Adenovirus Carrying Gene Encoding Haliotis discus discus Sialic Acid Binding Lectin Induces Cancer Cell Apoptosis
Abstract
:1. Introduction
2. Results and Discussion
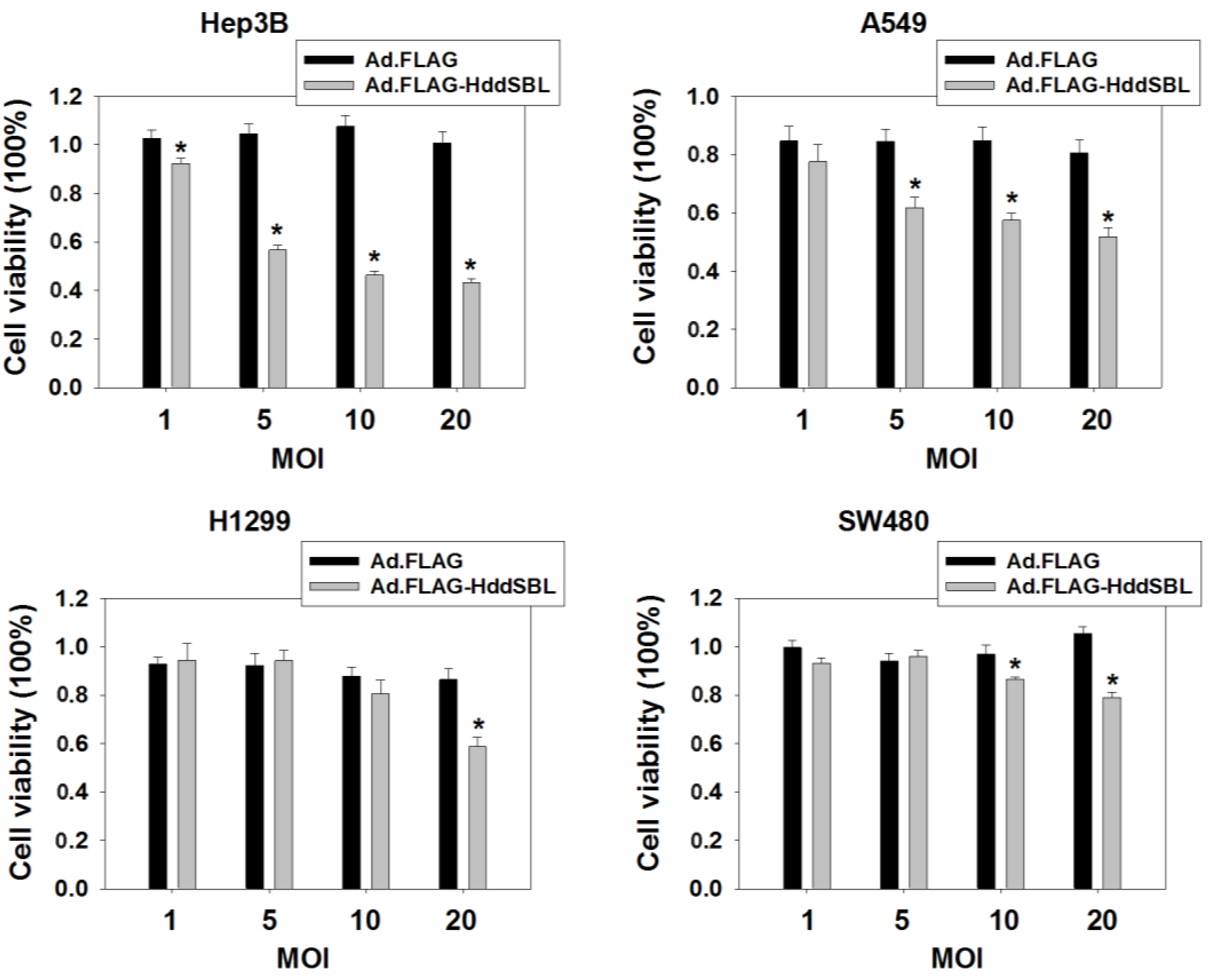
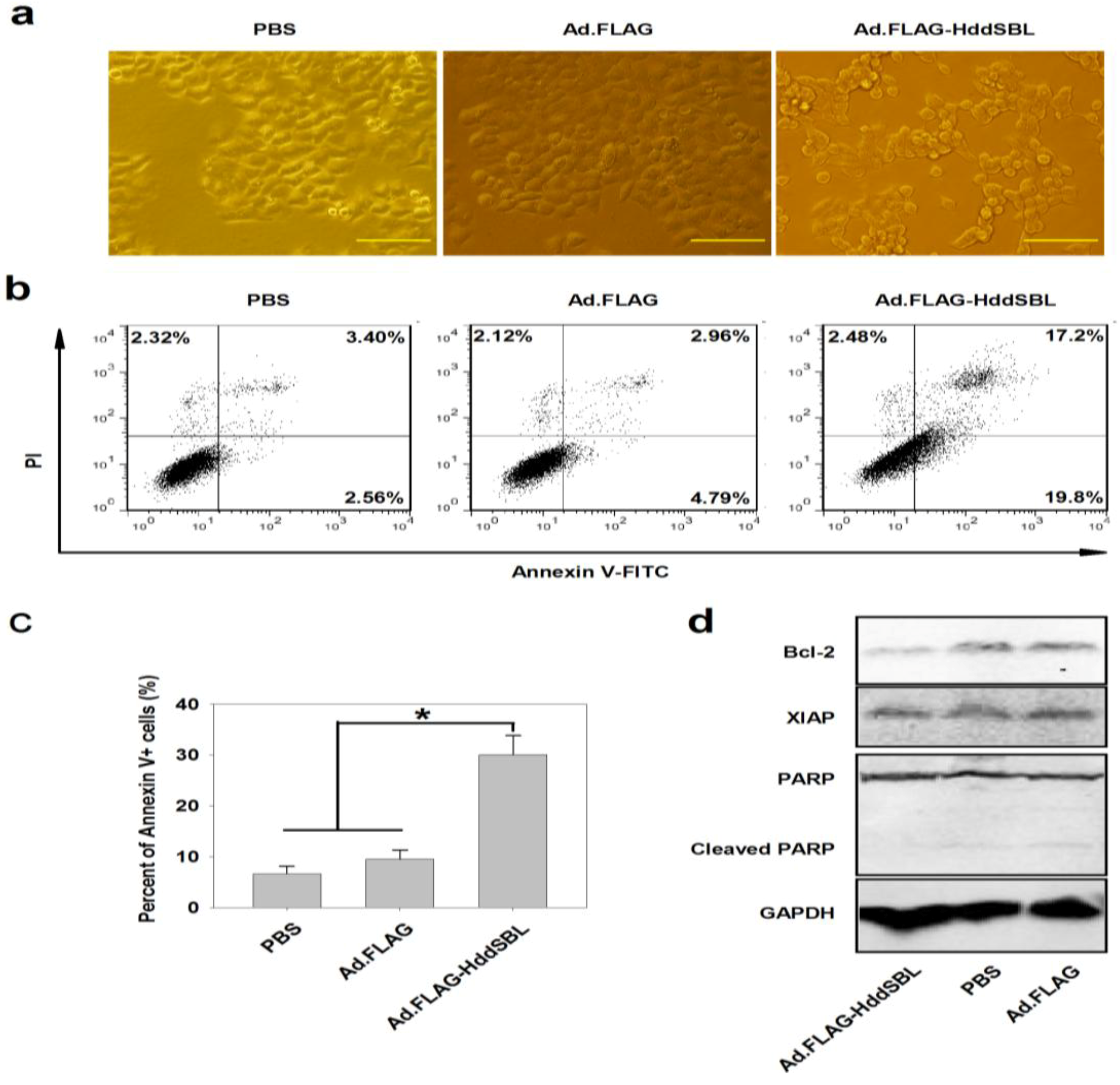
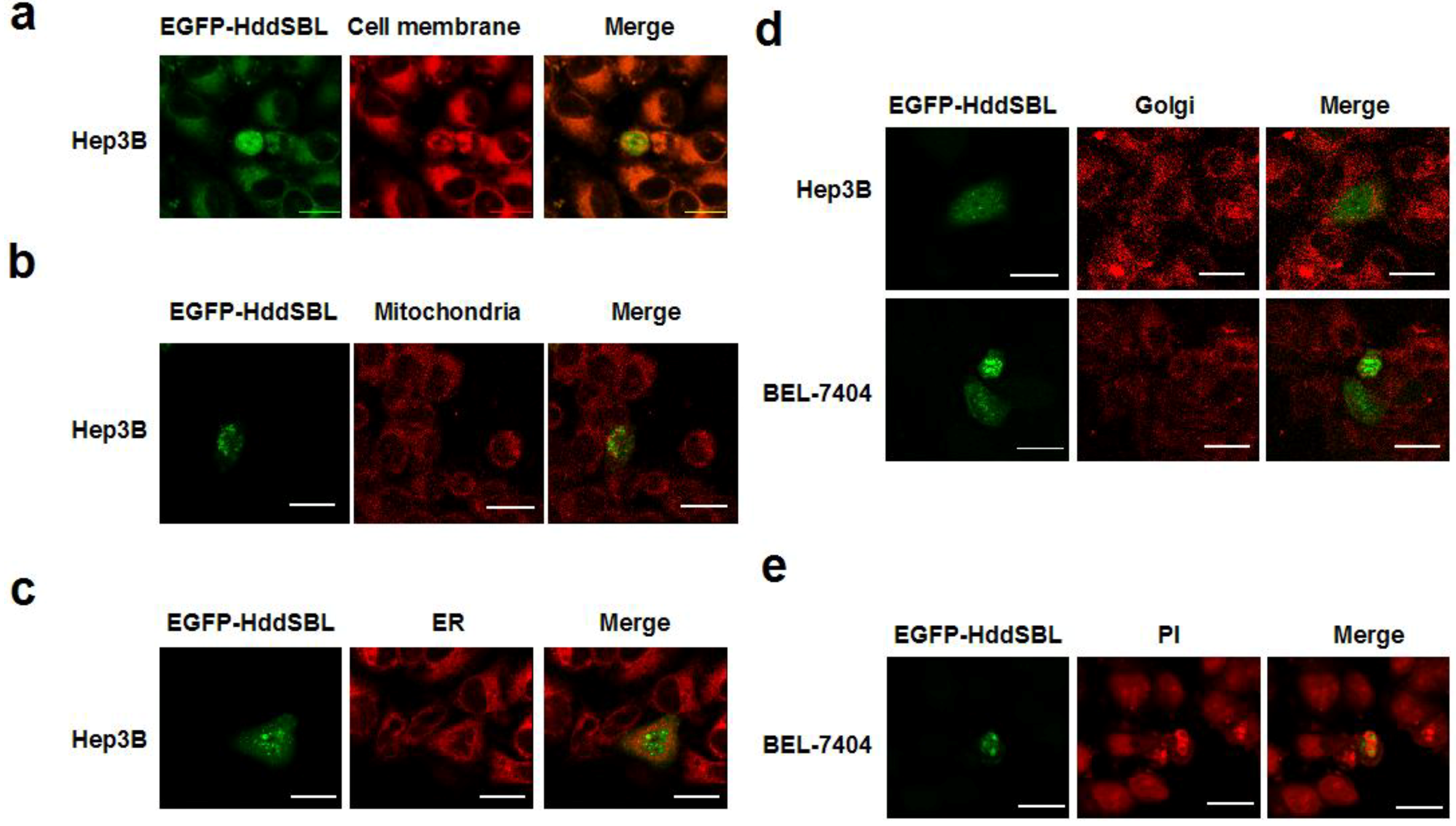
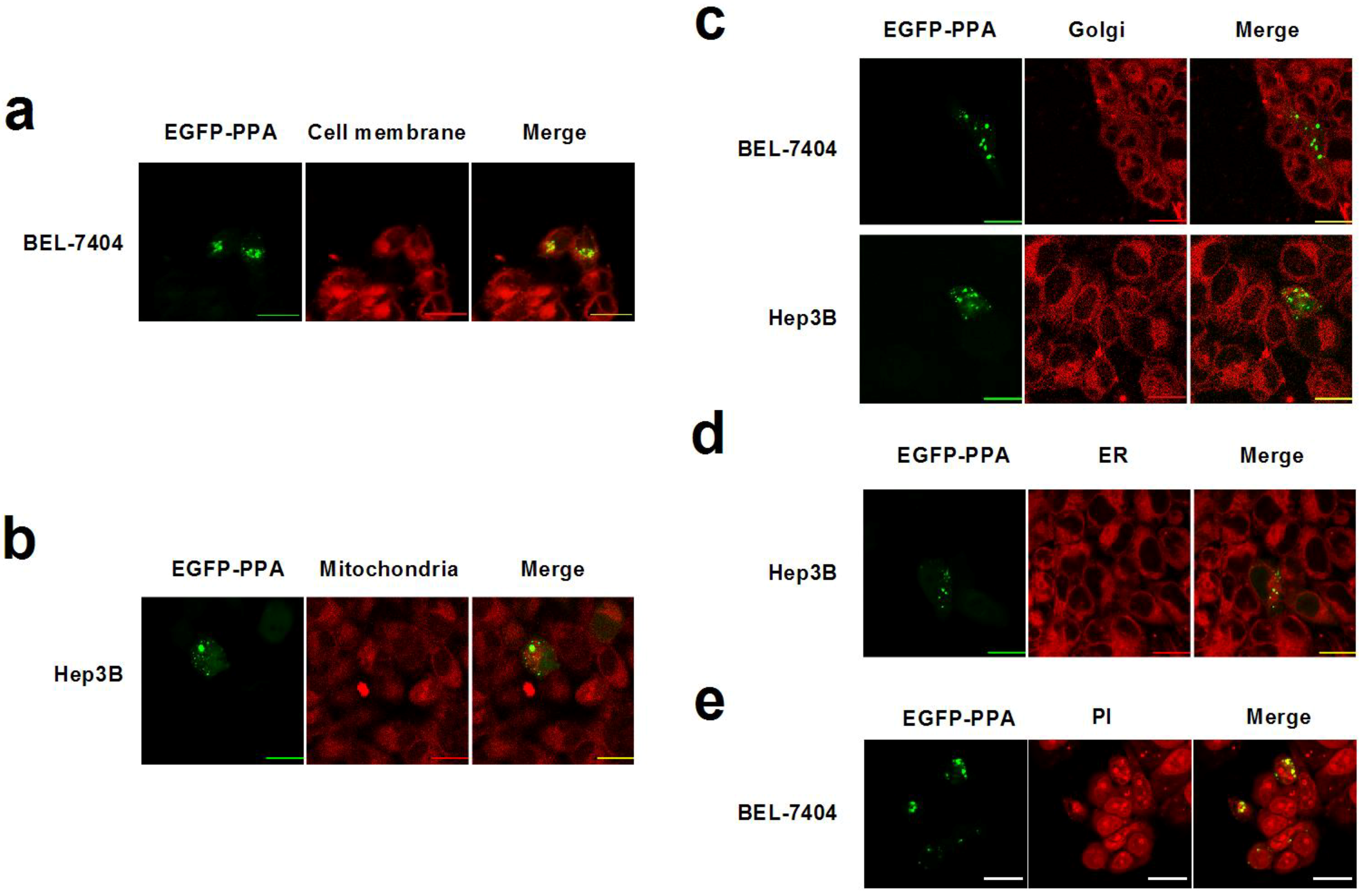
3. Experimental Section
3.1. Cell Culture and Transfection
3.2. Plasmid Construction
3.4. Subcellular Staining and Colocalization Study
3.5. Cytotoxicity Detection and Flow Cytometry Assay
3.6. Western Blotting Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sharon, N.; Lis, H. Lectins as cell recognition molecules. Science 1989, 246, 227–234. [Google Scholar]
- Sharon, N. Lectins: Carbohydrate-specific reagents and biological recognition molecules. J. Biol. Chem. 2007, 282, 2753–2764. [Google Scholar] [CrossRef]
- Ochoa-Alvarez, J.A.; Krishnan, H.; Shen, Y.; Acharya, N.K.; Han, M.; McNulty, D.E.; Hasegawa, H.; Hyodo, T.; Senga, T.; Geng, J.G.; et al. Plant lectin can target receptors containing sialic acid, exemplified by podoplanin, to inhibit transformed cell growth and migration. PLoS One 2012, 7, e41845. [Google Scholar]
- Liu, B.; Min, M.W.; Bao, J.K. Induction of apoptosis by concanavalin a and its molecular mechanisms in cancer cells. Autophagy 2009, 5, 432–433. [Google Scholar] [CrossRef]
- Liu, B.; Cheng, Y.; Bian, H.J.; Bao, J.K. Molecular mechanisms of polygonatum cyrtonema lectin-induced apoptosis and autophagy in cancer cells. Autophagy 2009, 5, 253–255. [Google Scholar] [CrossRef]
- Chen, K.; Yang, X.; Wu, L.; Yu, M.; Li, X.; Li, N.; Wang, S.; Li, G. Pinellia pedatisecta agglutinin targets drug resistant K562/ADR leukemia cells through binding with sarcolemmal membrane associated protein and enhancing macrophage phagocytosis. PLoS One 2013, 8, e74363. [Google Scholar]
- Lu, Q.; Li, N.; Luo, J.; Yu, M.; Huang, Y.; Wu, X.; Wu, H.; Liu, X.Y.; Li, G. Pinellia pedatisecta agglutinin interacts with the methylosome and induces cancer cell death. Oncogenesis 2012, 1, e29. [Google Scholar] [CrossRef]
- Jandus, C.; Simon, H.U.; von Gunten, S. Targeting siglecs—A novel pharmacological strategy for immuno- and glycotherapy. Biochem. Pharmacol. 2011, 82, 323–332. [Google Scholar] [CrossRef]
- O’Neill, A.S.; van den Berg, T.K.; Mullen, G.E. Sialoadhesin—A macrophage-restricted marker of immunoregulation and inflammation. Immunology 2013, 138, 198–207. [Google Scholar] [CrossRef]
- Kopatz, J.; Beutner, C.; Welle, K.; Bodea, L.G.; Reinhardt, J.; Claude, J.; Linnartz-Gerlach, B.; Neumann, H. Siglec-h on activated microglia for recognition and engulfment of glioma cells. Glia 2013, 61, 1122–1133. [Google Scholar] [CrossRef]
- Jellusova, J.; Nitschke, L. Regulation of B cell functions by the sialic acid-binding receptors Siglec-G and CD22. Front Immunol. 2011, 2, 96. [Google Scholar]
- McMillan, S.J.; Sharma, R.S.; McKenzie, E.J.; Richards, H.E.; Zhang, J.; Prescott, A.; Crocker, P.R. Siglec-E is a negative regulator of acute pulmonary neutrophil inflammation and suppresses cd11b beta2-integrin-dependent signaling. Blood 2013, 121, 2084–2094. [Google Scholar]
- Tuscano, J.M.; Kato, J.; Pearson, D.; Xiong, C.; Newell, L.; Ma, Y.; Gandara, D.R.; O’Donnell, R.T. CD22 antigen is broadly expressed on lung cancer cells and is a target for antibody-based therapy. Cancer Res. 2012, 72, 5556–5565. [Google Scholar] [CrossRef]
- Hoelzer, D. Anti-CD22 therapy in acute lymphoblastic leukaemia. Lancet Oncol. 2012, 13, 329–331. [Google Scholar] [CrossRef]
- Klaas, M.; Oetke, C.; Lewis, L.E.; Erwig, L.P.; Heikema, A.P.; Easton, A.; Willison, H.J.; Crocker, P.R. Sialoadhesin promotes rapid proinflammatory and type I Ifn responses to a sialylated pathogen, Campylobacter jejuni. J. Immunol. 2012, 189, 2414–2422. [Google Scholar] [CrossRef]
- Ma, D.Y.; Suthar, M.S.; Kasahara, S.; Gale, M., Jr.; Clark, E.A. CD22 is required for protection against west nile virus infection. J. Virol. 2013, 87, 3361–3375. [Google Scholar] [CrossRef]
- Crocker, P.R.; McMillan, S.J.; Richards, H.E. CD33-related siglecs as potential modulators of inflammatory responses. Ann. N. Y. Acad. Sci. 2012, 1253, 102–111. [Google Scholar]
- Haso, W.; Lee, D.W.; Shah, N.N.; Stetler-Stevenson, M.; Yuan, C.M.; Pastan, I.H.; Dimitrov, D.S.; Morgan, R.A.; FitzGerald, D.J.; Barrett, D.M.; et al. Anti-CD22-chimeric antigen receptors targeting B-cell precursor acute lymphoblastic leukemia. Blood 2013, 121, 1165–1174. [Google Scholar] [CrossRef]
- Schweizer, A.; Wohner, M.; Prescher, H.; Brossmer, R.; Nitschke, L. Targeting of CD22-positive B-cell lymphoma cells by synthetic divalent sialic acid analogues. Eur. J. Immunol. 2012, 42, 2792–2802. [Google Scholar] [CrossRef]
- Herrmann, H.; Cerny-Reiterer, S.; Gleixner, K.V.; Blatt, K.; Herndlhofer, S.; Rabitsch, W.; Jager, E.; Mitterbauer-Hohendanner, G.; Streubel, B.; Selzer, E.; et al. CD34(+)/CD38(−) stem cells in chronic myeloid leukemia express Siglec-3 (CD33) and are responsive to the CD33-targeting drug gemtuzumab/ozogamicin. Haematologica 2012, 97, 219–226. [Google Scholar] [CrossRef]
- Liu, B.; Wu, J.M.; Li, J.; Liu, J.J.; Li, W.W.; Li, C.Y.; Xu, H.L.; Bao, J.K. Polygonatum cyrtonema lectin induces murine fibrosarcoma l929 cell apoptosis and autophagy via blocking ras-raf and pi3k-akt signaling pathways. Biochimie 2010, 92, 1934–1938. [Google Scholar] [CrossRef]
- Lu, C.X.; Nan, K.J.; Lei, Y. Agents from amphibians with anticancer properties. Anticancer Drugs 2008, 19, 931–939. [Google Scholar] [CrossRef]
- Yang, J.; Wei, X.; Liu, X.; Xu, J.; Yang, D.; Fang, J.; Hu, X. Cloning and transcriptional analysis of two sialic acid-binding lectins (SABLs) from razor clam Solen grandis. Fish Shellfish Immunol. 2012, 32, 578–585. [Google Scholar] [CrossRef]
- Li, C.; Yu, S.; Zhao, J.; Su, X.; Li, T. Cloning and characterization of a sialic acid binding lectins (SABL) from manila clam Venerupis philippinarum. Fish Shellfish Immunol. 2011, 30, 1202–1206. [Google Scholar] [CrossRef]
- Tatsuta, T.; Hosono, M.; Sugawara, S.; Kariya, Y.; Ogawa, Y.; Hakomori, S.; Nitta, K. Sialic acid-binding lectin (leczyme) induces caspase-dependent apoptosis-mediated mitochondrial perturbation in jurkat cells. Int. J. Oncol. 2013, 43, 1402–1412. [Google Scholar]
- Tatsuta, T.; Hosono, M.; Miura, Y.; Sugawara, S.; Kariya, Y.; Hakomori, S.; Nitta, K. Involvement of ER stress in apoptosis induced by sialic acid-binding lectin (leczyme) from bullfrog eggs. Int. J. Oncol. 2013, 43, 1799–1808. [Google Scholar]
- Wang, S.Y.; Yu, Q.J.; Bao, J.K.; Liu, B. Polygonatum cyrtonema lectin, a potential antineoplastic drug targeting programmed cell death pathways. Biochem. Biophys. Res. Commun. 2011, 406, 497–500. [Google Scholar] [CrossRef]
- Ohtsubo, K.; Marth, J.D. Glycosylation in cellular mechanisms of health and disease. Cell 2006, 126, 855–867. [Google Scholar]
- Mahon, P.J.; Mirza, A.M.; Iorio, R.M. Role of the two sialic acid binding sites on the newcastle disease virus HN protein in triggering the interaction with the F protein required for the promotion of fusion. J. Virol. 2011, 85, 12079–12082. [Google Scholar] [CrossRef]
- Mistry, N.; Inoue, H.; Jamshidi, F.; Storm, R.J.; Oberste, M.S.; Arnberg, N. Coxsackievirus A24 variant uses sialic acid-containing o-linked glycoconjugates as cellular receptors on human ocular cells. J. Virol. 2011, 85, 11283–11290. [Google Scholar] [CrossRef]
- Oshansky, C.M.; Pickens, J.A.; Bradley, K.C.; Jones, L.P.; Saavedra-Ebner, G.M.; Barber, J.P.; Crabtree, J.M.; Steinhauer, D.A.; Tompkins, S.M.; Tripp, R.A. Avian influenza viruses infect primary human bronchial epithelial cells unconstrained by sialic acid alpha2,3 residues. PLoS One 2011, 6, e21183. [Google Scholar] [CrossRef]
- Leung, H.S.; Li, O.T.; Chan, R.W.; Chan, M.C.; Nicholls, J.M.; Poon, L.L. Entry of influenza a virus with a alpha2,6-linked sialic acid binding preference requires host fibronectin. J. Virol. 2012, 86, 10704–10713. [Google Scholar] [CrossRef]
- Neu, U.; Hengel, H.; Blaum, B.S.; Schowalter, R.M.; Macejak, D.; Gilbert, M.; Wakarchuk, W.W.; Imamura, A.; Ando, H.; Kiso, M.; et al. Structures of merkel cell polyomavirus VP1 complexes define a sialic acid binding site required for infection. PLoS Pathog 2012, 8, e1002738. [Google Scholar] [CrossRef]
- Bassaganas, S.; Perez-Garay, M.; Peracaula, R. Cell surface sialic acid modulates extracellular matrix adhesion and migration in pancreatic adenocarcinoma cells. Pancreas 2014, 43, 109–117. [Google Scholar] [CrossRef]
- Meesmann, H.M.; Fehr, E.M.; Kierschke, S.; Herrmann, M.; Bilyy, R.; Heyder, P.; Blank, N.; Krienke, S.; Lorenz, H.M.; Schiller, M. Decrease of sialic acid residues as an eat-me signal on the surface of apoptotic lymphocytes. J. Cell Sci. 2010, 123, 3347–3356. [Google Scholar] [CrossRef]
- Sorensen, A.L.; Rumjantseva, V.; Nayeb-Hashemi, S.; Clausen, H.; Hartwig, J.H.; Wandall, H.H.; Hoffmeister, K.M. Role of sialic acid for platelet life span: Exposure of beta-galactose results in the rapid clearance of platelets from the circulation by asialoglycoprotein receptor-expressing liver macrophages and hepatocytes. Blood 2009, 114, 1645–1654. [Google Scholar] [CrossRef]
- Kontou, M.; Weidemann, W.; Bork, K.; Horstkorte, R. Beyond glycosylation: Sialic acid precursors act as signaling molecules and are involved in cellular control of differentiation of PC12 cells. Biol. Chem. 2009, 390, 575–579. [Google Scholar]
- Steirer, L.M.; Moe, G.R. An antibody to de-N-acetyl sialic acid containing-polysialic acid identifies an intracellular antigen and induces apoptosis in human cancer cell lines. PLoS One 2011, 6, e27249. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yang, X.; Wu, L.; Duan, X.; Cui, L.; Luo, J.; Li, G. Adenovirus Carrying Gene Encoding Haliotis discus discus Sialic Acid Binding Lectin Induces Cancer Cell Apoptosis. Mar. Drugs 2014, 12, 3994-4004. https://doi.org/10.3390/md12073994
Yang X, Wu L, Duan X, Cui L, Luo J, Li G. Adenovirus Carrying Gene Encoding Haliotis discus discus Sialic Acid Binding Lectin Induces Cancer Cell Apoptosis. Marine Drugs. 2014; 12(7):3994-4004. https://doi.org/10.3390/md12073994
Chicago/Turabian StyleYang, Xinyan, Liqin Wu, Xuemei Duan, Lianzhen Cui, Jingjing Luo, and Gongchu Li. 2014. "Adenovirus Carrying Gene Encoding Haliotis discus discus Sialic Acid Binding Lectin Induces Cancer Cell Apoptosis" Marine Drugs 12, no. 7: 3994-4004. https://doi.org/10.3390/md12073994
APA StyleYang, X., Wu, L., Duan, X., Cui, L., Luo, J., & Li, G. (2014). Adenovirus Carrying Gene Encoding Haliotis discus discus Sialic Acid Binding Lectin Induces Cancer Cell Apoptosis. Marine Drugs, 12(7), 3994-4004. https://doi.org/10.3390/md12073994



