1. Introduction
Reactive oxygen species (ROS) naturally occur as byproducts of aerobic metabolism. In microalgae under non-stress conditions, the production and scavenging of ROS remain in equilibrium [
1]. However, several environmental stress factors, such as pollution, drought, high temperature, excessive light intensity, and nutritional limitation may increase the production of ROS, thus inducing oxidative stress. The formation of these unstable, yet very reactive radicals, can trigger human diseases—e.g., cancer and cardiovascular diseases—owing to the damage caused in proteins, DNA and lipids [
1,
2].
Photosynthetic organisms, like microalgae, are able to counteract the aforementioned negative effects via a number of enzymatic and non-enzymatic mechanisms [
1]. Lipidic components as carotenoids and polyunsaturated fatty acids (PUFA) are two examples of non-enzymatic classes of molecules able to protect the organism from oxidative damage [
2,
3]. A particular interest has been received by these two families of compounds due their great potential in industrial formulation of nutra- and pharmaceutical products [
4]. PUFA, found in microalgae as components of polar and neutral lipids, include linoleic (18:2), α-linolenic (18:3), arachidonic (20:4), eicosapentaenoic (20:5) and docosahexaenoic (22:6), among others; they are valuable for humans due to their physiological roles in cells—as precursors and primary preventers of health conditions, e.g., as anti-inflammatory or neuroprotective agents [
5,
6,
7]. Besides being excellent singlet oxygen scavengers suitable for use as food colorants, carotenoids may be employed as dietary supplements in cosmetics and nutraceuticals [
8]. In particular, lutein has proven to alleviate cardiovascular diseases, some types of cancer and degenerative human diseases [
9]. Hence, combined extraction of these lipidic compounds appears crucial in attempts to maximize their extra added value in nutra- and pharmaceutical formulations.
The mode of recovery of functional ingredients from natural matrices should be carefully addressed. There is indeed a need to combine appropriate, selective, cost-effective, and environment-friendly extraction procedures with legal requirements regarding use of food-grade solvents and processes. Extraction costs of microalgal intracellular metabolites are normally high; the downstream separation stages may account for 50%–80% of the total production costs [
10]. Despite the worldwide increasing interest in lipidic components of microalgae, there is no optimum standardized method for their extraction. It has been established that efficient extraction of lipids is strongly dependent on the polarity of the organic solvent or solvent mixture employed [
11]; however, other issues such as location of compound inside the cell have to be addressed, depending on cell structures complexity.
Based on their physicochemical characteristics, microalgal lipids can be divided into two major types: polar lipids, e.g., phospholipids and glycolipids; and neutral/non-polar lipids, e.g., mono-, di- and tri-acylglycerols (TAG) and carotenoids [
11,
12]. Polar lipids are important structural components of cell membranes and organelles, where they apparently operate as signal molecules (or precursors thereof). Among non-polar lipids, TAG are the most widespread group of compounds aimed at storage—and are accumulated as cytoplasmic oil bodies [
7].
Carotenoids are hydrophobic molecules that, depending on their role, can be divided in two categories—primary and secondary ones. Primary carotenoids—including β-carotene and such xanthophylls as lutein, neoxanthin, violaxanthin, antheraxanthin, and zeaxanthin (in
Chlorophyta), are contained within the non-polar “pouches” of the thylakoid membrane, and are pigment-protein complexes of photosynthetic apparatus so they essentially do not interact with the hydrophilic environment [
13]. Secondary carotenoids, like astaxanthin, are often esterified by fatty acids and accumulated in ester form—being accumulated in oil bodies and plastoglobuli [
13].
Neutral lipids are extracted with relatively non-polar solvents, such as hexane, whereas membrane-associated lipids are more polar, thus demanding such polar solvents as ethanol or methanol to disrupt hydrogen bonds and electrostatic forces.
The efficiency of extraction of lipids is highly dependent on polarity of the organic solvent or solvent mixture used. In general, solvent mixtures containing a polar and a non-polar component are able to extract a greater amount of lipids [
12]. Hexane/isopropanol (3:2) has accordingly proven to be one of the best non-halogenated solvent mixtures to extract fatty acids in
Isochrysis galbana [
13]. By the same token, most extraction methods suitable for carotenoids resort to such organic solvents as hexane, ethanol, isopropanol, acetone, methanol, benzene, and petroleum ether [
14,
15]. Although carotenoids can be polar (e.g., lutein) and nonpolar (e.g., β-carotene or carotenoids in ester form), the former is easily dissolved in polar solvents (e.g., acetone), while the latter is easily dissolved in nonpolar solvents (e.g., petroleum ether or hexane) [
16].
Therefore, food GRAS (Generally Recognized as Safe) solvents with lower environmental impact and toxicity were selected for this work. Ethyl lactate was chosen as alternative to ethyl acetate and halogenated solvents. It is environment-friendly and fully biodegradable into CO
2 and water. Its use has been approved for food products by U.S. Food and Drug Administration, and its miscibility with both hydrophilic and hydrophobic compounds make it appropriate to extract a diverse range of metabolites, namely carotenoids (in their stereoisomeric forms) and PUFA [
17]. Ethanol and isopropanol, two short chain alcohols, have been proposed as alternative extracting solvents due to their greater safety and lack of regulatory problems, namely for extraction of carotenoids [
8].
In attempts to cover a large range of polarities consistent with the various lipidic components of interest in microalgae, the next five food grade solvents were selected based on literature searches including data on their relative polarities: hexane, 0.009; acetone, 0.355; ethyl lactate, 0.460; isopropanol, 0.617; and ethanol, 0.654. Experimentation was conducted with plain ethanol, plain acetone, a mixture of hexane/isopropanol (3:2) (v/v) and plain ethyl lactate.
Due to the absence of a standard extraction method for lipidic components, our motivation was to investigate the potential impact of the aforementioned food grade solvents upon extraction, and assess the bioactivity potential of the extracts afterwards. The target compounds were carotenoids and PUFA, and the tested species were representative of two levels of cell complexity, i.e., Gloeothece sp. (prokaryote) and Scenedesmus obliquus (Sc. obliquus) (M2-1) (eukaryote). The antioxidant scavenging capacity was measured by four distinct assays: total activity (2,2′-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid (ABTS+•) and 2,2-diphenyl-1-picrylhydrazyl (DPPH•) radicals), and superoxide (O2•−) and nitric oxide (•NO−) radicals. Our findings may be useful in efforts to design more selective extraction protocols, and further incorporation of the extract obtained in food or cosmetics formulation based on the antioxidant potential attained.
2. Results and Discussion
2.1. Microalgae Production and Harvesting
Microalgae species were selected based on earlier studies by Guedes
et al. [
18]. They found that intracellular extracts of
Sc. obliquus (strain M2-1) possess a high antioxidant capacity when compared with other strains of
Scenedesmus. Moreover, its scavenging activity was well correlated with protective effects against DNA oxidative damage, with no mutagenic effects. It was also found that the maximum production of antioxidant compounds took place in the plain exponential phase, coinciding with the maximum peak production of lutein and β-carotene—thus suggesting a correlation between antioxidant capacity and presence of those carotenoids. Additionally,
Sc.
obliquus (M2-1) was also shown to have high content in PUFA, namely linoleic acid C18:2 (
n-6) [
6,
18]. In the same study,
Gloeothece sp. was revealed to possess antioxidant potential and an interesting profile of PUFA [
6,
18]. The growth conditions selected for biomass production were 25 °C and pH 8, based in an earlier study [
19].
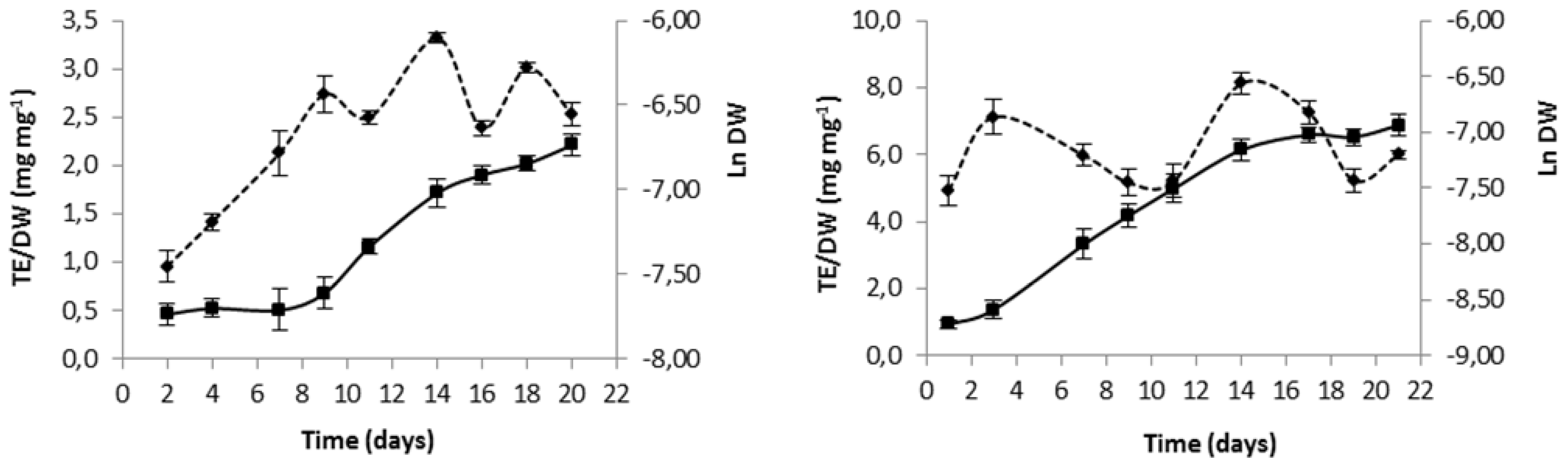
In order to fold best antioxidant potential of each microalga, culture time was selected based on growth curves and evolution in total antioxidant capacity (
Figure 1).
Figure 1.
Variation in time of biomass expressed as natural logarithm of dry weight (Ln DW) (mean ± standard deviation) (—), and variation of intracellular extract antioxidant capacity expressed as ratio of trolox equivalent (TE) antioxidant capacity to dry weight (DW) (mean ± standard deviation) (---), for Gloeothece sp. (A) and Scenedesmus obliquus (M2-1) (B).
Figure 1.
Variation in time of biomass expressed as natural logarithm of dry weight (Ln DW) (mean ± standard deviation) (—), and variation of intracellular extract antioxidant capacity expressed as ratio of trolox equivalent (TE) antioxidant capacity to dry weight (DW) (mean ± standard deviation) (---), for Gloeothece sp. (A) and Scenedesmus obliquus (M2-1) (B).
Inspection of
Figure 1 unfolds a maximum antioxidant intracellular capacity of both species in the intermediate exponential phase by 14 days of growth; hence, this was established as biomass harvesting day for subsequent use in lipidic extraction assays
2.2. Extracts Characterization
The principles underlying organic solvent extraction of microalgal lipidic compounds are anchored on the basic chemistry concept of “like dissolving like”. Due to the interactions between their long hydrophobic fatty acid chains, neutral lipids—such as TAG and carotenoids [
11]—contribute to weak van der Waals attractions between one another, leading to the formation of globules in the cytoplasm [
11].
A five-step protocol for organic solvent extraction has been proposed by Halim
et al. [
20], applicable to either non-polar or polar solvents. When a microalgal cell is exposed to a non-polar organic solvent, such as hexane: (1) the organic solvent penetrates through the cell membrane into the cytoplasm; (2) interacts with the neutral lipids via van der Waals forces alike; (3) an organic solvent-lipids complex is formed; (4) driven by a concentration gradient, the lipid complex diffuses across the cell membrane; and (5) said complex eventually crosses the static organic solvent film surrounding the cell into the bulk organic solvent. As a result, the neutral lipids are extracted out of the cells and remain dissolved in the non-polar organic solvent. A static organic solvent film is formed because the interaction between organic solvent and cell wall remains undisturbed for every rate of solvent flow or agitation. Some neutral lipids are, however, found in the cytoplasm complexed with polar lipids; such complexes are strongly linked via hydrogen bonds to proteins in the cell membrane. The van der Waals interactions between non-polar organic solvent and neutral lipids in the complex are insufficient to disrupt the membrane-based lipid-protein associations. Conversely, polar organic solvents (e.g., ethanol, isopropanol or acetone) can disrupt the lipid–protein associations by forming hydrogen bonds with the polar lipids in the complex [
11].
The mechanism of extraction of membrane-associated lipids by the mixture of non-polar/polar organic solvent follows the same major principles, except for minor differences arising from the solvent nature: (1) the organic solvent (both non-polar and polar) penetrates the cell membrane into the cytoplasm; (2) the solvent then interacts with the lipid complex—the non-polar organic solvent surrounds the lipid complex and enrolls in van der Waals associations with the neutral lipids of the complex, while the polar organic solvent surrounds the lipid complex and forms hydrogen bonds with the polar lipids in the complex, strong enough to counteract the lipid-protein associations binding the lipid complex to the cell membrane; (3) an organic solvent-lipid complex is formed, and dissociates away from the cell membrane; (4) the organic solvent-lipid complex diffuses across the cell membrane; and (5) said entity crosses the static organic solvent film surrounding the cell into the bulk organic solvent. Consequently, the addition of a polar organic solvent to a non-polar organic solvent facilitates extraction of membrane-associated neutral lipid complexes. However, the process inevitably leads to co-extraction of polar lipids [
11].
In this regard, it is expected that compound intracellular location affects its extractability by distinct solvents. Resorting to the solvents chosen, it was possible to produce extracts with different composition and, consequently, distinct antioxidant capacity, as discussed next in 2.2.1. Antioxidant Capacity section.
Numerous methods are used to assess the antioxidant capacity of natural compounds in biological systems. Two free radical scavenging methods commonly used involve ABTS
+• and DPPH
•, yet both such radicals are foreign to biological systems. ABTS
+• assays measures the relative ability of an antioxidant to scavenge the ABTS
+• generated in aqueous and organic solvents, as in ethanol: water 50:50 (
v/
v). Conversely, DPPH
• is widely used to determine antiradical/antioxidant capacities, but acts only upon species generated in a methanol phase. Comparatively, ABTS
+• also is more stable, so it can be used at different pH levels. DPPH
• may also suffer from color interference, for instance in the case of anthocyanins or carotenoids, which leads to underestimation of antioxidant capacity; moreover, it was reported that this method may be more sensitive to phenolic antioxidants over time [
20,
21]. Therefore, there is some controversy in the applicability of these assays for carotenoid antioxidant capacity assessment [
4,
21,
22]. In a report by Müller
et al. [
22], when comparing several methods to evaluate antioxidant capacity of carotenoids, DPPH
• did not show any scavenging capacity. However, this method seems appropriate to measure antioxidant capacity of poly-unsaturated fatty acids, as is the case of conjugated linoleic acid [
23]. Therefore, to avoid a misinterpretation of the total antiradical capacity of extracts, both DPPH
• and ABTS
+• assays were performed—thus allowed consistent confirmation of the relation between biochemical profile and results of said antioxidant assays (as described in the following sections).
Nitric oxide (
•NO
−) and superoxide (O
2•−) are two of the six major reactive oxygen species causing oxidative damage in the human body [
4]. The former is a short-lived free radical endogenously generated, involved in different physiological functions [
24]. It interacts with lipids, DNA and proteins, via direct oxidative reactions or via indirect radical-mediated mechanisms. Hence, any antioxidant scavenging capacity against this radical may unfold a similar capacity
in vivo and potential to prevent such diseases as chronic inflammatory diseases, cancer or neurodegenerative disorders [
25]. On the other hand, superoxide radical is the first product of oxygen univalent reduction. Its biological significance derives from its ability to generate other more reactive species, like hydroxyl radical (
•OH) and peroxynitrite (ONOO
−), and induce major damages
in vivo [
26].
All extracts of both microalgae acted as scavengers of ABTS
+•, DPPH
•, O
2•− and
•NO
−, in a concentration-dependent manner, with topical exceptions. Data can be compared through calculation of inhibitory concentration (IC) values, as acquired by plotting inhibitory scavenging percentages for various extract concentrations. Extracts from the two microalgae exhibited distinct behavior for each scavenging assay (
Table 1).
Regarding ABTS+•, acetonic extracts of both Gloeothece sp. and Sc. obliquus (M2-1) attained the best IC50 values: 63 and 41 μg·mL−1, respectively. On other hand, the most active in scavenging DPPH• were the hexane:isopranol (3:2) extract of Scenedesmus obliquus and ethanol the extract of Gloeothece sp. (IC25 of 194 and 274 μg·mL−1, respectively). Therefore, with respect to synthetic reactive species, Scenedesmus obliquus (M2-1) conveyed the best results compared to Gloeothece sp.; however, the other three extracts of the latter displayed the best results in the assay against ABTS+•.
In what concerns reactive species with biological significance, acetone and hexane:isopranol (3:2) extracts of Gloeothece sp. have strong activity against •NO−, both being quite similar (IC25 values of 6 and 7 μg·mL−1, respectively). On the other hand, only the ethanol and hexane:isopranol (3:2) extracts of Scenedesmus obliquus (M2-1) exhibited antioxidant capacity against this reactive nitrogen species (IC25 values of 15 and 20 μg·mL−1, respectively). Ethanol extracts of Gloeothece sp. and ethyl lactate extracts of Scenedesmus obliquus (M2-1) exhibited the best activities against O2•−, described by IC25 of 54 and 300 μg·mL−1, respectively. It is thus possible to conclude that each solvent system exerts different scavenging activity because of its composition. In order to establish some relationship between the observed activity and the lipidic composition, carotenoids and PUFA were quantified.
Table 1.
Comparison of antioxidant capacity of Gloeothece sp. and Scenedesmus obliquus (M2-1) extracts, in terms of IC (µg·mL−1) toward radicals ABTS+•, DPPH•, •NO− and O2•−.
Table 1.
Comparison of antioxidant capacity of Gloeothece sp. and Scenedesmus obliquus (M2-1) extracts, in terms of IC (µg·mL−1) toward radicals ABTS+•, DPPH•, •NO− and O2•−.
| Antioxidant Activity (µg·mL−1) |
|---|
| | Solvent | ABTS+• | DPPH• | •NO− | O2•− |
|---|
| IC50 | IC50 | IC25 | IC50 | IC25 | IC50 | IC25 |
|---|
| Gloeothece sp. | Ethanol | 75 | 629 | 274 | - | 23 | 247 | 54 |
| Ethyl lactate | 129 | - | 927 | 82 | 25 | - | - |
| Acetone | 63 | 850 | 310 | 22 | 6 | 1394 | 278 |
| HI (3:2) | 276 | - | 789 | 25 | 7 | 1183 | 357 |
| Scenedesmus obliquus (M2-1) | Ethanol | 87 | - | 633 | - | 15 | 637 | 416 |
| Ethyl lactate | 195 | 878 | 261 | - | - | 520 | 300 |
| Acetone | 41 | - | 488 | - | - | 826 | 620 |
| HI (3:2) | 648 | 412 | 194 | 60 | 20 | 1236 | 513 |
Lipidic Composition
As explained above, solvent polarity plays an important role on extractability of lipidic compounds due to the basic chemistry concept of “like dissolving like”. Moreover, it is important to remember that the cell location of the lipidic component is crucial for extraction because it needs to reach the compound into the cell.
As stated before, xanthophylls are relatively hydrophobic molecules typically associated with membranes and/or involved in non-covalent binding to specific proteins. Primary carotenoids are structural and functional components of the photosynthetic apparatus, typically confined to the thylakoid membrane complex—with proteins only being disrupted by polar organic solvents able to form hydrogen bonds [
14,
27]. Secondary carotenoids are produced in large quantities by microalgal cells, only after exposure to specific environmental stimuli (carotenogenesis), being usually found in lipid vesicles—in either the plastid stroma or the cytosol [
28].
In prokaryotic microalgae, such as
Gloeothece sp., most xanthophylls are associated with chlorophyll-binding polypeptides of the photosynthetic apparatus [
29]. In most green microalgae, carotenes and xanthophylls are synthesized within plastids, accumulating therein only. However, secondary xanthophylls in some green microalgae accumulate in the cytoplasm, which raises the possibility of an extra-plastidic site for carotenoid biosynthesis. Alternatively, xanthophylls synthesized in the chloroplast may be exported, and consequently accumulate in the cytoplasm—so, they may be found in essentially all cellular compartments [
29].
Prokaryotes and eukaryotes exhibit several structural differences in cell wall in terms of mechanical barrier. As it happens with several other members of the
Chlorococcales family, the trilaminar structure of the outer wall layers of eukaryotic
Scenedesmus species is composed of cellulose in the inner wall layers, and insoluble, acetolysis-resistant, lipid-containing biopolymerstermed algaenans localized in the trilaminar outer layer, thus contributing to cell wall rigidity [
30,
31]. Furthermore, prokaryotic
Gloeothece species hold a typical Gram-negative cell wall, mainly of polysaccharide nature, which differs in thickness and consistency [
32].
Besides solvent polarity, the cell structural complexity, including cell location of metabolites, of the two microalga under scrutiny affects lipidic component extractability. However, it is possible to propose a correlation between affinity of carotenoids for acetone and PUFA for ethanol (
Table 2 and
Table 3). At a first glance,
Gloeothece sp. extracts entail higher variety of carotenoids and higher total amount of PUFAs than their
Scenedesmus obliquus (M2-1) counterparts. Species of the
Scenedesmus genus possess particularly resistant cell walls, so extraction of carotenoids and fatty acids becomes notoriously difficult [
33].
Acetone is a solvent widely used in pigment extraction, as it extracts most photosynthetic pigments with a wide range of polarity [
28,
34,
35]. Our results indicate that acetonic extracts are the richest in carotenoids, particularly lutein. In
Gloeothece sp., the lutein content corresponds to
ca. 78% of the total quantified carotenoids (1.424 ± 0.079 µg
lutein·g
DryWeight−1—see
Table 2) and in
Scenedesmus obliquus (M2-1) corresponds to
ca. 47% (1.392 ± 0.034 µg
lutein·g
DW−1—see
Table 3). Conversely, violaxanthin and neoxanthin possess a significant expression in acetonic extract of
Scenedesmus obliquus (M2-1), 22.7% and 25.5% of the total quantified carotenoids, respectively (
Table 3). However, acetone is not selective only for carotenoids, since PUFA are also extracted. In acetonic extract of
Scenedesmus obliquus (M2-1), the content of PUFA ranges from 50% in the case of oleic acid to 71% of linoleic acid in the ethanol extract, and linolenic acid is even more concentrated in acetonic extract (
Table 3). This provides evidence of the dependence of the solvent ability to extract the feedstock species, as emphasized before [
14].
Ethanol affinity for PUFA is clear; for example, it extracts 3–7.8-fold more linolenic acid from
Gloeothece sp. than the other solvents (
Table 2). Ethanol is also able to extract 10-fold more linolelaidic acid from both
Gloeothece sp. and
Scenedesmus obliquus (M2-1) than ethyl lactate (
Table 2). Ethanol can extract carotenoids as well, but at a lower rate; for instance,
Gloeothece sp. ethanol extract contains 1.5–3-fold less carotenoids than its acetonic counterpart, although an exception occurs in what concerns to violaxanthin that is extracted to three-fold higher extent than with acetone (
Table 2).
Ethanol has a different behavior in extracting carotenoids from Scenedesmus obliquus (M2-1), as it extracts three-fold less lutein and 1.7-fold less neoxanthin. Due to its lower affinity for carotenoids, it was not possible to quantify the remaining carotenoids.
Ethyl lactate has been proposed to extract carotenoids, particularly lutein, from plant material [
17]; however, its performance in the microalgae under the processing conditions used is below expectation, in view of the low level of extraction of carotenoids. Still, ethyl lactate showed some selectivity for lutein in both species (
Table 2 and
Table 3). Ethyl lactate was able to extract PUFA as γ-linolenic acid from
Spirulina sp. [
36]. Ethyl lactate indeed extracted 6.185 ± 0.265 mg
FattyAcids·g
DW−1 from
Gloeothece sp., 55% of that corresponding to linolenic acid; furthermore, it was the only solvent that extracted linolelaidic acid to detectable levels (
Table 2). On the contrary, ethyl lactate performance toward PUFA extraction from
Scenedesmus obliquus (M2-1) rated the poorest—see
Table 3.
Previous studies have proven that hexane:isopropanol (3:2) mixture is one of the best non-halogenated solvent mixtures to extract fatty acids [
14]. However, it only led to a reasonable result regarding the extraction of oleic and
cis-vaccenic acid from
Gloeothece sp. (
Table 2), and, surprisingly, of the xanthophyll violaxanthin. With respect to
Scenedesmus obliquus (M2-1), this solvent extracted 1.849 ± 0.156 mg
FA·g
DW−1 of total PUFA (
Table 3). In addition to carotenoids and PUFA, hexane:isopropanol (3:2) has been claimed to extract more non-lipids (e.g., proteins and carbohydrates) than plain hexane, due to the polar nature of isopropanol [
14]—which may have contributed to the low recovery of PUFA and carotenoids.
Table 2.
Gloeothece sp. extracts lipidic profile in terms of carotenoids (µgcarotenoid·gDry Weight−1) and PUFA (mgFattyAcids·gDW−1) (mean ± standard deviation).
Table 2.
Gloeothece sp. extracts lipidic profile in terms of carotenoids (µgcarotenoid·gDry Weight−1) and PUFA (mgFattyAcids·gDW−1) (mean ± standard deviation).
| | Carotenoids (µgcarotenoid·gDW−1) | | PUFA (mgFA·gDW−1) | |
|---|
| Solvent | Violaxanthin | Neoxanthin | Lutein | α-Carotene | β-Carotene | Total Carotenoids | Oleic | cis-Vaccenic | Linoleic | Linolelaidic | Linolenic | Total PUFA |
|---|
| Ethanol | 0.181 ± 0.004 | 0.114 ± 0.004 | 0.822 ± 0.021 a | 0.018 ± 0.001 | 0.122 ± 0.006 | 1.258 ± 0.022 b | 0.771 ± 0.064 c | - | 2.250 ± 0.198 | - | 10.100 ± 0.212 | 13.219± 0.233 |
| Ethyl lactate | 0.067 ± 0.002 | 0.043 ± 0.001 | 0.424 ± 0.030 | - | 0.050 ± 0.002 | 0.584 ± 0.031 | 1.007 ± 0.192 | 0.264 ± 0.074 | 1.267 ± 0.200 | 0.201 ± 0.046 | 3.406 ± 0.111 | 6.185 ± 0.265 |
| Acetone | 0.058 ± 0.005 | 0.180 ± 0.013 | 1.424 ± 0.079 | 0.057 ± 0.004 | 0.251 ± 0.004 | 1.806 ± 0.080 | 0.773 ± 0.054 c | - | 0.255 ± 0.30 | - | 1.286 ± 0.064 | 2.317 ± 0.106 |
| HI (3:2) | 0.220 ± 0.008 | 0.086 ± 0.004 | 0.868 ± 0.015 a | 0.056 ± 0.003 | 0.067 ± 0.002 | 1.301 ± 0.014 b | 1.352 ± 0.032 | 0.689 ± 0.038 | 0.538 ± 0.098 | - | 2.631 ± 0.119 | 5.216 ± 0.126 |
Table 3.
Scenedesmus obliquus (M2-1) extracts lipidic profile in terms of carotenoids (µgcarotenoid·gDry Weight) and PUFA (mgFatty Acids·gDW−1) (mean ± standard deviation).
Table 3.
Scenedesmus obliquus (M2-1) extracts lipidic profile in terms of carotenoids (µgcarotenoid·gDry Weight) and PUFA (mgFatty Acids·gDW−1) (mean ± standard deviation).
| | Carotenoids (µgcarotenoid·g DW−1) | PUFA (mgFA·gDW−1) |
|---|
| Solvent | Violaxanthin | Neoxanthin | Lutein | β-Criptoxantin | α-Carotene | β-Carotene | Total Carotenoids | Oleic | Linoleic | Linolelaidic | Linolenic | Total PUFA |
|---|
| Ethanol | - | 0.439 ± 0.019 | 0.464 ± 0.011 a | - | - | - | 0.904 ± 0.019 | 0.889 ± 0.060 | 1.045 ± 0.097 | 1.045 ± 0.097 | 0.932 ± 0.088 | 2.888 ± 0.078 |
| Ethyl lactate | - | - | 0.156 ± 0.012 | - | - | - | 0.156 ± 0.012 | 0.320 ± 0.070 | 0.465 ± 0.012 | 0.147 ± 0.021 | 0.522 ± 0.078 | 1.454 ± 0.073 |
| Acetone | 0.674 ± 0.057 | 0.759 ± 0.053 | 1.392 ± 0.034 | 0.019 ± 0.001 | 0.022 ± 0.011 | 0.100 ± 0.004 | 2.970 ± 0.068 | 0.427 ± 0.076 b | 0.752 ± 0.22 a | - | 1.199 ± 0.089 | 2.381 ± 0.122 |
| HI (2:1) | 0.020 ± 0.001 | 0.357 ± 0.009 | 0.420 ± 0.034 a | - | - | - | 0.797 ± 0.030 | 0.518 ± 0.055 b | 0.734 ± 0.075 a | - | 0.577 ± 0.049 | 1.849 ± 0.156 |
2.3. Relation of Antioxidant Capacity with Carotenoid and PUFA Contents
There are a number of reports on the evaluation of antioxidant capacity in prokaryotic and eukaryotic microalgae compounds from lipophilic and hydrophilic nature [
19,
37,
38], but most of them have not performed antioxidant scavenging assays in lipid-rich extracts. An important and well-known class of antioxidants from microalgae are carotenoids, and they are already produced to commercial scale (e.g., astaxanthin from
Haematococcus sp. and β-carotene from
Dunaliella sp.) for use as additive in food and feed, as well as in cosmetics and as food supplements [
39]. Flavonoids, sterol, reducing sugars and tannins may also exert antiradical or antioxidant capacities in alcoholic extracts [
21]. Their co-extraction may provide an explanation for some unexpected results of antioxidant capacity obtained with ethyl lactate and hexane:isopropanol (3:2) extracts from
Scenedesmus obliquus (M2-1). One should take into account that synergic or antagonic interactions may occur between the compounds found in an extract. Hence, high amounts of a known antioxidant compound do not necessarily imply a high antioxidant activity, in view of the crude nature of the extracts obtained.
ABTS
+• assay was used before to evaluate the antioxidant capacity of carotenoid rich extracts (namely in lutein and β-carotene [
40]. Upon inspection of
Table 1,
Table 2 and
Table 3, it is possible to reach some conclusions: acetonic extracts of both microalgae species attained the best IC
50 values in this assay and they contain the highest levels of carotenoids, namely of lutein and β-carotene. IC
50 values found for ethyl lactate extracts and its selectivity to lutein suggests that this xanthophyll may be responsible for the main antioxidant capacity of these extracts.
With regard to results in
Table 1,
Table 2 and
Table 3, one realizes that it is not always possible to make a correlation between carotenoids content and antiradical capacity, which is supported by the fact that some studies revealed that DPPH
• does not detect carotenoids antioxidant capacity [
4,
21,
22]. Furthermore, this assay was used to quantify the antioxidant capacity of conjugated linoleic acid [
23]. Nevertheless, one concludes that ethanol extract of
Gloeothece sp. is particularly rich in linoleic and linolenic acids, which, besides lutein, may contribute to the best IC
25 values attained against O
2•− [
23].
Ethanolic and acetonic extracts from Gloeothece sp. seem interesting from an antioxidant point of view. In terms of scavenging capacity, ethanolic extract attained the best results against DPPH• and O2•−, while acetonic was the most effective against ABTS•+ and •NO−. These extracts have distinct contents of carotenoids and PUFA, which may explain the paired results. Ethanol extract is indeed richer in PUFA (13.219 ± 0.233 mgFA·gDW−1—76.4% corresponding to linolenic acid and 17% linoleic acid) than in carotenoids (1.258 ± 0.022 µgcarotenoid·gDW−1—65.3% lutein and 9.7% β-carotene); and acetonic extract is richer in carotenoids (1.806 ± 0.080 µgcarotenoid·gDW–1—78.8% lutein and 13.9% β-carotene) than in PUFA (2.317 ± 0.106 mgFA·gDW−1—55.5% linolenic acid and 11% linoleic acid). This pattern was not observed in Scenedesmus obliquus (M2-1) extracts; in fact, each extract exhibited a particular antioxidant activity. Acetonic extract was the most interesting in the ABTS•+ assay, possibly due to its distinctive content in lutein (1.392 ± 0.034 µgcarotenoid·gDW−1—46.8% of total carotenoids). On the other hand, hexane:isopropanol (3:2) extract exhibited a great activity in the DPPH• assay and ethanolic extract in the •NO− assay, but these two extracts have three-fold less carotenoids than the acetonic extract, although PUFA within the same magnitude. Ethyl lactate exhibited the best IC25 in O2•− assay (300 µg·mL−1) between Scenedesmus obliquus (M2-1) extracts, perhaps due to the great affinity of this solvent to lutein, which may exert an influence on its antioxidant capacity.
4. Conclusions
Concerning total antioxidant capacity, ethanol Gloeothece sp. extracts performed best results in DPPH• and O2•− assays, possibly due to its content in PUFA (76.4% of linolenic acid) and carotenoids (65.3% of lutein and 9.7% of β-carotene). Similarly, the acetonic extract attained good results in ABTS+• and •NO− assays, and probably for the same reasons, it content in carotenoids (78.8% of lutein and 13.9% of β-carotene) and PUFA (55.5% of linolenic acid and 11% of linoleic acid). Gloeothece sp. is a prokaryotic microalga poorly studied so far, so findings of this study may justify further exploitation of its antioxidant potential once it appears promising toward nutraceutical formulations.
Scenedesmus obliquus (M2-1) also seems to be a promising source of antioxidant-rich extracts. Acetone extract exhibited the best antioxidant capacity in ABTS+• assay, likely associated with its carotenoids content, 47% of which is lutein. Note that the hexane:isopropanol (3:2) extract also demonstrated the best result of antioxidant capacity in DPPH• assay.
Solvents used in extraction of lipidic components seems to be critical for the antioxidant performance—which appears to hinge, in particular, on the balance between carotenoids and PUFAs. However, further studies are warranted to confirm whether said compounds are by themselves responsible for the good performance recorded in antioxidant assays, or some form of interaction/synergism exists between them.
In terms of lipidic components extraction, in general, acetone is the most suitable to extract carotenoids, and ethanol stands out in PUFA extraction, regardless of the microalgae species.