Abstract
Biofouling, an undesirable accumulation of organisms on sea-immersed structures such as ship hulls and fishing nets, is a serious economic issue whose effects include oil wastage and clogged nets. Organotin compounds were utilized since the 1960s as an antifouling material; however, the use of such compounds was later banned by the International Maritime Organization (IMO) due to their high toxicity toward marine organisms, resulting in masculinization and imposex. Since the ban, there have been extensive efforts to develop environmentally benign antifoulants. Natural antifouling products obtained from marine creatures have been the subject of considerable attention due to their potent antifouling activity and low toxicity. These antifouling compounds often contain isocyano groups, which are well known to have natural antifouling properties. On the basis of our previous total synthesis of natural isocyanoterpenoids, we envisaged the installation of an isocyano functional group onto glucosamine to produce an environmentally friendly antifouling material. This paper describes an effective synthetic method for various glucosamine-based isocyanides and evaluation of their antifouling activity and toxicity against cypris larvae of the barnacle Amphibalanus amphitrite. Glucosamine isocyanides with an ether functionality at the anomeric position exhibited potent antifouling activity, with EC50 values below 1 μg/mL, without detectable toxicity even at a high concentration of 10 μg/mL. Two isocyanides had EC50 values of 0.23 and 0.25 μg/mL, comparable to that of CuSO4, which is used as a fouling inhibitor (EC50 = 0.27 μg/mL).
1. Introduction
Many essential human activities, such as transportation and fishing, are carried out in the ocean, and submarine structures—seawater intakes pipes for power plants, breeding nets, and so on—are also widely used. Ocean-based activities such as offshore energy generation will continue to increase in the future [1]. Any structure immersed in the ocean, including ship hulls and fishing nets, will encounter biofouling, that is, an undesirable accumulation of organisms such as barnacles and mussels. Biofouling can have serious economic consequences [2], including fuel waste—up to 40% [3]—and clogging of pipes and nets [4]. Removal of the fouling organisms for annual maintenance must be done at huge cost [5]. Since the 1960s, organotin compounds such as tributyltin (TBT) and triphenyltin (TPT) were widely employed to prevent fouling [6]. However, organotin compounds were suspected to be toxic, and biological consequences for marine organisms included growth inhibition, masculinization of shellfish, abnormal shell development, brittle shells, poor weight gain, and a disorder known as imposex in snails [7]. Harmful effects of TBT were also reported for marine organisms such as fish [8,9], crustaceans [10,11], and particularly mollusks [12]. These unfavorable aspects drove the International Maritime Organization (IMO) to ban the use of organotin compounds on ships worldwide in 2008 [13]. Alternative antifouling agents in current use mainly contain copper [6], and there have been some reports of negative consequences for the ocean environment and ecosystem [14,15]. The development of an environmentally friendly antifouling agent that is suitable for the ocean environment is therefore a priority.
In current research on green antifouling agents [16], natural products obtained from marine creatures have attracted much attention [17,18,19,20,21,22,23,24]. In particular, isocyano compounds are well-known examples of natural antifouling products. For example, 10-isocyano-4-cadinene (1), obtained from nudibranchs of the family Phyllidiidae by Okino, shows strong antifouling activity (50% effective concentration (EC50) = 0.14 μg/mL) against cypris larvae of the barnacle Amphibalanus amphitrite, comparable to CuSO4 (EC50 = 0.27 μg/mL), along with low toxicity (50% lethal concentration (LC50) >10 μg/mL) [25], as shown in Figure 1. Our group achieved the first total synthesis of 1 and examined the biological activity of a synthetic sample against the same cypris larvae, obtaining similar activities (EC50 = 0.06 μg/mL, LC50 > 10 μg/mL) compared to those of the natural compound [26,27,28]. Alcohol 2 and the nitrile 3, synthetic intermediates in the total synthesis of 1, were found to have less potency than 1. Other isocyanide compounds such as 3-isocyanotheonellin (4) also exhibited potent antifouling profiles [29,30,31,32,33]. These results clearly indicate that the isocyano group confers potent antifouling properties. We envisaged a compound in which the isocyano functionality was installed on a glucosamine unit (5), obtained from crustaceans such as crabs and shrimp as a cheap and abundant biomass platform, through organic synthesis, because 5 possesses an amino group, which is essential for introduction of the isocyano group. In addition, the four hydroxy groups in 5 enable easy access to a wide variety of synthetic isocyanides. Furthermore, the compound, which is derived from a sugar-based structure, was expected to have low toxicity. In this study, glucosamine-based isocyanides (6) were designed and synthesized in short reaction sequences. The antifouling activities and toxicities of the synthetic compounds 6 against the cypris larvae of Amphibalanus amphitrite were evaluated to prove the feasibility of these compounds as alternative environmentally friendly antifouling agents, including through investigation of substituent effects at the C-1 and C-3, 4, and 6 positions.
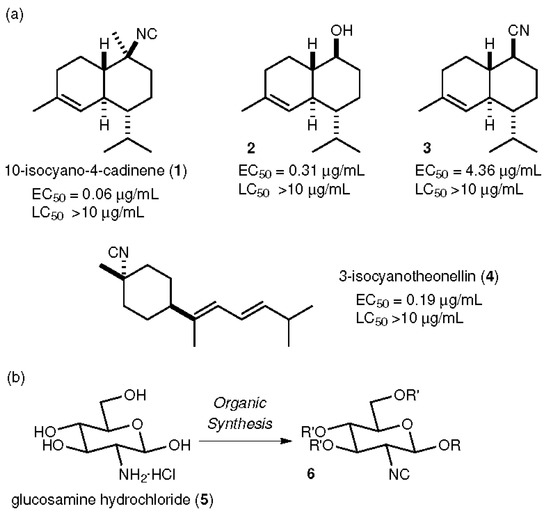
Figure 1.
Materials containing isocyano groups exhibit potent antifouling properties. (a) Previously reported natural products and derivatives with antifouling properties, along with their antifouling activities. (b) Synthetic plan for glucosamine-based isocyanides.
2. Results
2.1. Compounds with Ester Groups at the C-1 Position
Initially, the tetraacetate 6a and the tetrapalmitate 6b were prepared from d-glucosamine hydrochloride (5) by a sequence of formamide formation with HCO2Me and NaOMe [34], the acylation of hydroxy groups with Ac2O or palmitoyl chloride (PalCl), respectively, and the dehydration of formamides 7a and 7b with POCl3 and Et3N [35] (Figure 2). The antifouling activities of 6a and 6b were evaluated against the cypris larvae of Amphibalanus amphitrite after a 48 h incubation period, showing moderate activity (EC50 = 5.54 and 6.36 μg/mL, respectively) and very low toxicity (lethal larva was not observed at the concentration of 10 μg/mL).
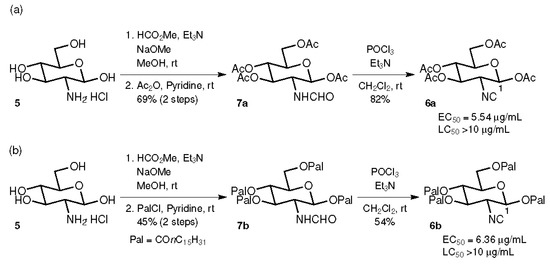
Figure 2.
Synthesis and biological activity of tetraacylated glucosamine-based isocyanides. (a) Synthesis of tetraacetate 6a. (b) Synthesis of tetrapalmitate 6b.
2.2. Compounds with Ether at the C-1 Position
Next, the effect of replacement of the C-1 ester groups of 6a and 6b with ether groups upon antifouling activity and toxicity against Amphibalanus amphitrite was examined. Phenyl (Ph), benzyl (CH2Ph), allyl (CH2CH = CH2), isopropyl (CH(CH3)2, iPr), and 2,2,2-trifluoroethyl (CH2CF3) groups were chosen as the substituent R of the C-1 alkoxy group. Figure 3 summarizes the synthesis and biological activities of isocyanides 6c–g. For the synthesis of 6c–g, the commercially available d-glucosamine derivative 8 was employed as a starting compound because glycosidation reactions between formamide 7a and alcohols such as isopropyl alcohol under various conditions did not afford the desired glycosidation products. In glycosidation reactions of 8 promoted by BF3·OEt2 (Step A), two solvent systems (CH2Cl2/room temperature, and MeCN/80 °C) were employed for optimization of chemical yield. Phenyl glycoside 9c and trifluoroethyl glycoside 9g were obtained in higher yields by allowing 8 to react with phenol and 2,2,2-trifluoroethanol at room temperature in CH2Cl2. However, treatment of 8 with benzyl alcohol, allyl alcohol, and isopropyl alcohol at 80 °C in MeCN produced benzyl glycoside 9d, allyl glycoside 9e, and isopropyl glycoside 9f in better yields. During each of the glycosidation reactions, the β-anomer was formed as the sole glycosidation product. The β-configurations at the anomeric position (C-1) of 9c–g were determined based on coupling constant analysis. Thus, a large vicinal coupling constant, J12 = 9 Hz (axial/axial relationship between the vicinal methine protons at C-1 and 2), was observed in all the 1H NMR spectra. The conversion of 9c–g into formamides 7c–g was accomplished in the following three steps (Step B): (i) removal of the phthaloyl and acetyl groups with ethylenediamine (EDA); (ii) a formamide formation reaction with HCO2Me and NaOMe; and (iii) acetylation of the three resulting hydroxyl groups with Ac2O and pyridine. Isocyanides 6c–g were derived from 7c–g using the same dehydration protocol with POCl3 and Et3N (Step C) that was used in the preparation of 7a and 7b. The biological activities of synthetic 6c–g against the cypris larvae of Amphibalanus amphitrite were then evaluated to reveal enhanced potency for 6c–g (EC50 = 0.23–0.71 μg/mL) relative to 6a and 6b (EC50 = 5.54 and 6.36 μg/mL, respectively). Furthermore, it was noteworthy that mortality among the cypris larvae was not observed during the assay, even at a high concentration of 10 μg/mL.
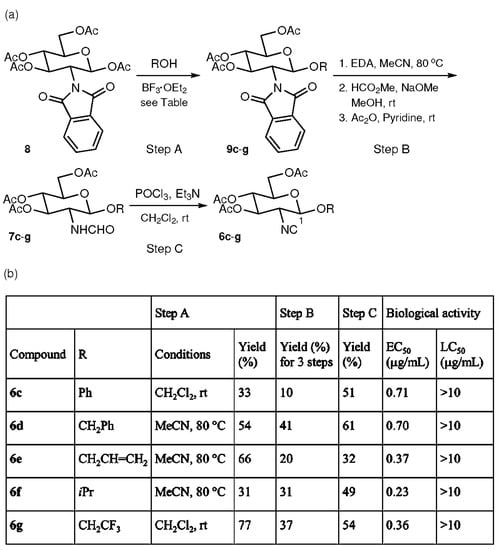
Figure 3.
Synthesis and biological activities of glucosamine-based isocyanides. (a) Synthesis of isocyanides 6c–g. (b) Table of reactions and biological activities.
2.3. Effects of Substituents at the C-3, 4, and 6 Positions
Further modifications were made by replacing the acetyl groups at C-3, 4, and 6 of the isopropyl glycoside 6f with other substituents. As illustrated in Figure 4, after removal of the phthaloyl and acetyl groups of 9f followed by formamide formation, octanoyl (COnC7H15), palmitoyl (COnC15H31), benzoyl (COPh), and TBS (SiMe2tBu) groups were installed to afford isopropyl glycosides 7h–k, respectively. Each of 7h–k was transformed to the corresponding isocyanide (6h–k) under dehydration conditions that were the same as those used for the preparation of 6a–g. Isocyanides 6h–k were tested against Amphibalanus amphitrite and exhibited similar antifouling activities (EC50 = 0.25–0.81 μg/mL) to 6f (EC50 = 0.23 μg/mL). Again, none of 6h–k showed detectable toxicity (LC50 > 10 μg/mL).
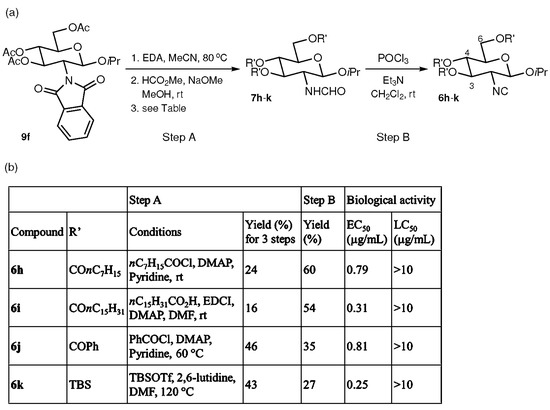
Figure 4.
Synthesis and biological activity of glycosylated glucosamine-based isocyanides. (a) Synthesis of isocyanides 6h–k. (b) Table of reactions and biological activities.
3. Discussion
These procedures are short syntheses (up to 5 steps from commercially available materials) allowing access to various glucosamine-based isocyanides using inexpensive reagents. In contrast, 27 steps were required for the total synthesis of 1. Although the chemical yields should be improved by further optimization, the synthetic scheme seems to be a reliable one for the synthesis of various isocyanides with latent antifouling activity. All of the samples obtained in the current study showed not only antifouling profiles but also extremely low toxicity against the cypris larvae of Amphibalanus amphitrite. Compounds 6a and 6b with the ester group at the C-1 position had EC50 values above 5 μg/mL, but 6c–g with the ether group at the C-1 position were more active, with EC50 values below 1 μg/mL. Compounds 6f and 6k exhibited EC50 values of 0.23 and 0.25 μg/mL, comparable to that of CuSO4, which is used as a fouling inhibitor (EC50 = 0.27 μg/mL) [27]. Substituent effects at the C-1 position of glucosamine were clearly evident in the higher EC50 values of 6a and 6b compared to 6c–g, suggesting that more detailed investigations of ether substituents at this position may enable the discovery of more potent compounds, and that substituents at the C-1 position affect the antifouling mechanism through steric or electronic effects [36,37] (Figure 2 and Figure 3). On the other hand, no significant effect on antifouling activity was obtained by changing the substituents attached to the three hydroxy groups at C-3, 4, and 6 (Figure 4). This result is advantageous from the point of view of the development of nontoxic and environmentally benign antifouling agents through the synthesis of functionalized glucosamine derivatives—for example, monomers for the synthesis of polymers that can be used in combination with paint—since it is possible to introduce various functional groups at the C-3, 4, and 6 positions without significant loss of antifouling activity.
The results obtained in this study are promising in terms of the development of green antifouling agents for practical use. Further preparation of a wide variety of glucosamine-based isocyanides along with relevant field assay and biodegradability test will be reported in our laboratory.
4. Materials and Methods
Optical rotations were obtained using a Horiba SEPA-300 instrument (Horiba, Kyoto, Japan). IR spectra were recorded on a JASCO FT/IR 4100 spectrometer using an NaCl cell. 1H and 13C NMR spectra were recorded using a JNM-EX 400 (400 MHz and 100 MHz) spectrometer (see Supplementary Materials). N,N-dimethylformamide (DMF), methanol (MeOH), and acetonitrile (MeCN) were purchased from Kanto Chemical Co. Inc. (Tokyo, Japan). Dichloromethane (CH2Cl2) was distilled from CaH2. All commercially available reagents were employed as received. Analytical TLC was carried out using pre-coated silica gel plates (TLC silica gel 60 F254). The silica gel used for column chromatography was Wakogel 60N 63–212 μm.
4.1. General Procedure for the Preparation of Formamides 7a and 7b
To a solution of d-glucosamine hydrochloride (5) (1.0 equiv) in MeOH (0.40 M) and HCO2Me (1.0 M), NaOMe (1.4 equiv) and Et3N (1.0 equiv) was added at room temperature under an Ar atmosphere. The mixture was stirred at room temperature overnight and then concentrated in vacuo. To a solution of the crude formamide in pyridine (15 equiv), Ac2O (6.0 equiv) for 7a or palmitoyl chloride (6.0 equiv) for 7b was added at room temperature under an Ar atmosphere. After stirring at room temperature overnight, MeOH (6.0 equiv) was added at 0 °C. The mixture was extracted with EtOAc, washed successively with 1.0 M NaOH, 3.0 M HCl, and brine, dried over Na2SO4, filtered, and concentrated in vacuo. The residue was purified by silica gel column chromatography (EtOAc/hexane = 40:60) to yield 7a or 7b, respectively.
4.2. General Procedure for the Preparation of Isocyanides 6a–k
To a solution of each of the series of formamides 7a–k (1.0 equiv) in CH2Cl2 (0.20 M), Et3N (9.0 equiv) and POCl3 (3.0 equiv) were added dropwise at 0 °C under an Ar atmosphere. After stirring at 0 °C for 10 min, the mixture was warmed to room temperature, stirred at room temperature for 1 h, quenched with saturated NaHCO3, and extracted with EtOAc. The combined organic extracts were washed with brine, dried over Na2SO4, filtered, and concentrated in vacuo. The residue was purified by silica gel column chromatography (EtOAc/hexane = 10:90) to yield isocyanides 6a–k, respectively.
4.3. General Procedure for the Preparation of Glycosides 9d–f
To a solution of 8 (1.0 equiv) in MeCN (0.25 M), benzyl alcohol, allyl alcohol, or isopropyl alcohol (1.0 equiv) and BF3·OEt2 (3.0 equiv) were added at room temperature under an Ar atmosphere. The mixture was stirred at 80 °C overnight, quenched with saturated aqueous NaHCO3, and extracted with EtOAc. The combined organic extracts were washed with brine, dried over Na2SO4, filtered, and concentrated in vacuo. The residue was then purified by silica gel column chromatography (EtOAc/hexane = 20:80) to yield 9d–f, respectively.
4.4. General Procedure for the Preparation of Glycosides 9c and 9g
To a solution of 8 (1.0 equiv) in CH2Cl2 (0.20 M), phenol or 2,2,2-trifluoroethanol (2.0 equiv) and BF3·OEt2 (2.0 equiv) were added at room temperature under an Ar atmosphere. The mixture was stirred at room temperature overnight, quenched with saturated aqueous NaHCO3, and extracted with AcOEt. The combined extracts were washed with brine, dried over Na2SO4, filtered, and concentrated in vacuo. The residue was then purified by silica gel column chromatography (EtOAc/hexane = 30:70) to yield 9c or 9g, respectively.
4.5. General Procedure for the Preparation of Formamides 7c–g from Glycosides 9c–g
To a solution of each of 9c–g (1.0 equiv) in MeCN (0.20 M), EDA (4.7 equiv) was added at room temperature under an Ar atmosphere. The mixture was stirred at 80 °C overnight and concentrated in vacuo. To a solution of the crude amine in MeOH (0.40 M) and HCO2Me (0.40 M), NaOMe (1.2 equiv) was added at room temperature under an Ar atmosphere. After stirring at room temperature overnight, DOWEX was added for neutralization. The mixture was filtered through a celite pad and concentrated in vacuo. To a solution of the crude formamide in pyridine (30 equiv), Ac2O (25 equiv) was added at room temperature under an Ar atmosphere. After stirring at room temperature overnight, MeOH (25 equiv) was added at 0 °C. The mixture was extracted with EtOAc, washed successively with 1.0 M NaOH (25 equiv), 3.0 M HCl, and brine, dried over Na2SO4, filtered, and concentrated in vacuo. The residue was purified by silica gel column chromatography (EtOAc/hexane = 40:60) to yield 7c–g, respectively.
4.6. Synthesis of 7h
To a solution of crude formamide in pyridine (50 equiv) and DMAP (0.50 equiv), obtained from 9f in two steps, octanoyl chloride (6.0 equiv) was added at room temperature under an Ar atmosphere. The mixture was stirred for 24 h, quenched with 3.0 M HCl (60 equiv), diluted with AcOEt, washed with brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column chromatography (EtOAc:Hexane = 5:95) to yield 7h (24% over three steps) as a white solid.
4.7. Synthesis of 7i
To a solution of crude formamide obtained from 9f in two steps, DMF (0.050 M), palmitic acid (5.0 equiv), DMAP (0.50 equiv), and EDCI (5.0 equiv) were added at room temperature under an Ar atmosphere. The mixture was stirred for 20 h, supplemented with EtOH (2.0 equiv) and EDCI (2.0 equiv), quenched with saturated aqueous NH4Cl, diluted with AcOEt, washed with brine, dried over Na2SO4, filtered, and concentrated in vacuo. The residue was purified by silica gel column chromatography (EtOAc:hexane = 10:90) to yield 7i (16% over three steps) as a white solid.
4.8. Synthesis of 7j
To the solution of crude formamide in pyridine (15 equiv) and DMAP (0.50 equiv), obtained from 9f in two steps, benzoyl chloride (5.0 equiv) was added at 60 °C under an Ar atmosphere. The mixture was stirred for 24 h, quenched with 3.0 M HCl (60 equiv), diluted with AcOEt, washed with brine, dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by silica gel column chromatography (EtOAc:hexane = 30:70) to yield 7j (46% over three steps) as a white solid.
4.9. Synthesis of 7k
To the solution of crude formamide, obtained from 9f in two steps, in DMF (0.050 M) and 2,6-Lutidine (15 equiv), TBSOTf (20 equiv) was added at 120 °C under an Ar atmosphere. The mixture was stirred for 1.5 h, quenched with saturated aqueous NaHCO3, diluted with AcOEt, washed with brine, dried over Na2SO4, filtered, and concentrated in vacuo. The residue was purified by silica gel column chromatography (EtOAc:hexane = 10:90) to yield 7k (43% over three steps) as a white solid.
4.10. Antifouling Assay
Adult barnacles (Amphibalanus amphitrite) procured from oyster farms at Lake Hamana and a pier at Shimizu bay, Shizuoka, were kept in an aquarium at 20 °C and fed on Artemia salina nauplii. Broods were released as I–II stage nauplii upon immersion in seawater after drying overnight. The nauplii thus obtained were cultured in filtered natural seawater (salinity 28) containing penicillin G (20 μg/mL) and streptomycin sulfate (30 μg/mL) and were fed on the diatom Chaetoceros gracilis at concentrations of 40 × 104 cells/mL. Larvae reached the cyprid stage in 5 days. The cyprids were collected and stored at 4 °C until use (0 days old).
The test compounds were dissolved in ethanol and aliquots of the solution were transferred to wells of a 24-well polystyrene culture plate and air-dried. Four wells were used for each concentration. To each well, filtered seawater (2.0 mL, salinity 28) and six 2-day-old cyprids were added. The plates were kept in the dark at 25 °C for 48 h. The numbers of cyprids that attached, metamorphosed, died, and did not settle were counted under a microscope. Three or four trials were done for each concentration. Probit analysis was used to calculate the EC50 values.
Supplementary Materials
The following are available online at www.mdpi.com/1660-3397/15/7/203/s1; characterization of all compounds and 1H and 13C NMR data are available.
Acknowledgments
This research was supported by the Environment Research and Technology Development Fund (1RF-1601) Environmental Restoration and Conservation Agency of Japan.
Author Contributions
Y.H., I.S.N., J.S., and T.M. prepared compounds; Y.N. and E.Y. evaluated biological activities. T.U. conceived and designed the research and analyzed the experimental data. T.U., Y.N., and F.M. wrote the paper. All of the authors reviewed and approved the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Global Wind Energy Council. Global Wind Report Annual Market Update 2015; GWEC: Brussels, Belgium, 2015; Available online: http://www.gwec.net/wp-content/uploads/vip/GWEC-Global-Wind-2015-Report_April-2016_22_04.pdf (accessed on 20 April 2017).
- Fitridge, I.; Dempster, T.; Guenther, J.; de Nys, R. The impact and control of biofouling in marine aquaculture: A review. Biofouling 2012, 28, 649–669. [Google Scholar] [CrossRef] [PubMed]
- Champ, M.A. A review of organotin regulatory strategies, pending actions, related costs and benefits. Sci. Total Environ. 2000, 258, 21–71. [Google Scholar] [CrossRef]
- Flemming, H.-C. Reverse osmosis membrane biofouling. Exp. Therm. Fluid Sci. 1997, 14, 382–391. [Google Scholar] [CrossRef]
- Bhadury, P.; Wright, P.C. Exploitation of marine algae: Biogenic compounds for potential antifouling applications. Planta 2004, 219, 561–578. [Google Scholar] [CrossRef] [PubMed]
- Brooks, S.J.; Waldock, M. Copper Biocides in the Marine Environment. In Ecotoxicology of Antifouling Biocides; Arai, T., Harino, H., Ohji, M., Langston, W.J., Eds.; Springer: Tokyo, Japan, 2009; pp. 413–430. [Google Scholar]
- Horiguchi, T.; Shiraishi, H.; Shimizu, M.; Yamazaki, S.; Morita, M. Imposex in Japanese gastropods (Neogastropoda and Mesogastropoda): Effects of tributyltin and triphenyltin from antifouling paints. Mar. Pollut. Bull. 1995, 31, 402–405. [Google Scholar] [CrossRef]
- Shimasaki, Y.; Kitano, T.; Oshima, Y.; Inoue, S.; Imada, N.; Honjo, T. Tributyltin causes masculinization in fish. Environ. Toxicol. Chem. 2003, 22, 141–144. [Google Scholar] [CrossRef] [PubMed]
- McAllister, B.G.; Kime, D.E. Early life exposure to environmental levels of the aromatase inhibitor tributyltin causes masculinisation and irreversible sperm damage in zebrafish (Danio rerio). Aquat. Toxicol. 2003, 65, 309–316. [Google Scholar] [CrossRef]
- Weis, J.S.; Perlmutter, J. Effects of tributyltin on activity and burrowing behavior of the fiddler crab, Uca pugilator. Estuaries 1987, 10, 342–346. [Google Scholar] [CrossRef]
- Weis, J.S.; Kim, K. Tributyltin is a teratogen in producing deformities in limbs of the fiddler crab, Uca pugilator. Arch. Environ. Contam. Toxicol. 1988, 17, 583–587. [Google Scholar] [CrossRef] [PubMed]
- Terlizzi, A.; Delos, A.L.; Garaventa, F.; Faimali, M.; Gerace, S. Limited effectiveness of marine protected areas: Imposex in Hexaplex trunculus (Gastropoda, Muricidae) populations from Italian marine reserves. Mar. Pollut. Bull. 2004, 48, 188–192. [Google Scholar] [CrossRef] [PubMed]
- Evans, S.M. TBT or not TBT?: That is the question. Biofouling 1999, 14, 117–129. [Google Scholar] [CrossRef]
- Gibbs, P.E.; Bryan, G.W.J. Reproductive failure in populations of the dog-whelk, Nucella lapillus, caused by imposex induced by tributyltin from antifouling paints. Mar. Biol. Assoc. UK 1986, 66, 767–777. [Google Scholar] [CrossRef]
- Gibbs, P.E.; Bryan, G.W. TBT-induced imposex in neogastropod snails: Masculinization to mass extinction. In Tributyltin: Case Study of an Environmental Contaminant; de Mora, S.J., Ed.; Cambridge University Press: Cambridge, UK, 1996; pp. 212–236. [Google Scholar]
- Chambers, L.D.; Stokes, K.R.; Walsh, F.C.; Wood, R.J.K. Modern approaches to marine antifouling coatings. Surf. Coat. Technol. 2006, 201, 3642–3652. [Google Scholar] [CrossRef]
- Steinberg, P.D.; de Nys, R.; Kjelleberg, S. Chemical cues for surface colonization. J. Chem. Ecol. 2002, 28, 1935–1951. [Google Scholar] [CrossRef] [PubMed]
- Fusetani, N. Biofouling and antifouling. Nat. Prod. Rep. 2004, 21, 94–104. [Google Scholar] [CrossRef] [PubMed]
- Raveendran, T.V.; Limna Mol, V.P. Natural product antifoulants. Curr. Sci. 2009, 97, 508–520. [Google Scholar]
- Maréchal, J.-P.; Hellio, C. Challenges for the development of new non-toxic antifouling solutions. Int. J. Mol. Sci. 2009, 10, 4623–4637. [Google Scholar] [CrossRef] [PubMed]
- Qian, P.-Y.; Xu, Y.; Fusetani, N. Natural products as antifouling compounds: Recent progress and future perspectives. Biofouling 2009, 26, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Fusetani, N. Antifouling marine natural products. Nat. Prod. Rep. 2011, 28, 400–410. [Google Scholar] [CrossRef] [PubMed]
- White, D.E.; Stewart, I.C.; Grubbs, R.H.; Stoltz, B.M. The catalytic asymmetric total synthesis of elatol. J. Am. Chem. Soc. 2008, 130, 810–811. [Google Scholar] [CrossRef] [PubMed]
- Umezawa, T.; Oguri, Y.; Matsuura, H.; Yamazaki, S.; Suzuki, M.; Yoshimura, E.; Furuta, T.; Nogata, Y.; Serisawa, Y.; Matsuyama-Serisawa, K.; et al. Omaezallene from red alga Laurencia sp.: Structure elucidation, total synthesis, and antifouling activity. Angew. Chem. Int. Ed. 2014, 53, 3909–3912. [Google Scholar] [CrossRef] [PubMed]
- Okino, T.; Yoshimura, E.; Hirota, H.; Fusetani, N. New antifouling sesquiterpenes from four nudibranchs of the family Phyllidiidae. Tetrahedron 1996, 52, 9447–9454. [Google Scholar] [CrossRef]
- Nishikawa, K.; Nakahara, H.; Shirokura, Y.; Nogata, Y.; Yoshimura, E.; Umezawa, T.; Okino, T.; Matsuda, F. Total synthesis of 10-isocyano-4-cadinene and determination of and its absolute configuration. Org. Lett. 2010, 12, 904–907. [Google Scholar] [CrossRef] [PubMed]
- Nishikawa, K.; Nakahara, H.; Shirokura, Y.; Nogata, Y.; Yoshimura, E.; Umezawa, T.; Okino, T.; Matsuda, F. Total synthesis of 10-isocyano-4-cadinene and its stereoisomers and evaluations of antifouling activities. J. Org. Chem. 2011, 76, 6558–6573. [Google Scholar] [CrossRef] [PubMed]
- Nishikawa, K.; Umezawa, T.; Garson, M.J.; Matsuda, F. Confirmation of the configuration of 10-isothiocyanato-4-cadinene diastereomers through synthesis. J. Nat. Prod. 2012, 75, 2232–2235. [Google Scholar] [CrossRef] [PubMed]
- Gulavita, N.K.; De Silva, E.D.; Hagadone, M.R.; Karuso, P.; Scheuer, P.J.; Van Duyne, G.D.; Clardy, J. Nitrogenous bisabolene sesquiterpenes from marine invertebrates. J. Org. Chem. 1986, 51, 5136–5139. [Google Scholar] [CrossRef]
- Kassühlke, K.E.; Potts, B.C.M.; Faulkner, D.J. New nitrogenous sesquiterpenes from two Philippine nudibranchs, Phyllidia pustulosa and P. varicosa, and from a Palauan sponge, Halichondria cf. lendenfeldi. J. Org. Chem. 1991, 56, 3747–3750. [Google Scholar] [CrossRef]
- Kitano, Y.; Ito, T.; Suzuki, T.; Nogata, Y.; Shinshima, K.; Yoshimura, E.; Chiba, K.; Tada, M.; Sakaguchi, I. Synthesis and antifouling activity of 3-isocyanotheonellin and its analogues. J. Chem. Soc. Perkin Trans. 1 2002, 2251–2255. [Google Scholar] [CrossRef]
- Nogata, Y.; Kitano, Y.; Yoshimura, E.; Shinshima, K.; Sakaguchi, I. Antifouling activity of simple synthetic isocyanides against larvae of the barnacle Balanus amphitrite. Biofouling 2004, 20, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, T.; Wagatsuma, H.; Kominami, Y.; Nogata, Y.; Yoshimura, E.; Chiba, K.; Kitano, Y. Anti-barnacle activity of isocyanides derived from amino acids. Chem. Biodivers. 2016, 13, 1502–1510. [Google Scholar] [CrossRef] [PubMed]
- Grenouillat, N.; Vauzeilles, B.; Beau, J.-M. Lipid analogs of the nodulation factors using the Ugi/Passerini multicomponent reactions: Preliminary studies on the carbohydrate monomer. Heterocycles 2007, 73, 891–901. [Google Scholar]
- Tavecchia, P.; Trumtel, M.; Veyrières, A.; Sinay, P. Glycosidations with N-formylamino sugar: A new approach to 2′-deoxy-β-disaccharides. Tetrahedron Lett. 1989, 43, 2533–2536. [Google Scholar] [CrossRef]
- Kitano, Y.; Nogata, Y.; Matsumura, K.; Yoshimura, E.; Chiba, K.; Tada, M.; Sakaguchi, I. Design and synthesis of anti-barnacle active fluorescence-labeled probe compounds and direct observation of the target region in barnacle cypris larvae for dimethyl-isocyanoalkyl compounds. Tetrahedron 2005, 61, 9969–9973. [Google Scholar] [CrossRef]
- Fujiwara, S.; Akima, C.; Nogata, Y.; Yoshimura, E.; Chiba, K.; Kitano, Y. Bio-organic and anti-barnacle studies of fluorescence-labeled probe compounds against cyprids of barnacles. J. Exp. Mar. Biol. Ecol. 2013, 445, 88–92. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).