C.; Kim, J.; Ko, Y.-J.; Kang, H.-K.; Yoo, E.-S. Sargachromanol G inhibits osteoclastogenesis by suppressing the activation NF-κB and MAPKs in RANKL-induced RAW 264.7 cells. Biochem. Biophys. Res. Commun. 2013, 434, 892–897. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-A.; Karadeniz, F.; Ahn, B.-N.; Kwon, M.S.; Mun, O.-J.; Bae, M.J.; Seo, Y.; Kim, M.; Lee, S.-H.; Kim, Y.Y.; et al. Bioactive quinone derivatives from the marine brown alga Sargassum thunbergii induce anti-adipogenic and pro-osteoblastogenic activities. J. Sci. Food Agric. 2016, 96, 783–790. [Google Scholar] [CrossRef] [PubMed]
- Jeong, Y.T.; Baek, S.H.; Jeong, S.C.; Yoon, Y.D.; Kim, O.H.; Oh, B.C.; Jung, J.W.; Kim, J.H. Osteoprotective Effects of Polysaccharide-Enriched Hizikia fusiforme Processing Byproduct In Vitro and In Vivo Models. J. Med. Food 2016, 19, 805–814. [Google Scholar] [CrossRef] [PubMed]
- Surget, G.; Roberto, V.P.; Le Lann, K.; Mira, S.; Guérard, F.; Laizé, V.; Poupart, N.; Cancela, M.L.; Stiger-Pouvreau, V. Marine green macroalgae: A source of natural compounds with mineralogenic and antioxidant activities. J. Appl. Phycol. 2017, 29, 575–584. [Google Scholar] [CrossRef]
- Ryu, B.; Li, Y.-X.; Kang, K.-H.; Kim, S.-K.; Kim, D.G. Floridoside from Laurencia undulata promotes osteogenic differentiation in murine bone marrow mesenchymal cells. J. Funct. Foods 2015, 19, 505–511. [Google Scholar] [CrossRef]
- Gorman, D.M.O.; Carroll, C.O.; Carmody, R.J. Evidence that Marine-derived, Multi-mineral, Aquamin Inhibits the NF-k B Signaling Pathway in Vitro. Phyther. Res. 2011, 4, 630–632. [Google Scholar]
- Yonezawa, T.; Mase, N.; Sasaki, H.; Teruya, T.; Hasegawa, S.; Cha, B.-Y.; Yagasaki, K.; Suenaga, K.; Nagai, K.; Woo, J.-T. Biselyngbyaside, isolated from marine cyanobacteria, inhibits osteoclastogenesis and induces apoptosis in mature osteoclasts. J. Cell. Biochem. 2012, 113, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Kita, M.; Kondo, M.; Koyama, T.; Yamada, K.; Matsumoto, T.; Lee, K.H.; Woo, J.T.; Uemura, D. Symbioimine Exhibiting Inhibitory Effect of Osteoclast Differentiation, from the Symbiotic Marine Dinoflagellate Symbiodinium sp. J. Am. Chem. Soc. 2004, 126, 4794–4795. [Google Scholar] [CrossRef] [PubMed]
- Minamida, M.; Kumagai, K.; Ulanova, D.; Akakabe, M.; Konishi, Y.; Tominaga, A.; Tanaka, H.; Tsuda, M.; Fukushi, E.; Kawabata, J.; et al. Amphirionin-4 with Potent Proliferation-Promoting Activity on Bone Marrow Stromal Cells from a Marine Dinoflagellate Amphidinium Species. Org. Lett. 2014, 16, 4858–4861. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.H.T.; Qian, Z.-J.; Nguyen, V.-T.; Choi, I.-W.; Heo, S.-J.; Oh, C.H.; Kang, D.-H.; Kim, G.H.; Jung, W.-K. Tetrameric peptide purified from hydrolysates of biodiesel byproducts of Nannochloropsis oculata induces osteoblastic differentiation through MAPK and Smad pathway on MG-63 and D1 cells. Process Biochem. 2013, 48, 1387–1394. [Google Scholar] [CrossRef]
- Merceron, C.; Portron, S.; Vignes-Colombeix, C.; Rederstorff, E.; Masson, M.; Lesoeur, J.; Sourice, S.; Sinquin, C.; Colliec-Jouault, S.; Weiss, P.; et al. Pharmacological modulation of human mesenchymal stem cell chondrogenesis by a chemically oversulfated polysaccharide of marine origin: Potential application to cartilage regenerative medicine. Stem Cells 2012, 30, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kwak, H.B.; Pi, S.; You, H.; Byeon, S.R.; Ying, Y.; Luesch, H.; Hong, J.; Kim, S.H. In Vitro and In Vivo Osteogenic Activity of Largazole. Am. Chem. Soc. Med. Chem. Lett. 2011, 2, 248–251. [Google Scholar] [CrossRef] [PubMed]
- Byun, M.R.; Kim, A.R.; Hwang, J.-H.; Sung, M.K.; Lee, Y.K.; Hwang, B.S.; Rho, J.-R.; Hwang, E.S.; Hong, J.-H. Phorbaketal A stimulates osteoblast differentiation through TAZ mediated Runx2 activation. FEBS Lett. 2012, 586, 1086–1092. [Google Scholar] [CrossRef] [PubMed]
- Kinugawa, M.; Fukuzawa, S.; Tachibana, K. Skeletal protein protection: The mode of action of an anti-osteoporotic marine alkaloid, norzoanthamine. J. Bone Miner. Metab. 2009, 27, 303–314. [Google Scholar] [CrossRef] [PubMed]
- Abramovitch-gottlib, L.; Sc, M.; Geresh, S.; Vago, R.; Ph, D. Biofabricated Marine Hydrozoan: A Bioactive Crystalline Material Promoting Ossification of Mesenchymal Stem Cells. Tissue Eng. 2006, 12, 729–739. [Google Scholar] [CrossRef] [PubMed]
- DiCarlo, B.B.; Hu, J.C.; Gross, T.; Vago, R.; Athanasiou, K.A. Biomaterial effects in articular cartilage tissue engineering using polyglycolic acid, a novel marine origin biomaterial, IGF-I, and TGF-β1. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2009, 223, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A.; Isa, Y.; Uehara, T. Proteins of calcified endoskeleton: II partial amino acid sequences of endoskeletal proteins and the characterization of proteinaceous organic matrix of spicules from the alcyonarian, Synularia polydactyla. Proteomics 2005, 5, 885–893. [Google Scholar] [CrossRef] [PubMed]
- Helman, Y.; Natale, F.; Sherrell, R.M.; Lavigne, M.; Starovoytov, V.; Gorbunov, M.Y.; Falkowski, P.G. Extracellular matrix production and calcium carbonate precipitation by coral cells in vitro. Proc. Natl. Acad. Sci. USA 2008, 105, 54–58. [Google Scholar] [CrossRef] [PubMed]
- Kariya, Y.; Mulloy, B.; Imai, K.; Tominaga, A.; Kaneko, T.; Asari, A.; Suzuki, K.; Masuda, H.; Kyogashima, M.; Ishii, T. Isolation and partial characterization of fucan sulfates from the body wall of sea cucumber Stichopus japonicus and their ability to inhibit osteoclastogenesis. Carbohydr. Res. 2004, 339, 1339–1346. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.H.T.; Qian, Z.-J.; Jung, W.-K. Beneficial Effect of Abalone Intestine Gastro-Intestinal Digests on Osteoblastic MG-63 Cell Differentiation. J. Aquat. Food Prod. Technol. 2014, 23, 436–446. [Google Scholar] [CrossRef]
- Weiss, I.M.; Göhring, W.; Fritz, M.; Mann, K. Perlustrin, a Haliotis laevigata (abalone) nacre protein, is homologous to the insulin-like growth factor binding protein N-terminal module of vertebrates. Biochem. Biophys. Res. Commun. 2001, 285, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.M.; Kim, B.J.; Shim, J.-H.; Kang, K.S.; Kim, K.-J.; Rhie, J.W.; Cha, H.J.; Cho, D.-W. Enhancement of bone regeneration through facile surface functionalization of solid freeform fabrication-based three-dimensional scaffolds using mussel adhesive proteins. Acta Biomater. 2012, 8, 2578–2586. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, D.V.; Silva, T.S.; Cordeiro, O.D.; Cavaco, S.I.; Simes, D.C. Identification of proteins with potential osteogenic activity present in the water-soluble matrix proteins from Crassostrea gigas nacre using a proteomic approach. Sci. World J. 2012, 2012, 765909. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.J.; Milet, C.; Peduzzi, J.; Pereira, L.; Haigle, J.; Barthelemy, M.; Lopez, E. Effect of water-soluble matrix fraction extracted from the nacre of Pinctada maxima on the alkaline phosphatase activity of cultured fibroblasts. J. Exp. Zool. 2000, 288, 327–334. [Google Scholar] [CrossRef]
- Kim, H.; Lee, K.; Ko, C.-Y.; Kim, H.-S.; Shin, H.-I.; Kim, T.; Lee, S.H.; Jeong, D. The role of nacreous factors in preventing osteoporotic bone loss through both osteoblast activation and osteoclast inactivation. Biomaterials 2012, 33, 7489–7496. [Google Scholar] [CrossRef] [PubMed]
- Bédouet, L.; Marie, A.; Dubost, L.; Péduzzi, J.; Duplat, D.; Berland, S.; Puisségur, M.; Boulzaguet, H.; Rousseau, M.; Milet, C.; et al. Proteomics analysis of the nacre soluble and insoluble proteins from the oyster Pinctada margaritifera. Mar. Biotechnol. 2007, 9, 638–649. [Google Scholar] [CrossRef] [PubMed]
- Lao, Y.; Zhang, X.; Zhou, J.; Su, W.; Chen, R.; Wang, Y.; Zhou, W.; Xu, Z.-F. Characterization and in vitro mineralization function of a soluble protein complex P60 from the nacre of Pinctada fucata. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2007, 148, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Benkendorff, K. Molluscan biological and chemical diversity: Secondary metabolites and medicinal resources produced by marine molluscs. Biol. Rev. 2010, 85. [Google Scholar] [CrossRef] [PubMed]
- Grienke, U.; Silke, J.; Tasdemir, D. Bioactive compounds from marine mussels and their effects on human health. Food Chem. 2014, 142, 48–60. [Google Scholar] [CrossRef] [PubMed]
- Marie, B.; Marin, F.; Marie, A.; Bédouet, L.; Dubost, L.; Alcaraz, G.; Milet, C.; Luquet, G. Evolution of nacre: Biochemistry and proteomics of the shell organic matrix of the cephalopod Nautilus macromphalus. Chembiochem 2009, 10, 1495–1506. [Google Scholar] [CrossRef] [PubMed]
- Silve, C.; Lopez, E.; Vidal, B.; Smith, D.C.; Camprasse, S.; Camprasse, G.; Couly, G. Nacre Initiâtes Biomineralization by Human Osteoblasts Maintained in Vitro. Calcif. Tissue Int. 1992, 51, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Atlan, G.; Balmain, N.; Berland, S.; Vidal, B.; Lopez, E. Reconstruction of human maxillary defects with nacre powder: Histological evidence for bone regeneration. C. R. Acad. Sci. III 1997, 320, 253–258. [Google Scholar] [CrossRef]
- Westbroek, P.; Marin, F. A marriage of bone and nacre. Nature 1998, 392, 861–862. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, R.; Singha, P.K.; Dey, S. Water soluble bioactives of nacre mediate antioxidant activity and osteoblast differentiation. PLoS ONE 2013, 8, e84584. [Google Scholar] [CrossRef] [PubMed]
- Liao, H.; Mutvei, H.; Hammarström, L.; Wurtz, T.; Li, J. Tissue responses to nacreous implants in rat femur: An in situ hybridization and histochemical study. Biomaterials 2002, 23, 2693–2701. [Google Scholar] [CrossRef]
- Zhang, C.; Li, S.; Ma, Z.; Xie, L.; Zhang, R. A novel matrix protein p10 from the nacre of pearl oyster (Pinctada fucata) and its effects on both CaCO3 crystal formation and mineralogenic cells. Mar. Biotechnol. 2006, 8, 624–633. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liu, S.; Xie, L.; Zhang, R.; Wang, Z. Pinctada fucata mantle gene 3 (PFMG3) promotes differentiation in mouse osteoblasts (MC3T3-E1). Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2011, 158, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Bédouet, L.; Duplat, D.; Marie, A.; Dubost, L.; Berland, S.; Rousseau, M.; Milet, C.; Lopez, E. Heterogeneity of proteinase inhibitors in the water-soluble organic matrix from the oyster nacre. Mar. Biotechnol. 2007, 9, 437–449. [Google Scholar] [CrossRef] [PubMed]
- Rousseau, M.; Boulzaguet, H.; Biagianti, J.; Duplat, D.; Milet, C.; Lopez, E.; Bédouet, L. Low molecular weight molecules of oyster nacre induce mineralization of the MC3T3-E1 cells. J. Biomed. Mater. Res. A 2008, 85, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.; Blake, C.; Berges, J.A.; Maggs, C.A. Environmental tolerances of free-living coralline algae (maerl): Implications for European marine conservation. Biol. Conserv. 2004, 120, 279–289. [Google Scholar] [CrossRef]
- Green, D.W.; Padula, M.P.; Santos, J.; Chou, J.; Milthorpe, B.; Ben-Nissan, B. A therapeutic potential for marine skeletal proteins in bone regeneration. Mar. Drugs 2013, 11, 1203–1220. [Google Scholar] [CrossRef] [PubMed]
- Palacios, C. The role of nutrients in bone health, from A to Z. Crit. Rev. Food Sci. Nutr. 2006, 46, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Gorman, D.M.O.; Tierney, C.M.; Brennan, O.; Brien, F.J.O. The Marine-derived, Multi-mineral formula, Aquamin, Enhances Mineralisation of Osteoblast Cells In Vitro. Phyther. Res. 2012, 380, 375–380. [Google Scholar]
- Widaa, A.; Brennan, O.; Gorman, D.M.O.; Brien, F.J.O. The Osteogenic Potential of the Marine-Derived Multi-Mineral Formula Aquamin Is Enhanced by the Presence of Vitamin D. Phyther. Res. 2014, 684, 678–684. [Google Scholar] [CrossRef] [PubMed]
- Aslam, M.N.; Kreider, J.M.; Paruchuri, T.; Bhagavathula, N.; DaSilva, M.; Zernicke, R.F.; Goldstein, S.A.; Varani, J. A mineral-rich extract from the red marine algae Lithothamnion calcareum preserves bone structure and function in female mice on a Western-style diet. Calcif. Tissue Int. 2010, 86, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Brennan, O.; Stenson, B.; Widaa, A.; O’Gorman, D.M.; O’Brien, F.J. Incorporation of the natural marine multi-mineral dietary supplement Aquamin enhances osteogenesis and improves the mechanical properties of a collagen-based bone graft substitute. J. Mech. Behav. Biomed. Mater. 2015, 47, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, B.D.; Cate, R.E.; O’Connor-Robison, C.I. A Marine Mineral Supplement Alters Markers of Bone Metabolism in Yearling Arabians. J. Equine Vet. Sci. 2010, 30, 419–424. [Google Scholar] [CrossRef]
- Frestedt, J.L.; Kuskowski, M.A.; Zenk, J.L. A natural seaweed derived mineral supplement (Aquamin F) for knee osteoarthritis: A randomised, placebo controlled pilot study. Nutr. J. 2009, 8, 7. [Google Scholar] [CrossRef] [PubMed]
- Frestedt, J.L.; Walsh, M.; Kuskowski, M.A.; Zenk, J.L. A natural mineral supplement provides relief from knee osteoarthritis symptoms: A randomized controlled pilot trial. Nutr. J. 2008, 7, 9. [Google Scholar] [CrossRef] [PubMed]
- Zenk, J.L.; Frestedt, J.L.; Kuskowski, M.A. Effect of Calcium Derived from Lithothamnion sp. on Markers of Calcium Metabolism in Premenopausal Women. J. Med. Food 2018, 21, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Fitton, J.H. Therapies from fucoidan; multifunctional marine polymers. Mar. Drugs 2011, 9, 1731–1760. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.W.; Baek, S.-H.; Lee, S.-H.; Kim, T.-H.; Kim, S.-Y. Fucoidan, a Sulfated Polysaccharide, Inhibits Osteoclast Differentiation and Function by Modulating RANKL Signaling. Int. J. Mol. Sci. 2014, 15, 18840–18855. [Google Scholar] [CrossRef] [PubMed]
- Changotade, S.I.T.; Korb, G.; Bassil, J.; Barroukh, B.; Willig, C.; Colliec-Jouault, S.; Durand, P.; Godeau, G.; Senni, K. Potential effects of a low-molecular-weight fucoidan extracted from brown algae on bone biomaterial osteoconductive properties. J. Biomed. Mater. Res. Part A 2008, 87, 666–675. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.-D.; Yoon, W.-J.; Woo, K.-M.; Baek, J.-H.; Lee, G.; Cho, J.-Y.; Ryoo, H.-M. Molecular regulation of matrix extracellular phosphoglycoprotein expression by bone morphogenetic protein-2. J. Biol. Chem. 2009, 284, 25230–25240. [Google Scholar] [CrossRef] [PubMed]
- Pereira, J.; Portron, S.; Dizier, B.; Vinatier, C.; Masson, M.; Sourice, S.; Galy-Fauroux, I.; Corre, P.; Weiss, P.; Fischer, A.-M.; et al. The in vitro and in vivo effects of a low-molecular-weight fucoidan on the osteogenic capacity of human adipose-derived stromal cells. Tissue Eng. Part A 2014, 20, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Schmidt, H.; Pavleska, D.; Wermann, T.; Seekamp, A.; Fuchs, S. Crude Fucoidan Extracts Impair Angiogenesis in Models Relevant for Bone Regeneration and Osteosarcoma via Reduction of VEGF and SDF-1. Mar. Drugs 2017, 15, 186. [Google Scholar] [CrossRef] [PubMed]
- Hwang, P.-A.; Hung, Y.-L.; Phan, N.N.; Hieu, B.-T.-N.; Chang, P.-M.; Li, K.-L.; Lin, Y.-C. The in vitro and in vivo effects of the low molecular weight fucoidan on the bone osteogenic differentiation properties. Cytotechnology 2016, 68, 1349–1359. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.S.; Kang, H.-J.; Park, J.-Y.; Lee, J. Fucoidan promotes osteoblast differentiation via JNK- and ERK-dependent BMP2-Smad 1/5/8 signaling in human mesenchymal stem cells. Exp. Mol. Med. 2015, 47, e128. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, S.; Hashizume, M.; Hokari, Y.; Nakagawa, T.; Igarashi, A.; Yamaguchi, M. Characterization of Active Component in Marine Alga Sargassum horneri Extract in Stimulating Bone Calcification in Vitro. J. Health Sci. 2004, 50, 634–639. [Google Scholar] [CrossRef]
- Uchiyama, S.; Yamaguchi, M. Anabolic Effect of Marine Alga Sargassum Horneri Extract on Bone Components in the Femoral-diaphyseal and -metaphyseal Tissues of Young and Aged Rats in Vivo. J. Health Sci. 2002, 48, 325–330. [Google Scholar] [CrossRef]
- Uchiyama, S.; Yamaguchi, M. Preventive Effect of Marine Alga Sargassum Horneri Extract on Bone Loss in Streptozotocin-Diabetic Rats in Vivo. J. Health Sci. 2003, 49, 149–155. [Google Scholar] [CrossRef]
- Matsumoto, T.; Hokari, Y.; Hashizume, M.; Yamaguchi, M. Effect of Sargassum horneri Extract on Circulating Bone Metabolic Markers: Supplemental Intake Has an Effect in Healthy Humans. J. Health Sci. 2008, 54, 50–55. [Google Scholar] [CrossRef]
- Das, S.K.; Ren, R.; Hashimoto, T.; Kanazawa, K. Fucoxanthin Induces Apoptosis in Osteoclast-like Cells Differentiated from RAW264.7 Cells. J. Agric. Food Chem. 2010, 58, 6090–6095. [Google Scholar] [CrossRef] [PubMed]
- Carson, M.A.; Nelson, J.; Cancela, M.L.; Laizé, V.; Gavaia, P.J.; Rae, M.; Heesch, S.; Verzin, E.; Maggs, C.; Gilmore, B.F.; et al. Red algal extracts from Plocamium lyngbyanum and Ceramium secundatum stimulate osteogenic activities in vitro and bone growth in zebrafish larvae. Sci. Rep. 2018, 8, 7725. [Google Scholar] [CrossRef] [PubMed]
- Tarasco, M.; Laizé, V.; Cardeira, J.; Cancela, M.L.; Gavaia, P.J. The zebrafish operculum: A powerful system to assess osteogenic bioactivities of molecules with pharmacological and toxicological relevance. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2017, 197, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, K.; Yada, M.; Tsuji, T.; Kuramoto, M.; Uemura, D. Suppressive Effect of Norzoanthamine Hydrochloride on Experimental Osteoporosis in Ovariectomized Mice. Biol. Pharm. Bull. 1999, 22, 920–924. [Google Scholar] [CrossRef] [PubMed]
- Akakabe, M.; Kumagai, K.; Tsuda, M.; Konishi, Y.; Tominaga, A.; Tsuda, M.; Fukushi, E.; Kawabata, J. Amphirionin-5, a novel linear polyketide from a cultured marine dinoflagellate Amphidinium species with a potent cell proliferation-promoting activity. Tetrahedron Lett. 2014, 55, 3491–3494. [Google Scholar] [CrossRef]
- Lee, K.; Silva, E.A.; Mooney, D.J. Growth factor delivery-based tissue engineering: General approaches and a review of recent developments. J. R. Soc. Interface 2011, 8, 153–170. [Google Scholar] [CrossRef] [PubMed]
- Rogers, A.D. The biology of Lophelia pertusa (Linnaeus 1758) and other deep-water reef-forming corals and impacts from human activities. Int. Rev. Hydrobiol. 1999, 84, 315–406. [Google Scholar] [CrossRef]
- Goffredo, S.; Vergni, P.; Reggi, M.; Caroselli, E.; Sparla, F.; Levy, O.; Dubinsky, Z.; Falini, G. The skeletal organic matrix from Mediterranean coral Balanophyllia europaea influences calcium carbonate precipitation. PLoS ONE 2011, 6, e22338. [Google Scholar] [CrossRef] [PubMed]
- Zoccola, D.; Moya, A.; Béranger, G.E.; Tambutté, E.; Allemand, D.; Carle, G.F.; Tambutté, S. Specific expression of BMP2/4 ortholog in biomineralizing tissues of corals and action on mouse BMP receptor. Mar. Biotechnol. 2009, 11, 260. [Google Scholar] [CrossRef] [PubMed]
- Kramarsky-Winter, E.; Loya, Y. Tissue regeneration in the coral Fungia granulosa: The effect of extrinsic and intrinsic factors. Mar. Biol. 2000, 137, 867–873. [Google Scholar] [CrossRef]
- Hughes, T.P.; Graham, N.A.J.; Jackson, J.B.C.; Mumby, P.J.; Steneck, R.S. Rising to the challenge of sustaining coral reef resilience. Trends Ecol. Evol. 2010, 25, 633–642. [Google Scholar] [CrossRef] [PubMed]
- Harriott, V.J. Can corals be harvested sustainably? Ambio 2003, 32, 130–133. [Google Scholar] [CrossRef] [PubMed]
- Proksch, P.; Edrada-Ebel, R.; Ebel, R. Drugs from the Sea—Opportunities and Obstacles. Mar. Drugs 2003, 1, 5–17. [Google Scholar] [CrossRef]
- Singh, A.; Nigam, P.S.; Murphy, J.D. Mechanism and challenges in commercialisation of algal biofuels. Bioresour. Technol. 2011, 102, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Saltonstall, K. Cryptic invasion by a non-native genotype of the common reed, Phragmites australis, into North America. Proc. Natl. Acad. Sci. USA 2002, 99, 2445–2449. [Google Scholar] [CrossRef] [PubMed]
- Christensen, K.; Doblhammer, G.; Rau, R.; Vaupel, J.W. Ageing populations: The challenges ahead. Lancet 2009, 374, 1196–1208. [Google Scholar] [CrossRef]
| Genus and Species | General Description | Extract/Bioactive | Reference Example |
|---|---|---|---|
| Numerous assorted species | Brown algae | Fucoidan | [21] |
| Numerous assorted species | Brown algae | Fucoidan (low molecular weight) | [22] |
| Numerous assorted species | Brown seaweed | Fucoxanthin | [23] |
| Sargassum horneri | Brown algae | Raw extract | [24] |
| Sargassum siliquastrum | Brown algae | Sargachromanol G | [25] |
| Sargassum thunbergii | Brown algae | Quinone derivatives | [26] |
| Hizikia fusiforme | Brown algae | Water by-product | [27] |
| Cladophora rupestris | Green algae | Crude extract | [28] |
| Codium fragile | Green algae | Crude extract | [28] |
| Laurencia undulata | Red algae | Floridoside | [29] |
| Lithothamnion corallioides | Calcareous red algae | Aquamin | [30] |
| Lyngbya sp. | Cyanobacteria | Macrolide | [31] |
| Symbiodinium sp. | Dinoflagellate | Symbioimine | [32] |
| Amphidinium sp. | Dinoflagellate | Polyketide | [33] |
| Nannochloropsis oculata | Microalgae | Peptide | [34] |
| Alteromonas infernus | Prokaryote | Polysaccharide | [35] |
| Symploca sp. | Cyanobacterium | Largazole (depsipeptide) | [36] |
| Phorbas sp. | Sponge | Phorbaketal A | [37] |
| Zoanthus sp. | Zoanthid | Norzoanthamine | [38] |
| Millepora dichotoma | Hydrocoral | Bioactive material | [39] |
| Porites lutea | Stony coral | Bioactive material | [40] |
| Porites lutea | Stony coral | Biomatrix | [40] |
| Synularia polydactyla | Alcyonarian coral | Proteins | [41] |
| Xenia elongate | Soft coral | Coral cells | [42] |
| Montipora digitata | Hard coral | Coral cells | [42] |
| Apostichopus japonicus | Sea cucumber | Fucan sulphate | [43] |
| Haliotis discus hannai | Abalone | Digested intestines | [44] |
| Haliotis laevigata | Abalone | Perlucin protein | [45] |
| Numerous assorted species | Mussels | Adhesive protein | [46] |
| Crassostrea gigas | Oyster | Protein Nacre (water soluble matrix) | [47] |
| Pinctada maxima | Pearl oyster | Individual proteins Low molecular weight molecules. | [48] |
| Pteria martensii | Pearl oyster | Nacre (water soluble matrix) | [49] |
| Pinctada margaritifera | Oyster | Proteinase inhibitor Proteins Nacre (water soluble matrix) | [50] |
| Pinctada fucata | Akoya pearl oyster | Pinctada fucata mantle gene 3 Protein p10 and other novel proteins | [51] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carson, M.A.; Clarke, S.A. Bioactive Compounds from Marine Organisms: Potential for Bone Growth and Healing. Mar. Drugs 2018, 16, 340. https://doi.org/10.3390/md16090340
Carson MA, Clarke SA. Bioactive Compounds from Marine Organisms: Potential for Bone Growth and Healing. Marine Drugs. 2018; 16(9):340. https://doi.org/10.3390/md16090340
Chicago/Turabian StyleCarson, Matthew A., and Susan A. Clarke. 2018. "Bioactive Compounds from Marine Organisms: Potential for Bone Growth and Healing" Marine Drugs 16, no. 9: 340. https://doi.org/10.3390/md16090340
APA StyleCarson, M. A., & Clarke, S. A. (2018). Bioactive Compounds from Marine Organisms: Potential for Bone Growth and Healing. Marine Drugs, 16(9), 340. https://doi.org/10.3390/md16090340





