1. Introduction
Angiogenesis, regarded as a process of forming new blood vessels, is essential for tissue repair and organ growth. However, an imbalance in the process is also highly related to numerous malignant, inflammatory, ischemic, and infectious disorders [
1,
2]. A number of signaling networks have been studied in the regulation of angiogenesis, including PI3K/AKT signaling and RAS/MAPK signaling [
3,
4,
5]. These signalings are activated by growth factors or angiogenesis inducers such as fibroblast growth factor (FGF) and vascular endothelial growth factor (VEGF) through binding to their corresponding receptors. This binding initiates the phosphorylation of receptors, resulting in the activation of downstream signaling pathways and inducing the VEGF and HIF-1 expression, thereby stimulating the angiogenesis [
5,
6]. Thus, negative regulation of the interaction between growth factors and receptors may be an effective therapy strategy for the modulation of neovascularization.
Previously, the highly sulfated polysaccharides heparin and heparan sulfate extracted from animal tissues have been demonstrated to be the most potent anti-angiogenic agents due to an efficient binding to VEGF and FGF1 [
7]. Intriguingly, fucoidans, a set of sulfated polysaccharides extracted from brown algae, attract more attention for its effective regulation of angiogenesis. In a previous report, a fucoidan-like polysaccharide STPC2 was isolated from a brown alga
Sargassum thunbergii. STPC2 could significantly inhibited tube formation and migration of human umbilical vein vascular endothelial cells (HUVECs), but the underlying mechanism of anti-angiogenesis has not been elucidated well. In this study, we aimed to demonstrate its structural determinants and the underlying mechanism for its regulation on angiogenesis.
3. Discussion
In this study, we further studied the structure-dependent anti-angiogenesis activity of STPC2 and elucidate the possible underlying mechanism for its regulation on angiogenesis. Firstly, we successfully prepared its desulfated product STPC2-DeS, and analyze the sulfate content. Compared to that of the native polysaccharide, the sulfate content in STPC2-DeS decreased significantly from 27.8% to 2.4%. With the remove of sulfate, its molecular weight was also reduced to 44.5 kD, but the sugar composition in STPC2-DeS did not show any distinct alteration. These results suggested that the desulfation was almost successful and did not make significant structural changes in STPC2-DeS, which guaranteed the reliability of our following experiments.
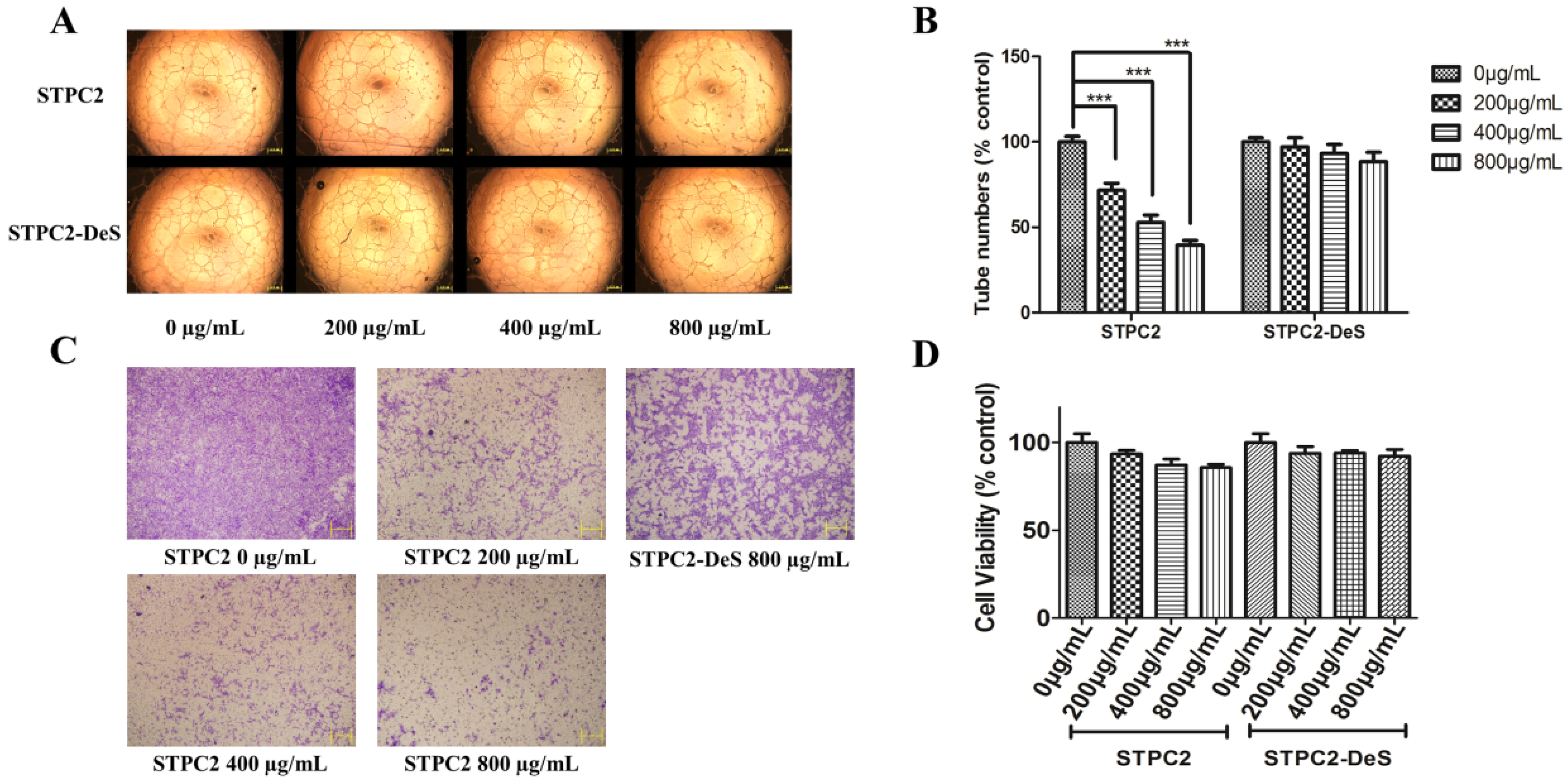
Considering the substantial sulfate group in fucoidan and other special chemical groups appearing in its structure, fucoidan has gained more and more interest in recent years. However, the details of structure-dependent activity have not been illustrated very well. Generally, it was considered that high-molecular-weight fucoidans (
Mw > 30 kD) with a high degree of sulfation exhibited a potent anti-angiogenic effect, whereas low-molecular-weight fucoidans (
Mw < 15 kD) promoted angiogenesis [
7]. However, the actual situation was more complicated for the structure–activity studies of fucoidans, not only for the difference of molecular weight, but also for the existence of sulfation pattern and degree of sulfation, and other specific hexuronic acid. In order to study the structural determinants in STPC2, we compared the effect of native and desulfated polysaccharides on the tube formation. The results indicated that STPC2 showed a significant inhibition on the capillary network formation in a dose-dependent manner, whereas STPC2-DeS failed to inhibit the tube formation even at the high concentration (800 µg/mL). Thus, it verified our hypothesis that sulfation in STPC2 had a critical role in its regulation on angiogenesis. In addition, in the transwell assay, an obvious interference of endothelial cells infiltrating into the lower well was observed in the STPC2 treatment. In contrast, STPC2-DeS did not show the inhibition of endothelial cell migration in transwell assay.
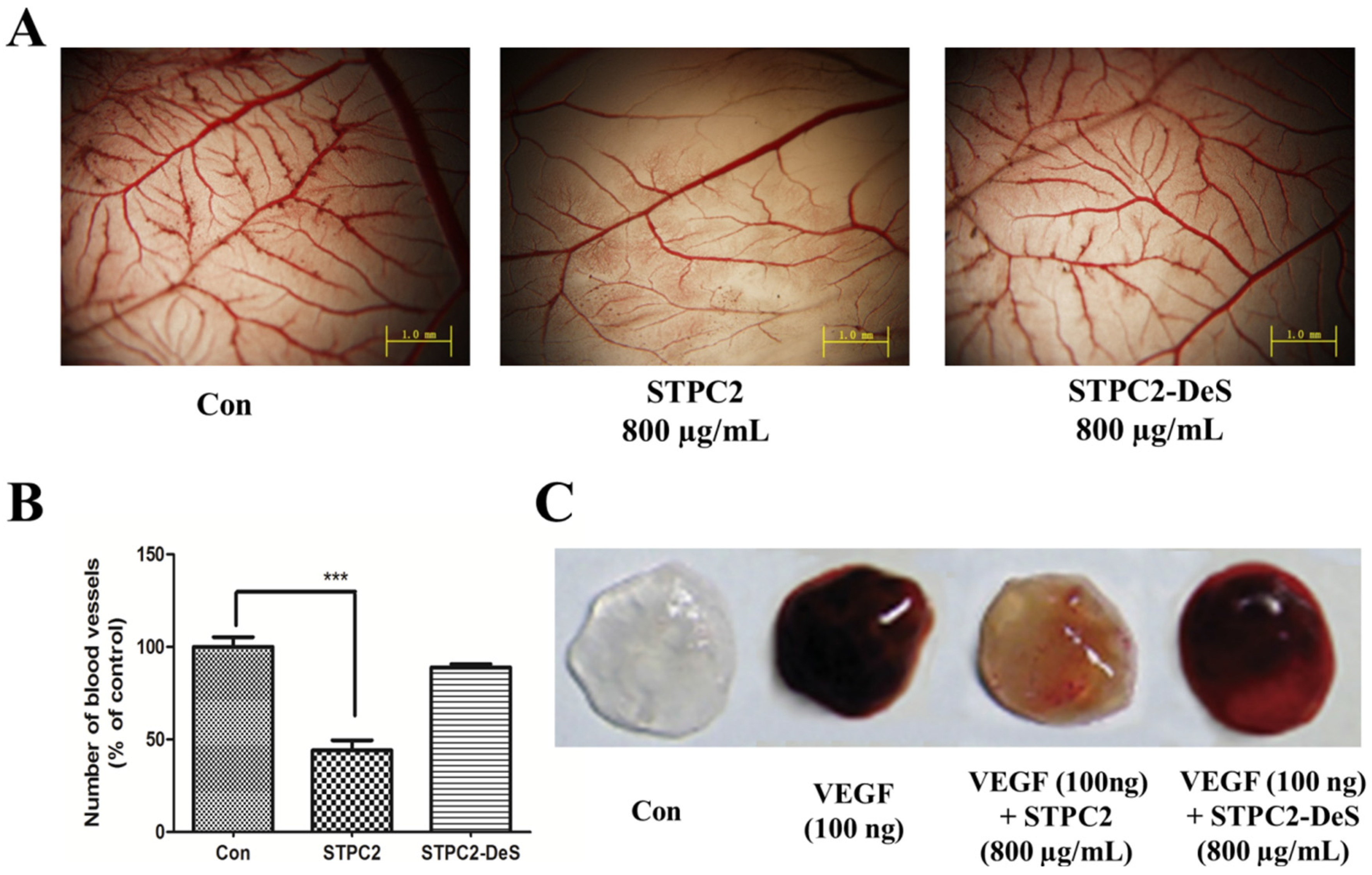
Given the visibility, accessibility, and rapid developmental growth of the chorioallantoic membrane (CAM) assay, an in vivo platform for experimentation, this approach has been widely used for the study of vascular development and angiogenesis, tumor growth and metastasis, drug distribution and toxicology [
17]. Because of its rapid vascular growth, CAM has been the good model to evaluate the effect of chemical compounds on growing vessels. Many pro- and anti-angiogenic compounds have been tested by quantifying the morphological alteration of the CAM vasculature. To further uncover the structure-dependent anti-angiogenic activity, we performed an in vivo CAM assay. It was found that STPC2 significantly inhibited the blood vessel density in the CAM assay. Meanwhile, we also applied a matrigel plug assay to verify the in vivo effect of STPC2. Matrigel, the basement membrane extract, has been extensively used in in vivo angiogenesis study. Matrigel was regarded to maintain its integrity after subcutaneously injected into mice and form a gel, the vessels could be formed via the stimulation of VEGF. In the matrigel plug assay, STPC2 was found to repress the VEGF-induced neovessel formation. On the contrary, STPC2-DeS did not exhibit the inhibition of new blood vessel growth and formation in the CAM assay and matrigel plug assay. Based on these multiple strategies of angiogenesis studies, these in vitro and in vivo results evidently demonstrated that sulfate groups in STPC2 played a crucial role in its anti-angiogenesis activity.
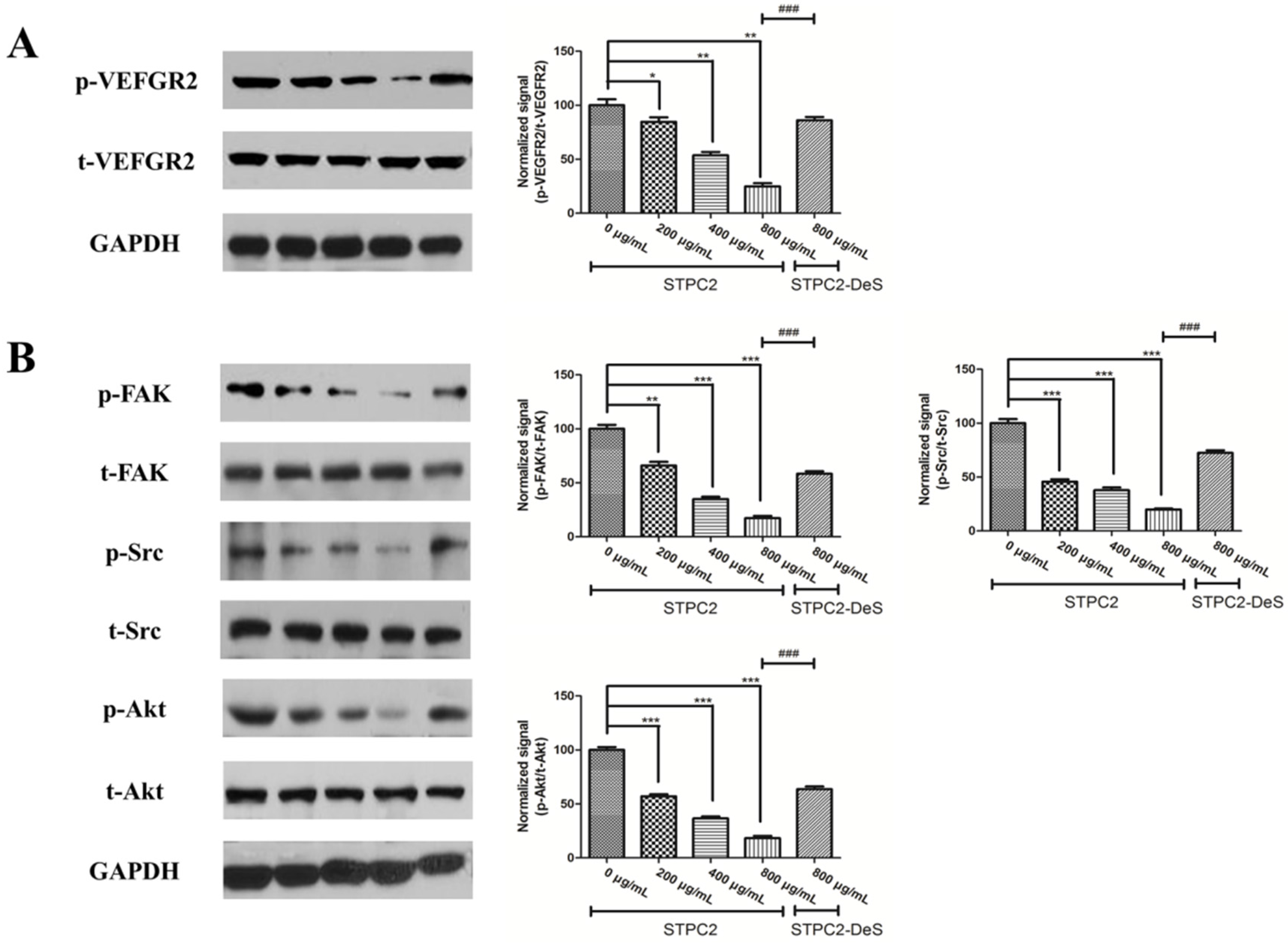
Angiogenic growth factors such as fibroblast growth factors (FGFs) and vascular endothelial growth factors (VEGFs) have been considered as the targets to inhibit deregulated blood vessel formation [
18]. In the previous study [
8], STPC2 could downregulate the expression of hypoxia-inducible-factor-1α (HIF-1α) and VEGF, which might impede vascular permeability. However, the molecular mechanism of STPC2 regulating the VEGF expression to affect the anti-angiogenic activity has been illustrated well.
The high-affinity cognate endothelial receptors for VEGF have also been identified. VEGFR2 is exclusively localized on endothelial cells during embryogenic development and regarded as a key role in endothelial cell differentiation and vasculogenesis [
19,
20]. These receptors possess intrinsic cytoplasmic enzymatic activity, which can be activated through ligand binding. The binding initiates receptor dimerization and autophosphorylation of tyrosine residues, and further provides docking sites for downstream signaling proteins [
21]. In order to uncover the molecular mechanism, we detected the alteration of VEGFR2 and its related downstream signaling molecules. Intriguingly, we found that its phosphorylation was significantly suppressed in the presence of STPC2, but not for STPC2-DeS. In addition, the experimental results also revealed that STPC2 repressed FAK, Src, and Akt activation. It substantially demonstrated that STPC2 exhibited the strong anti-angiogenic activity by the downregulation of VEGFR2 activity, and further modulated the activation of its downstream proteins.
In order to further elucidate the details of interaction between VEGF and VEGFR2 with STPC2, we analyzed the binding affinity of STPC2–VEGF and STPC2–VEGFR2 via SPR assay. In the SPR experiment, the
KD value of STPC2–VEGF (8.02 × 10
−6) was approximately three-fold less than that of STPC2–VEGFR2 (2.67 × 10
−5), which indicated that STPC2 showed a strong binding to VEGF compared to VEGFR2. In addition, the
KD value of STPC2-DeS to VEGF (2.92 × 10
−5) was approximately 3.6-fold larger than that of STPC2–VEGF (8.02 × 10
−6), which indicated the remove of sulfation to some extent reduced the affinity of STPC2 binding to VEGF. By reference to the protein database bank, the α-Fucose, α-
D-Mannose,
N-acetyl-
D-glucosamine and sulfation have been recognized as the ligands of VEGF (DOI: 10.2210/pdb4kzn/pdb). Substantial evidence has shown that β-
D-Mannose,
N-acetyl-
D-glucosamine and sulfation appeared as unique ligands in the crystal structures of VEGFR2 complex [
22,
23]. Hence, we thought the sulfated groups and α-Fucose in the structure of STPC2 might contribute to the interaction of STPC2 with VEGF. On the contrary, the interaction of STPC2 with its receptor VEGFR2 might function only based on sulfation groups. Thus, we speculated that the interaction of STPC2 with VEGF was more intensive than that of STPC2 with VEGFR2.
4. Material and Methods
4.1. Materials
Sargassum thunbergii (3.0 kg) was collected from Yantai, Shandong province (China). DEAE-cellulose 52 and Sephacryl S-300 HR were purchased from GE Healthcare Life Sciences (Shanghai, China). Other analytical chemical regents were supplied from Sinopharm (Shanghai, China).
4.2. Preparation of Desulfated STPC2
The native polysaccharide, STPC2, was prepared and characterized in the previous publication [
8]. Briefly, all the brown alga was extracted from the boiling-water and subjected to precipitation with ethanol. The obtained crude polysaccharide was further purified by CaCl
2 precipitation and chromatographyed on DEAE-cellulose and Sephacryl S-300 column to yield the homogeneous polysaccharide STPC2. After preliminarily identified the molecular weight, sugar composition, and degree of sulfation of the fresh-fractioned STPC2, the batch of the same physicochemical properties as previously reported was used to prepare its desulfated derivative. Briefly, STPC2 (500 mg) was dissolved in 50 mL of distilled water and loaded on a 732 sulphonic exchange resin column (H
+) (1.6 cm × 50 cm). The eluate was subjected to neutralization using pyridine and lyophilization. The subsequent process was reference to the previous report [
9]. Importantly, the final desulfated product was dialyzed against water with a membrane of small molecular weight cut off (MWCO, 0.1–0.5 kD). The retentate was vacuum-dried to give the desulfated derivative, named as STPC2-DeS.
4.3. Physicochemical Properties
High performance gel permeation chromatography (HPGPC) (Agilent, Shanghai, China) was used to determine the homogeneity and molecular weight of polysaccharides. The details were conducted as the previous report [
8]. Furthermore, sulfate content was estimated with elemental analysis, carried out on an Elementar Vario EL CUBE machine (Elementar, Germany).
4.4. Determination of Sugar Composition
Sugar composition analysis was performed as previously described [
8]. Briefly, native or desulfated polysaccharide was hydrolyzed with 2 M TFA at 125 °C for 2 h. After removing extra TFA with methanol, the hydrolyzates were reduced with sodium borohydride for 3 h at room temperature. After neutralized with AcOH and evaporated to dryness, the residue was converted into alditol acetates with Ac
2O for 1 h at 100 °C. The GC-MS analysis was referenced to the previous report [
9]. To quantify the uronic acid content in the structure, both were subjected to carboxyl-reduction referenced by the publication [
24]. The carboxyl reduced products were hydrolyzed and analyzed again as described above.
4.5. Infrared Spectroscopy (IR)
The IR spectrum was analyzed on a Nicolet 6700 FT-IR spectrometer (Thermo Scientific, Waltham, MA, USA). Desulfated polysaccharide (STPC2-DeS, 2 mg) was ground with KBr and then pressed into pellets, and further scanned from 4000 to 400 cm−1.
4.6. Cell Lines and Culture Conditions
Human umbilical vein vascular endothelial cells (HUVECs) were obtained from the Cell Bank of the Chinese Academy of Sciences, and cultured in RPMI 1640 medium (Gibco BRL, Gaithersburg, MA, USA) supplemented with 10% FBS, penicillin (50 U/mL), and streptomycin (50 µg/mL). All the cells were incubated at 37 °C with 5% CO2 in a humidified atmosphere.
4.7. Tube Formation Assay of HUVEC
The effect of STPC2 and STPC2-DeS on the formation of capillary tube-like structures was evaluated in a matrigel-based assay as previously reported [
25]. Briefly, a 96-well plate pre-coated with 50 μL matrigel each well was solidified at 37 °C for 1 h. Human umbilical vein vascular endothelial cells (HUVECs) were trypsinized, and supplemented with RPMI 1640 only or containing different doses of STPC2 or STPC2-DeS (0, 200, 400, and 800 µg/mL). The plate was incubated again for additional 24 h. The enclosed capillary network was photographed by an Olympus IX51 digital camera (Tokyo, Japan). Quantification of the inhibition of tube formation was analyzed by Image J software (NIH, Bethesda, MD, USA).
4.8. Cell Migration by Transwell Assay
The migration of HUVECs was evaluated by a transwell assay in a 24-well, 8-μm-pore size transwell plate (Costar, Cambridge, MA, USA). HUVECs (2 × 105 cells/well) were seeded in the upper chamber in 100 μL of serum free medium containing different concentrations of STPC2 (0, 200, 400, and 800 µg/mL) or STPC2-DeS (800 µg/mL). The lower chamber was maintained with same medium including 10% FBS. After incubated for 8 h, the migrated cells were stained with 0.1% crystal violet. While the non-migrated cells in the upper surface of the membrane were removed using a cotton swab. Migrated cells were then photographed by a microscope (Olympus BX510, Tokyo, Japan).
4.9. Chick Embryo Chorioallantoic Membrane Assay
The chorioallantoic membrane (CAM) assay, an in vivo platform for the study of vascular development and angiogenesis was conducted as previously described [
17]. Briefly, fertilized chick eggs were incubated at 37 °C in a humid atmosphere. After one week, the eggshell was cracked and peeled away over the airspace to open a small window. A 0.5 cm × 0.5 cm sheet of filter paper soaked with saline (control) or saline containing STPC2 (800 µg/mL) or STPC2-DeS (800 µg/mL) was covered on the chorioallantoic membrane. The eggs were sealed tightly and maintained in the incubator for another 48 h. Finally, the CAMs were taken to photograph with a microscope (Olympus BX510, Japan) after fixation (methanol:acetone = 1:1).
4.10. Matrigel Plug Assay
The experiment was carried out as described previously [
26]. C57/BL6 mice were got from Shanghai Laboratory Animal Center of the Chinese Academy of Sciences. All the protocols were carried out in accordance with the Guide for the Care and approved by the Animal Ethics Committee of Hospital. Briefly, we subcutaneously injected 0.5 mL of matrigel containing 100 ng VEGF into the ventral area of mice (
n = 5 per group). Control group mice were only injected with matrigel, whereas the experimental group mice were administrated with STPC2 (10 mg/kg) or STPC2-DeS (10 mg/kg) via tail vein injection every day for two weeks. After the mice were sacrificed, the matrigel plugs were excised and photographed.
4.11. Cell Viability Assay of STPC2 and STPC2-DeS
The cytotoxicity of STPC2 and STPC2-Des was evaluated on the HUVEC cells by the MTT assay. HUVEC cells (5 × 103 cells/ well) were seeded on 96-well plates in triplicate for 24 h. The medium was exchanged with different concentrations (0 µg/mL, 200 µg/mL, 400 µg/mL, 800 µg/mL) of STPC2 and STPC2-DeS, respectively, dissolved in corresponding medium for another 48 h. Subsequently, 20 µL of 5 mg/mL MTT was added into each well and incubated for 4 h at 37 °C. The medium was discarded and the formazan crystal was dissolved in 200 µL of DMSO. The absorption at 490 nm was recorded to calculate the cell viability ratio.
4.12. Western Blotting
HUVEC cells were serum-starved for overnight and then incubated with different concentrations of STPC2 (0, 200, 400, and 800 µg/mL) or STPC2-DeS (800 µg/mL) for 1 h. After that, the cells were lysed with RIPA buffer, according to the manufacturer’s instruction (Cell Signaling Technologies, Shanghai, China). The concentrations of proteins were determined by BCA protein assay kit (Beyotime, Haimen, Jiangsu, China). Equivalent amounts of proteins from different treatments were electrophoresised on SDS-PAGE gels and transferred to a polyvinylidene difluoride (PVDF) membrane (Bio-Rad, Hercules, CA, USA). Blots were performed with epitope-specific primary and secondary antibodies. Anti-VEGFR2 antibody, anti-VEGFR2 (phospho Y1054/1059) antibody, anti-FAK anbibody, anti-FAK (phospho Y397) antibody, anti-Src antibody, and anti-Src (phospho Y418) antibody were got from Abcam. Anti-Akt and phospho-Akt (Thr308) antibodies were purchased from Cell signaling technology. Finally, enhanced chemiluminescence (ECL) reagent was applied for signal detection. The data were quantified and normalized using image J software.
4.13. Surface Plasmon Resonance Analysis
To further uncover the details of interference of STPC2 with the interaction between VEGF and VEGFR2, surface plasmon resonance (SPR) analysis was used to study their respective affinities. The SPR experiment was carried out as previous report [
27]. Briefly, proteins (VEGF or VEGFR2) were immobilized on a CM5 sensor chip by the amine coupling method [
28]. For interaction determination, different concentrations of STPC2 or STPC2-DeS (50, 100, 200, 400, 800, and 1000 µg/mL) were injected into to the chips. All procedures were performed in HBS-EP (0.15 M NaCl, 0.01 M HEPES, 3 mM EDTA and 0.005% surfactant P20, pH 7.4) running buffer. Eventually, kinetic parameters were analyzed by BIACORE T200 Evaluation Software Version 1.0 (GE Healthcare, Uppsala, Sweden).
4.14. Statistical Analysis
All data were expressed as means ± S.D. p-value of the difference between groups was measured using Student’s t test for pair comparison or one-way ANOVA for multiple comparisons. * p < 0.05, ** p < 0.01, and *** p < 0.001 were considered statistically significant.