New Benzoxazine Secondary Metabolites from an Arctic Actinomycete
Abstract
:1. Introduction
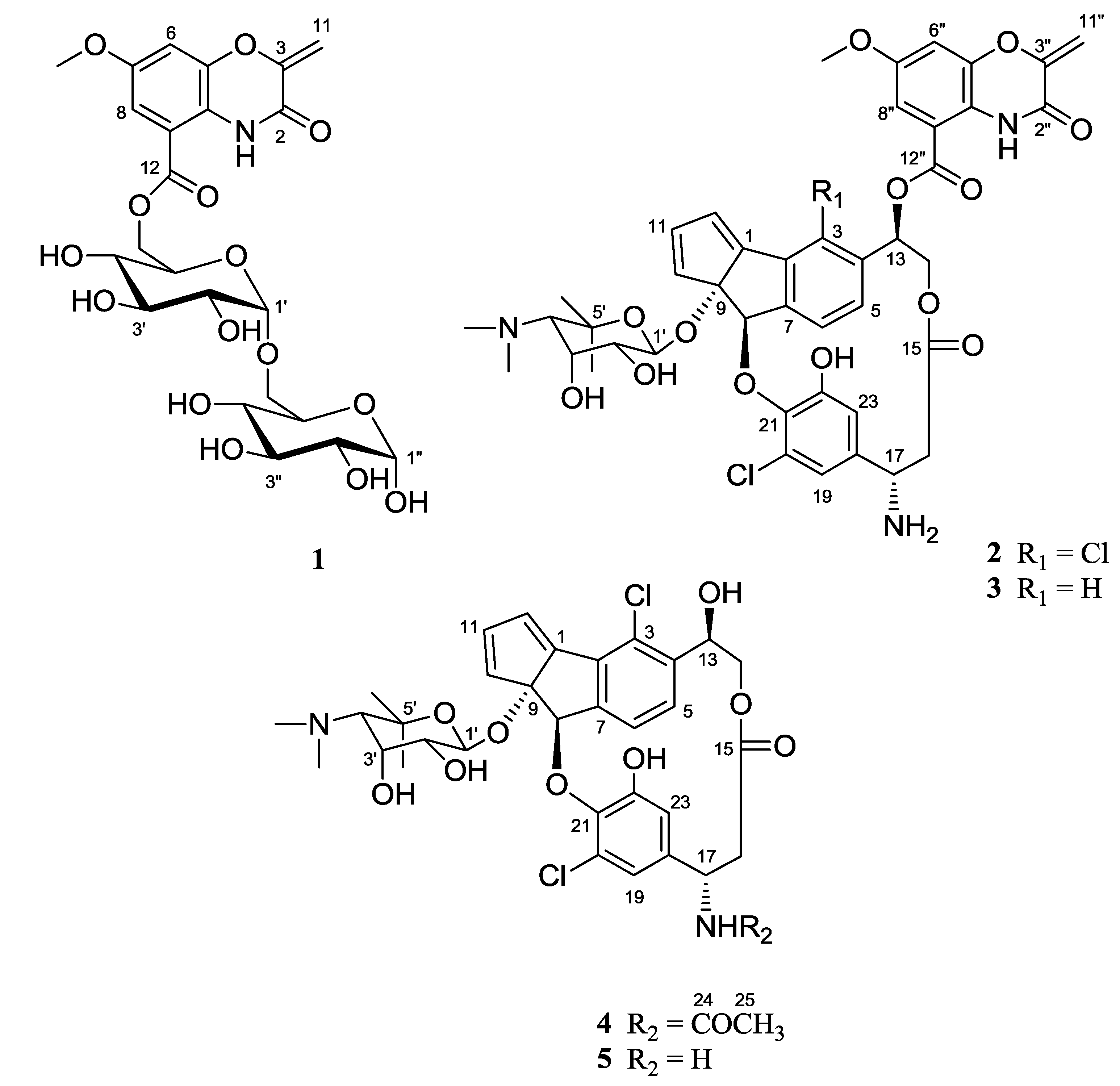
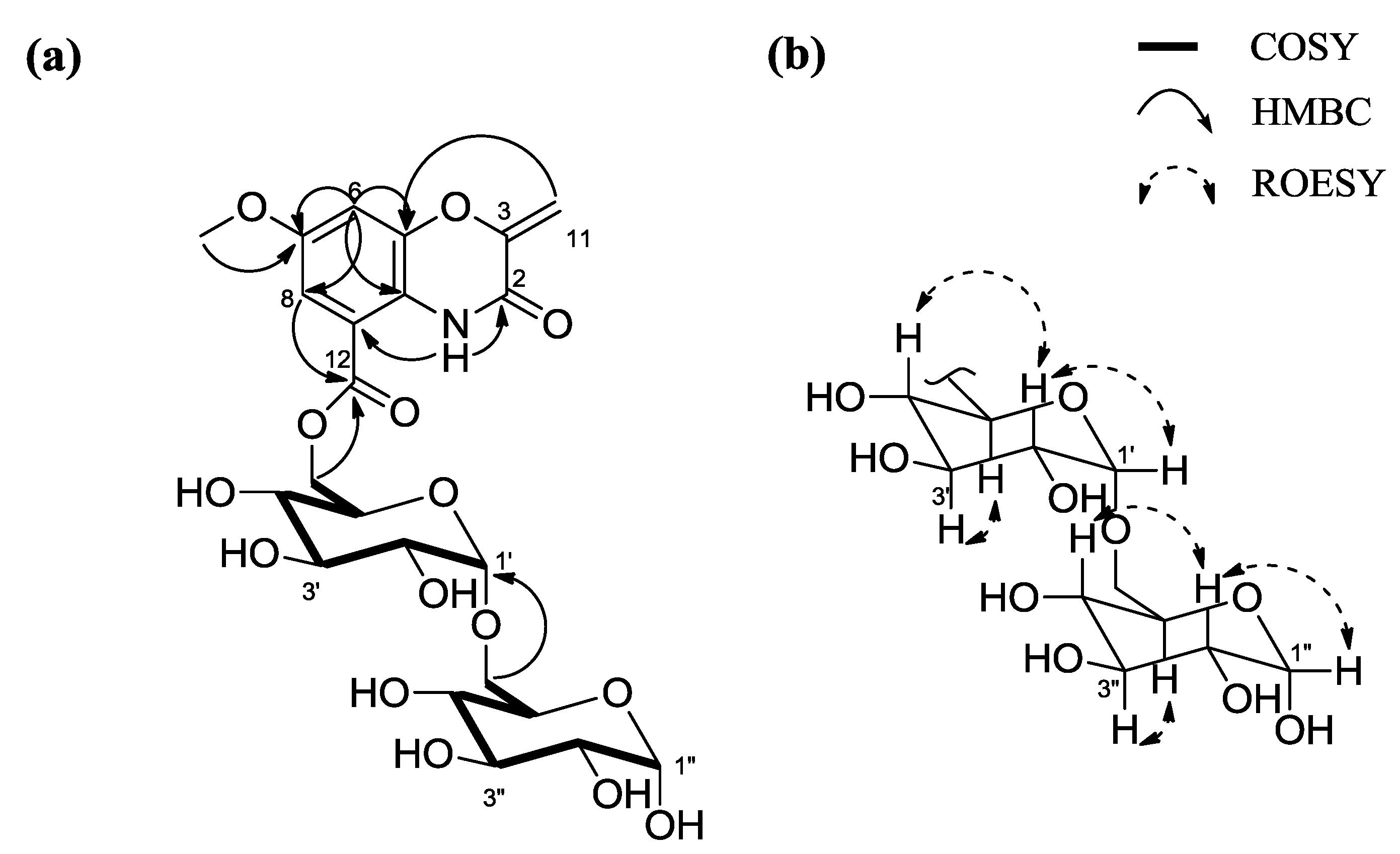
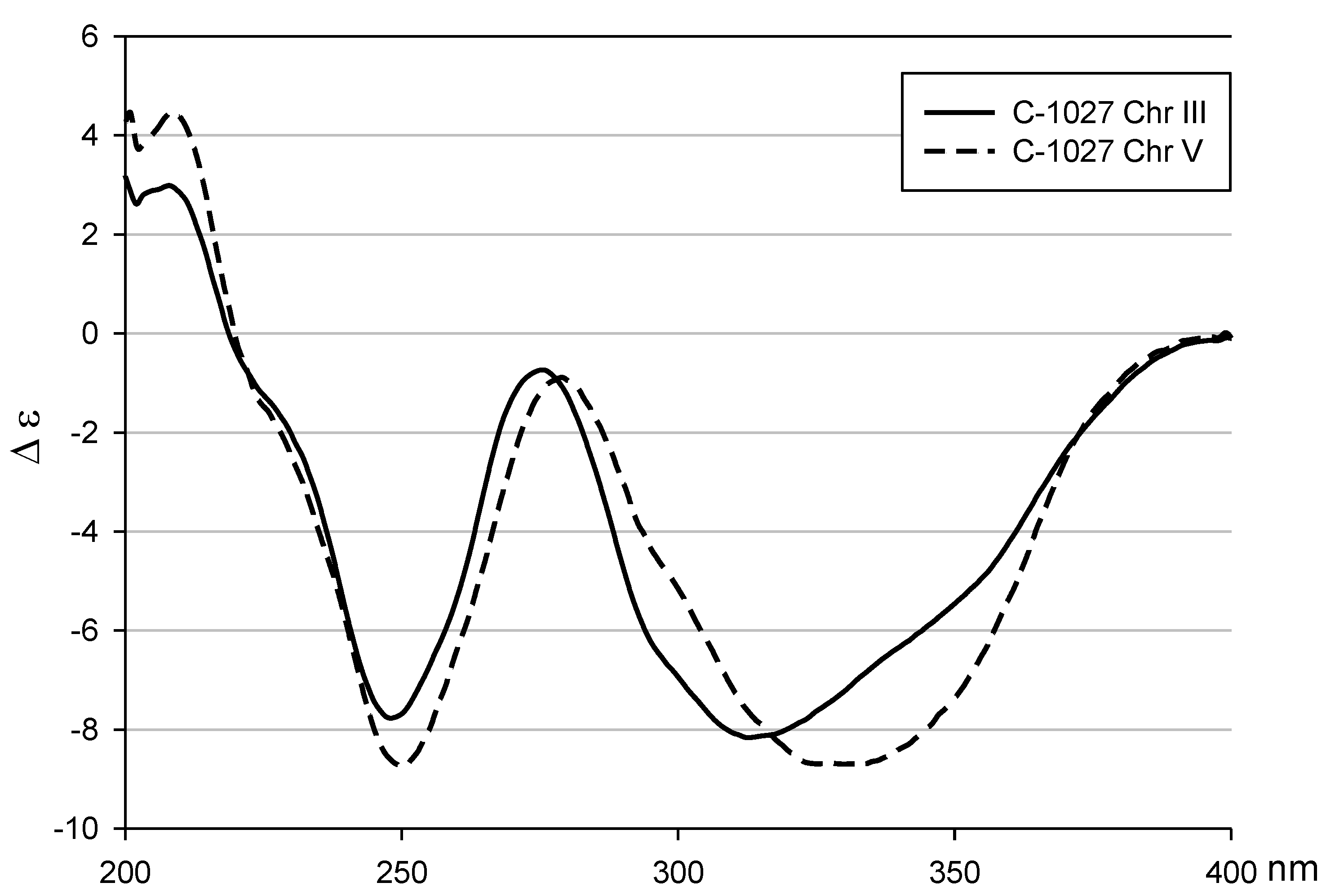
2. Results and Discussion
2.1. Structural Elucidation
| No. | δH a | Mult (J in Hz) | δC b | |
|---|---|---|---|---|
| 2 | 154.5 | C | ||
| 3 | 147.4 | C | ||
| 5 | 142.2 | C | ||
| 6 | 7.05 | d (2.5) | 107.2 | CH |
| 7 | 154.3 | C | ||
| 8 | 7.13 | d (2.5) | 108.6 | CH |
| 9 | 114.5 | C | ||
| 10 | 120.7 | C | ||
| 11a 11b | 5.48 5.14 | d (2.0) d (2.0) | 98.7 | CH2 |
| 12 | 165.3 | C | ||
| 1′ | 4.95 | d (3.5) | 93.7 | CH |
| 2′ | 3.35 | dd (9.5, 3.5) | 71.3 | CH |
| 3′ | 3.58 | dd (9.5, 9.5) | 72.8 | CH |
| 4′ | 3.20 | dd (9.5, 9.5) | 70.2 | CH |
| 5′ | 4.13 | ddd (9.5, 5.5, 2.0) | 69.2 | CH |
| 6′a 6′b | 4.49 4.40 | dd (11.5, 2.0) dd (11.5, 5.5) | 64.9 | CH2 |
| 1″ | 4.91 | d (3.5) | 93.7 | CH |
| 2″ | 3.23 | dd (9.5, 3.5) | 71.6 | CH |
| 3″ | 3.56 | dd (9.5, 9.5) | 72.7 | CH |
| 4″ | 3.14 | dd (9.5, 9.5) | 70.0 | CH |
| 5″ | 3.67 | ddd (9.5, 4.5, 2.0) | 72.6 | CH |
| 6″a 6″b | 3.54 3.47 | dd (12.0, 2.0) dd (12.0, 4.5) | 60.6 | CH2 |
| 1-NH | 10.11 | s | ||
| 7-OMe | 3.76 | s | 55.7 | CH3 |
| No. | δH a | Mult (J in Hz) | δC b | |
|---|---|---|---|---|
| 1 | 148.8 | C | ||
| 2 | 134.2 | C | ||
| 3 | 126.1 | C | ||
| 4 | 136.1 | C | ||
| 5 | 7.03 | d (8.0) | 130.0 | CH |
| 6 | 7.33 | d (8.0) | 125.9 | CH |
| 7 | 149.0 | C | ||
| 8 | 6.02 | s | 83.5 | CH |
| 9 | 99.7 | C | ||
| 10 | 6.73 | d (5.0) | 135.6 | CH |
| 11 | 6.74 | dd (5.0, 1.5) | 138.5 | CH |
| 12 | 6.85 | d (1.5) | 129.9 | CH |
| 13 | 6.01 | dd (10.5, 5) | 74.0 | CH |
| 14a 14b | 4.57 4.16 | d (10.5, 10) d (10.5, 5) | 62.8 | CH2 |
| 15 | 167.9 | C | ||
| 16a 16b | 3.01 2.36 | d (12.5) d (12.5) | 41.3 | CH2 |
| 17 | 4.23 | d (12.5) | 51.2 | CH |
| 18 | 133.6 | C | ||
| 19 | 6.87 | d (1.5) | 114.3 | CH |
| 20 | 129.6 | C | ||
| 21 | 139.7 | C | ||
| 22 | 152.4 | C | ||
| 23 | 6.17 | d (1.5) | 114.6 | CH |
| 1′ | 4.54 | d (8.0) | 92.9 | CH |
| 2′ | 2.97 | dd (8.0, 3.0) | 69.3 | CH |
| 3′ | 4.09 | br s | 67.4 | CH |
| 4′ | 3.16 | br s | 69.2 | CH |
| 5′ | 74.9 | C | ||
| 2″ | 154.5 | C | ||
| 3″ | 147.3 | C | ||
| 5″ | 142.1 | C | ||
| 6″ | 7.12 | d (2.5) | 107.4 | CH |
| 7″ | 154.6 | C | ||
| 8″ | 7.29 | 109.1 | CH | |
| 9″ | 113.5 | C | ||
| 10″ | 121.2 | C | ||
| 11″a 11″b | 5.48 5.16 | s s | 98.9 | CH2 |
| 12″ | 164.7 | C | ||
| 4′-NMe2 4′-NMe2 | 2.86 2.86 | br s br s | 44.3 42.7 | CH3 CH3 |
| 6′-Meα 6′-Meβ | 1.44 1.46 | s s | 31.3 21.3 | CH3 CH3 |
| 7″-OMe | 3.79 | s | 55.8 | CH3 |
| 17-NH2 | 9.08 | |||
| 22-OH | 8.56 | s | ||
| 2′-OH | 5.12 | |||
| 3′-OH | 5.49 | CH | ||
| 1″-NH | 10.05 | s |
2.2. Bioactivity Results
2.2.1. Inhibition of Candida albicans Isocitrate Lyase
2.2.2. Evaluation of Anti-Proliferative Activity
3. Experimental Section
3.1. General Experimental Procedures
3.2. Bacterial Material, Cultivation and Extraction
3.3. Isolation of Arcticoside (1) and C-1027 Chromophore-V (2)
3.3.1. Arcticoside (1)
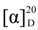 +19 (c 0.1 in MeOH); IR (KBr) 3388, 2965, 2938, 1680, 1615, 1507, 1259, 1053, 1032 cm−1; UV (MeOH) λmax (log ε) 346 (0.29), 220 (1.28), 209 (1.31) nm; 1H and 13C NMR (see Table 1); and HRFABMS [M + H]+ m/z 559.1527 (calcd for C23H29NO15, 559.1537).
+19 (c 0.1 in MeOH); IR (KBr) 3388, 2965, 2938, 1680, 1615, 1507, 1259, 1053, 1032 cm−1; UV (MeOH) λmax (log ε) 346 (0.29), 220 (1.28), 209 (1.31) nm; 1H and 13C NMR (see Table 1); and HRFABMS [M + H]+ m/z 559.1527 (calcd for C23H29NO15, 559.1537).3.3.2. C-1027 Chromophore-V (2)
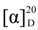 −412 (c 0.2 in MeOH); IR (KBr) 3428, 2933, 1683, 1438, 1366, 1207, 1135, 1051, 1028, 1007, 841 cm−1; UV (MeOH) λmax (log ε) 336 (0.19), 210 (1.42) 206 (1.43) nm; 1H and 13C NMR (see Table 2); and HRFABMS [M + H]+ m/z 880.2259 (calcd for C43H4435Cl2N3O13, 880.2251).
−412 (c 0.2 in MeOH); IR (KBr) 3428, 2933, 1683, 1438, 1366, 1207, 1135, 1051, 1028, 1007, 841 cm−1; UV (MeOH) λmax (log ε) 336 (0.19), 210 (1.42) 206 (1.43) nm; 1H and 13C NMR (see Table 2); and HRFABMS [M + H]+ m/z 880.2259 (calcd for C43H4435Cl2N3O13, 880.2251).3.4. Acid Hydrolysis and GC Analysis of Arcticoside (1)
3.5. Inhibition of the Isocitrate Lyase (ICL) Activity Assay
3.6. Anti-Proliferative Activity Assay
4. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bérdy, J. Thoughts and facts about antibiotics: Where we are now and where we are heading. J. Antibiot. 2012, 65, 385–395. [Google Scholar] [CrossRef]
- Ortholand, J.-Y.; Ganesan, A. Natural products and combinatorial chemistry: Back to the future. Curr. Opin. Chem. Biol. 2004, 8, 271–280. [Google Scholar] [CrossRef]
- Pettit, R.K. Culturability and secondary metabolite diversity of extreme microbes: Expanding contribution of deep sea and deep-sea vent microbes to natural product discovery. Mar. Biotechnol. 2011, 13, 1–11. [Google Scholar] [CrossRef]
- Andrianasolo, E.H.; Haramaty, L.; Rosario-Passapera, R.; Bidle, K.; White, E.; Vetriani, C.; Falkowski, P.; Lutz, R. Ammonificins A and B, hydroxyethylamine chroman derivatives from a cultured marine hydrothermal vent bacterium, Thermovibrio ammonificans. J. Nat. Prod. 2009, 72, 1216–1219. [Google Scholar] [CrossRef]
- Park, H.B.; Kwon, H.C.; Lee, C.-H.; Yang, H.O. Glionitrin A, an antibiotic–antitumor metabolite derived from competitive interaction between abandoned mine microbes. J. Nat. Prod. 2009, 72, 248–252. [Google Scholar] [CrossRef]
- Um, S.; Kim, Y.-J.; Kwon, H.; Wen, H.; Kim, S.-H.; Kwon, H.C.; Park, S.; Shin, J.; Oh, D.-C. Sungsanpin, a lasso peptide from a deep-sea streptomycete. J. Nat. Prod. 2013, 76, 873–879. [Google Scholar] [CrossRef]
- Gao, X.; Lu, Y.; Xing, Y.; Ma, Y.; Lu, J.; Bao, W.; Wang, Y.; Xi, T. A novel anticancer and antifungus phenazine derivative from a marine actinomycete BM-17. Microbiol. Res. 2012, 167, 616–622. [Google Scholar] [CrossRef]
- Ivanova, V.; Lyutskanova, D.; Kolarova, M.; Aleksieva, K.; Raykovska, V.; Stoilova-Disheva, M. Structural elucidation of a bioactive metabolites produced by Streptomycetes avidinii SB9 strain, isolated from permafrost soil in Spitsbergen, Arctic. Biotechnol. Biotechnol. Equip. 2010, 24, 2092–2095. [Google Scholar] [CrossRef]
- Minami, Y.; Yoshida, K.; Azuma, R.; Saeki, M.; Otani, T. Structure of an aromatization product of C-1027 chromophore. Tetrahedron Lett. 1993, 34, 2633–2636. [Google Scholar] [CrossRef]
- Nam, S.-J.; Gaudencio, S.P.; Kauffman, C.A.; Jensen, P.R.; Kondratyuk, T.P.; Marler, L.E.; Pezzuto, J.M.; Fenical, W. Fijiolides A and B, inhibitors of TNF-α-induced NFκB activation, from a marine-derived sediment bacterium of the genus Nocardiopsis. J. Nat. Prod. 2010, 73, 1080–1086. [Google Scholar] [CrossRef]
- Pretsch, E.; Bȕhlmann, P.; Affolter, C. Structure Determination of Organic Compounds—Tables of Spectral Data; Springer: New York, NY, USA, 2000; p. 153. [Google Scholar]
- Wahidulla, S.; Bhattacharjee, J.J. Benzoxazinoids from Acanthus illicifolius. J. Indian Inst. Sci. 2001, 81, 485–489. [Google Scholar]
- Sakawa, K.; Yoshida, K. Isolation and characterization of non-protein chromophore and its degradation product from antibiotic C-1027. J. Antibiot. 1991, 44, 564–568. [Google Scholar]
- Kumada, Y.; Miwa, T.; Naoi, N.; Watanabe, K.; Naganawa, H.; Takita, T.; Umezawa, H.; Nakamura, H.; Iitaka, Y. A degradation product of the chromophore of auromomycin. J. Antibiot. 1983, 36, 200–202. [Google Scholar] [CrossRef]
- Pretsch, E.; Bȕhlmann, P.; Affolter, C. Structure Determination of Organic Compounds—Tables of Spectral Data; Springer: New York, NY, USA, 2000; p. 176. [Google Scholar]
- Oh, D.-C.; Williams, P.G.; Kauffman, C.A.; Jensen, P.R.; Fenical, W. Cyanosporasides A and B, chloro- and cyano-cyclopenta[a]indene glycosides from the marine actinomycete “Salinispora pacifica”. Org. Lett. 2006, 8, 1021–1024. [Google Scholar] [CrossRef]
- Buchanan, G.O.; Williams, P.G.; Feling, R.H.; Kauffman, C.A.; Jensen, P.R.; Fenical, W. Sporolides A and B: Structurally unprecedented halogenated macrolides from the marine actinomycete Salinispora tropica. Org. Lett. 2005, 7, 2731–2734. [Google Scholar]
- Perrin, C.L.; Rodgers, B.L.; O’Connor, J.M. Nucleophilic addition to a p-benzyne derived from an enediyne: A new mechanism for halide incorporation into biomolecules. J. Am. Chem. Soc. 2007, 129, 4795–4799. [Google Scholar] [CrossRef]
- Yoshida, K.; Minami, Y.; Azuma, R.; Saeki, M.; Otani, T. Structure and cycloaromatization of a novel enediyne, C-1027 chromophore. Tetrahedron Lett. 1993, 34, 2637–2640. [Google Scholar] [CrossRef]
- Dunn, M.F.; Ramirez-Trujillo, J.A.; Hernandez-Lucas, I. Major roles of isocitrate lyase and malate synthase in bacterial and fungal pathogenesis. Microbiology 2009, 155, 3166–3175. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Moon, K.; Ahn, C.-H.; Shin, Y.; Won, T.H.; Ko, K.; Lee, S.K.; Oh, K.-B.; Shin, J.; Nam, S.-I.; Oh, D.-C. New Benzoxazine Secondary Metabolites from an Arctic Actinomycete. Mar. Drugs 2014, 12, 2526-2538. https://doi.org/10.3390/md12052526
Moon K, Ahn C-H, Shin Y, Won TH, Ko K, Lee SK, Oh K-B, Shin J, Nam S-I, Oh D-C. New Benzoxazine Secondary Metabolites from an Arctic Actinomycete. Marine Drugs. 2014; 12(5):2526-2538. https://doi.org/10.3390/md12052526
Chicago/Turabian StyleMoon, Kyuho, Chan-Hong Ahn, Yoonho Shin, Tae Hyung Won, Keebeom Ko, Sang Kook Lee, Ki-Bong Oh, Jongheon Shin, Seung-Il Nam, and Dong-Chan Oh. 2014. "New Benzoxazine Secondary Metabolites from an Arctic Actinomycete" Marine Drugs 12, no. 5: 2526-2538. https://doi.org/10.3390/md12052526
APA StyleMoon, K., Ahn, C.-H., Shin, Y., Won, T. H., Ko, K., Lee, S. K., Oh, K.-B., Shin, J., Nam, S.-I., & Oh, D.-C. (2014). New Benzoxazine Secondary Metabolites from an Arctic Actinomycete. Marine Drugs, 12(5), 2526-2538. https://doi.org/10.3390/md12052526





