Type II Collagen and Gelatin from Silvertip Shark (Carcharhinus albimarginatus) Cartilage: Isolation, Purification, Physicochemical and Antioxidant Properties
Abstract
:1. Introduction
2. Results and Discussion
2.1. Biochemical Composition of Shark Cartilage
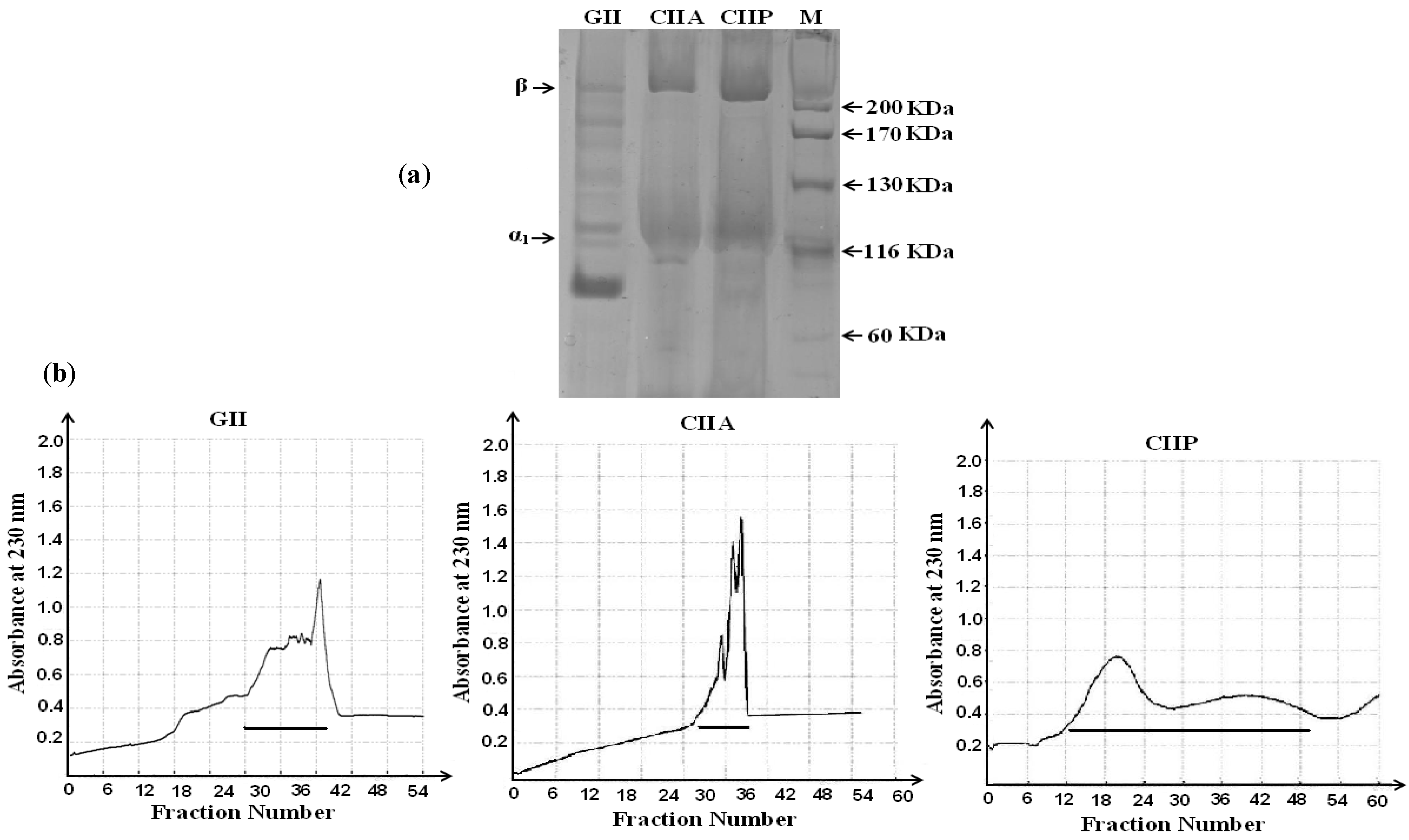
2.2. Protein Pattern
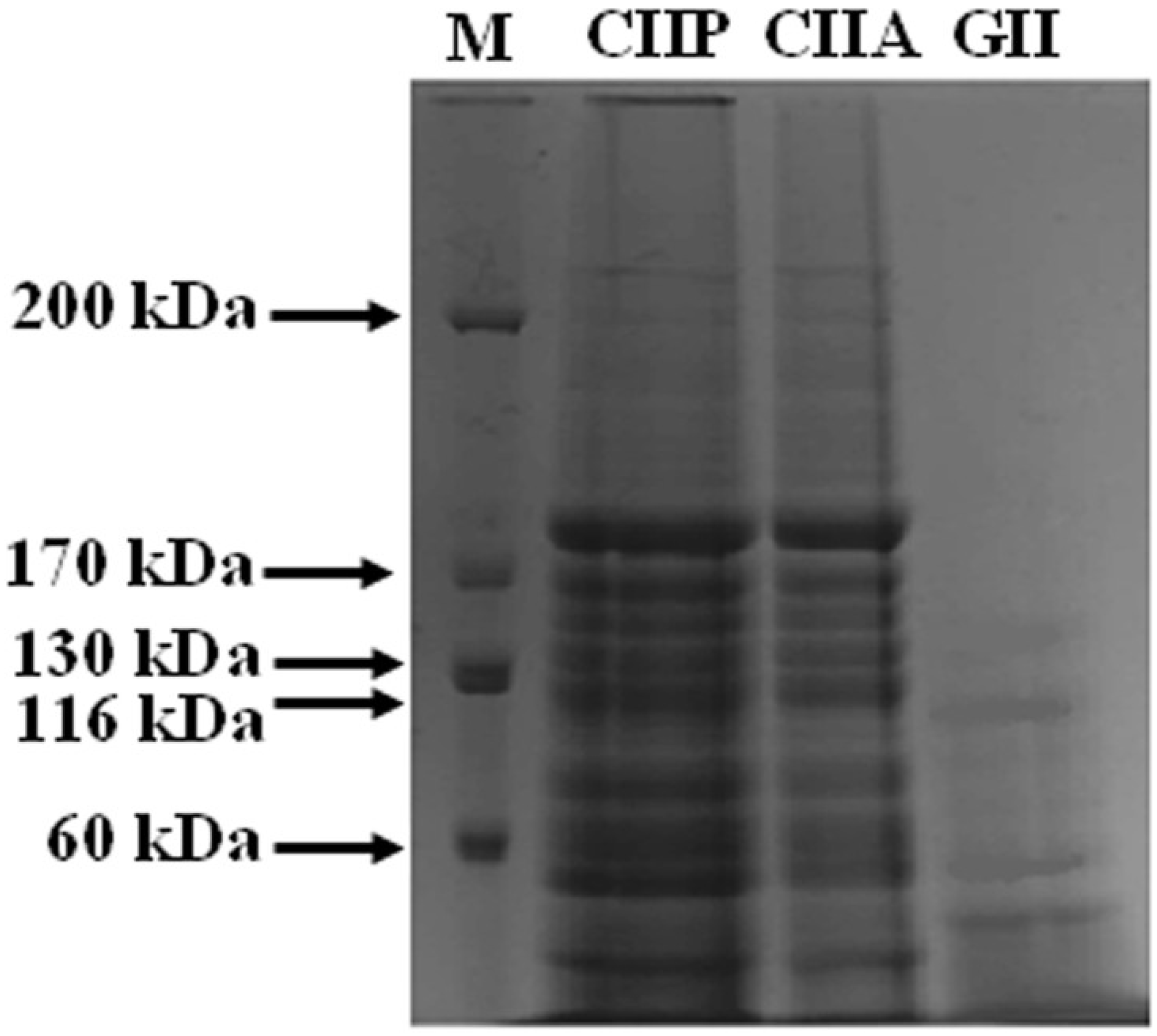
2.3. Peptide Mapping
2.4. Maximum Absorption
2.5. Amino Acid Composition
| Amino Acids | Shark Cartilage | CIIA | CIIP | GII |
|---|---|---|---|---|
| Hyp | 49.96 | 47.50 | 49.25 | 51.28 |
| Asp | 51.84 | 42.91 | 45.76 | 39.84 |
| Thr | 27.26 | 23.54 | 25.57 | 21.85 |
| Ser | 35.62 | 36.37 | 38.27 | 32.62 |
| Glu | 86.45 | 71.57 | 76.20 | 71.70 |
| Pro | 93.61 | 103.30 | 106.78 | 102.31 |
| Gly | 310.47 | 326.90 | 319.69 | 353.12 |
| Ala | 122.87 | 133.92 | 132.63 | 140.45 |
| Cys | 5.88 | 3.93 | 4.26 | 3.74 |
| Val | 25.63 | 25.14 | 25.43 | 22.65 |
| Met | 13.69 | 10.68 | 13.54 | 12.34 |
| Ile | 18.55 | 21.19 | 21.81 | 16.94 |
| Leu | 40.16 | 30.01 | 29.95 | 25.16 |
| Tyr | 15.13 | 8.75 | 7.19 | 2.73 |
| Phe | 19.51 | 18.95 | 14.97 | 14.66 |
| Lys | 22.34 | 30.93 | 29.37 | 27.35 |
| His | 9.98 | 9.33 | 9.38 | 8.15 |
| Arg | 50.96 | 55.05 | 49.87 | 53.14 |
| Total | 1000 | 1000 | 1000 | 1000 |
| Imino acid | 143.58 | 150.81 | 156.03 | 153.59 |
2.6. Relative Viscosity

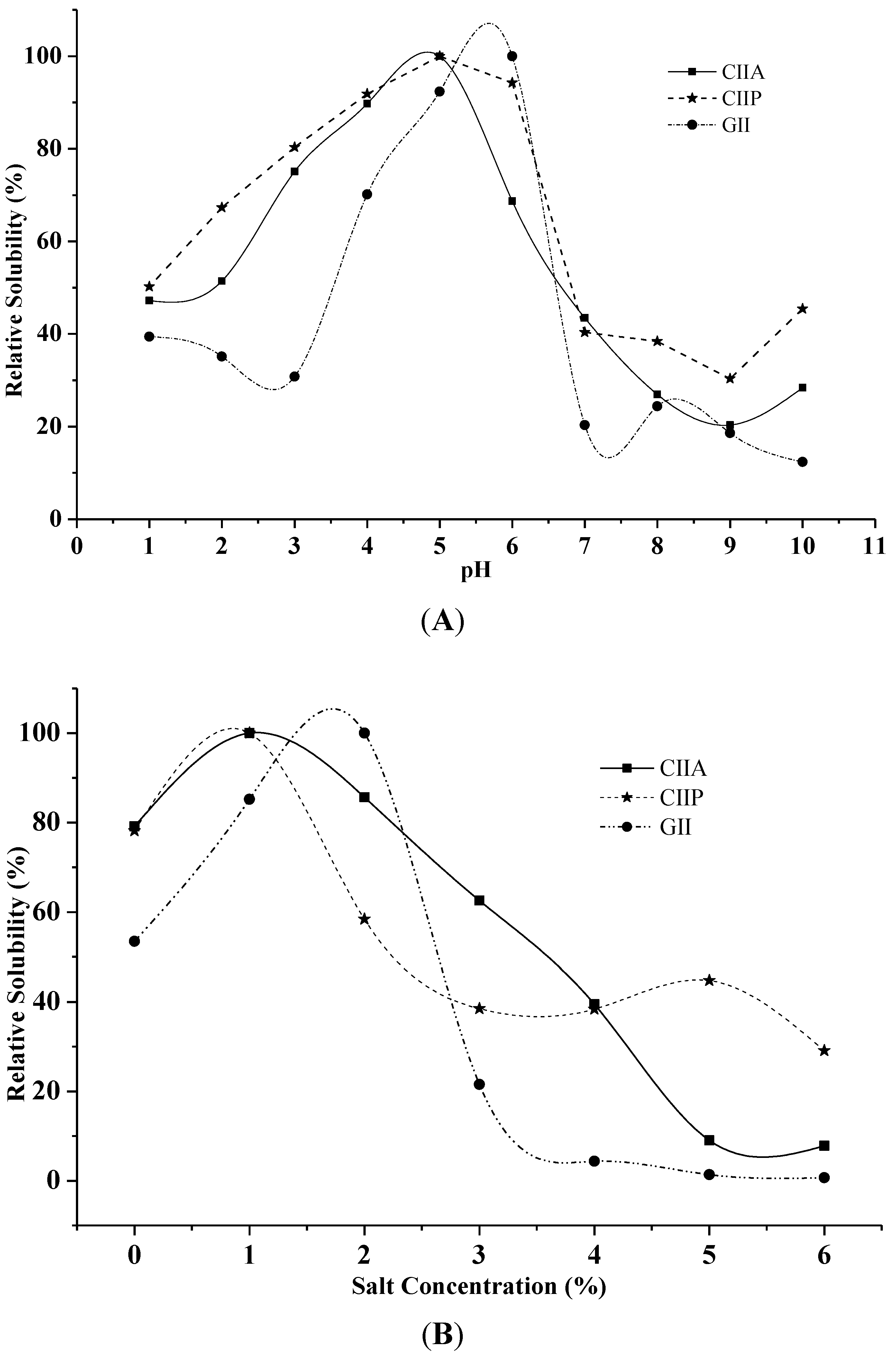
2.7. Effect of pH and NaCl Concentration on Solubility
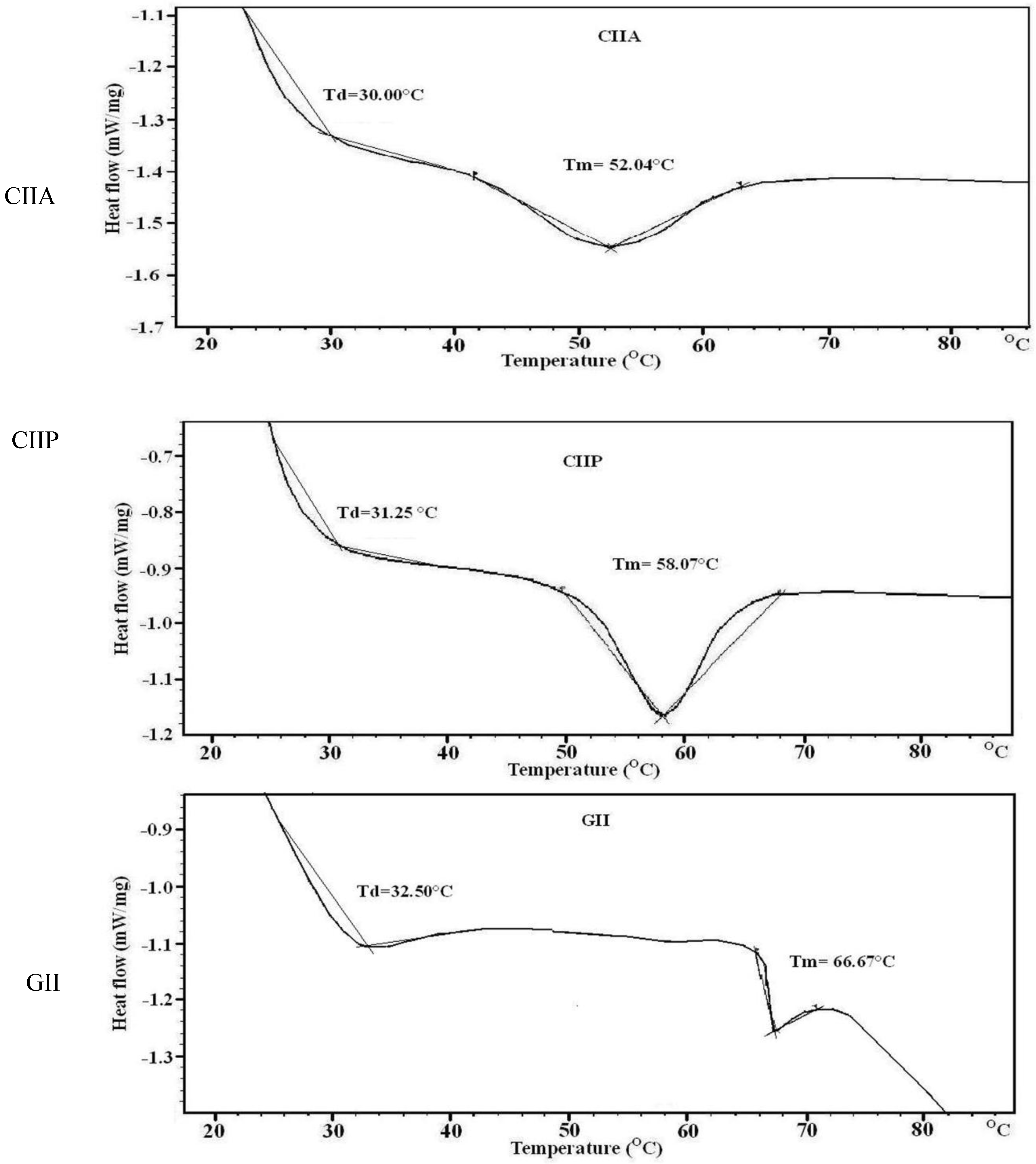
2.8. Thermal Stability
| Sample | DPPH Radical Scavenging (%) | Reducing Power (Absorbance at 700 nm) | DSC | |
|---|---|---|---|---|
| Denaturation Temp (°C) | Melting Temp (°C) | |||
| CIIA | 20.08 | 0.22 | 30.00 | 52.04 |
| CIIP | 24.77 | 0.24 | 31.25 | 58.07 |
| GII | 16.56 | 0.20 | 32.50 | 66.67 |
| BHT | 65.72 | 0.28 | - | - |
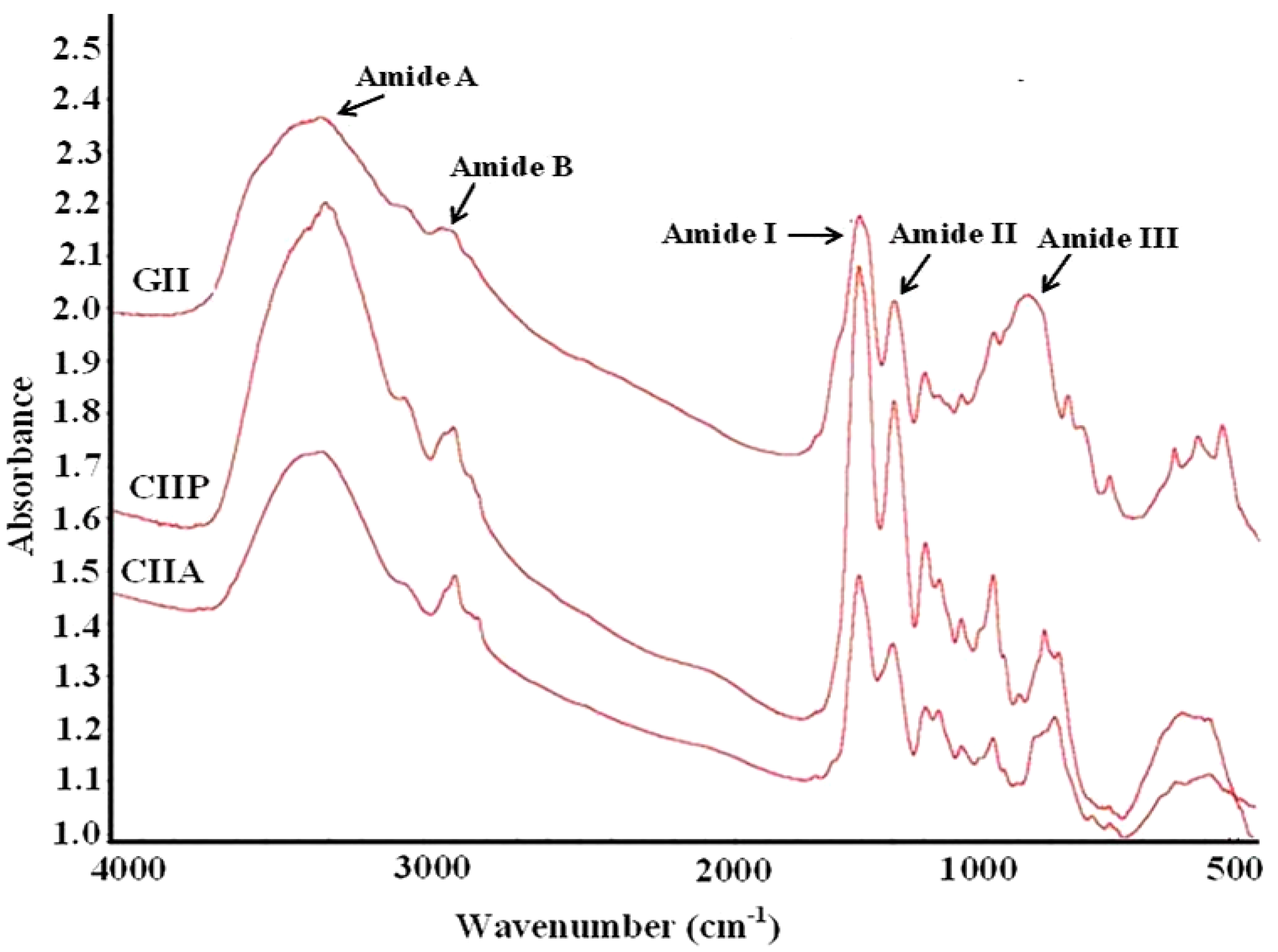
2.9. FTIR Spectra
| Peak Wavenumber (cm−1) | |||
|---|---|---|---|
| CIIP | CIIA | GII | Assignment |
| 3331.13 | 3340.38 | 3350.73 | Amide A: NH stretch coupled with a hydrogen bond |
| 2932.28 | 2927.42 | 2948.13 | Amide B: CH2 asymmetrical stretch |
| 2882.11 | 2855.34 | - | CH3-symmetric stretch: mainly proteins |
| 1796.47 | 1734.76 | - | Carbonyl C=O stretch: lipids |
| 1659.84 | 1659.77 | 1657.77 | Amide I: C=O stretch/hydrogen bond coupled with COO– |
| 1550.67 | 1554.23 | 1551.26 | Amide II: NH bend coupled with an CN stretch |
| 1452.09 | 1452.16 | 1453.79 | CH2 bend |
| 1338.83 | 1338.22 | 1338.47 | CH2 wag of proline |
| 1239.69 | 1240.40 | 1237.81 | Amide III: NH bend coupled with an CN stretch |
| 1157.44 | 1161.1 | 1129.96 | CO-O-C asymmetric stretch: glycogen and nucleic acids |
| 1079.18 | 1079.52 | - | C–O stretch |
| 876.16 | 874.26 | 874.30 | Skeletal stretch |
| 616.96 | 611.58 | 597.11 | Skeletal stretch |
2.10. CD Spectra
 type II pepsin soluble collagen;
type II pepsin soluble collagen;  type II acid soluble collagen;
type II acid soluble collagen;  GII-type II gelatin.
GII-type II gelatin.
 type II pepsin soluble collagen;
type II pepsin soluble collagen;  type II acid soluble collagen;
type II acid soluble collagen;  GII-type II gelatin.
GII-type II gelatin.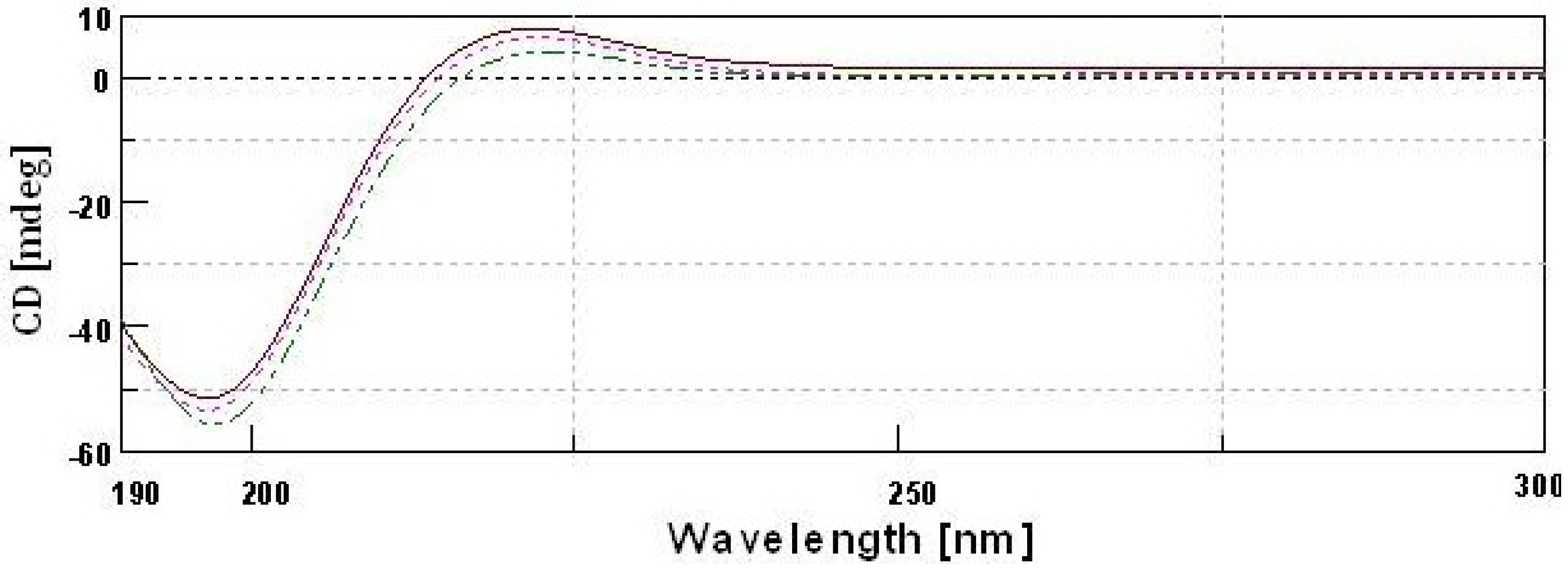
2.11. Antioxidant Activity
2.12. Microscopic Structure
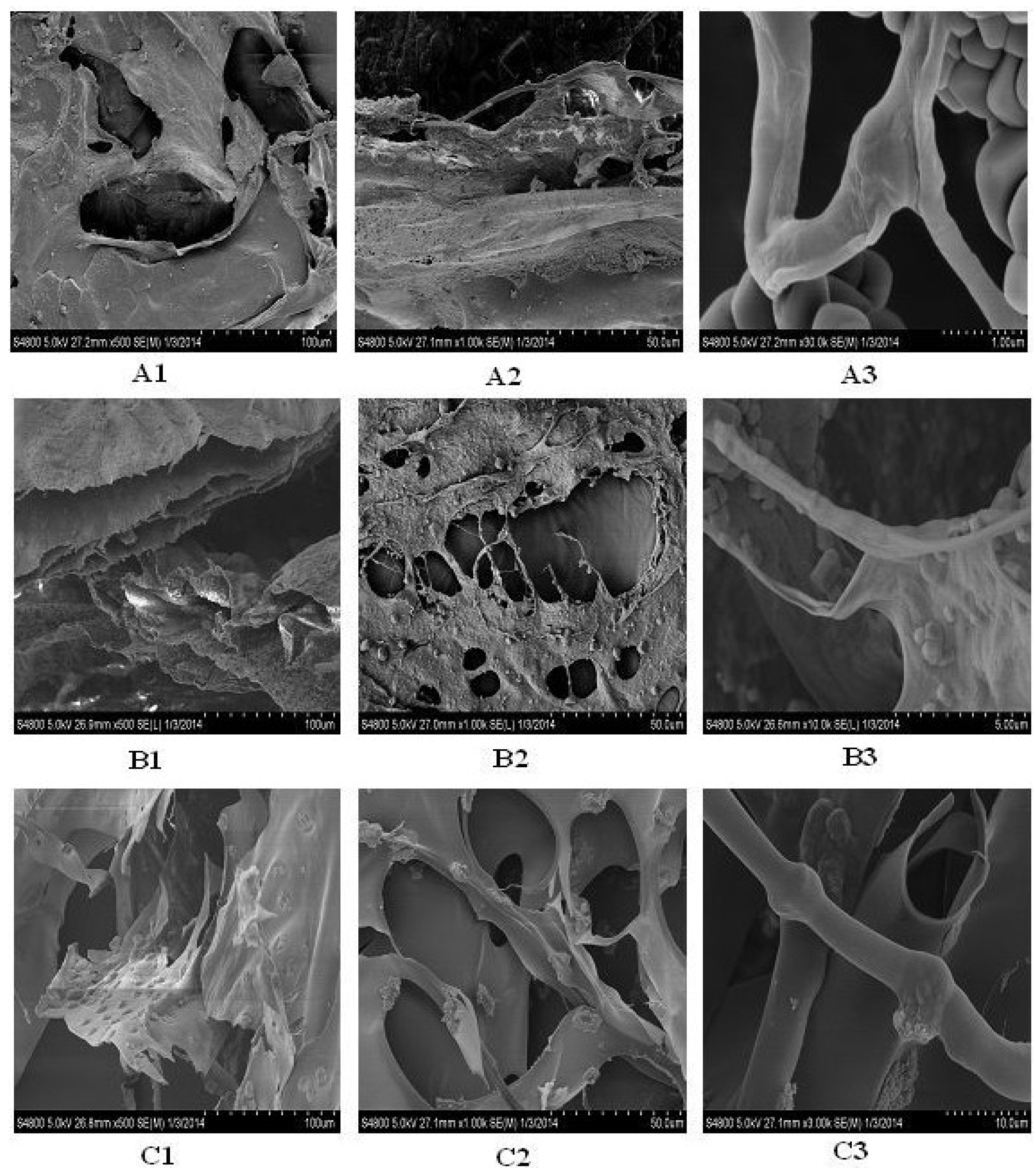
3. Experimental Section
3.1. Raw Materials
3.2. Isolation of Collagen and Gelatin
3.2.1. Pretreatment of Shark Cartilage
3.2.2. Isolation of ASC and PSC
3.2.3. Isolation of Gelatin
3.3. Purification by Gel Filtration Chromatography
3.4. Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis (SDS-PAGE)
3.5. Peptide Mapping
3.6. Viscosity
3.7. Collagen Solubility Test
3.7.1. Effect of pH
3.7.2. Effect of NaCl
3.8. UV Absorption Spectrum
3.9. Amino Acid Profiling
3.10. Fourier Transform Infrared Spectroscopy (FTIR)
3.11. Circular Dichroism (CD)
3.12. Scanning Electron Microscopy (SEM)
3.13. Thermal Stability
3.14. Antioxidant Activity
3.14.1. DPPH Radical Scavenging Assay
3.14.2. Reducing Power
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Poole, A.R. Cartilage in Health and Disease. In Arthritis and Allied Conditions: A Textbook of Rheumatology, 15th ed.; Koopman, W.J., Moreland, L.W., Eds.; Williams and Wilkins: Baltimore, MD, USA, 2005; pp. 223–269. [Google Scholar]
- Ogawa, M.; Moody, M.W.; Portier, R.J.; Bell, J.; Schexnayder, M.A.; Losso, J.N. Biochemical properties of black drum and sheepshead seabream skin collagen. J. Agric. Food Chem. 2003, 51, 8088–8092. [Google Scholar]
- Jeya Shakila, R.; Jeevithan, E.; Varatharajakumar, A.; Jeyasekaran, G.; Sukumar, D. Functional characterization of gelatin extracted from bones of red snapper and grouper in comparison with mammalian gelatin. LWT Food. Sci. Technol. 2012, 48, 30–36. [Google Scholar] [CrossRef]
- Jeevithan, E.; Jeya Shakila, R.; Varatharajakumar, A.; Jeyasekaran, G.; Sukumar, D. Physico- functional and mechanical properties of chitosan and calcium salts incorporated fish gelatin scaffolds. Int. J. Biol. Macromol. 2013, 60, 262–267. [Google Scholar] [CrossRef]
- Chirita, M. Mechanical properties of collagen biomimetic films formed in the presence of calcium, silica and chitosan. J. Bionic Eng. 2008, 5, 149–158. [Google Scholar] [CrossRef]
- Li, Z.R.; Wang, B.; Chi, C.F.; Zhang, Q.H.; Gong, Y.D.; Tang, J.J.; Luo, H.Y.; Ding, G.F. Isolation and characterization of acid soluble collagens and pepsin soluble collagens from the skin and bone of Spanish mackerel (Scomberomorous niphonius). Food Hydrocoll. 2013, 31, 103–113. [Google Scholar] [CrossRef]
- Chen, L.; Bao, B.; Wang, N.; Xie, J.; Wu, W.H. Oral Administration of Shark Type II Collagen Suppresses Complete Freund’s Adjuvant-Induced Rheumatoid Arthritis in Rats. Pharmaceuticals 2012, 5, 339–352. [Google Scholar]
- Zhao, W.; Tong, T.; Wang, L.; Li, P.P.; Chang, Y.; Zhang, L.L.; Wei, W. Chicken type II collagen induced immune tolerance of mesenteric lymph node lymphocytes by enhancing beta2-adrenergic receptor desensitization in rats with collagen-induced arthritis. Int. Immunopharmacol. 2011, 11, 12–18. [Google Scholar] [CrossRef]
- Zhu, P.; Li, X.Y.; Wang, H.K.; Jia, J.F.; Zheng, Z.H.; Ding, J.; Fan, C.M. Oral administration of type-II collagen peptide 250–270 suppresses specific cellular and humoral immune response in collagen-induced arthritis. Clin. Immunol. 2007, 122, 75–84. [Google Scholar]
- Garcia, G.; Komagata, Y.; Slavin, A.J.; Maron, R.; Weiner, H.L. Suppression of collagen-induced arthritis by oral or nasal administration of type II collagen. J. Autoimmun. 1999, 13, 315–324. [Google Scholar] [CrossRef]
- Youn, J.; Hwang, S.H.; Ryoo, Z.Y.; Lynes, M.A.; Paik, D.J.; Chung, H.S.; Kim, H.Y. Metallothionein suppresses collagen-induced arthritis via induction of TGF-b and down-regulation of proinflammatory mediators. Clin. Exp. Immunol. 2002, 129, 232–239. [Google Scholar] [CrossRef]
- Zhong, Y.; Khan, M.A.; Shahidi, F. Compositional characteristics and antioxidant properties of Fresh and Processed Sea Cucumber (Cucumaria frondosa). J. Agric. Food Chem. 2007, 55, 1188–1192. [Google Scholar] [CrossRef]
- Zhu, B.; Dong, X.; Zhou, D.; Gao, Y.; Yang, J.; Li, D.; Zhao, X.; Rena, T.; Yea, W.; Tana, H.; Wua, H.; Yu, C. Physicochemical properties and radical scavenging capacities of pepsin-solubilized collagen from sea cucumber (Stichopus japonicas). Food Hydrocoll. 2012, 28, 182–188. [Google Scholar] [CrossRef]
- Zhuang, Y.; Zhao, X.; Li, B. Optimization of antioxidant activity by response surface methodology in hydrolysates of jellyfish (Rhopilema esculentum) umbrella collagen. J. Zhejiang Univ. Sci. B 2009, 10, 572–579. [Google Scholar] [CrossRef]
- Mendis, E.; Rajapakse, N.; Byun, H.G.; Kim, S.K. Investigation of jumbo squid (Dosidicus gigas) skin gelatin peptides for their in vitro antioxidant effects. Life Sci. 2005, 77, 2166–2178. [Google Scholar] [CrossRef]
- Je, J.Y.; Qian, Z.J.; Byun, H.G.; Kim, S.K. Purification and characterization of an antioxidant peptide obtained from tuna backbone protein by enzymatic hydrolysis. Process Biochem. 2007, 42, 840–846. [Google Scholar] [CrossRef]
- Merly, L.; Smith, S.L. Collagen type II, alpha 1 protein: a bioactive component of shark cartilage. Int. Immunopharmacol. 2013, 15, 309–315. [Google Scholar] [CrossRef]
- Liang, Q.; Wang, L.; Sun, W.; Wang, Z.; Xu, J.; Ma, H. Isolation and characterization of collagen from the cartilage of Amur sturgeon (Acipenser schrenckii). Process Biochem. 2014, 49, 318–323. [Google Scholar] [CrossRef]
- Muyonga, J.H.; Cole, C.G.B.; Duodu, K.G. Characterisation of acid soluble collagen from skins of young and adult Nile perch (Lates niloticus). Food Chem. 2004, 85, 81–89. [Google Scholar] [CrossRef]
- Cho, S.M.; Kwak, K.S.; Park, D.C.; Gu, Y.S.; Ji, C.I.; Jang, D.H.; Lee, Y.B.; Kim, S.B. Processing optimization and functional properties of gelatin from shark (Isurus oxyrinchus) cartilage. Food Hydrocoll. 2004, 18, 573–579. [Google Scholar] [CrossRef]
- Kittiphattanabawon, P.; Benjakul, S.; Visessanguan, W.; Shahidi, F. Isolation and characterization of collagen from the cartilages of brownbanded bamboo shark (Chiloscyllium punctatum) and blacktip shark (Carcharhinus limbatus). LWT Food Sci. Technol. 2010, 43, 792–800. [Google Scholar] [CrossRef]
- Cao, H.; Shi, F.X.; Xu, F.; Yu, J.S. Molecular structure and physicochemical properties of pepsin-solubilized type II collagen from the chick sternal cartilage. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 1427–1437. [Google Scholar]
- Helen, E.; Cahir, A. Spatial organization of type I and II collagen in the canine meniscus. J. Orthop. Res. 2005, 23, 142–149. [Google Scholar] [CrossRef]
- Vercaigne-Marko, D.; Kosciarz, E.; Nedjar-Arroume, N.; Guillochon, D. Improvement of Staphylococcus aureus-V8-protease hydrolysis of bovine haemoglobin by its adsorption on to a solid phase in the presence of SDS: peptide mapping and obtention of two haemopoietic peptides. Biotechnol. Appl. Biochem. 2000, 31, 127–134. [Google Scholar]
- Jongjareonrak, A.; Benjakul, S.; Visessanguan, W.; Nagai, T.; Tanaka, M. Isolation and characterisation of acid and pepsin-solubilised collagens from the skin of brownstripe red snapper (Lutjanus vitta). Food Chem. 2005, 93, 475–484. [Google Scholar] [CrossRef]
- Veeruraj, A.; Arumugam, M.; Balasubramanian, T. Isolation and characterization of thermostable collagen from the marine eel-fish (Evenchelys macrura). Process Biochem. 2013, 48, 1592–1602. [Google Scholar] [CrossRef]
- Duan, R.; Zhang, J.; Du, X.; Yao, X.; Konno, K. Properties of collagen from skin, scale and bone of carp (Cyprinus carpio). Food Chem. 2009, 112, 702–706. [Google Scholar] [CrossRef]
- Kittiphattanabawon, P.; Benjakul, S.; Visessanguan, W.; Nagai, T.; Tanaka, M. Characterisation of acid-soluble collagen from skin and bone of bigeye snapper (Priacanthus tayenus). Food Chem. 2005, 89, 363–372. [Google Scholar] [CrossRef]
- Bae, I.; Osatomi, K.; Yoshida, A.; Osako, K.; Yamaguchi, A.; Hara, K. Biochemical properties of acid-soluble collagens extracted from the skins of underutilized fishes. Food Chem. 2008, 108, 49–54. [Google Scholar] [CrossRef]
- Woo, J.W.; Yu, S.J.; Cho, S.M.; Lee, Y.B.; Kim, S.B. Extraction optimization and properties of collagen from yellowfin tuna (Thunnus albacares) dorsal skin. Food Hydrocoll. 2008, 22, 879–887. [Google Scholar] [CrossRef]
- Wang, L.; Liang, Q.; Chen, T.; Wang, Z.; Xu, J.; Ma, H. Characterization of collagen from the skin of Amur sturgeon (Acipenser schrenckii). Food Hydrocoll. 2014, 38, 104–109. [Google Scholar] [CrossRef]
- Ikoma, T.; Kobayashi, H.; Tanaka, J.; Walsh, D.; Mann, S. Physical properties of type I collagen extracted from fish scales of Pagrus major and Oreochromis niloticas. Int. J. Biol. Macromol. 2003, 32, 199–204. [Google Scholar] [CrossRef]
- Usha, R.; Ramasami, T. Structure and conformation of intramolecularly cross-linked collagen. Colloids Surf. B 2005, 41, 21–24. [Google Scholar] [CrossRef]
- Aewsiri, T.; Benjakul, S.; Visessanguan, W.; Wierenga, P.A.; Gruppen, H. Improvement of foaming properties of cuttlefish skin gelatin by modification with N-hydroxysuccinimide esters of fatty acid. Food Hydrocoll. 2011, 25, 1277–1284. [Google Scholar] [CrossRef]
- Nikoo, M.; Benjakul, S.; Bashari, M.; Alekhorshied, M.; Cissouma, A.I.; Yanga, N.; Xu, X. Physicochemical properties of skin gelatin from farmed Amur sturgeon (Acipenser schrenckii) as influenced by acid pretreatment. Food Biosci. 2014, 5, 19–26. [Google Scholar] [CrossRef]
- Chang, C.Y.; Wu, K.C.; Chiang, S.H. Antioxidant properties and protein compositions of porcine haemoglobin hydrolysates. Food Chem. 2007, 100, 1537–1543. [Google Scholar] [CrossRef]
- Tang, C.H.; Peng, J.; Zhen, D.W.; Chen, Z. Physicochemical and antioxidant properties of buckwheat (Fagopyrum esculentum Moench) protein hydrolysates. Food Chem. 2009, 115, 672–678. [Google Scholar] [CrossRef]
- Aszodi, A.; Hunziker, E.B.; Olsen, B.R.; Fassler, R. The role of collagen II and cartilage fibril-associated molecules in skeletal development. Osteoarthr. Cartil. 2001, 9, 150–159. [Google Scholar]
- Joseph, G.; Jun, O.; Teruo, M. Biodegradable honeycomb collagen scaffold for dermal tissue engineering. J. Biomed. Mater. Res. Part A 2008, 87, 1103–1111. [Google Scholar]
- Jansson, K.; Haegerstrand, A.; Kratz, G. A biodegradable bovine collagen membrane as a dermal template for human in vivo wound healing. Scandinavian J. Plast. Reconstr. Surg. Hand Surg. 2001, 35, 369–375. [Google Scholar]
- Zhang, Y.; Liu, W.T.; Li, G.Y.; Shi, B.; Miao, Y.Q.; Wu, X.H. Isolation and partial characterization of pepsin-soluble collagen from the skin of grass carp (Ctenopharyngodon idella). Food Chem. 2007, 103, 906–912. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis; Association of Official Ananlytical Chemists Inc.: Arlington, VA, USA, 2000. [Google Scholar]
- Bergman, I.; Loxley, R. Two improved and simplified methods for the spectrophotometric determination of hydroxyproline. Anal. Chem. 1963, 35, 1961–1965. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with Folin phenol reagent. J. Biol. Chem. 1951, 193, 256–275. [Google Scholar]
- Rochdi, A.; Foucat, L.; Renou, J.P. NMR and DSC studies during thermal denaturation of collagen. Food Chem. 2000, 69, 295–299. [Google Scholar] [CrossRef]
- Shimada, K.; Fujikawa, K.; Yahara, K.; Nakamura, T. Antioxidative properties of xanthan on the autoxidation of soybean oil in cycloextrin emulsion. J. Agric. Food Chem. 1992, 40, 945–948. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jeevithan, E.; Bao, B.; Bu, Y.; Zhou, Y.; Zhao, Q.; Wu, W. Type II Collagen and Gelatin from Silvertip Shark (Carcharhinus albimarginatus) Cartilage: Isolation, Purification, Physicochemical and Antioxidant Properties. Mar. Drugs 2014, 12, 3852-3873. https://doi.org/10.3390/md12073852
Jeevithan E, Bao B, Bu Y, Zhou Y, Zhao Q, Wu W. Type II Collagen and Gelatin from Silvertip Shark (Carcharhinus albimarginatus) Cartilage: Isolation, Purification, Physicochemical and Antioxidant Properties. Marine Drugs. 2014; 12(7):3852-3873. https://doi.org/10.3390/md12073852
Chicago/Turabian StyleJeevithan, Elango, Bin Bao, Yongshi Bu, Yu Zhou, Qingbo Zhao, and Wenhui Wu. 2014. "Type II Collagen and Gelatin from Silvertip Shark (Carcharhinus albimarginatus) Cartilage: Isolation, Purification, Physicochemical and Antioxidant Properties" Marine Drugs 12, no. 7: 3852-3873. https://doi.org/10.3390/md12073852
APA StyleJeevithan, E., Bao, B., Bu, Y., Zhou, Y., Zhao, Q., & Wu, W. (2014). Type II Collagen and Gelatin from Silvertip Shark (Carcharhinus albimarginatus) Cartilage: Isolation, Purification, Physicochemical and Antioxidant Properties. Marine Drugs, 12(7), 3852-3873. https://doi.org/10.3390/md12073852








