Intracellular Immunohistochemical Detection of Tetrodotoxin in Pleurobranchaea maculata (Gastropoda) and Stylochoplana sp. (Turbellaria)
Abstract
:1. Introduction
2. Results and Discussion
2.1. Pleurobranchaea Maculata
2.1.1. Mantle
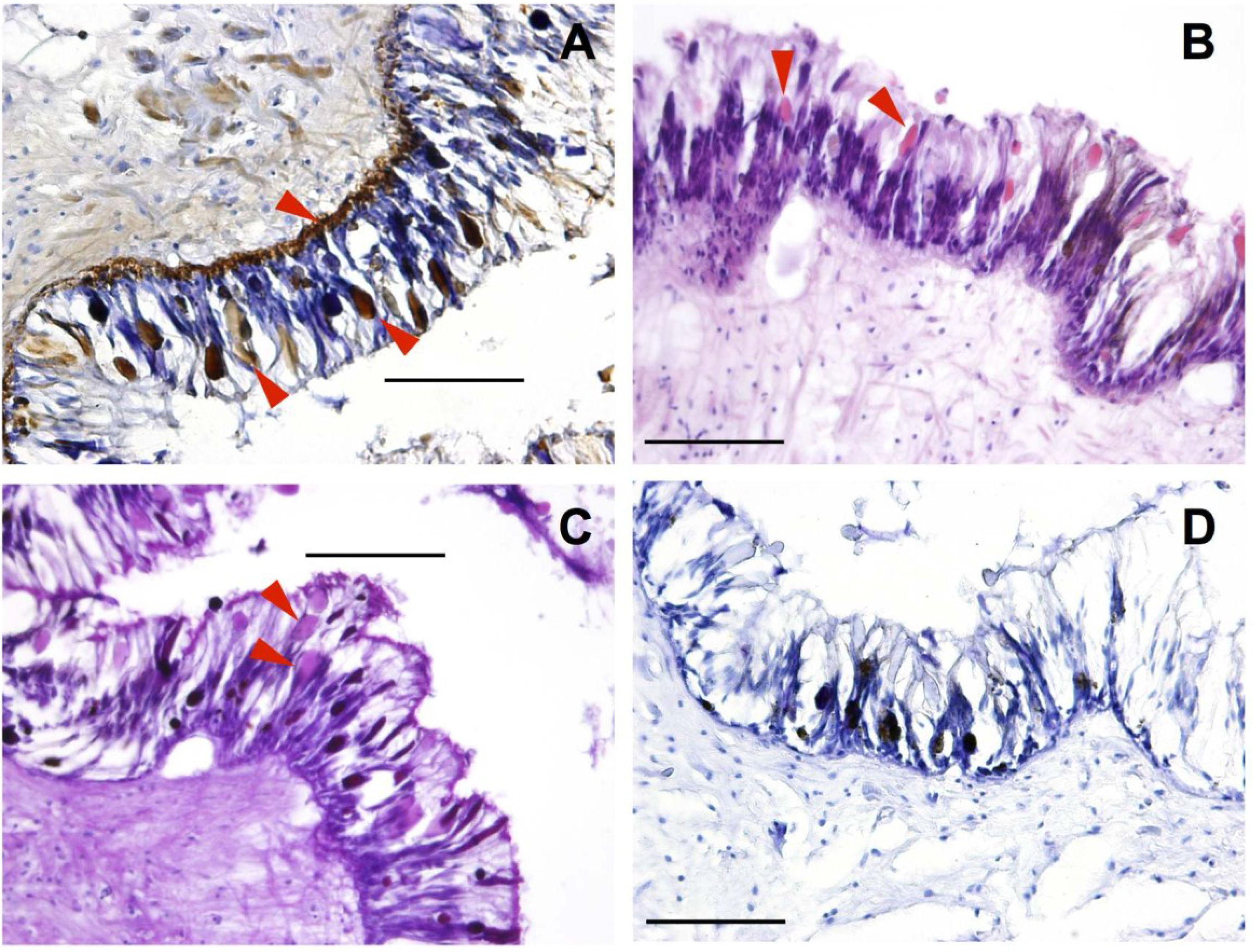
2.1.2. Reproductive and Digestive Tissue
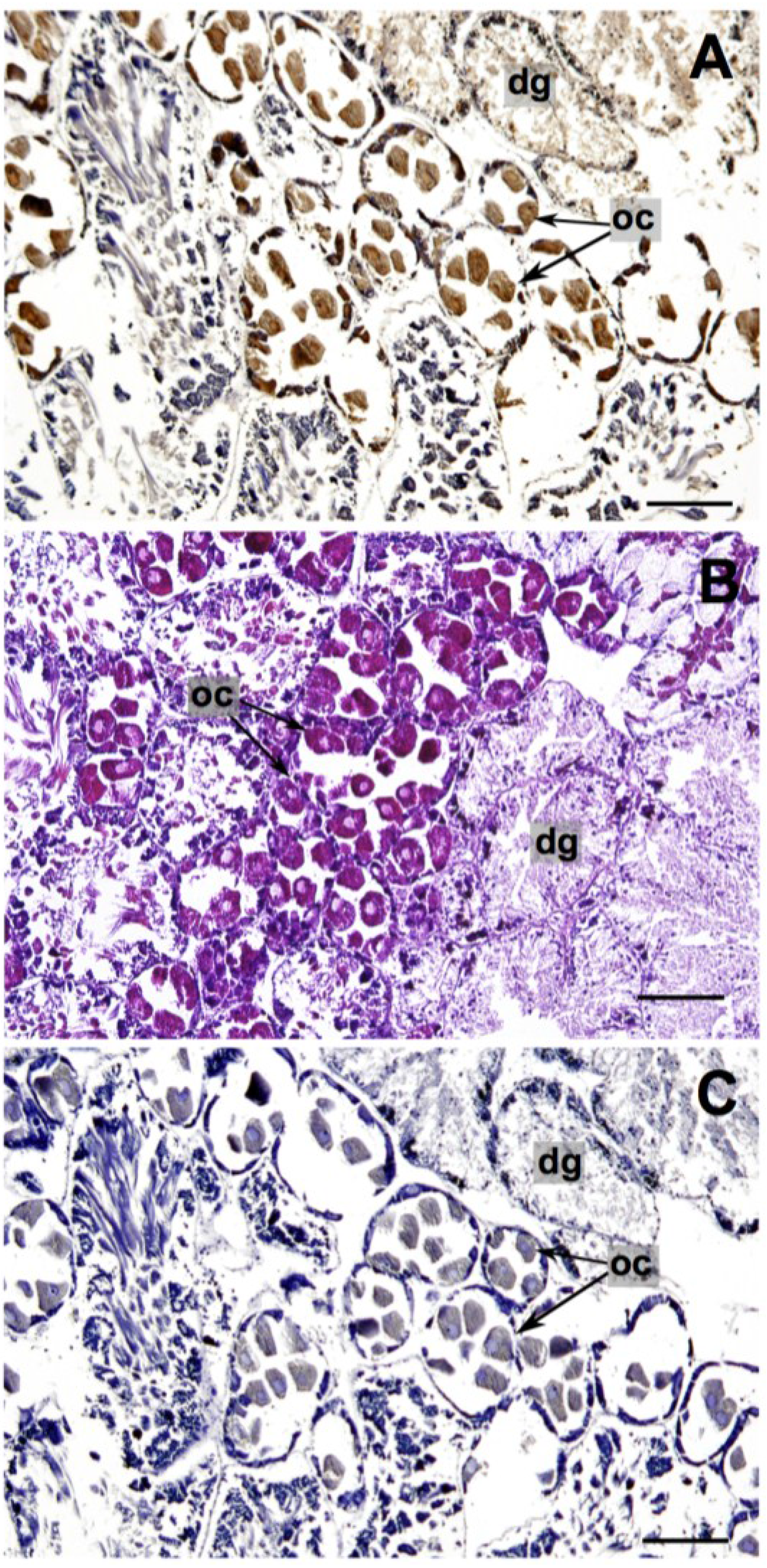
2.2. Stylochoplana sp.
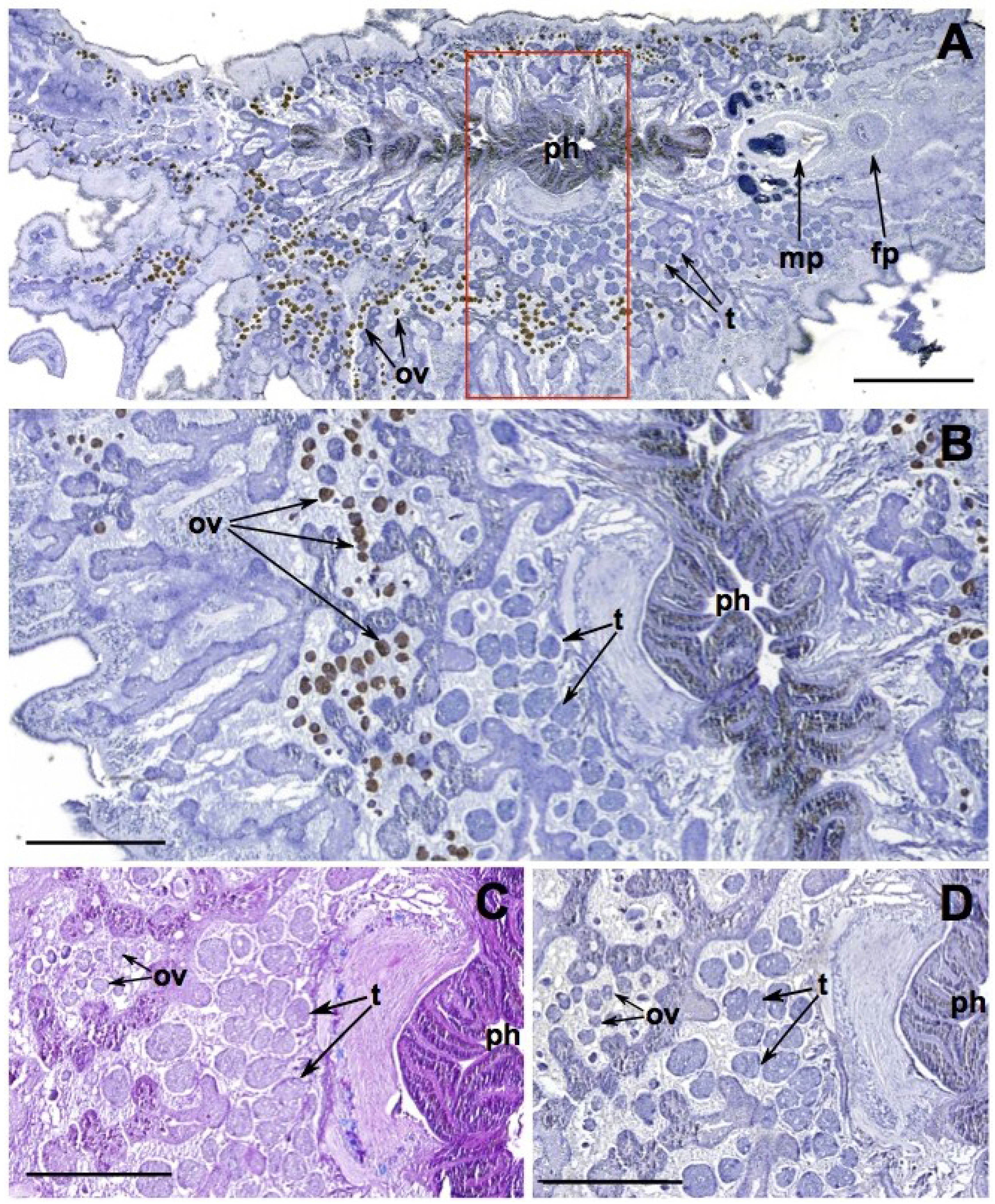
2.3. Offspring
| Sample | TTX |
|---|---|
| P.M egg mass (−eggs) | ND |
| P.M. egg mass (+eggs) | 3.7 mg·kg−1 |
| P.M. larvae | 48.3 pg·indivdual−1 |
| S.S. egg masses | 108 ± 2 mg·kg−1 |
3. Experimental Section
3.1. Specimen Collection
3.2. Histochemistry
| Step | Solution | Time (min) |
|---|---|---|
| 1. | 3% H2O2/10% methanol | 10 |
| 2. | 1× PBS | 10 × 3 |
| 3. | Normal Goat Serum (Vector Labs) | 20 |
| 4. | 1× PBS | 10 × 3 |
| 5. | mAB T20G10 * | Overnight at 4 °C |
| 6. | 1× PBS | 10 × 3 |
| 7. | Biotinylated secondary antibody (anti-rabbit IgG) * | 60 |
| 8. | 1× PBS | 10 × 3 |
| 9. | VECTASTAIN® ABC reagent * | 60 |
| 10. | 1× PBS | 10 × 3 |
| 11. | DAB | 2–5 |
| 12. | Deionized H2O | 5 |
| 13. | Counterstain (Gill’s II Hematoxylin) | 2 |
3.3. Tetrodotoxin Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Noguchi, T.; Arakawa, O.; Takatani, T. TTX accumulation in pufferfish. Comp. Biochem. Physiol. D Genomics Proteomics 2006, 1, 145–152. [Google Scholar] [CrossRef]
- Noguchi, T.; Arakawa, O. Tetrodotoxin—Distribution and accumulation in aquatic organisms, and cases of human intoxication. Mar. Drugs 2008, 6, 220–242. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, T.; Ebesu, J.S.M. Puffer poisoning: Epidemiology and treatment. J. Toxicol. Toxin Rev. 2001, 20, 1–10. [Google Scholar] [CrossRef]
- Chau, R.; Kalaitzis, J.A.; Neilan, B.A. On the origins and biosynthesis of tetrodotoxin. Aquat. Toxicol. 2011, 104, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, K.; Noguchi, T. Distribution and origin of tetrodotoxin. Toxin Rev. 2001, 20, 11–33. [Google Scholar] [CrossRef]
- Stokes, A.N.; Ducey, P.K.; Neuman-Lee, L.; Hanifin, C.T.; French, S.S.; Pfrender, M.E.; Brodie, E.D., III; Brodie, E.D., Jr. Confirmation and distribution of tetrodotoxin for the first time in terrestrial invertebrates: Two terrestrial flatworm species (Bipalium adventitium and Bipalium kewense). PLoS One 2014, 9, e100718. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-J.; Yu, R.-C.; Luo, X.; Zhou, M.-J.; Lin, X.-T. Toxin-screening and identification of bacteria isolated from highly toxic marine gastropod Nassarius semiplicatus. Toxicon 2008, 52, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.L.; Xie, L.P.; Xia, G.L.; Zhang, J.F.; Nie, Y.C.; Hu, J.C.; Wang, S.J.; Zhang, R.Q. A new tetrodotoxin-producing actinomycete, Nocardiopsis dassonvillei, isolated from the ovaries of puffer fish Fugu rubripes. Toxicon 2005, 45, 851–859. [Google Scholar] [CrossRef] [PubMed]
- Yasumoto, T.; Yasumura, D.; Yotsu, M.; Michishita, T.; Endo, A.; Kotaki, Y. Bacterial production of tetrodotoxin and anhydrotetrodotoxin. Agric. Biol. Chem. 1986, 50, 793–795. [Google Scholar] [CrossRef]
- Daly, J.W.; Padgett, W.L.; Saunders, R.L.; Cover, J.F. Absence of tetrodotoxins in a captive-raised riparian frog, Atelopus varius. Toxicon 1997, 35, 705–709. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, T.; Maruyama, J.; Hashimoto, K.; Narita, H. Tetrodotoxin in the starfish Astropecten polyacanthus, in association with toxification of a trumpet shell, “Boshubora” Charonia sauliae. Bull. Jpn. Soc. Sci. Fish 1982, 48, 1173–1177. [Google Scholar] [CrossRef]
- Wood, S.A.; Casas, M.; Taylor, D.; McNabb, P.; Salvitti, L.; Ogilvie, S.; Cary, S.C. Depuration of tetrodotoxin and changes in bacterial communities in Pleurobranchea maculata adults and egg masses maintained in captivity. J. Chem. Ecol. 2012, 38, 1342–1350. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.A.; Hwang, D.F.; Tsai, Y.H.; Chen, H.C.; Jeng, S.S.; Noguchi, T.; Ohwada, K.; Hashimoto, K. Microflora and tetrodotoxin-producing bacteria in a gastropod, Niotha clathrata. Food Chem. Toxicol. 1995, 33, 929–934. [Google Scholar] [CrossRef] [PubMed]
- Hwang, D.F.; Arakawa, O.; Saito, T.; Noguchi, T.; Simidu, U.; Tsukamoto, K.; Shida, Y.; Hashimoto, K. Tetrodotoxin-producing bacteria from the blue-ringed octopus Octopus maculosus. Mar. Biol. 1989, 100, 327–332. [Google Scholar] [CrossRef]
- Noguchi, T.; Hwang, D.F.; Arakawa, O.; Sugita, H.; Deguchi, Y.; Shida, Y.; Hashimoto, K. Vibrio alginolyticus, a tetrodotoxin-producing bacterium, in the intestines of the fish Fugu vermicularis vermicularis. Mar. Biol. 1987, 94, 625–630. [Google Scholar] [CrossRef]
- Yang, G.; Xu, J.; Liang, S.; Ren, D.; Yan, X.; Bao, B. A novel TTX-producing Aeromonas isolated from the ovary of Takifugu obscurus. Toxicon 2010, 56, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Cardall, B.L.; Brodie, E.D.; Hanifin, C.T. Secretion and regeneration of tetrodotoxin in the rough-skin newt (Taricha granulosa). Toxicon 2004, 44, 933–938. [Google Scholar] [CrossRef] [PubMed]
- Hanifin, C.T.; Brodie, E.D., III; Brodie, E.D., Jr. Tetrodotoxin levels of the rough-skin newt, Taricha granulosa, increase in long-term captivity. Toxicon 2002, 40, 1149–1153. [Google Scholar] [CrossRef] [PubMed]
- Mebs, D.; Arakawa, O.; Yotsu-Yamashita, M. Tissue distribution of tetrodotoxin in the red-spotted newt Notophthalmus viridescens. Toxicon 2010, 55, 1353–1357. [Google Scholar] [CrossRef] [PubMed]
- Tsuruda, K.; Arakawa, O.; Kawatsu, K.; Hamano, Y.; Takatani, T.; Noguchi, T. Secretory glands of tetrodotoxin in the skin of the Japanese newt Cynops pyrrhogaster. Toxicon 2002, 40, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Tanu, M.B.; Mahmud, Y.; Arakawa, O.; Takatani, T.; Kajihara, H.; Kawatsu, K.; Hamano, Y.; Asakawa, M.; Miyazawa, K.; Noguchi, T. Immunoenzymatic visualization of tetrodotoxin (TTX) in Cephalothrix species (Nernertea: Anopla: Palaeonemertea: Cephalotrichidae) and Planocera reticulata (Platyhelminthes: Turbellaria: Polycladida: Planoceridae). Toxicon 2004, 44, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Mahmud, Y.; Arakawa, O.; Ichinose, A.; Tanu, M.B.; Takatani, T.; Tsuruda, K.; Kawatsu, K.; Hamano, Y.; Noguchi, T. Intracellular visualization of tetrodotoxin (TTX) in the skin of a puffer Tetraodon nigroviridis by immunoenzymatic technique. Toxicon 2003, 41, 605–611. [Google Scholar] [CrossRef] [PubMed]
- Mahmud, Y.; Okada, K.; Takatani, T.; Kawatsu, K.; Hamano, Y.; Arakawa, O.; Noguchi, T. Intra-tissue distribution of tetrodotoxin in two marine puffers Takifugu vermicularis and Chelonodon patoca. Toxicon 2003, 41, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Tanu, M.B.; Mahmud, Y.; Takatani, T.; Kawatsu, K.; Hamano, Y.; Arakawa, O.; Noguchi, T. Localization of tetrodotoxin in the skin of a brackishwater puffer Tetraodon steindachneri on the basis of immunohistological study. Toxicon 2002, 40, 103–106. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.L.; Stark, M.R.; Caldwell, R.L. Microdistribution of tetrodotoxin in two species of blue-ringed octopuses (Hapalochlaena lunulata and Hapalochlaena fasciata) detected by fluorescent immunolabeling. Toxicon 2012, 60, 1307–1313. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.L.; Hanifin, C.T.; Brodie, E.D., Jr.; Brodie, E.D., III. Tetrodotoxin affects survival probability of rough-skinned newts (Taricha granulosa) faced with TTX-resistant garter snake predators (Thamnophis sirtalis). Chemoecology 2010, 20, 285–290. [Google Scholar] [CrossRef]
- McNabb, P.; Selwood, A.I.; Munday, R.; Wood, S.A.; Taylor, D.I.; MacKenzie, L.A.; van Ginkel, R.; Rhodes, L.L.; Cornelisen, C.; Heasman, K.; et al. Detection of tetrodotoxin from the grey side-gilled sea slug—Pleurobranchaea maculata, and associated dog neurotoxicosis on beaches adjacent to the Hauraki Gulf, Auckland, New Zealand. Toxicon 2010, 56, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Wood, S.A.; Taylor, D.I.; McNabb, P.; Walker, J.; Adamson, J.; Cary, S.C. Tetrodotoxin concentrations in Pleurobranchaea maculata: Temporal, spatial and individual variability from New Zealand populations. Mar. Drugs 2012, 10, 163–176. [Google Scholar] [CrossRef] [PubMed]
- Salvitti, L.R.; Wood, S.A.; Taylor, D.I.; McNabb, P.; Cary, S.C. First identification of tetrodotoxin (TTX) in the flatworm Stylochoplana sp.; a source of TTX for the sea slug Pleurobranchaea maculata. Toxicon 2015, 95, 23–29. [Google Scholar] [CrossRef]
- Rivera, V.R.; Poli, M.A.; Bignami, G.S. Prophylaxis and treatment with a monoclonal antibody of tetrodotoxin poisoning in mice. Toxicon 1995, 33, 1231–1237. [Google Scholar] [CrossRef] [PubMed]
- Putz, A.; Koenig, G.M.; Waegele, H. Defensive strategies of Cladobranchia (Gastropoda, Opisthobranchia). Nat. Prod. Rep. 2010, 27, 1386–1402. [Google Scholar] [CrossRef] [PubMed]
- Wagele, H.; Klussmann-Kolb, A. Opisthobranchia (Mollusca, Gastropoda)—More than just slimy slugs. Shell reduction and its implications on defence and foraging. Front. Zool. 2005, 2, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Wagele, H.; Ballesteros, M.; Avila, C. Defensive glandular structures in opisthobranch molluscs—From histology to ecology. In Oceanography and Marine Biology—An Annual Review; Gibson, R.N., Atkinson, R.J.A., Gordon, J.D.M., Eds.; Taylor & Francis: Abingdon, UK, 2006; pp. 197–276. [Google Scholar]
- Thompson, T.E. Detection of epithelial acid secretions in marine molluscs: Review of techniques, and new analytical methods. Comp. Biochem. Physiol. A Physiol. 1983, 74, 615–621. [Google Scholar] [CrossRef]
- Thompson, T.E. Acid secretion in the Pacific Ocean gastropods. Aust. J. Zool. 1969, 17, 755–764. [Google Scholar] [CrossRef]
- Cattaneo-Vietti, R.; Angelini, S.; Gaggero, L.; Lucchetti, G. Mineral composition of nudibranch spicules. J. Molluscan Stud. 1995, 61, 331–337. [Google Scholar] [CrossRef]
- Derby, C.D. Escape by inking and secreting: Marine molluscs avoid predators through a rich array of chemicals and mechanisms. Biol. Bull. 2007, 213, 274–289. [Google Scholar] [CrossRef] [PubMed]
- Pawlik, J.R. Marine invertebrate chemical defenses. Chem. Rev. 1993, 93, 1911–1922. [Google Scholar] [CrossRef]
- Willan, R.C. New Zealand side-gilled sea slugs (Opisthobranchia: Notaspidea: Pluerobranchidae). Malacologia 1983, 23, 221–270. [Google Scholar]
- Itoi, S.; Yoshikawa, S.; Tatsuno, R.; Suzuki, M.; Asahina, K.; Yamamoto, S.; Takanashi, S.; Tomohiro, T.; Arakawa, O.; Sakakura, Y.; et al. Difference in the localization of tetrodotoxin between the female and male pufferfish Takifugu niphobles, during spawning. Toxicon 2012, 60, 1000–1004. [Google Scholar] [CrossRef] [PubMed]
- Mosher, H.S.; Fuhrman, F.A.; Buchwald, H.D.; Fischer, H.G. Tarichatoxin-tetrodotoxin: A potent neurotoxin. Sci. New Ser. 1964, 144, 1100–1110. [Google Scholar]
- Pires, O.R.; Sebben, A.; Schwartz, E.F.; Largura, S.W.R.; Bloch, C.; Morales, R.A.V.; Schwartz, C.A. Occurrence of tetrodotoxin and its analogues in the Brazilian frog Brachycephalus ephippium (Anura: Brachycephalidae). Toxicon 2002, 40, 761–766. [Google Scholar] [CrossRef] [PubMed]
- Yotsu, M.; Iorizzi, M.; Yasumoto, T. Distribution of tetrodotoxin, 6-epitetrodotoxin, and 11-deoxytetrodotoxin in newts. Toxicon 1990, 28, 238–241. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.L.; Hanifin, C.T.; Brodie, E.D., Jr.; Caldwell, R.L. Ontogeny of tetrodotoxin levels in blue-ringed octopuses: Maternal investment and apparent independent production in offspring of Hapalochlaena lunulata. J. Chem. Ecol. 2011, 37, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Itoi, S.; Yoshikawa, S.; Asahina, K.; Suzuki, M.; Ishizuka, K.; Takimoto, N.; Mitsuoka, R.; Yokoyama, N.; Detake, A.; Takayanagi, C.; et al. Larval pufferfish protected by maternal tetrodotoxin. Toxicon 2014, 78, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Khor, S.; Wood, S.A.; Salvitti, L.; Taylor, D.I.; Adamson, J.; McNabb, P.; Cary, S.C. Investigating diet as the source of tetrodotoxin in Pleurobranchaea maculata. Mar. Drugs 2013, 12, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, K.; Jeon, J.K.; Noguchi, T.; Ito, K.; Hashimoto, K. Distribution of tetrodotoxin in the tissues of the flatworm Planocera multitentaculata (Platyhelminthes). Toxicon 1987, 25, 975–980. [Google Scholar] [CrossRef] [PubMed]
- Ritson-Williams, R.; Yotsu-Yamashita, M.; Paul, V.J. Ecological functions of tetrodotoxin in a deadly polyclad flatworm. Proc. Natl. Acad. Sci. USA 2006, 103, 3176–3179. [Google Scholar] [CrossRef] [PubMed]
- Hanifin, C.T.; Brodie, E.D., III; Brodie, E.D., Jr. Tetrodotoxin levels in eggs of the rough-skin newt, Taricha granulosa, are correlated with female toxicity. J. Chem. Ecol. 2003, 29, 1729–1739. [Google Scholar] [CrossRef] [PubMed]
- Kungsuwan, A.; Nagashima, Y.; Noguchi, T.; Shida, Y.; Suvapeepan, S.; Suwansakornkul, P.; Hashimoto, K. Tetrodotoxin in the horseshoe crab Carcinoscorpius rotundicauda inhabiting Thailand. Nippon Suisan Gakkaishi 1987, 53, 261–266. [Google Scholar] [CrossRef]
- Tanu, M.B.; Noguchi, T. Tetrodotoxin as a toxic principle in the horseshoe crab Carcinoscorpius rotundicauda collected from Bangladesh. J. Food Hyg. Soc. Jpn. 1999, 40, 426–430. [Google Scholar] [CrossRef]
- Sheumack, D.D.; Howden, M.E.H.; Spence, I. Occurrence of a tetrodotoxin-like compound in the eggs of the venomous blue-ringed octopus (Hapalochlaena maculosa). Toxicon 1984, 22, 811–812. [Google Scholar] [CrossRef] [PubMed]
- Pavelka, L.A.; Kim, Y.H.; Mosher, H.S. Tetrodotoxin and tetrodotoxin-like compounds from the eggs of the Costa Rican frog, Atelopus chiriquiensis. Toxicon 1977, 15, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, K. Tetrodotoxin as a pheromone. Nature 1995, 378, 563–564. [Google Scholar] [CrossRef] [PubMed]
- Nagashima, Y.; Mataki, I.; Toyoda, M.; Nakajima, H.; Tsumotoo, K.; Shimakura, K.; Shioma, K. Change in tetrodotoxin content of the puffer fish Takifugu rubripes during seed production from fertilized eggs to juveniles. Food Hyg. Saf. Sci. 2010, 51, 48–51. [Google Scholar] [CrossRef]
- Newman, L.J.; Cannon, L.R.G. Marine Flatworms: The World of Polyclad Flatworms; CSIRO Publishing: Melbourne, Victoria, Australia, 2003. [Google Scholar]
- Kiernan, J. Histological and Histochemical Methods: Theory and Practice, 4th ed.; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2008. [Google Scholar]
- Ho, B.; Yeo, D.S.A.; Ding, J.L. A tetrodotoxin neutralizing system in the hemolymph of the horseshoe-crab, Carcinoscorpius rotundicauda. Toxicon 1994, 32, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Llewellyn, L.E. Haemolymph protein in xanthid crabs: Its selective binding of saxitoxin and possible role in toxin bioaccumulation. Mar. Biol. 1997, 128, 599–606. [Google Scholar] [CrossRef]
- Hwang, P.-A.; Tsai, Y.-H.; Lin, H.-P.; Hwang, D.-F. Tetrodotoxin-binding proteins isolated from five species of toxic gastropods. Food Chem. 2007, 103, 1153–1158. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salvitti, L.R.; Wood, S.A.; Winsor, L.; Cary, S.C. Intracellular Immunohistochemical Detection of Tetrodotoxin in Pleurobranchaea maculata (Gastropoda) and Stylochoplana sp. (Turbellaria). Mar. Drugs 2015, 13, 756-769. https://doi.org/10.3390/md13020756
Salvitti LR, Wood SA, Winsor L, Cary SC. Intracellular Immunohistochemical Detection of Tetrodotoxin in Pleurobranchaea maculata (Gastropoda) and Stylochoplana sp. (Turbellaria). Marine Drugs. 2015; 13(2):756-769. https://doi.org/10.3390/md13020756
Chicago/Turabian StyleSalvitti, Lauren R., Susanna A. Wood, Leigh Winsor, and Stephen Craig Cary. 2015. "Intracellular Immunohistochemical Detection of Tetrodotoxin in Pleurobranchaea maculata (Gastropoda) and Stylochoplana sp. (Turbellaria)" Marine Drugs 13, no. 2: 756-769. https://doi.org/10.3390/md13020756
APA StyleSalvitti, L. R., Wood, S. A., Winsor, L., & Cary, S. C. (2015). Intracellular Immunohistochemical Detection of Tetrodotoxin in Pleurobranchaea maculata (Gastropoda) and Stylochoplana sp. (Turbellaria). Marine Drugs, 13(2), 756-769. https://doi.org/10.3390/md13020756






