Sulfated Seaweed Polysaccharides as Multifunctional Materials in Drug Delivery Applications
Abstract
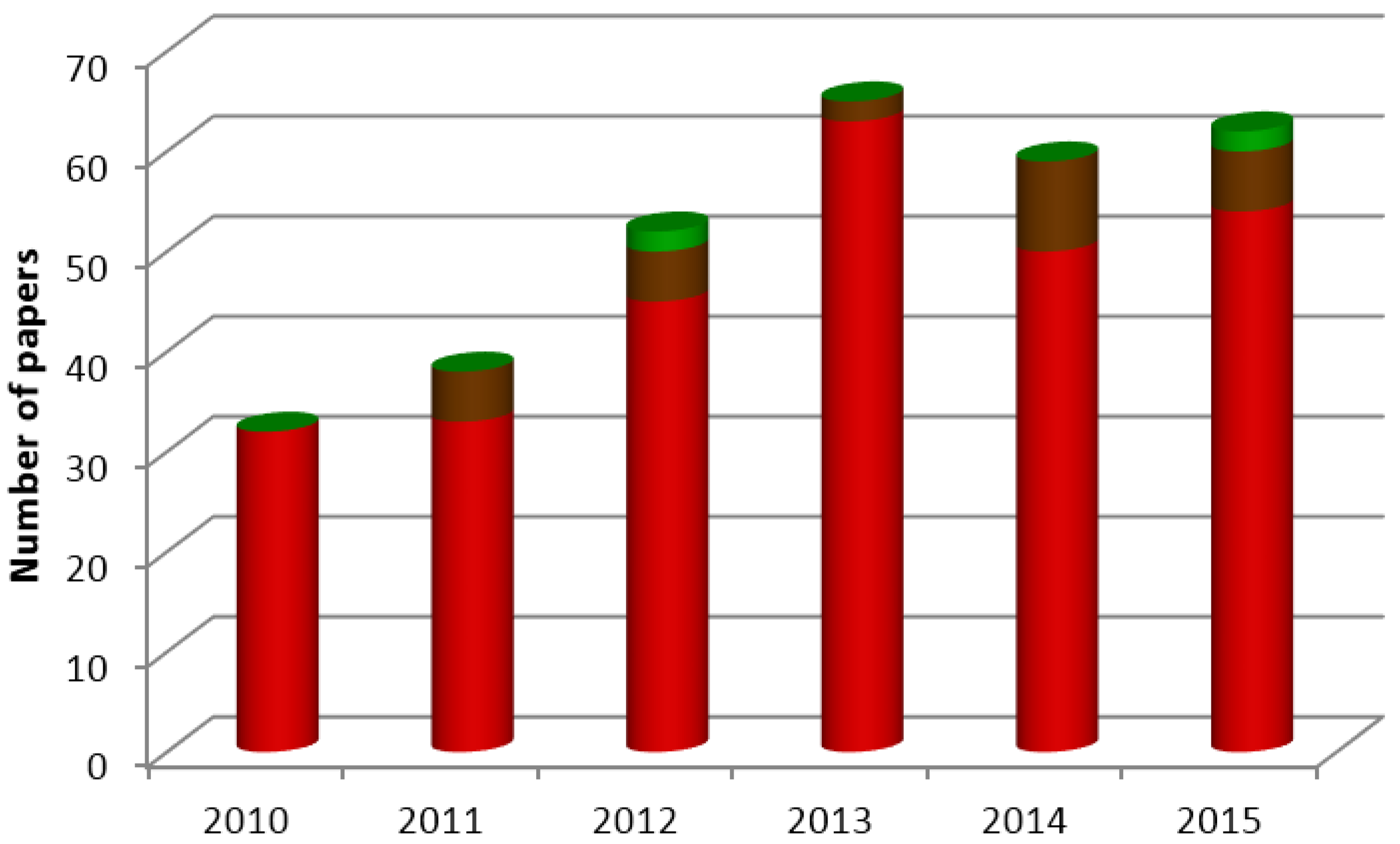
:1. Introduction
2. Carrageenan: Sulfated Polysaccharide of Red Seaweeds
2.1. Origin, Extraction and Processing
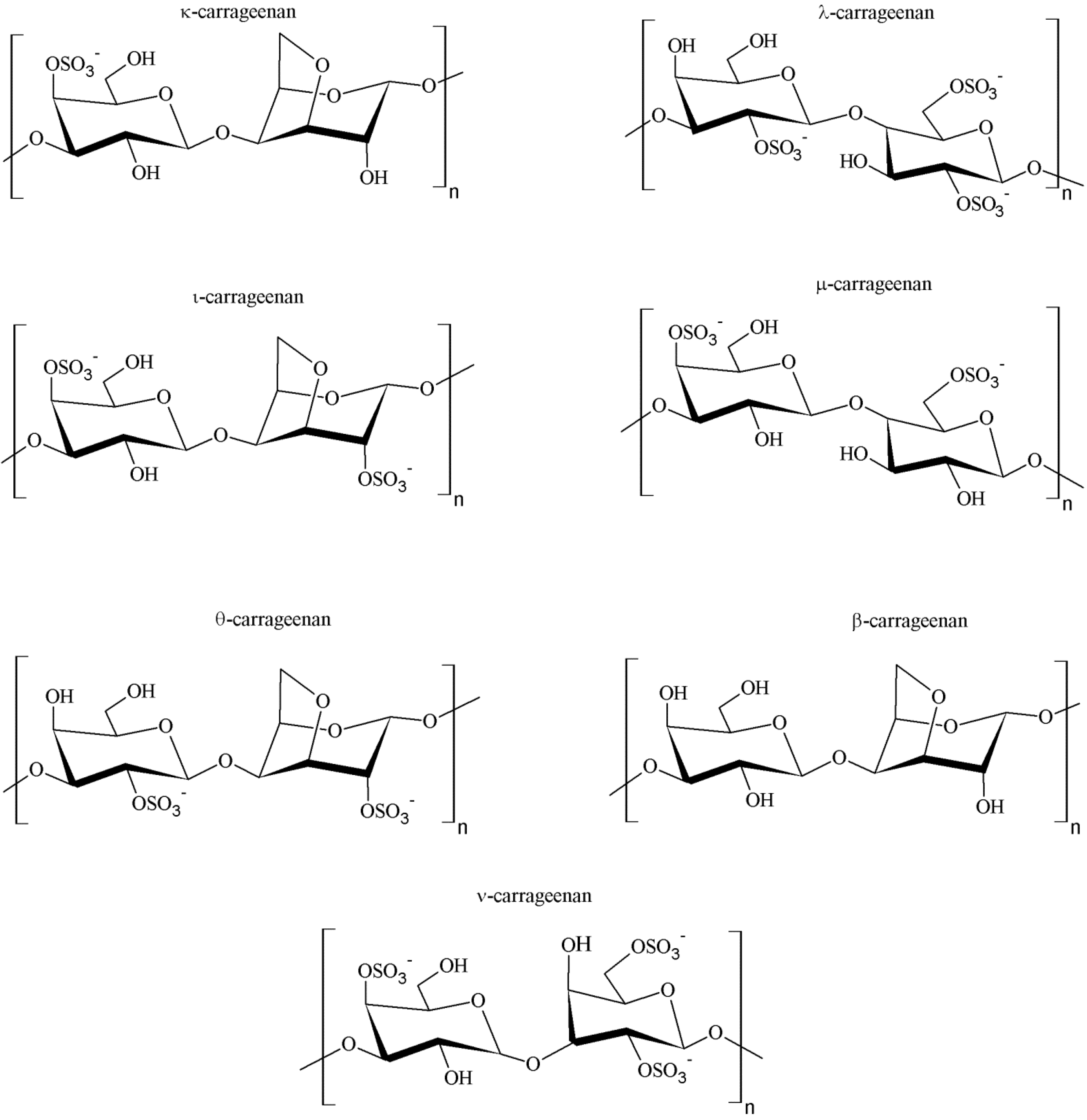
2.2. Chemical Structure and Types of Carrageenan
2.3. Physicochemical Properties
2.4. Biological Activity
3. Fucoidan: Sulfated Polysaccharide of Brown Seaweeds
3.1. Origin, Extraction and Processing
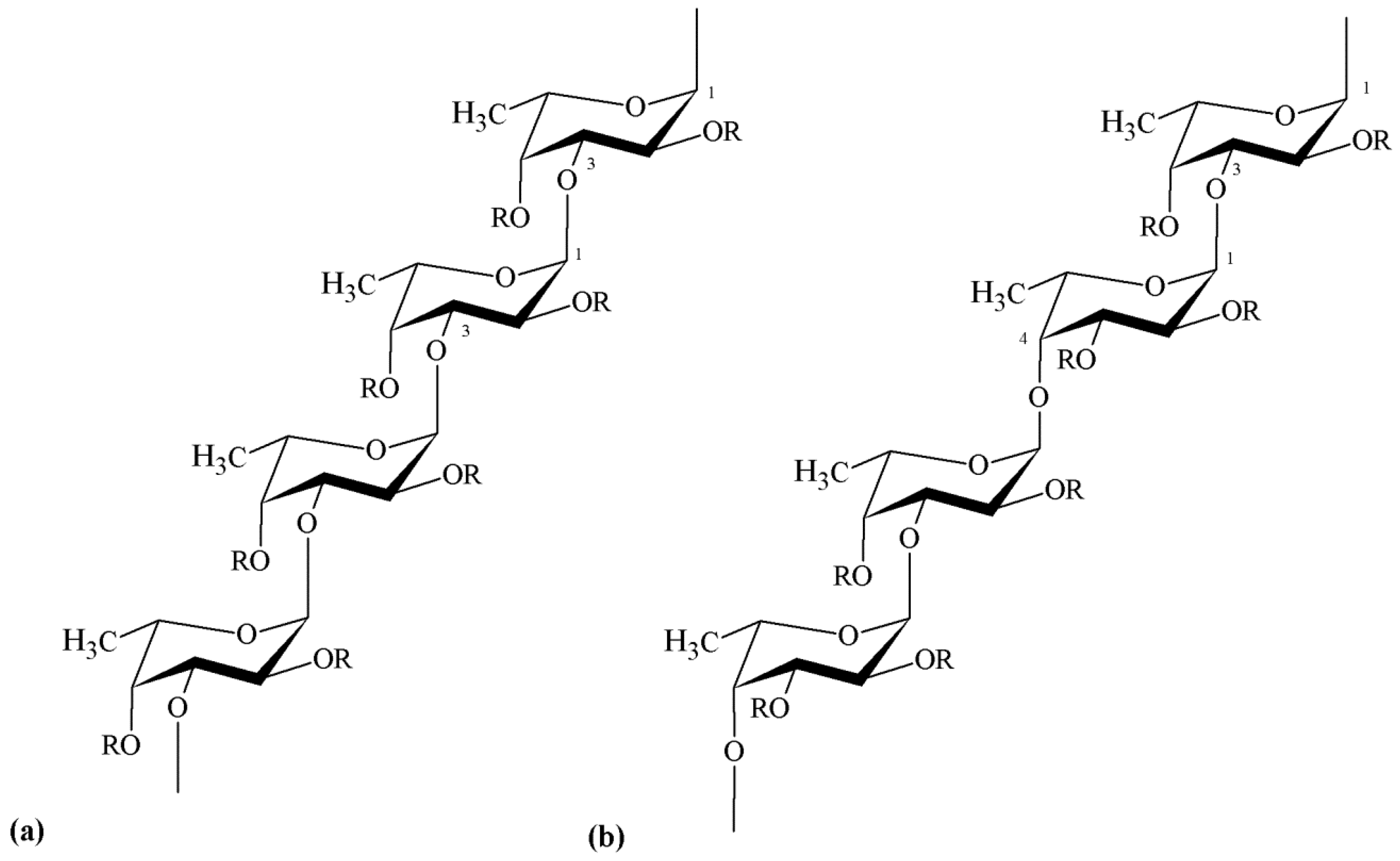
3.2. Chemical Structure
3.3. Physicochemical Properties
3.4. Biological Activity
4. Ulvan: Sulfated Polysaccharide of Green Seaweeds
4.1. Origin, Extraction and Processing
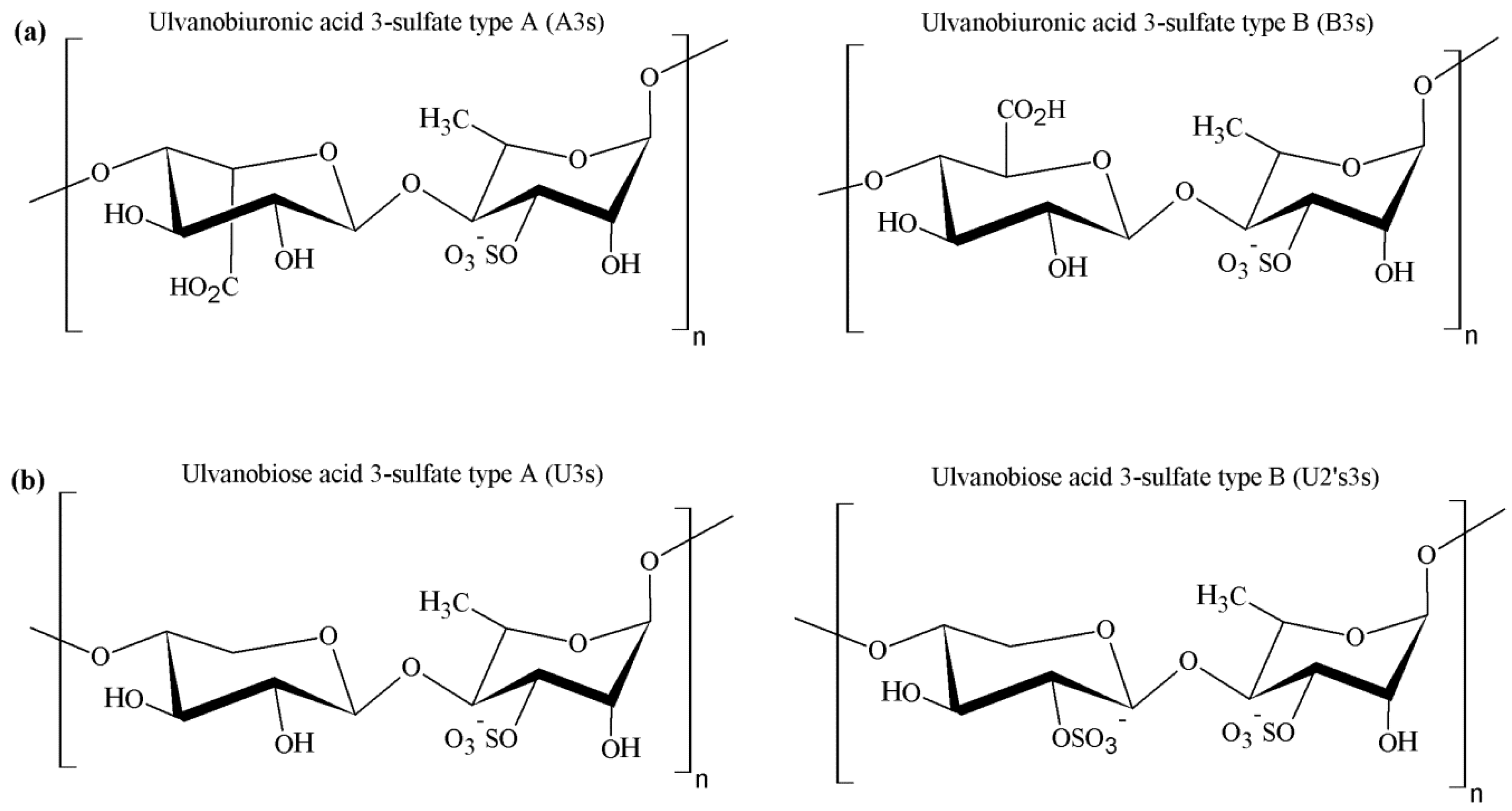
4.2. Chemical Structure
4.3. Physicochemical Properties
4.4. Biological Activity
5. Drug Delivery Systems Based on Seaweed Sulfated Polysaccharides
5.1. Nano and Microparticles
5.1.1. Nanoparticles
5.1.2. Microparticles
5.2. Hydrogel and Beads
5.3. Other Drug Delivery Systems
6. The Application of Sulfated Polysaccharides in Targeted Drug Delivery
7. Conclusions
Acknowledgments
Conflicts of Interest
References
- Percival, E. The polysaccharides of green, red and brown seaweeds: Their basic structure, biosynthesis and function. Br. Phycol. J. 1979, 14, 103–117. [Google Scholar] [CrossRef]
- McHugh, D.J. A Guide to the Seaweed Industry; FAO Fisheries Technical Paper 441; Food and Agriculture Organization of the United Nations: Rome, Italy, 2003. [Google Scholar]
- Mclachlan, J. Macroalgae (seaweeds): Industrial resources and their utilization. Plant Soil 1985, 157, 137–157. [Google Scholar] [CrossRef]
- Dai-Hung, N.; Se-Kwon, K. Sulfated polysaccharides as bioactive agents from marine algae. Int. J. Biol. Macromol. 2013, 62, 70–75. [Google Scholar]
- Wijesekara, I.; Pangestuti, R.; Se-Kwon, K. Biological activities and potential health benefits of sulfated polysaccharides derived from marine algae. Carbohydr. Polym. 2011, 84, 14–21. [Google Scholar] [CrossRef]
- Silva, T.H.; Alves, A.; Popa, E.G.; Reys, L.L.; Gomes, M.E.; Sousa, R.A.; Silva, S.S.; Mano, J.F.; Reis, R.L. Marine algae sulfated polysaccharides for tissue engineering and drug delivery approaches. Biomatter 2012, 2, 278–289. [Google Scholar] [CrossRef] [PubMed]
- Carragenan Properties and Specifications. Available online: http://www.webcitation.org/6eT9TLtpS (accessed on 12 January 2016).
- Necas, J.; Bartosikova, L. Carrageenan: A review. Vet. Med. 2013, 58, 187–205. [Google Scholar]
- Li, L.; Ni, R.; Shao, Y.; Mao, S. Carrageenan and its applications in drug delivery. Carbohydr. Polym. 2014, 103, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Rowe, R.C. Handbook of Pharmaceutical Excipients; Pharmaceutical Press: London, UK, 2006; pp. 124–126. [Google Scholar]
- Fucoidan Product. Available online: http://www.webcitation.org/6eTAI2JT1 (accessed on 12 January 2016).
- Descamps, V.; Colin, S.; Lahaye, M.; Jam, M.; Richard, C.; Potin, P.; Barbeyron, T.; Yvin, J.C.; Kloareg, B. Isolation and culture of a marine bacterium degrading the sulfated fucans from marine brown algae. Mar. Biotechnol. 2006, 8, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Tsutomu, F.; Yusuke, S.; Shigeru, M.; Kazue, T.; Takashi, K.; Tomohiko, S.; Yoshinori, N.; Yoshinori, T. Fucoidan is the active component of Fucus vesiculosus that promotes contraction of fibroblast-populated collagen gels. Biol. Pharm. Bull. 2000, 23, 1180–1184. [Google Scholar]
- Duarte, M.E.R.; Cardoso, M.A.; Noseda, M.D.; Cerezo, A.S. Structural studies on fucoidans from the brown seaweed Sargassum stenophyllum. Carbohydr. Res. 2001, 333, 281–293. [Google Scholar] [CrossRef]
- Bakunina, I.I.; Nedashkovskaia, O.I.; Alekseeva, S.A.; Ivanova, E.P.; Romanenko, L.A.; Gorshkova, N.M.; Isakov, V.V.; Zviagintseva, T.N.; Mikhaĭlov, V.V. Degradation of fucoidan by the marine proteobacterium Pseudoalteromonas citrea. Mikrobiologiia 2002, 71, 49–55. [Google Scholar] [PubMed]
- Woo-Jung, K.; Sung-Min, K.; Hyun Guell, K.; Hye-Rim, O.; Kyung-Bok, L.; Yoo-Kyung, L.; Yong-Il, P. Purification and anticoagulant activity of a fucoidan from Korean Undaria pinnatifida sporophyll. Algae 2007, 22, 247–252. [Google Scholar]
- Marais, M.F.; Joseleau, J.P. A fucoidan fraction from Ascophyllum nodosum. Carbohydr. Res. 2001, 336, 155–159. [Google Scholar] [CrossRef]
- Albuquerque, I.R.L.; Queiroz, K.C.S.; Alves, L.G.; Santos, E.A.; Leite, E.L.; Rocha, H.A.O. Heterofucans from Dictyota menstrualis have anticoagulant activity. Braz. J. Med. Biol. Res. 2004, 37, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Bilan, M.I.; Zakharova, A.N.; Grachev, A.A.; Shashkov, A.S.; Nifantiev, N.E.; Usov, A.I. Polysaccharides of algae: 60. Fucoidan from the Pacific brown alga Analipus japonicus (Harv.) Winne (Ectocarpales, Scytosiphonaceae). Bioorg. Khim. 2007, 33, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Lahaye, M.; Robic, A. Structure and function properties of Ulvan, a polysaccharide from green seaweeds. Biomacromolecules 2007, 8, 1765–1774. [Google Scholar] [CrossRef] [PubMed]
- Huimin, Q.; Tingting, Z.; Quanbin, Z.; Zhien, L.; Zengqin, Z.; Ronge, X. Antioxidant activity of different molecular weight sulfated polysaccharides from Ulva pertusa Kjellm (Chlorophyta). J. Appl. Phycol. 2005, 17, 527–534. [Google Scholar]
- Siddhanta, A.K.; Goswami, A.M.; Ramavat, B.K.; Mody, K.H.; Mairh, O.P. Water soluble polysaccharides of marine algal species of Ulva (Ulvales, Chlorophyta) of Indian waters. Indian J. Mar. Sci. 2001, 30, 166–172. [Google Scholar]
- Therkelsen, G.H. Carrageenan. In Industrial Gums: Polysaccharides and Their Derivatives; Whistler, R.L., Bemiller, J.M., Eds.; Academic Press: West Lafayette, IN, USA, 1993; pp. 145–180. [Google Scholar]
- Thomas, W.R. Production, properties and uses of carrageenan. In Thickening and Gelling Agents for Food; Imeson, A., Ed.; Chapman and Hall: London, UK, 1997; pp. 45–59. [Google Scholar]
- Prajapati, V.D.; Maheriya, P.M.; Jani, G.K.; Solanki, H.K. Carrageenan: A natural seaweed polysaccharide and its applications. Carbohydr. Polym. 2014, 105, 97–112. [Google Scholar] [CrossRef] [PubMed]
- De Ruiter, G.A.; Rudolph, B. Carrageenan biotechnology. Trends Food Sci. Technol. 1997, 8, 389–395. [Google Scholar] [CrossRef]
- Hawkes, M.W. Reproductive strategies. In Biology of the Red Algae; Cole, K.M., Sheath, R.G., Eds.; Press Syndicate of the University of Cambridge: New York, NY, USA, 1990; pp. 455–476. [Google Scholar]
- Stanley, N. Production, properties and uses of carrageenan. In Production and Utilization of Products from Commercial Seaweeds; McHugh, D.J., Ed.; Food and Agriculture Organization of the United Nations: Rome, Italy, 1987; pp. 116–146. [Google Scholar]
- Normah, O.; Nazarifah, I. Production of semi-refined carrageenan from locally available red seaweed, Eucheuma cottonii on a laboratory scale. J. Trop. Agric. Food Sci. 2003, 31, 207–213. [Google Scholar]
- Hilliou, L. Hybrid carrageenan: Isolation, chemical structure and gel properties. In Advances in Food and Nutrition Research, Marine Carbohydrates: Fundamentals and Application, Part A; Se-Kwon, K., Ed.; Elsevier Science: Whaltman, MA, USA, 2014; pp. 17–44. [Google Scholar]
- Dewi, E.N.; Java, C. Characterization and quality of semi refined carrageenan (SCR) products from different coastal waters based on fourier transform infrared technique. J. Coast. Dev. 2012, 16, 25–31. [Google Scholar]
- Varadarajan, S.A.; Nazaruddin, R.; Arbakariya, A.; Mamot, S. Development of high yielding carragenan extraction method from Eucheuma cotonii using cellulase and Aspergillus niger. In Proceedings of Prosiding Seminar Kimia Bersama UKM-ITB VIII9, Bangi, Malaysia, 11 June 2009; pp. 461–469.
- Barbeyron, T.; Michel, G.; Potin, P.; Henrissat, B.; Kloareg, B. ι-Carrageenases constitute a novel family of glycoside hydrolases, unrelated to that of κ-carrageenases. J. Biol. Chem. 2000, 275, 35499–35505. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Pascual, N.; Alemán, M.C.; Gómez-Guillén, M.C.; Monteiro, M. Enzyme-assisted extraction of κ/ι-hybrid carrageenan from Mastocarpus stellatus for obtaining bioactive ingredients and their application for edible active films development. Food Funct. 2014, 5, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Rhein-Knudsen, N.; Ale, M.T.; Meyer, A.S. Seaweed hydrocolloid production: An update on enzyme assisted extraction and modification technologies. Mar. Drugs 2015, 13, 3340–3359. [Google Scholar] [CrossRef] [PubMed]
- Tuvikene, R.; Truus, K.; Vaher, M.; Kailas, T.; Martin, G.; Kersen, P. Extraction and quantification of hybrid carrageenans from the biomass of the red algae Furcellaria lumbricalis and Coccotylus truncatus. Proc. Estonian Acad. Sci. Chem. 2006, 55, 40–53. [Google Scholar]
- Lahaye, M. Developments on gelling algal galactans, their structure and physico-chemistry. J. Appl. Phycol. 2001, 13, 173–184. [Google Scholar] [CrossRef]
- Guangling, J.; Guangli, Y.; Junzeng, Z.; Ewart, H.S. Chemical structures and bioactivities of sulfated polysaccharides from marine algae. Mar. Drugs 2011, 9, 196–233. [Google Scholar]
- CP Kelco ApS, GENU® Carrageenan Book; CP Kelco—A Huber Company: Lille Skensved, Denmark, 2002; pp. 1–26.
- Thrimawithana, T.R.; Young, S.; Dunstan, D.E.; Alany, R.G. Texture and rheological characterization of kappa and iota carrageenan in the presence of counter ions. Carbohydr. Polym. 2010, 82, 69–77. [Google Scholar] [CrossRef]
- Venugopal, V. Polysaccharide from seaweed and microalgae. In Marine Polysaccharides: Food Applications; Zollo, S., Ed.; Taylor and Francis Group: Boca Raton, FL, USA, 2011; pp. 111–122. [Google Scholar]
- Lai, V.M.F.; Wong, P.A.L.; Li, C.Y. Effects of cation properties on sol-gel transition and gel properties of κ-carrageenan. J. Food Sci. 2000, 65, 1332–1337. [Google Scholar] [CrossRef]
- Jouanneau, D.; Guibet, M.; Boulenguer, P.; Mazoyer, J.; Smietana, M.; Helbert, W. New insights into the structure of hybrid κ-/µ-carrageenan and its alkaline conversion. Food Hydrocoll. 2010, 24, 452–461. [Google Scholar] [CrossRef]
- Van de Velde, F. Structure and function of hybrid carrageenans. Food Hydrocoll. 2008, 22, 727–734. [Google Scholar] [CrossRef]
- Hilliou, L.; Larotonda, F.D.S.; Abreu, P.; Ramos, A.M.; Sereno, A.M.; Gonçalves, M.P. Effect of extraction parameters on the chemical structure and gel properties of κ/ι-hybrid carrageenans obtained from Mastocarpus stellatus. Biomol. Eng. 2006, 23, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Bo, Y.; Guangli, Y.; Xia, Z.; Weina, R.; Guangling, J.; Lianhua, F.; Yuehua, W.; Guanhua, D.; Tiller, C.; Girouard, G.; et al. Structural characterisation and bioactivities of hybrid carrageenan-like sulphated galactan from red alga Furcellaria lumbricalis. Food Chem. 2011, 124, 50–57. [Google Scholar]
- Van de Velde, F.; Antipova, A.S.; Rollema, H.S.; Burova, T.V.; Grinberg, N.V.; Pereira, L.; Gilsenan, P.M.; Tromp, R.H.; Rudolph, B.; Grinberg, V.Y. The structure of κ/ι-hybrid carrageenans II. Coil-helix transition as a function of chain composition. Carbohydr. Res. 2005, 340, 1113–1129. [Google Scholar] [CrossRef] [PubMed]
- Eccles, R.; Meier, C.; Jawad, M.; Weinmüllner, R.; Grassauer, A.; Prieschl-Grassauer, E. Efficacy and safety of an antiviral iota-carrageenan nasal spray: A randomized, double-blind, placebo-controlled exploratory study in volunteers with early symptoms of the common cold. Respir. Res. 2010, 11, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhang, P.; Hao, C.; Zhang, X.-E.; Cui, Z.-Q.; Guan, H.-S. In vitro inhibitory effect of carrageenan oligosaccharide on influenza A H1N1 virus. Antivir. Res. 2011, 92, 237–246. [Google Scholar] [CrossRef] [PubMed]
- De S.F-Tischera, P.C.; Talarico, L.; Noseda, M.; Guimaraes, S.M.P.; Damonte, E.; Duarte, M. Chemical structure and antiviral activity of carrageenans from Meristiella gelidium against herpes simplex and dengue virus. Carbohydr. Polym. 2006, 63, 459–465. [Google Scholar]
- Yuan, H.; Song, J.; Li, X.; Li, N.; Dai, J. Immunomodulation and antitumor activity of kappa-carrageenan oligosaccharides. Cancer Lett. 2006, 243, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Zhou, G.; Sun, Y.; Xin, H.; Zhang, Y.; Li, Z.; Xu, Z. In vivo antitumor and immunomodulation activities of different molecular weight lambda-carrageenans from Chondrus ocellatus. Pharmacol. Res. 2004, 50, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Güven, K.C.; Özsoy, Y.; Ulutin, O.N. Anticoagulant, fibrinolytic and antiaggregant activity of carrageenans and alginic acid. Bot. Mar. 1991, 34, 429–432. [Google Scholar] [CrossRef]
- Carlucci, M.J.; Pujol, C.A.; Ciancia, M.; Noseda, M.D.; Matulewicz, M.C.; Damonte, E.B.; Cerezo, A.S. Antiherpetic and anticoagulant properties of carrageenans from the red seaweed Gigartina skottsbergii and their cyclized derivatives: Correlation between structure and biological activity. Int. J. Biol. Macromol. 1997, 20, 97–105. [Google Scholar] [CrossRef]
- Panlasigui, L.N.; Baello, O.Q.; Dimatangal, J.M.; Dumelod, B.D. Blood cholesterol and lipid-lowering effects of carrageenan on human volunteers. Asia Pac. J. Clin. Nutr. 2003, 12, 209–214. [Google Scholar] [PubMed]
- Pangestuti, R.; Kim, S. Biological activities of carrageenan. In Advances in Food and Nutrition Research; Se-Kwon, K., Ed.; Waltham, MA, USA, 2014; Volume 72, pp. 113–124. [Google Scholar]
- Bo, L.; Fei, L.; Xinjun, W.; Ruixiang, Z. Fucoidan: Structure and bioactivity. Molecules 2008, 13, 1671–1695. [Google Scholar]
- Holtkamp, A.D.; Kelly, S.; Ulber, R.; Lang, S. Fucoidans and fucoidanases-focus on techniques for molecular structure elucidation and modification of marine polysaccharides. Appl. Microbiol. Biotechnol. 2009, 82, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Vo, T.-S.; Kim, S.-K. Fucoidans as a natural bioactive ingredient for functional foods. J. Funct. Foods 2013, 5, 16–27. [Google Scholar] [CrossRef]
- Wijesinghe, W.A.J.P.; Jeon, Y. Biological activities and potential industrial applications of fucose rich sulfated polysaccharides and fucoidans isolated from brown seaweeds: A review. Carbohydr. Polym. 2012, 88, 13–20. [Google Scholar] [CrossRef]
- Fitton, J.H.; Stringer, D.N.; Karpiniec, S.S. Therapies from fucoidan: An update. Mar. Drugs 2015, 13, 5920–5946. [Google Scholar] [CrossRef] [PubMed]
- Usov, A.I.; Zelinski, N.D. Chemical structures of algal polysaccharides. In Functional Ingredients from Algae for Foods and Nutraceuticals; Domínguez, H., Ed.; Elsevier Science: Cambridge, UK, 2013; pp. 45–49. [Google Scholar]
- Bernardi, G.; Springer, F. Properties of highly purified fucan. J. Biol. Chem. 1962, 237, 75–80. [Google Scholar] [PubMed]
- Percival, E.G.V.; Ross, A.G. Fucoidin. Part I. The isolation and purification of fucoidin from brown seaweeds. J. Chem. Soc. 1950, 717–720. [Google Scholar] [CrossRef]
- Ponce, N.M.A.; Pujol, C.A.; Damonte, E.B. Fucoidans from the brown seaweed Adenocystis utricularis: Extraction methods , antiviral activity and structural studies. Carbohydr. Res. 2003, 338, 153–165. [Google Scholar] [CrossRef]
- Bilan, M.I.; Grachev, A.A.; Ustuzhanina, N.E.; Shashkov, A.S.; Nifantiev, N.E.; Usov, A.I. A highly regular fraction of a fucoidan from the brown seaweed Fucus distichus L. Carbohydr. Res. 2004, 339, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Chizhov, A.O.; Dell, A.; Morris, H.R.; Haslam, S.M.; McDowell, R.A.; Shashkov, A.S.; Nifant’ev, N.E.; Khatuntseva, E.A.; Usov, A.I. A study of fucoidan from the brown seaweed Chorda filum. Carbohydr. Res. 1999, 320, 108–119. [Google Scholar] [CrossRef]
- Xiangdong, Q.; Amarasekara, A.; Doctor, V. Effect of oversulfation on the chemical and biological properties of fucoidan. Carbohydr. Polym. 2006, 63, 224–228. [Google Scholar]
- Kusaykin, M.I.; Chizhov, A.O.; Grachev, A.A.; Alekseeva, S.A.; Bakunina, I.Y.; Nedashkovskaya, O.I.; Sova, V.V.; Zvyagintseva, T.N. A comparative study of specificity of fucoidanases from marine microorganisms and invertebrates. J. Appl. Phycol. 2006, 18, 369–373. [Google Scholar] [CrossRef]
- Skriptsova, A.V. Fucoidans of brown algae: Biosynthesis, localization, and physiological role in thallus. Russ. J. Mar. Biol. 2015, 41, 145–156. [Google Scholar] [CrossRef]
- Hahn, T.; Lang, S.; Ulber, R.; Muffler, K. Novel procedures for the extraction of fucoidan from brown algae. Process Biochem. 2012, 47, 1691–1698. [Google Scholar] [CrossRef]
- Ale, M.T.; Mikkelsen, J.D.; Meyer, A.S. Important determinants for fucoidan bioactivity: A critical review of structure-function relations and extraction methods for fucose-containing sulfated polysaccharides from brown seaweeds. Mar. Drugs 2011, 9, 2106–2130. [Google Scholar] [CrossRef] [PubMed]
- Bilan, M.I.; Grachev, A.A.; Ustuzhanina, N.E.; Shashkov, A.S.; Nifantiev, N.E.; Usov, A.I. Structure of a fucoidan from the brown seaweed Fucus evanescens. Carbohydr. Res. 2002, 337, 719–730. [Google Scholar] [CrossRef]
- Li, B.; Wei, X.J.; Sun, J.L.; Xu, S.Y. Structural investigation of a fucoidan containing a fucose-free core from the brown seaweed, Hizikia fusiforme. Carbohydr. Res. 2006, 341, 1135–1146. [Google Scholar] [CrossRef] [PubMed]
- Chevolot, L.; Mulloy, B.; Ratiskol, J.; Foucault, A.; Colliec-Jouault, S. A disaccharide repeat unit is the major structure in fucoidans from two species of brown algae. Carbohydr. Res. 2001, 330, 529–535. [Google Scholar] [CrossRef]
- Bilan, M.I.; Shashkov, A.S.; Usov, A.I. Structure of a sulfated xylofucan from the brown alga Punctaria plantaginea. Carbohydr. Res. 2014, 393, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Bilan, M.I.; Grachev, A.A.; Shashkov, A.S.; Nifantiev, N.E.; Usov, A.I. Structure of a fucoidan from the brown seaweed Fucus serratus L. Carbohydr. Res. 2006, 341, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Usui, T.; Asari, K.; Takashi, M. Isolation of highly purified “fucoidan” from Eisenia bicyclis and its anticoagulant and antitumor activities. Agric. Biol. Chem. 1980, 44, 1965–1966. [Google Scholar] [CrossRef]
- Xing, R.; Liu, S.; Yu, H.; Chen, X.; Qin, Y.; Li, K.; Li, P. Extraction and separation of fucoidan from Laminaria japonica with chitosan as extractant. Biomed. Res. Int. 2013, 1, 1–4. [Google Scholar]
- Colliec, S.; Boisson-Vidal, C.; Jozefonvicz, J. A low molecular weight fucoidan fraction from the brown seaweed Pelvetia canaliculata. Phytochemistry 1994, 35, 697–700. [Google Scholar] [CrossRef]
- Pomin, V.H. Fucanomics and galactanomics: Marine distribution, medicinal impact, conceptions, and challenges. Mar. Drugs 2012, 10, 793–811. [Google Scholar] [CrossRef] [PubMed]
- Zvyagintseva, T.N.; Shevchenko, N.M.; Chizhov, A.O.; Krupnova, T.N.; Sundukova, E.V.; Isakov, V.V. Water-soluble polysaccharides of some far-eastern brown seaweeds. Distribution, structure, and their dependence on the developmental conditions. J. Exp. Mar. Biol. Ecol. 2003, 294, 1–13. [Google Scholar] [CrossRef]
- Synytsya, A.; Copicová, J.; Woo, J.K.; Yong, P., II. Marine algal biotechnology. In Springer Handbook of Marine Biotechnology; Se-kwon, K., Ed.; Springer: London, UK; New York, NY, USA, 2015; pp. 1–8. [Google Scholar]
- Mulloy, B.; Ribeiro, A.C.; Aves, A.P.; Vieira, R.P.; Mourão, P.A.S. Sulfated fucans from Echinoderms have a regular tetrasaccharide repeating unit defined by specific patterns of sulfation at the 0–2 and 0–4 positions. J. Biol. Chem. 1994, 269, 22113–22123. [Google Scholar] [PubMed]
- Rupérez, P.; Ahrazem, O.; Leal, J.A. Potential antioxidant capacity of sulfated polysaccharides from the edible marine brown seaweed Fucus vesiculosus. J. Agric. Food Chem. 2002, 50, 840–845. [Google Scholar] [CrossRef] [PubMed]
- Black, W.A.P. The seasonal variation in the combined l-fucose content of the common British Laminariaceae and Fucaceae. J. Sci. Food Agric. 1954, 5, 445–448. [Google Scholar] [CrossRef]
- Soeda, S.; Ohmagari, Y.; Shimeno, H.; Nagamatsu, A. Preparation of oversulfated fucoidan fragments and evaluation of their antithrombotic activities. Thromb. Res. 1993, 72, 247–256. [Google Scholar] [CrossRef]
- Ellouali, M.; Boisson-Vidal, C.; Durand, P.; Jozefonvicz, J. Antitumor activity of low molecular weight fucans extracted from brown seaweed Ascophyllum nodosum. Anticancer Res. 1993, 13, 2011–2019. [Google Scholar] [PubMed]
- Taylor, P.; Nishino, T.; Aizu, Y.; Nagumo, T. The relationship between the molecular weight and the anticoagulant activity of two types of fucan sulfates from the brown seaweed Ecklonia kurome. Agric. Biol. Chem. 1991, 55, 791–796. [Google Scholar]
- Anastyuk, S.D.; Shevchenko, N.M.; Nazarenko, E.L.; Imbs, T.I.; Gorbach, V.I.; Dmitrenok, P.S.; Zvyagintseva, T.N. Structural analysis of a highly sulfated fucan from the brown alga Laminaria cichorioides by tandem MALDI and ESI mass spectrometry. Carbohydr. Res. 2010, 345, 2206–2212. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Jasso, R.M.; Mussatto, S.I.; Pastrana, L.; Aguilar, C.N.; Teixeira, J.A. Extraction of sulfated polysaccharides by autohydrolysis of brown seaweed Fucus vesiculosus. J. Appl. Phycol. 2013, 25, 31–39. [Google Scholar] [CrossRef]
- Nardella, A.; Boisson-vidal, C.; Blondin, C.; Durand, P. Anticoagulant low molecular weight fucans produced by radical process and ion exchange chromatography of high molecular weight fucans extracted from the brown seaweed Ascophyllum nodosum. Carbohydr. Res. 1996, 289, 201–208. [Google Scholar] [CrossRef]
- Silchenko, A.S.; Kusaykin, M.I.; Kurilenko, V.V.; Zakharenko, A.M.; Isakov, V.V.; Zaporozhets, T.S.; Gazha, A.K.; Zvyagintseva, T.N. Hydrolysis of fucoidan by fucoidanase isolated from the marine bacterium, Formosa algae. Mar. Drugs 2013, 11, 2413–2430. [Google Scholar] [CrossRef] [PubMed]
- Jae-Geun, K.; Kil-Suk, J.; Jin-Hee, P. Rheological properties of fucoidans from Laminaria religiosa, Sporophylls of Undaria pinnatifida, Hizikia fusiforme and Sagassum fulvellum in Korea. Korean J. Fish. Aquat. Sci. 1997, 30, 329–333. [Google Scholar]
- Tako, M. Rheological characteristics of fucoidan isolated from commercially cultured Cladosiphon okamuranus. Bot. Mar. 2003, 46, 465. [Google Scholar] [CrossRef]
- MyoungLae, C.; Won-Seok, C.; Sangguan, Y. Steady and dynamic shear rheology of fucoidan-buckwheat starch mixtures. Starch J. 2009, 61, 282–290. [Google Scholar]
- Rioux, L.; Turgeon, S.L.; Beaulieu, M. Rheological characterisation of polysaccharides extracted from brown seaweeds. J. Sci. Food Agric. 2007, 87, 1630–1638. [Google Scholar] [CrossRef]
- Do-Yeong, K.; Weon-Sun, S. Roles of fucoidan, an anionic sulfated polysaccharide on BSA-stabilized oil-in-water emulsion. Macromol. Res. 2009, 17, 128–132. [Google Scholar]
- Sezer, A.D.; Cevher, E.; Hatıpoğlu, F.; Oğurtan, Z.; Baş, A.L.; Akbuğa, J. Preparation of fucoidan-chitosan hydrogel and its application as burn healing accelerator on rabbits. Biol. Pharm. Bull. 2008, 31, 2326–2333. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.M.; Kim, J.-K.; Cho, T.-S. Applications of ophthalmic biomaterials embedded with fucoidan. J. Ind. Eng. Chem. 2012, 18, 1197–1201. [Google Scholar] [CrossRef]
- Kwak, J. Fucoidan as a marine anticancer agent in preclinical development. Mar. Drugs 2014, 12, 851–870. [Google Scholar] [CrossRef] [PubMed]
- Jung-Bum, L.; Hayashi, K.; Hashimoto, M.; Nakano, T.; Hayashi, T. Novel antiviral fucoidan from Sporophyll of Undaria pinnatifida (Mekabu). Chem. Pharm. Bull. 2004, 52, 1091–1094. [Google Scholar]
- Young, H.; Ho, M.; Park, C.; Jin, C.; Gi-Young, K.; Il-Whan, C.; Deuk, N.; Taek-Jeong, N.; Kyu, T.; Hyun, Y. Anti-inflammatory effects of fucoidan through inhibition of NF-κB, MAPK and Akt activation in lipopolysaccharide-induced BV2 microglia cells. Food Chem. Toxicol. 2011, 49, 1745–1752. [Google Scholar]
- Cumashi, A.; Ushakova, N.A.; Preobrazhenskaya, M.E.; D’Incecco, A.; Piccoli, A.; Totani, L.; Tinari, N.; Morozevich, G.E.; Berman, A.E.; Bilan, M.I.; et al. A comparative study of the anti-inflammatory, anticoagulant, antiangiogenic, and antiadhesive activities of nine different fucoidans from brown seaweeds. Glycobiology 2007, 17, 541–552. [Google Scholar] [CrossRef] [PubMed]
- Croci, D.O.; Cumashi, A.; Ushakova, N.A.; Preobrazhenskaya, M.E.; Piccoli, A.; Totani, L.; Ustyuzhanina, N.E.; Bilan, M.I.; Usov, A.I.; Grachev, A.A.; et al. Fucans, but not fucomannoglucuronans, determine the biological activities of sulfated polysaccharides from Laminaria saccharina brown seaweed. PLoS ONE 2011, 6, e17283. [Google Scholar] [CrossRef] [PubMed]
- Kuznetsova, T.A.; Besednova, N.N.; Mamaev, A.N.; Momot, A.P.; Shevchenko, N.M.; Zvyagintseva, T.N. Anticoagulant activity of fucoidan from brown algae Fucus evanescens of the Okhotsk Sea. Bull. Exp. Biol. Med. 2003, 136, 471–473. [Google Scholar] [CrossRef] [PubMed]
- Raghavendran, H.R.B.; Srinivasan, P.; Rekha, S. Immunomodulatory activity of fucoidan against aspirin-induced gastric mucosal damage in rats. Int. Immunopharmacol. 2011, 11, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Teng, H.; Yang, Y.; Wei, H.; Liu, Z.; Liu, Z.; Ma, Y.; Gao, Z.; Hou, L.; Zou, X. Fucoidan suppresses hypoxia-induced lymphangiogenesis and lymphatic metastasis in mouse hepatocarcinoma. Mar. Drugs 2015, 13, 3514–3530. [Google Scholar] [CrossRef] [PubMed]
- Senthilkumar, K.; Manivasagan, P.; Venkatesan, J.; Kim, S.-K. Brown seaweed fucoidan: Biological activity and apoptosis, growth signaling mechanism in cancer. Int. J. Biol. Macromol. 2013, 60, 366–374. [Google Scholar] [CrossRef] [PubMed]
- Hayden, H.S.; Blomster, J.; Maggs, C.A.; Silva, P.C.; Stanhope, M.J.; Waaland, J.R. Linnaeus was right all along: Ulva and Enteromorpha are not distinct genera. Eur. J. Phycol. 2003, 38, 277–294. [Google Scholar] [CrossRef]
- Fleurence, J. L’habilitation des algues en alimentation humaine: Le point sur la réglementation française. Ind. Aliment. Agric. 1991, 108, 501–502. [Google Scholar]
- Abd-El-Baky, H.H.; El-Baz, F.K.; El-Baroty, G.S. Potential biological properties of sulphated polysaccharides extracted from the macroalgae Ulva lactuca L. Acad. J. Cancer Res. 2009, 2, 1–11. [Google Scholar]
- Stadnik, M.J.; De Freitas, M.B. Algal polysaccharides as source of plant resistance inducers. Trop. Plant Pathol. 2014, 39, 111–118. [Google Scholar] [CrossRef]
- Qia, H.; Huang, L.; Liu, X.; Liu, D.; Zhang, Q.; Liu, S. Antihyperlipidemic activity of high sulfate content derivative of polysaccharide extracted from Ulva pertusa (Chlorophyta). Carbohydr. Polym. 2012, 87, 1637–1640. [Google Scholar] [CrossRef]
- Feng Wang, F.; Wang, X.; Liu, X.; Hou, Y.; Zhang, Q. Extraction of the polysaccharides from five algae and their potential antioxidant activity in vitro. Carbohydr. Polym. 2010, 82, 118–121. [Google Scholar]
- Leiro, J.M.; Castro, R.; Arranz, J.A.; Lamas, J. Immunomodulating activities of acidic sulphated polysaccharides obtained from the seaweed Ulva rigida C. Agardh. Int. Immunopharmacol. 2007, 7, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Wenjun, M.; Xiaoxue, Z.; Yi, L.; Huijuan, Z. Sulfated polysaccharides from marine green algae Ulva conglobata and their anticoagulant activity. J. Appl. Phycol. 2006, 18, 9–14. [Google Scholar]
- Hernández-Garibay, E.; Zertuche-González, J.A.; Pacheco-Ruíz, I. Isolation and chemical characterization of algal polysaccharides from the green seaweed Ulva clathrata (Roth) C. Agardh. J. Appl. Phycol. 2011, 23, 537–542. [Google Scholar] [CrossRef]
- Robic, A.; Sassi, J.F.; Dion, P.; Lerat, Y.; Lahaye, M. Seasonal variability of physicochemical and rheological properties of ulvan in two ulva species (chlorophyta) from the Brittany coast. J. Phycol. 2009, 45, 962–973. [Google Scholar] [CrossRef]
- Robic, A.; Sassi, J.F.; Lahaye, M. Impact of stabilization treatments of the green seaweed Ulva rotundata (Chlorophyta) on the extraction yield, the physico-chemical and rheological properties of ulvan. Carbohydr. Polym. 2008, 74, 344–352. [Google Scholar] [CrossRef]
- Percival, E.; Wold, J.K. The acid polysaccharide from the green seaweed Ulva lactuca. Part II. The xite of the ester sulphate. J. Chem. Soc. 1963, 5459–5468. [Google Scholar] [CrossRef]
- Yanomoto, M. Physicochemical studies on sulfated polysaccharides extracted from seaweeds at various temperatures. Agric. Biol. Chem. 1980, 44, 589–593. [Google Scholar] [CrossRef]
- Brading, J.W.E.; Georg-Plant, M.M.T.; Hardy, D.M. The polysaccharide from the alga Ulva lactuca. Purification, hydrolysis, and methylation of the polysaccharide. J. Chem. Soc. 1954, 319–324. [Google Scholar] [CrossRef]
- Sarker, S.D.; Latif, Z.; Gray, A.I. Natural products isolation. In Methods in Biotechnology; Sarker, S.D., Latif, Z., Gray, A.I., Eds.; Humana Press: Totowa, NJ, USA, 2005; pp. 1–26. [Google Scholar]
- Costa, C.; Alves, A.; Pinto, P.R.; Sousa, R.A.; Da Silva, E.A.B.; Reis, R.L.; Rodrigues, A.E. Characterization of ulvan extracts to assess the effect of different steps in the extraction procedure. Carbohydr. Polym. 2012, 88, 537–546. [Google Scholar] [CrossRef]
- Alves, A.; Caridade, S.G.; Mano, J.F.; Sousa, R.A.; Reis, R.L. Extraction and physico-chemical characterization of a versatile biodegradable polysaccharide obtained from green algae. Carbohydr. Res. 2010, 345, 2194–2200. [Google Scholar] [CrossRef] [PubMed]
- Paradossi, G.; Cavalieri, F.; Pizzoferrato, L.; Liquori, A.M. A physico-chemical study on the polysaccharide ulvan from hot water extraction of the macroalga Ulva. Int. J. Biol. Macromol. 1999, 25, 309–315. [Google Scholar] [CrossRef]
- Quemener, B.; Lahaye, M.; Bobin-Dubigeon, C. Sugar determination in ulvans by a chemical-enzymatic method coupled to high performance anion exchange chromatography. J. Appl. Phycol. 1997, 9, 179–188. [Google Scholar] [CrossRef]
- Chattopadhyay, K.; Mandal, P.; Lerouge, P.; Driouich, A.; Ghosal, P.; Ray, B. Sulphated polysaccharides from Indian samples of Enteromorpha compressa (Ulvales, Chlorophyta): Isolation and structural features. Food Chem. 2007, 104, 928–935. [Google Scholar] [CrossRef]
- Lahaye, M.; Axelos, M.A.V. Gelling properties of water-soluble polysaccharides from proliferating marine green seaweeds (Ulva spp.). Carbohydr. Polym. 1993, 22, 261–265. [Google Scholar] [CrossRef]
- Robic, A.; Gaillard, C.; Sassi, J.F.; Leral, Y.; Lahaye, M. Ultrastructure of ulvan: A polysaccharide from green seaweeds. Biopolymers 2009, 91, 652–664. [Google Scholar] [CrossRef] [PubMed]
- Robic, A.; Bertrand, D.; Sassi, J.F.; Lerat, Y.; Lahaye, M. Determination of the chemical composition of ulvan, a cell wall polysaccharide from Ulva spp. (Ulvales, Chlorophyta) by FT-IR and chemometrics. J. Appl. Phycol. 2009, 21, 451–456. [Google Scholar] [CrossRef]
- Alves, A.; Sousa, R.A.; Reis, R.L. Processing of degradable ulvan 3D porous structures for biomedical applications. J. Biomed. Mater. 2012, 101, 998–1006. [Google Scholar] [CrossRef] [PubMed]
- Barros, A.A.A.; Alves, A.; Nunes, C.; Coimbra, M.A.; Pires, R.A.; Reis, R.L. Carboxymethylation of ulvan and chitosan and their use as polymeric components of bone cements. Acta Biomater. 2013, 9, 9086–9097. [Google Scholar] [CrossRef] [PubMed]
- Robic, A.; Rondeau-Mouro, C.; Sassi, J.F.; Lerat, Y.; Lahaye, M. Structure and interactions of ulvan in the cell wall of the marine green algae Ulva rotundata (Ulvales, Chlorophyceae). Carbohydr. Polym. 2009, 77, 206–216. [Google Scholar] [CrossRef]
- Yaich, H.; Garna, H.; Besbes, S.; Barthélemy, J.-P.; Paquot, M.; Blecker, C.; Attia, H. Impact of extraction procedures on the chemical, rheological and textural properties of ulvan from Ulva lactuca of Tunisia coast. Food Hydrocoll. 2014, 40, 53–63. [Google Scholar] [CrossRef]
- Haug, A. The influence of borate and calcium on the gel formation of a sulfated polysaccharide from Ulva lactuca. Acta Chem. Scand. B 1976, 30, 562–566. [Google Scholar] [CrossRef] [PubMed]
- Lahaye, M.; Ray, B.; Baumberger, S.; Quemener, B.; Axelos, M.A.V. Chemical characterisation and gelling properties of cell wall polysaccharides from species of ulva (Ulvales, Chlorophyta). Hydrobiologia 1996, 326, 473–480. [Google Scholar] [CrossRef]
- Pengzhan, Y.; Li, N.; Liu, X.; Zhou, G.; Zhang, Q.; Li, P. Antihyperlipidemic effects of different molecular weight sulfated polysaccharides from Ulva pertusa (Chlorophyta). Pharmacol. Res. 2003, 48, 543–549. [Google Scholar] [CrossRef]
- Ivanova, V.; Rouseva, R.; Kolarova, M.; Serkedjieva, J.; Rachev, R.; Manolova, N. Isolation of a polysaccharide with antiviral effect from Ulva lactuca. Prep. Biochem. 1994, 24, 83–97. [Google Scholar] [CrossRef] [PubMed]
- Cassolato, J.E.F.; Noseda, M.D.; Pujol, C.A.; Pellizzari, F.M.; Damonte, E.B.; Duarte, M.E.R. Chemical structure and antiviral activity of the sulfated heterorhamnan isolated from the green seaweed Gayralia oxysperma. Carbohydr. Res. 2008, 343, 3085–3095. [Google Scholar] [CrossRef] [PubMed]
- Arsul, V.A.; Lahoti, S.R. Natural polysaccharides as pharmaceutical excipients. World J. Pharm. Res. 2014, 3, 3776–3790. [Google Scholar]
- Rajamma, A.; Yogesha, H.; Sateesha, S. Natural gums as sustained release carriers: Development of gastroretentive drug delivery system of ziprasidone HCl. DARU J. Pharm. Sci. 2012, 20. [Google Scholar] [CrossRef]
- Beneke, C.E.; Viljoen, A.M.; Hamman, J.H. Polymeric plant-derived excipients in drug delivery. Molecules 2009, 14, 2602–2620. [Google Scholar] [CrossRef] [PubMed]
- Ghanam, D.; Kleinebudde, P. Suitability of κ-carrageenan pellets for the formulation of multiparticulate tablets with modified release. Int. J. Pharm. 2011, 409, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Kranz, H.; Jürgens, K.; Pinier, M.; Siepmann, J. Drug release from MCC- and carrageenan-based pellets: Experiment and theory. Eur. J. Pharm. Biopharm. 2009, 73, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Ige, P.P.; Rajput, P.; Pardeshi, C.; Kawade, R.; Swami, B.; Mahajan, H.; Nerkar, P.; Belgamwar, V.; Surana, S.; Gattani, S. Development of pellets of nifedipine using HPMC K15 M and κ-carrageenan as mucoadhesive sustained delivery system and in vitro evaluation. Iran. Polym. J. 2013, 22, 911–921. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Y. Preparation of kappa-carrageenan-chitosan polyelectrolyte gel beads. Zhongguo Zhong Yao Za Zhi 2012, 37, 466–470. (In Chinese) [Google Scholar] [PubMed]
- Sankalia, M.G.; Mashru, R.C.; Sankalia, J.M.; Sutariya, V. Stability improvement of alpha-amylase entrapped in kappa-carrageenan beads: Physicochemical characterization and optimization using composite index. Int. J. Pharm. 2006, 312, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Ozsoy, Y.; Bergişadi, N. Preparation of mefenamic acid sustained release beads based on kappa-carrageenan. Boll. Chim. Farm. 1999, 139, 120–123. [Google Scholar]
- Mohamadnia, Z.; Zohuriaan-Mehr, M.J.; Kabiri, K.; Jamshidi, A.; Mobedi, H. Ionically cross-linked carrageenan-alginate hydrogel beads. J. Biomater. Sci. Polym. Ed. 2008, 19, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Mahdavinia, G.R.; Rahmani, Z.; Karami, S.; Pourjavadi, A. Magnetic/pH-sensitive κ-carrageenan/sodium alginate hydrogel nanocomposite beads: Preparation, swelling behavior, and drug delivery. J. Biomater. Sci. Polym. Ed. 2014, 25, 1891–1906. [Google Scholar] [CrossRef] [PubMed]
- Piyakulawat, P.; Praphairaksit, N.; Chantarasiri, N.; Muangsin, N. Preparation and evaluation of chitosan/carrageenan beads for controlled release of sodium diclofenac. AAPS PharmSciTech 2007, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.-H.; Choi, H.-K. Preparation of various solid-lipid beads for drug delivery of enrofloxacin. Drug Deliv. 2004, 11, 365–370. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, R.V.; Boppana, R.; Krishna Mohan, G.; Mutalik, S.; Kalyane, N.V. pH-responsive interpenetrating network hydrogel beads of poly(acrylamide)-G-carrageenan and sodium alginate for intestinal targeted drug delivery: Synthesis, in vitro and in vivo evaluation. J. Colloid Interface Sci. 2012, 367, 509–517. [Google Scholar] [CrossRef] [PubMed]
- Popa, E.G.; Gomes, M.E.; Reis, R.L. Cell delivery systems using alginate–carrageenan hydrogel beads and bibers for regenerative medicine applications. Biomacromolecules 2011, 12, 3952–3961. [Google Scholar] [CrossRef] [PubMed]
- Santo, V.E.; Frias, A.M.; Carida, M.; Cancedda, R.; Gomes, M.E.; Mano, J.F.; Reis, R.L. Carrageenan-based hydrogels for the controlled delivery of PDGF-BB in bone tissue engineering applications. Biomacromolecules 2009, 10, 1392–1401. [Google Scholar] [CrossRef] [PubMed]
- Rosas-Durazo, A.; Lizardi, J.; Higuera-Ciapara, I.; Argüelles-Monal, W.; Goycoolea, F.M. Development and characterization of nanocapsules comprising dodecyltrimethylammonium chloride and κ-carrageenan. Colloids Surf. B Biointerfaces 2011, 86, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Daniel-da-Silva, A.L.; Ferreira, L.; Gil, A.M.; Trindade, T. Synthesis and swelling behavior of temperature responsive κ-carrageenan nanogels. J. Colloid Interface Sci. 2011, 355, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, S.; da Costa, A.M.R.; Grenha, A. Chitosan/carrageenan nanoparticles: Effect of cross-linking with tripolyphosphate and charge ratios. Carbohydr. Polym. 2012, 89, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Grenha, A.; Gomes, M.E.; Rodrigues, M.; Santo, V.E.; Mano, J.F.; Neves, N.M.; Reis, R.L. Development of new chitosan/carrageenan nanoparticles for drug delivery applications. J. Biomed. Mater. Res. A 2010, 92, 1265–1272. [Google Scholar] [CrossRef] [PubMed]
- Dionísio, M.; Cordeiro, C.; Remuñán-López, C.; Seijo, B.; Rosa-da-Costa, A.M.; Grenha, A. Pullulan-based nanoparticles as carriers for transmucosal protein delivery. Eur. J. Pharm. Sci. 2013, 50, 102–113. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, S.; Cordeiro, C.; Seijo, B.; Remuñán-López, C.; Grenha, A. Hybrid nanosystems based on natural polymers as protein carriers for respiratory delivery: Stability and toxicological evaluation. Carbohydr. Polym. 2015, 123, 369–380. [Google Scholar] [CrossRef] [PubMed]
- Briones, A.V.; Sato, T. Encapsulation of glucose oxidase (GOD) in polyelectrolyte complexes of chitosan-carrageenan. React. Funct. Polym. 2010, 70, 19–27. [Google Scholar] [CrossRef]
- D’Souza, A.A.; Devarajan, P.V. Bioenhanced oral curcumin nanoparticles: Role of carbohydrates. Carbohydr. Polym. 2016, 136, 1251–1258. [Google Scholar] [CrossRef] [PubMed]
- Raman, M.; Devi, V.; Doble, M. Biocompatible ι-carrageenan-γ-maghemite nanocomposite for biomedical applications—Synthesis, characterization and in vitro anticancer efficacy. J. Nanobiotechnol. 2015, 13, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Devi, N.; Maji, T.K. Genipin crosslinked chitosan-κ-carrageenan polyelectrolyte nanocapsules for the controlled delivery of isoniazid. Int. J. Polym. Mater. 2010, 59, 828–841. [Google Scholar] [CrossRef]
- Tomoda, K.; Asahiyama, M.; Ohtsuki, E.; Nakajima, T.; Terada, H.; Kanebako, M.; Inagi, T.; Makino, K. Preparation and properties of carrageenan microspheres containing allopurinol and local anesthetic agents for the treatment of oral mucositis. Colloids Surf. B Biointerfaces 2009, 71, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Devi, N.; Maji, T.K. Microencapsulation of isoniazid in genipin-crosslinked gelatin-A-κ-carrageenan polyelectrolyte complex. Drug Dev. Ind. Pharm. 2010, 36, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Patil, R.T.; Speaker, T.J. Water-based microsphere delivery system for proteins. J. Pharm. Sci. 2000, 89, 9–15. [Google Scholar] [CrossRef]
- Bonferoni, M.C.; Chetoni, P.; Giunchedi, P.; Rossi, S.; Ferrari, F.; Burgalassi, S.; Caramella, C. Carrageenan-gelatin mucoadhesive systems for ion-exchange based ophthalmic delivery: In vitro and preliminary in vivo studies. Eur. J. Pharm. Biopharm. 2004, 57, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Gavini, E.; Bonferoni, M.C.; Rassu, G.; Sandri, G.; Rossi, S.; Salis, A.; Porcu, E.P.; Giunchedi, P. Engineered microparticles based on drug-polymer coprecipitates for ocular-controlled delivery of ciprofloxacin: Influence of technological parameters. Drug Dev. Ind. Pharm. 2015, 19, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Leong, K.H.; Chung, L.Y.; Noordin, M.I.; Onuki, Y.; Morishita, M.; Takayama, K. Lectin-functionalized carboxymethylated kappa-carrageenan microparticles for oral insulin delivery. Carbohydr. Polym. 2011, 86, 555–565. [Google Scholar] [CrossRef]
- Suzuki, S.; Lim, J.K. Microencapsulation with carrageenan-locust bean gum mixture in a multiphase emulsification technique for sustained drug release. J. Microencapsul. 1994, 11, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Bosio, V.E.; Cacicedo, M.L.; Calvignac, B.; León, I.; Beuvier, T.; Boury, F.; Castro, G.R. Synthesis and characterization of CaCO3-biopolymer hybrid nanoporous microparticles for controlled release of doxorubicin. Colloids Surf. B Biointerfaces 2014, 123, 158–169. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Ferreiro, A.; González Barcia, M.; Gil-Martínez, M.; Vieites-Prado, A.; Lema, I.; Argibay, B.; Blanco Méndez, J.; Lamas, M.J.; Otero-Espinar, F.J. In vitro and in vivo ocular safety and eye surface permanence determination by direct and magnetic resonance imaging of ion-sensitive hydrogels based on gellan gum and kappa-carrageenan. Eur. J. Pharm. Biopharm. 2015, 94, 342–351. [Google Scholar] [CrossRef] [PubMed]
- Selvakumaran, S.; Muhamad, I.I.; Abd Razak, S.I. Evaluation of kappa carrageenan as potential carrier for floating drug delivery system: Effect of pore forming agents. Carbohydr. Polym. 2016, 135, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Singh, A.; Singh, R. Polyvinyl pyrrolidone/carrageenan blend hydrogels with nanosilver prepared by gamma radiation for use as an antimicrobial wound dressing. J. Biomater. Sci. Polym. Ed. 2015, 26, 1269–1285. [Google Scholar] [CrossRef] [PubMed]
- Varghese, J.S.; Chellappa, N.; Fathima, N.N. Gelatin-carrageenan hydrogels: Role of pore size distribution on drug delivery process. Colloids Surf. B Biointerfaces 2014, 113, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Jonganurakkun, B.; Nodasaka, Y.; Sakairi, N.; Nishi, N. DNA-based gels for oral delivery of probiotic bacteria. Macromol. Biosci. 2006, 6, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Chenxi, L.; Chunyan, L.; Zheshuo, L.; Qiuhong, L.; Xueying, Y.; Yu, L.; Lu, W. Enhancement in bioavailability of ketorolac tromethamine via intranasal in situ hydrogel based on poloxamer 407 and carrageenan. Int. J. Pharm. 2014, 474, 123–133. [Google Scholar]
- Hezaveh, H.; Muhamad, I.I. Impact of metal oxide nanoparticles on oral release properties of pH-sensitive hydrogel nanocomposites. Int. J. Biol. Macromol. 2012, 50, 1334–1340. [Google Scholar] [CrossRef] [PubMed]
- Popa, E.; Reis, R.; Gomes, M. Chondrogenic phenotype of different cells encapsulated in κ-carrageenan hydrogels for cartilage regeneration strategies. Biotechnol. Appl. Biochem. 2012, 59, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Cao, J.; Lei, D.L.; Cheng, X.B.; Zhou, H.Z.; Hou, R.; Zhao, Y.H.; Cui, F.Z. Application of nerve growth factor by gel increases formation of bone in mandibular distraction osteogenesis in rabbits. Br. J. Oral Maxillofac. Surg. 2010, 48, 515–519. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, C.V.; Moinuddin, Z.; Patil-Sen, Y.; Littlefield, R.; Hood, M. Lipid-hydrogel films for sustained drug release. Int. J. Pharm. 2015, 479, 416–421. [Google Scholar] [CrossRef] [PubMed]
- Pascalau, V.; Popescu, V.; Popescu, G.L.; Dudescu, M.C.; Borodi, G.; Dinescu, A.; Perhaita, I.; Paul, M. The alginate/k-carrageenan ratio’s influence on the properties of the cross-linked composite films. J. Alloys Compd. 2012, 536, 418–423. [Google Scholar] [CrossRef]
- Bajpai, S.K.; Dehariya, P.; Singh Saggu, S.P. Investigation of moisture sorption, permeability, cytotoxicity and drug release behavior of carrageenan/poly vinyl alcohol films. J. Macromol. Sci. A 2015, 52, 243–251. [Google Scholar] [CrossRef]
- Kianfar, F. Formulation development of a carrageenan based delivery system for buccal drug delivery using ibuprofen as a model drug. J. Biomater. Nanobiotechnol. 2011, 2, 582–595. [Google Scholar] [CrossRef]
- Khan, S.; Boateng, J.S.; Mitchell, J.; Trivedi, V. Formulation, characterisation and stabilisation of buccal films for paediatric drug delivery of omeprazole. AAPS PharmSciTech 2015, 16, 800–810. [Google Scholar] [CrossRef] [PubMed]
- Boateng, J.; Mani, J.; Kianfar, F. Improving drug loading of mucosal solvent cast films using a combination of hydrophilic polymers with amoxicillin and paracetamol as model drugs. Biomed. Res. Int. 2013, 2013, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Boateng, J.S.; Pawar, H.V.; Tetteh, J. Polyox and carrageenan based composite film dressing containing anti-microbial and anti-inflammatory drugs for effective wound healing. Int. J. Pharm. 2013, 441, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Pawar, H.V.; Tetteh, J.; Boateng, J.S. Preparation, optimisation and characterisation of novel wound healing film dressings loaded with streptomycin and diclofenac. Colloids Surf. B Biointerfaces 2013, 102, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Kianfar, F.; Chowdhry, B.Z.; Antonijevic, M.D.; Boateng, J.S. Novel films for drug delivery via the buccal mucosa using model soluble and insoluble drugs. Drug Dev. Ind. Pharm. 2012, 38, 1207–1220. [Google Scholar] [CrossRef] [PubMed]
- Naim, S.; Samuel, B.; Chauhan, B.; Paradkar, A. Effect of potassium chloride and cationic drug on swelling, erosion and release from kappa-carrageenan matrices. AAPS PharmSciTech 2004, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Panyoyai, N.; Bannikova, A.; Small, D.M.; Kasapis, S. Controlled release of thiamin in a glassy κ-carrageenan/glucose syrup matrix. Carbohydr. Polym. 2015, 115, 723–731. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Zaveri, T.; Ziegler, G.R.; Hayes, J.E. User preferences in a carrageenan-based vaginal drug delivery system. PLoS ONE 2013, 8, e54975. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, R.V.; Nagathan, V.V.; Biradar, P.R.; Naikawadi, A.A. Simvastatin loaded composite polyspheres of gellan gum and carrageenan: In vitro and in vivo evaluation. Int. J. Biol. Macromol. 2013, 57, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Kianfar, F.; Antonijevic, M.; Chowdhry, B.; Boateng, J.S. Lyophilized wafers comprising carrageenan and pluronic acid for buccal drug delivery using model soluble and insoluble drugs. Colloids Surf. B Biointerfaces 2013, 103, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Pawar, H.V.; Boateng, J.S.; Ayensu, I.; Tetteh, J. Multifunctional medicated lyophilised wafer dressing for effective chronic wound healing. J. Pharm. Sci. 2014, 103, 1720–1733. [Google Scholar] [CrossRef] [PubMed]
- Boateng, J.S.; Auffret, A.D.; Matthews, K.H.; Humphrey, M.J.; Stevens, H.N.E.; Eccleston, G.M. Characterisation of freeze-dried wafers and solvent evaporated films as potential drug delivery systems to mucosal surfaces. Int. J. Pharm. 2010, 389, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yao, W.; Wang, S.; Di, G.; Zheng, Q.; Chen, A. Preparation and characterization of fucoidan-chitosan nanospheres by the sonification method. J. Nanosci. Nanotechnol. 2014, 14, 3844–3849. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Wu, S.; Wu, J.Y.; Wen, D.; Mi, F. Preparation of fucoidan-shelled and genipin-crosslinked chitosan beads for antibacterial application. Carbohydr. Polym. 2015, 126, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-C.; Lam, U.-I. Chitosan/fucoidan pH sensitive nanoparticles for oral delivery system. J. Chin. Chem. Soc. 2011, 58, 779–785. [Google Scholar] [CrossRef]
- Lee, E.J.; Lim, K.H. Polyelectrolyte complexes of chitosan self-assembled with fucoidan: An optimum condition to prepare their nanoparticles and their characteristics. Korean J. Chem. Eng. 2014, 31, 664–675. [Google Scholar] [CrossRef]
- Lee, E.J.; Lim, K.-H. Formation of chitosan-fucoidan nanoparticles and their electrostatic interactions: Quantitative analysis. J. Biosci. Bioeng. 2016, 121, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Shao-Jung, W.; Trong-Ming, D.; Cheng-Wei, L.; Fwu-Long, M. Delivery of berberine using chitosan/fucoidan-taurine conjugate nanoparticles for treatment of defective intestinal epithelial tight junction barrier. Mar. Drugs 2014, 12, 5677–5697. [Google Scholar]
- Huang, Y.-C.; Li, R.-Y. Preparation and characterization of antioxidant nanoparticles composed of chitosan and fucoidan for antibiotics delivery. Mar. Drugs 2014, 12, 4379–4398. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-C.; Liu, T.J. Mobilization of mesenchymal stem cells by stromal cell-derived factor-1 released from chitosan/tripolyphosphate/fucoidan nanoparticles. Acta Biomater. 2012, 8, 1048–1056. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, A.C.; Bourbon, A.I.; Cerqueira, M.A.; Maricato, É.; Nunes, C.; Coimbra, M.A.; Vicente, A.A. Chitosan/fucoidan multilayer nanocapsules as a vehicle for controlled release of bioactive compounds. Carbohydr. Polym. 2015, 115, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.H.; Tang, D.W.; Hsieh, H.Y.; Wu, W.S.; Lin, B.X.; Chuang, E.Y.; Sung, H.W.; Mi, F.L. Nanoparticle-induced tight-junction opening for the transport of an anti-angiogenic sulfated polysaccharide across Caco-2 cell monolayers. Acta Biomater. 2013, 9, 7449–7459. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Jeong, D.; Na, K. Doxorubicin loading fucoidan acetate nanoparticles for immune and chemotherapy in cancer treatment. Carbohydr. Polym. 2013, 94, 850–856. [Google Scholar] [CrossRef] [PubMed]
- Dantas-Santos, N.; Almeida-Lima, J.; Vidal, A.A.J.; Gomes, D.L.; Oliveira, R.M.; Pedrosa, S.S.; Pereira, P.; Gama, F.M.; Rocha, H.A.O. Antiproliferative activity of fucan nanogel. Mar. Drugs 2012, 10, 2002–2022. [Google Scholar] [CrossRef] [PubMed]
- Sezer, A.D.; Akbuğa, J. Fucosphere-new microsphere carriers for peptide and protein delivery: Preparation and in vitro characterization. J. Microencapsul. 2006, 23, 513–522. [Google Scholar] [CrossRef] [PubMed]
- Sezer, A.D.; Akbuğa, J. The design of biodegradable ofloxacin-based core-shell microspheres: Influence of the formulation parameters on in vitro characterization. Pharm. Dev. Technol. 2012, 17, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Sezer, A.D.; Akbuğa, J. Comparison on in vitro characterization of fucospheres and chitosan microspheres encapsulated plasmid DNA (pGM-CSF): Formulation design and release characteristics. AAPS PharmSciTech 2009, 10, 1193–1199. [Google Scholar] [CrossRef] [PubMed]
- Sezer, A.D.; Cevher, E.; Hatipoǧlu, F.; Oǧurtan, Z.; Baş, A.L.; Akbuǧa, J. The use of fucosphere in the treatment of dermal burns in rabbits. Eur. J. Pharm. Biopharm. 2008, 69, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Hwang, S.; Lee, J. pH-responsive hydrogels from moldable composite microparticles prepared by coaxial electro-spray drying. Chem. Eng. J. 2011, 169, 348–357. [Google Scholar] [CrossRef]
- Nakamura, S.; Nambu, M.; Ishizuka, T.; Hattori, H.; Kanatani, Y.; Takase, B.; Kishimoto, S.; Amano, Y.; Aoki, H.; Kiyosawa, T.; et al. Effect of controlled release of fibroblast growth factor-2 from chitosan/fucoidan micro complex-hydrogel on in vitro and in vivo vascularization. J. Biomed. Mater. Res. A 2008, 85, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Dash, M.; Sangram, K.K.; Bartoli, C.; Morelli, A.; Smet, P.F.; Dubruel, P.; Chiellini, F. Biofunctionalization of ulvan scaffolds for bone tissue engineering. ACS Appl. Mater. Interfaces 2014, 6, 3211–3218. [Google Scholar] [CrossRef] [PubMed]
- Toskas, G.; Heinemann, S.; Heinemann, C.; Cherif, C.; Rolf-Dieter, H.; Roussis, V.; Hanke, T. Ulvan and ulvan/chitosan polyelectrolyte nanofibrous membranes as a potential substrate material for the cultivation of osteoblasts. Carbohydr. Polym. 2012, 89, 997–1002. [Google Scholar] [CrossRef] [PubMed]
- Toskas, G.; Rolf-Dieter, H.; Laourine, E.; Cherif, C.; Smyrniotopoulos, V.; Roussis, V. Nanofibers based on polysaccharides from the green seaweed Ulva rigida. Carbohydr. Polym. 2011, 84, 1093–1102. [Google Scholar] [CrossRef]
- Kikionis, S.; Ioannou, E.; Toskas, G.; Roussis, V. Electrospun biocomposite nanofibers of ulvan/PCL and ulvan/PEO. J. Appl. Polym. Sci. 2015, 132, 1–5. [Google Scholar] [CrossRef]
- Alves, A.; Pinho, E.D.; Neves, N.M.; Sousa, R.A.; Reis, R.L. Processing ulvan into 2D structures: Cross-linked ulvan membranes as new biomaterials for drug delivery applications. Int. J. Pharm. 2012, 426, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Raveendran, S.; Yoshida, Y.; Maekawa, T.; Kumar, D.S. Pharmaceutically versatile sulfated polysaccharide based bionano platforms. Nanomedicine 2013, 9, 605–626. [Google Scholar] [CrossRef] [PubMed]
- Mohanraj, V.J.; Chen, Y. Nanoparticles—A Review. Trop. J. Lournal Pharm. Res. 2006, 5, 561–573. [Google Scholar] [CrossRef]
- Nikam, A.P.; Mukesh, P.R.; Haudhary, S.P. Nanoparticles—An overview. J. Drug Deliv. Ther. 2014, 3, 1121–1127. [Google Scholar]
- Srikanth, K.; Gupta, V.R.M.; Manvi, S.R.; Devanna, N. Particulate carrier systems. Int. Res. J. Pharm. 2012, 3, 22–26. [Google Scholar]
- Lankalapalli, S.; Kolapalli, V.R.M. Polyelectrolyte complexes: A review of their applicability in drug delivery technology. Indian J. Pharm. Sci. 2009, 71, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Chan, H.-K.; Kwok, P.C.L. Production methods for nanodrug particles using the bottom-up approach. Adv. Drug Deliv. Rev. 2011, 63, 406–416. [Google Scholar] [CrossRef] [PubMed]
- Chiellini, F.; Piras, A.M.; Errico, C.; Chiellini, E. Micro/nanostructured polymeric systems for biomedical and pharmaceutical applications. Nanomedicine 2008, 3, 367–393. [Google Scholar] [CrossRef] [PubMed]
- Tapia, C.; Escobar, Z.; Costa, E.; Sapag-Hagar, J.; Valenzuela, F.; Basualto, C.; Gai, M.N.; Yazdani-Pedram, M. Comparative studies on polyelectrolyte complexes and mixtures of chitosan-alginate and chitosan-carrageenan as prolonged diltiazem clorhydrate release systems. Eur. J. Pharm. Biopharm. 2004, 57, 65–75. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, J.; Zhao, Z.; Li, J.; Zhang, R.; Yao, F. Formation and characterization of natural polysaccharide hollow nanocapsules via template layer-by-layer self-assembly. J. Colloid Interface Sci. 2012, 379, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Bulmer, C.; Margaritis, A.; Xenocostas, A. Encapsulation and controlled release of recombinant human erythropoietin from chitosan-carrageenan nanoparticles. Curr. Drug Deliv. 2012, 9, 527–537. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Wang, Q. Recent development of chitosan-based polyelectrolyte complexes with natural polysaccharides for drug delivery. Int. J. Biol. Macromol. 2014, 64, 353–367. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Bulmer, C.; Margaritis, A. Characterization of novel composite alginate chitosan-carrageenan nanoparticles for encapsulation of BSA as a model drug delivery system. Curr. Drug Deliv. 2015, 12, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Senthil, V.; Kumar, R.S.; Nagaraju, C.V.V.; Jawahar, N.; Ganesh, G.N.K.; Gowthamarajan, K. Design and development of hydrogel nanoparticles for mercaptopurine. J. Adv. Pharm. Technol. Res. 2010, 1, 334–337. [Google Scholar] [CrossRef] [PubMed]
- Dul, M.; Paluch, K.J.; Kelly, H.; Healy, A.M.; Sasse, A.; Tajber, L. Self-assembled carrageenan/protamine polyelectrolyte nanoplexes-Investigation of critical parameters governing their formation and characteristics. Carbohydr. Polym. 2015, 123, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Cheow, W.S.; Kiew, T.Y.; Hadinoto, K. Amorphous nanodrugs prepared by complexation with polysaccharides: Carrageenan versus dextran sulfate. Carbohydr. Polym. 2015, 117, 549–558. [Google Scholar] [CrossRef] [PubMed]
- Kurosaki, T.; Kitahara, T.; Kawakami, S.; Nishida, K.; Nakamura, J.; Teshima, M.; Nakagawa, H.; Kodama, Y.; To, H.; Sasaki, H. The development of a gene vector electrostatically assembled with a polysaccharide capsule. Biomaterials 2009, 30, 4427–4434. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-C.; Yang, Y.-T. Effect of basic fibroblast growth factor released from chitosan-fucoidan nanoparticles on neurite extension. J. Tissue Eng. Regen. Med. 2013. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.J.; Lim, K.-H. Relative charge density model on chitosan-fucoidan electrostatic interaction: Qualitative approach with element analysis. J. Biosci. Bioeng. 2015, 119, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, L.C.; Garcia, T.; Mori, M.; Sandri, G.; Bonferoni, M.C.; Finotelli, P.V.; Cinelli, L.P.; Caramella, C.; Cabral, L.M. Preparation and characterization of polysaccharide-based nanoparticles with anticoagulant activity. Int. J. Nanomed. 2012, 7, 2975–2986. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Bachelet-Violette, L.; Rouzet, F.; Beilvert, A.; Autret, G.; Maire, M.; Menager, C.; Louedec, L.; Choqueux, C.; Saboural, P.; et al. Ultrasmall superparamagnetic iron oxide nanoparticles coated with fucoidan for molecular MRI of intraluminal thrombus. Nanomedicine 2015, 10, 73–87. [Google Scholar] [CrossRef] [PubMed]
- Lira, M.C.B.; Santos-Magalhães, N.S.; Nicolas, V.; Marsaud, V.; Silva, M.P.C.; Ponchel, G.; Vauthier, C. Cytotoxicity and cellular uptake of newly synthesized fucoidan-coated nanoparticles. Eur. J. Pharm. Biopharm. 2011, 79, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Tengdelius, M.; Gurav, D.; Konradsson, P.; Påhlsson, P.; Griffith, M.; Oommen, O.P. Synthesis and anticancer properties of fucoidan-mimetic glycopolymer coated gold nanoparticles. Chem. Commun. 2015, 51, 8532–8535. [Google Scholar] [CrossRef] [PubMed]
- Alves, A.; Duarte, A.R.C.; Mano, J.F.; Sousa, R.A.; Reis, R.L. PDLLA enriched with ulvan particles as a novel 3D porous scaffold targeted for bone engineering. J. Supercrit. Fluids 2012, 65, 32–38. [Google Scholar] [CrossRef]
- Covis, R.; Vives, T.; Gaillard, C.; Benoit, M.; Benvegnu, T. Interactions and hybrid complex formation of anionic algal polysaccharides with a cationic glycine betaine-derived surfactant. Carbohydr. Polym. 2015, 121, 436–448. [Google Scholar] [CrossRef] [PubMed]
- Hoare, T.R.; Kohane, D.S. Hydrogels in drug delivery: Progress and challenges. Polymer 2008, 49, 1993–2007. [Google Scholar] [CrossRef]
- Purnama, A.; Aid-Launais, R.; Haddad, O.; Maire, M.; Mantovani, D.; Letourneur, D.; Hlawaty, H.; Le Visage, C. Fucoidan in a 3D scaffold interacts with vascular endothelial growth factor and promotes neovascularization in mice. Drug Deliv. Transl. Res. 2015, 5, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Sezer, A.D.; Hatipoğlu, F.; Oğurtan, Z.; Baş, A.L.; Akbuga, J. Evaluation of fucoidan-chitosan hydrogels on superficial dermal burn healing in rabbit: An in vivo study. J. Biotechnol. 2005, 118, S77–S77. [Google Scholar]
- Murakami, K.; Aoki, H.; Nakamura, S.; Nakamura, S.; Takikawa, M.; Hanzawa, M.; Kishimoto, S.; Hattori, H.; Tanaka, Y.; Kiyosawa, T.; et al. Hydrogel blends of chitin/chitosan, fucoidan and alginate as healing-impaired wound dressings. Biomaterials 2010, 31, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Kanno, K.; Akiyoshi, K.; Nakatsuka, T.; Watabe, Y.; Yukimura, S.; Ishihara, H.; Shin, N.; Kawasaki, Y.; Yano, D. Biocompatible hydrogel from a green tide-forming chlorophyta. J. Sustain. Dev. 2012, 5, 38–45. [Google Scholar] [CrossRef]
- Tran, T.T.-D.; Ngo, D.K.-P.; Vo, T.V.; Tran, P.H.-L. Design of sustained release tablet containing fucoidan. Curr. Drug Deliv. 2015, 12, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Bonferoni, M.C.; Rossi, S.; Ferrari, F.; Caramella, C. Development of oral controlled-release tablet formulations based on diltiazem-carrageenan complex. Pharm. Dev. Technol. 2004, 9, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Rosario, N.L.; Ghaly, E.S. Matrices of water-soluble drug using natural polymer and direct compression method. Drug Dev. Ind. Pharm. 2002, 28, 975–988. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V. Controlled-release tablets from carrageenans: Effect of formulation, storage and dissolution factors. Eur. J. Pharm. Biopharm. 2001, 51, 241–248. [Google Scholar] [CrossRef]
- Liang, L.; Linlin, W.; Jinfeng, L.; Shan, J.; Yitong, W.; Xin, Z.; Ding, J.; Yu, T.; Mao, S. Insights into the mechanisms of chitosan-anionic polymers-based matrix tablets for extended drug release. Int. J. Pharm. 2014, 476, 253–265. [Google Scholar]
- Elviri, L.; DeRobertis, S.; Baldassarre, S.; Bettini, R. Desorption electrospray ionization high-resolution mass spectrometry for the fast investigation of natural polysaccharide interactions with a model drug in controlled release systems. Rapid Commun. Mass Spectrom. 2014, 28, 1544–1552. [Google Scholar] [CrossRef] [PubMed]
- Bettini, R.; Bonferoni, M.C.; Colombo, P.; Zanelotti, L.; Caramella, C. Drug release kinetics and front movement in matrix tablets containing diltiazem or metoprolol/λ-carrageenan complexes. Biomed. Res. Int. 2014, 2014, 671532. [Google Scholar] [CrossRef] [PubMed]
- Buchholcz, G.; Kelemen, A.; Sovány, T.; Pintye-Hódi, K. Matrix tablets based on a carrageenan with the modified-release of sodium riboflavin 5’-phosphate. Pharm. Dev. Technol. 2015, 20, 676–683. [Google Scholar] [CrossRef] [PubMed]
- Jagdale, S.C.; Suryawanshi, V.M.; Pandya, S.V.; Kuchekar, B.S.; Chabukswar, A.R. Development of press-coated, floating-pulsatile drug delivery of lisinopril. Sci. Pharm. 2014, 82, 423–440. [Google Scholar] [CrossRef] [PubMed]
- Ito, I.; Ito, A.; Unezaki, S. Preparation and evaluation of gelling granules to improve oral administration. Drug Discov. Ther. 2015, 9, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Janaswamy, S.; Gill, K.L.; Campanella, O.H.; Pinal, R. Organized polysaccharide fibers as stable drug carriers. Carbohydr. Polym. 2013, 94, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Boateng, J.; Catanzano, O. Advanced therapeutic dressings for effective wound healing—A Review. J. Pharm. Sci. 2015, 104, 3653–3680. [Google Scholar] [CrossRef] [PubMed]
- Mayet, N.; Choonara, Y.E.; Kumar, P.; Tomar, L.K.; Tyagi, C.; Du Toit, L.C.; Pillay, V. A comprehensive review of advanced biopolymeric wound healing systems. J. Pharm. Sci. 2014, 103, 2211–2230. [Google Scholar] [CrossRef] [PubMed]
- Pielesz, A. Temperature-dependent FTIR spectra of collagen and protective effect of partially hydrolysed fucoidan. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2014, 118, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Sezer, A.D.; Hatipoglu, F.; Cevher, E.; Ogurtan, Z.; Bas, A.L.; Akbuga, J. Chitosan film containing fucoidan as a wound dressing for dermal burn healing: Preparation and in vitro/in vivo evaluation. APPS Pharm. Sci. Tech. 2007, 8, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Murakami, K.; Ishihara, M.; Aoki, H.; Nakamura, S.; Nakamura, S.I.; Yanagibayashi, S.; Takikawa, M.; Kishimoto, S.; Yokoe, H.; Kiyosawa, T.; et al. Enhanced healing of mitomycin C-treated healing-impaired wounds in rats with hydrosheets composed of chitin/chitosan, fucoidan, and alginate as wound dressings. Wound Repair Regen. 2010, 18, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Yanagibayashi, S.; Kishimoto, S.; Ishihara, M.; Murakami, K.; Aoki, H.; Takikawa, M.; Fujita, M.; Sekido, M.; Kiyosawa, T. Novel hydrocolloid-sheet as wound dressing to stimulate healing-impaired wound healing in diabetic db/db mice. Biomed. Mater. Eng. 2012, 22, 301–310. [Google Scholar] [PubMed]
- Manish, G.; Vimukta, S. Pulsatile drug delivery system: A review. Int. Res. J. Pharm. 2011, 1, 135–138. [Google Scholar]
- Pillai, O.; Panchagnula, R. Polymers in drug delivery. Curr. Opin. Chem. Biol. 2001, 5, 447–451. [Google Scholar] [CrossRef]
- Fahmy, T.M.; Fong, P.M.; Goyal, A.; Saltzman, W.M. Targeted for drug delivery. Mater. Today 2005, 8, 18–26. [Google Scholar] [CrossRef]
- Rani, K.; Paliwal, S. A review on targeted drug delivery: Its entire focus on advanced therapeutics and diagnostics. Sch. J. App. Med. Sci. 2014, 2, 328–331. [Google Scholar]
- Kang, B.; Opatz, T.; Landfester, K.; Wurm, F.R. Carbohydrate nanocarriers in biomedical applications: Functionalization and construction. Chem. Soc. Rev. 2015, 44, 8301–8325. [Google Scholar] [CrossRef] [PubMed]
- Medina, S.H.; Tekumalla, V.; Chevliakov, M.V.; Shewach, D.S.; Ensminger, W.D.; El-Sayed, M.E.H. N-acetylgalactosamine-functionalized dendrimers as hepatic cancer cell-targeted carriers. Biomaterials 2011, 32, 4118–4129. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Ziebell, M.R.; Prestwich, G.D. A hyaluronic acid-taxol antitumor bioconjugate targeted to cancer cells. Biomacromolecules 2000, 1, 208–218. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Yin, L.; Song, Y.; Tang, C.; Yin, C. Optimization of multifunctional chitosan-siRNA nanoparticles for oral delivery applications, targeting TNF-α silencing in rats. Acta Biomater. 2015, 17, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Fievez, V.; Plapied, L.; des Rieux, A.; Pourcelle, V.; Freichels, H.; Wascotte, V.; Vanderhaeghen, M.-L.; Jerôme, C.; Vanderplasschen, A.; Marchand-Brynaert, J.; et al. Targeting nanoparticles to M cells with non-peptidic ligands for oral vaccination. Eur. J. Pharm. Biopharm. 2009, 73, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Chono, S.; Tanino, T.; Seki, T.; Morimoto, K. Efficient drug targeting to rat alveolar macrophages by pulmonary administration of ciprofloxacin incorporated into mannosylated liposomes for treatment of respiratory intracellular parasitic infections. J. Control. Release 2008, 127, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, V.L.; Lee, Y.C.; Schlesinger, P.H.; Stahl, P.D. l-Fucose-terminated glycoconjugates are recognized by pinocytosis receptors on macrophages. Proc. Natl. Acad. Sci. USA 1981, 78, 1019–1022. [Google Scholar] [CrossRef] [PubMed]
- Hwang, S.M.; Kim, D.D.; Chung, S.J.; Shim, C.K. Delivery of ofloxacin to the lung and alveolar macrophages via hyaluronan microspheres for the treatment of tuberculosis. J. Control. Release 2008, 129, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, S.; Grenha, A. Activation of macrophages: Establishing a role for polysaccharides in drug delivery strategies envisaging antibacterial therapy. Curr. Pharm. Des. 2015, 21, 4869–4887. [Google Scholar] [CrossRef] [PubMed]
- Mahor, S.; Dash, B.C.; O’Connor, S.; Pandit, A. Mannosylated polyethyleneimine-hyaluronan nanohybrids for targeted gene delivery to macrophage-like cell lines. Bioconjug. Chem. 2012, 23, 1138–1148. [Google Scholar] [CrossRef] [PubMed]
- Nimje, N.; Agarwal, A.; Saraogi, G.K.; Lariya, N.; Rai, G.; Agrawal, H.; Agrawal, G.P. Mannosylated nanoparticulate carriers of rifabutin for alveolar targeting. J. Drug Target. 2015, 17, 777–787. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Chen, L.; Cao, Q.-R.; Chen, D.; Cui, J. Preparation and evaluation of mannose receptor mediated macrophage targeting delivery system. J. Control. Release 2011, 152, 190–191. [Google Scholar] [CrossRef] [PubMed]
- Asthana, G.S.; Asthana, A.; Kohli, D.V.; Vyas, S.P. Mannosylated chitosan nanoparticles for delivery of antisense oligonucleotides for macrophage targeting. Biomed. Res. Int. 2014, 2014, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Makino, K.; Yamamoto, N.; Higuchi, K.; Harada, N.; Ohshima, H.; Terada, H. Phagocytic uptake of polystyrene microspheres by alveolar macrophages: Effects of the size and surface properties of the microspheres. Colloids Surf. B Biointerfaces 2003, 27, 33–39. [Google Scholar] [CrossRef]
- Maretti, E.; Rossi, T.; Bondi, M.; Croce, M.A.; Hanuskova, M.; Leo, E.; Sacchetti, F.; Iannuccelli, V. Inhaled solid lipid microparticles to target alveolar macrophages for tuberculosis. Int. J. Pharm. 2014, 462, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Chong, A.S.; Parish, C.R. Cell surface receptors for sulphated polysaccharides: A potential marker for macrophage subsets. Immunology 1986, 58, 277–284. [Google Scholar] [PubMed]
- Hirota, K.; Terada, H. Endocytosis of particle formulations by macrophages and its application to clinical treatment. In Molecular Regulation of Endocytosis; Ceresa, B., Ed.; InTech: Rijeka, Croatia, 2012; pp. 413–428. [Google Scholar]
- Geiser, M. Update on macrophage clearance of inhaled micro- and nanoparticles. J. Aerosol Med. Pulm. Drug Deliv. 2010, 23, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Aderem, A.; Underhill, D.M. Mechanisms of phagocytosis in macrophages. Annu. Rev. Immunol. 1999, 17, 593–623. [Google Scholar] [CrossRef] [PubMed]
- Tabata, Y.; Ikada, Y. Effect of the size and surface charge of polymer microspheres on their phagocytosis by macrophage. Biomaterials 1988, 9, 356–362. [Google Scholar] [CrossRef]
- Jiang, Z.; Ueno, M.; Nishiguchi, T.; Abu, R.; Isaka, S.; Okimura, T.; Yamaguchi, K.; Oda, T. Importance of sulfate groups for the macrophage-stimulating activities of ascophyllan isolated from the brown alga Ascophyllum nodosum. Carbohydr. Res. 2013, 380, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Heinzelmann, M.; Polk, H.C., Jr.; Frederick, N. Modulation of lipopolysaccharide-induced monocyte activation by heparin-binding protein and fucoidan. Infect. Immun. 1998, 66, 5842–5847. [Google Scholar] [PubMed]
- Bhattacharyya, S.; Gill, R.; Ling Chen, M.; Zhang, F.; Linhardt, R.J.; Dudeja, P.K.; Tobacman, J.K. Toll-like receptor 4 mediates induction of the Bcl10-NFκB-interleukin-8 inflammatory pathway by carrageenan in human intestinal epithelial cells. J. Biol. Chem. 2008, 283, 10550–10558. [Google Scholar] [CrossRef] [PubMed]
- Nacife, V.P.; Soeiro, M.D.; Araújo-Jorge, T.C.; Castro-Faria Neto, H.C.; Meirelles, M.D. Ultrastructural, immunocytochemical and flow cytometry study of mouse peritoneal cells stimulated with carrageenan. Cell Struct. Funct. 2000, 25, 337–350. [Google Scholar] [PubMed]
- Sugita-Konishi, Y.; Yamashita, S.; Amano, F.; Shimizu, M. Effects of carrageenans on the binding, phagocytotic, and killing abilities of macrophages to salmonella. Biosci. Biotechnol. Biochem. 2003, 67, 1425–1428. [Google Scholar] [CrossRef] [PubMed]
- Kar, S.; Sharma, G.; Das, P.K. Fucoidan cures infection with both antimony-susceptible and-resistant strains of Leishmania donovani through Th1 response and macrophage-derived oxidants. J. Antimicrob. Chemother. 2011, 66, 618–625. [Google Scholar] [CrossRef] [PubMed]
- Teruya, T.; Tatemoto, H.; Konishi, T.; Tako, M. Structural characteristics and in vitro macrophage activation of acetyl fucoidan from Cladosiphon okamuranus. Glycoconj. J. 2009, 26, 1019–1028. [Google Scholar] [CrossRef] [PubMed]
- Zedong, J.; Okimura, T.; Yamaguchi, K.; Oda, T. The potent activity of sulfated polysaccharide, ascophyllan, isolated from Ascophyllum nodosum to induce nitric oxide and cytokine production from mouse macrophage RAW264.7 cells: Comparison between ascophyllan and fucoidan. Nitric Oxide Biol. Chem. 2011, 25, 407–415. [Google Scholar]
- Yang, J.W.; Yonn, S.Y.; Oh, S.J.; Kim, S.K.; Kang, K.W. Bifunctional effects of fucoidan on the expression of inducible nitric oxide synthase. Biochem. Biophys. Res. Commun. 2006, 346, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Dumas, B.; Jaulneau, V.; Lafitte, C.; Jacquet, C.; Fournier, S.; Salamagne, S.; Briand, X.; Esquerré-Tugayé, M.T. Ulvan, a sulfated polysaccharide from green algae, activates plant immunity through the jasmonic acid signaling pathway. J. Biomed. Biotechnol. 2010, 2010, 525291. [Google Scholar] [CrossRef]
- Lee, D.-G.; Hyun, J.-W.; Kang, K.-A.; Lee, J.-O.; Lee, S.-H.; Ha, B.-J.; Ha, J.-M.; Lee, E.Y.; Lee, J.-H. Ulva lactuca: A potential seaweed for tumor treatment and immune stimulation. Biotechnol. Bioprocess Eng. 2004, 9, 236–238. [Google Scholar] [CrossRef]
- Castro, R.; Piazzon, M.C.; Zarra, I.; Leiro, J.; Noya, M.; Lamas, J. Stimulation of turbot phagocytes by Ulva rigida C. Agardh polysaccharides. Aquaculture 2006, 254, 9–20. [Google Scholar] [CrossRef]

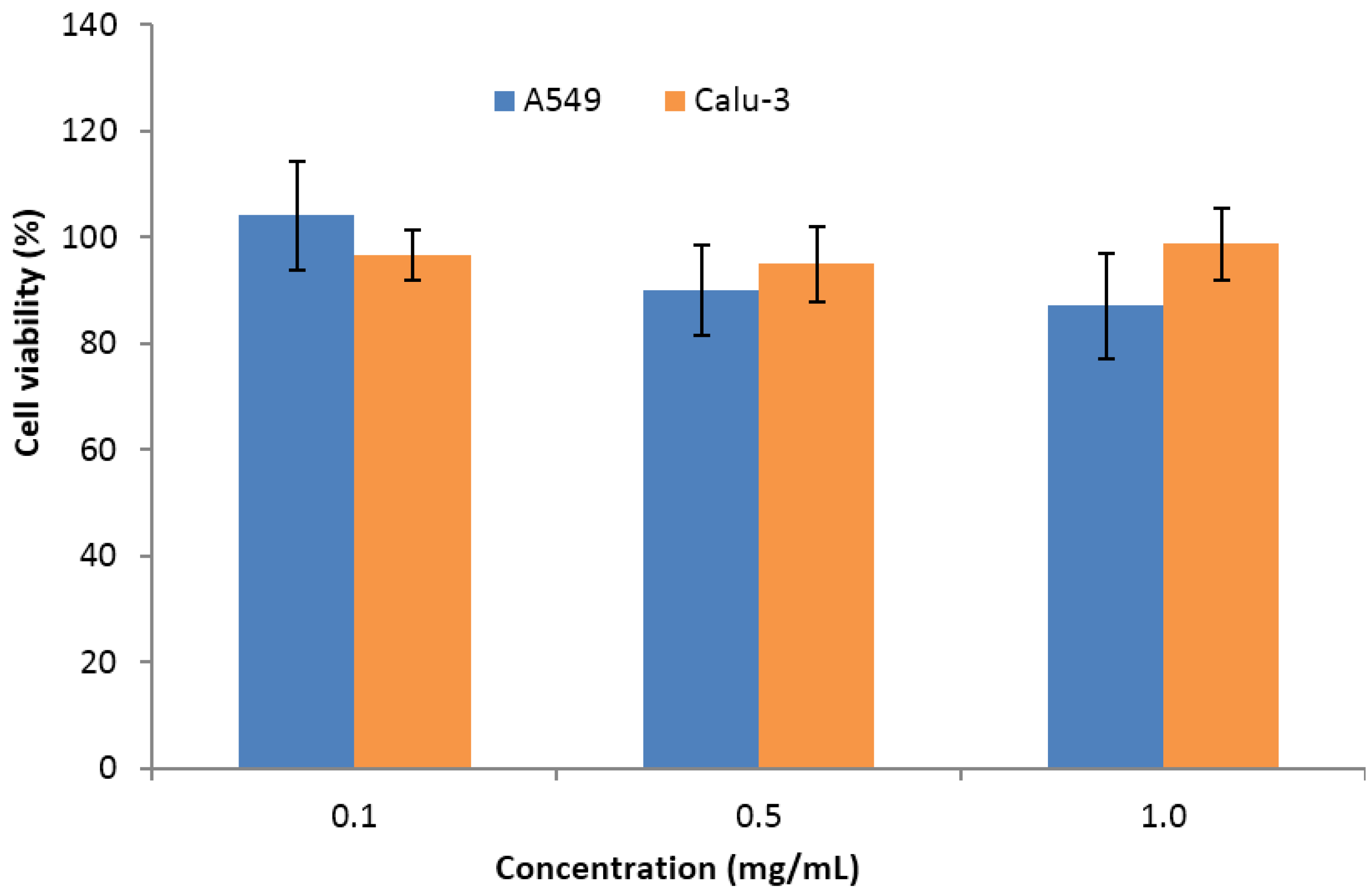
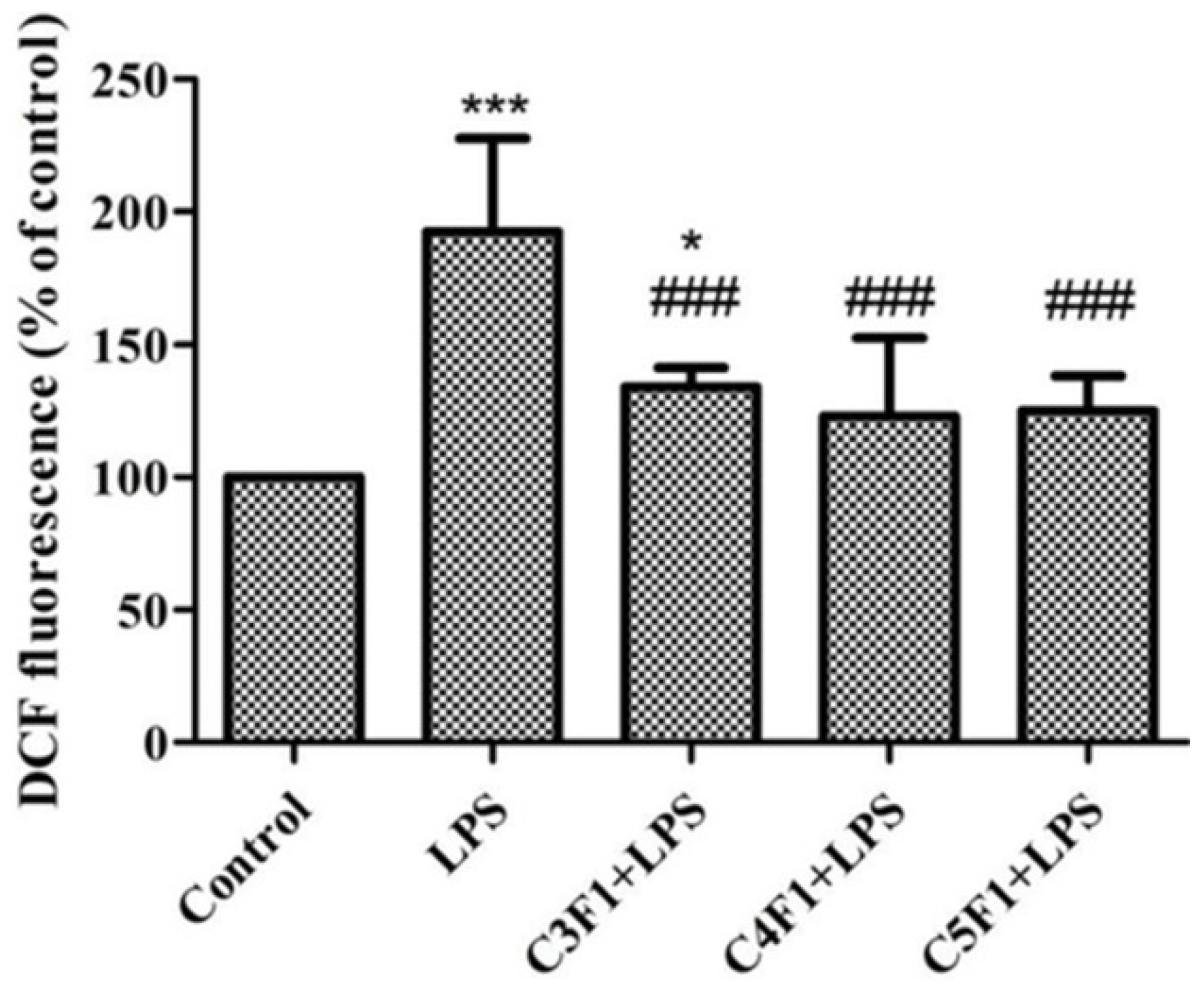
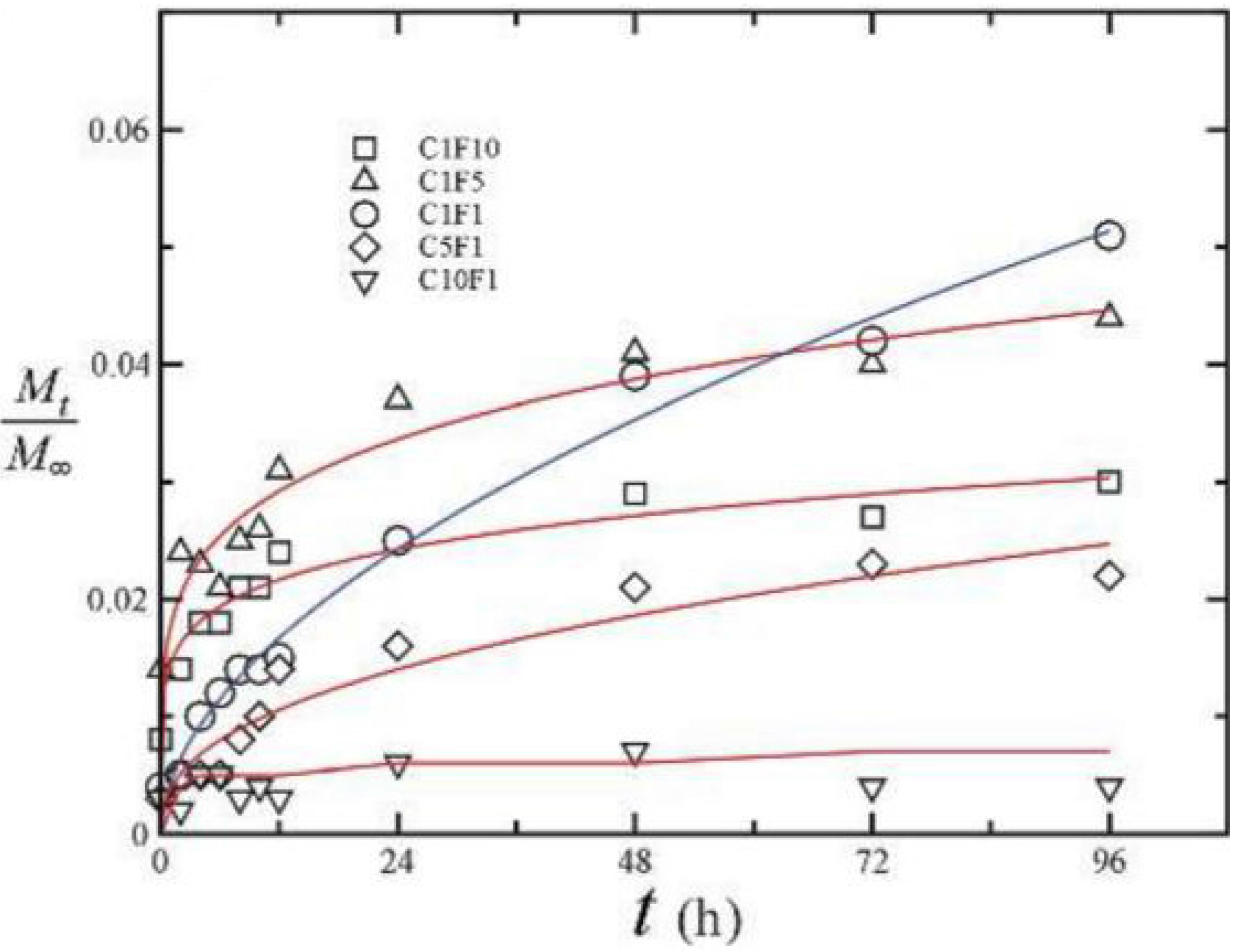
| Sulfated Polysaccharide | Marine Algae Group | Main Genera | Molecular Weight (kDa) | Solubility in Water | Viscosity (cps, in Water) | pH in Aqueous Solution | References |
|---|---|---|---|---|---|---|---|
| Carrageenan | Rhodophyceae | Chondrus Euchema Furcellaria Gigartina Hypnea Iridae Kappaphycus | 100–1000 | * κ-, ι- and λ-carrageenan soluble at 80 °C | 5–800 (1.5% w/v, 75 °C) | 7.0–10.0 | [2,7,8,9,10] |
| Fucoidan | Phaeophyceae | Analipus Chorda Dictyota Fucus Kjellmaniella Pelvetia Sargassum Undaria | 10–950 | 10 mg/mL (F. vesiculosus) | n.a. | n.a. | [11,12,13,14,15,16,17,18,19] |
| Ulvan | Chlorophyceae | Enteromorpha Ulva | 1.14 to > 2 × 106 | n.a. | 18–100 (1.6% w/v, Ulva spp.) | 7.5 (Ulva spp.) | [20,21,22] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cunha, L.; Grenha, A. Sulfated Seaweed Polysaccharides as Multifunctional Materials in Drug Delivery Applications. Mar. Drugs 2016, 14, 42. https://doi.org/10.3390/md14030042
Cunha L, Grenha A. Sulfated Seaweed Polysaccharides as Multifunctional Materials in Drug Delivery Applications. Marine Drugs. 2016; 14(3):42. https://doi.org/10.3390/md14030042
Chicago/Turabian StyleCunha, Ludmylla, and Ana Grenha. 2016. "Sulfated Seaweed Polysaccharides as Multifunctional Materials in Drug Delivery Applications" Marine Drugs 14, no. 3: 42. https://doi.org/10.3390/md14030042
APA StyleCunha, L., & Grenha, A. (2016). Sulfated Seaweed Polysaccharides as Multifunctional Materials in Drug Delivery Applications. Marine Drugs, 14(3), 42. https://doi.org/10.3390/md14030042







