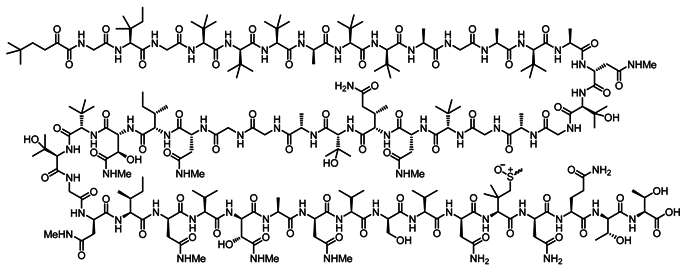
Salarin C, A Potent Inducer of Apoptosis
Yoel Kashman 1, Lee Zur 1, Ashgan Bishara 1, Maurice Aknin 2, Drorit Neumann 3 and Nathalie Ben-Califa 3
1 School of chemistry, Tel Aviv University, Ramat Aviv 69978, Israel
2 Laboratorie de Chimîe des Substances Naturelles et des Aliments, Faculté des Sciences et Techniques, Université de Réunion, 15 Avenue Rene Cassin, B.P. 7151, 97715 Saint Denis, Cedex 9, France
3 Department of Cell and Developmental Biology, Tel Aviv University, Ramat Aviv 69978, Israel
Four groups of novel nitrogen atom containing compounds have been isolated from the Madagascan sponge Fascaplysinopsis sp. The chemical content of the various samples collected in Salary bay (100 km north to Tulear) changed significantly from one site to the other suggesting the metabolites to origin from guest micro organisms. A notion supported by resemblance of several of the compounds to earlier reported micro organism metabolites. The sponge extract was tested for its effect on proliferation of the K562 leukemia cell line. Salarin C was the most effective, among the salarins tested, in inhibiting cell proliferation, as measured by the MTT assay, exhibiting inhibitory activity down to 0.005 µg/mL. Once purified, salarin C becomes very unstable and changes under light in the air to salarin A. Possessing eight functional groups complicates the chemistry of the salarins. Next to transformation of the oxazole ring to a triacetylamine, the second sensitive moiety is the vinylepoxide. The latter becomes a good site for chemical transformations and inter alia may explain the biogenesis of tausalarin C. Namely, opening of the vinylepoxide of salarin A by nucleophilic attack of pre-taumycin A.
New Polycyclic Xanthones Isolated from Marine Actinomadura sp.
Librada M Cañedo, Carmen Schleissner, Ana M Peñalver and Fernando de la Calle
Carmen Cuevas Pharmamar, Madrid 28770, Spain
Historically, terrestrial microorganisms have been a plentiful source of structurally diverse bioactive substances, and have provided important contributions to the discovery of pharmaceutically useful compounds, many of them isolated from actinomycetes [1]. Marine derived bacteria constitute a new and promising source of unique metabolites with considerable pharmaceutical and therapeutic potential [2,3], in particular, marine actinobacteria are an attractive resource for new bioactive compounds screening [4]. In the context of our interest in discovering new cytotoxic compounds from marine sources, three new polycyclic xanthones PM140035, PM140036 and PM140108, with general structure 1, have been isolated from marine bacteria belonging to the genus Actinomadura. Polycyclic xanthones are a family of polyketides which are characterized by their highly oxygenated angular hexacyclic frameworks [5]. A literature search for related compounds led to the closely related core structure of IB-00208 which was also isolated from a marine derived species of Actinomadura [6]. Citreamycins, cervinomycins and simaomicins also contain a 1,4-dioxygenated xanthone subunit, and are structurally related to 1 but belong to the family of xanthone derivatives with a cyclic amide, which is rare amongst aromatic polyketides [7]. More details about the producer microorganism, isolation and spectroscopic data leading to the structure determination of these new polycyclic xanthones and their biological activities will be reported.
References
Bauer, A.; Brönstrup, M. Industrial natural product chemistry for drug discovery and development. Nat. Prod. Rep. 2014, 31, 35–60.
Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2015, 32, 116–211.
Molinski, T.F.; Dalisay, D.S.; Lievens, S.L.; Saludes, J.P. Drug development from marine natural products. Nat. Rev. Drug Discov. 2009, 8, 69–85.
Subramani, R.; Aalbersberg, W. Marine actinomycetes: An ongoing source of novel bioactive metabolites. Microbiol. Res. 2012, 167, 571–580.
Winter, D.K.; Sloman, D.L.; Porco, J.A., Jr. Polycyclic xanthone natural products: Structure, biological activity and chemical synthesis. Nat. Prod. Rep. 2013, 30, 382–391.
Rodríguez, J.C.; Puentes, J.L.F.; Baz, J.P.; Cañedo, L.M. IB-00208, a New Cytotoxic Polycyclic Xanthone Produced by a Marine-derived Actinomadura II. Isolation, Physico-chemical Properties and Structure Determination. J. Antibiot. 2003, 56, 318–321.
Hopp, D.C.; Milanowski, D.J.; Rhea, J.; Jacobsen, D.; Rabenstein, J.; Smith, C.; Romari, K.; Clarke, M.; Francis, L.; Irigoyen, M.; et al. Citreamicins with Potent Gram-Positive Activity. J. Nat. Prod. 2008, 71, 2032–2035.
Pretrichodermamide C and N-methylpretrichodermamide B, Two New Cytotoxic Epidithiodiketopiperazines from Hyper Saline Lake Derived Penicillium sp.
Raha Orfali 1,2, Amal Ali 1, Weaam Ebrahim 1,3, Mohamed Abdel-Aziz 4, Werner Muller 5, WenHan Lin 6, Georgios Daletos 1 and Peter Proksch 1
1 Heinrich-Heine-University, Dusseldorf, Germany
2 King Saud University, Riyadh, Saudi Arabia
3 Mansoura University, Mansoura, Egypt
4 National Research Center, Cairo, Egypt
5 Medical Center of Johannes Gutenberg University, Mainz, Germany
6 Peking University, Beijing, China
The aim of this study is to isolate and structural elucidate bioactive fungal secondary metabolites from a hypersaline ecosystem. Epidithiodiketopiperazines (ETPs) are a diverse group of natural products characterized by a disulfide linkage across the dioxopiperazine ring. These metabolites possess a broad spectrum of biological activities, including antibacterial, antiviral and antifungal activities. Ascomycetes are known to accumulate numerous new and bioactive secondary metabolites including ETPs. The genus Penicillium comprises more than 300 known species and contains a highly diversified array of active compounds. Thousands of Penicillium isolates have probably already been screened in bioprospecting programmes since the discovery of penicillin, and still new biologically active secondary metabolites continue to be discovered from these fungi indicating their importance as a source for novel bioactive molecules with interest to the pharmaceutical industry. In this study, a fungal strain WN-11-1-3-1-2, identified as Penicillium sp., was isolated from the sediment of Wadi El-Natrun Lake (a hyper saline lake), Egypt, 80 km northwest of Cairo. The crude ethyl acetate extract of the fungus was subjected to different chromatographic techniques to yield two new epidithiodiketopiperazines (ETPs) derivatives, pretrichodermamide C (1) and N-methylpretrichodermamide B (2). The structures of (1) and (2) were unambiguously determined on the basis of one- and two-dimensional NMR spectroscopy and by high-resolution mass spectrometry, as well as by comparison with the literature. Compound (2) showed pronounced cytotoxicity against the murine lymphoma L5178Y cell line with an IC50 value of 2 mM. The results presented here suggest that halotolerant fungi from hypersaline environments are a rich source of bioactive secondary metabolites which could have implications for drug discovery in the future.
Antimalarial Endoperoxide Polyketides from the Chinese Marine sponge Plakortis cfr. simplex
Giuseppina Chianese 1, Marco Persico 1, Fan Yang 2, Hou-Wen Lin 2, Nicoletta Basilico 3, Silvia Parapini 4, Donatella Taramelli 4, Orazio Taglialatela-Scafati 1 and Caterina Fattorusso 1
1 Department of Pharmacy, University of Naples Federico II, Naples, Italy
2 Key Laboratory for Marine Drugs, Department of Pharmacy, Renji Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai, China
3 Dipartimento di Scienze Biomediche, Chirurgiche e Odontoiatriche, Università di Milano, Milan, Italy
4 Dipartimento di Scienze Farmacologiche e Biomolecolari Università di Milano, Milan, Italy
Marine sponges of the genus Plakortis (Demospongiae, Plakinidae) have been intensively investigated for their secondary metabolites over the last decades. The most prominent and peculiar class of Plakortis metabolites is given by propionate- and butyrate-based polyketides, exemplified by the simple 1,2-dioxane plakortin [1,2], often endowed with a promising antimalarial activity [3]. An intense research activity on Plakortis metabolites led to postulate a likely mechanism of action [4], basing on the information coming from natural and synthetically prepared analogues. Chemical investigation of the organic extract obtained from the sponge Plakortis simplex collected in the South China Sea afforded five new polyketide endoperoxides four of which containing a 1,2-dioxene ring, never evaluated before for antimalarial activity. The stereostructures of these metabolites have been deduced on the basis of spectroscopic analysis and chemical conversion. The isolated endoperoxide derivatives have been tested for their in vitro antimalarial activity against Plasmodium falciparum strains, showing IC50 values in the low micromolar range. Other apolar fractions of the same organic extract afforded novel polyketide-based metabolites, testifying the incredible chemical diversity produced by this organism. Some examples will be provided in this presentation.
References
Higgs, M.D.; Faulkner, D. John. Plakortin, an antibiotic from Plakortis halichondrioides. J. Org. Chem. 1978, 43, 3454–3457.
Fattorusso, E.; Taglialatela-Scafati, O; Di Rosa, M; Ianaro, An. Metabolites from the Sponge Plakortis simplex. Part 3: Isolation and Stereostructure of Novel Bioactive Cycloperoxides and Diol Analogues. Tetrahedron 2000, 56, 7959–7967.
Fattorusso, E.; Parapini S.; Campagnuolo, C.; Basilico, N.; Taglialatela-Scafati, O; Taramelli, D. Activity against Plasmodium falciparum of cycloperoxide compounds obtained from the sponge Plakortis simplex. J. Antimicrob. Chemother. 2002, 50, 883–888.
Taglialatela-Scafati, O.; Fattorusso, E.; Romano, A,; Scala, F.; Barone, V.; Cimino, P.; Stendardo, E.; Catalanotti,B.; Persicoa, M.; Fattorusso, C. Insight into the mechanism of action of plakortins, simple 1,2-dioxane antimalarials. Org. Biomol. Chem. 2010, 8, 846–856.
New 36-Membered Antifungal Macrolides from Streptomyces caniferus
Rodney Lacret, Daniel Oves-Costales, Caridad Díaz, Ignacio Pérez-Victoria, Jesús Martín, Mercedes de la Cruz, Nuria de Pedro, Francisca Vicente and Olga Genilloud
Fundación Medina, Armilla, Granada, Spain
Macrolides constitute one of the most interesting groups of natural products, mainly produced by actinomycetes and fungi. They exhibit various biological activities, including antitumor, antifungal, antiparasitic, citotoxic and immunosuppressant activities. Bioactive macrolides containing a 32 or 36-membered macrocyclic lactones have been described in the past and include, among others, brasilinolide, liposidolide, and novonestmycins A and B. However, macrolides linked to sugar derivatives and a 1,4-naphtoquinone are fairly uncommon. At present, they are restricted to the axenomycins and langkolide, isolated from culture broths of Streptomyces lisandri and Streptomyces sp. Acta 3062. As part of the PharmaSea project, over 400 actinomycetes were grown on carefully selected fermentation media, and their fermentation extracts were assayed against clinically relevant pathogenic microbial strains. In this work, we detected that acetone extracts from culture broths of Streptomyces caniferus (CA-271066) possesed antifungal activity against Aspergillus fumigatus and Candida albicans. Herein we report the bioassay guided isolation, structural elucidation, antifungal and citotoxic properties of a family of new macrolides (structurally related to axenomycins) isolated from culture broths of this actinomycete. The producing strain was isolated from an Ascidia collected off-shore in Sao Tome and Principe and it was grown in APM9-modified medium. The extraction of culture broths with acetone afforded an aqueous crude extract (ACE), which displayed antifungal activity. Liquid-liquid extraction of ACE with ethyl acetate followed by reversed-phase semipreparative HPLC yielded several new 36-membered antifungal macrolides as the compounds responsible for the observed bioactivity. Structural elucidation of the family of compounds was based on 1D and 2D NMR and High Resolution Mass Spectrometry (ESI-TOF). Their relative stereochemistry was determined by J-based configuration analysis combined with the presence of key NOESY correlations and data reported for similar compounds.
Novel Adociaquinone Derivatives from the Indonesian Marine Sponge Xestospongia sp.
Fei He 1, Linh H. Mai 1, Arlette Longeon 1, Brent R. Copp 2, Nadège Loaëc 3, Amandine Bescond 3, Laurent Meijer 3 and Marie-Lise Bourguet-Kondracki 1
1 Laboratoire Molécules de Communication et Adaptation des Micro-organismes, UMR 7245 CNRS/MNHN, Muséum National d’Histoire Naturelle, 57 rue Cuvier (C.P. 54), 75005 Paris, France
2 School of Chemical Sciences, University of Auckland, Private Bag 92019, Auckland 1142, New Zealand
3 ManRos Therapeutics, Perharidy Research Center, 29680 Roscoff, France
Marine sponges of the genus Xestospongia have proved to be an extremely rich source of secondary metabolites with unprecedented molecular structures and various bioactivities. Adocia-, halena- and xesto-quinone are the three main quinone-type skeletons identified from sponges of the genus Xestospongia. Among the most significant compounds, adociaquinones A and B, first isolated from the sponge Adocia sp. and then from the Philippine sponge Xestospongia sp. revealed inhibition of topoisomerase II in catalytic DNA unwinding and decatenation assays as well as inhibition of enzyme in the potassium sodium dodecyl sulfate assay. Previous investigations on the South Pacific Xestospongia sp. by our group led to the isolation of a series of halenaquinone-type compounds, including xestosaprol C methylacetal, 3-ketoadociaquinones A and B, tetrahydrohalenaquinones A and B, halenaquinol sulfate, halenaquinone and orhalquinone. Orhalquinone demonstrated significant inhibitory activities against both human and yeast farnesyltransferase enzymes, with IC50 values of 0.4 μM [1]. In the frame of the European Bluegenics program, we have chemically investigated the Indonesian sponge of Xestospongia sp. collected off North Sulawesi because its methanol crude extract had showed kinase inhibition as well as antimicrobial and antioxidant activities. Bio-guided fractionation of the extract led to the isolation of seven new adociaquinone derivatives 1a–4c, together with seven known compounds, adociaquinone A and B, secoadociaquinones A and B, 15-chloro-14-hydroxyxestoquinone, 14-chloro-15-hydroxyxestoquinone and xestoquinol sulfate. The known compounds were identified by comparison of their spectroscopic data with those of the literature. The isolation and structural elucidation of the new compounds as well as their biological activities will be presented and discussed [2].
References
Longeon, A.; Copp, B.R.; Roué, M.; Dubois, J.; Valentin, A.; Petek, S.; Debitus, C.; Bourguet-Kondracki, M.-L. New bioactive halenaquinone derivatives from South Pacific marine sponges of the genus Xestospongia. Bioorg. Med. Chem. 2010, 18, 6006–6011.
He, F.; Mai, L.H.; Longeon, A.; Copp, B.R.; Loaëc, N.; Bescond, A.; Meijer, L.; Bourguet-Kondracki, M.-L. Novel Adociaquinone Derivatives from the Indonesian Sponge Xestospongia sp. Mar. Drugs 2015, 13, 2617–2628.
Erythrins, New Toxic Metabolites from the Marine Ciliate Pseudokeronopsis erythrina Used as Chemical Defense against Predators
Andrea Anesi 1, Federico Buonanno 2, Graziano DiGiuseppe 3, Claudio Ortenzi 2 and Graziano Guella 1
1 University of Trento, Trento, Italy
2 University of Macerata, Macerata, Italy
3 University of Pisa, Pisa, Italy
Marine protozoa are known for their ability to produce a vast and chemically diverse array of secondary metabolites that are involved in different ecological functions. Morphospecies belonging to genus Euplotes have been extensively studied for their ability to produce chemically diverse secondary metabolites and, interestingly, it was found that strains belonging to same genetic clades were characterized by a different profile of bioactive compounds [1]. From the genus Pseudokeronopsis only two classes of pigments have been so far isolated, keronopsins as defensive molecules of Pseudokeronopsis rubra [2] and, more recently, keronopsamides from cell culture of the marine ciliate Pseudokeronopsis riccii [3]. We report here on the characterization of new secondary metabolites, erythrins, produced by cell cultures of Pseudokeronopsis erythrina (Ciliophora, Hypotricha). Their structure have been elucidated by extensive NMR and high resolution MS measurements and are characterized by a central 4-hydroxy-unsaturated delta-lactone ring bearing an alkyl saturated chain at C(2) and a butyl -benzenoid group at C(5). The simultaneous presence of the corresponding 4-sulphate analogues has also been ascertained and a reasonable proposal of their biosynthesis will be reported. Cold-shock treatment has been performed to induce the discharge of these metabolites from cell pigment granules. The analysis of cytotoxic activity on a panel of free-living ciliates and micro-invertebrates, together with some observation on the defensive behavior by P. erythrina, indicated that erythrins are very effective for its chemical defence.
References
Guella, G.; Skropeta, D.; di Giuseppe, G.; Dini, F. Structures, biological activities and phylogenetic relationships of terpenoids from marine ciliates of the genus Euplotes. Mar. Drugs 2010, 8, 2080–2116.
Höfle, G.; Pohlan, S.; Uhlig, G.; Kabbe, K.; Schumacher, D. Keronopsins A and B, Chemical Defence Substances of the Marine Ciliate Pseudokeronopsis rubra (Protozoa): Identification by Ex Vivo HPLC. Angew. Chem. Int. Ed. Engl. 1994, 33, 1495–1497.
Guella, G.; Frassanito, R.; Mancini, I.; Sandron, T.; Modeo, L.; Verni, F.; Dini, F.; Petroni, G. Keronopsamides, a new class of pigments from marine ciliates. Eur. J. Org. Chem. 2010, 2010, 427–434.
Cyclic Peptides from the Indonesian Marine Sponge Callyspongia aerizusa with Potent and Selective Antitubercular Activity
Georgios Daletos 1, Rainer Kalscheuer 2, Rudolf Hartmann 3 and Peter Proksch 1
1 Institute for Pharmaceutical Biology and Biotechnology, Heinrich-Heine-University, Duesseldorf, Germany
2 Institute for Medical Microbiology and Hospital Hygiene, Heinrich-Heine-University, Duesseldorf, Germany
3 Institute of Complex Systems: Strukturbiochemie, Forschungszentrum Juelich, Juelich, Germany
Chemical investigation of the Indonesian sponge Callyspongia aerizusa afforded 13 cyclic peptide derivatives, namely callyaerins. The planar structures of the isolated compounds were unambiguously elucidated on the basis of 1D and 2D NMR spectroscopic data and MS interpretation. The absolute configurations of their constituent amino acid residues were determined using Marfey’s method. The basic structural unit of the callyaerins comprises a cyclic peptide with a linear peptide side chain, both of variable size, linked through a non-proteinogenic (Z)-2,3-diaminoacrylic acid (DAA) functional group. The peptides are unusual in containing a considerable number of proline residues, of which one proline is always positioned at the beginning of the side chain, while all others are found in the ring system. All compounds were investigated in vitro against Mycobacterium tuberculosis, as well as against THP-1 (human acute monocytic leukemia), and MRC-5 (human fetal lung fibroblast) cell lines in order to assess their general cytotoxicity. Callyaerins were found to inhibit M. tuberculosis at low micromolar concentrations making these compounds interesting candidates for further studies.
Epipyrones from the Marine-Derived Fungus Epicoccum Nigrum Link Inhibit the Proteases Cathepsin K and S
Peter Hufendiek 1, Stefan Kehraus 1, Michael Gütschow 2 and Gabriele M. König 1
1 Institute of Pharmaceutical Biology, University of Bonn, Bonn, Germany
2 Institute of Pharmaceutical Institute, Pharmaceutical Chemistry I, University of Bonn, Bonn, Germany
Fungi belonging to the Ascomycota are an excellent source of bioactive compounds, among others, antibiotics, immunosuppressants and cholesterol lowering agents. This project aims to identify new fungal secondary metabolites useful as lead structures in pharmacology, especially compounds which show enzyme inhibitory activity. Since the marine environment and fungi occurring there are not yet well researched concerning the presence of pharmacologically active compounds, our project targets fungi isolated from marine algae. Here we focus on a marine-derived strain of the fungus Epicoccum nigrum, isolated from the surface of a green alga. Cultivation and fractionation of the crude extract led to the isolation of the epipyrones, which are isomeric polyketides with an unusual C-glycosyl-moiety. Biological testing revealed selective activity against two different cysteine proteases, i.e., cathepsin K and S. The IC50-values are 11.4 and 6.6 μM, respectively. Cathepsin K inhibitors may be used to combat osteoporosis, whereas cathepsin S plays a role in tumor proliferation. Interestingly, no activity was seen against cathepsin B and L. For the serine proteases human leukocyte elastase and trypsin, lower or no activity was measured, underlining the selectivity of the compounds. Since the epipyrones tend to isomerize quickly, we aimed to block isomerization by acetylation of the glycosyl-unit. Indeed, acetylation led to one single isomer, which will also be tested against the aforementioned targets, to compare the inhibitory activity. In conclusion, the epipyrones were shown to exhibit selective activity against cathepsin K and S. The role of these enzymes in osteoporosis and tumor growth renders the epipyrones interesting lead compounds for further studies.
Acknowledgments
This research is funded by the NRW International Graduate Research School BIOTECH-PHARMA.
Isocoumarins and Cyclic Hexapeptide from the Sponge-Associated Fungus Aspergillus similanensis sp. nov. Kufa 0013
Chadaporn Prompanya 1, Tida Dethoup 2, Madalena Pinto 3 and Anake Kijjoa 1
1 Instituto de Ciências Biomédicas Abel Salazar and CIIMAR, Universidade do Porto, Porto, Portugal
2 Department of Plant Pathology, Faculty of Agriculture, Kasetsart University, Bangkok, Thailand
3 Laboratório de Química Orgânica e Farmacêutica, Departamento de Ciências Químicas, Faculdade de Farmácia, Universidade do Porto, Porto, Portugal
In our ongoing search for new natural products with antibacterial and anticancer activities produced by the marine-derived fungi of the genera Neosartorya and Aspergillus, we have investigated the secondary metabolites of a Thai collection of a new species of Aspergillus, which we have named Aspergillus similanensis (KUFA0013), isolated from the marine sponge Rhabdermia sp., collected from the Similan Islands in Southern Thailand. The ethyl acetate extract of the culture of this fungus furnished several isocoumarine derivatives including new isocoumarins similanpyrones A (1), B (2) and C (3), a new chevalone analog, chevalone E (4), a new pyripyropene analog, pyripyropene T (5), and a new cyclic peptide, named similanamide (6) [1,2]. Some of the isolated compounds were evaluated for their antimicrobial activity against Gram-positive and Gram-negative bacteria and multidrug-resistant isolates from the environment.
Acknowledgments
This work was partially supported by the Project MARBIOTECH (reference NORTE-07-0124-FEDER-000047).
References
Prompanya, C.; Dethoup, T.; Bessa, L.J.; Pinto, M.M.M.; Gales, L.; Costa, P.M.; Silva, A.M.S.; Kijjoa, A. New Isocoumarin Derivatives and Meroterpenoids from the Marine Sponge-Associated Fungus Aspergillus similanensis sp. nov. KUFA 0013 Mar. Drugs 2014, 12, 5160–5173.
Prompanya, C.; Fernandes, C.; Cravo, S.; Pinto, M.M.M.; Dethoup, T.; Silva, A.M.S.; Kijjoa, A. A New Cyclic Hexapeptide and a New Isocoumarin Derivative from the Marine Sponge-Associated Fungus Aspergillus similanensis KUFA 0013. Mar. Drugs 2015, 13, 1432–1450.
Indole Alkaloids and Dihydroisocoumarin from the Alga-Associated Fungus Neosartorya takakii KUFC 7898
War War May Zin 1, Suradet Buttachon 1, Jamrearn Buaruang 2 and Anake Kijjoa 1
1 Instituto de Ciências Biomédicas Abel Salazar and CIIMAR, Universidade do Porto, Porto, Portugal
2 Division of Environmental Science, Faculty of Science, Ramkhamhaeng University, Bangkok, Thailand
In our ongoing search for new natural products with antibacterial activity produced by the marine-derived fungi of the genus Neosartorya [1,2], we have investigated the secondary metabolites of a Thai collection of Neosartorya takakii KUFC 7898, isolated from the marine alga Amphiroa sp., collected from Samaesarn Island in the Gulf of Thailand. The ethyl acetate extract of its culture furnished so far, besides the indole alkaloids aszonalenin (1a), acetylaszonalenin (1b), tryptoquivalines F (2a), H (2b), L (2c) and a new tryptoquivaline derivative which we have named tryptoquivaline U (3d), aszonapyrone A (3) and a dihydroisocoumarin derivative, 6-hydroxymellein (4). The structures of the compounds were established by 1D and 2D NMR spectral analysis and HRMS. All the compounds were evaluated for their antibacterial activity against Gram positive (Staphylococcus aureus ATCC 25923 and Bacillus subtilis ATCC 6633) and Gram negative (Escherichia coli ATCC 25922 and Pseudomonas aeruginosa ATCC 27853) bacteria as well as multidrug-resistant isolates from the environment.
Acknowledgments
This work was partially supported by the Project MARBIOTECH (reference NORTE-07-0124-FEDER-000047).
References
Eamvijarn, A.; Nelson M. Gomes, N.M.; Dethoup, T.; Buaruang, J.; Manoch, L.; Silva, A.; Pedro, M.; Marini, I.; Roussis, V.; et al. Bioactive meroditerpenes and indole alkaloids from the soil fungus Neosartorya fischeri (KUFC 6344), and the marine-derived fungi Neosartorya laciniosa (KUFC 7896) and Neosartorya tsunodae (KUFC 9213). Tetrahedron 2013, 69, 8583–8591.
Gomes, N.M.; Bessa, L.J.; Buttachon, S.; Costa, P.M.; Buaruang, J.; Dethoup, T.; Silva, A.M.S.; Kijjoa, A. Antibacterial and Antibiofilm Activities of Tryptoquivalines and Meroditerpenes Isolated from the Marine-Derived Fungi Neosartorya paulistensis, N. laciniosa, N. tsunodae, and the Soil Fungi N. fischeri and N. siamensis. Mar. Drugs 2014, 12, 822–839.
Anti-Inflammatory Activity of Tanzawaic Acid Derivatives from a Marine-derived Fungus Penicillium steckii 108YDC142
Chien Fang, Soo-Jin Heo, Hyi-Seung Lee, Yeon-Ju Lee, Jong Seok Lee and Hee Jae Shin
Korea Institute of Ocean Science and Technology, Ansan, Korea
Chemical investigation of a marine-derived fungus Penicillium steckii 108YDC142, isolated from a marine sponge sample collected at Wangdolcho, East Sea, Korea, resulted in the discovery of a new tanzawaic acid derivative (1), together with four known analogues, tanzawaic acids A (2), C (3), D (4), and K (5). Their structures were determined by the detailed analysis of NMR and MS data, along with chemical methods. These compounds significantly inhibited the nitric oxide (NO) production and the new tanzawaic acid derivative (1) inhibited the lipopolysaccharide (LPS)-induced inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2) proteins and mRNA expressions in RAW 264.7 macrophages. Additionally, compound 1 reduced the mRNA levels of inflammatory cytokines, including tumor necrosis factor-α, interleukin (IL)-1β, and IL-6. Taken together, the results of this study demonstrate that the new tanzawaic acid derivative inhibits LPS-induced inflammation.
Nocardiomycins A–C, New Cytotoxic Cyclic Depsipeptides Isolated from Marine Actinomycetes
Marta Pérez, Cristina Lillo, Rogelio Fernández, Librada Cañedo, Paz Zuñiga, Pilar Rodríguez, Simon Munt and Carmen Cuevas
Pharmamar, Colmenar Viejo/Madrid, Spain
Cyclic depsipeptides have emerged as a very important class of bioactive compounds from marine derived bacteria. Several of these compounds have been disclosed to have cytotoxic, antiviral and/or antifungal properties. Specifically, rakicidins A, B and D [1–4] showed cytotoxic activity against the murine carcinoma colon 26-L5 tumour cells, and vinylamycin [5,6], exhibited antimicrobial activities against Gram-positive bacteria including MRSA. In an effort to discover new cytotoxic compounds from marine sources, three new lipopeptides named nocardiomycin A (1), nocardiomycin B (2) and nocardiomycin C (3) have been isolated from different marine bacteria belonging to the order Actinomycetales. These compounds, structurally related to the reported rakicidins, are 15-membered depsipeptides consisting of three amino acids and a 3-hydroxyfatty acid. One of these amino acids corresponds to the rare unusual 4-amino-2,4-pentadienoate, only found in a few examples of secondary metabolites of Actinomycetes. Details of the producer microorganisms, isolation and spectroscopic data leading to the structure determination of these new cytotoxic compounds, as well as their biological properties will be presented.
References
McBrien, K.D.; Berry, R.L.; Lowe, S.E.; Neddermann, K.M.; Bursuker, I.; Huang, S.; Klohr, S.E.; Leet, J.E. Rakicidins, New Cytotoxic Lipopeptides from Micromonospora sp. Fermentation, Isolation and Characterization. J. Antibiot. 1995, 48, 1446–1452.
Igarashi, Y.; Shimasaki, R.; Miyanaga, S.; Oku, N.; Onaka, H.; Sakurai, H.; Saiki, I.; Kitani, S.; Nihira, T.; Wimonsiravude, W. et al. Rakicidin D, an inhibitor of tumor cell invasion from marine-derived Streptomyces sp. J. Antibiot. 2010, 63, 563–565.
Oku, N.; Matoba, S.; Yamazaki, Y.M.; Shimasaki, R.; Miyanaga, S.; Igarashi, Y. Complete Stereochemistry and Preliminary Structure-Activity Relationship of Rakicidin A, a Hypoxia-Selective Cytotoxin from Micromonospora sp. J. Nat. Prod. 2014, 77, 2561–2565.
Sang, F.; Li, D.; Sun, X.; Cao, X.; Wang, L.; Sun, J.; Sun, B.; Wu, L.; Yang, G.; Chu, X.; et al. Total Synthesis and Determination of the Absolute Configuration of Rakicidin A. J. Am. Chem. Soc. 2014, 136, 15787–15791.
Igarashi, M.; Shida, T.; Sasaki, Y.; Kinoshita, N.; Naganawa, H.; Hamada, M.; Takeuchi, T. Vinylamycin, a new depsipeptide antibiotic, from Streptomyces sp. J. Antibiot. 1999, 52, 873–879.
Carr, G.; Poulsen, M.; Klassen, J.L.; Hou, Y.; Wyche, T.P.; Bugni, T.S.; Currie, C.R.; Clardy, J. Microtermolides A and B from Termite-Associated Streptomyces sp. and Structural Revision of Vinylamycin. J. Org. Lett. 2012, 14, 2822–2825.
New Cyclopeptides Isolated from Lissoclinum patella
Rogelio Fernández, Elena Gómez, Marta Pérez and Carmen Cuevas
Pharmamar, Colmenar Viejo/Madrid, Spain
Ascidians from the genus Lissoclinum are a rich source of a variety of cytotoxic cyclic peptides characterized by the presence of oxazole and thiazole moieties. Some of these peptides contain threonine and serine residues whose side chains have been modified as dimethylallyl ethers. They include the cyclopeptides Nairaiamide, Patellin, Trunkamide A, Mollamide, Hexamollamide and Comoramide [1]. In the course of our screening program to isolate novel compounds with antitumor properties from marine sources, we have isolated two new cyclopeptides, named Wetamide A and B from the ascidian Lissoclinum patella collected off the coast of Wetar in the Southwest islands of Indonesia. These compounds were obtained by bioassay-guided fractionation of an organic extract of the organism, using VLC RP-18 chromatography and reverse phase semi-preparative HPLC. Structure elucidation of these new metabolites was carried out by spectroscopic methods including MS, 1H, 13C and 2D-NMR. The stereochemistry of the amino acids was determined by hydrolysis followed by derivatization with Marfey’s reagent and comparison with commercial standards by HPLC-MS [2].
Acknowledgments
Udayana University of Bali. Indonesia. Sebastiano Gulinello (Expedition Department Logistic Coordinator). Ministry of Marine Affairs and Fisheries. Republic of Indonesia.
References
Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2015, 32, 116–211, and previous papers in this series.
Marfey, P. Determination of d-amino acids. II. Use of a bifunctional reagent, 1,5-difluoro-2,4-dinitrobenzene. Carlsberg. Res. Commun. 1984, 49, 591–596.
Chemical Studies of a Sample of Hexadella sp. from the Arafura Sea (Indonesia)
Rogelio Fernández, Patricia Gema Cruz, Marta Perez and Carmen Cuevas
Pharmamar, Colmenar Viejo/Madrid, Spain
Marine sponges of the order Verongida are a rich source of brominated tyrosine metabolites, many of which have exhibited diverse biological activities [1–4]. Chemical modification occurs both on the side chain and aromatic ring of the brominated tyrosine precursors giving rise to a broad range of biosynthetically related compounds. In the course of our screening program to search for new antitumour compounds from marine organisms, we have isolated two new bromotyrosine derivatives PM140657 (1) and PM140674 (2), as well as the known pseudoceratin A (3) [5], from a sample of Hexadella sp. collected off the coast of Arafura (Indonesia). The structures of all the compounds were elucidated by analysis of their 1D and 2D-NMR spectra and comparison with data reported for other bromotyrosine derivatives. PM140674 exhibits micromolar cytotoxicity against several cell lines, including lung (A549), colon (HT29), breast (MDA-MB-231) and pancreas (PSN1).
Acknowledgments
Udayana University of Bali. Indonesia. Sebastiano Gulinello (Expedition Department Logistic Coordinator). Ministry of Marine Affairs and Fisheries. Indonesia.
References
Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2015, 32, 116–211, and previous papers in this series.
Xu, M.; Andrews, K.T.; Birrell, G.W.; Tran, T.L.; Camp, D.; Davis, R.A.; Quinn, R.J. Psammaplysin H, a new antimalarial bromotyrosine alkaloid from a marine sponge of the genus Pseudoceratina. Bioorg. Med. Chem. Lett. 2011, 21, 846–848.
Shaker, K.H.; Zinecker, H.; Ghani, M.A.; Imhoff, J.F.; Schneider, B. Bioactive metabolites from the sponge Suberea sp. Chem. Biodivers. 2010, 7, 2880–2887.
Buchanan, M.S.; Carroll, A.R.; Wessling, D.; Jobling, M.; Avery, V.M.; Davis, R.A.; Feng, Y.; Hooper, J.N.A.; Quinn, R.J. Clavatadines C–E, Guanidine Alkaloids from the Australian Sponge Suberea clavata. J. Nat. Prod. 2009, 72, 973–975.
Jang, J.-H.; van Soest, R.W.; Fusetani, N.; Matsunaga, S. Pseudoceratins A and B, antifungal bicyclic bromotyrosine-derived metabolites from the marine sponge Pseudoceratina purpurea. J. Org. Chem. 2007, 72, 1211–1217.
Bioactive Metabolites from Marine-Derived Actinobacteria from the East Mediterranean
Panagiota Georgantea 1, Leto-Aikaterini Tziveleka 1, Eleni Mavrogonatou 2, Eniko Rab 1,3, Dimitris Kekos 3, Harris Pratsinis 2, Dimitris Kletsas 2, Vassilios Roussis 1 and Efstathia Ioannou 1
1 Department of Pharmacognosy and Chemistry of Natural Products, Faculty of Pharmacy, University of Athens, Athens, Greece
2 Laboratory of Cell Proliferation and Aging, Institute of Biosciences and Applications, National Centre for Scientific Research “Demokritos”, Athens, Greece
3 Biotechnology Laboratory, School of Chemical Engineering, National Technical University of Athens, Athens, Greece
The screening of microbial natural products represents an important route to the discovery of novel anticancer and antibiotic agents. Diverse actinobacteria isolated from unique ecosystems have been shown to produce bioactive compounds which exert their influence by processes that are not compromised by existing multidrug-resistance pathways. In order to obtain new strains likely to produce novel metabolites, examination of samples from different habitats and extreme environments is necessary. The East Mediterranean basin is a geomorphologically and biologically unique marine ecosystem that has not been investigated so far for its microbiota as producers of secondary metabolites. In search of new bioactive secondary metabolites from marine microorganisms found in the Greek seas, we have selectively isolated more than 900 actinobacterial strains from sediments and macroorganisms from the Aegean and the Ionian Seas. On the basis of preliminary screening of the chemical profiles of small scale liquid cultures of numerous actinobacterial strains with LC-DAD-MS and NMR, in conjunction with evaluation of their antibacterial and cytotoxic activities, strains BI0048 and BI0383 were selected for further chemical investigation. The large scale liquid cultures of strains BI0048 and BI0383 afforded two crude extracts which were subjected to multi-step fractionations that have led so far to the isolation of 24 and 16 compounds, respectively. Among these, two polyketides are new natural products. The structure elucidation and the assignment of the relative configurations of the compounds were based on analyses of their spectroscopic data and comparison with literature data. The isolated metabolites are currently being evaluated for their antibacterial and cytotoxic activities.
Acknowledgments
This work was supported by the project aristeia-2587 “biomaract”, which is implemented under the “aristeia” action of the operational programme “education and lifelong learning” and is co-funded by the European Social Fund (ESF) and national resources.
Prevezanes and Related Diterpenes from Laurencia glandulifera and Evaluation of Their Anti-Inflammatory Activity
Maria Harizani 1, Gerasimos Konidaris 1, Maria Daskalaki 2, Sotirios Kampranis 3, Christos Tsatsanis 2, Vassilios Roussis 1 and Efstathia Ioannou 1
1 Department of Pharmacognosy and Chemistry of Natural Products, Faculty of Pharmacy, University of Athens, Athens, Greece
2 Laboratory of Clinical Chemistry, University of Crete Medical School, Heraklion, Greece
3 Mediterranean Agronomic Institute of Chania, Chania, Greece
Despite the extensive investigations on the chemical profile of numerous Laurencia species, this cosmopolitan genus still represents a prolific source of metabolites, often exhibiting antibacterial, antifungal, insecticidal and/or cytotoxic activities. Laurencia biosynthesizes a wide spectrum of secondary metabolites, including sesquiterpenes, diterpenes, triterpenes and C15 acetogenins which are frequently characterized by the presence of halogen atoms. Previous investigations on Laurencia glandulifera have led to the isolation of the brominated diterpene neorogioltriol which has exhibited very significant levels of analgesic and anti-inflammatory activity [1]. In search of new congeners, specimens of L. glandulifera were collected in Kefalonia Island in the Ionian Sea, Greece, at a depth of 1–2 m in April of 2014. Extraction of the fresh algal tissues with mixtures of CH2Cl2/MeOH afforded a residue that was subjected to a series of chromatographic separations. Eight diterpenes of the prevezane skeleton have been isolated until now. Among these, one metabolite features a new carbon skeleton. The structures and relative configurations of the isolated natural products were determined on the basis of extensive analysis of their 1D and 2D NMR and MS data. The isolated metabolites are currently being evaluated for their anti-inflammatory activity by a cell-based assay which measures the extent of macrophage activation by bacterial lipopolysaccharide (LPS).
Acknowledgments
This work was supported by the project GSRT-EPANII-11ΣYN-3-770 “NRG”, which is implemented under the “Cooperation 2011” Action of the Operational Programme “Competitiveness and Entrepreneurship” and is co-funded by the European Social Fund (ESF) and National Resources
Reference
Chatter, R.; Kladi, M.; Tarhouni, S.; Maatoug, R.; Kharrat, R.; Vagias, C.; Roussis, V. Neorogioltriol: A brominated diterpene with analgesic activity from Laurencia glandulifera. Phytochem. Lett. 2009, 2, 25–28.
New Cytotoxic Sesquiterpenes from the Brazilian Red Alga Laurencia catarinensis
Miriam Falkenberg 1,2, Efstathia Ioannou 1, Cintia Lhullier 1,2, Tauana Wanke 1,2, Ana Claudia Philippus 2, Lucas F.O. Vieira 2, Panagiota Papazafiri 3 and Vassilios Roussis 1
1 Department of Pharmacognosy and Chemistry of Natural Products, Faculty of Pharmacy, Athens, Greece
2 Laboratory of Medicinal Chemistry and Natural Products, Federal University of Santa, Florianópolis, Brazil
3 Department of Animal and Human Physiology, Faculty of Biology, School of Science, Athens, Greece
The complex Laurencia (Rhodomelaceae) includes species with a wide distribution throughout the world and represents a prolific source of new secondary metabolites. Besides fulfilling ecological needs of the seaweeds, the halogenated terpenes and acetogenins that are frequently encountered in Laurencia species, exhibit a wide range of pharmacological activities, e.g., antiinflammatory, cytotoxic, and antibacterial. Previous studies on Laurencia species have indicated that the chemodiversity observed in the species level may be affected by environmental factors. In the course of our ongoing investigations towards the isolation of bioactive marine metabolites, we have previously investigated the chemical composition of Laurencia catarinensis Cordeiro-Marino & Fujii, collected off Ilha do Arvoredo, Santa Catarina, Southern Brazil [1] and reported the isolation of seven new and seven known metabolites, most of them structurally related to caespitol. Recent collections of this species off Ilha do Xavier from the state of Santa Catarina afforded a crude extract with a different chemical profile. In order to study the magnitude of the difference in the chemical profiles, extracts obtained from collections performed from Ilha do Arvoredo and Ilha do Xavier were investigated in depth, leading to the isolation of a number of minor metabolites. Herein, we report the isolation and structure elucidation of nine new and 14 known metabolites reported for the first time from L. catarinensis. Among the two different populations, only four metabolites were found as common constituents. The isolated compounds were evaluated for their in vitro cytotoxicity against three human tumor cell lines, namely HT29, MCF7, and A431, exhibiting variable levels of activity.
Reference
Lhullier, C.; Falkenberg, M.; Ioannou, E.; Quesada, A.; Papazafiri, P.; Horta, P.A.; Schenkel, E.P.;Vagias, C.; Roussis, V. Cytotoxic halogenated metabolites from the Brazilian red alga Laurencia catarinensis. J. Nat. Prod. 2010, 73, 27–32.
Studies on the Red Sea Sponge Haliclona sp. for Its Chemical and Cytotoxic Properties
Ali El Gamal 1,2, Shaza Al-Massarani 1, Mansour Al-Said 1, Maged Abdel-Kader 3, Hazem Ghabbour 1, Hoong-Kun Fun 1, Wael Abdel-Mageed 1, Abdelkader Ashour 1, Ashok Kumar 1 and Adnan Al-Rehaily 1
1 King Saud University, Riyadh, Saudi Arabia
2 Mansoura University, El Mansoura, Egypt
3 Salman Abdulaziz University, Al-kharj, Saudi Arabia
The Red Sea is characterized by a great diversity of living organisms [1]. Previous chemical studies of marine sponges belonging to the genus Haliclona led to the isolation of a variety of bioactive secondary metabolites, including alkaloids [2], macrolides [3], polyacetylenes [4], polyketides [5] and peptides [6]. The total cytotoxic alcoholic extract of a sponge belonging to genus Haliclona, collected from the eastern coast of the Red Sea, Jeddah, Saudi Arabia, was subjected to intensive chromatographic fractionation and purification guided by cytotoxic bioassay toward various cancer cell lines. This investigation resulted in the isolation of a new indole alkaloid, 1-(1H-indol-3-yloxy) propan-2-ol (1), and the previously synthesized pyrrolidine alkaloid, (2R,3S,4R,5R) pyrrolidine-(1-hydroxyethyl)-3,4-diol hydrochloride (4), isolated here naturally for the first time. In addition, six known compounds—tetillapyrone (2), nortetillapyrone (3), 2-methyl maleimide-5-oxime (5), Maleimide-5-oxime (6), 5-(hydroxymethyl) dihydrofuran-2(3H)-one (7), and Ergosta-5,24(28)-dien-3-ol (8) were also identified. The structures of these isolated compounds were elucidated based on extensive examination of their spectroscopic data including 1D and 2D NMR. Compound 5 is reported here for the first time from the genus Haliclona, while compound 7 is not yet reported from a marine source. X-ray single-crystal structure determination was performed to determine the absolute configuration of compound 4. Unfortunately most of the isolated compounds exhibited weak cytotoxic activity against HepG-2, Daoy, and HeLa cancer cell lines.
References
Lira, N.S.; Montes, R.C.; Barbosa-Filho, J.M. Brominated compounds from marine sponges of the genus Aplysina and a compilation of their 13C NMR spectral data. Mar. Drugs 2011, 9, 2316–2368.
Rashid, M.A.; Gustafson, K.R.; Boyd, M.R. A new isoquinoline alkaloid from the marine sponge Haliclona species. J. Nat. Prod. 2001, 64, 1249–1250.
Liu, Y.-H.; Wang, B.; Liu, D.-Y.; Li, L.-D.; Fei, L.N.C. Chemistry and Biological activities of marine sponge Halichlona. JTO 2008, 27, 70–82.
Alarif, W.M.; Abdel-Lateff, A.; Al-Lihaibi, S.S.; Ayyad, S.E.N.; Badria, F.A. A New Cytotoxic Brominated Acetylenic Hydrocarbon from the Marine Sponge Haliclona sp. with a Selective Effect against Human Breast Cancer. Z. Naturforsch. C 2013, 68, 70–75.
Kennedy, J.; Codling, C.E.; Jones, B.V.; Dobson, A.D.W.; Marchesi, J.R. Diversity of microbes associated with the marine sponge, Haliclona simulans, isolated from Irish waters and identification of polyketide synthase genes from the sponge metagenome. Environ. Microbiol. 2008, 10, 1888–1902.
Rashid, M.A.; Gustafson, K.R.; Boswell, J.L.; Boyd, M.R. Haligramides A and B, Two New Cytotoxic Hexapeptides from the Marine Sponge Haliclona nigra. J. Nat. Prod. 2000, 63, 956–959.
Two New Xenicanes from the Soft Coral Clavularia sp.
Carlos Jiménez 2, Carlos Urda 1, Marta Pérez 1, Rogelio Fernández 1, Jaime Rodriguez 2 and Carmen Cuevas 1
1 PharmaMar S.A., Colmenar Viejo, Madrid, Spain
2 Universidade da Coruña, A Coruña, Spain
Soft corals belonging to the order Alcyonacea (subclass Octocorallia), are rich sources of xenicane-type compounds which are characterized by a dihydropyran ring fused to nine-membered ring. Four families with different functionalities have been identified within this structural class and they are represented by the xenicins, xenialactols, and xeniolides A and B [1–3]. In this communication, we describe the isolation of two new xenicanes, named PM090004 and PM090082, from a soft coral belonging to the genus Clavularia, collected off the coast of Okuza (Tanzania). Only one xenicane has been reported from this genus so far [4]. These new compounds were obtained by column chromatography and semipreparative HPLC purification from an organic extract of this organism. Their planar structures were determined by 1D and 2D NMR and HRESIMS techniques while their relative stereochemistries were elucidated by comparison of their chemical shifts and coupling constants with the literature values of their congeners, as well as by NOESY experiments. Both new xenicanes were moderately cytotoxic against a panel of different tumour cell lines.
Acknowkedgments
P. J. Ruysenaars (The Pemba channel fishing club Shimoni) and Ministry of Livestock and Fisheries Development. Fisheries Department (Republic of Tanzania).
References
Ishigami, S.; Goto, Y.; Inoue, N.; Kawazu, S.; Matsumoto, Y.; Imahara, Y.; Tarumi, M.; Nakai, H.; Fusetani, N.; Nakao, Y. Cristaxenicin A, an Antiprotozoal Xenicane Diterpenoid from the Deep Sea Gorgonian Acanthoprimnoa cristata. J. Org. Chem. 2012, 77, 10962–10966.
Lin, Y.-S.; Fazary, A.E.; Chen, C.-H.; Kuo, Y.-H.; Shen, Y.-C. Asterolaurins G – J, New Xenicane Diterpenoids from the Taiwanese Soft Coral Asterospicularia laurae. Helv. Chim. Acta 2011, 94, 273–281.
Andrianasolo, E.H.; Haramaty, L.; Degenhardt, K.; Mathew, R.; White, E.; Lutz, R.; Falkowski, P. Induction of apoptosis by diterpenes from the soft coral Xenia elongate. J. Nat. Prod. 2007, 70, 1551–1557.
Wang, S.; Huang, M.; Duh, C. Cytotoxic constituents from the formosan soft coral Clavularia inflata var. luzoniana. J. Nat. Prod. 2006, 69, 1411–1416.
Multiplicity Editing in Long-Range Heteronuclear Correlation NMR Experiments: Application to Natural Products
Josep Saurí 1, Eduard Sistaré 2, Michel Frederich 3, Alembert T Tchinda 4, Teodor Parella 2, R. Thomas Williamson 1 and Gary E. Martin 1
1 NMR Structure Elucidation, Process and Analytical Chemistry, Merck & Co. Inc., Rahway, NJ 07065, USA
2 Servei de Ressonància Magnètica Nuclear, Universitat Autònoma de Barcelona, E-08193 Bellaterra, Barcelona, Spain
3 Laboratory of Pharmacognosy, Department of Pharmacy, CIRM, University of Liège, B36, 4000 Liège, Belgium
4 Center for Studies on Medicinal Plants and Traditional Medicine, Institute of Medical Research and Medicinal Plants Studies (IMPM), P.O. Box 6163, Yaoundé, Cameroon
Even C/CH2 and odd CH/CH3 carbon-multiplicity information can be directly distinguished from the relative positive/negative phase of cross-peaks in a novel ME (Multiplicity-Edited)-selHSQMBC experiment. The method can be extended by a TOCSY propagation step, and is also fully compatible for the simultaneous and precise determination of long-range heteronuclear coupling constants. In addition, broadband homonuclear decoupling techniques can also be incorporated to enhance sensitivity and signal resolution by effective collapse of J(HH) multiplets. Strychnine, taxol, staurosporine, and sungucine are utilized as model compounds to demonstrate the usefulness of these techniques.
Homodecoupled 1,1- and 1,n-Adequate NMR Experiments: Application to the Structural Elucidation of Proton-Deficient Natural Products
Josep Saurí 1, Wolfgang Bermel 2, Alexei V. Buevich 1, Maged H.M. Sharaf 3, Paul L. Schiff 4, Teodor Parella 5, R. Thomas Williamson 1 and Gary E. Martin 1
1 NMR Structure Elucidation, Process and Analytical Chemistry, Merck & Co. Inc., Rahway, NJ 07065, USA
2 Bruker Biospin GmbH, Silberstreifen, 76287 Rheinstetten, Germany
3 American Herbal Products Association, Silver Spring, MD 20910, USA
4 Department of Pharmaceutical Sciences, School of Pharmacy University of Pittsburgh, Pittsburgh, PA 15261, USA
5 Servei de Ressonància Magnètica Nuclear, Universitat Autònoma de Barcelona, E-08193 Bellaterra, Barcelona, Spain
Pure shift NMR methods have recently been the subject of intense research focus. By collapsing homonuclear proton-proton couplings, resolution and experimental sensitivity both increase. Cryptospirolepine is the most structurally complex alkaloid discovered thus far from any Cryptolepis. Characterization of several degradants of the original sample a decade after the initial report called the validity of the originally proposed structure in question. We now wish to report the development of improved homodecoupled variants of 1,1- and 1,n-ADEQUATE (HD-ADEQUATE) and the utilization of these techniques in resolving long-standing structural questions associated with crytospirolepine. In addition, we evaluate the combination of NUS and homonuclear decoupling for the acquisition of both 1JCC and nJCC homonuclear coupling constants in related J-modulated ADEQUATE experiments.
Extending Long-Range Heteronuclear NMR Connectivities by Modified HSQMBC Experiments
Josep Saurí 2, Núria Marcó 1, R. Thomas Williamson 2, Gary E. Martin 2 and Teodor Parella 1
1 Servei de Ressonància Magnètica Nuclear, Universitat Autònoma de Barcelona, E-08193 Bellaterra, Barcelona, Spain
2 NMR Structure Elucidation, Process and Analytical Chemistry, Merck & Co. Inc., 126 E. Lincoln Avenue, Rahway, NJ 07065, USA
The detection of long-range heteronuclear correlations associated with J(CH) coupling values smaller than 1–2 Hz is a challenge in the structural analysis of small molecules and natural products. HSQMBC-COSY and HSQMBC-TOCSY pulse schemes are evaluated as complementary NMR methods to standard HMBC/HSQMBC experiments. The re-optimization of the interpulse delay and the incorporation of an additional J(HH) transfer step in the HSQMBC pulse scheme can favor the sensitive observation of traditionally missing or very weak correlations and, in addition, facilitates the detection of a significant number of still longer-range connectivities to both protonated and non-protonated carbons under optimal sensitivity conditions. A comparative 1H–13C study is performed using strychnine as a model compound and several examples are also provided including 1H–15N applications.
The Defensive Chemistry of the Irish Nudibranch Archidoris pseudoargus (Gastropoda Opisthobranchia)
Ryan Young 1,2 and Bill Baker 1,2
1 National University of Ireland, Galway, Co Galway, Ireland
2 University of South Florida, Tampa, FL, USA
Historically, marine natural products from the Republic of Ireland have been greatly underrepresented in the literature despite having a coastline of over 4500 miles. Archidoris pseudoargus is a soft-bodied, slow moving Dorid nudibranch which inhabits the coastal waters of Ireland and the United Kingdom. Nudibranchs are a good source of new chemical diversity, employing these secondary metabolites to deter predation. In this study we have identified new chemistry as well as used a metabolomics approach to identify the origin of said chemistry. In early Spring, mature adults come together to reproduce and shortly thereafter to oviposit on the subtidal rocky shoreline. These egg sacs can be brightly colored and exposed to predation, yet none of the many surrounding predators appear to feed on these nutrient rich egg masses. We have investigated whether the defensive chemistry of the parents is responsible for protecting the egg masses.
Structural Elucidation of Secondary Metabolites in Laminaria digitata
Anne Vissers and Jean-Paul Vincken
Harry Gruppen Lab. Food Chemistry, Wageningen University, Wageningen, Gelderland, The Netherlands
For future protein supply, Laminaria digitata is a promising source due to its relatively high protein content (15% w/w DM) and robustness in North-Sea growing conditions. In order to apply the seaweed proteins in the feed industry, extraction is needed. The first step in extraction of proteins requires cell rupture, as a result of which cell walls and membranes are broken, allows proteins and secondary defence metabolites to contact each other and interact. Phlorotannins, polymers of 1,3,5-trihydroxy benzene are important secondary metabolites exclusively found in brown seaweeds. The monomers can connect to each other via carbon-carbon and via ether connections and polymers ranging between 10 and 100 kDa are formed (Amsler 2008). Due to the large size and abundance of hydroxyl groups, the molecules can bind non-covalently to proteins via hydrogen bridges and hydrophobic interactions with the tannin rings, and form phlorotannin-protein complexes with limited solubility. An additional group of secondary metabolites is represented by the terpenoids. In order to study the phlorotannin-protein complexation potential during the process of protein extraction, a detailed investigation on phlorotannins content, degree of polymerisation and structure is needed. Chromatographic analysis of phlorotannins forms a challenge due to the structural diversity and similar polarities of these molecules. For investigation, tannins were extracted from Laminaria digitata in 80% methanol and partitioned with ethyl acetate. The ethyl acetate phase was subjected to Reversed Phase flash chromatography to separate co-extracted phlorotannins and terpenoids. Pools of phlorotannins up to a degree of polymerisation of 15 subunits, and the terpenoid pool were analysed using RP-UHPLC-MS. The fractionation by preparative chromatography prior to RP-UHPLC analysis greatly facilitated identification and quantification of the various phlorotannin and terpenoid structures.
Initial Attempts in the Purification of the Mediterranean Sponge Crambe tailliezi
Siguara B. L. Silva 1,2, Erwan Poupon 2 and Olivier P. Thomas 1
1 Nice Institute of Chemistry-PCRE, UMR 7272 CNRS, University Nice Sophia Antipolis, Parc Valrose, 28 Avenue Valrose, 06108 Nice, France
2 BioCIS, Faculté de Pharmacie, University Paris-Sud, 5 rue J-B Clément, 92296 Châtenay-Malabry, France
First described in 1982 by Vacelet and Boury-Esnault, Crambe tailliezi (Crambeidae, Poecilosclerida, Demospongiae) is an encrusting cream sponge found in the Mediterranean and the Macaronesian Sea where its abundance seems to increase along the French Riviera (between 15 and 40 m). Even if the sister species C. crambe has been widely studied before, there is no work concerning the isolation and identification of chemicals from C. tailliezi. Considering the very interesting alkaloids isolated from Crambeidae sponges, such as the crambescins and crambecidins from C. crambe, we decided to investigate the chemodiversity of C. tailliezi. The first results on the isolation and structure elucidation of the major compounds present in this species were found to be extremely complicated. However, we were able to recognize guanidine alkaloids similar to the already described batzelladines. This would be the first report of bioactive batzelladine derivatives in a Mediterranean sponge as most of them were found in Caribbean sponges like from species of the genus Batzella or Monanchora. The NMR and HRMS analyses also allowed the identification of the already described compounds crambescidin 816 and crambescidin 800. These compounds have been first isolated from C. crambe and it is worth noticing that both C. crambe and C. tailliezi share common secondary metabolites, what could raise interesting questions about the divergent evolutionary history of metabolic pathways in these two sister species.
Bioactive Compounds from Marine Bacteria Recovered from Sediments Collected at the St. Peter and St. Paul Archipelago, Brazil
Alison Batista Silva 1, Elthon G Ferreira 2, Karine Pires 2, Paula C Jimenez 3, Letícia Veras Costa-Lotufo 4, Otília Deusdência Pessoa 1, Edilberto R Silveira 1 and Maria Conceição M Torres 1
1 Departamento de Química Orgânica e Inorgânica, Universidade Federal do Ceará, Fortaleza, Ceará, Brazil
2 Instituto de Ciências do Mar, Universidade Federal do Ceará, Fortaleza, Ceará, Brazil
3 Departamento de Ciências do Mar, Universidade Federal de São Paulo, Santos, São Paulo, Brazil
4 Instituto de Ciências Biomédicas, Universidade de São Paulo, São Paulo, Brazil
The extension, localization and ecological aspects of the Brazilian coastal zone makes it a remarkable reservoir of compounds with pharmacological potential. Marine actonomycete have shown to be an attractive source of structurally diversified bioactive compounds. The main purpose of this study was to prospect antitumor compounds through a cytotoxicity-guided fractionation approach of the actinomycete strains Salinispora arenicola and Streptomyces sp. recovered from sediments collected at the Saint Peter and Saint Paul’s Archipelago (SPSPA)—Brazil. Fractionation of the ethyl acetate crude extract obtained from S. arenicola (IC50 0.55 μg/mL) resulted in two cytotoxic fractions. HR-LCMS dereplication analysis of such fractions combined with comparison in the AntiMarin database indicated the presence of staurosporine derivatives (1–4), a well known group of anticancer compounds. Chromatographic separations of the inactive fractions led to the isolation of a new rifamycin derivative, 3-(2′-oxo-propyl)-rifamycin S (5), in addition to five known metabolites, including 6-methoxy-1-methylisatin, 3-hydroxy-6-methoxy-1-methylindolin-2-one and three diketopiperazines. The structures of these compounds were elucidated through a series of 1D and 2D NMR experiments and HRMS. Bioassay-guided fractionation of the ethyl acetate extract from Streptomyces sp. (IC50 of 2.27 μg/mL) afforded highly cytotoxic antibiotics piericidin A (6) and glycopiericidin A (7), as well as three known diketopiperazines from the active fractions. The identification of these compounds was carried out by analyses HR-LCMS, followed by query to the AntiMarin data base. These results highlight the biotechnological potential of the actonomycete strains recovered from St. Peter and St. Paul Archipelago.
Secondary Metabolites and Their Biological Acitivity of the Marine-Derived Fungus Stemphylium globuliferum
Jan Schrör 1, Peter Hufendiek 1, Stefan Kehraus 1, Michael Gütschow 2 and Gabriele M. König 1
1 University of Bonn, Institute of Pharmaceutical Biology, Bonn, Germany
2 University of Bonn, Pharmaceutical Institute, Pharmaceutical Chemistry I, Bonn, Germany
Fungi are well known to be a rich source of structurally complex and biologically active compounds. The aim of the current study was to investigate metabolites of the marine-derived fungus Stemphylium globuliferum. The strain was isolated from the brown alga Petalonia zosterifolia collected in the Baltic Sea. Investigation of the extract revealed several metabolites belonging to two structural classes, i.e., dialkylresorcinols and macrolides. Thus, after separation via vacuum liquid chromatography and further purification using HPLC we gained the resorcinol-type metabolite 4-butyl-3,5-dihydroxy-benzoic acid, a new natural product. Another compound isolated was stemphol, a 2,5-dialkylresorcinol. This known metabolite showed activity against multi-resistant Staphylococcus aureus, Enterococcus faecium as well as against Candida albicans. In addition, a weak inhibitory activity toward the protease humane leucocyte elastase (HLE) was found. A third metabolite was the 14-membered lactone coriolide, which was formerly reported from butterflies. This macrolide was active against Bacillus megaterium. Furthermore, coriolide displayed a specific inhibitory activity towards HLE with an IC50 of 5 μg/mL, while its activity towards cathepsin B and L was about 10-fold weaker. The isolated metabolites from the fungus Stemphylium globuliferum showed a promising antibiotic activity against selected microorganisms as well as towards the protease HLE.
Isolation and Structural Elucidation of Two Novel Pinnatifidenyne-Derived Acetogenins from Laurencia viridis
Adrián Morales Amador 1,2, Caterina Rodríguez de Vera 1,2, Olivia Márquez Fernández 3, Antonio Hernández Daranas 1,4, José Javier Fernández 1,2, Manuel Norte 1,2 and María Luis Souto 1,2
1 University Institute of Bio-Organic Chemistry “Antonio González”, Center for Biomedical Research of the Canary Islands (CIBICAN), University of La Laguna, La Laguna 39206, Canary Islands, Spain
2 Department of Organic Chemistry, University of La Laguna, La Laguna 39206, Canary Islands, Spain
3 Laboratorio de Alta Tecnología de Xalapa LATEX—Universidad Veracruzana, Xalapa, Veracruz, Mexico
4 Department of Chemical Engineering and Pharmaceutical Technology, University of La Laguna, La Laguna, Tenerife, Spain
The red alga of the genus Laurencia (Rhodomelaceae) include many species widely distributed around the world, being the main red algal genus chemically studied in the last 50 years [1]. They are producers of a vast range of interesting halogenated secondary metabolites, including sesquiterpenes, diterpenes, triterpenes, and acetogenines. Many of these compounds are unique in terms of structural or biological diversity, often exhibiting antibacterial, antifungal, antiviral, antifeedant, antifouling, cytotoxic, antiproliferative, anti-inflammatory, ichthyotoxic and insecticidal activity [2]. As part of our continuing interest on the chemistry of the genus Laurencia, we report the isolation of two new C15 acetogenins from Laurencia viridis [3,4]. The structures were elucidated on the basis of detailed analysis of 1D and 2D NMR data and revealed that these two compounds are interesting variations on the pinnatifidenyne structure [5].
Acknowledgments
To MINECO SAF2011-28883-C03-01 and FP7-REGPOT-2012-CT2012-316137-IMBRAIM projects. C.R. de Vera to MINECO FPU program. The research group acknowledge the financing granted to ULL by Agencia Canaria de Investigación, Innovación y Sociedad de la Información, being 85% cofinanced by the European Social Fund.
References
Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinset, M.R. Marine natural products. Nat. Prod. Rep. 2015, 32, 116–211, and earlier reviews in this series.
Wang, B.G.; Gloer, J.B.; Ji, N.Y.; Zhao, J.C. Halogenated organic molecules of Rhodomelaceae origin: Chemistry and biology. Chem. Rev. 2013, 113, 3632–3685.
Gutiérrez-Cepeda, A.; Daranas, A.H.; Fernández, J.J.; Norte, M.; Souto, M.L. Stereochemical Determination of Five-Membered Cyclic Ether Acetogenins Using a Spin-Spin Coupling Constant Approach and DFT Calculations. Mar. Drugs 2014, 12, 4031–4044.
Gutiérrez-Cepeda, A.; Fernández, J.J.; Gil, L.V.; López-Rodríguez, M.; Norte, M.; Souto, M.L. Nonterpenoid C15 acetogenins from Laurencia marilzae. J. Nat. Prod. 2011, 74, 441–448.
Norte, M.; Fernández, J.J.; Cataldo, F.; González, A.G. E-Dihydrorhodophytin, AC 15 acetogenin from the red alga Laurencia pinnatifida. Phytochemistry 1989, 28, 647–649.
Cytotoxic Anomoian B and Aplyzanzine B, New Bromotyrosine Alkaloids from Two Indonesian Sponges
Jaime Rodríguez 1, Guillermo Tarazona 2, Patricia G. Cruz 2, Rogelio Fernández 2, Marta Penas 2, Carmen Cuevas 2 and Carlos Jiménez 1
1 Departamento de Química Fundamental, Facultade de Ciencias and Centro de Investigaciones Científicas Avanzadas (CICA) Universidade da Coruña, 15071 A Coruña, Spain
2 Medicinal Chemistry Department, PharmaMar S.A.U., Pol. Ind. La Mina Norte, Avda. De los Reyes, 1, 28770-Colmenar Viejo, Madrid, Spain
Marine sponges belonging to the order Verongida are devoid of spicules [1], and are difficult to characterize. As such, bromotyrosine derivatives that are characteristic of these types of sponges have been used as chemotaxonomic makers, and have proved a useful tool to facilitate taxonomy identification [2]. Examples of such compounds that have been used for taxonomic work are anomoian A [3] and aplyzanzine A [4]. In a continuation of our investigation of new marine natural products, we now report the isolation of two new bromotyrosine derivatives, anomoian B and aplyzanzine B, that may also have utility as new chemotaxonomic makers and that have been isolated from two different Verongida sponges, collected off the coast of Indonesia. The structures of anomoian B and aplyzanzine B were determined by 1D and 2D NMR experiments and confirmed by high-resolution mass spectrometry. The stereochemistry of the new molecules has been assigned by comparison with literature models [3]. Both compounds showed moderated cytotoxic activity against a panel of different cancer cell lines [5], and their mechanism of action is currently being studied.
References
Ehrlich, H.; Ilan, M.; Maldonado, M.; Muricy, G.; Bavestrello, G.; Kljajic, Z.; Carballo, J.L. Three-dimensional chitin-based scaffolds from Verongida sponges (Demospongiae: Porifera). Part I. Isolation and identification of chitin. Int. J. Biol. Macromol. 2010, 47, 132–140.
Gotsbacher, M.; Karuso, P. New Antimicrobial Bromotyrosine Analogues from the Sponge Pseudoceratina purpurea and Its Predator Tylodina corticalis. Mar. Drugs 2015, 13, 1389–1409.
Kottakota, S.K.; Evangelopoulos, D.; Alnimr, A.; Bhakta, S.; McHugh, T.D.; Gray, M.; Groundwater, P.W.; Marrs, E.C.L.; Perry, J.D.; Spilling, C.D.; et al. Synthesis and biological evaluation of purpurealidin E-derived marine sponge metabolites: aplysamine-2, aplyzanzine A, and suberedamines A and B. J. Nat. Prod. 2012, 75, 1090–1101.
Evan, T.; Rudi, A.; Ilan, M.; Kashman, Y. Aplyzanzine A, a new dibromotyrosine derivative from a Verongida sponge. J. Nat. Prod. 2001, 64, 226–227.
Carter, D.C.; Moore, R.E.; Mynderse, J.S.; Niemczura, W.P.; Todd, J.S. Structure of majusculamide C, a cyclic depsipeptide from Lyngbya majuscule. J. Org. Chem. 1984, 49, 236–241.
Antimicrobial Metabolites from South African Laurencia spp.
Jameel Fakee 1, Kim A. Durrel 2, Marilize le Roes-Hill 2, John J. Bolton 3 and Denzil R. Beukes 4
1 Rhodes University, Grahamstown, South Africa
2 Cape Peninsula University of Technology, Bellville, South Africa
3 University of Cape Town, Rondebosch, South Africa
4 University of the Western Cape, Bellville, South Africa
The rapid development of resistance against common antibiotics combined with the slow pace of new antimicrobial drug discovery and development present a significant health risk. We have therefore initiated a programme to explore the antimicrobial potential of South African marine organisms. A number of organic extracts from Laurencia spp. collected from the South African coast were screened against five biomedically relevant microorganisms, Acinetobacter baumannii, Enterococcus faecalis, Escherichia coli, Staphylococcus aureus subsp. aureus and Candida albicans. One of the more promising extracts was from the red alga, Laurencia corymbosa which was selected for further studies. Some 30 metabolites were isolated and characterized by spectroscopic methods. Two of the more active metabolites include the chamigrane (1), which showed a minimum inhibitory activity (MIC) of 1 μg/mL against E. faecalis while the cuparene (2) showed the same level of activity against A. baumannii.
In Vitro Shistossomicial Activity of the Ascidians Botrylloides giganteum, Didemnum sp. and Trididemnum orbiculatum from Brazil
Luis Claudio Kellner Filho 1, Rita Cássia Nascimento Pedroso 1, Gustavo Muniz Dias 2, Lizandra Guidi Magalhães 1, Marcio Luis Andrade e Silva 1, Wilson Roberto Cunha 1, Patricia Mendonça Pauletti 1, Victoria Helen Woolner 3, Peter Thomas Northcote 3 and Ana Helena Januario 1
1 Universidade de Franca, Franca, São Paulo, Brazil
2 Universidade Federal do ABC, São Bernando do Campo, São Paulo, Brazil
3 Victoria University of Wellington, Wellington, New Zealand
The ascidians Botrylloides giganteum, Didemnum sp. and Trididemnum orbiculatum marine species are widely distributed in the São Sebastião Channel, São Paulo, Brazil. Ascidias (Tunicata, Ascidiacea) have yielded a variety of structurally novel and pharmacologically interesting compounds however, studies related to potential schistosomicidal about these genera are non-existent. Schistosomiasis is one of the most significant neglected diseases in the world. The aim of this research was to evaluate the schistosomicidal potential of the crude methanolic extracts of B. giganteum (BG), Didemnum sp. (DS) and T. orbiculatum (TO) against adult worms of Shistosoma mansoni. The extracts were tested for viability and motor activity at the concentrations 50 and 100 μg/mL. Regarding the mobility, TO extract reduced on 100% of motor activity at 50 μg/mL after 72 h, while DS extract decreased the motor activity on 100% of parasites at 100 μg/mL after 24 h. In contrast, the BG extract was inactive. No extract elicited death of the parasites. Despite the inactivity, the proton NMR spectrum of a semi-purirfed fraction from the BO extract revealed interesting signals which prompted further investigation. Combinations of normal and reversed-phase column chromatography and HPLC-ELSD of BG extract conduced the isolation of the aliphatic sulfated compound (3Z,6Z,9Z)-3,6,9-Dodecatrien-1-yl hydrogen sulfate (1). The structural elucidation of 1 was established by NMR spectroscopic and mass spectrometric analysis TOF-MS (−): m/z 259.1009 (C12H19O4S). This compound was previously isolated as a kairomone secreted by the crustacean Daphnia magna.
Search for New Natural Products in Marine Actinomycetales: First Insights into the Secondary Metabolites of Williamsia maris
Andrea Lubich 1, Liselotte Krenn 1, Martin Zehl 1,2 and Hanspeter Kaehlig 3
1 Department of Pharmacognosy, Faculty of Life Sciences, University of Vienna, Vienna, Austria
2 Department of Pharmaceutical Chemistry, Faculty of Life Sciences, University of Vienna, Vienna, Austria
3 Institute of Organic Chemistry, University of Vienna, Vienna, Austria
The marine environment is recognized as the space with the highest biodiversity on earth and is hence the richest source for new lead structures in drug development. In this connection, microbes are supposed to produce the largest number and variety of marine secondary metabolites. Among microorganisms, bacteria of the order Actinomycetales are most promising for the elucidation of new structures as 45% of all previously discovered microbial secondary metabolites were isolated from members of this order. This fact prompted us to focus our search for new natural products on marine Actinomycetales. In our project, the scarcely examined strain Williamsia maris, which was initially isolated from the Sea of Japan, was obtained from DMSZ (Braunschweig, Germany) and has been cultivated in a GYM-medium (0.4% glucose, 0.4% yeast extract, 1% malt extract) for 14 days. By centrifugation, the cells were removed and the resin Amberlite XAD-16 was added to the fermentation broth to adsorb released metabolites. After two days of incubation, the adsorbate was eluted from the resin with acetone to gain 1.5 g of dried crude extract. This extract was fractionated by SPE on RP-18 cartridges into 18 fractions. Subsequent size-exclusion chromatography led to the isolation of three compounds. Their structures were determined by high-resolution MS and NMR spectroscopy as lumichrome, indole-3-carbaldehyde and a 3-O-methyl mannose polysaccharide. LC-MS analysis of another fraction of the extract indicates the presence of two cyclic dipeptides, with the masses and fragmentation patterns correlating with those of cyclo(Ile-Pro) and cyclo(Leu-Pro). All five substances are reported in the strain Williamsia maris for the first time.
Industrial Biotechnology and Polymers and Biomolecules
Biocare Marine: Biomolecules of the Sea for Environmental Remediation and Healthcare
Christine Delbarre-Ladrat, Laetitia Kolypczuk, Delphine Passerini, Frédérique Chevalier, Jacqueline Ratiskol, Corinne Sinquin, Agata Zykwinska, Françoise Leroi and Sylvia Colliec-Jouault
Ifremer, Nantes, France
BioCare marine was a two-year collaborative research project (2012–2014) funded under the EU, Interreg 2-Seas programme. It aimed to isolate, characterise and sustainably utilise marine biomolecules from seaweeds, fish, oysters, shrimps and bacteria. The biomolecules were evaluated to be integrated into functional products for human healthcare and environmental applications. The ocean represents a vast and relatively untapped resource where the organisms therein have evolved a myriad of mechanisms to survive in this changing and demanding environment. These include antimicrobial substances to help out-compete neighbouring organisms, polysaccharides to prevent dehydration and provide structure and defences against toxic metal poisoning. The scientific objectives of Biocare Marine were to: (i) discover and functionalise new antimicrobial compounds (ii) construct novel wound dressings and tissue regeneration scaffolds using marine biomolecules that hold great promise for the treatment of chronic wounds, and for the remodelling and reconstruction of skin in burn victims (iii) utilise the specialised polysaccharides produced by marine bacteria to construct heavy metal capture-systems using an advanced technology platform. Toxic heavy metals cause environmental and health damage and present a significant threat to human wellbeing. Our consortium consisted of the University of Brighton (UK), University of Gent (Gent, Belgium), Ifremer (a French governmental laboratory, Plouzanr, France) and Polymaris (a marine biotech company, Morlaix, France). The poster will be particularly dedicated on bacteria and molecules from Ifremer collection.
Bioremediation of Heavy Metals Using Cyanothece sp. CCY 0110 Cultures or Its Released Polysaccharides (RPS)
Rita Mota 1,2, Federico Rossi 3, Sara B. Pereira 1, Ângela Brito 1,2, Roberto de Philippis 3,4, and Paula Tamagnini 1,2
1 i3S—Instituto de Investigação e Inovação em Saúde & IBMC—Instituto de Biologia Molecular e Celular, Universidade do Porto, Porto, Portugal
2 Departamento de Biologia, Faculdade de Ciências, Universidade do Porto, Porto, Portugal
3 Department of Agrifood Production and Environmental Sciences, University of Florence, Florence, Italy
4 Institute of Ecosystem Study (ISE), National Research Council (CNR), Sesto Fiorentino, Italy
Many cyanobacteria can produce extracellular polymeric substances (EPS) that can remain associated to the cell or be released into the environment (RPS-released polysaccharides). The particular features of these polymers, namely the presence of two different uronic acids, sulphate groups and high number of different monosaccharides, makes them promising for biotechnological applications such as metal removal from polluted waters. Cyanothece sp. CCY 0110, a marine unicellular cyanobacterium, is among the most efficient RPS-producers. The polymer produced is remarkably thermostable, composed by nine different monosaccharides, and contains sulfate groups and peptides [1]. The effects of several heavy metals on the growth/survival, EPS production, ultrastructure and protein profiles of Cyanothece were also evaluated. The results showed that each metal affect the cells in a particular manner, triggering distinctive responses [2]. For optimization of the metal removal process, it is necessary to understand the interactions between cells/EPS with metal ions. Therefore, the affinity of various culture fractions for different metals in mono- and multi-metal systems was assessed. Our results clearly showed that RPS are the most efficient fraction in metal-adsorption. Moreover, an acid or basic pre-treatment of RPS increased the specific metal removal. Currently, major sites for metal binding are being identified by physicochemical analyses.
References
Mota, R.; Guimarães, R.; Büttel, Z.; Rossi, F.; Colica, G.; Silva, C.J.; Santos, C.; Gales, L.; Zille, A.; De Philippis, R.; et al. Production and characterization of extracellular carbohydrate polymer from Cyanothece sp. CCY 0110. Carbohydr. Polym. 2013, 92, 1408–1415.
Mota, R.; Pereira, S.B.; Meazzini, M.; Fernandes, R.; Santos, A.; Evans, C.A.; De Philippis, R.; Wright, P.C.; Tamagnini, P. Effects of heavy metals on Cyanothece sp. CCY 0110 growth, extracellular polymeric substances (EPS) production, ultrastructure and protein profiles. J. Proteom. 2015, 120, 75–94.
Anti-Coagulant Potential of Polysaccharide-Rich Fractions of Macroalgae and Influence of Depolymerization and Sulfation
Amandine Adrien 1,2, Nicolas Bridiau 1, Delphine Dufour 2, Stanislas Baudouin 2 and Thierry Maugard 1
1 LIENSs Laboratory, UMR 7266 CNRS-University of La Rochelle, La Rochelle, France
2 SEPROSYS, Séparations Procédés Systèmes, La Rochelle, France
Macroalgae have been used in traditional medicine for centuries and perceived as a food with great health benefits. They are an important source of polysaccharides, such as sulfated polysaccharides, which are responsible for many of their potential properties that might find relevance in nutraceutical, pharmaceutical and cosmeceutical applications. Nowadays, to treat health troubles linked to thrombo-embolic complication, most of the anticoagulant treatments are heparin-based. Despite its major anticoagulant activity, heparin can cause serious adverse events. Moreover, its low bioavailability makes such a treatment really expensive. Given the risks and high costs of these treatments, there is a compelling need for further investigation of new sources of anticoagulants. To study the anticoagulant potential of sulfated polysaccharides from macroalgae, different extracts were prepared using various extraction processes leading to enriched polysaccharides fractions. Extracts from six edible seaweeds, including brown (Laminaria digitata, Fucus vesiculosus, Himanthalia elongata, Ascophyllum nodosum), green (Ulva lactuca), and red (Chondrus crispus) macroalgae, were prepared and the biochemical composition of each extract was determined. The potential anticoagulant activity of each extract was also investigated at different scales, from the specific antithrombin-dependent pathway (anti-Xa and anti-IIa) to the intrinsic and/or common (Activated Partial Thromboplastin Time), extrinsic (Prothrombin Time) or common (Thrombin Time) anticoagulant pathways. Furthermore, the extract anticoagulant properties were compared with those of commercial anticoagulants: heparin and Lovenox®. To have a better understanding of the structure-function relations for the anticoagulant activity of sulfated polysaccharides, extracts from green macroalgae (Ulvas) were also prepared and purified according to a “green” industrial process using neither acid nor solvent. Those polysaccharides were then submitted to chemical modifications (sulfation) or physical modifications (depolymerization assisted by ultrasounds or ion exchange resin).
Identification, Expression and Characterization of Marine Polysaccharide Degrading Enzymes from Novel Bacteria Isolated from Intertidal Biotopes
Varsha Kale 1,2, Olafur Fridjonsson 1, Sesselja Omarsdottir 2, Solveig Petursdottir 1, Bryndis Bjornsdottir 1, Brynjar Ellertsson 1, Solveig Olafsdottir 1 and Gudmundur Hreggvidsson 1,2
1 Matis ohf, Reykjavik, Iceland
2 University of Iceland, Reykjavik, Iceland
Intertidal areas are often covered with algae containing a variety of complex polysaccharides as well as invertebrates harbouring “unconventional” structural polysaccharides. Organisms in these environments need to tolerate extreme conditions, e.g., wave action, radiation and fluctuating temperatures, osmolarity, and pH. Many of the adaptive strategies adopted by the organisms depend on polysaccharides of different structures and properties. Marine polysaccharides contain all the lignocellulose sugars, but also “rare sugars”, e.g., deoxy sugars, sugar acids and sugar alcohols, which are often complex, branched and highly substituted. As the polysaccharides are an abundant source of carbon, microbes from the unique intertidal biotopes are expected to be a valuable source of scientifically interesting and industrially applicable polysaccharide processing enzymes. A number of genomes from novel marine bacteria, isolated from intertidal areas, including geothermal biotopes, were sequenced. Following in-depth bioinformatic analysis, selected genes encoding various carbohydrate enzymes were expressed including enzymes capable of degrading complex marine polysaccharides, e.g., chondroitin sulfate, chitin, laminarin, fucoidan and alginate. Among those were the α-l-fucosidase Aful2 from Litorilinea aerophila MAT4131; chondroitin lyase ChoA1 and the sulfatase SulA2 from the Arthrobacter strain MAT3885. Both strains were isolated from marine environments following enrichment on chondroitin sulfate containing media. Functional analysis of the enzymes revealed that the fucosidase AfuL2 was active on natural substrates such as fucoidan from Laminaria digitata and Fucus vesiculosus. Addition of the sulfatase SulA2 improved the breakdown of the substrates. Furthermore, the release of fucose from sea cucumber derived fucosylated chondroitin sulfate, induced by AfuL2, was aided by co-incubation with SulA2 and the chondroitin lyase ChoA1. Accordingly, the marine enzymes were capable of reacting with sulfated polysaccharides in a concerted action.
Fucosterol from Padina Australis Protects Sh-Sy5y Cells against Amyloid-Induced Neurotoxicity by Enhancing the Expression of Neuroglobin
Li Zhe Wong 1, Sook Yee Gan 2 and Eng Lai Tan 2
1 School of Postgraduate Studies, International Medical University, Kuala Lumpur, Malaysia
2 Life Sciences, School of Pharmacy, International Medical University, Kuala Lumpur, Malaysia
Alzheimer’s disease (AD) is a neurodegenerative disease that leads to progressive loss of neurons which often results in deterioration of memory and cognitive function. The development of AD is highly associated with the formation of plaques within the dying cells in the brain and tangles within the neurons. Recent evidences highlighted the importance of neuroglobin (NGB) in ameliorating AD by protecting the neuronal cells. However, the underlying protective mechanisms remain to be elucidated. This study assessed the ability of fucosterol, a compound isolated from Padina australis, in inducing the expression of NGB gene in neuroblastoma cell line. SH-SY5Y cells were exposed to different concentrations of fucosterol in the presence of beta-amyloid. The effect on apoptosis was determined using Annexin V FITC staining and the expression of NGB was determined using real-time PCR based on SYBR Green. Flow cytometry confirmed the protective effects on SH-SY5Y cells were significant when 5 μg/mL of fucosterol (7.47% double positive for Annexin V and propidium iodide as compared to 9.88% in negative control). Furthermore, results from both real-time PCR and western blot showed that fucosterol significantly induced the expression of NGB in SH-SY5Y with 4 fold increase of NGB expression as compared to the negative control. Finally, we showed that inhibition of NGB through aldrithiol significantly decreased the viability of SH-SY5Y with 41.51% of cell populations under the late apoptosis. This study has thus provided evidences that fucosterol was able to protect SH-SY5Y by enhancing the expression of NGB.
Protein Isolation and Comparison from Various Green Sources
Emma Teuling 1, Peter Wierenga 1, Johan Schrama 2 and Harry Gruppen 1
1 Laboratory of Food Chemistry, Wageningen University, Wageningen, The Netherlands
2 Aquaculture and Fisheries Group, Wageningen University, Wageningen, The Netherlands
Green sources like algae and leaves are gaining interest as alternative protein sources for food and feed products. These green sources are biologically diverse, ranging from microcellular marine organisms to terrestrial multicellular organisms. The aim of this project is to get more insight into the similarities and differences between green sources and how these differences affect protein isolation and the techno-functionalities of the protein isolates obtained. Protein sources were selected from various unicellular photosynthetic phyla: Arthrospira sp. (cyanobacteria), Nannochloropsis gaditana (heterokontophyta) and Tetraselmis sp. (chlorophyta). The protein contents of these materials were found to be 37%, 42% and 57% for respectively Tetraselmis, Nannochloropsis and Arthrospira. The total carbohydrate content was similar for all sources, ranging from 8% to 11%. Total lipid contents ranged from 15% to 24%. Since all three organisms were protein-rich but contained different types of proteins and carbohydrates, they are good model organisms for comparing protein isolation from unicellular, green sources. A mild protein isolation process was developed, allowing application of the obtained protein isolates in food and feed. After cell disruption 33%, 61% and 80% of the total biomass protein was extracted as soluble protein from Tetraselmis, Nannochloropsis and Arthrospira, respectively. After further purification, the total yield (5%–9%) and purity (66%–74%) of the protein isolates were similar. The microalgal isolates obtained were colourless, while the cyanobacterial protein isolate had an intense blue colour. To conclude, the developed isolation process can be applied to obtain protein isolates from various sources with similar purities and yields. The effects of the differences in protein composition and gross composition of the isolates on the techno-functional properties in food will be assessed by screening their solubility, emulsifying, gelling and foaming properties. These results will provide a better understanding concerning the differences and similarities of proteins from various green sources.
Screening of New Anti-Microbials from Marine Bacteria
Laetitia Kolypczuk 1, Christine Delbarre-Ladrat 1, Michel Dion 2, Delphine Passerini 1, Frédérique Chevalier 1 and Françoise Leroi 1
1 IFREMER—BRM/EM3B, Nantes, France
2 University—UFIP, Nantes, France
Marine environment is a rich and relatively little studied source of new functional compounds as antimicrobials. Marine bacteria have developed a range of molecules to survive and colonize this very competitive and challenging environment. We have an important and original collection of more than 4000 marine bacteria strains isolated from deep-sea hydrothermal vents, from seafood products, from microalgae culture and from an hypersaline lake of Kenya. The aim of our study is to screen our collection to discover innovative anti-microbial activities to fight the growing threat of broad spectrum antibiotic resistant infections in human and animals health. All of our bacteria are screened against 15 selected targets bacteria, a range of Gram-positive and Gram-negative bacteria, pathogens for human, for aquaculture organisms or involved in food spoilage. Our first results are promising.
Collagen from Marine Sponges: Comparative Studies on Their Morphological and Biochemical Characterization
Leto-Aikaterini Tziveleka, Evangelia Foufa, Efstathia Ioannou and Vassilios Roussis
Department of Pharmacognosy and Chemistry of Natural Products, Faculty of Pharmacy, University of Athens, Athens, Greece
Marine organisms have been proven an inexhaustible source of potent bioactive compounds. Between the diverse and multiple pharmacological properties of marine metabolites, bioactive natural macromolecules have gained interest, since such biomacromolecules possess a wide range of biomedical applications. Collagen is a ubiquitous high molecular weight fibrous protein found in all multicellular organisms. Collagen polypeptide chains are organized in three α-helices wrapping around one another forming its characteristic triple helix tertiary structure. This natural biomaterial has a wide range of applications in the health-related fields. Three species of marine demosponges, Axinella cannabina, Suberites carnosus and Plakortis simplex, all collected from the Aegean Sea, were comparatively studied for the determination of their total collagen, intercellular collagen and spongin contents. The isolated collagens were morphologically, physicochemically and biochemically characterized. The yield of total collagen, intercellular collagen and spongin was respectively, 12.6%, 0.1%, and 4.2% dry weight for A. cannabina and 5.0%, 0.4%, and 10.4% dry weight for S. carnosus, while for P. simplex only 0.8% dry weight of spongin was isolated. Light microscopy observations showed fibrous structures, while scanning electron microscopy confirmed the presence of collagen in the isolated samples. The measured IR spectra were characteristic for proteins of the collagen class. The acid-base properties of the material were investigated by titration, which also showed that sponge collagen was insoluble in dilute acid media, while dispersion of collagen was facilitated in dilute basic media. Since collagen from cattle entails the risks of allergic reactions and possible connective tissue disorders, such as arthritis and lupus, and more seriously, of bovine spongiform encephalopathy and transmissible spongiform encephalopathy, a safer source can be marine sponges. Our results suggest that, if efficient aquaculture can be established, A. cannabina and S. carnosus should be considered as alternative sources of collagen.
Comparison of Microwave- and Freeze-Drying of Sargassum muticum for Fucoxanthin Extraction
Elena M. Balboa 1,2, Noelia Flórez 1,2, Enma Conde 1,2, Andrés Moure 1,2 and Herminia Domínguez 1,2
1 Department of Chemical Engineering, Faculty of Sciences, University of Vigo-Campus Ourense As Lagoas s/n, 32004 Ourense, Spain
2 CITI-Universidade de Vigo, Parque Tecnolóxico de Galicia, Rúa Galicia n° 2, 32900 Ourense, Spain
Sargassum muticum (Yendo) Fensholt is an invasive brown macroalga in European waters. In order to control its expansion and because its eradication is not feasible, huge amounts of algal biomass are collected from coastal waters with any valuable destination but the direct application to agricultural lands. S. muticum (Sm) biomass is available and susceptible to be valorized to obtain new and active fractions and compounds for cosmetic, pharmaceutical and food industries. As a brown macroalgae, Sm is a potential source for carotenoids extraction, as fucoxathin. Several activities for this pigment have been reported: antidiabetic, cholesterol-metabolism control, anti-obesity, antioxidant, cancer cells anti-proliferation, and anti-inflamatory [1]. Fucoxanthin-containing extracts have been obtained by absolute ethanol extraction of freeze-dried Sm as previously reported [2]. Recent data showed an increased availability of the components of Sm algal material by microwave extraction, caused by cell wall structure destruction [3]. Therefore, microwave extraction was proposed as a preparative process before fucoxanthin extraction and as alternative to freeze-drying. Sm was collected (July 2012, Praia Mourisca, Pontevedra, Spain) washed and stored until use. A multimode microwave extractor (NEOS-GR, MilestoneSrl, Italy) was used for algal drying by using different two-steps combinations of time-power profiles (5–10 min/200–800 W). Microwave-dried Sm samples were subjected to ethanol extraction and resulting extracts were compared to those obtained from freeze-dried Sm [2]. Differences on solubles and fucoxanthin extraction yields were determined gravimetrically and by HPLC using a fucoxanthin standard, respectively. Total phenolic content was measured g gallic acid equivalents/g extract [4], and radical scavenging capacity of extracts was expressed as g Trolox/g [5] extract and as IC50, DPPH (g/L) [6]. Results are discussed considering the moisture of initial Sm and the preferred use of cheaper and efficient technologies and solvent reduction.
References
Kanda, H.; Kamo, Y.; Machmudah, S.; Wahyudiono, E.Y.; Goto, M. Extraction of fucoxanthin from raw macroalgae excluding drying and cell wall disruption by liquefied dimethyl ether. Mar. Drugs 2014, 12, 2383–2396.
Conde, E.; Moure, A.; Dominguea, H. Supercritical CO2 extraction of fatty acids, phenolics and fucoxanthin from freeze-dried Sargassum muticum. J. Appl. Phycol. 2015, 27, 957–963.
Perez, L.; Conde, E.; Dominguea, H. Microwave hydrodiffusion and gravity processing of Sargassum muticum. Process Biochem. 2014, 49, 981–988.
Singleton, V.L.; Rossi, J.A., Jr. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Viticult. 1965, 16, 144–158.
Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237.
Von Gadpw, A.; Joubert, E.; Hansmann, C.F. Comparison of the antioxidant activity of rooibos tea (Aspalathus linearis) with green, oolong and black tea. Food Chem. 1997, 60, 73–77.
Different Methods Fucoidan Extraction from Sargassum muticum
Noelia Flórez 1,2, María Parada 1,2, Elena M. Balboa 1,2, M. J. González-Muñoz 1,2 and Herminia Domínguez 1,2
1 University of Vigo, Ourense, Spain
2 Citi-university of Vigo, Ourense, Spain
Sargassum muticum (Sm) is a marine brown seaweed that contains alginate, laminaran and sulfated polysaccharides known as fucoidans. Fucoidans are found in the cell walls but the amount varies seasonally and depends primarily on the algae species. Studies have indicated that fucoidans are increasing pharmaceutical interest due to their versatile bioactivity (1). Anticoagulant, antitumor, antiviral, antiinflamatory activities have been studied (2). Crude fucoidans can be extracted from brown algae with different technologies; hydrothermal processing (3) and enzymatic hydrolysis (4) are suitable for the fractionation of vegetal biomass. During these processes, simultaneous extraction and depolymerisation of the fucoidan take place. In this study, two aqueous extraction methods have been proposed and compared for the solubilization and extraction of polysaccharide components and fractions with antioxidant properties from the invasive brown alga Sargassum muticum. Liquors obtained from autohydrolysis and enzymatic methods were analyzed and compared. S. muticum was collected in Praia da Mourisca (Pontevedra, Spain) in June 2012, washed with water and stored at −18 °C before use. Sm was mixed with water at a liquid:solid ratio of 30:1 (w/w, dry basis) and heated in a stainless steel reactor up to 170 °C (Parr Instr. Co., Moline, IL, USA), the reactor was quickly cooled and the liquid and solid phases were separated by filtration. The residual alginate in liquid phase was precipitated by adding 1% CaCl2 (3). Enzymatic hydrolysis was performed with two different enzymes under different conditions. Liquors from both processes were stored at −18 °C until analysis. The extraction yield was gravimetrically measured and the glucose, xylose, galactose, arabinose and fucose content was determined by HPLC and sulfate content by gelatin-BaCl2 method.
Purification with Resins of the Phlorotannins in the Extracts Obtained by Microwave Hydrodiffusion and Gravity (Mhg) of Sargassum muticum
María Parada 1,2, Noelia Flórez 1,2, Elena M. Balboa 1,2 and Herminia Domínguez 1,2
1 University of Vigo, Ourense, Spain
2 Citi-University of Vigo, Ourense, Spain
Sargassum muticum (Sm) is an invasive brown macroalgae in the Atlantic coasts. Different aqueous based extraction processes have been tried to obtain soluble fractions from this algae, including pressurized hot water extraction, enzyme assisted aqueous extraction and microwave hydrodiffusion and gravity (MHG). MHG was selected on the basis of its simplicity and the possibility of using water as solvent or to perform a solvent free process. In this study was to recover phenolic compounds with antioxidant activity and a variety of biological actions from Sm, in a MHG equipment and the selective recovery and concentration of the phenolic compounds from the liquid phase separated has been proposed. Sm was collected in Cape Estai (Pontevedra, Spain) in June 2012, was washed with tap water, freezed and stored in closed plastic bags at −20 °C until use. Operation in microwave NEOS-GR extractor (Milestone S.r.l., Italy) was carried out. Extraction was performed in the presence of light in an open system at maximum temperature of 100 °C. Under these conditions, the phenolic compounds could be stable, and it is also expected that phloroglucinol derivatives would not be affected. Combinations of irradiation power and time during operation, phenolic compounds and radical scavengers were studied. Subsequent purification in commercial food grade non ionic polymeric resins was accomplished. Adsorption onto non-ionic food grade polymeric resins and further desorption with 68% aqueous ethanol solutions were performed operating under previously selected operational conditions. Several commercial food grade resins were screened with the aim of selecting the most suited for the practical recovery of phenolic compounds with radical scavenging activity. Under the optimized adsorption-desorption conditions a powdered yellowish product with 30% phologlucinol content, expressed as g PGE/100 g extract, was obtained and the radical scavenging capacity of one gram of product was equivalent to 1 g of Trolox.
Multi Component Extraction from Scottish Macroalgae
Kirsty Black 1,2
1 Marine Biopolymers Limited, Ayrshire, UK
2 Strathclyde University, Glasgow, UK
Marine Biopolymers Limited (MBL) is an SME founded in late 2009 specialising in the extraction of components from indigenous Scottish seaweeds. MBL has developed a disruptive process for the extraction of alginate, typically the largest component in brown seaweeds, reducing manufacturing time from 24 h or more in a traditional process to less than 12 h. Although its initial main focus is alginate, MBL intends to also bring a range of other, high value, seaweed components to the market, including polysaccharides such as Laminarin, and Fucoidan. The company is also involved in the EU FP7 project, SeaBioTech, and is researching a range of microbial organisms and their metabolites as well as other algae derived compounds with the aim of also commercialising these into high value products.
Preparation of Semisynthetic Derivatives of Bromosphaerol and Evalution of Their Antifouling Activity
Kyriakos Proussis 1,2, Efstathia Ioannou 1, Theodora Calogeropoulou 2, Silvia Morgana 3, Veronica Piazza 3, Marco Faimali 3, Maria Protopapa 4, Skarlatos Dedos 4, George Verriopoulos 4 and Vassilios Roussis 1
1 Department of Pharmacognosy and Chemistry of Natural Products, Faculty of Pharmacy, University of Athens, Athens, Greece
2 Institute of Biology, Medicinal Chemistry and Biotechnology, National Hellenic Research Foundation, Athens, Greece
3 Institute of Marine Sciences (ISMAR), CNR, Genoa, Italy
4 Department of Zoology-Marine Biology, Faculty of Biology, University of Athens, Athens, Greece
Biofouling represents one of the most serious problems in modern maritime industries and its control is one of the biggest challenges of marine biotechnology. In search of ecologically friendly, biodegradable antifouling agents, a number of marine natural products were evaluated for their inhibitory activity against the settlement of cyprids of Balanus amphitrite. Among them, the brominated diterpene bromosphaerol (1), isolated as the main constituent of the red alga Sphaerococcus coronopifolius, exhibited prominent inhibitory activity coupled with negligible toxicity against the barnacle nauplii [1]. To gain an insight on the structural features responsible for the antifouling properties of bromosphaerol and to further improve its selectivity index and effective incorporation in marine paints, a number of structural analogues (2–12) were designed and synthesized. The new derivatives are currently being evaluated for their inhibitory activity against the settlement of barnacle cyprids and for their toxicity against barnacle nauplii.
Acknowledgments
This work was supported by the project GSRT-EPANII-11ΣYN-5-1274 “MARIPAINTS”, which is implemented under the “COOPERATION 2011” Action of the Operational Programme “Competitiveness and Entrepreneurship” and is co-funded by the European Social Fund (ESF) and National Resources.
References
Piazza, V.; Roussis, V.; Garaventa, F.; Greco, G.; Smyrniotopoulos, V.; Vagias, C.; Faimali, M. Terpenes from the red alga Sphaerococcus coronopifolius inhibit the settlement of barnacles. Mar. Biotechnol. 2011, 13, 764–772.