Comparison of Cell Disruption Methods for Improving Lipid Extraction from Thraustochytrid Strains
Abstract
:1. Introduction
2. Results and Discussion
2.1. Biomass and Lipid Production
| Properties | Thraustochytrid strains | |
|---|---|---|
| Schizochytrium sp. S31 | Thraustochytrium sp. AMCQS5-5 | |
| Dry weight (g·L−1) | 4.06 | 3.23 |
| Biomass productivity (g L−1·day−1) | 0.88 | 0.64 |
| Average lipid content (mg·L−1) | 503.7 | 321.3 |
| Lipid productivity (mg·L−1·day−1) | 100.74 | 64.2 |
2.2. Lipid Extraction from Thraustochytrid by Organic Solvents
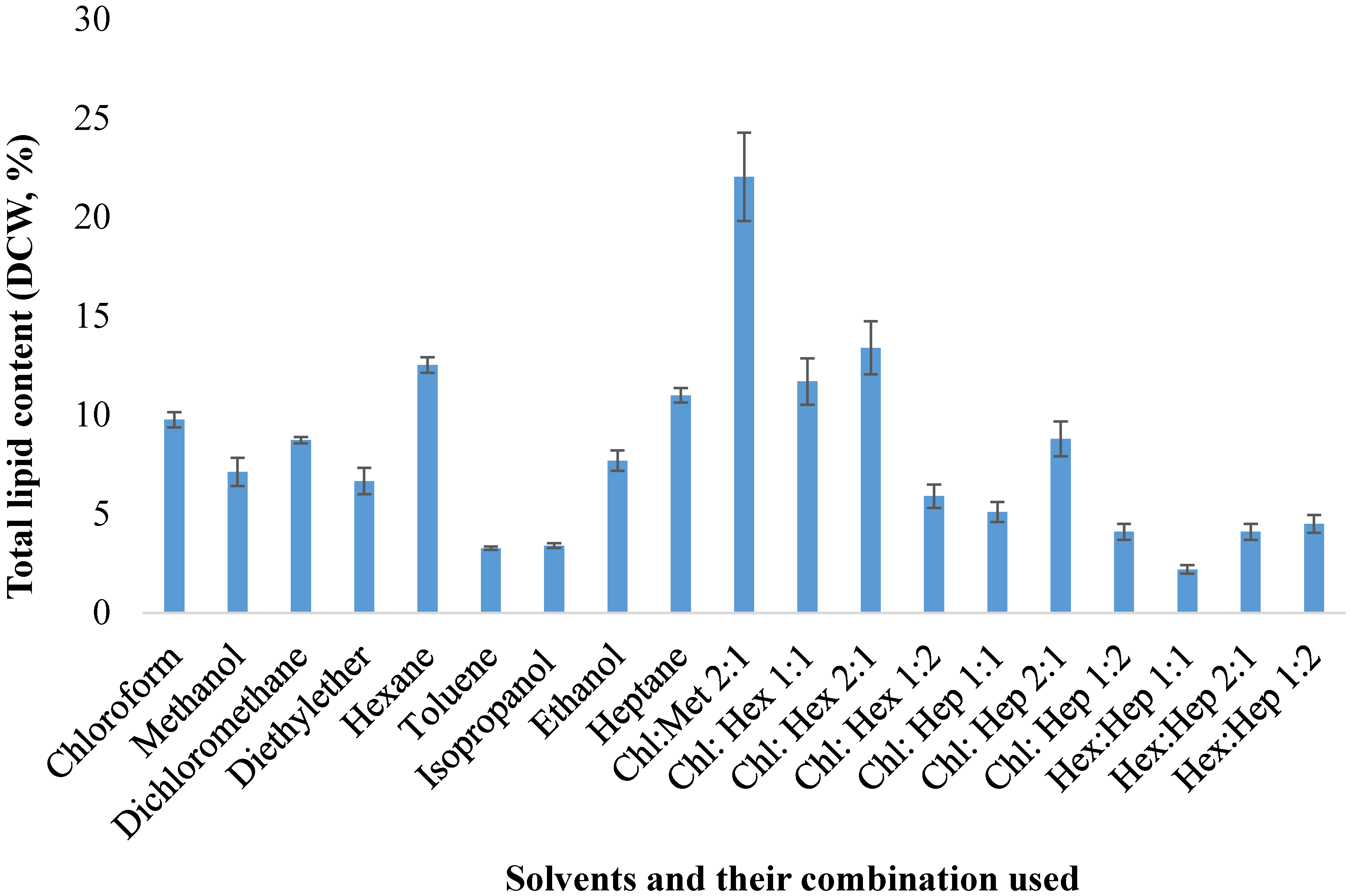
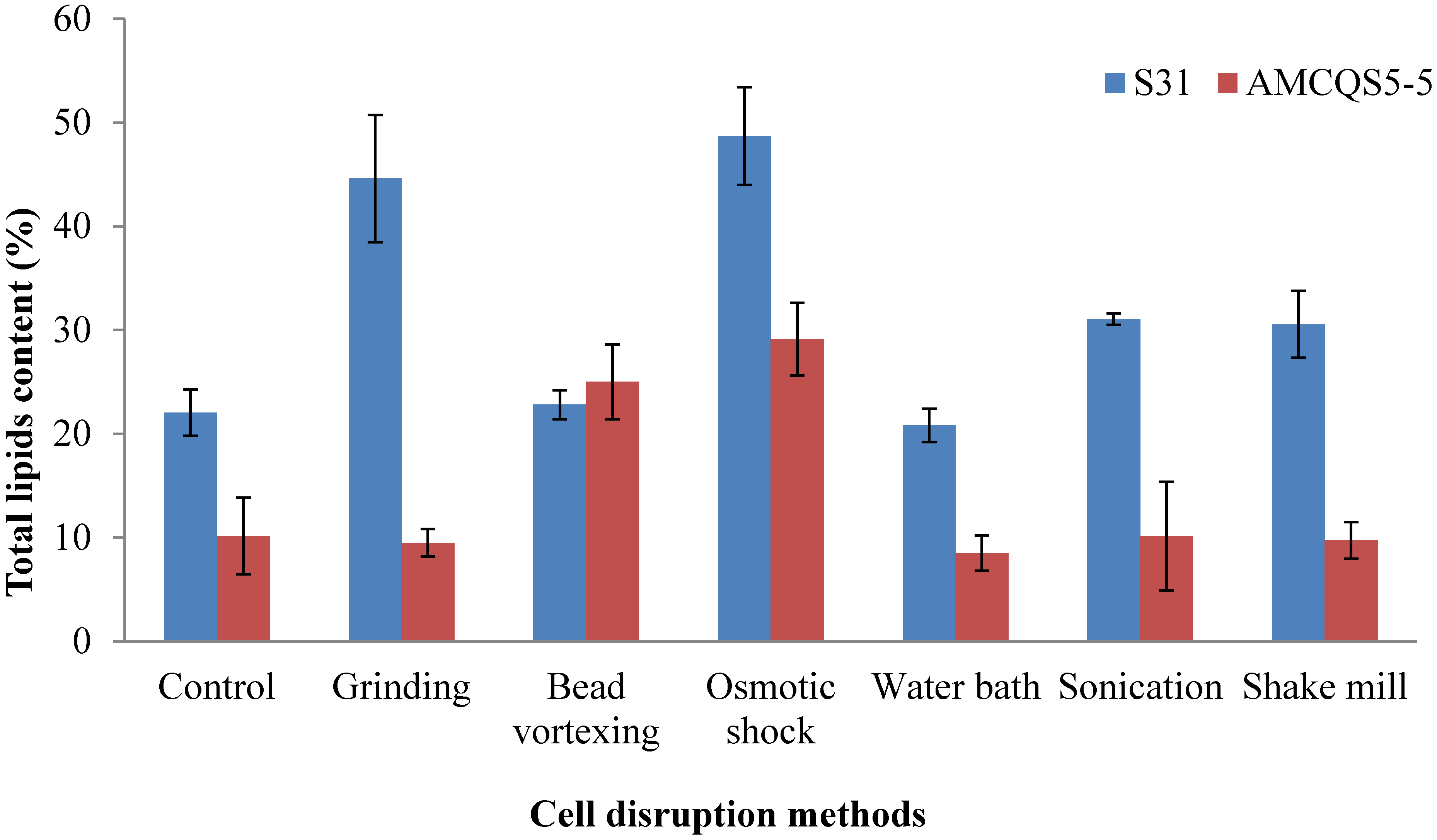
2.3. Comparison of Lipid Extraction Methods
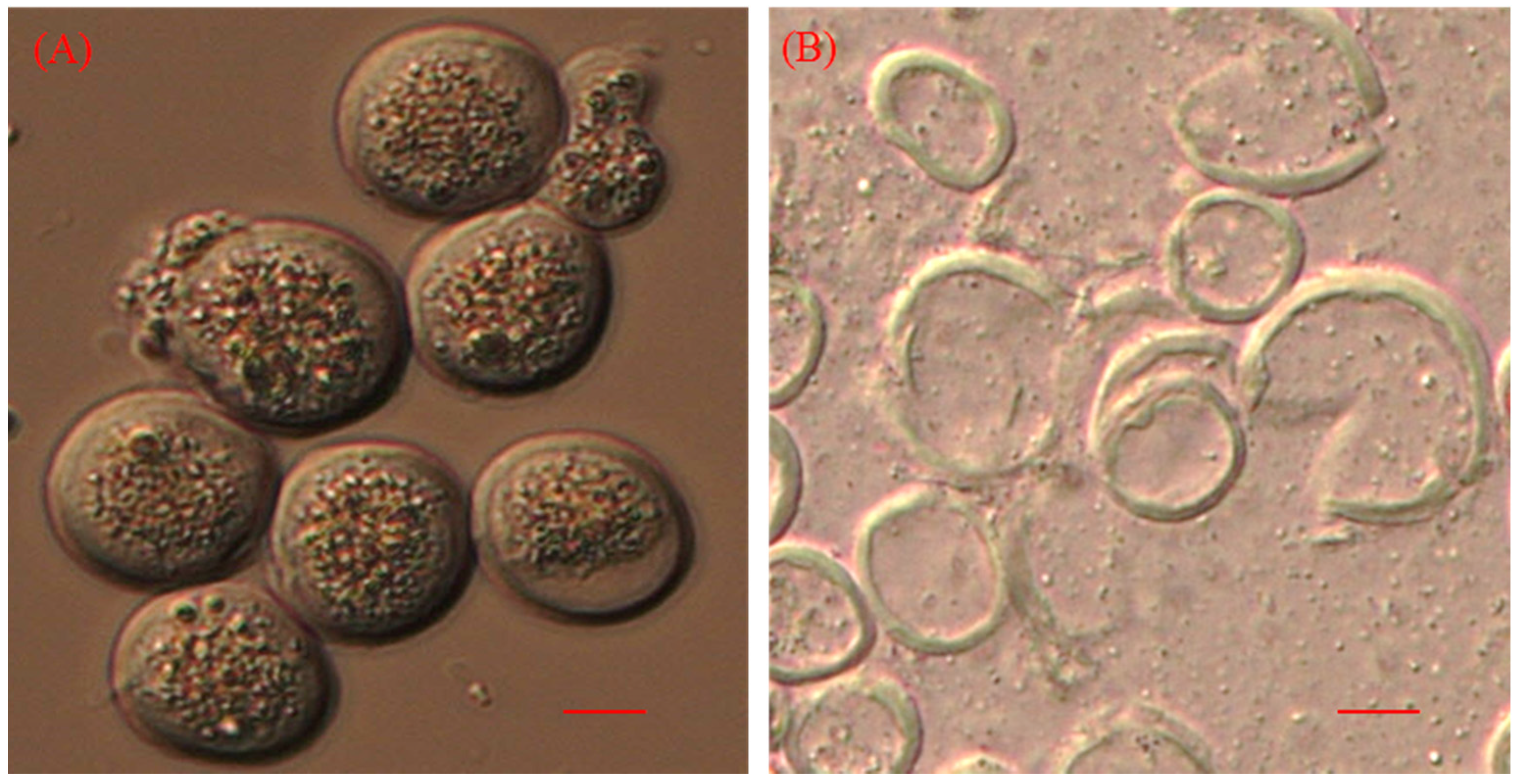
| No. | Cell disruption methods used | Efficient method | Organisms used | Lipid content (%) | Reference |
|---|---|---|---|---|---|
| 1 | Autoclaving | Microwaves | [9] | ||
| Bead beating | Botryococcus sp. | 28.6 | |||
| Microwaves | Chlorella vulgaris | 11 | |||
| Sonication | Scenedesmus sp. | 11.5 | |||
| Osmotic shock | |||||
| 2 | Sonication | Sonication | [31] | ||
| Osmotic shock | Chlorella sp. | 20.1 | |||
| Microwave | Nostoc sp. | 18.2 | |||
| Autoclave | Tolypothrix sp. | 14 | |||
| Bead beating | |||||
| 3 | Grinding | Grinding | Chlorella vulgaris | 29 | [17] |
| Sonication | |||||
| Bead milling | |||||
| Enzymatic lysis | |||||
| Microwaves | |||||
| 4 | Grinding | Osmotic shock | This study | ||
| Bead vortexing | Schizochytrium sp. S31 | 48.7 | |||
| Osmotic shock | |||||
| Water bath | Thraustochytrium sp. AMCQS5-5 | 29.1 | |||
| Sonication | |||||
| Shake mill |
| Cell disruption methods | Advantages | Disadvantages |
|---|---|---|
| Manual grinding | - Quickest and efficient - 2 min process | - Localised heating caused denaturation of molecules |
| Bead vortexing | - Can be established easily and relatively effective | - High heat generation, - Incomplete cell lysis |
| Osmotic shock | - Lower energy consumption - Easier scale-up | - Generation of waste salt water - Time consuming |
| Water bath | - Maximum disruption - Easy in handling at lab scale | - Increases the viscosity - Energy intensive |
| Sonication | - Faster extraction - Suitable for all cell type | - Damage chemical structure of molecules |
| Shake mill | - Rapid method | - High energy intensive - High heat generation |
2.4. Fatty Acid Composition of Extracted Lipid
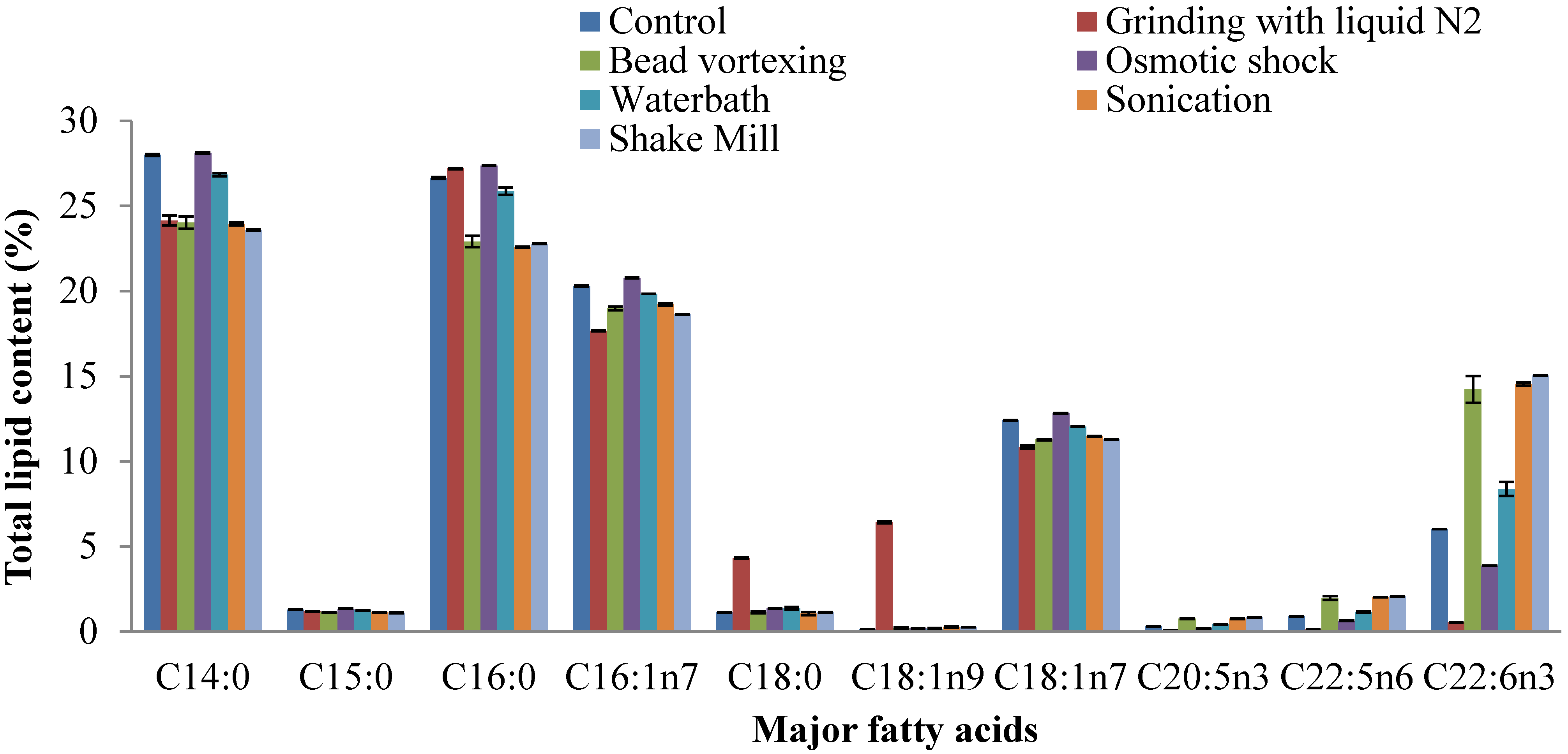
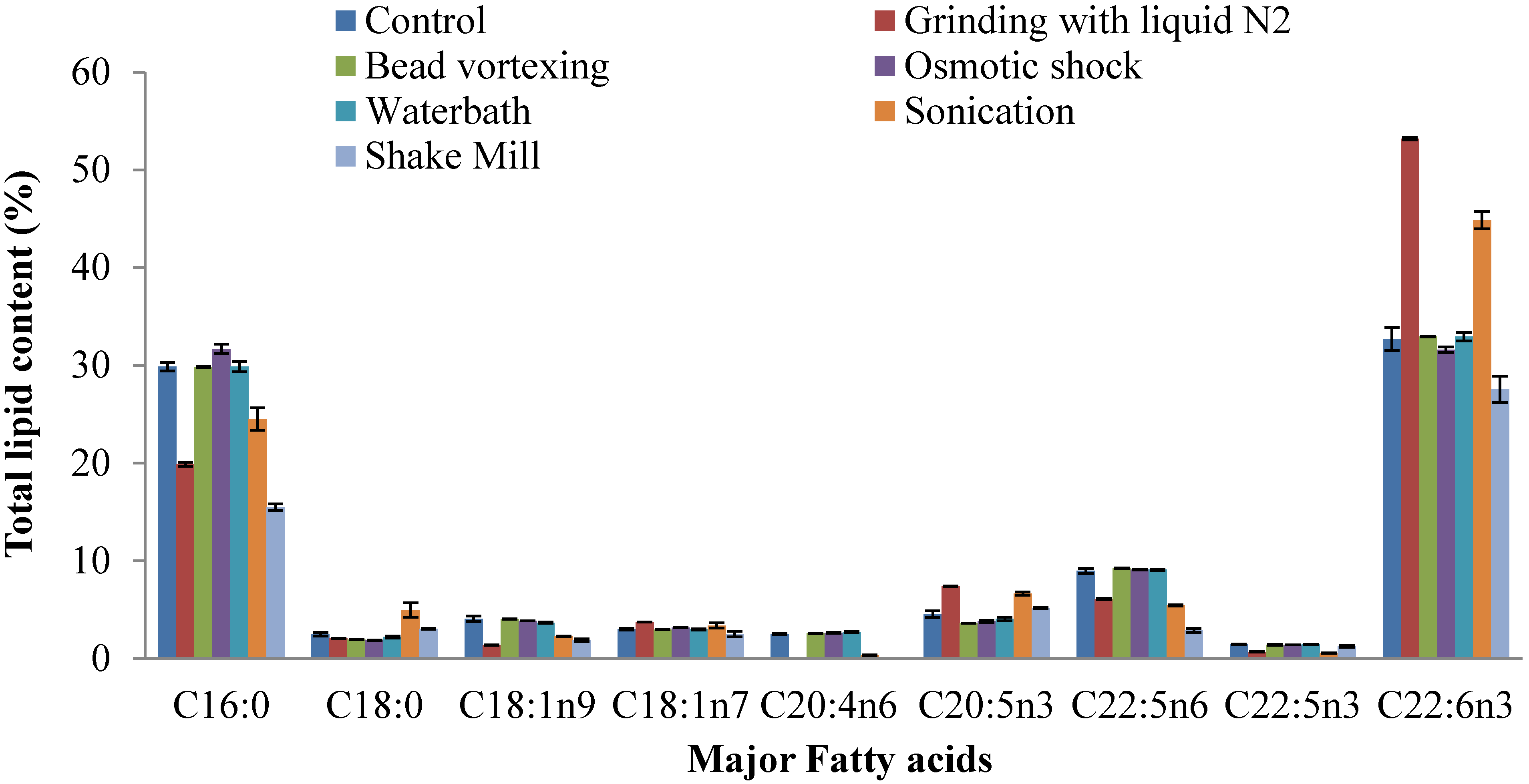
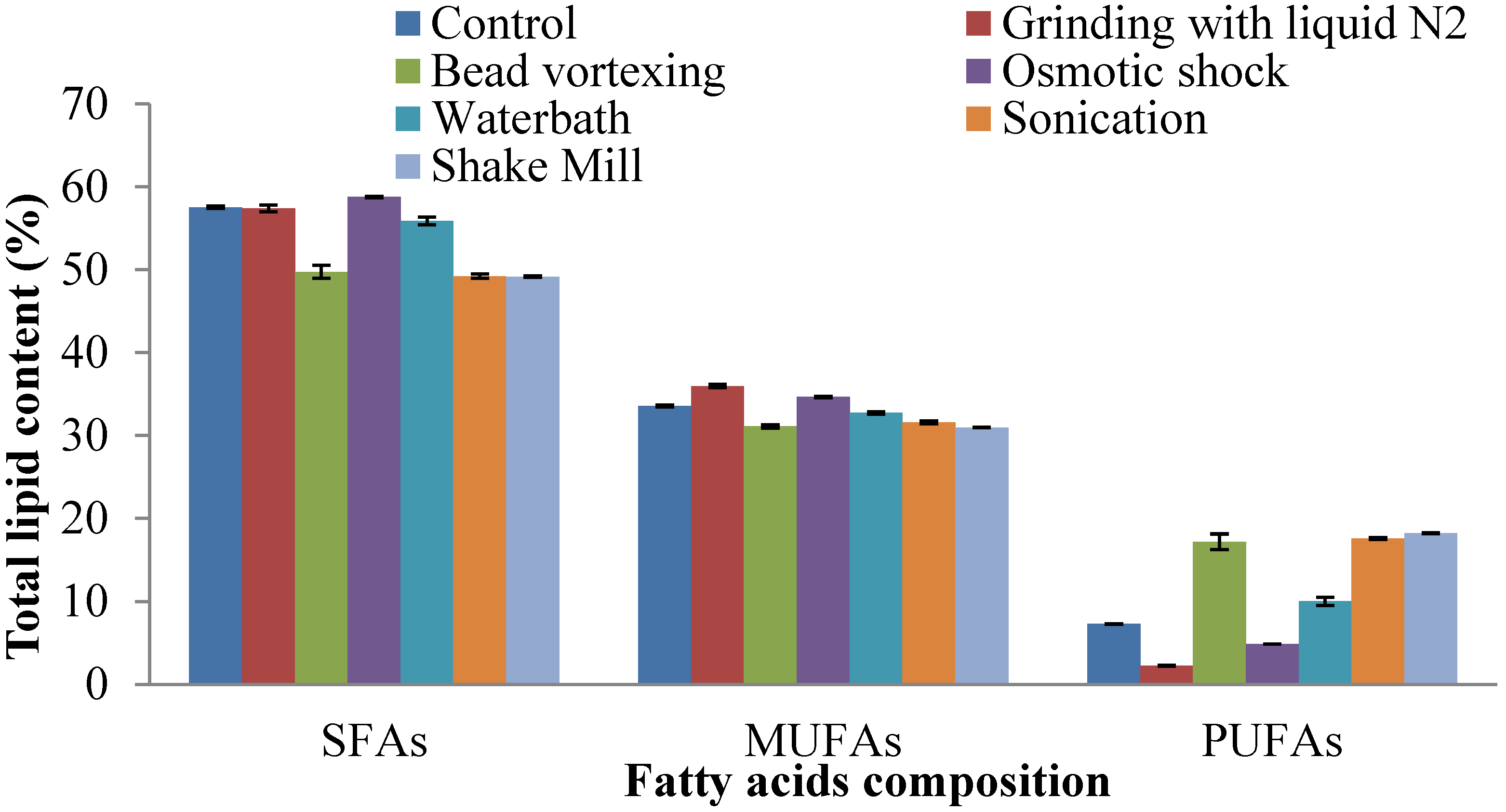
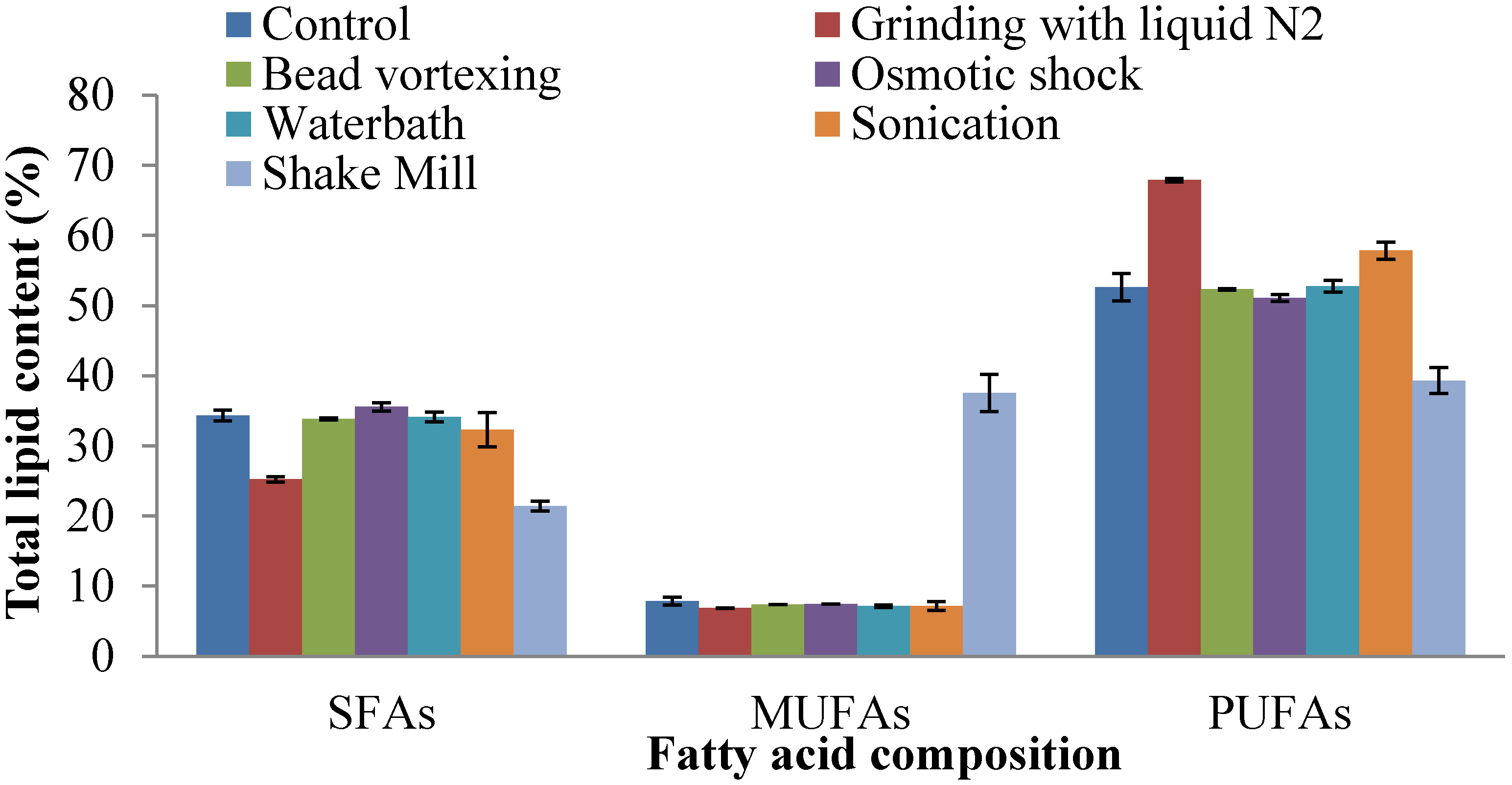
2.5. Prediction of Biodiesel Properties
| Properties | Units | Strain S31 | Strain AMCQS5-5 |
|---|---|---|---|
| Saturated fatty acid | % (m/m) | 59 | 44 |
| Monounsaturated fatty acids | % (m/m) | 35 | 4.6 |
| Polyunsaturated fatty acids | % (m/m) | 14 | 33 |
| Degree of unsaturation | 63 | 70 | |
| Saponification value | mg KOH/g oil | 233.98 | 164.73 |
| Iodine value | g I2/100 g | 98.61 | 157.44 |
| Cetane number | min | 47.44 | 44.01 |
| Long chain saturated factor | % (m/m) | 3.8 | 5.85 |
| Cold filter plugging point | oC | 4.54 | 1.90 |
| Cloud point | oC | 9.74 | 10.79 |
| Allylic position equivalents | 83 | 167.60 | |
| Bis-allylic position equivalents | 70 | 165.20 | |
| Oxidation stability | h | 6.5 | 1.65 |
| Higher heating value | oC | 42.14 | 32.33 |
| Kinematic viscosity | mm2/s | 1.28 | 0.94 |
| Density | kg/m3 | 0.94 | 0.73 |
2.6. Energy Analysis
| Cell Disruption Methods | Lipid Yield (%) | Energy Consumption (MJ·kg−1 Dry Mass) | Processing Time (min) |
|---|---|---|---|
| Control | 22.04 | Nil a | 0 |
| Manual grinding | 44.6 | ND b | 2 |
| Bead vortexing | 22.8 | 48 | 20 |
| Osmotic shock | 48.7 | 4.8 c | 2 |
| Water bath | 20.8 | 2400 | 20 |
| Sonication | 31.05 | 1200 | 20 |
| Shake mill | 30.5 | 690 | 5 |
3. Experimental Section
3.1. Chemicals
3.2. Strain Selection and Biomass Production
3.3. Lipid Extraction from Thraustochytrids by Organic Solvents
3.4. Cell Disruption for Lipid Extraction
3.4.1. Grinding with Liquid Nitrogen
3.4.2. Bead Vortexing
3.4.3. Sonication
3.4.4. Osmotic Shock
3.4.5. Water Bath
3.4.6. Shake Mill
3.5. Fatty Acids Methyl Esters (FAMEs) Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nogueira, L.A.H. Does biodiesel make sense? Energy 2011, 36, 3659–3666. [Google Scholar]
- Meher, L.C.; Vidya Sagar, D.; Naik, S.N. Technical aspects of biodiesel production by transesterification-A review. Renew. Sustainable Energy Rev. 2006, 10, 248–268. [Google Scholar]
- Lin, L.; Cunshan, Z.; Vittayapadung, S.; Xiangqian, S.; Mingdong, D. Opportunities and challenges for biodiesel fuel. Appl. Energy 2011, 88, 1020–1031. [Google Scholar]
- Chisti, Y. Biodiesel from microalgae. Biotech. Adv. 2007, 25, 294–306. [Google Scholar]
- Silitonga, A.S.; Masjuki, H.H.; Mahlia, T.M.I.; Ong, H.C.; Chong, W.T.; Boosroh, M.H. Overview properties of biodiesel diesel blends from edible and non-edible feedstock. Renew. Sustain. Energy Rev. 2013, 22, 346–360. [Google Scholar]
- Singh, A.; Nigam, P.S.; Murphy, J.D. Renewable fuels from algae: An answer to debatable land based fuels. Bioresour. Technol. 2011, 102, 10–16. [Google Scholar] [PubMed]
- McMillan, J.R.; Watson, I.A.; Ali, M.; Jaafar, W. Evaluation and comparison of algal cell disruption methods: Microwave, waterbath, blender, ultrasonic and laser treatment. Appl. Energy 2013, 103, 128–134. [Google Scholar] [CrossRef]
- Griffiths, M.J.; Harrison, S.T.L. Lipid productivity as a key characteristic for choosing algal species for biodiesel production. J. Appl. Phycol. 2009, 21, 493–507. [Google Scholar] [CrossRef]
- Lee, J.-Y.; Yoo, C.; Jun, S.-Y.; Ahn, C.-Y.; Oh, H.-M. Comparison of several methods for effective lipid extraction from microalgae. Bioresour. Technol. 2010, 101 (Suppl. 1), S75–S77. [Google Scholar] [CrossRef] [PubMed]
- Lewis, T.; Nichols, P.D.; McMeekin, T.A. Evaluation of extraction methods for recovery of fatty acids from lipid-producing microheterotrophs. J. Microbiol. Methods 2000, 43, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Yoon, B.-D.; Oh, H.-M. Rapid method for the determination of lipid from the green alga Botryococcus braunii. Biotechnol. Tech. 1998, 12, 553–556. [Google Scholar] [CrossRef]
- Ryckebosch, E.; Muylaert, K.; Foubert, I. Optimization of an Analytical Procedure for Extraction of Lipids from Microalgae. J. Am. Oil. Chem. Soc. 2012, 89, 189–198. [Google Scholar] [CrossRef]
- Ramluckan, K.; Moodley, K.G.; Bux, F. An evaluation of the efficacy of using selected solvents for the extraction of lipids from algal biomass by the soxhlet extraction method. Fuel 2014, 116, 103–108. [Google Scholar] [CrossRef]
- Kumar, R.R.; Rao, P.H.; Muthu, A. Lipid extraction methods from microalgae: A comprehensive review. Front. Energy Res. 2015, 2. [Google Scholar] [CrossRef]
- Jeon, J.-M.; Choi, H.-W.; Yoo, G.-C.; Choi, Y.-K.; Choi, K.-Y.; Park, H.-Y.; Park, S.-H.; Kim, Y.-G.; Kim, H.J.; Lee, S.H.; et al. New mixture composition of organic solvents for efficient extraction of lipids from Chlorella vulgaris. Biomass Bioenergy 2013, 59, 279–284. [Google Scholar] [CrossRef]
- Halim, R.; Harun, R.; Danquah, M.K.; Webley, P.A. Microalgal cell disruption for biofuel development. Appl. Energy 2012, 91, 116–121. [Google Scholar] [CrossRef]
- Zheng, H.; Yin, J.; Gao, Z.; Huang, H.; Ji, X.; Dou, C. Disruption of Chlorella vulgaris cells for the release of biodiesel-producing lipids: A comparison of grinding, ultrasonication, bead milling, enzymatic lysis, and microwaves. Appl. Biochem. Biotechnol. 2011, 164, 1215–1224. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.; Han, J.-I. Simultaneous treatment (cell disruption and lipid extraction) of wet microalgae using hydrodynamic cavitation for enhancing the lipid yield. Bioresour. Technol. 2015, 186, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Singh, D.; Barrow, C.J.; Puri, M. Exploring potential use of Australian thraustochytrids for the bioconversion of glycerol to omega-3 and carotenoids production. Biochem. Eng. J. 2013, 78, 11–17. [Google Scholar] [CrossRef]
- Vello, V.; Phang, S.-M.; Chu, W.-L.; Abdul Majid, N.; Lim, P.-E.; Loh, S.-K. Lipid productivity and fatty acid composition-guided selection of Chlorella strains isolated from Malaysia for biodiesel production. J. Appl. Phycol. 2014, 26, 1399–1413. [Google Scholar] [CrossRef]
- Rodolfi, L.; Chini Zittelli, G.; Bassi, N.; Padovani, G.; Biondi, N.; Bonini, G.; Tredici, M.R. Microalgae for oil: Strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol. Bioeng. 2009, 102, 100–112. [Google Scholar] [CrossRef] [PubMed]
- Salama, E.-S.; Kim, H.-C.; Abou-Shanab, R.I.; Ji, M.-K.; Oh, Y.-K.; Kim, S.-H.; Jeon, B.-H. Biomass, lipid content, and fatty acid composition of freshwater Chlamydomonas mexicana and Scenedesmus obliquus grown under salt stress. Bioprocess Biosyst. Eng. 2013, 36, 827–833. [Google Scholar] [CrossRef] [PubMed]
- Taoka, Y.; Nagano, N.; Okita, Y.; Izumida, H.; Sugimoto, S.; Hayashi, M. Effect of Tween 80 on the growth, lipid accumulation and fatty acid composition of Thraustochytrium aureum ATCC 34304. J. Biosci. Bioeng. 2011, 111, 420–424. [Google Scholar] [CrossRef] [PubMed]
- Olofsson, M.; Lamela, T.; Nilsson, E.; Bergé, J.-P.; del Pino, V.; Uronen, P.; Legrand, C. Combined effects of nitrogen concentration and seasonal changes on the production of lipids in Nannochloropsis oculata. Mar. Drugs 2014, 12, 1891–1910. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Schuur, B.; Kersten, S.R.A.; Brilman, D.W.F. Opportunities for switchable solvents for lipid extraction from wet algal biomass: An energy evaluation. Algal. Res 2015, 11, 271–283. [Google Scholar] [CrossRef]
- Kale, A. Method of Extracting Neutral Lipids with Two Solvents. Patent WO2013142694 A1, 26 September 2013. [Google Scholar]
- Shen, Y.; Pei, Z.; Yuan, W.; Mao, E. Effect of nitrogen and extraction method on algae lipid yield. Int. J. Agric. Biol. Eng. 2009, 2, 51–57. [Google Scholar]
- Dos Santos, R.R.; Moreira, D.M.; Kunigami, C.N.; Aranda, D.A.G.; Teixeira, C.M.L.L. Comparison between several methods of total lipid extraction from Chlorella vulgaris biomass. Ultrason. Sonochem. 2015, 22, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Puri, M.; Wilkens, S.; Mathur, A.S.; Tuli, D.K.; Barrow, C.J. Characterization of a new zeaxanthin producing strain of Chlorella saccharophila isolated from New Zealand marine waters. Bioresour. Technol. 2013, 143, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Li, Y.; Hu, X.; Su, W.; Zhong, M. Combined enzymatic and mechanical cell disruption and lipid extraction of green alga Neochloris oleoabundans. Int. J. Mol. Sci. 2015, 16, 7707–7722. [Google Scholar] [CrossRef] [PubMed]
- Prabakaran, P.; Ravindran, A.D. A comparative study on effective cell disruption methods for lipid extraction from microalgae. Lett. Appl. Microbiol. 2011, 53, 150–154. [Google Scholar] [CrossRef] [PubMed]
- Yoo, G.; Park, W.-K.; Kim, C.W.; Choi, Y.-E.; Yang, J.-W. Direct lipid extraction from wet Chlamydomonas reinhardtii biomass using osmotic shock. Bioresour. Technol. 2012, 123, 717–722. [Google Scholar] [CrossRef] [PubMed]
- Schutte, H.; Kula, M.R. Pilot-scale and process-scale techniques for cell disruption. Biotechnol. Appl. Biochem. 1990, 12, 599–620. [Google Scholar] [PubMed]
- Chisti, Y.; Moo-Young, M. Disruption of microbial cells for intracellular products. Enzyme Microb. Tech. 1986, 8, 194–204. [Google Scholar] [CrossRef]
- Arnal, J.M.; Sancho, M.; Iborra, I.; Gozálvez, J.M.; Santafé, A.; Lora, J. Concentration of brines from RO desalination plants by natural evaporation. Desalination 2005, 182, 435–439. [Google Scholar] [CrossRef]
- Sauer, T.; Galinski, E.A. Bacterial milking: A novel bioprocess for production of compatible solutes. Biotechnol. Bioeng. 1998, 57, 306–313. [Google Scholar] [CrossRef] [PubMed]
- Kar, J.R.; Singhal, R.S. Investigations on ideal mode of cell disruption in extremely halophilic Actinopolyspora halophila (MTCC 263) for efficient release of glycine betaine and trehalose. Biotechnol. Rep. 2015, 5, 89–97. [Google Scholar] [CrossRef]
- Cao, Y.; Liu, W.; Xu, X.; Zhang, H.; Wang, J.; Xian, M. Production of free monounsaturated fatty acids by metabolically engineered Escherichia coli. Biotechnol. Biofuels 2014, 7. [Google Scholar] [CrossRef] [PubMed]
- Hoekman, S.K.; Broch, A.; Robbins, C.; Ceniceros, E.; Natarajan, M. Review of biodiesel composition, properties, and specifications. Renew. Sustain. Energy Rev. 2012, 16, 143–169. [Google Scholar] [CrossRef]
- Ryckebosch, E.; Bruneel, C.; Muylaert, K.; Foubert, I. Microalgae as an alternative source of omega-3 long chain polyunsaturated fatty acids. Lipid Tech. 2012, 24, 128–130. [Google Scholar] [CrossRef]
- Singh, D.; Mathur, A.S.; Tuli, D.K.; Puri, M.; Barrow, C.J. Propyl gallate and butylated hydroxytoluene influence the accumulation of saturated fatty acids, omega-3 fatty acid and carotenoids in thraustochytrids. J. Funct. Foods 2015, 15, 186–192. [Google Scholar] [CrossRef]
- Talebi, A.F.; Mohtashami, S.K.; Tabatabaei, M.; Tohidfar, M.; Bagheri, A.; Zeinalabedini, M.; Hadavand Mirzaei, H.; Mirzajanzadeh, M.; Malekzadeh Shafaroudi, S.; Bakhtiari, S. Fatty acids profiling: A selective criterion for screening microalgae strains for biodiesel production. Algal Res. 2013, 2, 258–267. [Google Scholar] [CrossRef]
- Islam, M.A.; Magnusson, M.; Brown, R.J.; Ayoko, G.A.; Nabi, M.N.; Heimann, K. Microalgal species selection for biodiesel production based on fuel properties derived from fatty acid profiles. Energies 2013, 6, 5676–5702. [Google Scholar] [CrossRef]
- Martinez-Guerra, E.; Gude, V.V.; Mondala, A.; Holmes, W.; Hernandez, R. Microwave and ultrasound enhanced extractive-transesterification of algal lipids. Appl. Energy 2014, 129, 354–363. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Physiol. Pharm. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Gupta, A.; Vongsvivut, J.; Barrow, C.J.; Puri, M. Molecular identification of marine yeast and its spectroscopic analysis establishes unsaturated fatty acid accumulation. J. Biosci. Bioeng. 2012, 114, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Online osmotic pressure calculation hosted by Georgia State University, Atlanta, GA, USA. Available online: http://hyperphysics.phy-astr.gsu.edu/hbase/kinetic/ospcal.html (accessed on 19 July 2015).
- Christie, W.W.; Han, X. Lipid Analysis: Lipid Isolation, Separation, Identification and Lipidomic Analysis; Oily Press: Bridgewater, England, UK, 2010. [Google Scholar]
- Ackman, R.G. The gas chromatograph in practical analyses of common and uncommon fatty acids for the 21st century. Anal. Chim. Acta 2002, 465, 175–192. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Byreddy, A.R.; Gupta, A.; Barrow, C.J.; Puri, M. Comparison of Cell Disruption Methods for Improving Lipid Extraction from Thraustochytrid Strains. Mar. Drugs 2015, 13, 5111-5127. https://doi.org/10.3390/md13085111
Byreddy AR, Gupta A, Barrow CJ, Puri M. Comparison of Cell Disruption Methods for Improving Lipid Extraction from Thraustochytrid Strains. Marine Drugs. 2015; 13(8):5111-5127. https://doi.org/10.3390/md13085111
Chicago/Turabian StyleByreddy, Avinesh R., Adarsha Gupta, Colin J. Barrow, and Munish Puri. 2015. "Comparison of Cell Disruption Methods for Improving Lipid Extraction from Thraustochytrid Strains" Marine Drugs 13, no. 8: 5111-5127. https://doi.org/10.3390/md13085111
APA StyleByreddy, A. R., Gupta, A., Barrow, C. J., & Puri, M. (2015). Comparison of Cell Disruption Methods for Improving Lipid Extraction from Thraustochytrid Strains. Marine Drugs, 13(8), 5111-5127. https://doi.org/10.3390/md13085111






