Abstract
Cancer and infectious diseases continue to be a major public health problem, and new drugs are necessary. As marine organisms are well known to provide a wide range of original compounds, the aim of this study was to investigate the bioactivity of the main constituents of the cosmopolitan red alga, Sphaerococcus coronopifolius. The structure of several bromoditerpenes was determined by extensive spectroscopic analysis and comparison with literature data. Five molecules were isolated and characterized which include a new brominated diterpene belonging to the rare dactylomelane family and named sphaerodactylomelol (1), along with four already known sphaerane bromoditerpenes (2–5). Antitumor activity was assessed by cytotoxicity and anti-proliferative assays on an in vitro model of human hepatocellular carcinoma (HepG-2 cells). Antimicrobial activity was evaluated against four pathogenic microorganisms: Escherichia coli, Pseudomonas aeruginosa, Staphylococcus aureus and Candida albicans. Compound 4 exhibited the highest antimicrobial activity against S. aureus (IC50 6.35 µM) and compound 5 the highest anti-proliferative activity on HepG-2 cells (IC50 42.9 µM). The new diterpene, sphaerodactylomelol (1), induced inhibition of cell proliferation (IC50 280 µM) and cytotoxicity (IC50 720 µM) on HepG-2 cells and showed antimicrobial activity against S. aureus (IC50 96.3 µM).
Keywords:
red algae; Sphaerococcus; diterpenes; sphaerane; dactylomelane; HepG-2; pathogenic microorganisms 1. Introduction
Cancer and infectious diseases continue to be one of the major public health concerns, and consequently, there is a perpetual need for new chemotherapeutics to fight new diseases and drug resistance. In the last few decades, nature has played a significant role as a source of new drugs, and recent trends in drug research emphasize that the marine environment has a high potential for the discovery of new pharmaceuticals [1,2,3]. Marine ecosystems are among the richest and most complex ones in terms of biodiversity. Original chemical and physical conditions in such an environment provide conditions for the production of quite specific and potent active molecules. Among other reasons, marine organisms have been found to produce original and bioactive substances because they are living in an exigent, competitive and aggressive environment [4,5]. These characteristics render marine organisms ideal candidates as novel sources of both preexisting and unrecognized high value-added biomolecules with potential for providing sustainable economic and human benefits [6]. Marine algae have been one of the richest and promising sources of bioactive specialized metabolites that probably have diverse simultaneous functions for their producer and can act, for example, as antimicrobial, antifouling and herbivore deterrents or as ultraviolet-screening agents [7,8,9]. These defensive strategies can result in a high level of structural and chemical diversity for the metabolites, originating from different metabolic pathways with great pharmaceutical and biomedical potential [10,11]. Marine algae-originated compounds have been found to be associated with numerous health-promoting effects, including, in particular, anti-oxidative, anti-inflammatory, antiviral, antimicrobial or anticancer effects [10,12]. Well-documented bioactive metabolites of marine algae include mainly brominated phenols, polysaccharides, but especially, a large variety of terpenoids, several of them being halogenated [13,14,15,16].
Since its first chemical analysis in 1976, the cosmopolitan red alga, Sphaerococcus coronopifolius, has yielded a large number of interesting brominated cyclic diterpenes, most of them containing one or two bromine atoms [17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40]. Concerning the biological activities of these compounds, some of them have already demonstrated antibacterial activity against Gram-positive bacteria [33], and others have been assayed for their cytotoxicity against human lung cancer cell lines [37] or their antibacterial activity against multidrug-resistant and methicillin-resistant Staphylococcus aureus strains [38,39]. Several bromoditerpenes have also been screened against the model organism, Amphibalanus amphitrite, in order to evaluate their antifouling properties.
The aim of the present study was to address the antitumor and antimicrobial bioactivity characterization of the major brominated diterpenes isolated from samples of S. coronopifolius collected in the Atlantic, while most of the chemically-studied specimens of this algal species were collected from the Mediterranean or the Adriatic seas.
2. Results and Discussion
2.1. In Vitro Bioactivity-Guided Fractionation
The screening of antitumor and antimicrobial activities of compounds isolated from S. coronopifolius was performed for the methanol (MeOH) and dichloromethane (CH2Cl2) extracts of this alga. The CH2Cl2 extract exhibited the highest anti-proliferative and antimicrobial activities (Table 1 and Table 2, respectively). Consequently, in order to isolate and identify the compounds responsible for these biological activities, this extract was further studied. It was first fractionated by normal phase vacuum liquid chromatography (VLC) on silica with eluents of increasing polarities (from cyclohexane to EtOAc), yielding five fractions (F1–F5 through a 25% step). In the first bioassay, fraction F2 exhibited the strongest cytotoxicity (IC50 104 µg/mL) and anti-proliferative (19.8 µg/mL) activity (Table 1). On the other hand, fractions F1, F3, F4 and F5 demonstrated much more potency in the inhibition of HepG-2 cell proliferation than in the reduction of their viability. In antimicrobial assays (Table 2), the highest growth inhibition was measured against S. aureus for fractions F2 and F3 with IC50 of 5.10 and 5.39 µg/mL, respectively. In the antifungal assay, the highest activity was exhibited against C. albicans by fraction F2 with an IC50 of 53.9 µg/mL.

Table 1.
Cytotoxicity and anti-proliferative (IC50) effects induced on HepG-2 cells by crude extracts and VLC fractions of S. coronopifolius. IC50 values are expressed as the means of eight independent experiments with 95% confidence intervals.
| IC50 (µg/mL) | ||||
|---|---|---|---|---|
| Cytotoxicity | Anti-Proliferative | |||
| Crude extracts | MeOH | 470.6 (310.7–712.6) | 646.5 (398.4–1049.0) | |
| CH2Cl2 | 14.13 (8.12–24.60) | 32.32 (22.37–46.70) | ||
| VLC fractions | F1 | >1000 | 102.5 (68.08–154.2) | |
| F2 | 104.3 (81.82–132.9) | 19.78 (13.79–28.38) | ||
| F3 | >1000 | 70.17 (38.78–127.0) | ||
| F4 | >1000 | 36.68 (23.37–57.55) | ||
| F5 | >1000 | 39.32 (25.89–59.71) | ||
According to these results, several VLC fractions showed interesting effects against different targets, and it was decided to purify and structurally characterize the main components of fractions F2 and F3, due to their high anti-proliferative and antimicrobial effects, respectively.

Table 2.
Antimicrobial activities (IC50) against E. coli, P. aeruginosa, S. aureus and C. albicans of crude extracts and VLC fractions obtained from S. coronopifolius. IC50 values are expressed as the means of eight independent experiments with 95% confidence intervals.
| IC50 (µg/mL) | |||||
|---|---|---|---|---|---|
| E. coli | P. aeruginosa | S. aureus | C. albicans | ||
| Crude Extracts | MeOH | >1000 | >1000 | 73.65 (58.52–92.69) | >1000 |
| CH2Cl2 | 267.1 (231.5–308.1) | 363.1 (207.0–637.1) | 25.15 (13.47–46.96) | 435.9 (285.6–665.5) | |
| VLC Fractions | F1 | 107.0 (92.02–124.5) | 338.7 (248.3–461.9) | 16.49 (10.19–26.66) | 78.61 (58.12–106.3) |
| F2 | 228.4 (188.7–276.4) | 141.5 (117.6–170.4) | 5.10 (4.50–5.78) | 538.9 (308.4–941.6) | |
| F3 | >1000 | 599.9 (307.4–1171.0) | 5.39 (4.19–6.93) | >1000 | |
| F4 | 433.9 (358.5–525.1) | 436.4 (184.1–1035.0) | 6.45 (5.16–8.05) | >1000 | |
| F5 | 757.0 (614.5–932.6) | 422.8 (291.9–612.4) | 13.16 (9.98–17.35) | >1000 | |
2.2. Isolation and Structure Elucidation of the Major Compounds of Fractions F2 and F3
The HPLC purifications of the selected VLC fractions, F2 and F3, followed by the structural identification of the major compounds by NMR and MS led to the identification of five diterpenes belonging to two distinct families: one new dactylomelane (1) and four known sphaeranes (2–5) (Figure 1). It was not surprising to find compounds belonging to the sphaerane class of diterpenes in our samples. In comparison with literature data, we were thus able to identify, unambiguously, bromosphaerol (2) [31], 12S-hydroxybromosphaerol (3) [34], 12R-hydroxybromosphaerol (4) [34] and sphaerococcenol (5) [31] as additional major metabolites of the CH2Cl2 extract of S. coronopifolius harvested on the Portuguese coast.
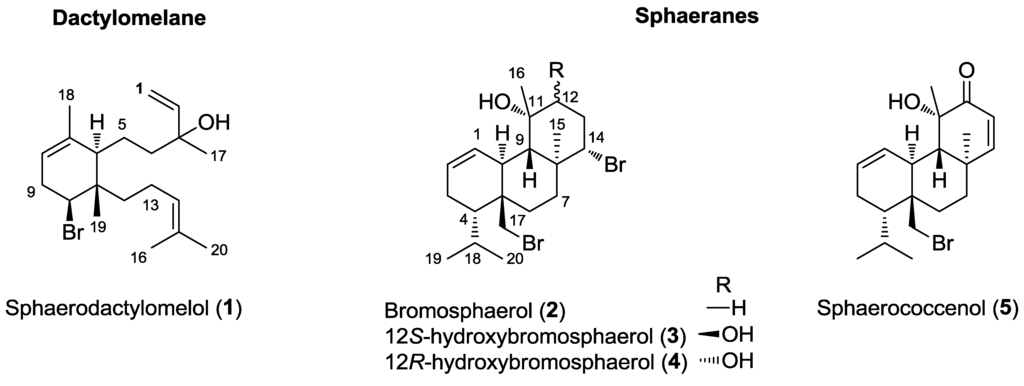
Figure 1.
Chemical structure of the five bromoditerpenes, 1–5, isolated from S. coronopifolius.
Figure 1.
Chemical structure of the five bromoditerpenes, 1–5, isolated from S. coronopifolius.
Compound 1 was isolated as an optically-active oil, and its (+)-HRESIMS spectrum (see Supplementary Information) revealed an isotopic pattern characteristic of the presence of one bromine atom at m/z 351.17117 and 353.16891 (intensities 1:1) that was consistent with the molecular formula C20H32Br+. The 1H NMR spectrum (see Supplementary Information) confirmed the presence of a diterpene with five characteristic methyl signals at δH 0.89 (s, H3-19), 1.30 (s, H3-17), 1.63 (s, H3-16), 1.69 (s, H3-20) and 1.70 (s, H3-18), the chemical shifts of the last three revealing three methyls substituted on olefinic double bonds (Table 3). A terminal monosubstituted double bond was evidenced by the vinylic signals at δH 5.92 (dd, J = 17.2 and 10.8 Hz, H-2), 5.22 (dd, J = 17.2 and 1.2 Hz, H-1a) and 5.09 (dd, J = 10.8 and 1.2 Hz, H-1b). Two additional trisubstituted double bonds were inferred from the NMR signals at δH 5.22–5.17 (m, H-8), δC 120.5 (CH, C-8) and 137.2 (qC, C-7) and at δH 5.12–5.08 (m, H-14), δC 124.3 (CH, C-14) and 131.8 (qC, C-15).

Table 3.
1H (500 MHz) and 13C (125 MHz) NMR data of sphaerodactylomelol (1).
| Atom n° | δH in ppm, mult. (J in Hz) | δC in ppm, mult. |
|---|---|---|
| 1 | 5.09, dd (10.8, 1.2) 5.22, dd (17.2, 1.2) | 112.3, CH2 |
| 2 | 5.92, dd (17.2, 10.8) | 144.9, CH |
| 3 | - | 73.6, qC |
| 4 | 1.83, td (13.1, 4.7) 1.46 m | 44.2, CH2 |
| 5 | 1.63, tdd (13.1, 4.7, 2.0) 1.38–1.29, m | 23.3, CH2 |
| 6 | 2.11–2.06, m | 45.0, CH |
| 7 | - | 137.2, qC |
| 8 | 5.22–5.17, m | 120.5, CH |
| 9 | 2.62–2.49, m | 35.3, CH2 |
| 10 | 4.31, dd (10.2, 6.2) | 61.5, CH |
| 11 | - | 41.4, qC |
| 12 | 1.57, ddd (14.4, 12.4, 5.1) 1.43 m | 38.6, CH2 |
| 13 | 2.03, dd (12.9, 5.8) 1.90–18.1, m | 21.5, CH2 |
| 14 | 5.12–5.08, m | 124.3, CH |
| 15 | - | 131.8, qC |
| 16 | 1.63, s | 17.9, CH3 |
| 17 | 1.30, s | 27.9, CH3 |
| 18 | 1.70, s | 22.4, CH3 |
| 19 | 0.89, s | 16.8, CH3 |
| 20 | 1.69, s | 25.9, CH3 |
It appeared that the terminal double bond was connected to a quaternary carbon, as evidenced by the absence of further scalar coupling from the vinylic protons. The H-1 and H-2/C-3 HMBC correlations indicated that the chemical shift of the quaternary carbon was δC 73.6 (qC, C-3), which was characteristic of an allylic alcohol (Figure 2a). The conclusion on the presence of an alcohol and not a bromine atom at this position was also given by the observation of a second heteroatom-substituted carbon at δH 4.31 (dd, J = 10.2 and 6.2 Hz, H-10), δC 61.5 (CH, C-10), with chemical shifts fully consistent with a brominated methine. Consequently, the isotopic pattern obtained by HRESIMS was the result of dehydration of the allylic and tertiary alcohol at C-3 [M − H2O + H]+, and the molecular formula of 1 was then determined to be C20H33BrO. In view of the NMR data, from the four unsaturations corresponding to this molecular formula, the last one was assigned to a cycle. As described in Figure 2a, full analysis of 1H-1H COSY and HMBC spectra (see Supplementary Information) allowed the establishment of the carbon skeleton of compound 1. More precisely, the closing of the precursor geranylgeranyl chain through the C-6/C-11 bond was ascertained by the key H3-19, H-10, H2-12/C-6 and H3-19, H-10, H-9/C-11 HMBC correlations, thus leading to the six-membered ring. The key H2-12/C-6, C-11, C-19 HMBC correlations placed the last H-12/H-13/H-14 spin coupled system at C-11, thus revealing the full planar structure of 1. We relied on the NOESY spectrum (see Supplementary Information) for the elucidation of the relative configuration of 1. H-10/H-6 nOe placed the bromine on the same side as the linear alkyl chain at C-6, while the second alkyl chain was placed on the other side, thanks to H3-18/H2-4 and H-10/H2-13 nOes (Figure 2b). The absolute configuration was not determined, due to the lack of simple assessment. However, we assume the same stereochemistry as the one determined for the known analogues, 2, 3 and 4.
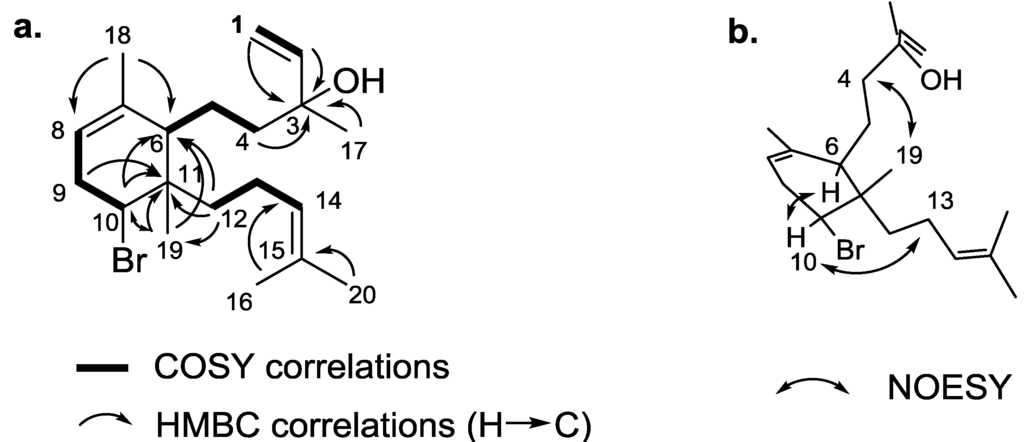
Figure 2.
Key COSY, HMBC (a) and NOESY (b) correlations for sphaerodactylomelol (1).
Figure 2.
Key COSY, HMBC (a) and NOESY (b) correlations for sphaerodactylomelol (1).
This compound does not belong to the usual sphaerane family found in this species, but it is another rare example of the dactylomelane family (Figure 1) [41]. To date, there are nine compounds of this class of diterpenes described in the literature. Eight of them are characterized by a bridged oxide between C-7 and C-10, thus leading to a 7-oxabicyclo[2.2.1]heptane moiety. These compounds were isolated from sea hares of the genus Aplysia, but also from a red alga of the genus Laurencia, thus confirming the diet of the sea hare [41,42,43]. The last compound of this chemical class was isolated from the same species, S. coronopifolius, harvested also along the Atlantic coast [33]. Its chemical structure is very similar to that of compound 1, differing by the presence of an allylic alcohol at C-14 instead of a dimethyl-substituted double bond. Interestingly, all of the compounds isolated from samples of S. coronopifolius collected in the Mediterranean and Adriatic seas belong to the sphaerane family, suggesting that the presence of dactylomelanes would be restricted to Atlantic specimens.
2.3. Antitumor and Antimicrobial Activities of Purified Compounds
The results of the anti-proliferative experiments demonstrated that all isolated compounds have anti-proliferative activity on HepG-2 cells at sub-toxic concentrations (Table 4). The highest inhibition of cell proliferation was exhibited by compound 5 and then compound 4 with IC50 of 42.9 and 105 μM, respectively. Sphaerococcenol A (5) has already demonstrated high cytotoxic activities against A549, OE21, PC-3 and LoVo cell lines, with LC50 of 3.7, 3.0, 3.7 and 2.8 µM, respectively [37]. However, this is the first report of activity against HepG-2 cells. In the same report, 12R-hydroxybromosphaerol (4) also showed cytotoxic activity against the four cell lines with LC50 of 28, 25, 26 and 26 μM, respectively. The absence of cytotoxicity was verified in the case of NSCLC-N6 (L16) and A549 cell lines [34]. On the other hand, the major reduction of HepG-2 cell viability was induced by the new compound 1 with an IC50 of 720 µM, which also showed an anti-proliferative activity (IC50 280 µM).

Table 4.
Cytotoxicity and anti-proliferative effects (IC50) induced on HepG-2 cells by bromoditerpenes 1–5 isolated from S. coronopifolius and drugs (positive controls). IC50 values are expressed as the means of eight independent experiments with 95% confidence intervals.
| IC50 (µM) | ||||
|---|---|---|---|---|
| Cytotoxicity | Anti-Proliferative | |||
| Bromoditerpenes | 1 | 719.85 (519.79–996.81) | 279.93 (206.78–378.74) | |
| 2 | >1000 | 203.33 (90.65–456.18) | ||
| 3 | >1000 | 291.42 (206.22–411.83) | ||
| 4 | >1000 | 104.83 (55.27–198.89) | ||
| 5 | >1000 | 42.87 (22.76–78.88) | ||
| Drugs (+) | Cisplatin | 454.6 (388.9–531.3) | 75.41 (61.78–92.05) | |
| Tamoxifen | >1000 | 45.68 (31.84–65.57) | ||
Regarding antimicrobial activity (Table 5), the highest growth inhibition against S. aureus was obtained for compounds 4 (IC50 6.35 µM), 2 (IC50 22.4 µM) and 1 (IC50 96.3 µM). This is the first report for an antimicrobial activity of 12R-hydroxybromosphaerol (4). For sphaerococcenol A (5), a previous report showed an antimalarial activity against the chloroquine-resistant Plasmodium falciparum FCB1 strains with an IC50 of 1 µM, but no results has been published on antimicrobial activities [33].
3. Experimental Section
3.1. General Experimental Procedures
UV and CD spectra were measured using a Jasco J-810 spectropolarimeter. Optical rotation was measured on a Perkin-Elmer 343 polarimeter, using a 100-mm microcell. IR spectra were recorded on a Bruker Tensor 27 spectrophotometer. NMR spectra were measured on a Bruker Avance 500-MHz spectrometer. Chemical shift values (δ) were reported in parts per million (ppm) relative to the appropriate internal solvent standard, and coupling constants (J-values) are given in Hertz and referenced to residual solvent signals (CDCl3 at δH 7.26 and δC 77.16). HPLC purifications were carried out on a Jasco LC-2000 system equipped with a PU-2087 Plus preparative pumping system and a UV-2075 Plus detector. HRESIMS data were conducted on an LTQ Orbitrap mass spectrometer (Thermo Fisher Scientific, Waltham, MA, USA).

Table 5.
Antimicrobial activities (IC50) against E. coli, P. aeruginosa, S. aureus and C. albicans of bromoditerpenes 1–5 isolated from S. coronopifolius and drugs (positive controls). IC50 values are expressed as the means of eight independent experiments with 95% confidence intervals.
| IC50 (µM) | |||||
|---|---|---|---|---|---|
| E. coli | P. aeruginosa | S. aureus | C. albicans | ||
| Bromoditerpenes | 1 | >100 | >100 | 96.30 (84.60–109.61) | >100 |
| 2 | >100 | >100 | 22.42 (15.44–32.57) | >100 | |
| 3 | >100 | >100 | >100 | >100 | |
| 4 | >100 | >100 | 6.35 (4.78–8.42) | >100 | |
| 5 | >100 | >100 | >100 | >100 | |
| Drugs (+) | Ampicillin | 6.42 (1.86–22.26) | - | 0.11 (0.08–0.15) | - |
| Bacitracin | >100 | - | 2.85 (2.36–3.44) | - | |
| Chloramphenicol | >100 | - | 80.49 (58.99–109.86) | - | |
| Oxytetracycline | 1.12 (0.65–1.89) | 2.13 (1.65–2.76) | 0.87 (0.59–1.32) | - | |
| Amphotericin b | - | - | - | >100 | |
| Flumequine | - | - | - | >100 | |
3.2. Sampling, Identification and Treatment of Algal Material
Sphaerococcus coronopifolius samples were collected freshly from Berlenga Nature Reserve (39°24'44.8"N 9°30'29.5"W), Peniche (Portugal), in June 2012, and immediately transported to the laboratory. The alga was then cleaned and washed with sea water to remove epiphytes, detritus and encrusting material, and the resulting algal material was freeze-dried (Scanvac Cool Safe, LaboGene, Lynge, Denmark). The dry algal material was ground and stored at −80 °C until further use.
3.3. Extraction and Fractionation of Algal Extract by Vacuum Liquid Chromatography (VLC)
Freeze-dried samples of S. coronopifolius (470 g) were sequentially extracted in a 1:4 biomass:solvent ratio with MeOH and then CH2Cl2 at constant stirring for 12 h. Liquid-liquid extraction was additionally performed for the MeOH extract, using n-hexane to remove fats. The CH2Cl2 extract (5.2 g) was further concentrated and subjected to normal phase vacuum liquid chromatography on silica gel 60 (0.06–0.2 mm), using cyclohexane with increasing amounts (25%) of ethyl acetate (EtOAc) as the mobile phase (5 fractions, each one with 400 mL of eluent). The dried fractions were stored at −20 °C until further use (F1 287 mg, F2 412 mg, F3 796 mg, F4 442 mg, F5 159 mg).
3.4. Purification of Bromoditerpenes
Fraction F2 (412 mg) was further purified by preparative reversed-phase HPLC (XSelect CSH Phenyl-Hexyl 19 mm × 250 mm, 5 μm, Waters) at a flow rate of 12 mL/min and with a mixture of eluents H2O/CH3CN: isocratic from 0 to 5 min (25:75) and then a linear gradient from 5 to 20 min (from 25:75 to 15:85). This first purification afforded 6 subfractions, F2f1–F2f6, from which only compound 2 (F2p6, tR 18.8 min, 23 mg) was pure. Fraction F2f1 (tR 12.6 min, 80 mg) was subjected to a second purification step on a semi-preparative reversed-phase column (Synergi Fusion-RP 80 A, 10 mm × 250 mm, 4 μm, Phenomenex, Torrance, CA, USA) with an isocratic mode H2O/CH3CN (27:73) and a flow rate of 4.5 mL/min to afford pure compounds 3 (F2f1p1, tR 17.5 min, 10 mg) and 5 (F2f1p2, tR 19.0 min, 26 mg). Subfraction F2f3 (tR 14.2 min, 9 mg) was purified on the same column with an isocratic mode H2O/CH3CN (20:80) at a flow rate of 5 mL/min to yield new compound 1 (F2f3p1, tR 18.0 min, 4.5 mg).
The purification of fraction F3 (796 mg) was performed with the same procedure previously used for fraction F2, starting with a preparative reversed-phase HPLC separation. The elution was performed using mixtures of H2O/CH3CN under the following conditions: isocratic step from 0 to 5 min (30:70) and then a linear gradient until 25 min (from 30:70 to 15:85). This purification afforded pure compound 4 (F3p1, tR 13.4 min, 19 mg), along with the four compounds previously purified from fraction F2.
3.5. Sphaerodactylomelol (1)
Colorless oil; [α]D20 −33.3 (c 0.15, CHCl3); UV (DAD) λmax 255 nm; ECD (c 2.68 × 10−4 M, CH2Cl2) λmax (Δε) 228 (0.95), 277 (−0.13) nm; IR (neat) σ 2968, 2927, 2855, 1457, 1437, 1377 cm−1; 1H and 13C NMR data, see Table 3; HRESIMS m/z 351.17117 and 353.16891 [M – OH]+ (50:50, calcd. for C20H32Br+, 351.16819 and 353.16614, Δ −8.5 and −7.8 ppm).
3.6. Biological Activities
3.6.1. Cytotoxicity and Anti-Proliferative Activities
The anti-proliferative activities were performed on an in vitro carcinoma model of a human hepatocellular cancer (HepG-2), acquired in the American Type Culture Collection (ATCC). HepG-2 cells were cultured in RPMI 1640 (Sigma, Saint-Louis, MO, USA) medium supplemented with 10% fetal bovine serum (FBS) (Gibco, Grand Island, NY, USA) supplemented with 100 U/mL penicillin G, 0.25 µg/mL amphotericin B and 100 µg/mL streptomycin (Sigma, USA). The cells medium was changed every 3 days, and the cells reached confluence after 5–6 days of initial seeding in plates of a 25-cm2 growth area at a concentration of 42.2 × 106 cells/ plate. For the subculture, cells were dissociated with trypsin-EDTA (Sigma, Saint-Louis, MO, USA), split into a 1:3 ratio and subcultured in Petri dishes with a 25-cm2 growth area. Cells were maintained in controlled conditions: 95% of humidified atmosphere, 5% of CO2 and 37 °C. Cells were seeded in 96-well plates, at a concentration of 4.4 × 104 cells/well, for the cytotoxicity and anti-proliferative assays.
Cytotoxicity was evaluated after the cells reached total confluence and anti-proliferative activity after 36 h of initial seeding. Cells were incubated with crude extracts, VLC fractions and purified compounds, previously sterile filtered (0.2 µm, Whatman, Little Chalfont, UK), during 24 h at 1 mg/mL. For the samples with the highest activity, dose-response assays were done (10–1000 µg/mL; 24 h). Cisplatin (Sigma, St. Loiu, MO, USA) and tamoxifen (Sigma, Shanghai, China) were used as positive controls. The effects were estimated by the colorimetric assay based on the conversion of tetrazolium dye (MTT) (Sigma, Seelze, Germany) into a blue formazan product by living mitochondria [44]. After the treatment with bromoterpenes compounds, the cells medium was removed, and cells were washed with Hank’s medium (medium composition, in mM: NaCl 137, KCl 5, MgSO4 0.8, Na2HPO4 0.33, KH2PO4 0.44, CaCl2 0.25, MgCl2 1.0, Tris HCl 0.15 and sodium butyrate 1.0, pH = 7.4). Cells were then incubated with MTT (1.2 mM), previously dissolved in Hank’s medium, during 4 h at 37 °C. The formazan products were dissolved in isopropanol (Panreac, Barcelona, Spain), contained 0.04 N HCl and were determined by the absorbance at 570 nm.
Results were expressed as IC50, defined as the concentration causing a 50% reduction or inhibition of cell viability and cell proliferation, respectively.
3.6.2. Antimicrobial Activities
Antimicrobial activities were evaluated by the capacity to inhibit E. coli (ATCC 25922), P. aeruginosa (ATCC 27853), S. aureus (ATCC 25923) and C. albicans (ATCC 10231) growth (OD 600nm). The crude extracts, VLC fractions, purified compounds and positives controls (ampicillin, bacitracin, chloramphenicol, oxytetracycline, amphotericin b and flumequine, from Sigma Aldrich, Canada) were prepared with sterile-filtered dimethylsulfoxide and stored at −20 °C. Tests were performed in 96-well plates at 37 °C for bacteria and 30 °C for fungi. Antimicrobial activity was expressed as IC50, defined as the concentration causing a 50% reduction of microorganism growth.
3.6.3. Data Analysis
IC50 were calculated from the analysis of non-linear regression using GraphPad Prism program even Y = 100/(1 + 10 (X − Log IC50)) equation.
4. Conclusions
In the present study, the chemical characterization of the major brominated diterpenes isolated from samples of S. coronopifolius collected in the Atlantic revealed five major compounds, including a new brominated dactylomelane diterpene, named sphaerodactylomelol (1), and four already known sphaerane bromoditerpenes, 2–5. These molecules exhibited interesting high antimicrobial and antitumor activities against different targets and open new therapeutic potential. To the best of our knowledge, this is the first study on the isolation, identification and bioactive screening of bromoditerpenes isolated from S. coronopifolius collected from the Atlantic. Nonetheless, future studies will deeply describe the antitumor and antimicrobial activities and the mechanisms of action of the isolated compounds.
Supplementary Files
Supplementary File 1Acknowledgments
This work was supported through funding provided by the European Commission Collaborative Project FP7 number 265896-www.bammbo.eu). One of the authors (G.C.) was partially supported by CNRS (Côte d’Azur delegation-DR 20). J.-M. Guigonis from the Plate-Forme Bernard Rossi of Nice is acknowledged for recording HRMS spectra.
Author Contributions
The authors Daniel Rodrigues and Celso Alves contributed equally to this work and did the main experiments (purification, identification, cytotoxicity, anti-proliferative, antimicrobial assays and drafted the manuscript). Joana Silva, André Horta and Susete Pinteus have been involved in the antimicrobial, cytotoxicity and antiproliferative assays. Gérald Culioli and Olivier Thomas designed and coordinated the purification and identification of compounds. Rui Pedrosa designed and coordinated the antimicrobial, cytotoxicity and anti-proliferative experiments and conceived the study.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Xiong, Z.-Q.; Wang, J.-F.; Hao, Y.-Y.; Wang, Y. Recent advances in the discovery and development of marine microbial natural products. Mar. Drugs 2013, 11, 700–717. [Google Scholar] [CrossRef] [PubMed]
- Sawadogo, W.; Schumacher, M.; Teiten, M.-H.; Cerella, C.; Dicato, M.; Diederich, M. A survey of marine natural compounds and their derivatives with anti-cancer activity reported in 2011. Molecules 2013, 18, 3641–3673. [Google Scholar] [CrossRef] [PubMed]
- Simmons, T.L.; Andrianasolo, E.; McPhail, K.; Flatt, P.; Gerwick, W.H. Marine natural products as anticancer drugs. Mol. Cancer Ther. 2005, 4, 333–342. [Google Scholar] [PubMed]
- Aneiros, A.; Garateix, A. Bioactive peptides from marine sources: pharmacological properties and isolation procedures. J. Chromatogr. B 2004, 803, 41–53. [Google Scholar] [CrossRef]
- Rocha, J.; Peixe, L.; Gomes, N.C. M.; Calado, R. Cnidarians as a source of new marine bioactive compounds—an overview of the last decade and future steps for bioprospecting. Mar. Drugs 2011, 9, 1860–1886. [Google Scholar] [CrossRef] [PubMed]
- Murray, P.M.; Moane, S.; Collins, C.; Beletskaya, T.; Thomas, O.P.; Duarte, A.W. F.; Nobre, F.S.; Owoyemi, I.O.; Pagnocca, F.C.; Sette, L.D.; et al. Sustainable production of biologically active molecules of marine based origin. N. Biotechnol. 2013, 30, 839–850. [Google Scholar] [CrossRef] [PubMed]
- Paul, V.J.; Puglisi, M.P. Chemical mediation of interactions among marine organisms. Nat. Prod. Rep. 2004, 21, 189–209. [Google Scholar] [CrossRef] [PubMed]
- Ianora, A.; Boersma, M.; Casotti, R.; Fontana, A.; Harder, J.; Hoffmann, F.; Pavia, H.; Potin, P.; Poulet, S.A.; Toth, G. New trends in marine chemical ecology. Estuar. Coast. 2006, 29, 531–551. [Google Scholar] [CrossRef]
- De Almeida, C.L.F.; de S. Falcão, H.; de M. Lima, G.R.; de A. Montenegro, C.; Lira, N.S.; de Athayde-Filho, P.F.; Rodrigues, L.C.; de Souza, M.d.F.V.; Barbosa-Filho, J.M.; Batista, L.M. Bioactivities from marine algae of the genus Gracilaria. Int. J. Mol. Sci. 2011, 12, 4550–4573. [Google Scholar] [CrossRef] [PubMed]
- Pangestuti, R.; Kim, S.-K. Neuroprotective effects of marine algae. Mar. Drugs 2011, 9, 803–818. [Google Scholar] [CrossRef] [PubMed]
- Joana Gil-Chávez, G.; Villa, J.A.; Fernando Ayala-Zavala, J.; Basilio Heredia, J.; Sepulveda, D.; Yahia, E.M.; González-Aguilar, G.A. Technologies for extraction and production of bioactive compounds to be used as nutraceuticals and food ingredients: an overview. Compr. Rev. Food Sci. Food Saf. 2013, 12, 5–23. [Google Scholar]
- Thomas, N.; Kim, S.-K. Beneficial effects of marine algal compounds in cosmeceuticals. Mar. Drugs 2013, 11, 146–164. [Google Scholar] [CrossRef] [PubMed]
- Viano, Y.; Bonhomme, D.; Ortalo-Magné, A.; Thomas, O.P.; Hattab, M.E.; Piovetti, L.; Blache, Y.; Culioli, G. Dictyotadimer A, a new dissymmetric bis-diterpene from a brown alga of the genus Dictyota. Tetrahedron Lett. 2011, 52, 1031–1035. [Google Scholar] [CrossRef]
- Lee, J.-C.; Hou, M.-F.; Huang, H.-W.; Chang, F.-R.; Yeh, C.-C.; Tang, J.-Y.; Chang, H.-W. Marine algal natural products with anti-oxidative, anti-inflammatory, and anti-cancer properties. Cancer Cell Int. 2013, 13, 55. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-X.; Himaya, S.; Kim, S.-K. Triterpenoids of marine origin as anti-cancer agents. Molecules 2013, 18, 7886–7909. [Google Scholar] [CrossRef] [PubMed]
- Cabrita, M.T.; Vale, C.; Rauter, A.P. Halogenated compounds from marine algae. Mar. Drugs 2010, 8, 2301–2317. [Google Scholar] [CrossRef] [PubMed]
- Fattorusso, E.; Magno, S.; Santacroce, C.; Sica, D.; di Blasio, B.; Pedone, C.; Impellizzeri, G.; Mangiafico, S.; Oriente, G. Bromosphaerol, a new bromine-containing diterpenoid from the red alga Sphaerococcus coronopifolius. Gazz. Chim. Ital. 1976, 106, 779–783. [Google Scholar]
- Fenical, W.; Finer, J.; Clardy, J. Sphaerococcenol A, a new rearranged bromo-diterpene from the red alga Sphaerococcus coronopifolius. Tetrahedron Lett. 1976, 731–734. [Google Scholar]
- Cafieri, F.; de Napoli, L.; Fattorusso, E.; Impellizzeri, G.; Piattelli, M.; Sciuto, S. Bromosphaerodiol, a minor bromo compound from the red alga Sphaerococcus coronopifolius. Experientia 1977, 33, 1549–1550. [Google Scholar] [CrossRef]
- Cafieri, F.; de Napoli, L.; Fattorusso, E.; Piattelli, M.; Sciuto, S. Presphaerol, a new rearranged diterpene from the red alga Sphaerococcus coronopifolius. Tetrahedron Lett. 1979, 20, 963–966. [Google Scholar] [CrossRef]
- Cafieri, F.; Fattorusso, E.; di Blasio, B.; Pedone, C. Diterpenes from the red alga Sphaerococcus coronopifolius. Structure of sphaerodiene and reassignment of structure for presphaerol. Tetrahedron Lett. 1981, 22, 4123–4126. [Google Scholar] [CrossRef]
- Cafieri, F.; Ciminiello, P.; Fattorusso, E.; Santacroce, C. 12S-hydroxybromosphaerol, a new bromoditerpene from the red alga Sphaerococcus coronopifolius. Experientia 1982, 38, 298–299. [Google Scholar] [CrossRef]
- Cafieri, F.; Ciminiello, P.; Santacroce, C.; Fattorusso, E. (1S)-1,2-Dihydro-1-hydroxybromosphaerol, a minor bromoditerpene from the red alga Sphaerococcus coronopifolius. Phytochemistry 1982, 21, 2412–2413. [Google Scholar] [CrossRef]
- Cafieri, F.; Ciminiello, P.; Santacroce, C.; Fattorusso, E. Three diterpenes from the red alga Sphaerococcus coronopifolius. Phytochemistry 1983, 22, 1824–1825. [Google Scholar] [CrossRef]
- Cafieri, F.; Fattorusso, E.; Santacroce, C. Bromocorodienol, a diterpenoid based on a novel bicyclic skeleton from the red alga Sphaerococcus coronopifolius. Tetrahedron Lett. 1984, 25, 3141–3144. [Google Scholar] [CrossRef]
- Cafieri, F.; Fattorusso, E.; Mayol, L.; Santacroce, C. Coronopifoliol, a diterpene based on an unprecedented tetracyclic skeleton from the red algae Sphaerococcus coronopifolius. J. Org. Chem. 1985, 50, 3982–3984. [Google Scholar] [CrossRef]
- Cafieri, F.; Fattorusso, E.; Mayol, L.; Santacroce, C. Structure of bromotetrasphaerol, a further irregular diterpene from the red alga Sphaerococcus coronopifolius. Tetrahedron 1986, 42, 4273–4276. [Google Scholar] [CrossRef]
- Bavoso, A.; Cafieri, F.; de Napoli, L.; di Blasio, B.; Fattorusso, E.; Pavone, V.; Santacroce, C. Isolation and structure determination of norsphaerol, a bis-nor-diterpene from the red alga Sphaerococcus coronopifolius. Gazz. Chim. Ital. 1987, 117, 87–89. [Google Scholar]
- Cafieri, F.; De Napoli, L.; Fattorusso, E.; Santacroce, C. Diterpenes from the red alga Sphaerococcus coronopifolius. Phytochemistry 1987, 26, 471–473. [Google Scholar] [CrossRef]
- Cafieri, F.; De Napoli, L.; Fattorusso, E.; Santacroce, C. Sphaeropyrane, a diterpene from the marine red alga Sphaerococcus coronopifolius. Phytochemistry 1988, 27, 621–623. [Google Scholar] [CrossRef]
- De Rosa, S.; De Stefano, S.; Scarpelli, P.; Zavodnik, N. Chemical studies of north Adriatic seaweeds. Part 3. Terpenes from the red alga Sphaerococcus coronopifolius of the north Adriatic Sea. Phytochemistry 1988, 27, 1875–1878. [Google Scholar]
- Cafieri, F.; Ciminiello, P.; Fattorusso, E.; Mangoni, A. Two novel bromoditerpenes from the red alga Sphaerococcus coronopifolius. Gazz. Chim. Ital. 1990, 120, 139–142. [Google Scholar]
- Etahiri, S.; Bultel-Ponce, V.; Caux, C.; Guyot, M. New bromoditerpenes from the red alga Sphaerococcus coronopifolius. J. Nat. Prod. 2001, 64, 1024–1027. [Google Scholar] [CrossRef] [PubMed]
- Smyrniotopoulos, V.; Quesada, A.; Vagias, C.; Moreau, D.; Roussakis, C.; Roussis, V. Cytotoxic bromoditerpenes from the red alga Sphaerococcus coronopifolius. Tetrahedron 2008, 64, 5184–5190. [Google Scholar] [CrossRef]
- Smyrniotopoulos, V.; Vagias, C.; Rahman, M.M.; Gibbons, S.; Roussis, V. Brominated diterpenes with antibacterial activity from the red alga Sphaerococcus coronopifolius. J. Nat. Prod. 2008, 71, 1386–1392. [Google Scholar] [CrossRef] [PubMed]
- Smyrniotopoulos, V.; Vagias, C.; Roussis, V. Sphaeroane and neodolabellane diterpenes from the red alga Sphaerococcus coronopifolius. Mar. Drugs 2009, 7, 184–195. [Google Scholar] [CrossRef] [PubMed]
- Smyrniotopoulos, V.; Vagias, C.; Bruyère, C.; Lamoral-Theys, D.; Kiss, R.; Roussis, V. Structure and in vitro antitumor activity evaluation of brominated diterpenes from the red alga Sphaerococcus coronopifolius. Bioorg. Med. Chem. 2010, 18, 1321–1330. [Google Scholar] [CrossRef] [PubMed]
- Smyrniotopoulos, V.; Vagias, C.; Rahman, M.M.; Gibbons, S.; Roussis, V. Ioniols I and II, tetracyclic diterpenes with antibacterial activity, from Sphaerococcus coronopifolius. Chem. Biodivers. 2010, 7, 666–676. [Google Scholar] [CrossRef] [PubMed]
- Smyrniotopoulos, V.; Vagias, C.; Rahman, M.M.; Gibbons, S.; Roussis, V. Structure and antibacterial activity of brominated diterpenes from the red alga Sphaerococcus coronopifolius. Chem. Biodivers. 2010, 7, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Piazza, V.; Roussis, V.; Garaventa, F.; Greco, G.; Smyrniotopoulos, V.; Vagias, C.; Faimali, M. Terpenes from the red alga Sphaerococcus coronopifolius inhibit the settlement of barnacles. Mar. Biotechnol. 2011, 13, 764–772. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, J.J.; Souto, M.L.; Gil, L.V.; Norte, M. Isolation of naturally occurring dactylomelane metabolites as Laurencia constituents. Tetrahedron 2005, 61, 8910–8915. [Google Scholar] [CrossRef]
- Estrada, D.M.; Ravelo, J.L.; Ruiz-Pérez, C.; Martín, J.D.; Solans, X. Dactylomelol, a new class of diterpene from the sea hare Aplysia dactylomela. Tetrahedron Lett. 1989, 30, 6219–6220. [Google Scholar] [CrossRef]
- Findlay, J.A.; Li, G. Novel terpenoids from the sea hare Aplysia punctata. Can. J. Chem. 2002, 80, 1697–1707. [Google Scholar] [CrossRef]
- Yuan, Y.V.; Walsh, N.A. Antioxidant and antiproliferative activities of extracts from a variety of edible seaweeds. Food Chem. Toxicol. 2006, 44, 1144–1150. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
