Alkaloids from the Sponge Stylissa carteri Present Prospective Scaffolds for the Inhibition of Human Immunodeficiency Virus 1 (HIV-1)
Abstract
:1. Introduction
2. Results and Discussion
2.1. Biological Activities of Fractions of S. carteri Extracts
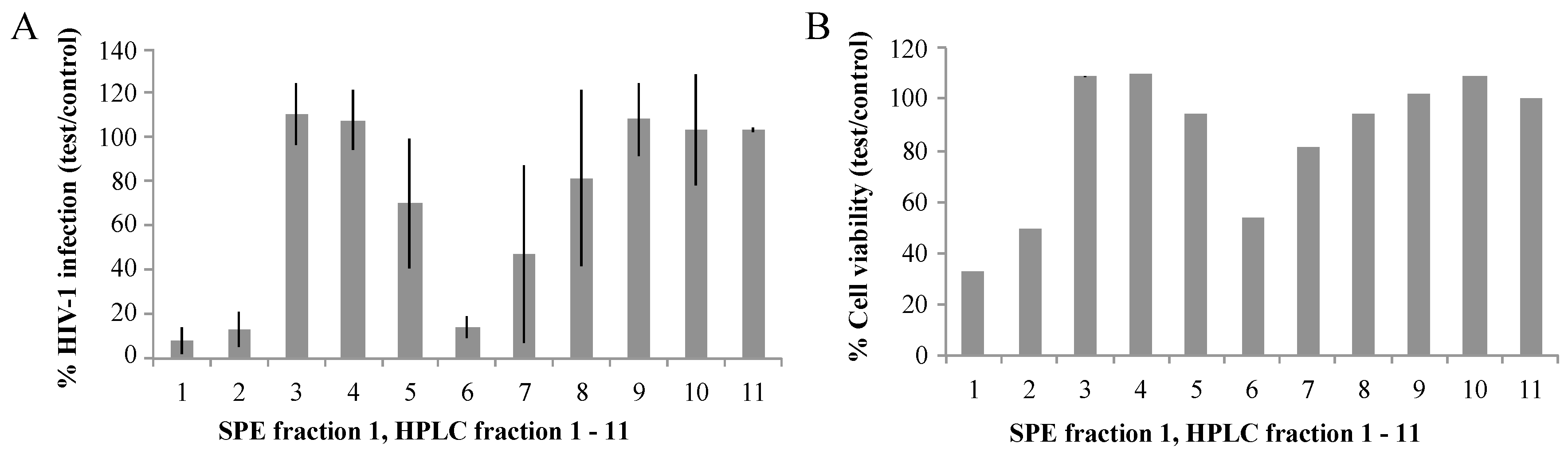
2.2. Identification of HIV-1 Candidate Inhibitors in HPLC Fractions 2 and 6
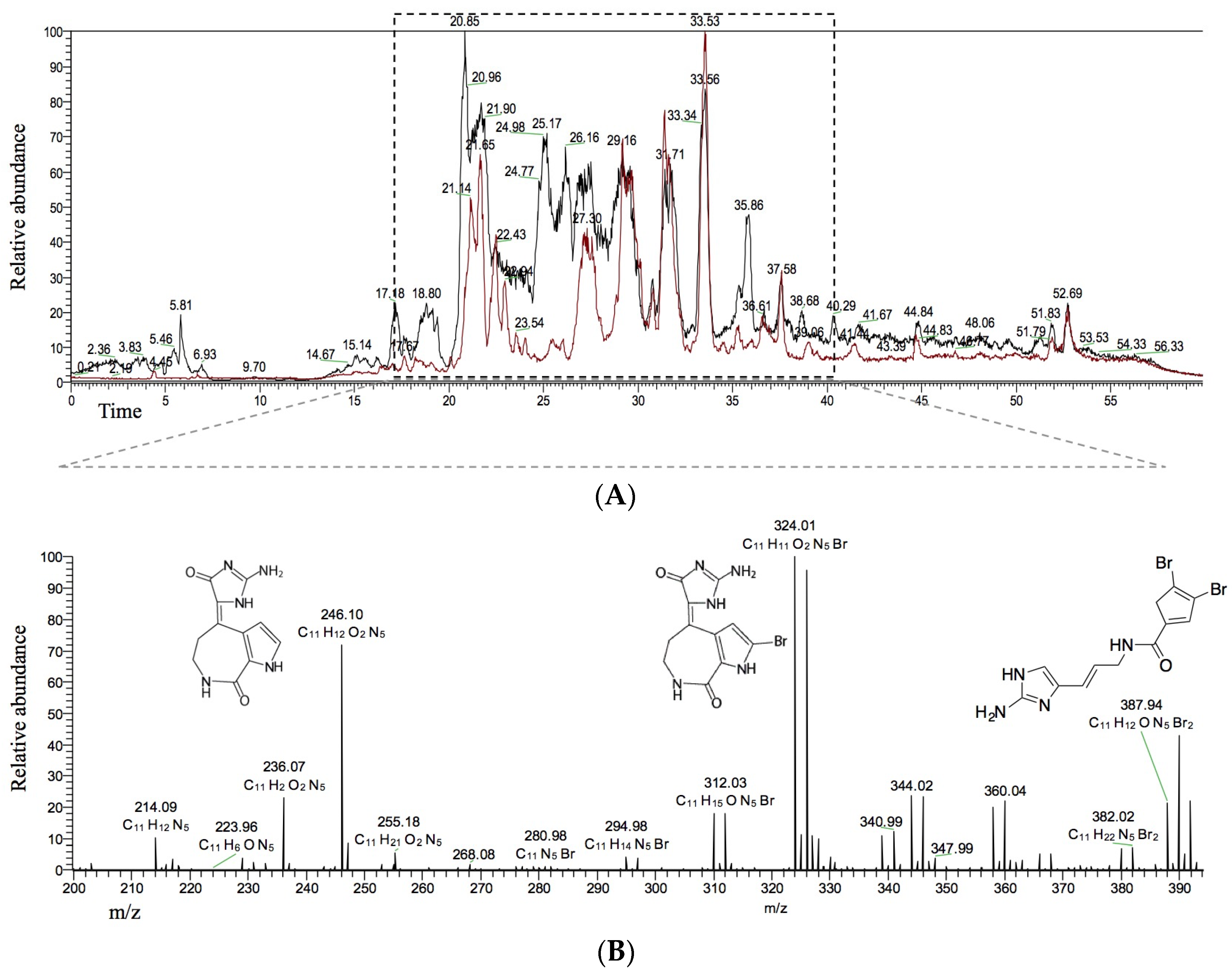
2.3. Three HIV-1 Candidate Inhibitors Hymenialdisine, Debromohymenialdisine, and Oroidin from S. carteri Inhibit HIV-1 Replication to Varying Degrees

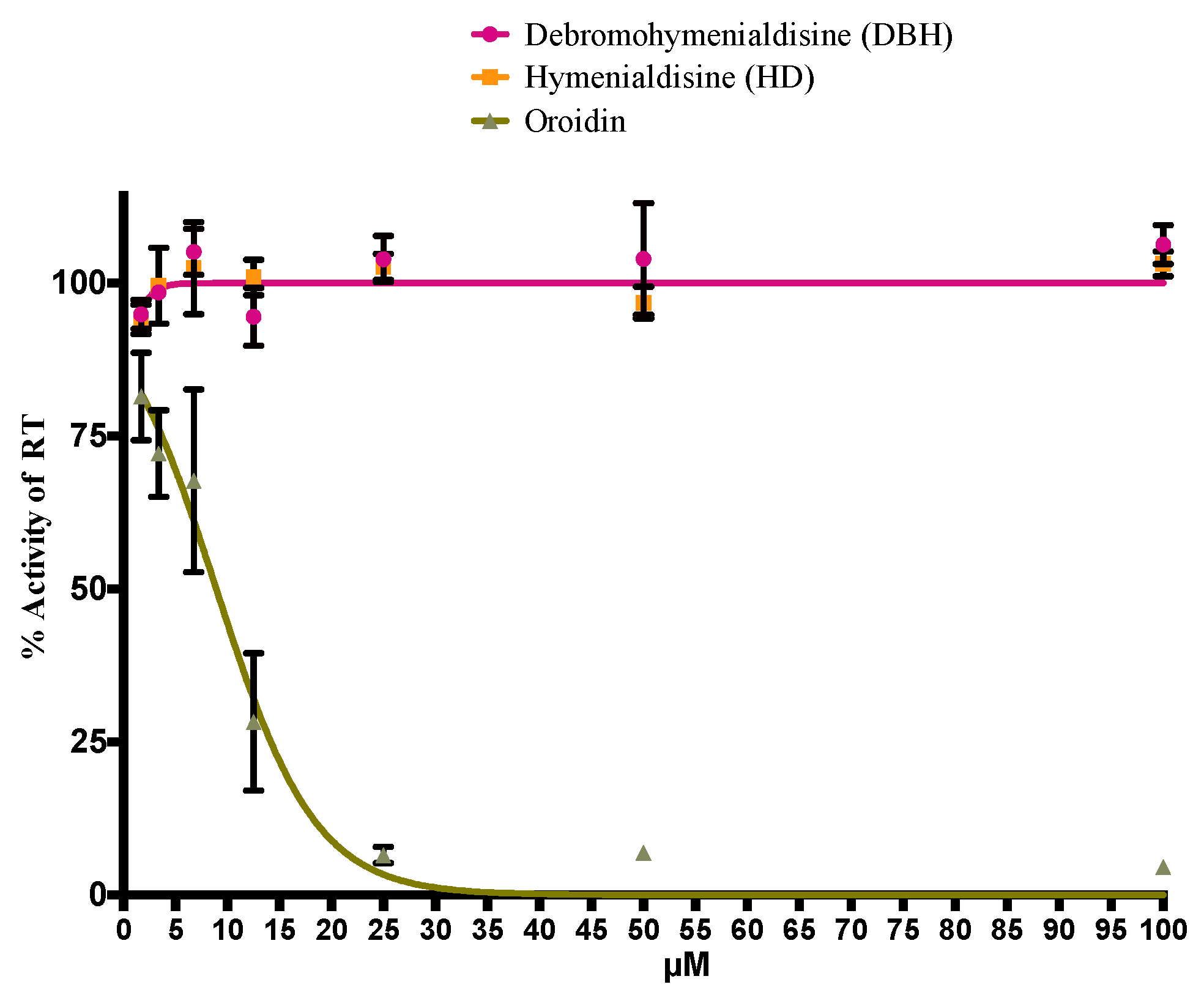
2.4. Oroidin from S. carteri Inhibits the HIV-1 Reverse Transcriptase (RT) in a Biochemical Assay
3. Experimental Section
3.1. S. carteri Specimen Collection and Sample Fractionation
3.1.1. Analytical Chemistry of Bioactive S. carteri Fractions
3.1.2. Single Compounds from Bioactive S. carteri Fractions
3.2. Human Immunodeficiency Virus, Full Virus Screening (EASY-HIT)
MTT Cell Proliferation Assay
3.3. Human Immunodeficiency Virus, Reverse Transcriptase Biochemical Test
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Joint United Nations Programme on HIV/AIDS (UNAIDS). Global Report: UNAIDS Report on the Global Aids Epidemics 2013; UNAIDS/JC2502/1/E; UNAIDS: Geneva, Switzerland, 2013. [Google Scholar]
- U.S. Food and Drug Administration (FDA). Antiretroviral Drugs Used in the Treatment of HIV Infection; U.S. Department of Health and Human Services: Silver Spring, MD, USA, 2015.
- Martinez, J.P.; Sasse, F.; Bronstrup, M.; Diez, J.; Meyerhans, A. Antiviral drug discovery: Broad-spectrum drugs from nature. Nat. Prod. Rep. 2015, 32, 29–48. [Google Scholar] [CrossRef] [PubMed]
- Clercq, D. Curious discoveries in antiviral drug development: The role of serendipity. Med. Res. Rev. 2015, 35, 698–719. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M.; Battershill, C.N. Therapeutic agents from the sea: Biodiversity, chemo-evolutionary insight and advances to the end of darwin’s 200th year. Diving Hyperb. Med. 2009, 39, 216–225. [Google Scholar] [PubMed]
- Butler, M.S.; Robertson, A.A.B.; Cooper, M.A. Natural product and natural product derived drugs in clinical trials. Nat. Prod. Rep. 2014, 31, 1612–1661. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.W.; Chen, J.H.; Hu, G.P.; Yu, J.C.; Zhu, X.; Lin, Y.C.; Chen, S.P.; Yuan, J. Statistical research on the bioactivity of new marine natural products discovered during the 28 years from 1985 to 2012. Mar. Drugs 2015, 13, 202–221. [Google Scholar] [CrossRef] [PubMed]
- Yarnold, J.E.; Hamilton, B.R.; Welsh, D.T.; Pool, G.F.; Venter, D.J.; Carroll, A.R. High resolution spatial mapping of brominated pyrrole-2-aminoimidazole alkaloids distributions in the marine sponge stylissa flabellata via MALDI-mass spectrometry imaging. Mol. Biosyst. 2012, 8, 2249–2259. [Google Scholar] [CrossRef] [PubMed]
- Keifer, P.A.; Schwartz, R.E.; Koker, M.E.S.; Hughes, R.G.; Rittschof, D.; Rinehart, K.L. Bioactive bromopyrrole metabolites from the caribbean sponge agelas conifera. J. Organ. Chem. 1991, 56, 2965–2975. [Google Scholar] [CrossRef]
- Breton, J.J.; Chabot-Fletcher, M.C. The natural product hymenialdisine inhibits interleukin-8 production in U937 cells by inhibition of nuclear factor-kappaB. J. Pharmacol. Exp. Ther. 1997, 282, 459–466. [Google Scholar] [PubMed]
- Kremb, S.; Helfer, M.; Heller, W.; Hoffmann, D.; Wolff, H.; Kleinschmidt, A.; Cepok, S.; Hemmer, B.; Durner, J.; Brack-Werner, R. EASY-HIT: HIV full-replication technology for broad discovery of multiple classes of HIV inhibitors. Antimicrob. Agents Chemother. 2010, 54, 5257–5268. [Google Scholar] [CrossRef] [PubMed]
- Helfer, M.; Koppensteiner, H.; Schneider, M.; Rebensburg, S.; Forcisi, S.; Müller, C.; Schmitt-Kopplin, P.; Schindler, M.; Brack-Werner, R. The root extract of the medicinal plant Pelargonium sidoides is a potent HIV-1 attachment inhibitor. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Kremb, S.; Helfer, M.; Kraus, B.; Wolff, H.; Wild, C.; Schneider, M.; Voolstra, C.R.; Brack-Werner, R. Aqueous extracts of the marine brown alga Lobophora variegata inhibit HIV-1 infection at the level of virus entry into cells. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Forenza, S.; Minale, L.; Riccio, R. New bromo-pyrrole derivatives from sponge agelas oroides. J. Chem. Soc. D Chem. Commun. 1971. [Google Scholar] [CrossRef]
- Sharma, G.M.; Buyer, J.S.; Pomerantz, M.W. Characterization of a yellow compound isolated from the marine sponge phakellia flabellata. J. Chem. Soc. Chem. Commun. 1980. [Google Scholar] [CrossRef]
- Cimino, G.; Derosa, S.; Destefano, S.; Mazzarella, L.; Puliti, R.; Sodano, G. Isolation and X-ray crystal structure of a novel bromo-compound from 2 marine sponges. Tetrahedron Lett. 1982, 23, 767–768. [Google Scholar] [CrossRef]
- Mohammed, R.; Peng, J.; Kelly, M.; Hamann, M.T. Cyclic heptapeptides from the jamaican sponge stylissa caribica. J. Nat. Prod. 2006, 69, 1739–1744. [Google Scholar] [CrossRef] [PubMed]
- Al-Mourabit, A.; Zancanella, M.A.; Tilvi, S.; Romo, D. Biosynthesis, asymmetric synthesis, and pharmacology, including cellular targets, of the pyrrole-2-aminoimidazole marine alkaloids. Nat. Prod. Rep. 2011, 28, 1229–1260. [Google Scholar] [CrossRef] [PubMed]
- Meijer, L.; Thunnissen, A.M.W.H.; White, A.W.; Garnier, M.; Nikolic, M.; Tsai, L.H.; Walter, J.; Cleverley, K.E.; Salinas, P.C.; Wu, Y.Z.; et al. Inhibition of cyclin-dependent kinases, GSK-3 beta and CK1 by hymenialdisine, a marine sponge constituent. Chem. Biol. 2000, 7, 51–63. [Google Scholar] [CrossRef]
- Debebe, Z.; Ammosova, T.; Breuer, D.; Lovejoy, D.B.; Kalinowski, D.S.; Kumar, K.; Jerebtsova, M.; Ray, P.; Kashanchi, F.; Gordeuk, V.R.; et al. Iron chelators of the di-2-pyridylketone thiosemicarbazone and 2-benzoylpyridine thiosemicarbazone series inhibit HIV-1 transcription: Identification of novel cellular targets-iron, cyclin-dependent kinase (CDK) 2, and CDK9. Mol. Pharmacol. 2011, 79, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Curman, D.; Cinel, B.; Williams, D.E.; Rundle, N.; Block, W.D.; Goodarzi, A.A.; Hutchins, J.R.; Clarke, P.R.; Zhou, B.B.; Lees-Miller, S.P.; et al. Inhibition of the G2 DNA damage checkpoint and of protein kinases Chk1 and Chk2 by the marine sponge alkaloid debromohymenialdisine. J. Biol. Chem. 2001, 276, 17914–17919. [Google Scholar] [CrossRef] [PubMed]
- Goh, W.C.; Rogel, M.E.; Kinsey, C.M.; Michael, S.F.; Fultz, P.N.; Nowak, M.A.; Hahn, B.H.; Emerman, M. HIV-1 Vpr increases viral expression by manipulation of the cell cycle: A mechanism for selection of Vpr in vivo. Nat. Med. 1998, 4, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Bugni, T.S.; Richards, B.; Bhoite, L.; Cimbora, D.; Harper, M.K.; Ireland, C.M. Marine natural product libraries for high-throughput screening and rapid drug discovery. J. Nat. Prod. 2008, 71, 1095–1098. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
O’Rourke, A.; Kremb, S.; Bader, T.M.; Helfer, M.; Schmitt-Kopplin, P.; Gerwick, W.H.; Brack-Werner, R.; Voolstra, C.R. Alkaloids from the Sponge Stylissa carteri Present Prospective Scaffolds for the Inhibition of Human Immunodeficiency Virus 1 (HIV-1). Mar. Drugs 2016, 14, 28. https://doi.org/10.3390/md14020028
O’Rourke A, Kremb S, Bader TM, Helfer M, Schmitt-Kopplin P, Gerwick WH, Brack-Werner R, Voolstra CR. Alkaloids from the Sponge Stylissa carteri Present Prospective Scaffolds for the Inhibition of Human Immunodeficiency Virus 1 (HIV-1). Marine Drugs. 2016; 14(2):28. https://doi.org/10.3390/md14020028
Chicago/Turabian StyleO’Rourke, Aubrie, Stephan Kremb, Theresa Maria Bader, Markus Helfer, Philippe Schmitt-Kopplin, William H. Gerwick, Ruth Brack-Werner, and Christian R. Voolstra. 2016. "Alkaloids from the Sponge Stylissa carteri Present Prospective Scaffolds for the Inhibition of Human Immunodeficiency Virus 1 (HIV-1)" Marine Drugs 14, no. 2: 28. https://doi.org/10.3390/md14020028
APA StyleO’Rourke, A., Kremb, S., Bader, T. M., Helfer, M., Schmitt-Kopplin, P., Gerwick, W. H., Brack-Werner, R., & Voolstra, C. R. (2016). Alkaloids from the Sponge Stylissa carteri Present Prospective Scaffolds for the Inhibition of Human Immunodeficiency Virus 1 (HIV-1). Marine Drugs, 14(2), 28. https://doi.org/10.3390/md14020028







