Ladder-Shaped Ion Channel Ligands: Current State of Knowledge
Abstract
1. Introduction
- (i)
- current taxonomy of these harmful algae;
- (ii)
- geographic distribution of the species under consideration in the new areas over the world;
- (iii)
- current state of knowledge of the spectrum of toxic compounds, produced by lineage Gambierdiscus and genus Karenia;
- (iv)
- toxins’ effects and their physiological mechanisms, especially interactions with ion channels;
- (v)
- intracellular functions of these substances in the marine algae Gambierdiscus and Karenia, which have been poorly studied until now.
2. Actual State of Taxonomy
3. New Data on the Geographic Distribution of Gambierdiscus Lineage and Genus Karenia
4. Common Problems of the Compounds, Produced by Gambierdiscus and Karenia
5. Compounds, Produced by Gambierdiscus and Their Physiological Effects
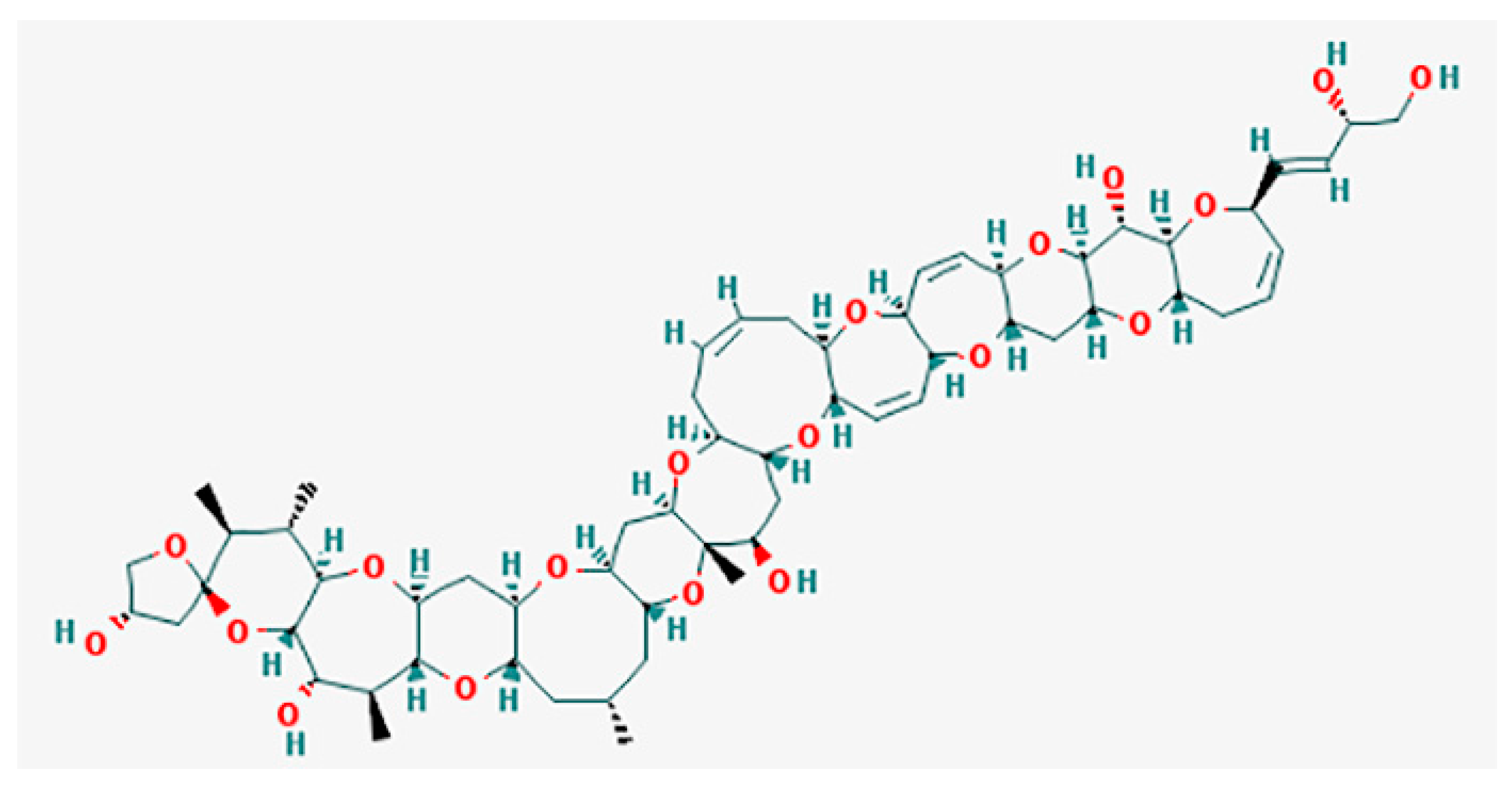
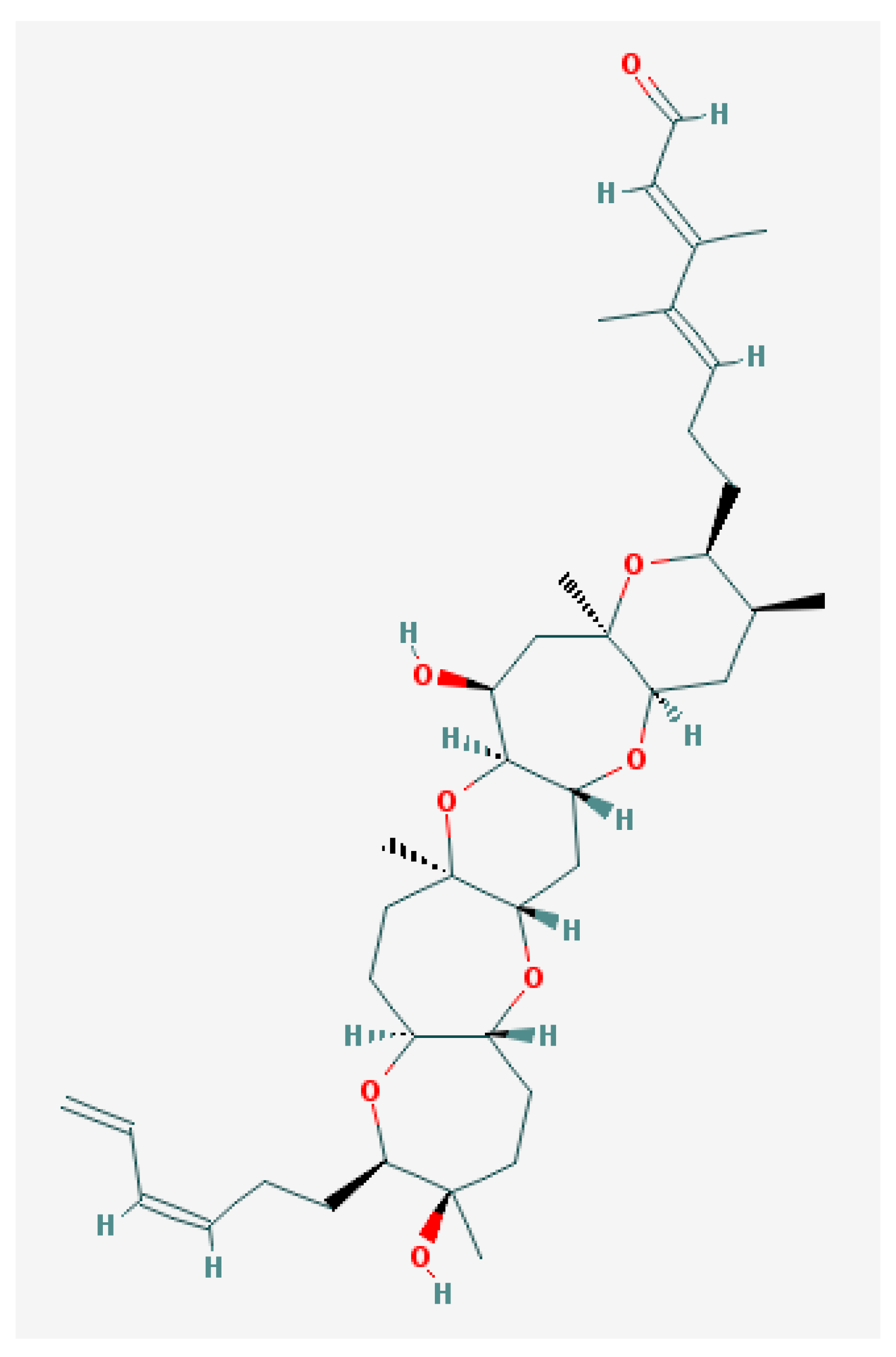
5.1. Ciguatoxins (CTXs)
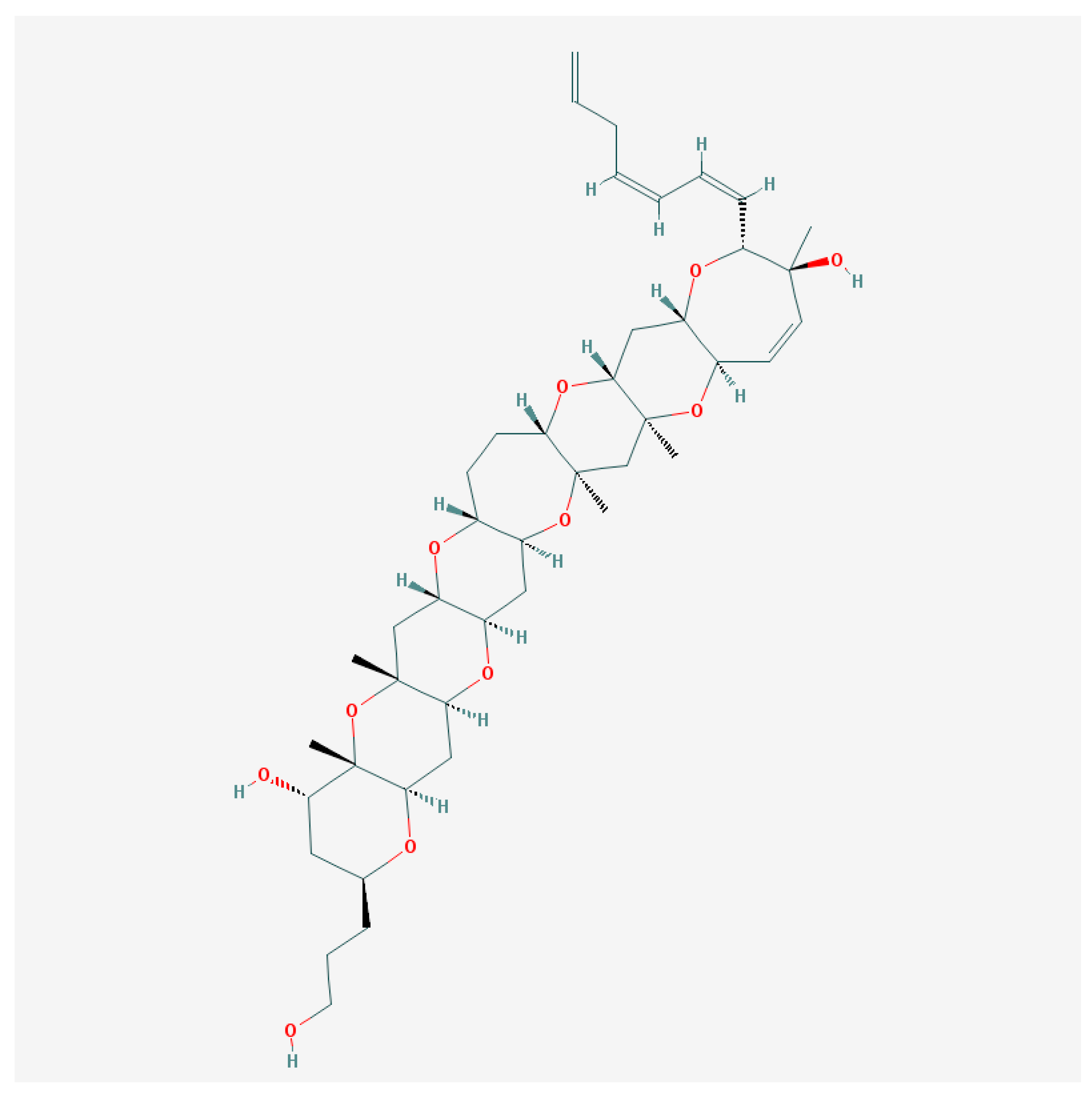
5.2. Gambierol
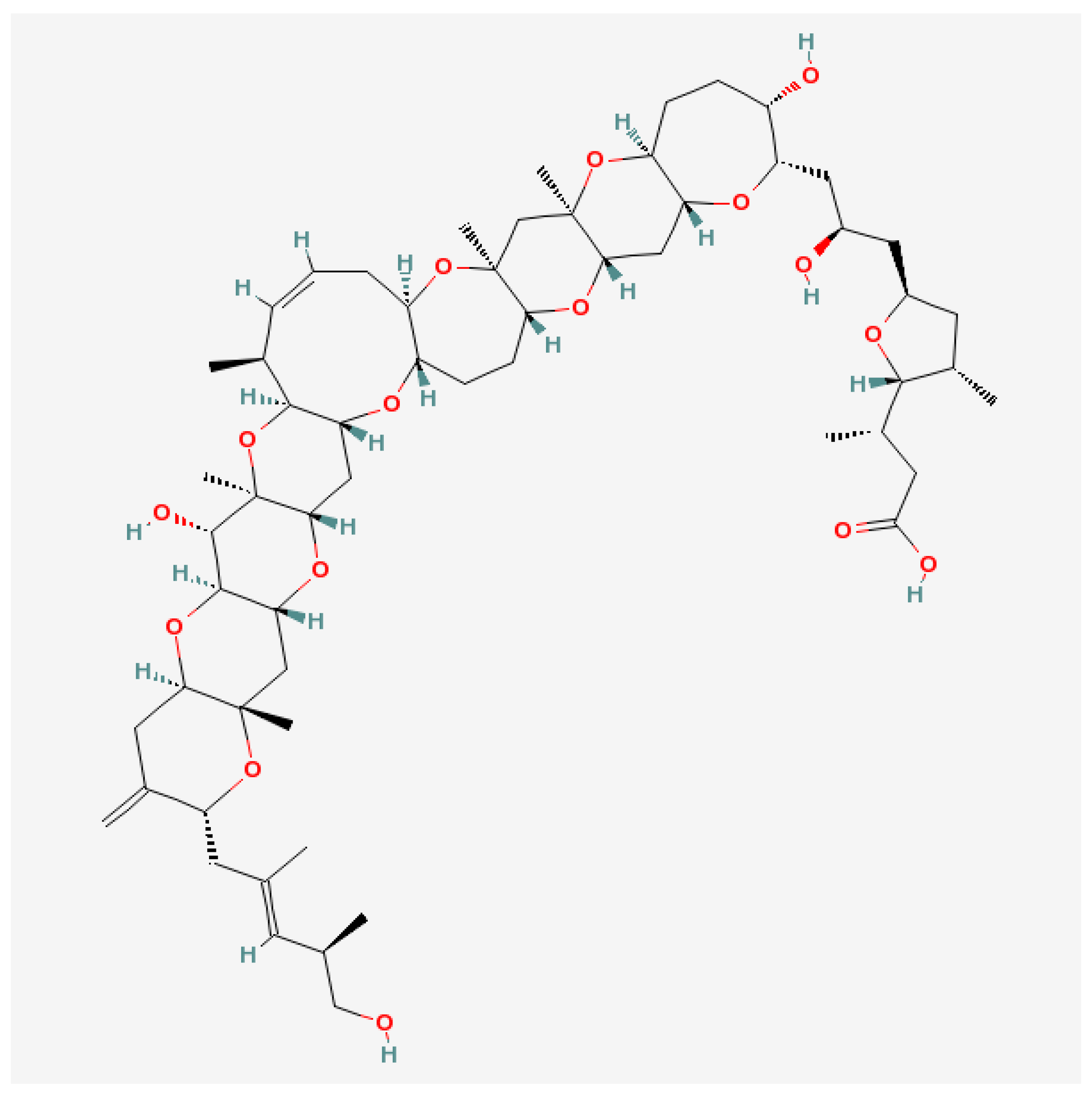
5.3. Maitotoxins
6. Compounds, Produced by Karenia and Their Physiological Effects
6.1. Brevetoxins and Others
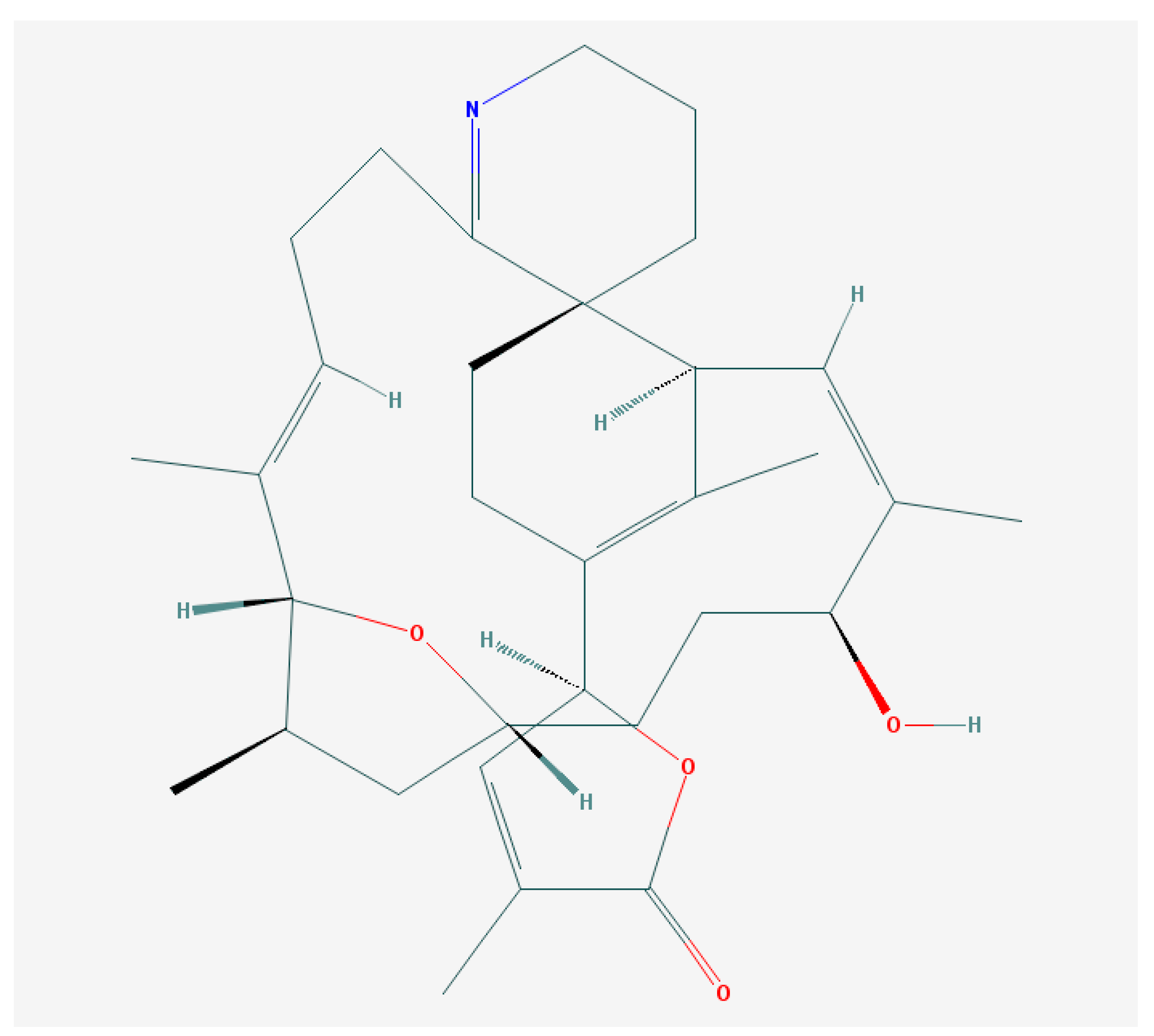
6.2. Brevenal—Dissident Ladder-Shape Compound
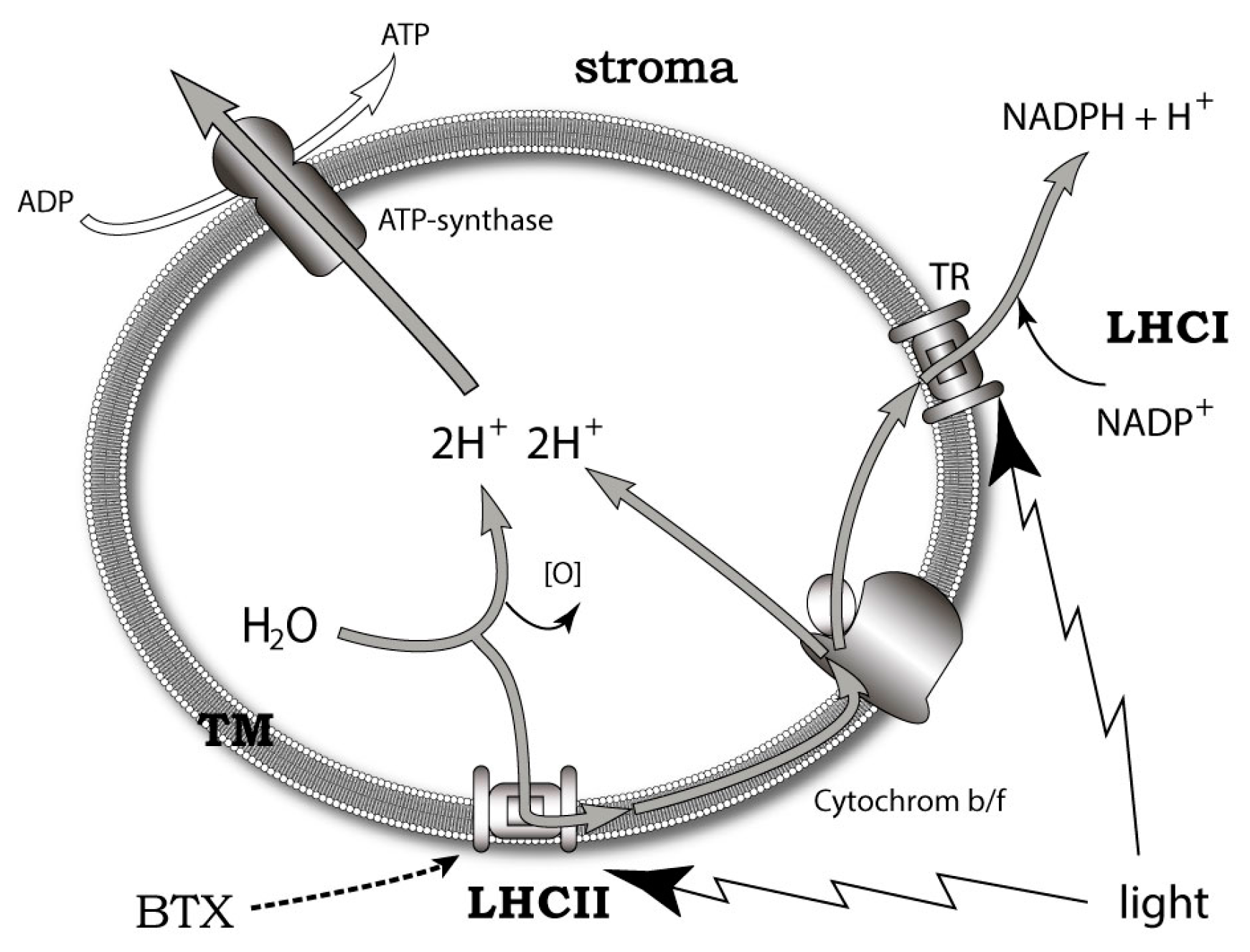
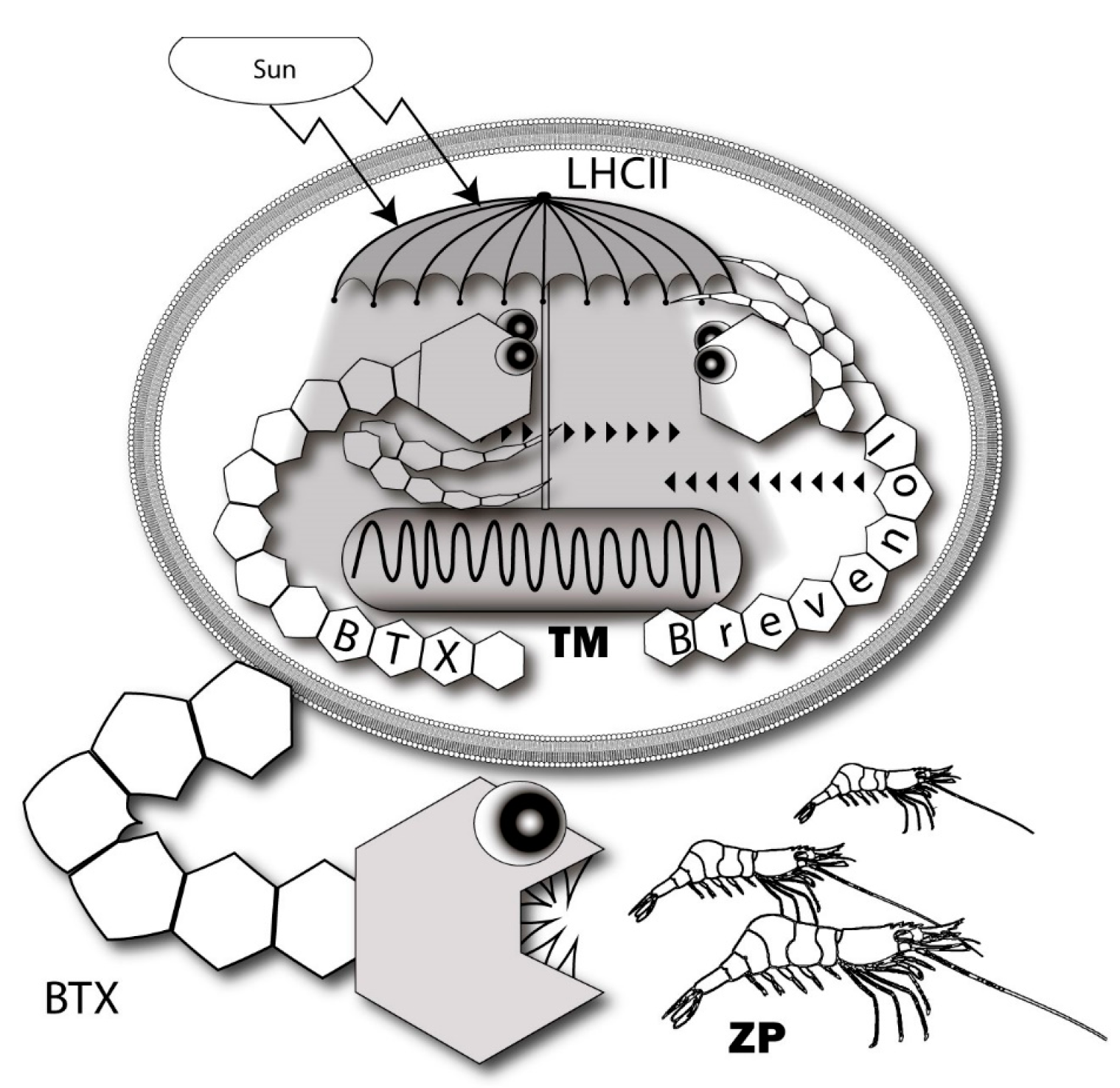
7. Possible Intrinsic Functions of Ladder-Shaped Toxins in Microalgae
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kohli, G.S.; Farrell, H.; Murray, S.A. Gambierdiscus, the cause of ciguatera fish poisoning: An increased human health threat influenced by climate change. In Climate Change and Marine and Freshwater Toxins; Botana, L.M., Louzao, M.C., Vilariño, N., Eds.; De Gruyter: Berlin, Germany, 2015; pp. 273–312. ISBN 9783110382617. [Google Scholar]
- Botana, L.M. Toxicological Perspective on Climate Change: Aquatic Toxins. Chem. Res. Toxicol. 2016, 29, 619–625. [Google Scholar] [CrossRef] [PubMed]
- Dickey, R.W.; Plakas, S.M. Ciguatera: A public health perspective. Toxicon 2010, 56, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Poletti, R.; Milandri, A.; Pompei, M. Algal biotoxins of marine origin: New indications from the European Union. Vet. Res. Commun. 2003, 27, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Maucher, J.M.; Briggs, L.R.; Podmore, C.; Ramsdell, J.S. Optimisation of blood collection card method/ELISA for monitoring exposure of bottlenose dolphin to brevetoxin-producing red tides. Environ. Sci. Technol. 2007, 41, 563–567. [Google Scholar] [CrossRef] [PubMed]
- Gebhard, E.; Levin, M.; Bogomolni, A.; De Guise, S. Immunomodulatory effects of brevetoxin (PbTx-3) upon in vitro exposure in bottlenose dolphins (Tursiops truncatus). Harmful Algae 2015, 44, 54–62. [Google Scholar] [CrossRef]
- Lombet, A.; Bidard, J.-N.; Lazdunski, M. Ciguatoxin and brevetoxins share a common receptor site on the neuronal voltage-dependent Na+ channel. FEBS Lett. 1987, 219, 355–359. [Google Scholar] [CrossRef]
- Nicholson, G.M.; Lewis, R.J. Ciguatoxins: Cyclic Polyether Modulators of Voltage-gated Ion Channel Function. Mar. Drugs 2006, 4, 82–118. [Google Scholar] [CrossRef]
- Arias, H.R. Marine toxins targeting ion channels. Mar. Drugs 2006, 4, 37–69. [Google Scholar] [CrossRef]
- Vetter, I.; Touska, F.; Hess, A.; Hinsbey, R.; Sattler, S.; Lampert, A.; Sergejeva, M.; Sharov, A.; Collins, L.S.; Eberhardt, M.; et al. Ciguatoxins activate specific cold pain pathways to elicit burning pain from cooling. EMBO J. 2012, 31, 3795–3808. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, K.; Deuis, J.R.; Inserra, M.; Collins, L.S.; Namer, B.; Cabot, P.J.; Reeh, P.W.; Lewis, R.J.; Vetter, I. Analgesic treatment of ciguatoxin-induced cold allodynia. Pain 2013, 154, 1999–2006. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.J. The changing face of ciguatera. Toxicon 2001, 39, 97–106. [Google Scholar] [CrossRef]
- Inoue, M.; Miyazaki, K.; Uehara, H.; Maruyama, M.; Hirama, M. First- and second-generation total synthesis of ciguatoxin CTX3C. Proc. Natl. Acad. Sci. USA 2004, 101, 12013–12018. [Google Scholar] [CrossRef] [PubMed]
- Voets, T. TRP channel blamed for burning cold after a tropical fish meal. EMBO J. 2012, 31, 3785–3787. [Google Scholar] [CrossRef] [PubMed]
- Parsons, M.L.; Aligizaki, K.; Bottein Dechraoui, M.-Y.; Fraga, S.; Morton, S.L.; Penna, A.; Rhodes, L. Gambierdiscus and Ostreopsis: Reassessment of the state of knowledge of their taxonomy, geography, ecophysiology, and toxicology. Harmful Algae 2012, 14, 107–129. [Google Scholar] [CrossRef]
- Mattei, C.; Vetter, I.; Eisenblätter, A.; Krock, B.; Ebbecke, M.; Desel, H.; Zimmermann, K. Ciguatera fish poisoning: A first epidemic in Germany highlights an increasing risk for European countries. Toxicon 2014, 91, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Litaker, R.W.; Vandersea, M.W.; Faust, M.A.; Kibler, S.R.; Chinain, M.; Holmes, M.J.; Holland, W.C.; Tester, P.A. Taxonomy of Gambierdiscus including four new species, Gambierdiscus caribaeus, Gambierdiscus carolinianus, Gambierdiscus carpenteri and Gambierdiscus ruetzleri (Gonyaulacales, Dinophyceae). Phycologia 2009, 45, 344–390. [Google Scholar] [CrossRef]
- Litaker, R.W.; Vandersea, M.W.; Faust, M.A.; Kibler, S.R.; Nau, A.W.; Holland, W.C.; Chinain, M.; Holmes, M.J.; Tester, P.A. Global distribution of ciguatera causing dinoflagellates in the genus Gambierdiscus. Toxicon 2010, 56, 711–730. [Google Scholar] [CrossRef] [PubMed]
- Kuno, S.; Kamikawa, R.; Yoshimatsu, S.; Sagara, T.; Nishio, S.; Sako, Y. Genetic diversity of Gambierdiscus spp. (Gonyaulacales, Dinophyceae) in Japanese coastal areas. Phycol. Res. 2010, 58, 44–52. [Google Scholar] [CrossRef]
- Jeong, H.J.; Lim, A.S.; Jang, S.H.; Yih, W.O.; Kang, N.S.; Lee, S.Y.; Yoon, Y.D.; Kim, H.S. First report of the epiphytic dinoflagellate Gambierdiscus caribaeus in the temperate waters off Jeju Island, Korea: Morphology and molecular characterization. J. Eukaryot. Microbiol. 2012, 59, 637–650. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, T.; Sato, S.; Tawong, W.; Sakanari, H.; Uehara, K.; Shah, M.M.R.; Suda, S.; Yasumoto, T.; Taira, Y.; Yamaguchi, H.; et al. Genetic diversity and distribution of the ciguatera-causing dinoflagellate Gambierdiscus spp. (Dinophyceae) in coastal areas of Japan. PLoS ONE 2013, 8, E60882. [Google Scholar] [CrossRef]
- Xu, Y.; Richlen, M.L.; Morton, S.L.; Mak, Y.L.; Chan, L.L.; Tekiau, A.; Anderson, D.M. Distribution, abundance and diversity of Gambierdiscus spp. from a ciguatera-endemic area in Marakei, Republic of Kiribati. Harmful Algae 2014, 34, 56–68. [Google Scholar] [CrossRef]
- Gomez, F.; Qiu, D.; Lopes, R.M.; Lin, S. Fukuyoa paulensis gen. et sp. nov., a new genus for the globular species of the dinoflagellate Gambierdiscus (Dinophyceae). PLoS ONE 2015, 10, E0119676. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, L.; Smith, K.F.; Verma, A.; Curley, B.G.; Harwood, D.T.; Murray, S.; Kohli, G.S.; Solomona, D.; Rongo, T.; Munday, R.; et al. A new species of Gambierdiscus (Dinophyceae) from the south-west Pacific: Gambierdiscus honu sp. nov. Harmful Algae 2017, 65, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Fraga, S.; Rodrıguez, F.; Caillaud, A.; Diogene, J.; Raho, N.; Zapata, M. Gambierdiscus excentricus sp. nov. (Dinophyceae), a benthic toxic dinoflagellate from the Canary Islands (NE Atlantic Ocean). Harmful Algae 2011, 11, 10–22. [Google Scholar] [CrossRef]
- Nishimura, T.; Sato, S.; Tawong, W.; Sakanari, H.; Yamaguchi, H.; Adachi, M. Morphology of Gambierdiscus scabrosus sp. nov. (Gonyaulacales): A new epiphitic dinoflagellate from coastal areas of Japan. J. Phycol. 2014, 50, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Fraga, S.; Rodrıguez, F. Genus Gambierdiscus in the Canary Islands (NE Atlantic Ocean) with description of Gambierdiscus silvae sp. nov., a new potentially toxic epiphytic benthic dinoflagellate. Protist 2014, 165, 839–853. [Google Scholar] [CrossRef] [PubMed]
- Fraga, S.; Rodríguez, F.; Riobó, P.; Bravo, I. Gambierdiscus balechii sp. nov (Dinophyceae), a new benthic toxic dinoflagellate from the Celebes Sea (SW Pacific Ocean). Harmful Algae 2016, 58, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.F.; Rhodes, L.; Verma, A.; Curley, B.G.; Harwood, D.T.; Kohli, G.S.; Solomona, D.; Rongo, T.; Munday, R.; Murray, S.A. A new Gambierdiscus species (Dinophyceae) from Rarotonga, Cook Islands: Gambierdiscus cheloniae sp. nov. Harmful Algae 2016, 60, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Kretzschmar, A.L.; Verma, A.; Harwood, D.T.; Hoppenrath, M.; Murray, S. Characterization of Gambierdiscus lapillus sp. nov. (Gonyaulacales, Dinophyceae): A new toxic dinoflagellate from the Great Barrier Reef (Australia). J. Phycol. 2017, 53, 283–297. [Google Scholar] [CrossRef] [PubMed]
- Murray, S.; Momigliano, P.; Heimann, K.; Blair, D. Molecular phylogenetics and morphology of Gambierdiscus yasumotoi from tropical eastern Australia. Harmful Algae 2014, 39, 242–252. [Google Scholar] [CrossRef]
- Rhodes, L.; Papiol, G.; Smith, K.; Harwood, T. Gambierdiscus cf. yasumotoi (Dinophyceae) isolated from New Zealand’s sub-tropical northern coastal waters. N. Z. J. Mar. Freshw. Res. 2014, 48, 303–310. [Google Scholar] [CrossRef]
- Kimura, K.; Okuda, S.; Nakayama, K.; Shikata, T.; Takahashi, F.; Yamaguchi, H.; Skamoto, S.; Yamaguchi, M.; Tomaru, Y. RNA Sequencing Revealed Numerous Polyketide Synthase Genes in the Harmful Dinoflagellate Karenia mikimotoi. PLoS ONE 2015, 10, E0142731. [Google Scholar] [CrossRef] [PubMed]
- Wear, R.G.; Gardner, J.P.A. Biological effects of the toxic algal bloom of February and March 1998 on the benthos of Wellington Harbour, New Zealand. Mar. Ecol. Prog. Ser. 2001, 218, 63–76. [Google Scholar] [CrossRef]
- Yang, Z.B.; Takayama, H.; Matsuoka, K.; Hodgkiss, I.J. Karenia digitata sp. nov. (Gymnodiniales, Dinophyceae), a new harmful algal bloom species from the coastal waters of west Japan and Hong Kong. Phycologia 2000, 39, 463–470. [Google Scholar] [CrossRef]
- Yang, Z.B.; Hodgkiss, I.J.; Hansen, G. Karenia longicanalis sp nov. (Dinophyceae): A new bloom-forming species isolated from Hong Kong, May 1998. Bot. Mar. 2001, 44, 67–74. [Google Scholar] [CrossRef]
- Haywood, A.J.; Steidinger, K.A.; Truby, E.W.; Bergquist, P.R.; Bergquist, P.L.; Adamson, J.; MacKenzie, L. Comparative morphology and molecular phylogenetic analysis of three new species of the genus Karenia (Dinophyceae) from New Zealand. J. Phycol. 2004, 40, 165–179. [Google Scholar] [CrossRef]
- Holland, P.T.; Shi, F.; Satake, M.; Hamamoto, Y.; Ito, E.; Beuzenberg, V.; McNabb, P.; Munday, R.; Briggs, L.; Truman, P.; et al. Novel toxins produced by the dinoflagellate Karenia brevisulcata. Harmful Algae 2012, 13, 47–57. [Google Scholar] [CrossRef]
- Adachi, R.; Fukuyo, Y. The thecal structure of a marine toxic dinoflagellate Gambierdiscus toxicus gen. et sp. nov. collected in a ciguatera-endemic area. Bull. Jpn. Soc. Sci. Fish 1979, 45, 67–71. [Google Scholar] [CrossRef]
- Caillaud, A.; de la Iglesia, P.; Darius, H.T.; Pauillac, S.; Aligizaki., K.; Fraga, S.; Chinain, M.; Diogène, J. Update on Methodologies Available for Ciguatoxin Determination: Perspectives to Confront the Onset of Ciguatera Fish Poisoning in Europe. Mar. Drugs 2010, 8, 1838–1907. [Google Scholar] [CrossRef] [PubMed]
- Cassell, R.T.; Chen, W.; Thomas, S.; Liu, L.; Rein, K.S. Brevetoxin, the dinoflagellate neurotoxin, localizes to thylakoid membranes and interacts with the light-harvesting complex II (LHCII) of photosystem II. Chembiochem 2015, 16, 1060–1067. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. Scientific Opinion on Marine Biotoxins in Shellfish—Emerging toxins: Brevetoxin Group. EFSA J. 2010, 1677, 1–29. [Google Scholar] [CrossRef]
- Turner, A.D.; Higgins, C.; Davidson, K.; Veszelovszki, A.; Payne, D.; Hungerford, J.; Higman, W. Potential Threats Posed by New or Emerging Marine Biotoxins in UK Waters and Examination of Detection Methodology Used in Their Control: Brevetoxins. Mar. Drugs 2015, 13, 1224–1254. [Google Scholar] [CrossRef] [PubMed]
- Visciano, P.; Schirone, M.; Berti, M.; Milandri, A.; Tofalo, R.; Suzzi, G. Marine Biotoxins: Occurrence, Toxicity, Regulatory Limits and Reference Methods. Front. Microbiol. 2016, 7, 1051. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, C. Biodiversity issues for the forthcoming tropical Mediterranean Sea. Hydrobiologia 2007, 580, 7–21. [Google Scholar] [CrossRef]
- Salat, J.; Pascual, J. The oceanographic and meteorological station at L’Estartit (NW Mediterranean). Tracking long-term hydrological change in the Mediterranean Sea. In Proceedings of the CIESM Workshop Ser., Monaco-Ville, Monaco, 22–24 April 2002; pp. 29–32. [Google Scholar]
- Bentur, Y.; Spanier, E. Ciguatoxin-like substances in edible fish on the eastern Mediterranean. Clin. Toxicol. 2007, 45, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Boada, L.D.; Zumbado, M.; Luzardo, O.P.; Almeida-Gonzalez, M.; Plakas, S.M.; Granade, H.R.; Abraham, A.; Jester, E.L.; Dickey, R.W. Ciguatera fish poisoning on the West Africa Coast: An emerging risk in the Canary Islands (Spain). Toxicon 2010, 56, 1516–1519. [Google Scholar] [CrossRef] [PubMed]
- Perez-Arellano, J.L.; Luzardo, O.P.; Brito, A.P.; Cabrera, M.H.; Zumbado, M.; Carranza, C.; Angel-Moreno, A.; Dickey, R.W.; Boada, L.D. Ciguatera fish poisoning, Canary Islands. Emerg. Infect. Dis. 2005, 11, 1981–1982. [Google Scholar] [CrossRef] [PubMed]
- Aligizaki, K.; Nikolaidis, G. Morphological identification of two tropical dinoflagellates of the genera Gambierdiscus and Sinophysis in the Mediterranean Sea. J. Biol. Res. 2008, 9, 75–82. [Google Scholar]
- Aligizaki, K.; Nikolaidis, G.; Fraga, S. Is Gambierdiscus expanding to new areas? Harmful Algae 2008, 36, 6–7. [Google Scholar]
- Llewellyn, L.E. Revisiting the association between sea surface temperature and the epidemiology of fish poisoning in the South Pacific: Reassessing the link between ciguatera and climate change. Toxicon 2010, 56, 691–697. [Google Scholar] [CrossRef] [PubMed]
- Laza-Martınez, A.; David, H.; Riobo, P.; Miguel, I.; Orive, E. Characterization of a Strain of Fukuyoa paulensis (Dinophyceae) from the Western Mediterranean Sea. J. Eukaryot. Microbiol. 2016, 63, 481–497. [Google Scholar] [CrossRef] [PubMed]
- Chan, T.Y.K. Ciguatera fish poisoning in East Asia and Southeast Asia. Mar. Drugs 2015, 13, 3466–3478. [Google Scholar] [CrossRef] [PubMed]
- Kohli, G.S.; Murray, S.A.; Neilan, B.A.; Rhodes, L.L.; Harwood, D.T.; Smith, K.F.; Meyer, L.; Capper, A.; Brett, S.; Hallegraeff, G.M. High abundance of the potentially maitotoxic dinoflagellate Gambierdiscus carpenteri in temperate waters of New South Wales, Australia. Harmful Algae 2014, 39, 134–145. [Google Scholar] [CrossRef]
- Murray, S.A.; Farrell, H.; Harwood, T.; Zammit, A. Is ciguatera moving south in Australia? Harmful Algae News 2016, 54, 5–6. [Google Scholar]
- Ajani, P.; Harwood, D.T.; Murray, S.A. Recent Trends in Marine Phycotoxins from Australian Coastal Waters. Mar. Drugs 2017, 15, 33. [Google Scholar] [CrossRef] [PubMed]
- Richlen, M.L.; Morton, S.L.; Barber, P.H.; Lobel, P.S. Phylogeography, morphological variation and taxonomy of the toxic dinoflagellate Gambierdiscus toxicus (Dinophyceae). Harmful Algae 2008, 7, 614–629. [Google Scholar] [CrossRef]
- Brand, L.E.; Compton, A. Long-term increase in Karenia brevis abundance along the Southwest Florida Coast. Harmful Algae 2007, 6, 232–252. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, L.L.; Haywood, A.J.; Ballantine, W.J.; MacKenzie, A.L. Algal blooms and climate anomalies in north-east New Zealand, August–December 1992. N. Z. J. Mar. Freshw. Res. 1993, 27, 419–430. [Google Scholar] [CrossRef]
- Jasperse, J.A. (Ed.) Marine Toxins and New Zealand Shellfish: Proceedings of a Workshop on Research Issues, 10–11 June 1993; Royal Society of New Zealand: Wellington, New Zealand, 1993. [Google Scholar]
- Todd, K. A Review of NSP Monitoring in New Zealand in Support of a New Programme; Cawthron Report No. 660; Marine Biotoxin Technical Committee: Nelson, New Zealand, 2003. [Google Scholar]
- Lewis, R.J.; Inserra, M.; Vetter, I.; Holland, W.C.; Hardison, D.R.; Tester, P.A.; Litaker, R.W. Rapid Extraction and Identification of Maitotoxin and Ciguatoxin-Like Toxins from Caribbean and Pacific Gambierdiscus Using a New Functional Bioassay. PLoS ONE 2016, 11, E0160006. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.; Rodriguez, I.; Barreiro, A.; Kaufmann, M.; Neto, A.I.; Hassouani, M.; Sabour, B.; Alfonso, A.; Botana, L.M.; Vasconcelos, V. First Report of Ciguatoxins in Two Starfish Species: Ophidiaster ophidianus and Marthasterias glacialis. Toxins 2015, 7, 3740–3757. [Google Scholar] [CrossRef] [PubMed]
- Pawlowiez, R.; Darius, H.T.; Cruchet, P.; Rossi, F.; Caillaud, A.; Laurent, D.; Chinain, M. Evaluation of seafood toxicity in the Australes archipelago (French Polynesia) using the neuroblastoma cell-based assay. Food Addit. Contam. Part A 2013, 30, 567–586. [Google Scholar] [CrossRef] [PubMed]
- Basti, L.; Nagai, S.; Go, J.; Okano, S.; Nagai, K.; Watanabe, R.; Suzuki, T.; Tanaka, Y. Differential inimical effects of Alexandrium spp. and Karenia spp. on cleavage, hatching, and two larval stages of Japanese pearl oyster Pinctada fucata martensii. Harmful Algae 2015, 43, 1–12. [Google Scholar] [CrossRef]
- Pearn, J. Neurology of ciguatera. J. Neurol. Neurosurg. Psychiatry 2001, 70, 4–8. [Google Scholar] [CrossRef] [PubMed]
- Inserra, M.C.; Israel, M.R.; Caldwell, A.; Castro, J.; Deuis, J.R.; Harrington, A.M.; Keramidas, A.; Garcia-Caraballo, S.; Maddern, J.; Erickson, A.; et al. Multiple sodium channel isoforms mediate the pathological effects of Pacific ciguatoxin-11. Sci. Rep. 2017, 7, 42810. [Google Scholar] [CrossRef] [PubMed]
- Hardison, D.R.; Holland, W.C.; McCall, J.R.; Bourdelais, A.J.; Baden, D.G.; Darius, H.T.; Chinain, M.; Tester, P.A.; Shea, D.; Quintana, H.A.F.; et al. Fluorescent Receptor Binding Assay for Detecting Ciguatoxins in Fish. PLoS ONE 2016, 11, E0153348. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. Scientific Opinion on Marine Biotoxins in Shellfish—Emerging toxins: Ciguatoxin Group. EFSA J. 2010, 1627, 1–38. [Google Scholar] [CrossRef]
- Watanabe, R.; Uchida, H.; Suzuki, T.; Matsushima, R.; Nagae, M.; Toyohara, Y.; Satake, M.; Oshima, Y.; Inoue, A.; Yasumoto, T. Gambieroxide, a novel epoxy polyether compound from the dinoflagellate Gambierdiscus toxicus GTP2 strain. Tetrahedron 2013, 69, 10299–10303. [Google Scholar] [CrossRef]
- Ciguatoxin. National Center for Biotechnology Information; PubChem Compound Database; CID=5311333. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/5311333 (accessed on 18 July 2017).
- Lewis, R.J.; Holmes, M.J. Origin and Transfer of Toxins Involved in Ciguatera. Comp. Biochem. Physiol. 1993, 106C, 615–628. [Google Scholar] [CrossRef]
- Murata, M.; Matsumori, N.; Konoki, K.; Oishi, T. Structural Features of Dinoflagellate Toxins Underlying Biological Activity as Viewed by NMR. Bull. Chem. Soc. Jpn. 2008, 81, 307–319. [Google Scholar] [CrossRef]
- Lewis, R.J.; Sellin, M.; Poli, M.A.; Norton, R.S.; MacLeod, J.K.; Sheil, M.M. Purification and characterization of ciguatoxins from moray eel (Lycodontis jauanicus, Muraenidae). Toxicon 1991, 29, 1115–1127. [Google Scholar] [CrossRef]
- Mafra, L.L.; Bricelj, V.M.; Fennel, K. Domoic acid uptake and elimination kinetics in oysters and mussels in relation to body size and anatomical distribution of toxin. Aquat. Toxicol. 2010, 100, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Lehane, L.; Lewis, R.J. Ciguatera: Recent advances but the risk remains. Int. J. Food Microbiol. 2000, 61, 91–125. [Google Scholar] [CrossRef]
- Meyer, L.; Capper, A.; Carter, S.; Simpfendorfer, C. An investigation into ciguatoxin bioaccumulation in sharks. Toxicon 2016, 119, 234–243. [Google Scholar] [CrossRef] [PubMed]
- Bottein Dechraoui, M.-Y.; Wacksman, J.J.; Ramsdell, J.S. Species Selective Resistance of Cardiac Muscle Voltage Gated Sodium Channels: Characterization of Brevetoxin and Ciguatoxin Binding Sites in Rats and Fish. Toxicon 2006, 48, 702–712. [Google Scholar] [CrossRef] [PubMed]
- Eisenblätter, A.; Lewis, R.; Dörfler, A.; Forster, C.; Zimmermann, K. Brain Mechanisms of Abnormal Temperature Perception in Cold Allodynia induced by Ciguatoxin. Ann. Neurol. 2017, 81, 104–116. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, J.; Liberona, J.L.; Molgo, J.; Jaimovich, E. Pacific ciguatoxin-1b effect over Na+ and K+ currents, inositol 1,4,5-triphosphate content and intracellular Ca2+ signals in cultured rat myotubes. Br. J. Pharmacol. 2002, 137, 1055–1062. [Google Scholar] [CrossRef] [PubMed]
- Birinyi-Strachan, L.C.; Gunning, S.J.; Lewis, R.J.; Nicholson, G.M. Block of voltage-gated potassium channels by Pacific ciguatoxin-1 contributes to increased neuronal excitability in rat sensory neurons. Toxicol. Appl. Pharmacol. 2005, 204, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Martin, V.; Vale, C.; Antelo, A.; Hirama, M.; Yamashita, S.; Vieytes, M.R.; Botana, L.M. Differential effects of ciguatoxin and maitotoxin in primary cultures of cortical neurons. Chem. Res. Toxicol. 2014, 27, 1387–1400. [Google Scholar] [CrossRef] [PubMed]
- Au, N.P.B.; Kumar, G.; Asthana, P.; Tin, C.; Mak, Y.L.; Chan, L.L.; Lam, P.K.S.; Ma, C.H.M. Ciguatoxin reduces regenerative capacity of axotomized peripheral neurons and delays functional recovery in pre-exposed mice after peripheral nerve injury. Sci. Rep. 2015, 6, 26809. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.; Au, N.P.; Lei, E.N.; Mak, Y.L.; Chan, L.L.; Lam, M.H.; Chan, L.L.; Lam, P.K.; Ma, C.H. Acute Exposure to Pacific Ciguatoxin Reduces Electroencephalogram Activity and Disrupts Neurotransmitter Metabolic Pathways in Motor Cortex. Mol. Neurobiol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Cao, B.; Wang, J.; Liu, J.; Tung, V.O.; Lam, P.K.; Chan, L.L.; Li, Y. Neurotoxicity and reactive astrogliosis in the anterior cingulate cortex in acute ciguatera poisoning. Neuromol. Med. 2013, 15, 310–323. [Google Scholar] [CrossRef] [PubMed]
- Ryan, J.C.; Bottein Dechraoui, M.-Y.; Morey, J.S.; Rezvani, A.; Levin, E.D.; Gordon, C.J.; Ramsdell, J.S.; Van Dolah, F.M. Transcriptional profiling of whole blood and serum protein analysis of mice exposed to the neurotoxin Pacific Ciguatoxin-1. Neurotoxicology 2007, 28, 1099–1109. [Google Scholar] [CrossRef] [PubMed]
- Ryan, J.C.; Morey, J.S.; Bottein Dechraoui, M.-Y.; Ramsdell, J.S.; Van Dolah, F.M. Gene expression profiling in brain of mice exposed to the marine neurotoxin ciguatoxin reveals an acute anti-inflammatory, neuroprotective response. BMC Neurosc. 2010, 11, 107. [Google Scholar] [CrossRef] [PubMed]
- Mattei, C.; Wen, P.J.; Nguyen-Huu, T.D.; Alvarez, M.; Benoit, E.; Bourdelais, A.J.; Lewis, R.J.; Baden, D.G.; Molgo, J.; Meunier, F.A. Brevenal Inhibits Pacific Ciguatoxin-1B-Induced Neurosecretion from Bovine Chromaffin Cells. PLoS ONE 2008, 3, e3448. [Google Scholar] [CrossRef] [PubMed]
- Nguyen-Huu, T.D.; Mattei, C.; Wen, P.J.; Bourdelais, A.J.; Lewis, R.J.; Benoit, E.; Baden, D.G.; Molgó, J.; Meunier, F.A. Ciguatoxin-induced catecholamine secretion in bovine chromaffin cells: Mechanism of action and reversible inhibition by brevenal. Toxicon 2010, 56, 792–796. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.J.; Norton, R.S.; Brereton, I.M.; Eccles, C.D. Ciguatoxin-2 is a diastereomer of ciguatoxin-3. Toxicon 1993, 31, 637–643. [Google Scholar] [CrossRef]
- Djouhri, L.; Fang, X.; Okuse, K.; Wood, J.N.; Berry, C.M.; Lawson, S.N. The TTX-resistant sodium channel Nav1.8 (SNS/PN3): Expression and correlation with membrane properties in rat nociceptive primary afferent neurons. J. Physiol. 2003, 550, 739–752. [Google Scholar] [CrossRef] [PubMed]
- Patel, R.; Brice, N.L.; Lewis, R.J.; Dickenson, A.H. Ionic mechanisms of spinal neuronal cold hypersensitivity in ciguatera. Eur. J. Neurosci. 2015, 42, 3004–3011. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, K.; Leffler, A.; Babes, A.; Cendan, C.M.; Carr, R.W.; Kobayashi, J.; Nau, C.; Wood, J.N.; Reeh, P.W. Sensory neuron sodium channel Nav1.8 is essential for pain at low temperatures. Nature 2007, 447, 856–859. [Google Scholar] [CrossRef] [PubMed]
- Lolignier, S.; Bonnet, C.; Gaudioso, C.; Noel, J.; Ruel, J.; Amsalem, M.; Ferrier, J.; Rodat-Despoix, L.; Bouvier, V.; Aissouni, Y.; et al. The Nav1.9 channel is a key determinant of cold pain sensation and cold allodynia. Cell Rep. 2015, 11, 1067–1078. [Google Scholar] [CrossRef] [PubMed]
- Mattei, C.; Marquais, M.; Schlumberger, S.; Molgó, J.; Vernoux, J.P.; Lewis, R.J.; Benoit, E. Analysis of Caribbean ciguatoxin-1 effects on frog myelinated axons and the neuromuscular junction. Toxicon 2010, 56, 759–767. [Google Scholar] [CrossRef] [PubMed]
- Sauviat, M.P.; Marquais, M.; Vernoux, J.P. Muscarinic effects of the Caribbean ciguatoxin C-CTX-1 on frog atrial heart muscle. Toxicon 2002, 40, 1155–1163. [Google Scholar] [CrossRef]
- Yamaoka, K.; Inoue, M.; Miyahara, H.; Miyazaki, K.; Hirama, M. A quantitative and comparative study of the effects of a synthetic ciguatoxin CTX3C on the kinetic properties of voltage-dependent sodium channels. Br. J. Pharmacol. 2004, 142, 879–889. [Google Scholar] [CrossRef] [PubMed]
- Yamaoka, K.; Inoue, M.; Miyazaki, K.; Hirama, M.; Kondo, C.; Kinoshita, E.; Miyoshi, H.; Seyama, I. Synthetic Ciguatoxins Selectively Activate Nav1.8-derived Chimeric Sodium Channels Expressed in HEK293 Cells. J. Biol. Chem. 2009, 284, 7597–7605. [Google Scholar] [CrossRef] [PubMed]
- Perez, S.; Vale, C.; Alonso, E.; Alfonso, C.; Rodriguez, P.; Otero, P.; Alfonso, A.; Vale, P.; Hirama, M.; Vieytes, M.R.; et al. A comparative study of the effect of ciguatoxins on voltage dependent Na+ and K+ channels in cerebellar neurons. Chem. Res. Toxicol. 2011, 24, 587–596. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, V.; Azevedo, J.; Silva, M.; Ramos, V. Effects of Marine Toxins on the Reproduction and Early Stages Development of Aquatic Organisms. Mar. Drugs 2010, 8, 59–79. [Google Scholar] [CrossRef] [PubMed]
- Allemand, D.; De Renzis, G.; Payan, P.; Girard, J.-P. Regulatory and Energetic Role of Na+ in Amino Acid Uptake by Fertilized Sea Urchin Eggs. Dev. Biol. 1986, 118, 19–27. [Google Scholar] [CrossRef]
- Shmukler, Y.; Nikishin, D. Transmitters in Blastomere Interactions. In Cell Interaction; Gowder, S., Ed.; InTech: Rijeka, Croatia, 2012; pp. 31–65. ISBN 978-953-51-0792-7. [Google Scholar]
- Buznikov, G.A.; Slotkin, T.A.; Lauder, J.M. Sea urchin embryos and larvae as biosensors for neurotoxicants. Curr. Protoc. Toxicol. 2003, Chapter 1, 1.6.1–1.6.24. [Google Scholar] [CrossRef]
- Yan, M.; Leung, P.T.; Ip, J.C.; Cheng, J.P.; Wu, J.J.; Gu, J.R.; Lam, P.K. Developmental toxicity and molecular responses of marine medaka (Oryzias melastigma) embryos to ciguatoxin P-CTX-1 exposure. Aquat. Toxicol. 2017, 185, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Edmunds, J.S.G.; McCarthy, R.A.; Ramsdell, J.S. Ciguatoxin reduces larval survivability in finfish. Toxicon 1999, 37, 1827–1832. [Google Scholar] [CrossRef]
- Colman, J.R.; Bottein Dechraoui, M.-Y.; Dickey, R.W.; Ramsdell, J.S. Characterization of the developmental toxicity of Caribbean ciguatoxins in finfish embryos. Toxicon 2004, 44, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Nagai, H.; Murata, M.; Torigoe, K.; Satake, M.; Yasumoto, T. Gambieric acids, new potent antifungal substances with unprecedented polyether structures from a marine dinoflagellate Gambierdiscus toxicus. J. Org. Chem. 1992, 57, 5448–5453. [Google Scholar] [CrossRef]
- Satake, M.; Murata, M.; Yasumoto, T. Gambierol—A new toxic polyether compound isolated from the marine dinoflagellate Gambierdiscus toxicus. J. Am. Chem. Soc. 1993, 115, 361–362. [Google Scholar] [CrossRef]
- Rodriguez, I.; Genta-Jouve, G.; Alfonso, C.; Calabro, K.; Alonso, E.; Sanchez, J.A.; Alfonso, A.; Thomas, O.P.; Botana, L.M. Gambierone, a ladder-shaped polyether from the dinoflagellate Gambierdiscus belizeanus. Org. Lett. 2015, 17, 2392–2395. [Google Scholar] [CrossRef] [PubMed]
- Holmes, M.J.; Lewis, R.J. Purification and characterisation of large and small maitotoxins from cultured Gambierdiscus toxicus. Nat. Toxins 1994, 2, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Hossen, V.; Soliño, L.; Leroy, P.; David, E.; Velge, P.; Dragacci, S.; Krys, S.; Flores Quintana, H.; Diogène, J. Contribution to the risk characterization of ciguatoxins: LOAEL estimated from eight ciguatera fish poisoning events in Guadeloupe (French West Indies). Environ. Res. 2015, 143, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Fuwa, H.; Kainuma, N.; Tachibana, K.; Tsukano, C.; Satake, M.; Sasaki, M. Diverted total synthesis and biological evaluation of gambierol analogues: Elucidation of crucial structural elements for potent toxicity. Chemistry 2004, 10, 4894–4909. [Google Scholar] [CrossRef] [PubMed]
- Ghiaroni, V.; Sasaki, M.; Fuwa, H.; Rossini, G.P.; Scalera, G.; Yasumoto, T.; Pietra, P.; Bigiani, A. Inhibition of voltage-gated potassium currents by gambierol in mouse taste cells. Toxicol. Sci. 2005, 85, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Cuypers, E.; Abdel-Mottaleb, Y.; Kopljar, I.; Rainier, J.D.; Raes, A.L.; Snyders, D.J.; Tytgat, J. Gambierol, a toxin produced by the dinoflagellate Gambierdiscus toxicus, is a potent blocker of voltage-gated potassium channels. Toxicon 2008, 51, 974–983. [Google Scholar] [CrossRef] [PubMed]
- Inoue, M.; Hirama, M.; Satake, M.; Sugiyama, K.; Yasumoto, T. Inhibition of brevetoxin binding to the voltage-gated sodium channel by gambierol and gambieric acid-A. Toxicon 2003, 41, 469–474. [Google Scholar] [CrossRef]
- Kopljar, I.; Labro, A.J.; Cuypers, E.; Johnson, H.W.B.; Rainier, J.D.; Tytgat, J.; Snyders, D.J. A polyether biotoxin binding site on the lipid-exposed face of the pore domain of Kv channels revealed by the marine toxin gambierol. Proc. Natl. Acad. Sci. USA 2009, 106, 9896–9901. [Google Scholar] [CrossRef] [PubMed]
- Kopljar, I.; Labro, A.J.; de Block, T.; Rainier, J.D.; Tytgat, J.; Snyders, D.J. The ladder-shaped polyether toxin gambierol anchors the gatingmachinery of Kv3.1 channels in the resting state. J. Gen. Physiol. 2013, 141, 359–369. [Google Scholar] [CrossRef] [PubMed]
- Gambierol. National Center for Biotechnology Information; PubChem Compound Database; CID=6442244. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/6442244 (accessed on 18 July 2017).
- Rubiolo, J.A.; Vale, C.; Martín, V.; Fuwa, H.; Sasaki, M.; Botana, L.M. Potassium currents inhibition by gambierol analogs prevents human T lymphocyte activation. Arch. Toxicol. 2015, 89, 1119–1134. [Google Scholar] [CrossRef] [PubMed]
- Konoki, K.; Suga, Y.; Fuwa, H.; Yotsu-Yamashita, M.; Sasaki, M. Evaluation of gambierol and its analogs for their inhibition of human Kv1.2 and cytotoxicity. Bioorg. Med. Chem. Lett. 2015, 25, 514–518. [Google Scholar] [CrossRef] [PubMed]
- Kopljar, I.; Grottesi, A.; de Block, T.; Rainier, J.D.; Tytgat, J.; Labro, A.J.; Snyders, D.J. Voltage-sensor conformation shapes the intra-membrane drug binding site that determines gambierol affinity in Kv channels. Neuropharmacology 2016, 107, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Cui, Y.; Busse, E.; Mehrotra, S.; Rainier, J.D.; Murray, T.F. Gambierol Inhibition of Voltage-Gated Potassium Channels Augments Spontaneous Ca Oscillations in Cerebrocortical Neurons. J. Pharmacol. Exp. Ther. 2014, 350, 615–623. [Google Scholar] [CrossRef] [PubMed]
- Alonso, E.; Fuwa, H.; Vale, C.; Suga, Y.; Goto, T.; Konno, Y.; Sasaki, M.; LaFerla, F.M.; Vieytes, M.R.; Gimenez-Llort, L.; et al. Design and Synthesis of Skeletal Analogues of Gambierol: Attenuation of Amyloid-β and Tau Pathology with Voltage-Gated Potassium Channel and N-Methyl-d-aspartate Receptor Implications. J. Am. Chem. Soc. 2012, 134, 7467–7479. [Google Scholar] [CrossRef] [PubMed]
- Alonso, E.; Vale, C.; Vieytes, M.R.; Laferla, F.M.; Giménez-Llort, L.; Botana, L.M. The cholinergic antagonist gymnodimine improves Aβ and tau neuropathology in an in vitro model of Alzheimer disease. Cell Physiol. Biochem. 2011, 27, 783–794. [Google Scholar] [CrossRef] [PubMed]
- Alonso, E.; Vieiram, A.C.; Rodriguez, I.; Alvariño, R.; Gegunde, S.; Fuwa, H.; Suga, Y.; Sasaki, M.; Alfonso, A.; Cifuentes, J.M.; et al. Tetracyclic truncated analog of the marine toxin gambierol modifies NMDA, tau and amyloid β expression in mice brains: Implications in AD pathology. ACS Chem. Neurosci. 2017. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Sasaki, M. Studies toward the total synthesis of gambieric acids, potent antifungal polycyclic ethers: Convergent synthesis of the CDEFG-ring system. Org. Lett. 2005, 7, 2441–2444. [Google Scholar] [CrossRef] [PubMed]
- Gambieric Acid A. National Center for Biotechnology Information; PubChem Compound Database; CID=46173842. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/46173842 (accessed on 18 July 2017).
- Maitotoxin. National Center for Biotechnology Information; PubChem Compound Database; CID=71460273. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/71460273 (accessed on 18 July 2017).
- Murata, M.; Yasumoto, T. The structure elucidation and biological activities of high molecular weight algal toxins: Maitotoxin, prymnesins and zooxanthellatoxins. Nat. Prod. Rep. 2000, 17, 293–314. [Google Scholar] [CrossRef] [PubMed]
- Nicolaou, K.C.; Frederick, M.O. On the structure of maitotoxin. Angew. Chem. Int. Ed. 2007, 46, 5278–5282. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.J. Ciguatera: Australian perspectives on a global problem. Toxicon 2006, 48, 799–809. [Google Scholar] [CrossRef] [PubMed]
- Reyes, J.G.; Sánchez-Cárdenas, C.; Acevedo-Castillo, W.; Leyton, P.; López-González, I.; Felix, R.; Gandini, M.A.; Treviño, M.B.; Treviño, C.L. Maitotoxin: An Enigmatic Toxic Molecule with Useful Applications in the Biomedical Sciences. In Seafood and Freshwater Toxins: Pharmacology, Physiology and Detection; Botana, L.M., Ed.; CRC Press: Boca Raton, FL, USA, 2014; pp. 677–694. ISBN 9781466505148. [Google Scholar]
- Kakizaki, A.; Takahashi, M.; Akagi, H.; Tachikawa, E.; Yamamoto, T.; Taira, E.; Yamakuni, T.; Ohizumi, Y. Ca2+ channel activating action of maitotoxin in cultured brainstem neurons. Eur. J. Pharmacol. 2006, 536, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Meunier, F.A.; Mattei, C.; Molgo, J. Marine toxins potently affecting neurotransmitter release. Prog. Mol. Subcell. Biol. 2009, 46, 159–186. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.Z.; Deckey, R.; Jiao, G.L.; Ren, H.F.; Li, M. Caribbean maitotoxin elevates [Ca(2+)]i and activates non-selective cation channels in HIT-T15 cells. World J. Diabetes 2013, 4, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Nicolaou, K.C.; Heretsch, P.; Nakamura, T.; Rudo, A.; Murata, M.; Konoki, K. Synthesis and Biological Evaluation of QRSTUVWXYZA’ Domains of Maitotoxin. J. Am. Chem. Soc. 2014, 136, 16444–16451. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, E.; Barrett, P. Calcium messenger system: An integrated view. Physiol. Rev. 1984, 61, 938–984. [Google Scholar]
- Allbritton, N.L.; Meyer, T.; Stryer, L. Range of messenger action of calcium ion and inositol 1,4,5-triphosphate. Science 1992, 258, 1812–1815. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Weiss, M.T.; Yin, J.; Frew, R.; Tenn, C.; Nelson, P.P.; Vair, C.; Sawyer, T.W. Role of the sodium hydrogen exchanger in maitotoxin-induced cell death in cultured rat cortical neurons. Toxicon 2009, 54, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Shono, Y.; Kamouchi, M.; Kitazono, T.; Kuroda, J.; Nakamura, K.; Hagiwara, N.; Ooboshi, H.; Ibayashi, S.; Iida, M. Change in intracellular pH causes the toxic Ca2+ entry via NCX1 in neuron- and glia-derived cells. Cell. Mol. Neurobiol. 2010, 30, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Darszon, A.; Nishigaki, T.; Beltran, C.; Trevino, C.L. Calcium channels in the development, maturation, and function of spermatozoa. Physiol. Rev. 2011, 91, 1305–1355. [Google Scholar] [CrossRef] [PubMed]
- Whitaker, M. Calcium signalling in early embryos. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2008, 363, 1401–1418. [Google Scholar] [CrossRef] [PubMed]
- O’Toole, C.M.; Arnoult, C.; Darszon, A.; Steinhardt, R.A.; Florman, H.M. Ca(2+) entry through store-operated channels in mouse sperm is initiated by egg ZP3 and drives the acrosome reaction. Mol. Biol. Cell 2000, 11, 1571–1584. [Google Scholar] [CrossRef] [PubMed]
- Jungnickel, M.K.; Marrero, H.; Birnbaumer, L.; Lemos, J.R.; Florman, H.M. Trp2 regulates entry of Ca2+ into mouse sperm triggered by egg ZP3. Nat. Cell Biol. 2001, 3, 499–502. [Google Scholar] [CrossRef] [PubMed]
- Treviño, C.L.; De la Vega-Beltrán, J.L.; Nishigaki, T.; Felix, R.; Darszon, A. Maitotoxin potently promotes Ca2+ influx in mouse spermatogenic cells and sperm, and induces the acrosome reaction. J. Cell. Physiol. 2006, 206, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Chávez, J.C.; De Blas, G.A.; De la Vega-Beltrán, J.L.; Nishigaki, T.; Chirinos, M.; González-González, M.E.; Larrea, F.; Solís, A.; Darszon, A.; Treviño, C.L. The opening of maitotoxin-sensitive calcium channels induces the acrosome reaction in human spermatozoa: Differences from the zona pellucida. Asian J. Androl. 2011, 13, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Reyes, J.; Osses, N.; Knox, M.; Darszon, A.; Trevino, C. Glucose and lactate regulate maitotoxin-activated Ca2+ entry in spermatogenic cells: The role of intracellular [Ca2+]. FEBS Lett. 2010, 584, 3111–3115. [Google Scholar] [CrossRef] [PubMed]
- Naar, J.P.; Flewelling, L.J.; Lenzi, A.; Abbott, J.P.; Granholm, A.; Jacocks, H.M.; Gannon, D.; Henry, M.; Pierce, R.; Baden, D.G.; et al. Brevetoxins, like ciguatoxins, are potent ichthyotoxic neurotoxins that accumulate in fish. Toxicon 2007, 50, 707–723. [Google Scholar] [CrossRef] [PubMed]
- Sun, P.; Leeson, C.; Zhi, X.; Leng, F.; Pierce, R.H.; Henry, M.S.; Rein, K.S. Characterization of an epoxide hydrolase from the Florida red tile dinoflagellate, Karenia brevis. Phytochemistry 2016, 122, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Brevetoxin A. National Center for Biotechnology Information; PubChem Compound Database; CID=10865808. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/10865808 (accessed 18 on July 2017).
- Brevetoxin B. National Center for Biotechnology Information; PubChem Compound Database; CID=10865865. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/10865865 (accessed 18 on July 2017).
- Nicolaou, K.C.; Frederick, M.O.; Aversa, R.J. The Continuing Saga of the Marine Polyether Biotoxins. Angew. Chem. Int. Ed. Engl. 2008, 47, 7182–7225. [Google Scholar] [CrossRef] [PubMed]
- Bottein Dechraoui, M.-Y.; Ramsdell, J.S. Type B Brevetoxins Show Tissue Selectivity for Voltage-Gated Sodium Channels: Comparison of Brain, Skeletal Muscle and Cardiac Sodium Channels. Toxicon 2003, 41, 919–927. [Google Scholar] [CrossRef]
- Cocilova, C.C.; Milton, S.L. Characterization of brevetoxin (PbTx-3) exposure in neurons of the anoxia-tolerant freshwater turtle (Trachemys scripta). Aquat. Toxicol. 2016, 180, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Kimm-Brinson, K.L.; Ramsdell, J.S. The Red Tide Toxin, Brevetoxin, Induces Embryo Toxicity and Developmental Abnormalities. Environ. Health Perspect. 2001, 109, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Silvestre, F.; Tosti, E. Impact of Marine Drugs on Animal Reproductive Processes. Mar. Drugs 2009, 7, 539–564. [Google Scholar] [CrossRef] [PubMed]
- Colman, J.R.; Ramsdell, J.S. The type B Brevetoxin (PbTx-3) adversely affects development, cardiovascular function, and survival in Medaka (Oryzias latipes) embryos. Environ. Health Perspect. 2003, 111, 1920–1925. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.Z.; Gobler, C.J. Lethal effects of Northwest Atlantic Ocean isolates of the dinoflagellate, Scrippsiella trochoidea, on Eastern oyster (Crassostrea virginica) and Northern quahog (Mercenaria mercenaria) Larvae. Mar. Biol. 2012, 159, 199–210. [Google Scholar] [CrossRef]
- Mooney, B.D.; de Salas, M.; Hallegraeff, G.M.; Place, A.R. Survey for Karlotoxin Production in 15 Species of Gymnodinioid Dinoflagellates (Kareniaceae, Dinophyta). J. Phycol. 2009, 45, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Furey, A.; Garcia, J.; O’Callaghan, K.; Lehane, M.; Amandi, M.J.; James, K.J. Brevetoxins: Structure, toxicology and origin. In Phycotoxins: Chemistry and Biochemistry; Botana, L.M., Ed.; Blackwell Publishing: Hoboken, NJ, USA, 2007; pp. 19–46. ISBN 9780813827001. [Google Scholar]
- Ramsdell, J.S. The molecular and integrative basis to brevetoxin toxicity. In Seafood and Freshwater Toxins: Pharmacology, Physiology, and Detection, 2nd ed.; Botana, L.M., Ed.; CRC Press: New York, NY, USA, 2008; pp. 519–545. ISBN 9780849374371. [Google Scholar]
- Brand, L.E.; Campbell, L.; Bresnan, E. Karenia: The biology and ecology of a toxic genus. Harmful Algae 2012, 14, 156–178. [Google Scholar] [CrossRef]
- Landsberg, J.H. The effects of harmful algal blooms on aquatic organisms. Rev. Fish. Sci. 2002, 10, 113–390. [Google Scholar] [CrossRef]
- Fowler, N.; Tomas, C.; Baden, D.; Campbell, L.; Bourdelais, A. Chemical analysis of Karenia papilionacea. Toxicon 2015, 101, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Shi, F.; McNabb, P.; Rhodes, L.; Holland, P.; Webb, S.; Adamson, J.; Immers, A.; Gooneratne, R.; Holland, J. The toxic effects of three dinoflagellate species from the genus Karenia on invertebrate larvae and finfish. N. Z. J. Mar. Freshw. Res. 2012, 46, 149–165. [Google Scholar] [CrossRef]
- Satake, M.; Shoji, M.; Oshima, Y.; Naoki, H.; Fujita, T.; Yasumoto, T. Gymnocin-A, a cytotoxic polyether from the notorious red tide dinoflagellate, Gymnodinium mikimotoi. Tetrahedron Lett. 2002, 43, 5829–5832. [Google Scholar] [CrossRef]
- Satake, M.; Tanaka, Y.; Ishikura, Y.; Oshima, Y.; Naoki, H.; Yasumoto, T. Gymnocin-B with the largest contiguous polyether rings from the red tide dinoflagellate, Karenia (formerly Gymnodinium) mikimotoi. Tetrahedron Lett. 2005, 46, 3537–3540. [Google Scholar] [CrossRef]
- Dorantes-Aranda, J.J.; Seger, A.; Mardones, J.I.; Nichols, P.D.; Hallegraeff, G.M. Progress in Understanding Algal Bloom-Mediated Fish Kills: The Role of Superoxide Radicals, Phycotoxins and Fatty Acids. PLoS ONE 2015, 10, e0133549. [Google Scholar] [CrossRef] [PubMed]
- Mooney, B.D.; Nichols, P.D.; de Salas, M.F.; Hallegraeff, G.M. Lipid, fatty acid and sterol composition of eight species of Kareniaceae (Dinophyta): Chemotaxonomy and putative lipid phycotoxins. J. Phycol. 2007, 43, 101–111. [Google Scholar] [CrossRef]
- Yang, W.; Zhang, N.; Cui, W.; Xu, Y.; Li, H.; Liu, J. Effects of co-existing microalgae and grazers on the production of hemolytic toxins in Karenia mikimotoi. Chin. J. Oceanol. Limnol. 2011, 29, 1155–1163. [Google Scholar] [CrossRef]
- Dang, L.X.; Li, Y.; Liu, F.; Zhang, Y.; Yang, W.D.; Li, H.Y.; Liu, J.S. Chemical Response of the Toxic Dinoflagellate Karenia mikimotoi Against Grazing by Three Species of Zooplankton. J. Eukaryot. Microbiol. 2015, 62, 470–480. [Google Scholar] [CrossRef] [PubMed]
- Chang, F.H. Toxic effects of three closely-related dinoflagellates, Karenia concordia, K. brevisulcata and K. mikimotoi (Gymnodiniales, Dinophyceae) on other microalgal species. Harmful Algae 2011, 10, 181–187. [Google Scholar] [CrossRef]
- Gentien, P.; Arzul, G. Exotoxins production by Gyrodinium cf. aureolum (Dinophyceae). J. Exp. Mar. Biol. Assoc. UK 1990, 70, 571–581. [Google Scholar] [CrossRef]
- Yamasaki, Y.; Kim, D.I.; Matsuyama, Y.; Oda, T.; Honji, T. Production of superoxide anion and hydrogen peroxide by the red tide dinoflagellate Karenia mikimotoi. J. Biosci. Bioeng. 2004, 97, 212–215. [Google Scholar] [CrossRef]
- Marshall, J.; De Salas, M.; Oda, T.; Hallegraeff, G. Superoxide production by marine microalgae. I. Survey of 37 species from 6 classes. Mar. Biol. 2005, 147, 533–540. [Google Scholar] [CrossRef]
- Marshall, J.; De Salas, M.; Oda, T.; Hallegraeff, G. Superoxide production by marine microalgae. II. Towards understanding ecological consequences and possible functions. Mar. Biol. 2005, 147, 541–549. [Google Scholar] [CrossRef]
- MacKenzie, L.A.; Haywood, A.J.; Adamson, J.; Truman, P.; Till, D.; Seki, T.; Satake, M.; Yasumoto, T. Gymnodimine Contamination of Shellfish in New Zealand. In Harmful and Toxic Algal Blooms; Yasumoto, T., Oshima, Y., Fukuyo, Y., Eds.; Intergovernmental Oceanographic Commission of UNESCO: Paris, France, 1996; pp. 97–100. [Google Scholar]
- McCarthy, M.; Bane, V.; García-Altares, M.; van Pelt, F.N.A.M.; Furey, A.; O’Halloran, J. Assessment of emerging biotoxins (pinnatoxin G and spirolides) at Europe’s first marine reserve: Lough Hyne. Toxicon 2015, 108, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Tsukano, C.; Ebine, M.; Sasaki, M. Convergent Total Synthesis of Gymnocin-A and Evaluation of Synthetic Analogues. J. Am. Chem. Soc. 2005, 127, 4326–4335. [Google Scholar] [CrossRef] [PubMed]
- Otero, A.; Chapela, M.-J.; Atanassova, M.; Vieites, J.M.; Cabado, A.G. Cyclic Imines: Chemistry and Mechanism of Action: A Review. Chem. Res. Toxicol. 2011, 24, 1817–1829. [Google Scholar] [CrossRef] [PubMed]
- Miles, C.O.; Wilkins, A.L.; Stirling, D.J.; MacKenzie, A.L. Gymnodimine C, an isomer of gymnodimine B, from Karenia selliformis. J. Agric. Food Chem. 2003, 51, 4838–4840. [Google Scholar] [CrossRef] [PubMed]
- Kharrat, R.; Servent, D.; Girard, E.; Ouanounou, G.; Amar, M.; Marrouchi, R.; Benoit, E.; Molgó, J. The marine phycotoxin gymnodimine targets muscular and neuronal nicotinic acetylcholine receptor subtypes with high affinity. J. Neurochem. 2008, 107, 952–963. [Google Scholar] [CrossRef] [PubMed]
- Gymnodimine A. National Center for Biotechnology Information; PubChem Compound Database; CID=11649137. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/11649137 (accessed on 18 July 2017).
- Bourne, Y.; Radic, Z.; Araoz, R.; Talley, T.T.; Benoit, E.; Servent, D.; Taylor, P.; Molgo, J.; Marchot, P. Structural determinants in phycotoxins and AChBP conferring high affinity binding and nicotinic AChR antagonism. Proc. Natl. Acad. Sci. USA 2010, 107, 6076–6081. [Google Scholar] [CrossRef] [PubMed]
- Stivala, C.E.; Benoit, E.; Araoz, R.; Servent, D.; Novikov, A.; Molgó, J.; Zakarian, A. Synthesis and Biology of Cyclic Imine Toxins, An Emerging Class of Potent, Globally Distributed Marine Toxins. Nat. Prod. Rep. 2015, 32, 411–435. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, M.; Hellström-Lindahl, E. Nicotinic receptor agonists and antagonists increase sAPPα secretion and decrease Aβ levels in vitro. Neurochem. Int. 2009, 54, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Chang, F.H. Gymnodinium brevisulcatum sp. nov. (Gymnodiniales, Dinophyceae), a new species isolated from the 1998 summer toxic bloom in Wellington Harbour, New Zealand. Phycologia 1999, 38, 377–384. [Google Scholar] [CrossRef]
- Seki, T.; Satake, M.; Mackenzie, L.; Kaspar, H.F.; Yasumoto, T. Gymnodimine, a new marine toxin of unprecedented structure isolated from New Zealand oysters and the dinoflagellate, Gymnodinium sp. Tetrahedron Lett. 1995, 36, 7093–7096. [Google Scholar] [CrossRef]
- Dragunow, M.; Trzoss, M.; Brimble, M.A.; Cameron, R.; Beuzenberg, V.; Holland, P.; Mountfort, D. Investigations into the cellular actions of the shellfish toxin gymnodimine and analogues. Environ. Toxicol. Pharmacol. 2005, 20, 305–312. [Google Scholar] [CrossRef] [PubMed]
- De Salas, M.F.; Bolch, C.S.; Hallegraeff, G.M. Karenia asterichroma sp. nov. (Gymnodiniales, Dinophyceae), a new dinoflagellate species associated with finfish aquaculture mortalities in Tasmania, Australia. Phycologia 2004, 43, 624–631. [Google Scholar] [CrossRef]
- De Salas, M.F.; Bolch, C.J.S.; Hallegraeff, G.M. Karenia umbella sp. nov. (Gymnodiniales, Dinophyceae), a new potentially ichthyotoxic dinoflagellate species from Tasmania, Australia. Phycologia 2004, 43, 166–175. [Google Scholar] [CrossRef]
- Brevenal. National Center for Biotechnology Information; PubChem Compound Database; CID=46886836. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/46886836 (accessed on 18 July 2017).
- Bourdelais, A.J.; Campbell, S.; Jacocks, H.; Naar, J.; Wright, J.L.; Carsi, J.; Baden, D.G. Brevenal is a natural inhibitor of brevetoxin action in sodium channel receptor binding assays. Cell Mol. Neurobiol. 2004, 24, 553–563. [Google Scholar] [CrossRef] [PubMed]
- Gold, E.P.; Jacocks, H.M.; Bourdelais, A.J.; Baden, D.G. Brevenal, a brevetoxin antagonist from Karenia brevis, binds to a previously unreported site on mammalian sodium channels. Harmful Algae 2013, 26, 12–19. [Google Scholar] [CrossRef] [PubMed]
- LePage, K.T.; Rainier, J.D.; Johnson, H.W.B.; Baden, D.G.; Murray, T.F. Gambierol Acts as a Functional Antagonist of Neurotoxin Site 5 on Voltage Gated Sodium Channels in Cerebellar Granule Neurons. J. Pharmacol. Exp. Ther. 2007, 323, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Cestèle, S.; Catterall, W.A. Molecular mechanisms of neurotoxin action on voltage-gated sodium channels. Biochimie 2000, 82, 883–892. [Google Scholar] [CrossRef]
- Bourdelais, A.J.; Jacocks, H.M.; Wright, J.L.C.; Bigwarfe, P.M.; Baden, D.G. A New Polyether Ladder Compound Produced by the Dinoflagellate Karenia. J. Nat. Prod. 2005, 68, 2–6. [Google Scholar] [CrossRef] [PubMed]
- Waggett, R.J.; Hardison, D.R.; Tester, P.A. Toxicity and nutritional inadequacy of Karenia brevis: Synergistic mechanisms disrupt top-down grazer control. Mar. Ecol. Prog. Ser. 2012, 444, 15–30. [Google Scholar] [CrossRef]
- Hong, J.; Talapatra, S.; Katz, J.; Tester, P.A.; Waggett, R.J.; Place, A.R. Algal Toxins Alter Copepod Feeding Behavior. PLoS ONE 2012, 7, E36845. [Google Scholar] [CrossRef] [PubMed]
- Sunda, W.G.; Burleson, C.; Hardison, D.R.; Morey, J.S.; Wang, Z.; Wolny, J.; Corcoran, A.A.; Flewelling, L.J.; Van Dolah, F.M. Osmotic stress does not trigger brevetoxin production in the dinoflagellate Karenia brevis. Proc. Natl. Acad. Sci. USA 2013, 110, 10223–10228. [Google Scholar] [CrossRef] [PubMed]
- Buznikov, G.A. Neurotransmitters in Embryogenesis; Harwood Academic Publ.: Chur, Switzerland, 1990; p. 526. ISBN 3718648776, 9783718648771. [Google Scholar]
- Buznikov, G.A.; Shmukler, Y.B.; Lauder, J.M. From oocyte to neuron: Do neurotransmitters function in the same way throughout development? Cell. Mol. Neurobiol. 1996, 16, 532–559. [Google Scholar] [CrossRef]
- Errera, R.M.; Campbell, L. Osmotic stress triggers toxin production by the dinoflagellate Karenia brevis. Proc. Natl. Acad. Sci. USA 2011, 108, 10597–10601. [Google Scholar] [CrossRef] [PubMed]
- Errera, R.M.; Campbell, L. Correction for Errera and Campbell, Osmotic stresstriggers toxin production by the dinoflagellate Karenia brevis. Proc. Natl. Acad. Sci. USA 2012, 109, 17723–17724. [Google Scholar] [CrossRef]
- Errera, R.M.; Bourdelais, A.; Drennan, M.A.; Dodd, E.B.; Henrichs, D.W.; Campbell, L. Variation in brevetoxin and brevenal content among clonalcultures of Karenia brevis may influence bloom toxicity. Toxicon 2010, 55, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Yi, S.-J.; Kim, K.H.; Choix, H.J.; Yoo, J.O.; Jung, H.-I.; Han, J.-A.; Kim, Y.-M.; Suh, I.B.; Ha, K.-S. [Ca2+]-dependent Generation of Intracellular Reactive Oxygen Species Mediates Maitotoxin-induced Cellular Responses in Human Umbilical Vein Endothelial Cells. Mol. Cells 2006, 21, 121–128. [Google Scholar] [PubMed]
- Pignol, B.; Auvin, S.; Carré, D.; Marin, J.-G.; Chabrier, P.-E. Calpain inhibitors and antioxidants act synergistically to prevent cell necrosis: Effects of the novel dual inhibitors (cysteine protease inhibitor and antioxidant) BN 82204 and its pro-drug BN 82270. J. Neurochem. 2006, 98, 1217–1228. [Google Scholar] [CrossRef] [PubMed]
- Rolton, A.; Soudant, P.; Vignier, J.; Pierce, R.; Henry, M.; Shumway, S.E.; Bricelj, V.M.; Volety, A.K. Susceptibility of gametes and embryos of the eastern oyster, Crassostrea virginica, to Karenia brevis and its toxins. Toxicon 2015, 99, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Matile, S.; Berova, N.; Nakanishi, K. Exciton coupled circular dichroic studies of self-assembled brevetoxin-porphyrin conjugates in lipid bilayers and polar solvents. Chem. Biol. 1996, 3, 379–392. [Google Scholar] [CrossRef]
- Zhou, J.; Fritz, L. Okadaic acid localizes to chloroplasts in the DSP-toxin-producing dinoflagellates Prorocentrum lima and Prorocentrum maculosum. Phycologia 1994, 33, 455–461. [Google Scholar] [CrossRef]
- Young, F.M.; Thomson, C.; Metcalf, J.S.; Lucocq, J.M.; Codd, G.A. Immunogold localisation of microcystins in cryosectioned cells of Microcystis. J. Struct. Biol. 2005, 151, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Rivas-San Vicente, M.; Plasencia, J. Salicylic acid beyond defence: Its role in plant growth and development. J. Exp. Bot. 2011, 62, 3321–3338. [Google Scholar] [CrossRef] [PubMed]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shmukler, Y.B.; Nikishin, D.A. Ladder-Shaped Ion Channel Ligands: Current State of Knowledge. Mar. Drugs 2017, 15, 232. https://doi.org/10.3390/md15070232
Shmukler YB, Nikishin DA. Ladder-Shaped Ion Channel Ligands: Current State of Knowledge. Marine Drugs. 2017; 15(7):232. https://doi.org/10.3390/md15070232
Chicago/Turabian StyleShmukler, Yuri B., and Denis A. Nikishin. 2017. "Ladder-Shaped Ion Channel Ligands: Current State of Knowledge" Marine Drugs 15, no. 7: 232. https://doi.org/10.3390/md15070232
APA StyleShmukler, Y. B., & Nikishin, D. A. (2017). Ladder-Shaped Ion Channel Ligands: Current State of Knowledge. Marine Drugs, 15(7), 232. https://doi.org/10.3390/md15070232






