Fucoidan Stimulates Monocyte Migration via ERK/p38 Signaling Pathways and MMP9 Secretion
Abstract
:1. Introduction
2. Results and Discussion
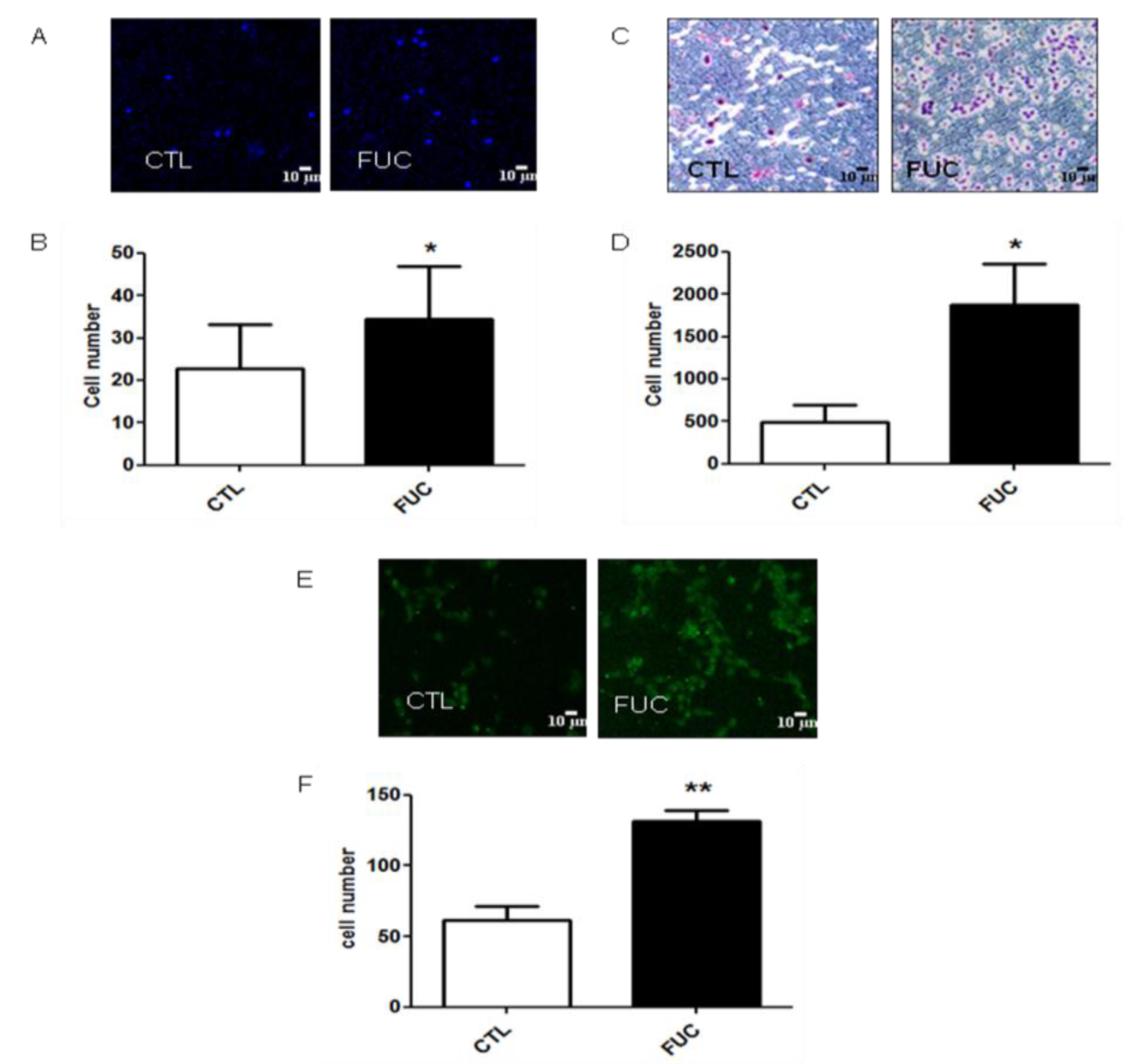
2.1. Fucoidan Pretreatment Enhances Monocyte Adhesion, Migration and Transmigration
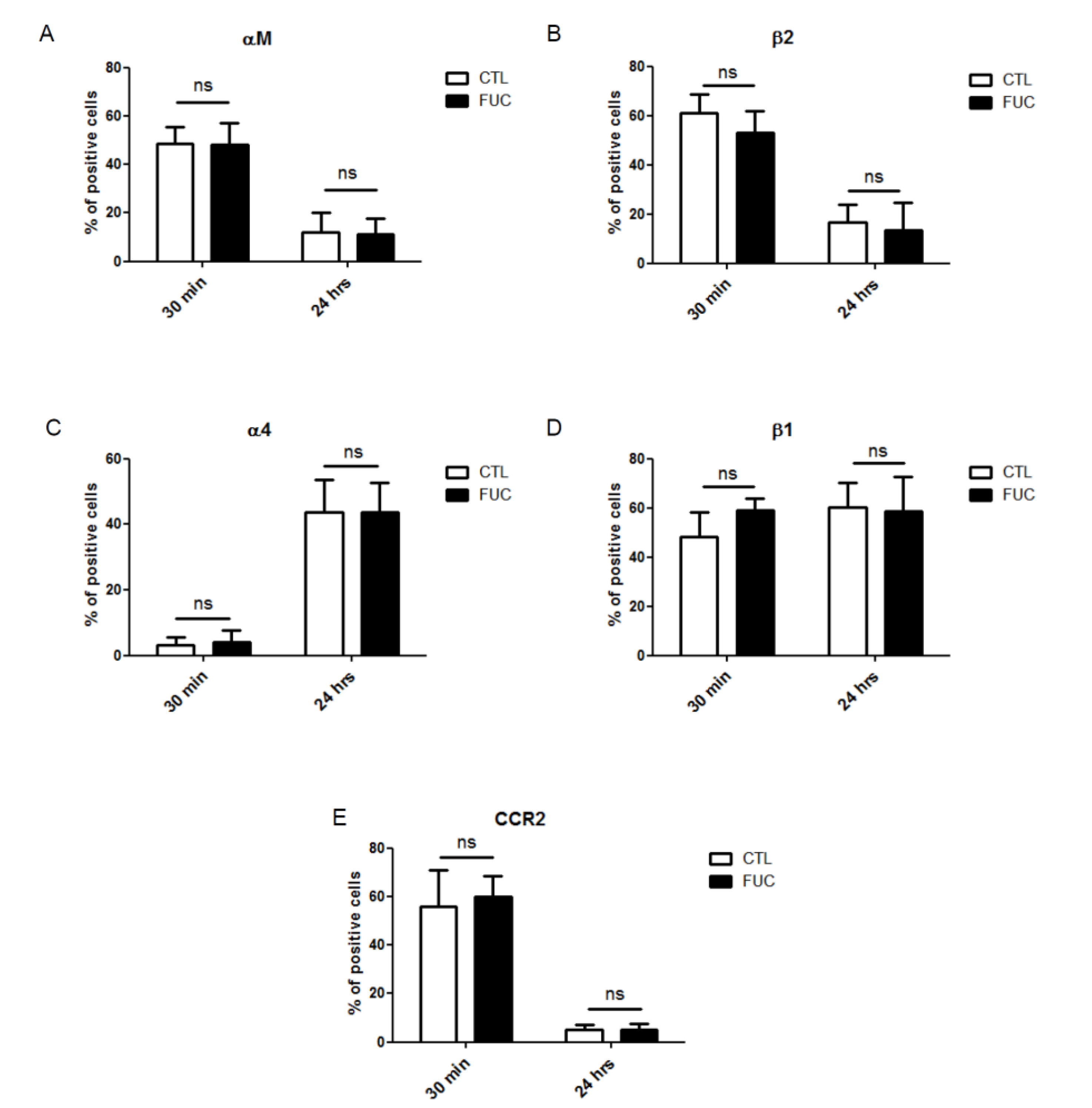
2.2. Fucoidan Stimulation of Monocyte Adhesion and Migration Is not Due to Modulation of Integrin Expression or CCR2 Receptor Expression
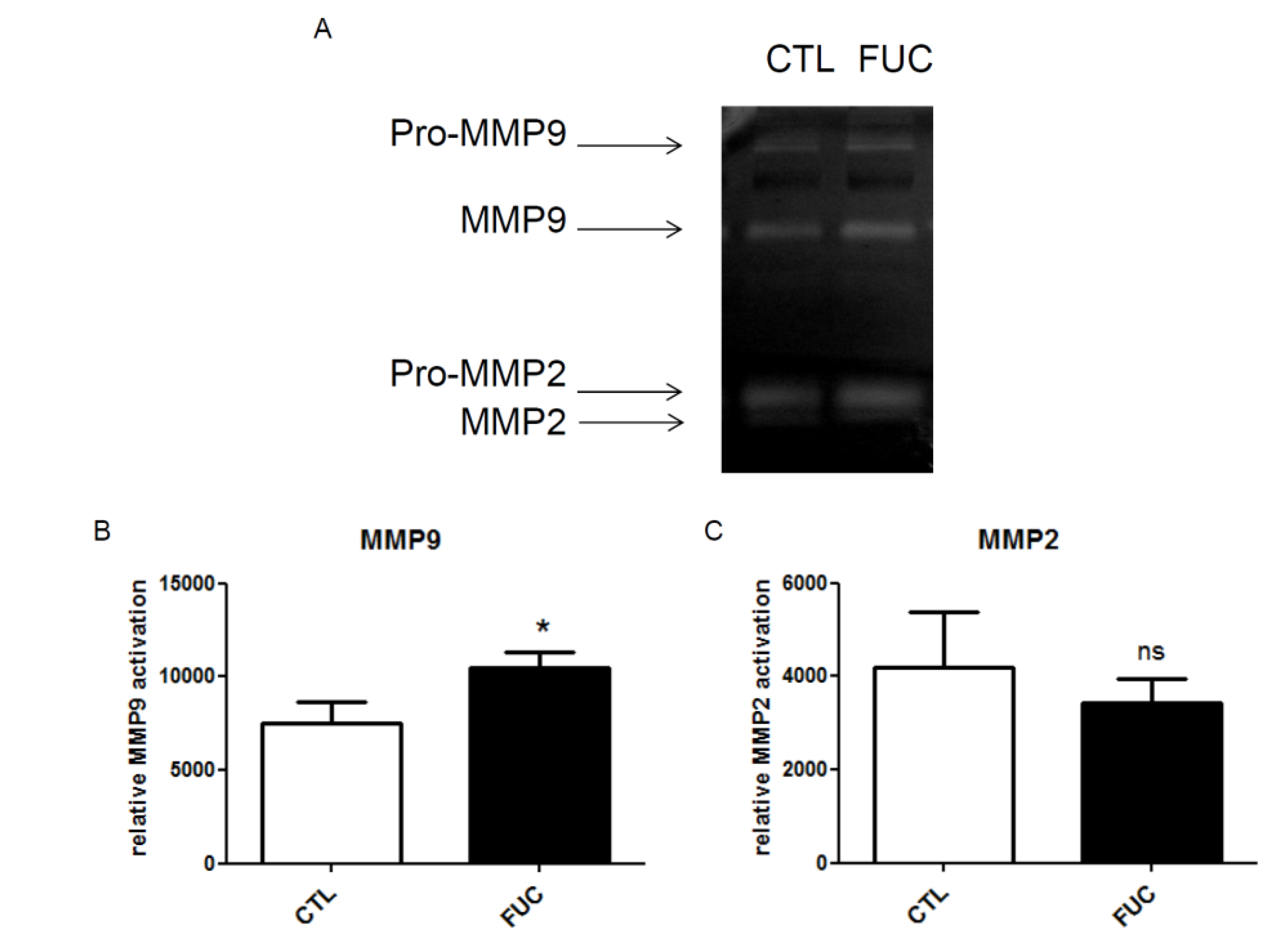
2.3. Fucoidan Enhances PBMC MMP9 Activity
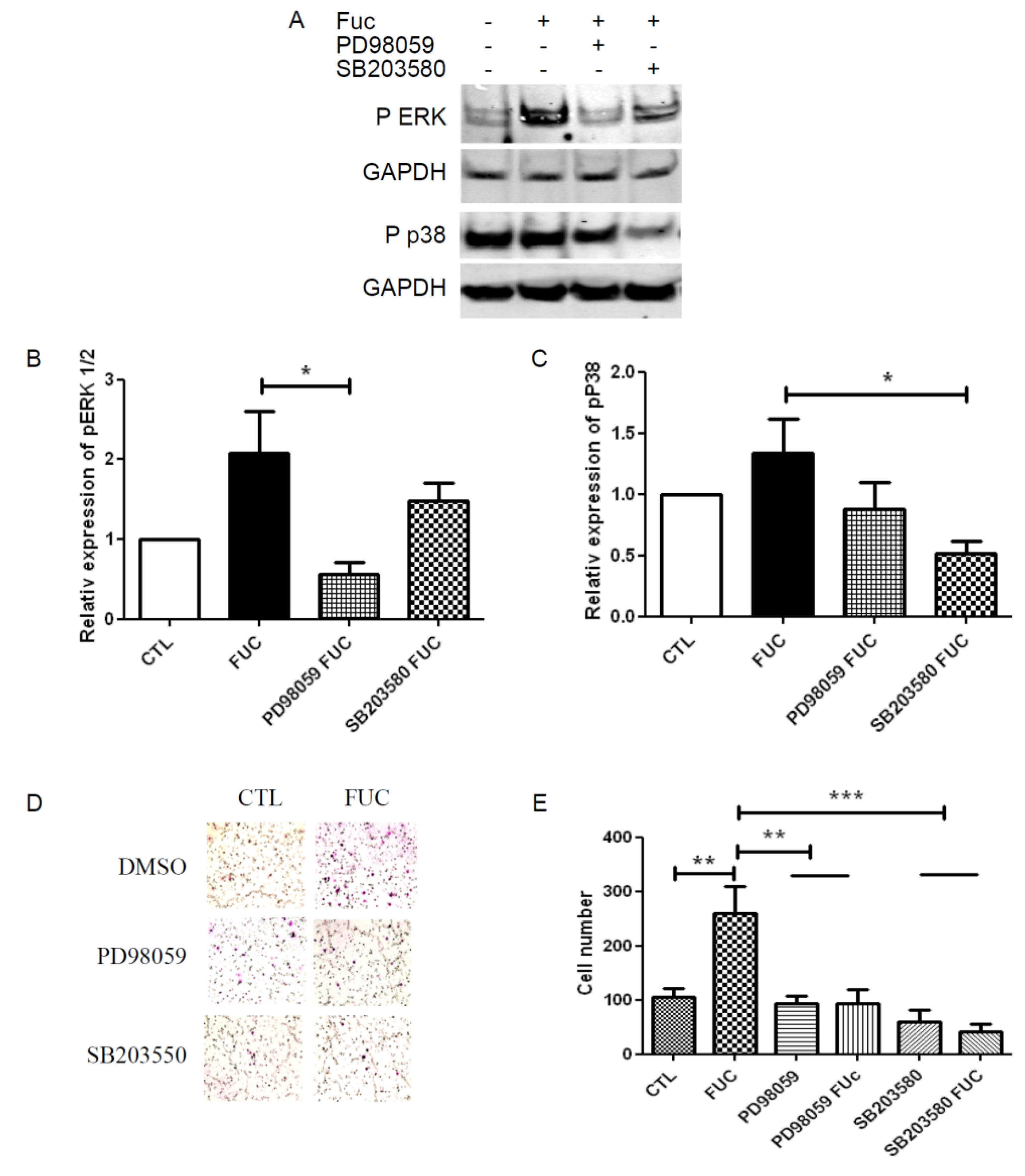
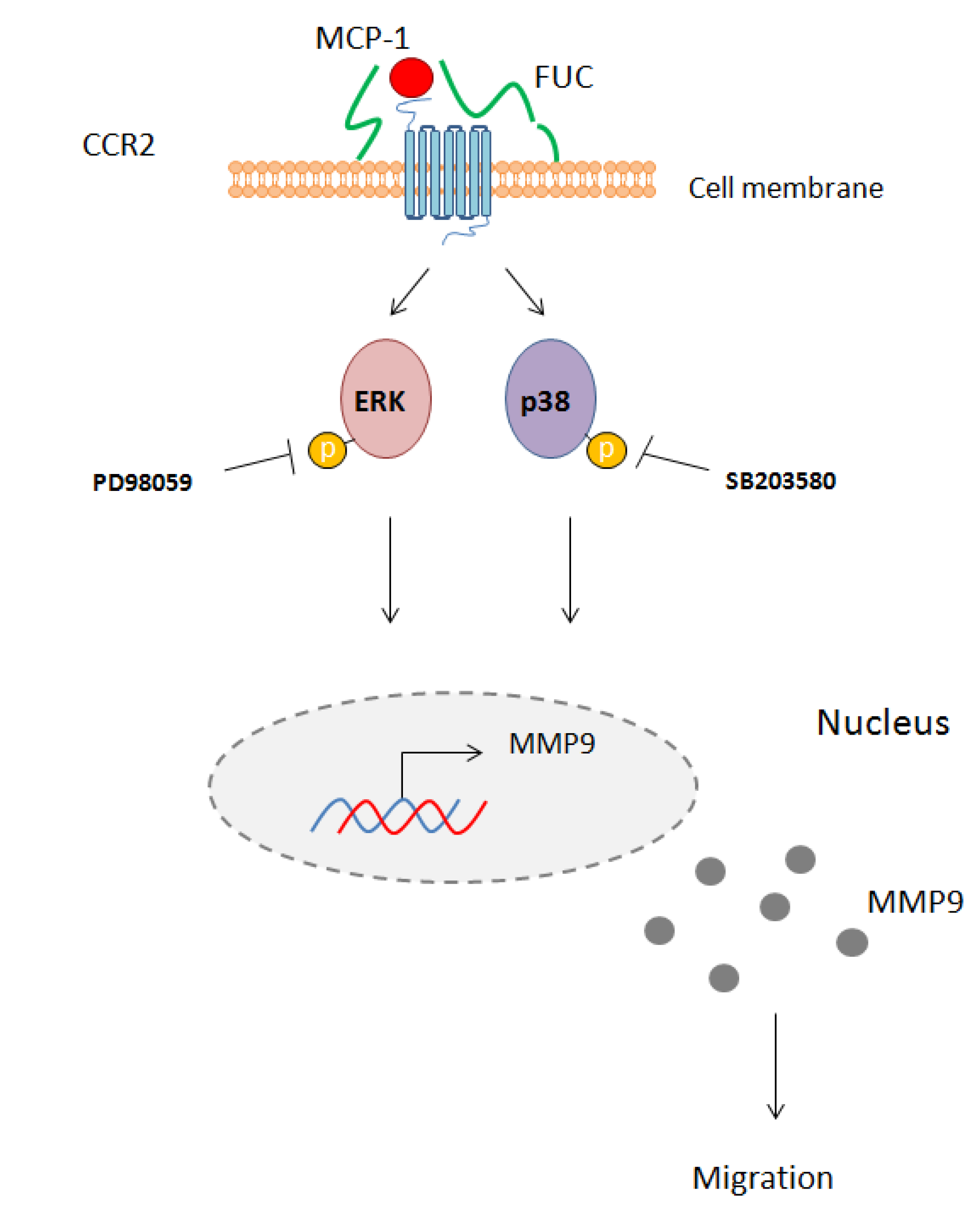
2.4. Fucoidan Enhancement of PBMC Migration Is Countered by ERK and p38 Pathway Inhibition
3. Discussion
4. Experimental
4.1. Reagents
4.2. Monocyte and HUVEC Isolation
4.3. Cell Adhesion Assay
4.4. Cell Migration Assay
4.5. Transmigration Assay
4.6. Western Blot
4.7. Flow Cytometry
4.8. Zymography
4.9. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Golomb, B.A.; Dang, T.T.; Criqui, M.H. Peripheral arterial disease: Morbidity and mortality implications. Circulation 2006, 114, 688–699. [Google Scholar] [CrossRef] [PubMed]
- Norgren, L.; Hiatt, W.R.; Dormandy, J.A.; Nehler, M.R.; Harris, K.A.; Fowkes, F.G.R.; Rutherford, R.B. TASC II Working Group Inter-society consensus for the management of peripheral arterial disease. Int. Angiol. J. Int. Union Angiol. 2007, 26, 81–157. [Google Scholar]
- Shanmugasundaram, M.; Ram, V.K.; Luft, U.C.; Szerlip, M.; Alpert, J.S. Peripheral arterial disease—What do we need to know? Clin. Cardiol. 2011, 34, 478–482. [Google Scholar] [CrossRef] [PubMed]
- Fowkes, F.G.R.; Rudan, D.; Rudan, I.; Aboyans, V.; Denenberg, J.O.; McDermott, M.M.; Norman, P.E.; Sampson, U.K.A.; Williams, L.J.; Mensah, G.A.; et al. Comparison of global estimates of prevalence and risk factors for peripheral artery disease in 2000 and 2010: A systematic review and analysis. Lancet 2013, 382, 1329–1340. [Google Scholar] [CrossRef]
- Rastan, A.; Krankenberg, H.; Baumgartner, I.; Blessing, E.; Müller-Hülsbeck, S.; Pilger, E.; Scheinert, D.; Lammer, J.; Beschorner, U.; Noory, E.; et al. Stent Placement vs. Balloon Angioplasty for Popliteal Artery Treatment: Two-Year Results of a Prospective, Multicenter, Randomized Trial. J. Endovasc. Ther. Off. J. Int. Soc. Endovasc. Spec. 2015, 22, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Varol, C.; Mildner, A.; Jung, S. Macrophages: Development and tissue specialization. Annu. Rev. Immunol. 2015, 33, 643–675. [Google Scholar] [CrossRef] [PubMed]
- McGinigle, K.L.; Kalbaugh, C.A.; Marston, W.A. Living in a medically underserved county is an independent risk factor for major limb amputation. J. Vasc. Surg. 2014, 59, 737–741. [Google Scholar] [CrossRef] [PubMed]
- Lau, J.F.; Weinberg, M.D.; Olin, J.W. Peripheral artery disease. Part 1: Clinical evaluation and noninvasive diagnosis. Nat. Rev. Cardiol. 2011, 8, 405–418. [Google Scholar] [CrossRef] [PubMed]
- Aerden, D.; Massaad, D.; von Kemp, K.; van Tussenbroek, F.; Debing, E.; Keymeulen, B.; van den Brande, P. The ankle-brachial index and the diabetic foot: A troublesome marriage. Ann. Vasc. Surg. 2011, 25, 770–777. [Google Scholar] [CrossRef] [PubMed]
- Weinberg, M.D.; Lau, J.F.; Rosenfield, K.; Olin, J.W. Peripheral artery disease. Part 2: Medical and endovascular treatment. Nat. Rev. Cardiol. 2011, 8, 429–441. [Google Scholar] [CrossRef] [PubMed]
- Novo, S.; Coppola, G.; Milio, G. Critical limb ischemia: Definition and natural history. Curr. Drug Targets Cardiovasc. Haematol. Disord. 2004, 4, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Bertelè, V.; Roncaglioni, M.C.; Pangrazzi, J.; Terzian, E.; Tognoni, E.G. Clinical outcome and its predictors in 1560 patients with critical leg ischaemia. Chronic Critical Leg Ischaemia Group. Eur. J. Vasc. Endovasc. Surg. Off. J. Eur. Soc. Vasc. Surg. 1999, 18, 401–410. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, I.; Chronos, N.; Comerota, A.; Henry, T.; Pasquet, J.-P.; Finiels, F.; Caron, A.; Dedieu, J.-F.; Pilsudski, R.; Delaère, P. Local gene transfer and expression following intramuscular administration of FGF-1 plasmid DNA in patients with critical limb ischemia. Mol. Ther. J. Am. Soc. Gene Ther. 2009, 17, 914–921. [Google Scholar] [CrossRef] [PubMed]
- Hiatt, W.R.; Hirsch, A.T.; Creager, M.A.; Rajagopalan, S.; Mohler, E.R.; Ballantyne, C.M.; Regensteiner, J.G.; Treat-Jacobson, D.; Dale, R.A.; Rooke, T. Effect of niacin ER/lovastatin on claudication symptoms in patients with peripheral artery disease. Vasc. Med. Lond. Engl. 2010, 15, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Nikol, S.; Baumgartner, I.; van Belle, E.; Diehm, C.; Visoná, A.; Capogrossi, M.C.; Ferreira-Maldent, N.; Gallino, A.; Wyatt, M.G.; Wijesinghe, L.D.; et al. TALISMAN 201 investigators Therapeutic angiogenesis with intramuscular NV1FGF improves amputation-free survival in patients with critical limb ischemia. Mol. Ther. J. Am. Soc. Gene Ther. 2008, 16, 972–978. [Google Scholar] [CrossRef] [PubMed]
- Belch, J.; Hiatt, W.R.; Baumgartner, I.; Driver, I.V.; Nikol, S.; Norgren, L.; van Belle, E. TAMARIS Committees and Investigators Effect of fibroblast growth factor NV1FGF on amputation and death: A randomised placebo-controlled trial of gene therapy in critical limb ischaemia. Lancet 2011, 377, 1929–1937. [Google Scholar] [CrossRef]
- Lederman, R.J.; Mendelsohn, F.O.; Anderson, R.D.; Saucedo, J.F.; Tenaglia, A.N.; Hermiller, J.B.; Hillegass, W.B.; Rocha-Singh, K.; Moon, T.E.; Whitehouse, M.J.; et al. TRAFFIC Investigators Therapeutic angiogenesis with recombinant fibroblast growth factor-2 for intermittent claudication (the TRAFFIC study): A randomised trial. Lancet 2002, 359, 2053–2058. [Google Scholar] [CrossRef]
- Rajagopalan, S.; Mohler, E.R.; Lederman, R.J.; Mendelsohn, F.O.; Saucedo, J.F.; Goldman, C.K.; Blebea, J.; Macko, J.; Kessler, P.D.; Rasmussen, H.S.; et al. Regional angiogenesis with vascular endothelial growth factor in peripheral arterial disease: A phase II randomized, double-blind, controlled study of adenoviral delivery of vascular endothelial growth factor 121 in patients with disabling intermittent claudication. Circulation 2003, 108, 1933–1938. [Google Scholar] [PubMed]
- Tateishi-Yuyama, E.; Matsubara, H.; Murohara, T.; Ikeda, U.; Shintani, S.; Masaki, H.; Amano, K.; Kishimoto, Y.; Yoshimoto, K.; Akashi, H.; et al. Therapeutic Angiogenesis using Cell Transplantation (TACT) Study Investigators Therapeutic angiogenesis for patients with limb ischaemia by autologous transplantation of bone-marrow cells: A pilot study and a randomised controlled trial. Lancet 2002, 360, 427–435. [Google Scholar] [CrossRef]
- Ishida, A.; Ohya, Y.; Sakuda, H.; Ohshiro, K.; Higashiuesato, Y.; Nakaema, M.; Matsubara, S.; Yakabi, S.; Kakihana, A.; Ueda, M.; et al. Autologous peripheral blood mononuclear cell implantation for patients with peripheral arterial disease improves limb ischemia. Circ. J. Off. J. Jpn. Circ. Soc. 2005, 69, 1260–1265. [Google Scholar] [CrossRef]
- Huang, P.; Li, S.; Han, M.; Xiao, Z.; Yang, R.; Han, Z.C. Autologous transplantation of granulocyte colony-stimulating factor-mobilized peripheral blood mononuclear cells improves critical limb ischemia in diabetes. Diabetes Care 2005, 28, 2155–2160. [Google Scholar] [CrossRef] [PubMed]
- Fadini, G.P.; Agostini, C.; Avogaro, A. Autologous stem cell therapy for peripheral arterial disease meta-analysis and systematic review of the literature. Atherosclerosis 2010, 209, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Moazzami, K.; Majdzadeh, R.; Nedjat, S. Local intramuscular transplantation of autologous mononuclear cells for critical lower limb ischaemia. Cochrane Database Syst. Rev. 2011, 12, CD008347. [Google Scholar] [PubMed]
- Silvestre, J.-S.; Smadja, D.M.; Lévy, B.I. Postischemic revascularization: From cellular and molecular mechanisms to clinical applications. Physiol. Rev. 2013, 93, 1743–1802. [Google Scholar] [CrossRef] [PubMed]
- Magri, D.; Vasilas, P.; Muto, A.; Fitzgerald, T.N.; Fancher, T.T.; Feinstein, A.J.; Nishibe, T.; Dardik, A. Elevated monocytes in patients with critical limb ischemia diminish after bypass surgery. J. Surg. Res. 2011, 167, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Capoccia, B.J.; Shepherd, R.M.; Link, D.C. G-CSF and AMD3100 mobilize monocytes into the blood that stimulate angiogenesis in vivo through a paracrine mechanism. Blood 2006, 108, 2438–2445. [Google Scholar] [CrossRef] [PubMed]
- Heil, M.; Ziegelhoeffer, T.; Pipp, F.; Kostin, S.; Martin, S.; Clauss, M.; Schaper, W. Blood monocyte concentration is critical for enhancement of collateral artery growth. Am. J. Physiol. Heart Circ. Physiol. 2002, 283, H2411–H2419. [Google Scholar] [CrossRef] [PubMed]
- Heil, M.; Ziegelhoeffer, T.; Wagner, S.; Fernández, B.; Helisch, A.; Martin, S.; Tribulova, S.; Kuziel, W.A.; Bachmann, G.; Schaper, W. Collateral artery growth (arteriogenesis) after experimental arterial occlusion is impaired in mice lacking CC-chemokine receptor-2. Circ. Res. 2004, 94, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Voskuil, M.; van Royen, N.; Hoefer, I.E.; Seidler, R.; Guth, B.D.; Bode, C.; Schaper, W.; Piek, J.J.; Buschmann, I.R. Modulation of collateral artery growth in a porcine hindlimb ligation model using MCP-1. Am. J. Physiol. Heart Circ. Physiol. 2003, 284, H1422–H1428. [Google Scholar] [CrossRef] [PubMed]
- Sarlon, G.; Zemani, F.; David, L.; Duong van Huyen, J.P.; Dizier, B.; Grelac, F.; Colliec-Jouault, S.; Galy-Fauroux, I.; Bruneval, P.; Fischer, A.M.; et al. Therapeutic effect of fucoidan-stimulated endothelial colony-forming cells in peripheral ischemia. J. Thromb. Haemost. JTH 2012, 10, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Zemani, F.; Silvestre, J.-S.; Fauvel-Lafeve, F.; Bruel, A.; Vilar, J.; Bieche, I.; Laurendeau, I.; Galy-Fauroux, I.; Fischer, A.M.; Boisson-Vidal, C. Ex vivo priming of endothelial progenitor cells with SDF-1 before transplantation could increase their proangiogenic potential. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 644–650. [Google Scholar] [CrossRef] [PubMed]
- Boisson-Vidal, C.; Zemani, F.; Caligiuri, G.; Galy-Fauroux, I.; Colliec-Jouault, S.; Helley, D.; Fischer, A.-M. Neoangiogenesis induced by progenitor endothelial cells: Effect of fucoidan from marine algae. Cardiovasc. Hematol. Agents Med. Chem. 2007, 5, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Luyt, C.-E.; Meddahi-Pellé, A.; Ho-Tin-Noe, B.; Colliec-Jouault, S.; Guezennec, J.; Louedec, L.; Prats, H.; Jacob, M.-P.; Osborne-Pellegrin, M.; Letourneur, D.; et al. Low-molecular-weight fucoidan promotes therapeutic revascularization in a rat model of critical hindlimb ischemia. J. Pharmacol. Exp. Ther. 2003, 305, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Nahrendorf, M.; Swirski, F.K.; Aikawa, E.; Stangenberg, L.; Wurdinger, T.; Figueiredo, J.-L.; Libby, P.; Weissleder, R.; Pittet, M.J. The healing myocardium sequentially mobilizes two monocyte subsets with divergent and complementary functions. J. Exp. Med. 2007, 204, 3037–3047. [Google Scholar] [CrossRef] [PubMed]
- Geissmann, F.; Jung, S.; Littman, D.R. Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity 2003, 19, 71–82. [Google Scholar] [CrossRef]
- Hartney, J.M.; Brown, J.; Chu, H.W.; Chang, L.Y.; Pelanda, R.; Torres, R.M. Arhgef1 regulates alpha5beta1 integrin-mediated matrix metalloproteinase expression and is required for homeostatic lung immunity. Am. J. Pathol. 2010, 176, 1157–1168. [Google Scholar] [CrossRef] [PubMed]
- Nourshargh, S.; Alon, R. Leukocyte migration into inflamed tissues. Immunity 2014, 41, 694–707. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, C.E.; Hoefer, I.E.; Meder, B.; Roth, H.; van Royen, N.; Breit, S.M.; Jost, M.M.; Aharinejad, S.; Hartmann, S.; Buschmann, I.R. Arteriogenesis depends on circulating monocytes and macrophage accumulation and is severely depressed in op/op mice. J. Leukoc. Biol. 2006, 80, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Arras, M.; Ito, W.D.; Scholz, D.; Winkler, B.; Schaper, J.; Schaper, W. Monocyte activation in angiogenesis and collateral growth in the rabbit hindlimb. J. Clin. Invest. 1998, 101, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Wolff, R.A.; Tomas, J.J.; Hullett, D.A.; Stark, V.E.; van Rooijen, N.; Hoch, J.R. Macrophage depletion reduces monocyte chemotactic protein-1 and transforming growth factor-beta1 in healing rat vein grafts. J. Vasc. Surg. 2004, 39, 878–888. [Google Scholar] [CrossRef] [PubMed]
- Schepers, A.; Eefting, D.; Bonta, P.I.; Grimbergen, J.M.; de Vries, M.R.; van Weel, V.; de Vries, C.J.; Egashira, K.; van Bockel, J.H.; Quax, P.H.A. Anti-MCP-1 gene therapy inhibits vascular smooth muscle cells proliferation and attenuates vein graft thickening both in vitro and in vivo. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 2063–2069. [Google Scholar] [CrossRef] [PubMed]
- Tatewaki, H.; Egashira, K.; Kimura, S.; Nishida, T.; Morita, S.; Tominaga, R. Blockade of monocyte chemoattractant protein-1 by adenoviral gene transfer inhibits experimental vein graft neointimal formation. J. Vasc. Surg. 2007, 45, 1236–1243. [Google Scholar] [CrossRef] [PubMed]
- Haroun-Bouhedja, F.; Lindenmeyer, F.; Lu, H.; Soria, C.; Jozefonvicz, J.; Boisson-Vidal, C. In vitro effects of fucans on MDA-MB231 tumor cell adhesion and invasion. Anticancer Res. 2002, 22, 2285–2292. [Google Scholar] [PubMed]
- Sadir, R.; Baleux, F.; Grosdidier, A.; Imberty, A.; Lortat-Jacob, H. Characterization of the stromal cell-derived factor-1alpha-heparin complex. J. Biol. Chem. 2001, 276, 8288–8296. [Google Scholar] [CrossRef] [PubMed]
- Thorlacius, H.; Vollmar, B.; Seyfert, U.T.; Vestweber, D.; Menger, M.D. The polysaccharide fucoidan inhibits microvascular thrombus formation independently from P- and l-selectin function in vivo. Eur. J. Clin. Invest. 2000, 30, 804–810. [Google Scholar] [CrossRef] [PubMed]
- Frenette, P.S.; Weiss, L. Sulfated glycans induce rapid hematopoietic progenitor cell mobilization: Evidence for selectin-dependent and independent mechanisms. Blood 2000, 96, 2460–2468. [Google Scholar] [PubMed]
- Cambien, B.; Pomeranz, M.; Millet, M.A.; Rossi, B.; Schmid-Alliana, A. Signal transduction involved in MCP-1-mediated monocytic transendothelial migration. Blood 2001, 97, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Ge, H.; Yuan, W.; Liu, J.; He, Q.; Ding, S.; Pu, J.; He, B. Functional Relevance of Protein Glycosylation to the Pro-Inflammatory Effects of Extracellular Matrix Metalloproteinase Inducer (EMMPRIN) on Monocytes/Macrophages. PLoS ONE 2015, 10, e0117463. [Google Scholar] [CrossRef] [PubMed]
- Ashida, N.; Arai, H.; Yamasaki, M.; Kita, T. Distinct signaling pathways for MCP-1-dependent integrin activation and chemotaxis. J. Biol. Chem. 2001, 276, 16555–16560. [Google Scholar] [CrossRef] [PubMed]
- McGilvray, I.D.; Tsai, V.; Marshall, J.C.; Dackiw, A.P.B.; Rotstein, O.D. Monocyte adhesion and transmigration induce tissue factor expression: Role of the mitogen-activated protein kinases. Shock 2002, 18, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.-C.; Ho, M.-Y.; Liang, S.-M.; Liang, C.-M. Recombinant protein rVP1 upregulates BECN1-independent autophagy, MAPK1/3 phosphorylation and MMP9 activity via WIPI1/WIPI2 to promote macrophage migration. Autophagy 2013, 9, 5–19. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Li, Y.; Teruya, K.; Katakura, Y.; Ichikawa, A.; Eto, H.; Hosoi, M.; Hosoi, M.; Nishimoto, S.; Shirahata, S. Enzyme-digested Fucoidan Extracts Derived from Seaweed Mozuku of Cladosiphon novae-caledoniae kylin Inhibit Invasion and Angiogenesis of Tumor Cells. Cytotechnology 2005, 47, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Kim, J.-S.; Kim, E. Fucoidan from Seaweed Fucus vesiculosus Inhibits Migration and Invasion of Human Lung Cancer Cell via PI3K-Akt-mTOR Pathways. PLoS ONE 2012, 7, e50624. [Google Scholar] [CrossRef] [PubMed]
- Teng, H.; Yang, Y.; Wei, H.; Liu, Z.; Liu, Z.; Ma, Y.; Gao, Z.; Hou, L.; Zou, X. Fucoidan Suppresses Hypoxia-Induced Lymphangiogenesis and Lymphatic Metastasis in Mouse Hepatocarcinoma. Mar. Drugs 2015, 13, 3514–3530. [Google Scholar] [CrossRef] [PubMed]
- Fitton, J.H. Therapies from fucoidan; Multifunctional marine polymers. Mar. Drugs 2011, 9, 1731–1760. [Google Scholar] [CrossRef] [PubMed]
- Ustyuzhanina, N.E.; Bilan, M.I.; Ushakova, N.A.; Usov, A.I.; Kiselevskiy, M.V.; Nifantiev, N.E. Fucoidans: Pro- or antiangiogenic agents? Glycobiology 2014, 24, 1265–1274. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, K.; Xue, C.; Zhao, X.; Mori, M.; Sugawara, T.; Hirata, T. Effects of middle molecular weight fucoidans on in vitro and ex vivo angiogenesis of endothelial cells. Int. J. Mol. Med. 2005, 15, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Zemani, F.; Benisvy, D.; Galy-Fauroux, I.; Lokajczyk, A.; Colliec-Jouault, S.; Uzan, G.; Fischer, A.M.; Boisson-Vidal, C. Low-molecular-weight fucoidan enhances the proangiogenic phenotype of endothelial progenitor cells. Biochem. Pharmacol. 2005, 70, 1167–1175. [Google Scholar] [CrossRef] [PubMed]
- Nardella, A.; Chaubet, F.; Boisson-Vidal, C.; Blondin, C.; Durand, P.; Jozefonvicz, J. Anticoagulant low molecular weight fucans produced by radical process and ion exchange chromatography of high molecular weight fucans extracted from the brown seaweed Ascophyllum nodosum. Carbohydr. Res. 1996, 289, 201–208. [Google Scholar] [CrossRef]
- Mulloy, B.; Gee, C.; Wheeler, S.F.; Wait, R.; Gray, E.; Barrowcliffe, T.W. Molecular weight measurements of low molecular weight heparins by gel permeation chromatography. Thromb. Haemost. 1997, 77, 668–674. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sapharikas, E.; Lokajczyk, A.; Fischer, A.-M.; Boisson-Vidal, C. Fucoidan Stimulates Monocyte Migration via ERK/p38 Signaling Pathways and MMP9 Secretion. Mar. Drugs 2015, 13, 4156-4170. https://doi.org/10.3390/md13074156
Sapharikas E, Lokajczyk A, Fischer A-M, Boisson-Vidal C. Fucoidan Stimulates Monocyte Migration via ERK/p38 Signaling Pathways and MMP9 Secretion. Marine Drugs. 2015; 13(7):4156-4170. https://doi.org/10.3390/md13074156
Chicago/Turabian StyleSapharikas, Elene, Anna Lokajczyk, Anne-Marie Fischer, and Catherine Boisson-Vidal. 2015. "Fucoidan Stimulates Monocyte Migration via ERK/p38 Signaling Pathways and MMP9 Secretion" Marine Drugs 13, no. 7: 4156-4170. https://doi.org/10.3390/md13074156
APA StyleSapharikas, E., Lokajczyk, A., Fischer, A.-M., & Boisson-Vidal, C. (2015). Fucoidan Stimulates Monocyte Migration via ERK/p38 Signaling Pathways and MMP9 Secretion. Marine Drugs, 13(7), 4156-4170. https://doi.org/10.3390/md13074156





