Pseudaboydins A and B: Novel Isobenzofuranone Derivatives from Marine Fungus Pseudallescheria boydii Associated with Starfish Acanthaster planci
Abstract
:1. Introduction
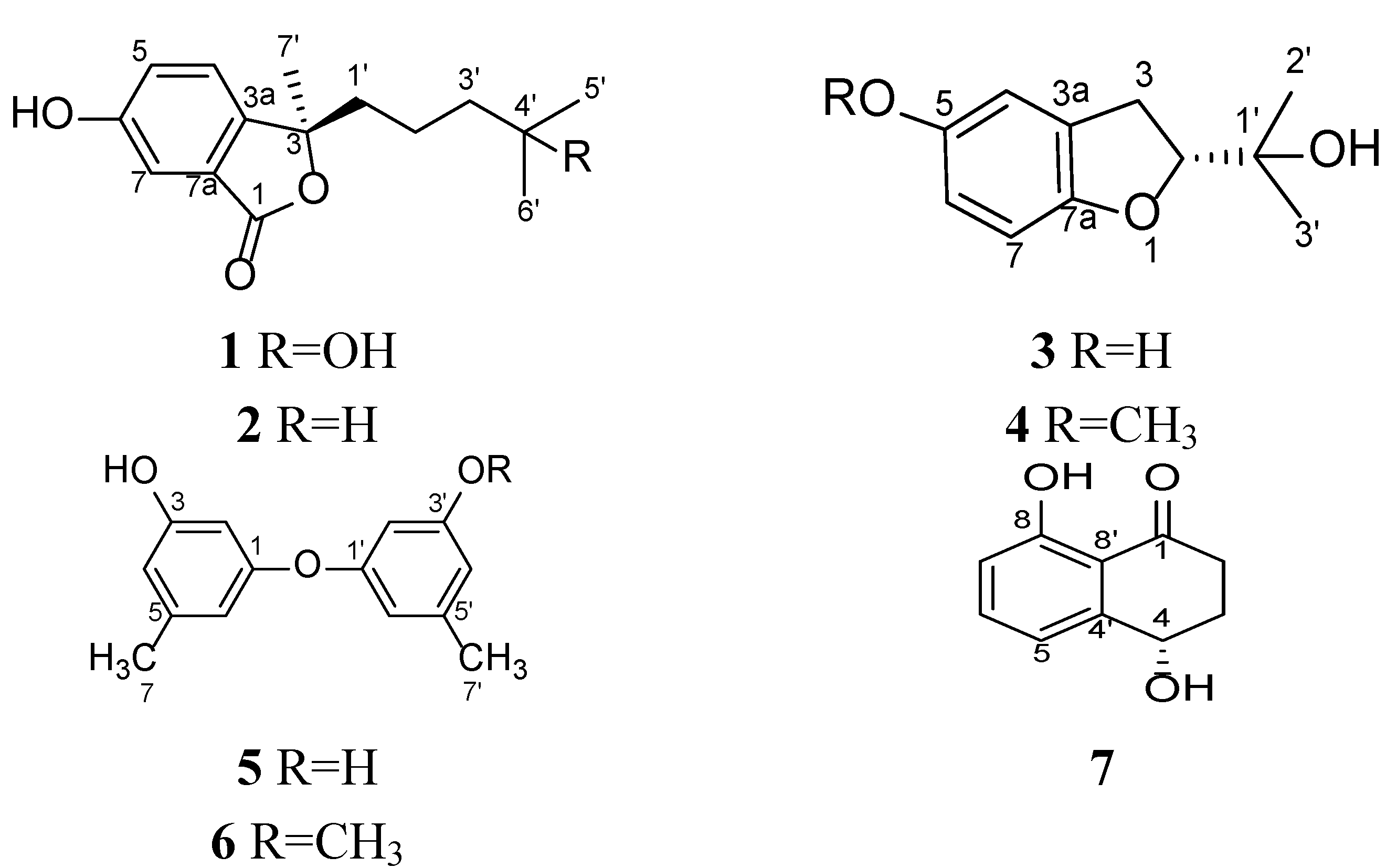
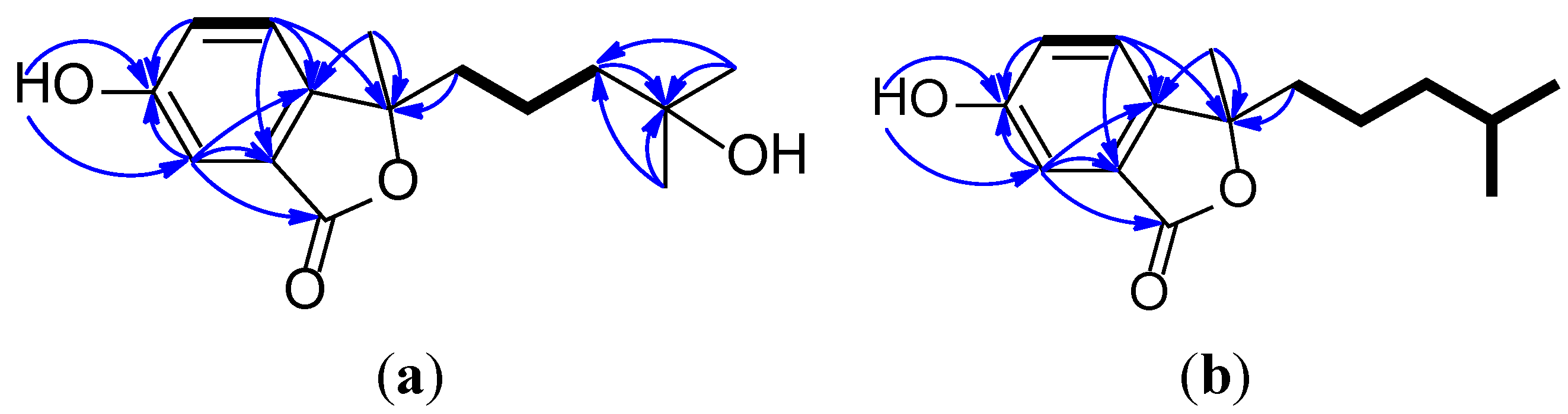
2. Results and Discussion
| Position | 1 | 2 | ||||
|---|---|---|---|---|---|---|
| δC | Type | δH, Multiple, ( J in Hz) | δC | Type | δH, Multiple, ( J in Hz) | |
| 1 | 171.8 | C | 171.6 | C | ||
| 2 | ||||||
| 3 | 77.7 | C | 79.1 | C | ||
| 3a | 137.1 | C | 135.5 | C | ||
| 4 | 124.7 | CH | 7.13, d (8.0) | 126.5 | CH | 7.08, d (8.0) |
| 5 | 121.5 | CH | 7.56, d (8.0) | 121.3 | CH | 7.54, d (8.0) |
| 6 | 157.1 | C | 156.1 | C | ||
| 7 | 119.0 | CH | 7.55, s | 119.3 | CH | 7.57, s |
| 7a | 129.7 | C | 129.8 | C | ||
| 1′ | 33.9 | CH2 | 2.45, brd (13.6) 1.65, m | 42.9 | CH2 | 1.92, dt (14.4, 8.0) 1.80, dt (14.4, 8.0) |
| 2′ | 16.7 | CH2 | 1.71, m | 21.7 | CH2 | 1.29, m |
| 3′ | 36.8 | CH2 | 1.52, m | 39.1 | CH2 | 1.16, m |
| 4′ | 75.4 | C | 27.8 | CH | 1.49, nine (6.4) | |
| 5′ | 32.0 | CH3 | 1.28, s | 22.6 | CH3 | 0.82, d (6.4) |
| 6′ | 24.8 | CH3 | 0.95, s | 22.4 | CH3 | 0.82, d (6.4) |
| 7′ | 31.4 | CH3 | 1.50, s | 29.0 | CH3 | 1.68, s |
| 6-OH | 9.33, brs | 9.34, brs | ||||
| 4′-OH | 1.80, brs | |||||
| Position | 3 a | 4 b | ||||
|---|---|---|---|---|---|---|
| δC | Type | δH, Multiple, (J in Hz) | δC | Type | δH, Multiple, (J in Hz) | |
| 1 | ||||||
| 2 | 88.6 | CH | 4.42, dd (9.6, 8.4) | 89.4 | CH | 4.58, dd (9.2, 8.8) |
| 3 | 30.5 | CH2 | 3.06, dd (16.0, 8.4); 2.98, dd (16.0, 9.6) | 31.2 | CH2 | 3.17, dd (16.0, 8.8); 3.10, dd (16.0, 9.2) |
| 3a | 128.1 | C | 128.1 | C | ||
| 4 | 111.9 | CH | 6.59, d (2.4) | 111.2 | CH | 6.75, d (1.6) |
| 5 | 150.9 | C | 154.1 | C | ||
| 6 | 113.3 | CH | 6.42, dd (8.4, 2.4) | 112.8 | CH | 6.64, dd (8.8, 1.6) |
| 7 | 108.3 | CH | 6.50, d (8.4) | 108.9 | CH | 6.68, d (8.8) |
| 7a | 152.5 | C | 153.7 | C | ||
| 1′ | 70.1 | C | 71.8 | C | ||
| 2′ | 24.7 | CH3 | 1.09, s | 23.9 | CH3 | 1.20, s |
| 3′ | 26.2 | CH3 | 1.11, s | 26.2 | CH3 | 1.33, s |
| 5-OH | 8.69, s | |||||
| 1′-OH | 4.50, s | 1.97, brs | ||||
| 5-OCH3 | 56.0 | CH3 | 3.75, s | |||
3. Experimental Section
3.1. General Experimental Procedures
3.2. Fungal Strain and Culture Method
3.3. Extraction and Isolation
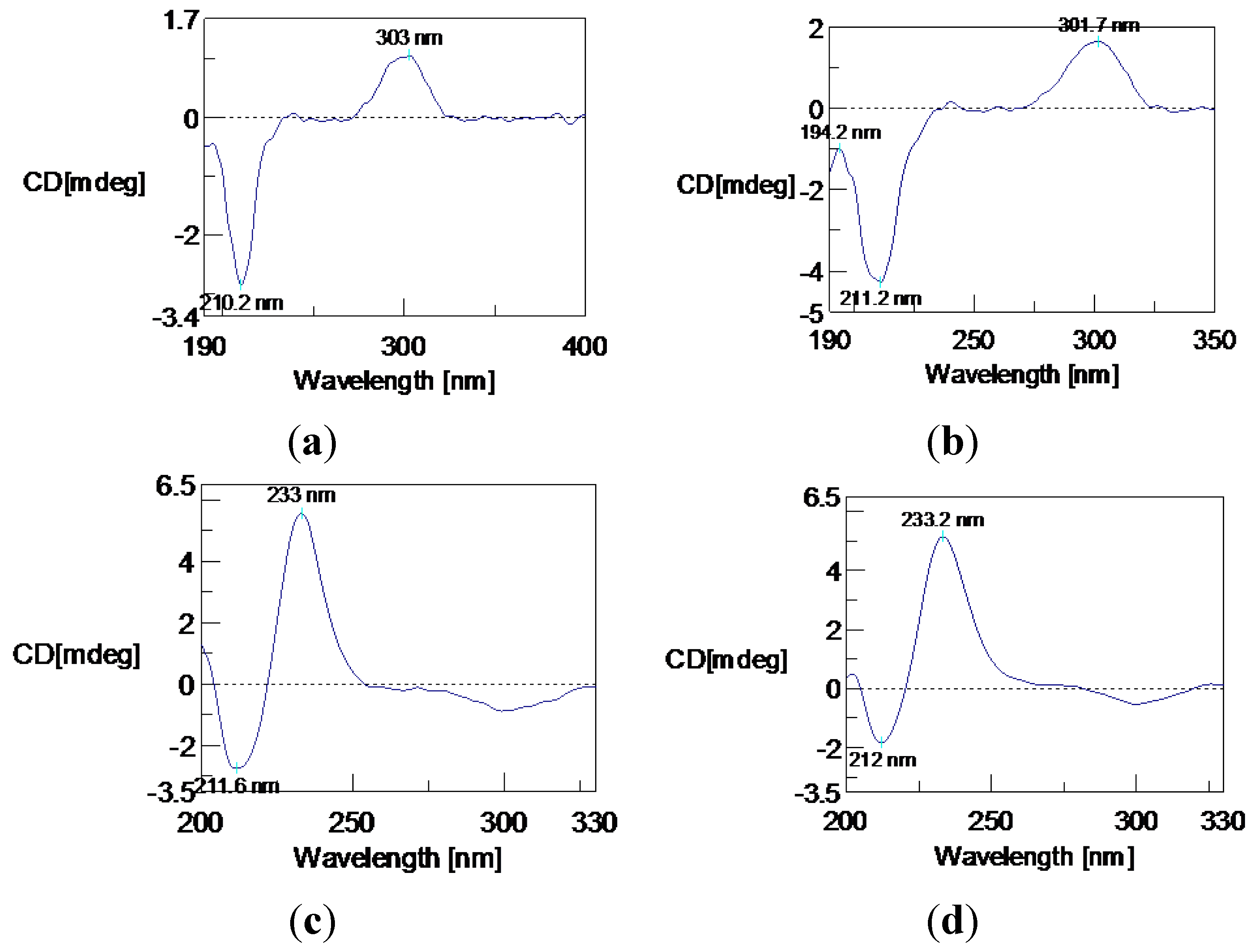
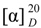 : +200° (c = 0.025, MeOH). CD (MeCN) λmax (Δε): 210.2 (−5.41), 303 (+2.01) nm. IR (KBr): 3217, 3073, 2978, 1680, 1574, 1507, 1419, 1389, 1354, 1287, 1230, 1161, 1065, 957, 891, 763 cm−1. UV (MeOH) λmax (ε): 217 nm (8152), 243 nm (7142), 298 nm (2247). 1H and 13C-NMR: Table 1. LREIMS: m/z 264, 246, 231, 203, 189, 181, 165, 147, 133, 119, 105, 91, 69. HREIMS: m/z 264.1355 [M]+ (calcd. for C15H20O4+: 264.1362).
: +200° (c = 0.025, MeOH). CD (MeCN) λmax (Δε): 210.2 (−5.41), 303 (+2.01) nm. IR (KBr): 3217, 3073, 2978, 1680, 1574, 1507, 1419, 1389, 1354, 1287, 1230, 1161, 1065, 957, 891, 763 cm−1. UV (MeOH) λmax (ε): 217 nm (8152), 243 nm (7142), 298 nm (2247). 1H and 13C-NMR: Table 1. LREIMS: m/z 264, 246, 231, 203, 189, 181, 165, 147, 133, 119, 105, 91, 69. HREIMS: m/z 264.1355 [M]+ (calcd. for C15H20O4+: 264.1362). 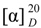 : +53.6° (c = 0.078, MeOH). CD (MeCN) λmax (Δε): 211.2 (−7.22), 301.7 (+2.78) nm. IR (KBr): 3377, 3073, 2952, 1687, 1575, 1510, 1467, 1380, 1294, 1223, 1127, 1052, 947, 890, 766 cm−1. UV (MeOH) λmax (ε): 219 nm (11,833), 244 nm (12,258), 299 nm (4202). 1H and 13C-NMR: Table 1. LREIMS: m/z 248, 233, 203, 192, 178, 165, 147, 133, 119, 105, 91, 77. HREIMS: m/z 248.1408 [M]+ (calcd. for C15H20O3+: 248.1412).
: +53.6° (c = 0.078, MeOH). CD (MeCN) λmax (Δε): 211.2 (−7.22), 301.7 (+2.78) nm. IR (KBr): 3377, 3073, 2952, 1687, 1575, 1510, 1467, 1380, 1294, 1223, 1127, 1052, 947, 890, 766 cm−1. UV (MeOH) λmax (ε): 219 nm (11,833), 244 nm (12,258), 299 nm (4202). 1H and 13C-NMR: Table 1. LREIMS: m/z 248, 233, 203, 192, 178, 165, 147, 133, 119, 105, 91, 77. HREIMS: m/z 248.1408 [M]+ (calcd. for C15H20O3+: 248.1412). 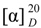 : +81.3° (c = 0.49, MeOH). CD (MeCN) λmax (Δε): 211.6 (−3.21), 233 (+6.53) nm. 1H and 13C-NMR: Table 2. LREIMS: m/z 194, 176, 161, 136, 123, 107, 77, 59.
: +81.3° (c = 0.49, MeOH). CD (MeCN) λmax (Δε): 211.6 (−3.21), 233 (+6.53) nm. 1H and 13C-NMR: Table 2. LREIMS: m/z 194, 176, 161, 136, 123, 107, 77, 59.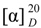 : +42.5° (c = 0.41, MeOH). CD (MeCN) λmax (Δε): 212 (−2.32), 233.2 (+6.48) nm. 1H and 13C-NMR: Table 2. LREIMS: m/z 208, 190, 175, 150, 135, 121, 107, 91, 77.
: +42.5° (c = 0.41, MeOH). CD (MeCN) λmax (Δε): 212 (−2.32), 233.2 (+6.48) nm. 1H and 13C-NMR: Table 2. LREIMS: m/z 208, 190, 175, 150, 135, 121, 107, 91, 77. 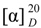 : −9.6° (c = 0.12, MeOH). 13C-NMR (CDCl3, 100 MHz) δC: 204.3 (C-1), 162.7 (C-8), 145.9 (C-4′), 137.0 (C-6), 117.8 (C-7), 117.4 (C-5), 115.2 (C-8′), 67.7 (C-4), 34.6 (C-2), 31.2 (C-3). 1H NMR (CDCl3, 400 MHz) δH: 12.41 (brs, 8-OH), 7.49 (dd, J = 8.4, 7.2 Hz, H-6), 7.02 (d, J = 7.2 Hz, H-5), 6.92 (d, J = 8.4 Hz, H-7), 4.91 (dd, J = 8.0, 4.0 Hz, H-4), 3.00 (ddd, J = 17.6, 8.4, 4.8 Hz, H-2a), 2.64 (ddd, J = 17.6, 8.0, 4.8 Hz, H-2b), 2.34 (dddd, J = 15.2, 8.4, 4.8, 4.0 Hz, H-3a), 2.19 (dddd, J = 15.2, 8.0, 8.0, 4.8 Hz, H-2b), 1.97 (s, 4-OH). LREIMS: m/z 178, 160, 150, 132, 121, 104, 93, 77, 65, 51.
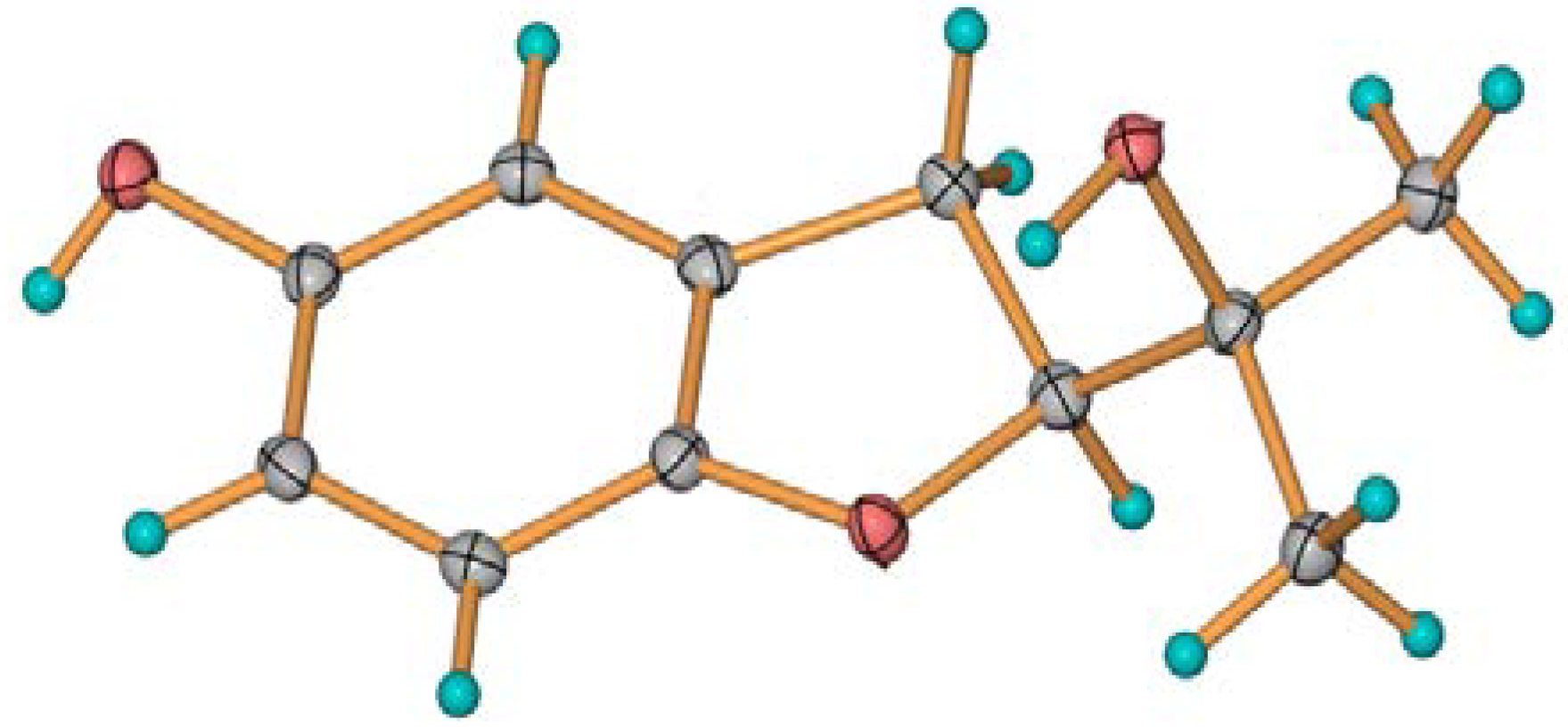
: −9.6° (c = 0.12, MeOH). 13C-NMR (CDCl3, 100 MHz) δC: 204.3 (C-1), 162.7 (C-8), 145.9 (C-4′), 137.0 (C-6), 117.8 (C-7), 117.4 (C-5), 115.2 (C-8′), 67.7 (C-4), 34.6 (C-2), 31.2 (C-3). 1H NMR (CDCl3, 400 MHz) δH: 12.41 (brs, 8-OH), 7.49 (dd, J = 8.4, 7.2 Hz, H-6), 7.02 (d, J = 7.2 Hz, H-5), 6.92 (d, J = 8.4 Hz, H-7), 4.91 (dd, J = 8.0, 4.0 Hz, H-4), 3.00 (ddd, J = 17.6, 8.4, 4.8 Hz, H-2a), 2.64 (ddd, J = 17.6, 8.0, 4.8 Hz, H-2b), 2.34 (dddd, J = 15.2, 8.4, 4.8, 4.0 Hz, H-3a), 2.19 (dddd, J = 15.2, 8.0, 8.0, 4.8 Hz, H-2b), 1.97 (s, 4-OH). LREIMS: m/z 178, 160, 150, 132, 121, 104, 93, 77, 65, 51.3.4. Crystal Structure Determination of 3
3.5. Cytotoxicity Assay
3.6. Antibacterial Activity Assay
4. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Li, H.J.; Jiang, W.H.; Liang, W.L.; Huang, J.X.; Mo, Y.F.; Ding, Y.Q.; Lam, C.K.; Qian, X.J.; Zhu, X.F.; Lan, W.J. Induced marine fungus Chondrostereum sp. as a means of producing new sesquiterpenoids Chondrosterins I and J by using glycerol as the carbon source. Mar. Drugs 2014, 12, 167–175. [Google Scholar] [CrossRef]
- Li, H.J.; Chen, T.; Xie, Y.L.; Chen, W.D.; Zhu, X.F.; Lan, W.J. Isolation and structural elucidation of Chondrosterins F-H from the marine fungus Chondrostereum sp. Mar. Drugs 2013, 11, 551–558. [Google Scholar] [CrossRef]
- Li, H.J.; Xie, Y.L.; Xie, Z.L.; Chen, Y.; Lam, C.K.; Lan, W.J. Chondrosterins A–E, triquinane-type sesquiterpenoids from soft coral-associated fungus Chondrostereum sp. Mar. Drugs 2012, 10, 627–638. [Google Scholar] [CrossRef]
- Li, H.J.; Lan, W.J.; Lam, C.K.; Yang, F.; Zhu, X.F. Hirsutane sesquiterpenoids from the marine-derived fungus Chondrostereum sp. Chem. Biodivers. 2011, 8, 317–324. [Google Scholar] [CrossRef]
- Lan, W.J.; Zhao, Y.; Xie, Z.L.; Liang, L.Z.; Shao, W.Y.; Zhu, L.P.; Yang, D.P.; Zhu, X.F.; Li, H.J. Novel sorbicillin analogues from the marine fungus Trichoderma sp. associated with the seastar Acanthaster planci. Nat. Prod. Commun. 2012, 7, 1337–1340. [Google Scholar]
- Zhao, Y.; Li, S.Q.; Li, H.J.; Lan, W.J. Lanostane triterpenoids from the fungus Ceriporia lacerate. Chem. Nat. Compd. 2013, 49, 653–656. [Google Scholar] [CrossRef]
- Xie, Z.L.; Li, H.J.; Wang, L.Y.; Liang, W.L.; Liu, W.; Lan, W.J. Trichodermaerin, a new lactone from the marine fungus Trichoderma erinaceum associated with the sea star Acanthaster planci. Nat. Prod. Commun. 2013, 8, 67–68. [Google Scholar]
- Chang, Y.C.; Deng, T.S.; Pang, K.L.; Hsiao, C.J.; Chen, Y.Y.; Tang, S.J.; Lee, T.H. Polyketides from the littoral plant associated fungus Pseudallescheria boydii. J. Nat. Prod. 2013, 76, 1796–1800. [Google Scholar]
- Su, H.J.; Lin, M.J.; Tsou, Y.J.; Ko, W.H. Pseudallin, a new antibiotic produced by the human pathogenic fungus Pseudallescheria boydii, with ecological significance. Bot. Stud. 2012, 53, 239–242. [Google Scholar]
- Ko, W.H.; Tsou, Y.J.; Ju, Y.M.; Hsieh, H.M.; Ann, P.J. Production of a fungistatic substance by Pseudallescheria boydii isolated from soil amended with vegetable tissues and its significance. Mycopathologia 2010, 169, 125–131. [Google Scholar] [CrossRef]
- Pavlaskova, K.; Nedved, J.; Kuzma, M.; Zabka, M.; Sulc, M.; Sklenar, J.; Novak, P.; Benada, O.; Kofronova, O.; Hajduch, M.; et al. Characterization of Pseudacyclins A–E, a suite of cyclic peptides produced by Pseudallescheria boydii. J. Nat. Prod. 2010, 73, 1027–1032. [Google Scholar]
- Nirma, C.; Eparvier, V.; Stien, D. Antifungal agents from Pseudallescheria boydii SNB-CN73 isolated from a Nasutitermes sp. Termite. J. Nat. Prod. 2013, 76, 988–991. [Google Scholar]
- Pinto, M.R.; Rodrigues, M.L.; Travassos, L.R.; Haido, R.M.T.; Wait, R.; Barreto-Bergter, E. Characterization of glucosylceramides in Pseudallescheria boydii and their involvement in fungal differentiation. Glycobiology 2002, 12, 251–260. [Google Scholar]
- Uchida, I.; Kuriyama, K. The π–π* circular dichroism of α, β-unsaturated γ-lactones. Tetrahedron Lett. 1974, 15, 3761–3764. [Google Scholar] [CrossRef]
- Gawronski, J.K.; van Oeveren, A.; van der Deen, H.; Leung, C.W.; Feringa, B.L. Simple circular dichroic method for the determination of absolute configuration of 5-substituted 2(5H)-furanones. J. Org. Chem. 1996, 61, 1513–1515. [Google Scholar] [CrossRef]
- Tanaka, M.; Nara, F.; Yamasato, Y.; Ono, Y.; Ogita, T. F-11334s, new inhibitors of membrane-bound neutral sphingomyelinase. J. Antibiot. 1999, 52, 827–830. [Google Scholar]
- Maria, D.B.; Giovanni, V.; Paola, V.F.; Giovanni, F. Fungal metabolites. 28. The chemistry of Lactarius fuliginosus and Lactarius picinus. Tetrahedron 1992, 48, 7331–7344. [Google Scholar] [CrossRef]
- Flack, H.D.; Bernardinelli, G. The use of X-ray crystallography to determine absolute configuration. Chirality 2008, 20, 681–690. [Google Scholar] [CrossRef]
- Takenaka, Y.; Tanahashi, T.; Nagakura, N.; Hamada, N. Phenyl ethers from cultured lichen mycobionts of Graphis scripta var. serpentina and G. rikuzensis. Chem. Pharm. Bull. 2003, 51, 794–797. [Google Scholar]
- Wang, J.F.; Lu, Z.Y.; Liu, P.P.; Wang, Y.; Li, J.; Hong, K.; Zhu, W.M. Cytotoxic polyphenols from the fungus Penicillium expansum 091006 endogenous with the mangrove plant Excoecaria agallocha. Planta Med. 2012, 78, 1861–1866. [Google Scholar]
- Talapatra, S.K.; Karmacharya, B.; de, S.C.; Talapatra, B. (−)-Regiolone, an α-tetralone from Juglans regia: Structure, stereochemistry and conformation. Phytochemistry 1988, 27, 3929–3932. [Google Scholar]
- Sheldrick, G.M. SADABS: Program for Empirical Absorption Correction of Area Detector Data; University of Göttingen: Göttingen, Germany, 1996. [Google Scholar]
- Sheldrick, G.M. SHELXTL 5.10 for Windows NT: Structure Determination Software Programs; Bruker Analytical X-ray Systems, Inc.: Madison, WI, USA, 1997. [Google Scholar]
- CCDC CIF Depository Request Form. Available online: http://www.ccdc.cam.ac.uk/cgi-bin/catreq.cgi (accessed on 26 May 2014).
- Logrado, L.P.; Santos, C.O.; Romeiro, L.A.S.; Costa, A.M.; Ferreira, J.R.O.; Cavalcanti, B.C.; Manoel de Moraes, O.; Costa-Lotufo, L.V.; Pessoa, C.; dos Santos, M.L. Synthesis and cytotoxicity screening of substituted isobenzofuranones designed from anacardic acids. Eur. J. Med. Chem. 2010, 45, 3480–3489. [Google Scholar]
- Huang, X.Z.; Zhu, Y.; Guan, X.L.; Tian, K.; Guo, J.M.; Wang, H.B.; Fu, G.M. A novel antioxidant isobenzofuranone derivative from fungus Cephalosporium sp. AL031. Molecules 2012, 17, 4219–4224. [Google Scholar]
- Tayone, W.C.; Honma, M.; Kanamaru, S.; Noguchi, S.; Tanaka, K.; Nehira, T.; Hashimoto, M. Stereochemical investigations of isochromenones and isobenzofuranones isolated from Leptosphaeria sp. KTC727. J. Nat. Prod. 2011, 74, 425–429. [Google Scholar] [CrossRef]
- Ding, G.; Liu, S.C.; Guo, L.D.; Zhou, Y.G.; Che, Y.S. Antifungal metabolites from the plant endophytic fungus Pestalotiopsis foedans. J. Nat. Prod. 2008, 71, 615–618. [Google Scholar]
- Strobel, G.; Ford, E.; Worapong, J.; Harper, J.K.; Arif, A.M.; Grant, D.M.; Fung, P.C.W.; Chau, R.M.W. Isopestacin, an isobenzofuranone from Pestalotiopsis microspora, possessing antifungal and antioxidant activities. Phytochemistry 2002, 60, 179–183. [Google Scholar]
- Youla, S.T.; Kelvin, K.O.; Alan, K.W. Phytotoxic metabolites of Phomopsis convolvulus, a host-specific pathogen of field bindweed. Can. J. Chem. 1992, 70, 2276–2284. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lan, W.-J.; Liu, W.; Liang, W.-L.; Xu, Z.; Le, X.; Xu, J.; Lam, C.-K.; Yang, D.-P.; Li, H.-J.; Wang, L.-Y. Pseudaboydins A and B: Novel Isobenzofuranone Derivatives from Marine Fungus Pseudallescheria boydii Associated with Starfish Acanthaster planci. Mar. Drugs 2014, 12, 4188-4199. https://doi.org/10.3390/md12074188
Lan W-J, Liu W, Liang W-L, Xu Z, Le X, Xu J, Lam C-K, Yang D-P, Li H-J, Wang L-Y. Pseudaboydins A and B: Novel Isobenzofuranone Derivatives from Marine Fungus Pseudallescheria boydii Associated with Starfish Acanthaster planci. Marine Drugs. 2014; 12(7):4188-4199. https://doi.org/10.3390/md12074188
Chicago/Turabian StyleLan, Wen-Jian, Wei Liu, Wan-Ling Liang, Zeng Xu, Xiu Le, Jun Xu, Chi-Keung Lam, De-Po Yang, Hou-Jin Li, and Lai-You Wang. 2014. "Pseudaboydins A and B: Novel Isobenzofuranone Derivatives from Marine Fungus Pseudallescheria boydii Associated with Starfish Acanthaster planci" Marine Drugs 12, no. 7: 4188-4199. https://doi.org/10.3390/md12074188
APA StyleLan, W.-J., Liu, W., Liang, W.-L., Xu, Z., Le, X., Xu, J., Lam, C.-K., Yang, D.-P., Li, H.-J., & Wang, L.-Y. (2014). Pseudaboydins A and B: Novel Isobenzofuranone Derivatives from Marine Fungus Pseudallescheria boydii Associated with Starfish Acanthaster planci. Marine Drugs, 12(7), 4188-4199. https://doi.org/10.3390/md12074188





