Sea Cucumbers Metabolites as Potent Anti-Cancer Agents
Abstract
:1. Introduction
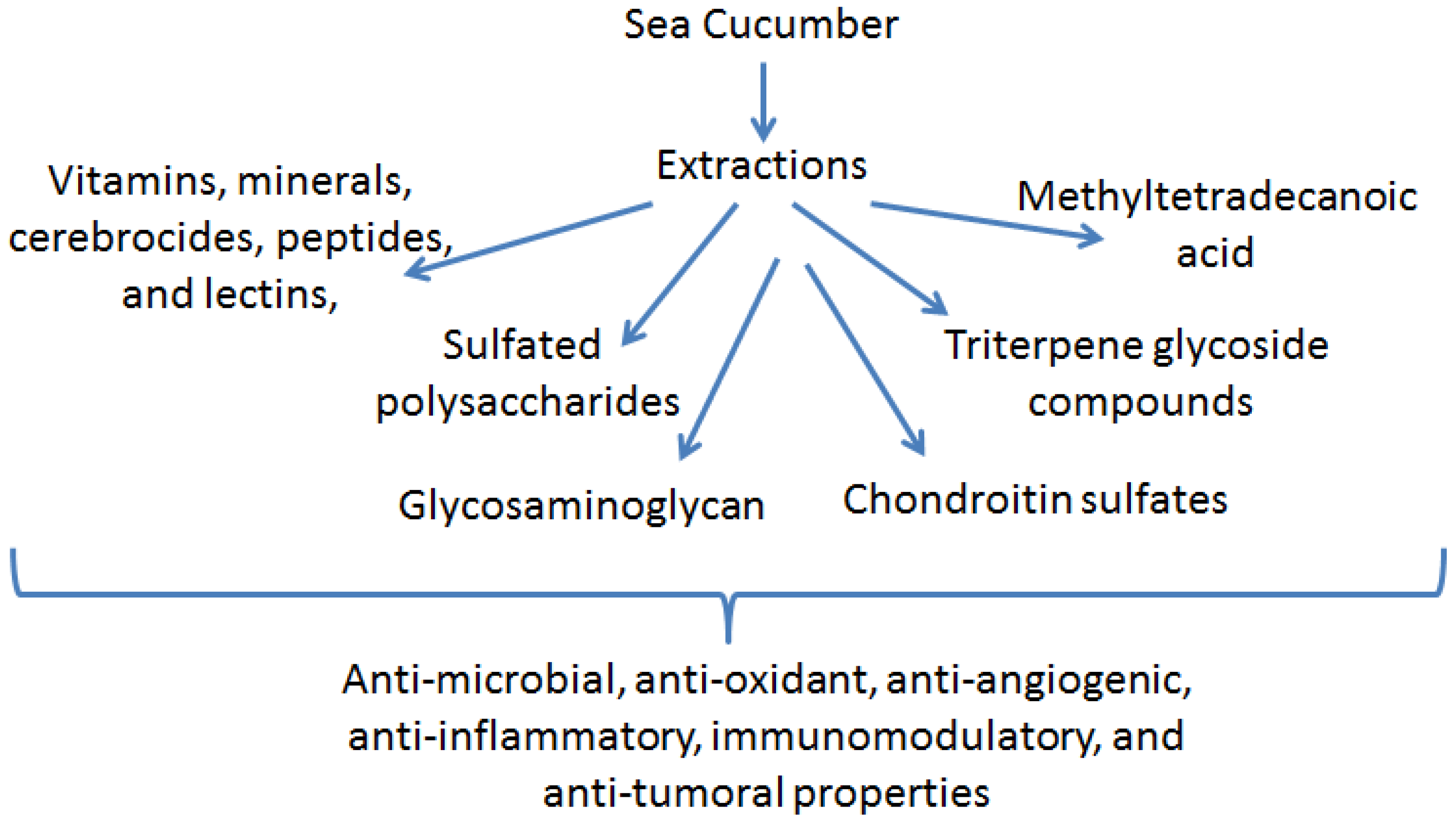
2. Anti-Inflammatory Function of Sea Cucumber Extracts

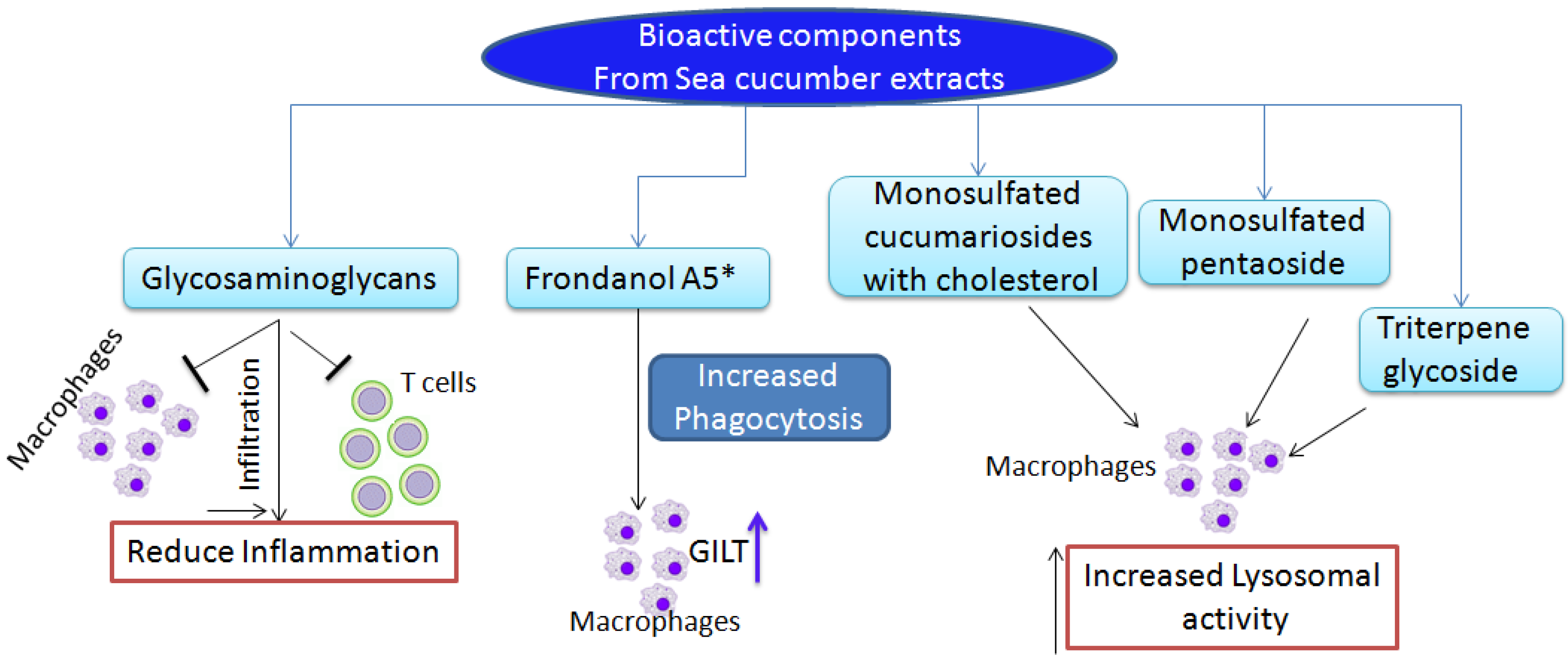
3. Immunostimulatory and Anti-Tumor Functions of Sea Cucumber Extracts
| Compound | Sea cucumber | Effects | Type of cancer | Refs. |
|---|---|---|---|---|
| Triterpene glycosides | Mensamaria intercedens | Anti-tumorigenic | mouse model of S180 sarcoma and mouse Lewis lung cancer cell lines | [51] |
| Hot water extract | Stichopus japonicas | Anti-proliferation cytotoxic | Human colon cancer CaCo2 cells | [52] |
| Organic extracts | Holothuria leucospilota, Holothuria scabra, Stichopus chloronotus | Anti-proliferation | human A549 non-small lung cancer cells and C33A cervical cancer cells | [29] |
| sulfated triterpene glycosides | Pearsonothuria graeffei | Invasion, migration, decreased VEGF, MMP9, increased TIMP-1, decreased NF-κB | human hepatocellular liver carcinoma cells (HepG2) and human endothelial cells (ECV-304) | [53] |
| Frondoside A | Cucumaria frondosa | Anti-proliferation | Pancreatic cancer cells | [54] |
| Frondoside A | Cucumaria frondosa | Tumor inhibition, anti-proliferation, apoptosis, increased p21 | Pancreatic cancer xenografts | [55] |
| Frondoside A + Gemcitabine | Cucumaria frondosa | Tumor inhibition, apoptosis, necrosis, Cas3,7 & 9 increase | Pancreatic cancer xenografts | [56] |
| Frondoside A | Cucumaria frondosa | Anti-proliferation, Cas3, 7 increase | Lung and breast cancer | [57] |
| Frondoside A | Cucumaria frondosa | Anti-angiogenesis, decreased CD31 | Lung cancer xenografts | [58] |
| Frondoside A + Cisplatin | Cucumaria frondosa | Tumor inhibition | Lung cancer xenografts | [58] |
| Frondoside A | Cucumaria frondosa | Anti-proliferation Migration and invasion, increase in p53, Cas3/7 | Breast cancer cells | [59] |
| Frondoside A | Cucumaria frondosa | Anti-tumor | Breast cancer xenografts | [59] |
| Frondoside A + Paclitaxel | Cucumaria frondosa | cytotoxic | Breast cancer cells | [59] |
| Frondoside A | Cucumaria frondosa | Anti-tumor and anti-metastatic, decrease ERK1/2 | syngeneic murine model of metastatic breast cancer using Line 66.1 | [60] |
| Polar fraction of Frondanol A5 | Cucumaria frondosa | Anti-proliferation, inhibition of cell cycle, induce apoptosis | Pancreatic cancer cells | [61] |
| Frondanol A5 | Cucumaria frondosa | Aberrant crypt inhibition, p21 in-crease, DNA fragmentation, apoptosis | AOM-induced rat colon cancer model | [30] |
| Frondanol A5 | Cucumaria frondosa | p21 increase, G2/Minhibition, apoptosis | Human colon cancer cells HCT116 | [30] |
| Frondanol A5 | Cucumaria frondosa | Inhibition of small intestinal and colon tumors, increase in GILT expression, macrophage phagocytosis | ApcMin/+ colon cancer model | [31] |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Newman, D.J.; Crag, G.M.; Snader, K.M. Natural products as sources of new drugs over the period 1981–2002. Nat. Prod. 2003, 66, 1022–1037. [Google Scholar] [CrossRef]
- Nagle, D.G.; Zhou, Y.D.; Mora, F.D.; Mohammed, K.A.; Kim, Y.P. Mechanism targeted discovery of antitumor marine natural products. Curr. Med. Chem. 2004, 11, 1725–1756. [Google Scholar] [CrossRef] [PubMed]
- Balunas, M.J.; Chai, H.B.; Kinghorn, A.D. Drug discovery from natural sources. AAPS J. 2006, 8, E239–E253. [Google Scholar] [CrossRef] [PubMed]
- Rajasekaran, A.; Sivagnanam, G.; Xavier, R. Nutraceuticals as therapeutic agents: A review research. J. Pharm. Technol. 2008, 1, 328–340. [Google Scholar]
- Adrian, T.E. Novel marine-derived anti-cancer agents. Curr. Pharm. Des. 2007, 13, 3417–3426. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society Publication. Cancer Facts & Figures 2006; American Cancer Society Publication: Atlanta, GA, USA, 2006. Available online: http://www.cancer.org (accessed on 10 January 2015).
- Kelly, M.S. Echinoderms: Their culture and bioactive compounds. Prog. Mol. Subcell. Biol. 2005, 39, 139–165. [Google Scholar] [PubMed]
- Costside bioresources. Available online: http://www.seacucumber.com (accessed on 16 February 2015).
- Mehmet, A.; Hüseyin, S.; Bekir, T.; Yilmaz, E.; Sevim, K. Proximate composition and fatty acid profile of three different fresh and dried commercial sea cucumbers from Turkey. Int. J. Food Sci. Technol. 2011, 46, 500–508. [Google Scholar] [CrossRef]
- Conand, C. Overview of Sea Cucumbers Fisheries over the Last Decade—What Possibilities for a Durable Management? In Echinoderm; Barker, M., Ed.; Swets & Zeitlinger: Lisse, The Netherlands, 2001; pp. 339–344. [Google Scholar]
- Conand, C.; Byrne, M. A review of recent developments in the world sea cucumber fisheries. Mar. Fish. Rev. 1993, 55, 1–13. [Google Scholar]
- Bruckner, A.W.; Johnson, K.A.; Field, J.D. Conservation strategies for sea cucumbers: Can a CITES Appendix II listing promote sustainable international trade? SPC Beche-de-mer Inf. Bull. 2003, 18, 24–33. [Google Scholar]
- Lawrence, A.J.; Afifi, R.; Ahmed, M.; Khalifa, S.; Paget, T. Bioactivity as an options value of sea cucumbers in the Egyptian Red Sea. Conserv. Biol. 2009, 24, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Shiell, G. Field observations of juvenile sea cucumbers. SPC Beche-de-mer Inf. Bull. 2004, 20, 6–11. [Google Scholar]
- Conand, C. Sea Cucumber Biology, Taxonomy, Distribution: Conversation Status. In Proceedings of the Convention on International Trade in Endangered Species of Wild Fauna and Flora Tech Workshop on the Conversation of Sea Cucumbers in the Families Holothuridae and Stichopodidae, Kuala Lumpur, Malaysia, 1–3 March 2004.
- Kerr, R.; Chen, Z. In vivo and in vitro biosynthesis of saponins in sea cucumbers (Holothuroidea). J. Nat. Prod. 1995, 58, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, T.; Togawa, K.; Higuchi, R.; Komori, T.; Sasaki, T. Constituents of Holothuroidea, II. Six newly identified biologically active triterpenoid glycoside sulfates from the sea cucumber Cucumaria echinata. Eur. J. Org. Chem. 1990, 1990, 453–460. [Google Scholar]
- Aminin, D.L.; Chaykina, E.L.; Agafonova, I.G.; Avilov, S.A.; Kalinin, V.I.; Stonik, V.A. Antitumor activity of the immunomodulatory lead Cumaside. Int. Immunopharmacol. 2010, 10, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Vieira, R.P.; Mulloy, B.; Mourão, P.A. Structure of a fucose-branched chondroitin sulphate from sea cucumber. Evidence for the presence of 3-O-sulfo-β-d-glucuronosyl residues. J. Biol. Chem. 1991, 266, 13530–13536. [Google Scholar] [PubMed]
- Mourao, P.A.S.; Pereira, M.S. Searching for alternatives to heparin: Sulfated fucans from marine invertebrates. Trends Cardiovasc. Med. 1999, 9, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Goad, L.J.; Garneau, F.X.; Simard, J.L.; ApSimon, J.W.; Girard, M. Isolation of Δ9(11)-sterols from the sea cucumber. Implications for holothurin biosynthesis. Tetrahedron Lett. 1985, 26, 3513–3516. [Google Scholar] [CrossRef]
- Mamelona, J.; Pelletier, E.M.; Lalancette, K.G.; Legault, J.; Karboune, S.; Kermasha, S. Quantification of phenolic contents and antioxidant capacity of Atlantic sea cucumber, Cucumaria frondosa. Food Chem. 2007, 104, 1040–1047. [Google Scholar] [CrossRef]
- Rafiuddin, A.M.; Venkateshwarlu, U.; Jayakumar, R. Multilayered peptide incorporated collagen tubules for peripheral nerve repair. Biomaterials 2004, 25, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, T.; Zaima, N.; Yamamoto, A.; Sakai, S.; Noguchi, R.; Hirata, T. Isolation of sphingoid bases of sea cucumber cerberosides and their cytotoxicity against human colon cancer cells. Biosci. Biotechnol. Biochem. 2006, 70, 2906–2912. [Google Scholar] [CrossRef] [PubMed]
- Mojica, E.R.E.; Merca, F.E. Lectin from the body walls of black sea cucumber (Holothuria atra Jäger). Philipp. J. Sci. 2004, 133, 77–85. [Google Scholar]
- Mojica, E.R.E.; Merca, F.E. Biological properties of lectin from sea cucumber (Holothuria scabra Jäger). J. Biol. Sci. 2005, 5, 472–477. [Google Scholar] [CrossRef]
- Mojica, E.R.E.; Merca, F.E. Isolation and partial characterization of a lectin from the internal organs of the sea cucumber (Holothuria scabra Jäger). Int. J. Zool. Res. 2005, 1, 59–65. [Google Scholar] [CrossRef]
- Tian, F.; Zhang, X.; Tong, Y.; Yi, Y.; Zhang, S.; Li, L.; Sun, P.; Lin, L.; Ding, J. PE, a new sulfated saponin from sea cucumber, exhibits anti-angiogenic and anti-tumor activities in vitro and in vivo. Cancer Biol. Ther. 2005, 4, 874–882. [Google Scholar] [CrossRef] [PubMed]
- Althunibat, O.Y.; Ridzwan, B.H.; Taher, M.; Jamaludin, M.D.; Ikeda, M.A.; Zali, B.I. In vitro antioxidant and antiproliferative activities of three Malaysian sea cucumber species. Eur. J. Sci. Res. 2009, 37, 376–387. [Google Scholar]
- Janakiram, N.B.; Mohammed, A.; Zhang, Y.; Choi, C.-I.; Woodward, C.; Collin, P.; Steele, V.E.; Rao, C.V. Chemopreventive effects of Frondanol A5, a Cucumaria frondosa extract, against rat colon carcinogenesis and inhibition of human colon cancer cell growth. Cancer Pre. Res. 2010, 3, 82–91. [Google Scholar] [CrossRef]
- Janakiram, N.B.; Mohammed, A.; Taylor, B.; Lightfoot, S.; Collin, P.D.; Steele, V.E.; Rao, C.V. Improved innate immune responses by Frondanol® A5, a sea cucumber extract, prevent intestinal tumorigenesis. Cancer Prev. Res. 2015, 8, 327–337. [Google Scholar] [CrossRef]
- Sebban, H.; Courtois, G. NF-kappaB and inflammation in genetic disease. Biochem. Pharmacol. 2006, 72, 1153–1160. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, T. Inflammation and cancer: A failure of resolution. Trends Pharmacol. Sci. 2007, 28, 162–165. [Google Scholar] [CrossRef] [PubMed]
- Hanada, T.; Yoshimura, A. Regulation of cytokine signaling and inflammation. Cytokine Growth Factor Rev. 2002, 13, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Henry, D.; McGettigan, P. Epidemiology overview of gastrointestinal and renal toxicity of NSAIDs. Int. J. Clin. Pract. 2003, 135, 43–49. [Google Scholar]
- Shaifuzain, A.R.; Amran, A.S.; Muzaffar, T.M.S.; Shaifulizan, A.R.; Hasnan, J. Effect of oral sea cucumber (Stichopus sp1) extract on fracture healing. Malays. J. Med. Sci. 2007, 14, 172. [Google Scholar]
- Fredalina, B.D.; Ridzwan, B.H.; Abidin, A.A.; Kaswandi, M.A.; Zaiton, H.; Zali, I.; Kittakoop, P.; Jais, A.M. Fatty acid compositions in local sea cucumber, Stichopus chloronotus, for wound healing. Gen. Pharmacol. 1999, 33, 337–340. [Google Scholar] [CrossRef] [PubMed]
- Croft, K.D.; Beilin, L.J.; Legge, F.M.; Vandongen, R. Effects of diets enriched in eicosapentaenoic or docosahexanoic acids on prostanoic metabolism in the rat. Lipids 1987, 22, 647–650. [Google Scholar] [CrossRef]
- Collin, P.D. Tissue Fraction of Sea Cucumber for the Treatment of Inflammation. U.S. Patent 5,770,205, 23 June 1998. [Google Scholar]
- Collin, P.D. Peptides Having Anti-Cancer and Anti-Inflammatory Activity. U.S. Patent 6,767,890, 27 July 2004. [Google Scholar]
- Whitehouse, M.W.; Fairlie, D.P.; Thong, Y.H. Anti-inflammatory activity of the isoquinoline alkaloid, tetrandrine, against established adjuvant arthritis in rats. Agents Actions 1994, 42, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Herencia, F.; Ubeda, A.; Ferrándiz, M.L.; Terencio, M.C.; Alcaraz, M.J.; García-Carrascosa, M.; Capaccioni, R.; Payá, M. Anti-inflammatory activity in mice of extracts from Mediterranean marine invertebrates. Life Sci. 1998, 62, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Belmiro, C.L.; Castelo-Branco, M.T.; Melim, L.M.; Schanaider, A.; Elia, C.; Madi, K.; Pavão, M.S.; de Souza, H.S. Unfractionated heparin and new heparin analogues from ascidians (chordate-tunicate) ameliorate colitis in rats. J. Biol. Chem. 2009, 284, 11267–11278. [Google Scholar] [CrossRef] [PubMed]
- Kozlowski, E.O.; Pavao, M.S. Effect of sulfated glycosaminoglycans on tumor invasion and metastasis. Front. Biosci. (Schol. Ed.) 2011, 13, 1541–1551. [Google Scholar] [CrossRef]
- Kozlowski, E.O.; Pavao, M.S.; Borsig, L. Ascidian dermatan sulfates attenuate metastasis, inflammation and thrombosis by inhibition of P-selectin. J. Thromb. Haemost. 2011, 9, 1807–1815. [Google Scholar] [CrossRef] [PubMed]
- Panagos, C.G.; Thomson, D.S.; Moss, C.; Hughes, A.D.; Kelly, M.S.; Liu, Y.; Chai, W.; Venkatasamy, R.; Spina, D.; Page, C.P.; et al. Fucosylated chondroitin sulfates from the body wall of the sea cucumber Holothuria forskali: Conformation, selectin binding, and biological activity. J. Biol. Chem. 2014, 289, 28284–28298. [Google Scholar] [CrossRef] [PubMed]
- Aminin, D.L.; Pinegin, B.V.; Pichugina, L.V.; Zaporozhets, T.S.; Agafonova, I.G.; Boguslavski, V.M.; Silchenko, A.S.; Avilov, S.A.; Stonik, V.A. Immunomodulatory properties of cumaside. Int. Immunopharmacol. 2006, 6, 1070–1082. [Google Scholar] [CrossRef] [PubMed]
- Aminin, D.L.; Agafonova, I.G.; Kalinin, V.I.; Silchenko, A.S.; Avilov, S.A.; Stonik, V.A.; Collin, P.D.; Woodward, C. Immunomodulatory properties of Frondoside A, a major triterpene glycoside from the North Atlantic commercially harvested sea cucumber Cucumaria frondosa. J. Med. Food. 2008, 11, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Agafonova, I.G.; Aminin, D.L.; Avilov, S.A.; Stonik, V.A. Influence of cucumariosides upon intracellular [Ca2+]i and lysosomal activity of macrophages. J. Agric. Food Chem. 2003, 51, 6982–6986. [Google Scholar] [CrossRef] [PubMed]
- Aminin, D.L.; Agafonova, I.G.; Berdyshev, E.V.; Isachenko, E.G.; Avilov, S.A.; Stonik, V.A. Immunomodulatory properties of cucumariosides from the edible Far-Eastern holothurian Cucumaria japonica. J. Med. Food 2001, 4, 127–135. [Google Scholar]
- Zou, Z.; Yi, Y.; Wu, H.; Wu, J.; Liaw, C.; Lee, K. Intercedensides A–C, three new cytotoxic triterpene glycosides from the sea cucumber Mensamaria intercedens Lampert. J. Natl. Prod. 2003, 66, 1055–1060. [Google Scholar] [CrossRef]
- Ogushi, M.; Yoshie-stark, M.; Suzuki, T. Cytostatic activity of hot water extracts from the sea cucumber in Caco-2. Food Sci. Technol. Res. 2005, 11, 202–206. [Google Scholar] [CrossRef]
- Zhao, Q.; Xue, Y.; Liu, Z.; Li, H.; Wang, J.; Li, Z.; Wang, Y.; Dong, P.; Xue, C. Differential effects of sulfated triterpene glycosides, holothurin A1, and 24-dehydroechinoside A, on antimetastasic activity via regulation of the MMP-9 signal pathway. J. Food Sci. 2010, 75, 280–288. [Google Scholar] [CrossRef]
- Li, X.; Roginsky, A.B.; Ding, X.-Z.; Woodward, C.; Collin, P.; Newman, R.A.; Bell, R.H.; Adrian, T.E., Jr. Review of the apoptosis pathways in pancreatic cancer and the anti-apoptotic effects of the novel sea cucumber compound, Frondoside A. Ann. N. Y. Acad. Sci. 2008, 1138, 181–198. [Google Scholar] [CrossRef] [PubMed]
- Al Shemaili, J.; Mensah-Brown, E.; Parekh, K.; Thomas, S.A.; Attoub, S.; Hellman, B.; Nyberg, F.; Adem, A.; Collin, P.; Adrian, T.E. Frondoside A enhances the antiproliferative effects of gemcitabine in pancreatic cancer. Eur. J. Cancer 2014, 50, 1391–1398. [Google Scholar] [CrossRef] [PubMed]
- Attoub, S.; Arafat, K.; Gélaude, A.; Al Sultan, M.A.; Bracke, M.; Collin, P.; Takahashi, T.; Adrian, T.E.; de Wever, O. Frondoside A suppressive effects on lung cancer survival, tumor growth, angiogenesis, invasion, and metastasis. PLoS ONE 2013, 8, e53087. [Google Scholar] [CrossRef] [PubMed]
- Al Marzouqi, N.; Iratni, R.; Nemmar, A.; Arafat, K.; Ahmed Al Sultan, M.; Yasin, J.; Collin, P.; Mester, J.; Adrian, T.E.; Attoub, S. Frondoside A inhibits human breast cancer cell survival, migration, invasion and the growth of breast tumor xenografts. Eur. J. Pharmacol. 2011, 668, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Kundu, N.; Collin, P.D.; Goloubeva, O.; Fulton, A.M. Frondoside A inhibits breast cancer metastasis and antagonizes prostaglandin E receptors EP4 and EP2. Breast Cancer Res. Treat. 2012, 132, 1001–1008. [Google Scholar] [CrossRef] [PubMed]
- Roginsky, A.B.; Ding, X.Z.; Woodward, C.; Ujiki, M.B.; Singh, B.; Bell, R.H., Jr.; Collin, P.; Adrian, T.E. Anti-pancreatic cancer effects of a polar extract from the edible sea cucumber, Cucumaria frondosa. Pancreas 2010, 39, 646–652. [Google Scholar] [CrossRef] [PubMed]
- Kale, V.; Freysdottir, J.; Paulsen, B.S.; Friðjónsso, O.H.; Hreggviðsson, G.O.; Omarsdottir, S. Sulphated polysaccharide from the sea cucumber Cucumaria frondosa affect maturation of human dendritic cells and their activation of allogeneic CD4(+) Tcells in vitro. Bioact. Carbohydr. Diet. Fibre 2013, 2, 108–117. [Google Scholar] [CrossRef]
- Himaya, S.W.A.; Ryu, B.; Qian, Z.-J.; Kim, S.-K. Sea cucumber, Stichopus japonicus ethyl acetate fraction modulates the lipopolysaccharide induced iNOS and COX-2 via MAPK signaling pathway in murine macrophages. Environ. Toxicol. Pharmacol. 2010, 30, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Avilov, S.A.; Kalinin, V.I.; Kalinovsky, A.I.; Dmitrenok, P.S.; Fedorov, S.N.; Stepanov, V.G.; Dong, Z.; Stonik, V.A. Constituents of the sea cucumber Cucumaria okhotensis. Structures of okhotosides B1–B3 and cytotoxic activities of some glycosides from this species. J. Natl. Prod. 2007, 71, 351–356. [Google Scholar] [CrossRef]
- Wu, F.J.; Xue, Y.; Liu, X.F.; Xue, C.H.; Wang, J.F.; Du, L.; Takahashi, K.; Wang, Y.M. The protective effect of eicosapentaenoic acid-enriched phospholipids from sea cucumber Cucumaria frondosa on oxidative stress in PC12 cells and SAMP8 mice. Neurochem. Int. 2014, 64, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Cansell, M.; Nacka, F.; Combe, N. Marine lipid-based liposomes increase in vivo FA bioavailability. Lipids 2003, 38, 551–559. [Google Scholar] [CrossRef] [PubMed]
- Lemaitre-Delaunay, D.; Pachiaudi, M.; Laville, J.; Pousin, M.; Armstrong, M.; Lagarde, M. Blood compartmental metabolism of docosahexaenoic acid (DHA) in humans after ingestion of a single dose of [13C]DHA in phosphatidylcholine. J. Lipid Res. 1999, 40, 1867–1874. [Google Scholar] [PubMed]
- Hu, S.; Xu, L.; Shi, D.; Wang, J.; Wang, Y.; Lou, Q.; Xue, C. Eicosapentaenoic acid-enriched phosphatidylcholine isolated from Cucumaria frondosa exhibits anti-hyperglycemic effects via activating phosphoinositide 3-kinase/protein kinase B signal pathway. J. Biosci. Bioeng. 2014, 117, 457–463. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janakiram, N.B.; Mohammed, A.; Rao, C.V. Sea Cucumbers Metabolites as Potent Anti-Cancer Agents. Mar. Drugs 2015, 13, 2909-2923. https://doi.org/10.3390/md13052909
Janakiram NB, Mohammed A, Rao CV. Sea Cucumbers Metabolites as Potent Anti-Cancer Agents. Marine Drugs. 2015; 13(5):2909-2923. https://doi.org/10.3390/md13052909
Chicago/Turabian StyleJanakiram, Naveena B., Altaf Mohammed, and Chinthalapally V. Rao. 2015. "Sea Cucumbers Metabolites as Potent Anti-Cancer Agents" Marine Drugs 13, no. 5: 2909-2923. https://doi.org/10.3390/md13052909
APA StyleJanakiram, N. B., Mohammed, A., & Rao, C. V. (2015). Sea Cucumbers Metabolites as Potent Anti-Cancer Agents. Marine Drugs, 13(5), 2909-2923. https://doi.org/10.3390/md13052909





