Signal Transducers and Activators of Transcription (STAT) Regulatory Networks in Marine Organisms: From Physiological Observations towards Marine Drug Discovery
Abstract
:1. Introduction
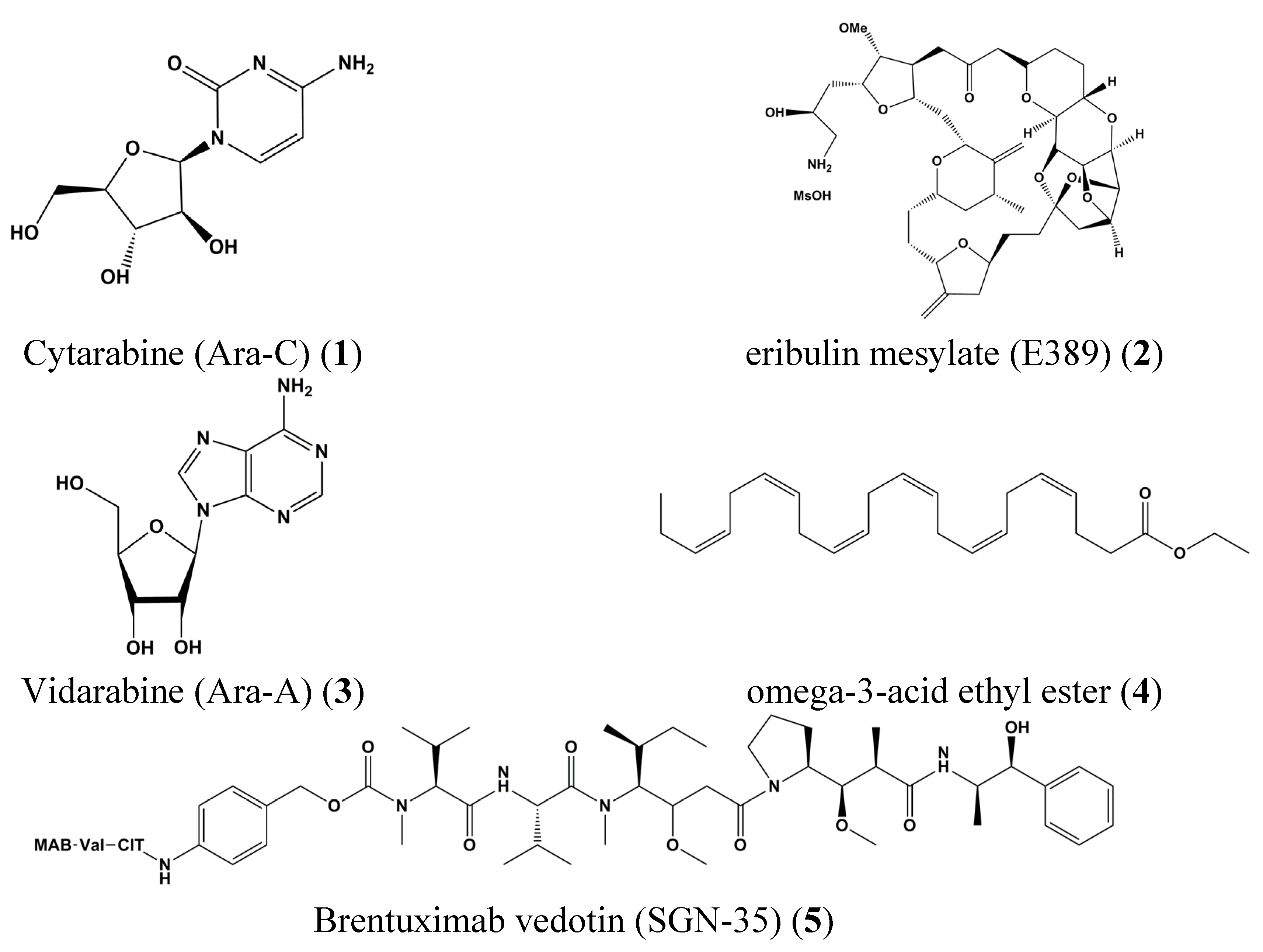
2. Past and Present of Drug Discovery in the Field of Cancer and Marine Influences
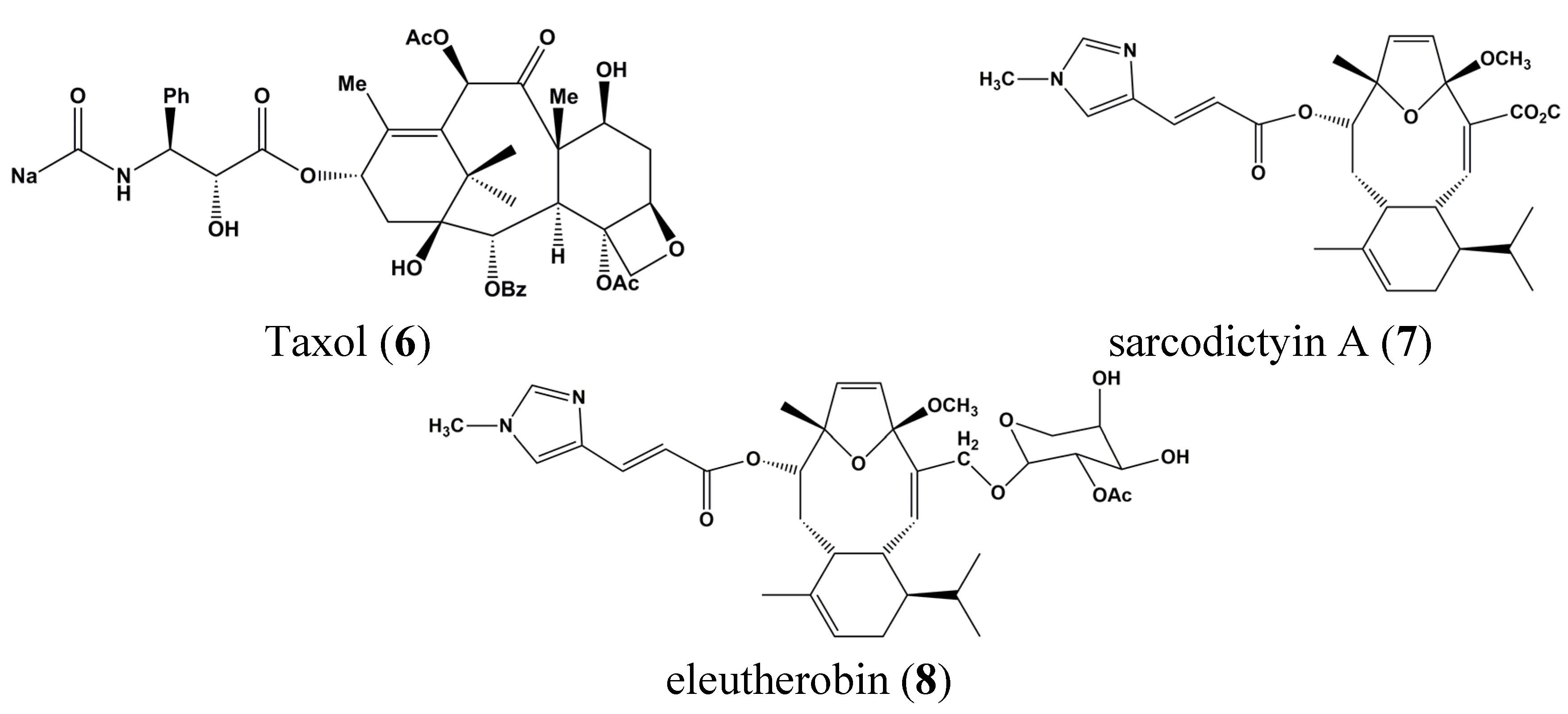
3. Clinical Trials of Marine-Derived Anticancer Drugs
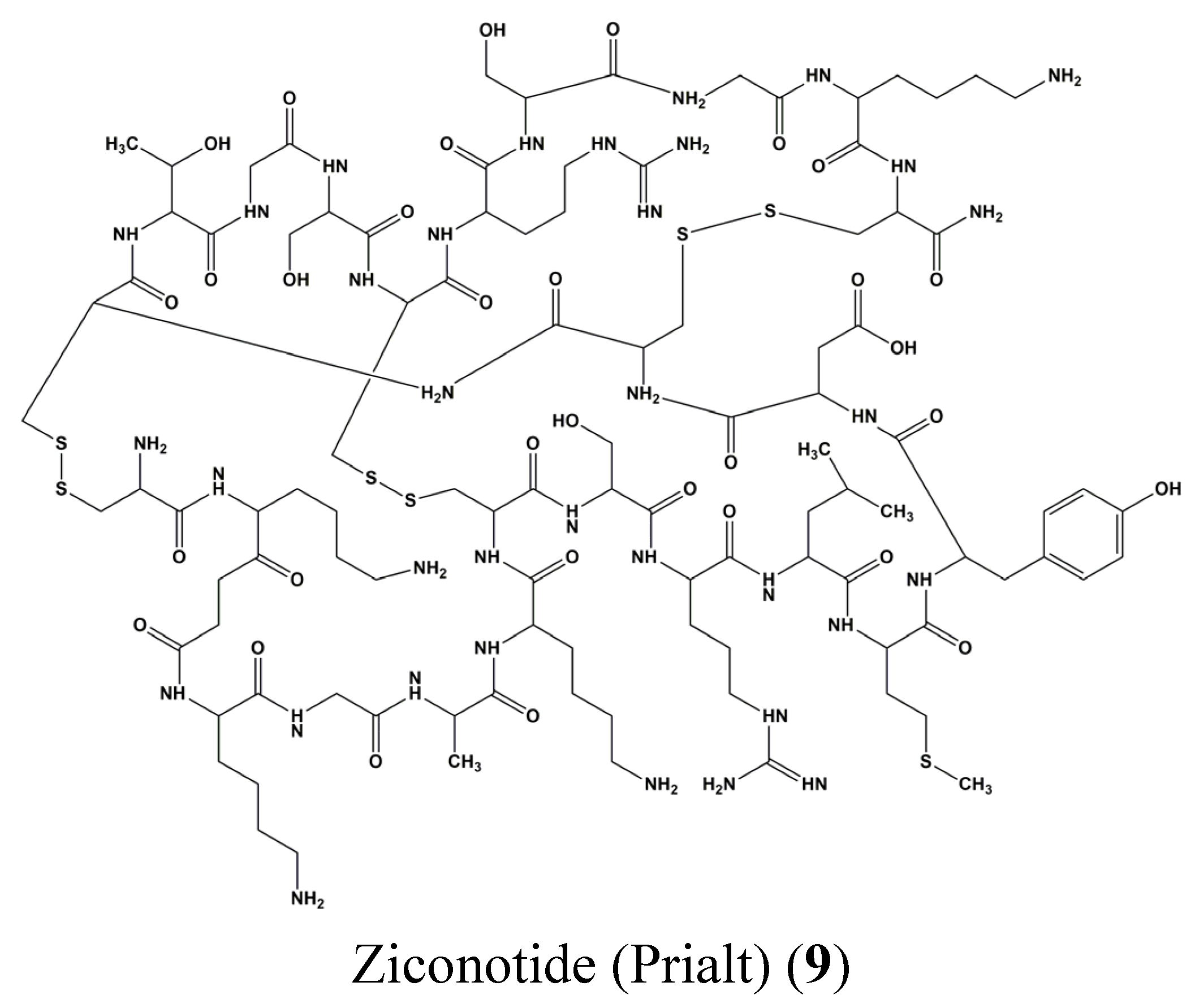
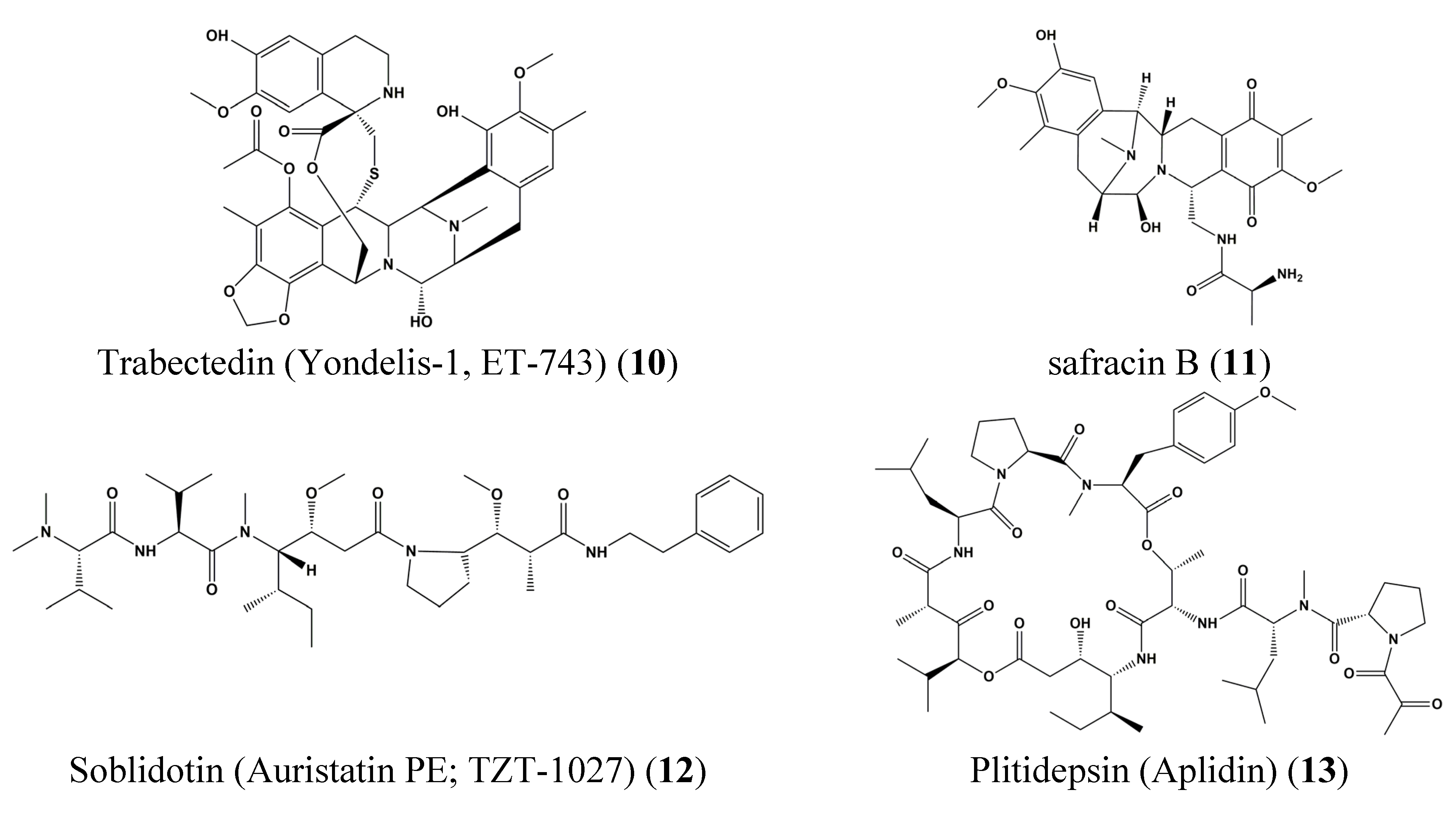
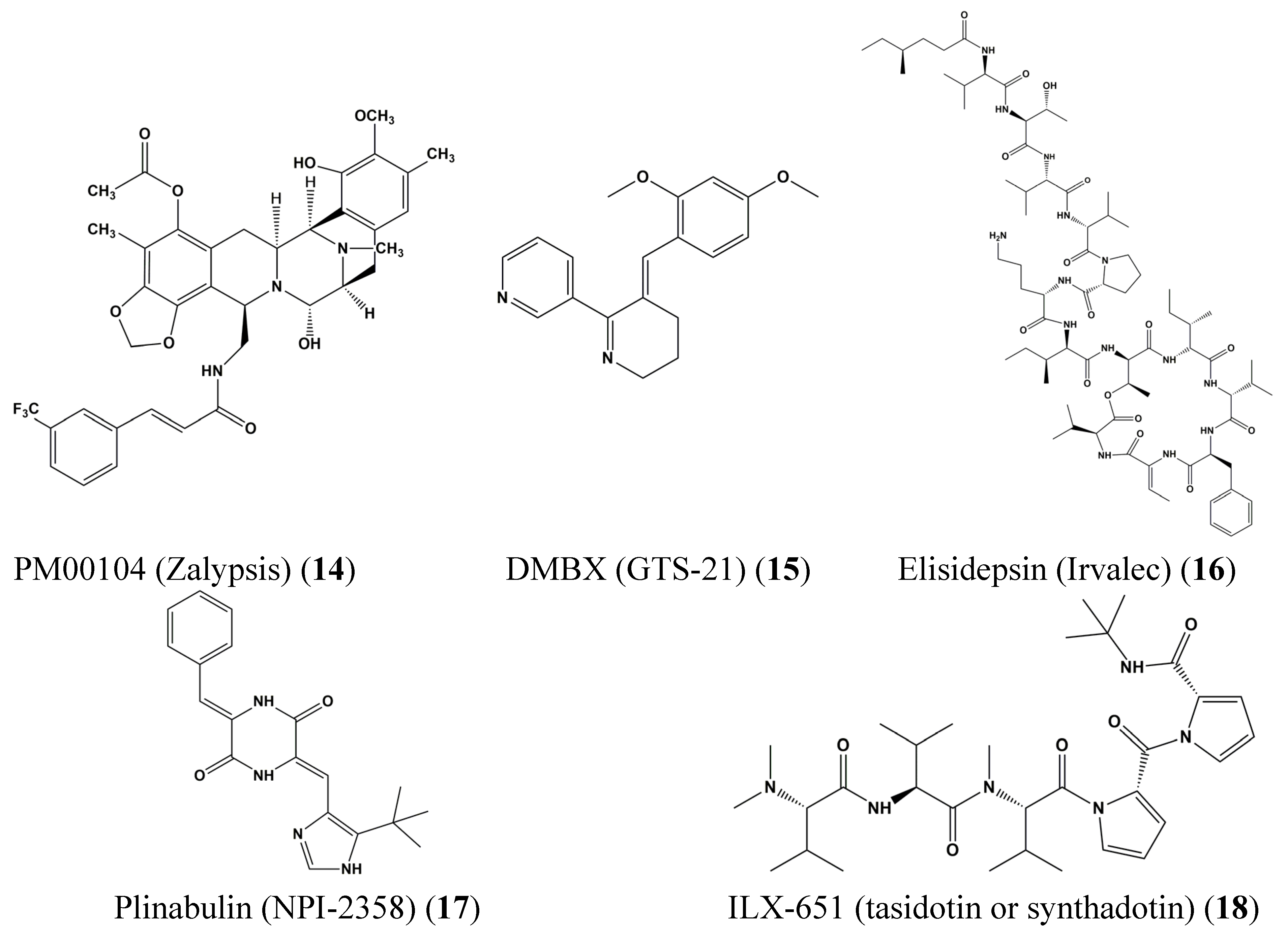
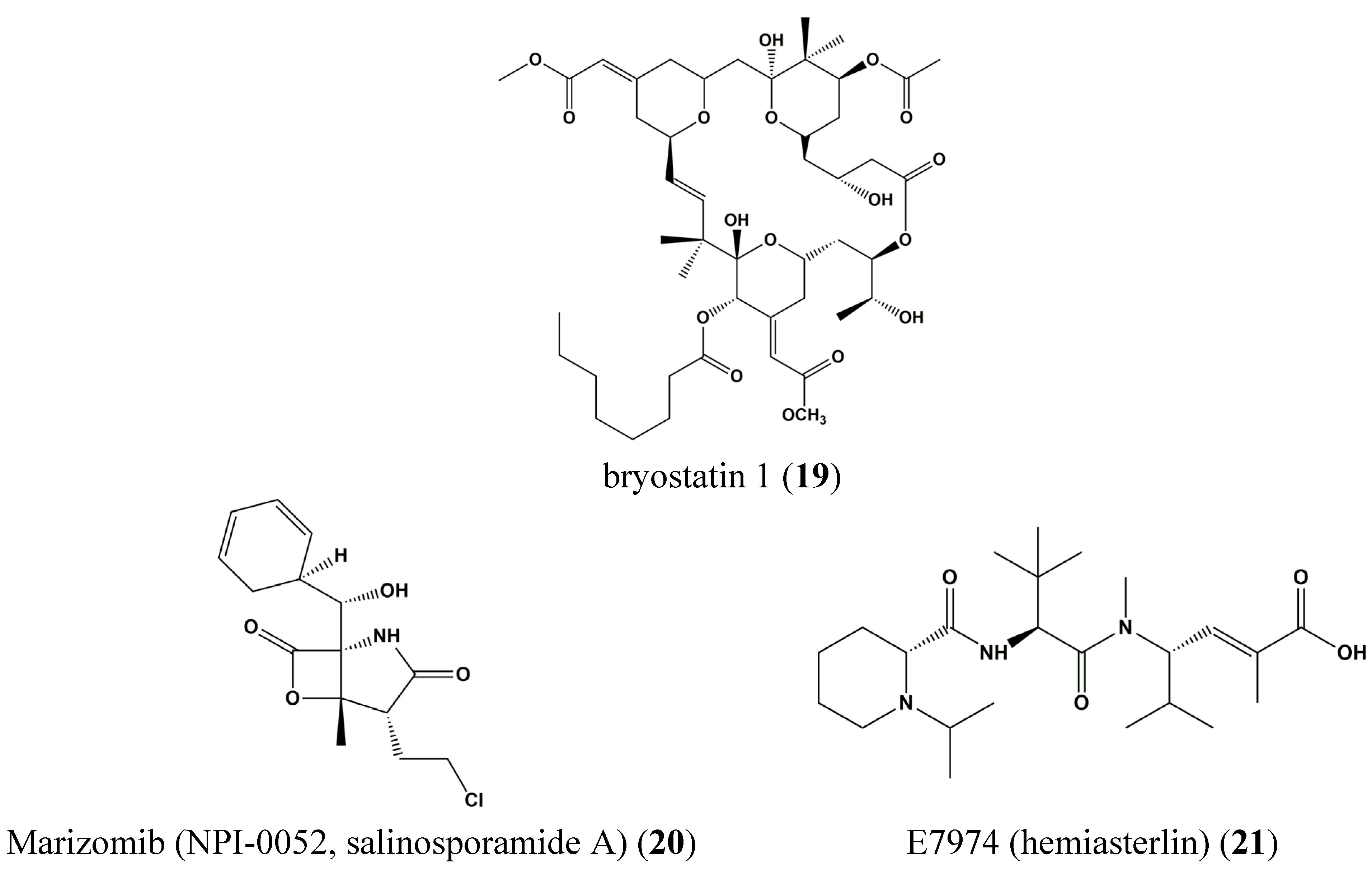
4. STAT Signaling in Health and Disease
4.1. JAK/STAT Signaling Pathways
4.2. JAK/STAT Signaling in Marine Organisms
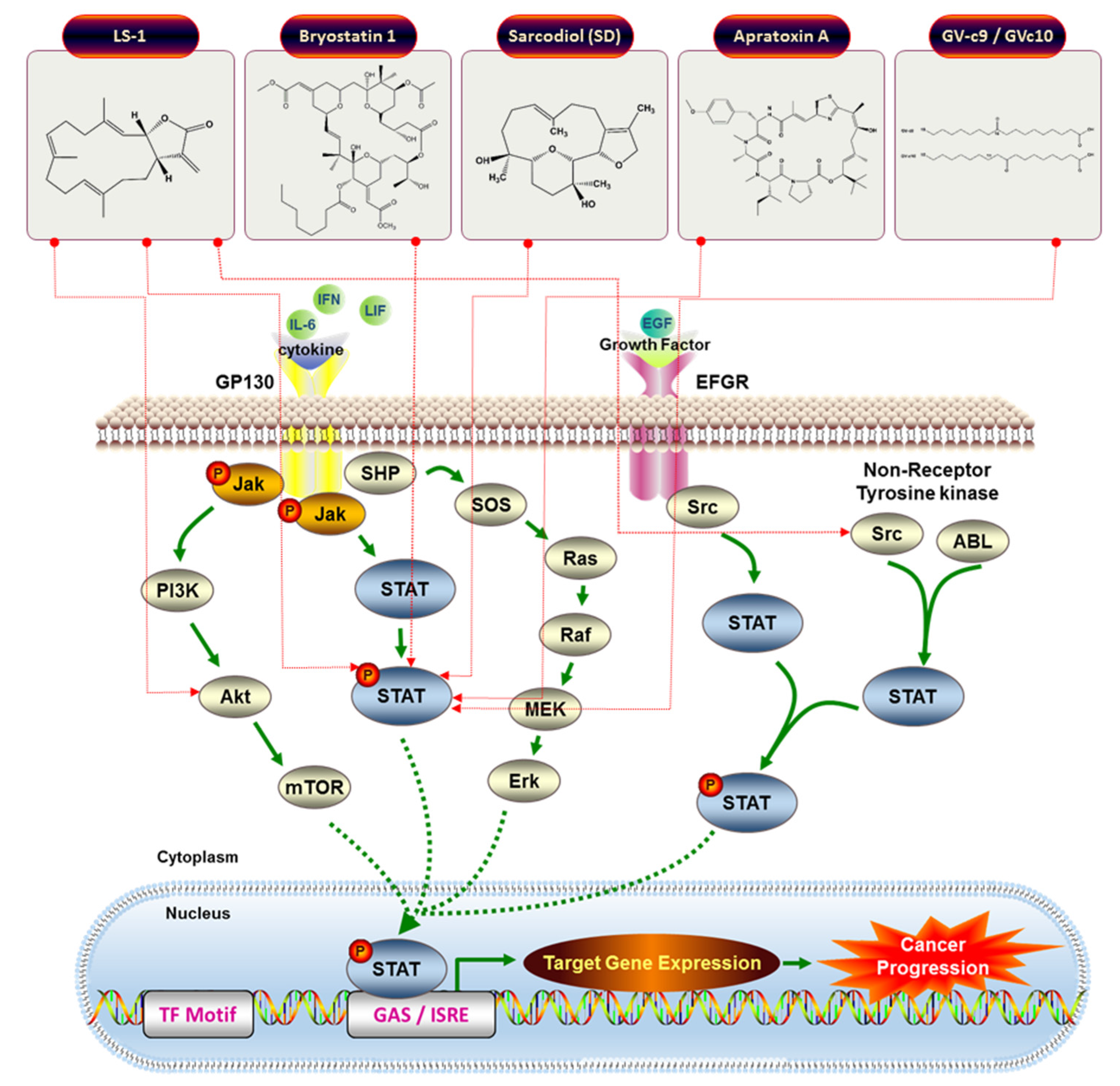
4.3. JAK/STAT-Inhibiting Anticancer Drugs from Marine Organisms
| N | Compound(s) | Disease model | Cell lines | Effect observed | STAT protein | Reference |
|---|---|---|---|---|---|---|
| 1 | Cytarabine (Ara-C) | Leukemia | K562 | Inhibition of STAT5 tyrosine phosphorylation | STAT5 | [26] |
| 15 | DMXBA (GTS-21) | Preadipocyte | 3T3-L1 | Reduction ASP-mediated chemokine MCP-1 secretion | STAT3 | [38] |
| 19 | Bryostatin 1 | Blood cancer | Chronic lymphocytic leukemia isolates cells | Induction of tyrosine phosphorylation, DNA binding of STAT1 | STAT1 | [70] |
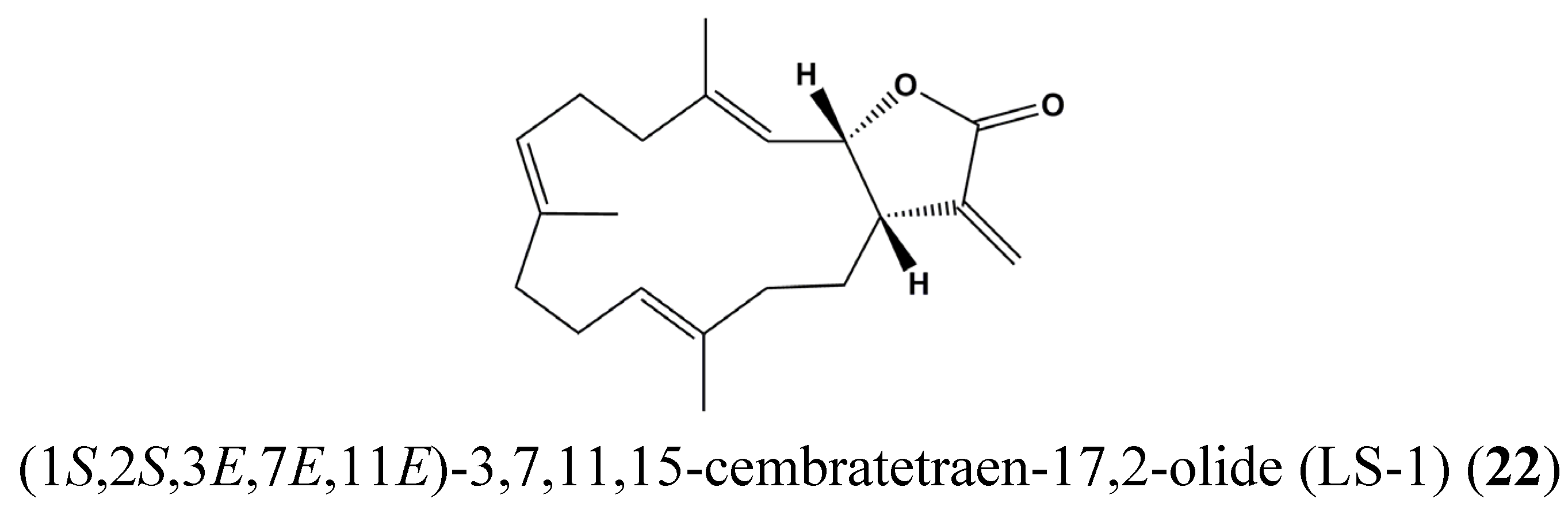
| 22 | (1S,2S,3E,7E,11E)-3,7,11,15-Cembratetraen-17,2-olide (LS-1) | Colon cancer | HT-29 | Disruption of mitochondrial membrane potential, ROS generation, Cell cycle arrest, De-phosphorylation of STAT3 | STAT3 | [71] |
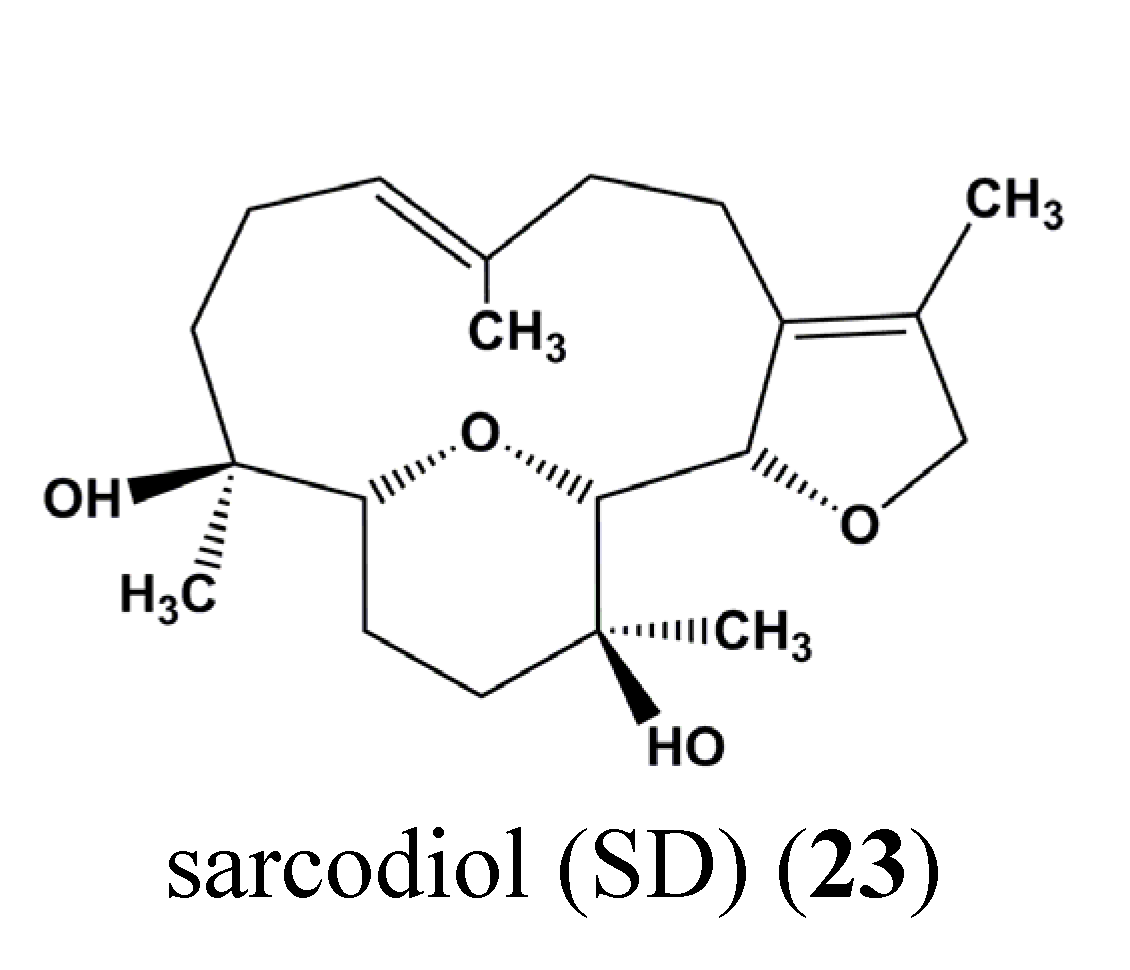
| 23 | Sarcodiol (SD) | Skin cancer | B16F10 | Inhibition of de novo DNA Synthesis, Induction of DNA fragmentation, Inhibition of STAT3 | STAT3 | [72] |
| 24 | Apratoxin A | Osteosarcoma, Breast cancer | U2OS, MCF7 | Inhibition of STAT3 tyrosine phosphorylation. Gp130 degradation | STAT3 | [73] |
| 25 | GV-c9, and GV-c10 | Macrophage | Raw 264.7 | Inhibition of inflammatory markers (IL-6, TNF-alpha, and nitric oxide) | STAT1 | [74] |


5. Conclusions and Perspectives
Acknowledgments
Conflicts of Interest
Abbreviations
| ALL | Acute lymphocytic leukemia |
| AML | Acute myelogenous leukemia |
| Bcr-Abl | Break point cluster–Abelson |
| CLL | Chronic lymphocytic leukemia |
| DNA | Deoxyribonucleic Acid |
| EGFR | Epidermal growth factor receptor |
| EMEA | European Medicines Agency |
| EsSOCS | Eriocheir sinensis suppressors of cytokine signaling |
| INF | interferon |
| IL | interleukin |
| JAK | Janus kinase |
| JfGPH | Japanese flounder glycoprotein 130 homologue |
| JNK | c-Jun N-terminal kinase |
| NCI | National Cancer Institute |
| NF-κB | nuclear factor-κB |
| p53 | tumor suppressor protein 53 |
| RbSTAT4 | Single transducer and activator of transcription 4 family identified from rock bream |
| ROS | reactive oxygen species |
| SD | Sarcodiol |
| SmSOCS3 | Scophthalmus maximus suppressors of cytokine signaling 3 |
| SOCS2 | suppressors of cytokine signaling 2 |
| STAT | Single transducer and activator of transcription |
| WHO | World Health Organization |
| WSSV | White spot syndrome virus |
References
- Sawadogo, W.R.; Schumacher, M.; Teiten, M.H.; Dicato, M.; Diederich, M. Traditional west african pharmacopeia, plants and derived compounds for cancer therapy. Biochem. Pharmacol. 2012, 84, 1225–1240. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, M.; Kelkel, M.; Dicato, M.; Diederich, M. A survey of marine natural compounds and their derivatives with anti-cancer activity reported in 2010. Molecules 2011, 16, 5629–5646. [Google Scholar] [CrossRef] [PubMed]
- Sawadogo, W.R.; Schumacher, M.; Teiten, M.H.; Cerella, C.; Dicato, M.; Diederich, M. A survey of marine natural compounds and their derivatives with anti-cancer activity reported in 2011. Molecules 2013, 18, 3641–3673. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, M.; Juncker, T.; Schnekenburger, M.; Gaascht, F.; Diederich, M. Natural compounds as inflammation inhibitors. Genes Nutr. 2011, 6, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Folmer, F.; Dicato, M.; Diederich, M. From the deepest sea shelf to the uppermost kitchen cabinet shelf: The quest for novel TNF-α inhibitors. Curr. Top Med. Chem. 2012, 12, 1392–1407. [Google Scholar] [CrossRef] [PubMed]
- Cragg, G.M.; Newman, D.J. Natural products: A continuing source of novel drug leads. Biochim. Biophys. Acta Gen. Subj. 2013, 1830, 3670–3695. [Google Scholar] [CrossRef] [PubMed]
- Teiten, M.H.; Gaascht, F.; Dicato, M.; Diederich, M. Anticancer bioactivity of compounds from medicinal plants used in European medieval traditions. Biochem. Pharmacol. 2013, 86, 1239–1247. [Google Scholar] [CrossRef] [PubMed]
- Cerella, C.; Teiten, M.H.; Radogna, F.; Dicato, M.; Diederich, M. From nature to bedside: Pro-survival and cell death mechanisms as therapeutic targets in cancer treatment. Biotechnol. Adv. 2014, 32, 1111–1122. [Google Scholar] [CrossRef] [PubMed]
- Folmer, F.; Jaspars, M.; Dicato, M.; Diederich, M. Marine natural products as targeted modulators of the transcription factor NF-κB. Biochem. Pharmacol. 2008, 75, 603–617. [Google Scholar] [CrossRef] [PubMed]
- Folmer, F.; Jaspars, M.; Solano, G.; Cristofanon, S.; Henry, E.; Tabudravu, J.; Black, K.; Green, D.H.; Kupper, F.C.; Aalbersberg, W.; et al. The inhibition of TNF-α-induced NF-κB activation by marine natural products. Biochem. Pharmacol. 2009, 78, 592–606. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M.; Battershill, C.N. Therapeutic agents from the sea: Biodiversity, chemo-evolutionary insight and advances to the end of Darwin’s 200th year. Diving Hyperb. Med. J. 2009, 39, 216–225. [Google Scholar]
- Folmer, F.; Jaspars, M.; Schumacher, M.; Dicato, M.; Diederich, M. Marine natural products targeting phospholipases A2. Biochem. Pharmacol. 2010, 80, 1793–1800. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, M.; Kelkel, M.; Dicato, M.; Diederich, M. Gold from the sea: Marine compounds as inhibitors of the hallmarks of cancer. Biotechnol. Adv. 2011, 29, 531–547. [Google Scholar] [CrossRef] [PubMed]
- Orlikova, B.; Legrand, N.; Panning, J.; Dicato, M.; Diederich, M. Anti-inflammatory and anticancer drugs from nature. Cancer Treat. Res. 2014, 159, 123–143. [Google Scholar] [PubMed]
- Schnekenburger, M.; Dicato, M.; Diederich, M. Epigenetic modulators from “the big blue”: A treasure to fight against cancer. Cancer Lett. 2014, 351, 182–197. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Mageed, W.M.; Milne, B.F.; Wagner, M.; Schumacher, M.; Sandor, P.; Pathom-aree, W.; Goodfellow, M.; Bull, A.T.; Horikoshi, K.; Ebel, R.; et al. Dermacozines, a new phenazine family from deep-sea dermacocci isolated from a Mariana Trench sediment. Org. Biomol. Chem. 2010, 8, 2352–2362. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.; Vieira, H.; Gaspar, H.; Santos, S. Marketed marine natural products in the pharmaceutical and cosmeceutical industries: Tips for success. Mar. Drugs 2014, 12, 1066–1101. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, M.; Cerella, C.; Eifes, S.; Chateauvieux, S.; Morceau, F.; Jaspars, M.; Dicato, M.; Diederich, M. Heteronemin, a spongean sesterterpene, inhibits TNFα -induced NF-κB activation through proteasome inhibition and induces apoptotic cell death. Biochem. Pharmacol. 2010, 79, 610–622. [Google Scholar] [CrossRef] [PubMed]
- Trecul, A.; Morceau, F.; Dicato, M.; Diederich, M. Dietary compounds as potent inhibitors of the signal transducers and activators of transcription (STAT) 3 regulatory network. Genes Nutr. 2012, 7, 111–125. [Google Scholar] [CrossRef] [PubMed]
- Cragg, G.M.; Newman, D.J.; Snader, K.M. Natural products in drug discovery and development. J. Nat. Prod. 1997, 60, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wu, Q.; Cui, G.H.; Chen, Y.Q.; Li, R. Deguelin blocks cells survival signal pathways and induces apoptosis of HL-60 cells in vitro. Int. J. Hematol. 2009, 89, 618–623. [Google Scholar] [CrossRef] [PubMed]
- Cragg, G.M. Paclitaxel (taxol): A success story with valuable lessons for natural product drug discovery and development. Med. Res. Rev. 1998, 18, 315–331. [Google Scholar] [CrossRef]
- van der Burg, M.E.; Onstenk, W.; Boere, I.A.; Look, M.; Ottevanger, P.B.; de Gooyer, D.; Kerkhofs, L.G.; Valster, F.A.; Ruit, J.B.; van Reisen, A.G.; et al. Long-term results of a randomised phase III trial of weekly versus three-weekly paclitaxel/platinum induction therapy followed by standard or extended three-weekly paclitaxel/platinum in European patients with advanced epithelial ovarian cancer. Eur. J. Cancer 2014, 50, 2592–2601. [Google Scholar] [CrossRef] [PubMed]
- Martin, M. Nab-paclitaxel dose and schedule in breast cancer. Breast Cancer Res. 2015, 17, 81. [Google Scholar] [CrossRef] [PubMed]
- Lammers, P.E.; Lu, B.; Horn, L.; Shyr, Y.; Keedy, V. Nab-paclitaxel in combination with weekly carboplatin with concurrent radiotherapy in stage III non-small cell lung cancer. Oncologist 2015, 20, 491–492. [Google Scholar] [CrossRef] [PubMed]
- Kindler, T.; Breitenbuecher, F.; Kasper, S.; Stevens, T.; Carius, B.; Gschaidmeier, H.; Huber, C.; Fischer, T. In Bcr-Abl-positive cells, STAT-5 tyrosine-phosphorylation integrates signals induced by imatinib mesylate and Ara-C. Leukemia 2003, 17, 999–1009. [Google Scholar] [CrossRef] [PubMed]
- Monga, M.; Sausville, E.A. Developmental therapeutics program at the NCI: Molecular target and drug discovery process. Leukemia 2002, 16, 520–526. [Google Scholar] [CrossRef] [PubMed]
- Nastrucci, C.; Cesario, A.; Russo, P. Anticancer drug discovery from the marine environment. Recent Pat. Anti-Cancer Drug Discov. 2012, 7, 218–232. [Google Scholar] [CrossRef]
- Pope, J.E.; Deer, T.R. Ziconotide: A clinical update and pharmacologic review. Expert Opin. Pharmacother. 2013, 14, 957–966. [Google Scholar] [CrossRef] [PubMed]
- Thomas, X. Chemotherapy of acute leukemia in adults. Expert Opin. Pharmacother. 2009, 10, 221–237. [Google Scholar] [CrossRef] [PubMed]
- Absalon, M.J.; Smith, F.O. Treatment strategies for pediatric acute myeloid leukemia. Expert Opin. Pharmacother. 2009, 10, 57–79. [Google Scholar] [CrossRef] [PubMed]
- Cuevas, C.; Francesch, A. Development of yondelis® (trabectedin, ET-743). A semisynthetic process solves the supply problem. Nat. Prod. Rep. 2009, 26, 322–337. [Google Scholar] [CrossRef] [PubMed]
- Erba, E.; Bergamaschi, D.; Bassano, L.; Damia, G.; Ronzoni, S.; Faircloth, G.T.; D’Incalci, M. Ecteinascidin-743 (ET-743), a natural marine compound, with a unique mechanism of action. Eur. J. Cancer 2001, 37, 97–105. [Google Scholar] [CrossRef]
- Mayer, A.M.; Glaser, K.B.; Cuevas, C.; Jacobs, R.S.; Kem, W.; Little, R.D.; McIntosh, J.M.; Newman, D.J.; Potts, B.C.; Shuster, D.E. The odyssey of marine pharmaceuticals: A current pipeline perspective. Trends Pharmacol. Sci. 2010, 31, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Yap, T.A.; Carden, C.P.; Kaye, S.B. Beyond chemotherapy: Targeted therapies in ovarian cancer. Nat. Rev. Cancer 2009, 9, 167–181. [Google Scholar] [CrossRef] [PubMed]
- Lippert, J.W., III. Vascular disrupting agents. Bioorg. Med. Chem. 2007, 15, 605–615. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, J.; Natsume, T.; Kobayashi, M. Comparison of the antivascular and cytotoxic activities of TZT-1027 (soblidotin) with those of other anticancer agents. Anticancer Drugs 2007, 18, 905–911. [Google Scholar] [PubMed]
- Wu, J.; Jiao, Z.Y.; Zhang, Z.; Tang, Z.H.; Zhang, H.H.; Lu, H.L.; Cianflone, K. Cross-talk between α7 nAChR-mediated cholinergic pathway and acylation stimulating protein signaling in 3T3-L1 adipocytes: Role of NFκB and STAT3. Biochem. Cell Biol. 2015, 93, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Freedman, R.; Olincy, A.; Buchanan, R.W.; Harris, J.G.; Gold, J.M.; Johnson, L.; Allensworth, D.; Guzman-Bonilla, A.; Clement, B.; Ball, M.P.; et al. Initial phase 2 trial of a nicotinic agonist in schizophrenia. Am. J. Psychiatry 2008, 165, 1040–1047. [Google Scholar] [CrossRef] [PubMed]
- Kox, M.; van Velzen, J.F.; Pompe, J.C.; Hoedemaekers, C.W.; van der Hoeven, J.G.; Pickkers, P. GTS-21 inhibits pro-inflammatory cytokine release independent of the toll-like receptor stimulated via a transcriptional mechanism involving JAK2 activation. Biochem. Pharmacol. 2009, 78, 863–872. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.D.; Williams, R.M. Chemistry and biology of the tetrahydroisoquinoline antitumor antibiotics. Chem. Rev. 2002, 102, 1669–1730. [Google Scholar] [CrossRef] [PubMed]
- Leal, J.F.; Garcia-Hernandez, V.; Moneo, V.; Domingo, A.; Bueren-Calabuig, J.A.; Negri, A.; Gago, F.; Guillen-Navarro, M.J.; Aviles, P.; Cuevas, C.; et al. Molecular pharmacology and antitumor activity of zalypsis in several human cancer cell lines. Biochem. Pharmacol. 2009, 78, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Kuznetsov, G.; TenDyke, K.; Towle, M.J.; Cheng, H.; Liu, J.; Marsh, J.P.; Schiller, S.E.; Spyvee, M.R.; Yang, H.; Seletsky, B.M.; et al. Tubulin-based antimitotic mechanism of E7974, a novel analogue of the marine sponge natural product hemiasterlin. Mol. Cancer Ther. 2009, 8, 2852–2860. [Google Scholar] [CrossRef] [PubMed]
- Feling, R.H.; Buchanan, G.O.; Mincer, T.J.; Kauffman, C.A.; Jensen, P.R.; Fenical, W. Salinosporamide A: A highly cytotoxic proteasome inhibitor from a novel microbial source, a marine bacterium of the new genus Salinospora. Angew. Chem. Int. Ed. Engl. 2003, 42, 355–357. [Google Scholar] [CrossRef] [PubMed]
- Fenical, W.; Jensen, P.R.; Palladino, M.A.; Lam, K.S.; Lloyd, G.K.; Potts, B.C. Discovery and development of the anticancer agent salinosporamide A (NPI-0052). Bioorg. Med. Chem. 2009, 17, 2175–2180. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, D.; Hideshima, T.; Anderson, K.C. A novel proteasome inhibitor NPI-0052 as an anticancer therapy. Br. J. Cancer 2006, 95, 961–965. [Google Scholar] [CrossRef] [PubMed]
- Shuai, K.; Stark, G.R.; Kerr, I.M.; Darnell, J.E., Jr. A single phosphotyrosine residue of STAT91 required for gene activation by interferon-gamma. Science 1993, 261, 1744–1746. [Google Scholar] [CrossRef] [PubMed]
- Darnell, J.E., Jr.; Kerr, I.M.; Stark, G.R. JAK-STAT pathways and transcriptional activation in response to IFNs and other extracellular signaling proteins. Science 1994, 264, 1415–1421. [Google Scholar] [CrossRef] [PubMed]
- Yeh, T.C.; Pellegrini, S. The Janus kinase family of protein tyrosine kinases and their role in signaling. Cell Mol. Life Sci. 1999, 55, 1523–1534. [Google Scholar] [CrossRef] [PubMed]
- Yamaoka, K.; Saharinen, P.; Pesu, M.; Holt, V.E., III; Silvennoinen, O.; O’Shea, J.J. The Janus kinases (Jaks). Genome Biol. 2004, 5, 253. [Google Scholar] [CrossRef] [PubMed]
- Shuai, K.; Horvath, C.M.; Huang, L.H.; Qureshi, S.A.; Cowburn, D.; Darnell, J.E., Jr. Interferon activation of the transcription factor STAT91 involves dimerization through SH2-phosphotyrosyl peptide interactions. Cell 1994, 76, 821–828. [Google Scholar] [CrossRef]
- Sadowski, H.B.; Shuai, K.; Darnell, J.E., Jr.; Gilman, M.Z. A common nuclear signal transduction pathway activated by growth factor and cytokine receptors. Science 1993, 261, 1739–1744. [Google Scholar] [CrossRef] [PubMed]
- Darnell, J.E., Jr. Stats and gene regulation. Science 1997, 277, 1630–1635. [Google Scholar] [CrossRef] [PubMed]
- Schindler, C.; Darnell, J.E., Jr. Transcriptional responses to polypeptide ligands: The JAK-STAT pathway. Annu. Rev. Biochem. 1995, 64, 621–651. [Google Scholar] [CrossRef] [PubMed]
- Bromberg, J.F. Activation of STAT proteins and growth control. Bioessays 2001, 23, 161–169. [Google Scholar] [CrossRef]
- Takeda, K.; Akira, S. STAT family of transcription factors in cytokine-mediated biological responses. Cytokine Growth Factor Rev. 2000, 11, 199–207. [Google Scholar] [CrossRef]
- Williams, J.G. STAT signalling in cell proliferation and in development. Curr. Opin. Genet. Dev. 2000, 10, 503–507. [Google Scholar] [CrossRef]
- Ihle, J.N. Cytokine receptor signalling. Nature 1995, 377, 591–594. [Google Scholar] [CrossRef] [PubMed]
- Imada, K.; Leonard, W.J. The JAK-STAT pathway. Mol. Immunol. 2000, 37, 1–11. [Google Scholar] [CrossRef]
- Kotenko, S.V.; Pestka, S. JAK-STAT signal transduction pathway through the eyes of cytokine class II receptor complexes. Oncogene 2000, 19, 2557–2565. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.X.; Leonard, W.J. The role of Stat5a and Stat5b in signaling by IL-2 family cytokines. Oncogene 2000, 19, 2566–2576. [Google Scholar] [CrossRef] [PubMed]
- Smithgall, T.E.; Briggs, S.D.; Schreiner, S.; Lerner, E.C.; Cheng, H.; Wilson, M.B. Control of myeloid differentiation and survival by Stats. Oncogene 2000, 19, 2612–2618. [Google Scholar] [CrossRef] [PubMed]
- Mui, A.L. The role of STATs in proliferation, differentiation, and apoptosis. Cell Mol. Life Sci. 1999, 55, 1547–1558. [Google Scholar] [CrossRef] [PubMed]
- Zeidler, M.P.; Bach, E.A.; Perrimon, N. The roles of the Drosophila JAK/STAT pathway. Oncogene 2000, 19, 2598–2606. [Google Scholar] [CrossRef] [PubMed]
- Ramana, C.V.; Chatterjee-Kishore, M.; Nguyen, H.; Stark, G.R. Complex roles of STAT1 in regulating gene expression. Oncogene 2000, 19, 2619–2627. [Google Scholar] [CrossRef] [PubMed]
- Hirano, T.; Ishihara, K.; Hibi, M. Roles of STAT3 in mediating the cell growth, differentiation and survival signals relayed through the IL-6 family of cytokine receptors. Oncogene 2000, 19, 2548–2556. [Google Scholar] [CrossRef] [PubMed]
- Dearolf, C.R. JAKs and STATs in invertebrate model organisms. Cell Mol. Life Sci. 1999, 55, 1578–1584. [Google Scholar] [CrossRef] [PubMed]
- Blasius, R.; Reuter, S.; Henry, E.; Dicato, M.; Diederich, M. Curcumin regulates signal transducer and activator of transcription (STAT) expression in K562 cells. Biochem. Pharmacol. 2006, 72, 1547–1554. [Google Scholar] [CrossRef] [PubMed]
- Blasius, R.; Dicato, M.; Diederich, M. Effect of curcumin treatment on protein phosphorylation in K562 cells. Ann. N. Y. Acad. Sci. 2007, 1095, 377–387. [Google Scholar] [CrossRef] [PubMed]
- Trecul, A.; Morceau, F.; Gaigneaux, A.; Orsini, M.; Chateauvieux, S.; Grandjenette, C.; Dicato, M.; Diederich, M. Polyphenol tri-vanillic ester 13c inhibits P-JAK2V617F and Bcr-Abl oncokinase expression in correlation with STAT3/STAT5 inactivation and apoptosis induction in human leukemia cells. Cancer Lett. 2013, 340, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Liongue, C.; O’Sullivan, L.A.; Trengove, M.C.; Ward, A.C. Evolution of JAK-STAT pathway components: Mechanisms and role in immune system development. PLoS ONE 2012, 7, e32777. [Google Scholar] [CrossRef] [PubMed]
- Vogl, C.; Shukla, P.; Ebersberger, I. Evolution of jak and stat proteins. In JAK-STAT Signaling: From Basics to Disease; Decker, T., Müller, M., Eds.; Springer: Vienna, Austria, 2012; pp. 99–114. [Google Scholar]
- Robertsen, B. The interferon system of Teleost fish. Fish Shellfish Immunol. 2006, 20, 172–191. [Google Scholar] [CrossRef] [PubMed]
- Santos, M.D.; Yasuike, M.; Kondo, H.; Hirono, I.; Aoki, T. A novel type-1 cytokine receptor from fish involved in the Janus kinase/signal transducers and activators of transcription (JAK/STAT) signal pathway. Mol. Immunol. 2007, 44, 3355–3363. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhao, J.; Zhang, H.; Gai, Y.; Wang, L.; Li, F.; Yang, J.; Qiu, L.; Song, L. The involvement of suppressors of cytokine signaling 2 (SOCS2) in immune defense responses of chinese mitten crab eriocheir sinensis. Dev. Comp. Immunol. 2010, 34, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Mu, Y.; Ding, F.; Cui, P.; Ao, J.; Hu, S.; Chen, X. Transcriptome and expression profiling analysis revealed changes of multiple signaling pathways involved in immunity in the large yellow croaker during aeromonas hydrophila infection. BMC Genom. 2010, 11, 506. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Xiao, Z.Z.; Sun, L. Suppressor of cytokine signaling 3 inhibits head kidney macrophage activation and cytokine expression in scophthalmus maximus. Dev. Comp. Immunol. 2011, 35, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Xiang, J. Signaling pathways regulating innate immune responses in shrimp. Fish Shellfish Immunol. 2013, 34, 973–980. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.G.; Huang, X.D.; Wang, Q.; Zhao, M.; Wu, S.Z.; He, M.X. Gene cloning and function analysis of cytokine-induced suppressor of cytokine signaling (SOCS) from pearl oyster pinctada fucata. Fish Shellfish Immunol. 2013, 34, 849–854. [Google Scholar] [CrossRef] [PubMed]
- Premachandra, H.K.; Elvitigala, D.A.; Bathige, S.D.; Whang, I.; Lee, Y.; de Zoysa, M.; Lee, J. Genomic structure and immunological response of an STAT4 family member from rock bream (Oplegnathus fasciatus). Fish Shellfish Immunol. 2013, 35, 1829–1837. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Zhang, Z.; Wang, S.; Li, H.; Zuo, H.; Xu, X.; Weng, S.; He, J.; Li, C. A janus kinase in the JAK/STAT signaling pathway from Litopenaeus vannamei is involved in antiviral immunity response. Fish Shellfish Immunol. 2015, 44, 662–673. [Google Scholar] [CrossRef] [PubMed]
- Battle, T.E.; Frank, D.A. STAT1 mediates differentiation of chronic lymphocytic leukemia cells in response to Bryostatin 1. Blood 2003, 102, 3016–3024. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.Y.; Boo, H.J.; Kang, J.I.; Kim, M.K.; Yoo, E.S.; Hyun, J.W.; Koh, Y.S.; Kim, G.Y.; Maeng, Y.H.; Hyun, C.L.; et al. (1S,2S,3E,7E,11E)-3,7,11,15-Cembratetraen-17,2-olide, a cembrenolide diterpene from soft coral Lobophytum sp., inhibits growth and induces apoptosis in human colon cancer cells through reactive oxygen species generation. Biol. Pharm. Bull. 2012, 35, 1054–1063. [Google Scholar] [CrossRef] [PubMed]
- Szymanski, P.T.; Kuppast, B.; Ahmed, S.A.; Khalifa, S.; Fahmy, H. Sarcophine-diol, a skin cancer chemopreventive agent, inhibits proliferation and stimulates apoptosis in mouse melanoma B16F10 cell line. Mar. Drugs 2012, 10, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Law, B.K.; Luesch, H. Apratoxin a reversibly inhibits the secretory pathway by preventing cotranslational translocation. Mol. Pharmacol. 2009, 76, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Dang, H.T.; Kang, G.J.; Yang, E.J.; Park, S.S.; Yoon, W.J.; Jung, J.H.; Kang, H.K.; Yoo, E.S. Two enone fatty acids isolated from Gracilaria verrucosa suppress the production of inflammatory mediators by down-regulating NF-kappaB and STAT1 activity in lipopolysaccharide-stimulated RAW 264.7 cells. Arch. Pharm. Res. 2009, 32, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Takada, K.; Takemoto, Y.; Yoshida, M.; Nogi, Y.; Okada, S.; Matsunaga, S. Gliotoxin analogues from a marine-derived fungus, Penicillium sp., and their cytotoxic and histone methyltransferase inhibitory activities. J. Nat. Prod. 2012, 75, 111–114. [Google Scholar] [CrossRef] [PubMed]
- Baud, M.G.; Leiser, T.; Haus, P.; Samlal, S.; Wong, A.C.; Wood, R.J.; Petrucci, V.; Gunaratnam, M.; Hughes, S.M.; Buluwela, L.; et al. Defining the mechanism of action and enzymatic selectivity of psammaplin a against its epigenetic targets. J. Med. Chem. 2012, 55, 1731–1750. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.-Y.; Orlikova, B.; Diederich, M. Signal Transducers and Activators of Transcription (STAT) Regulatory Networks in Marine Organisms: From Physiological Observations towards Marine Drug Discovery. Mar. Drugs 2015, 13, 4967-4984. https://doi.org/10.3390/md13084967
Lee J-Y, Orlikova B, Diederich M. Signal Transducers and Activators of Transcription (STAT) Regulatory Networks in Marine Organisms: From Physiological Observations towards Marine Drug Discovery. Marine Drugs. 2015; 13(8):4967-4984. https://doi.org/10.3390/md13084967
Chicago/Turabian StyleLee, Jin-Young, Barbora Orlikova, and Marc Diederich. 2015. "Signal Transducers and Activators of Transcription (STAT) Regulatory Networks in Marine Organisms: From Physiological Observations towards Marine Drug Discovery" Marine Drugs 13, no. 8: 4967-4984. https://doi.org/10.3390/md13084967
APA StyleLee, J.-Y., Orlikova, B., & Diederich, M. (2015). Signal Transducers and Activators of Transcription (STAT) Regulatory Networks in Marine Organisms: From Physiological Observations towards Marine Drug Discovery. Marine Drugs, 13(8), 4967-4984. https://doi.org/10.3390/md13084967







