High Levels of Structural Diversity Observed in Microcystins from Microcystis CAWBG11 and Characterization of Six New Microcystin Congeners
Abstract
:1. Introduction
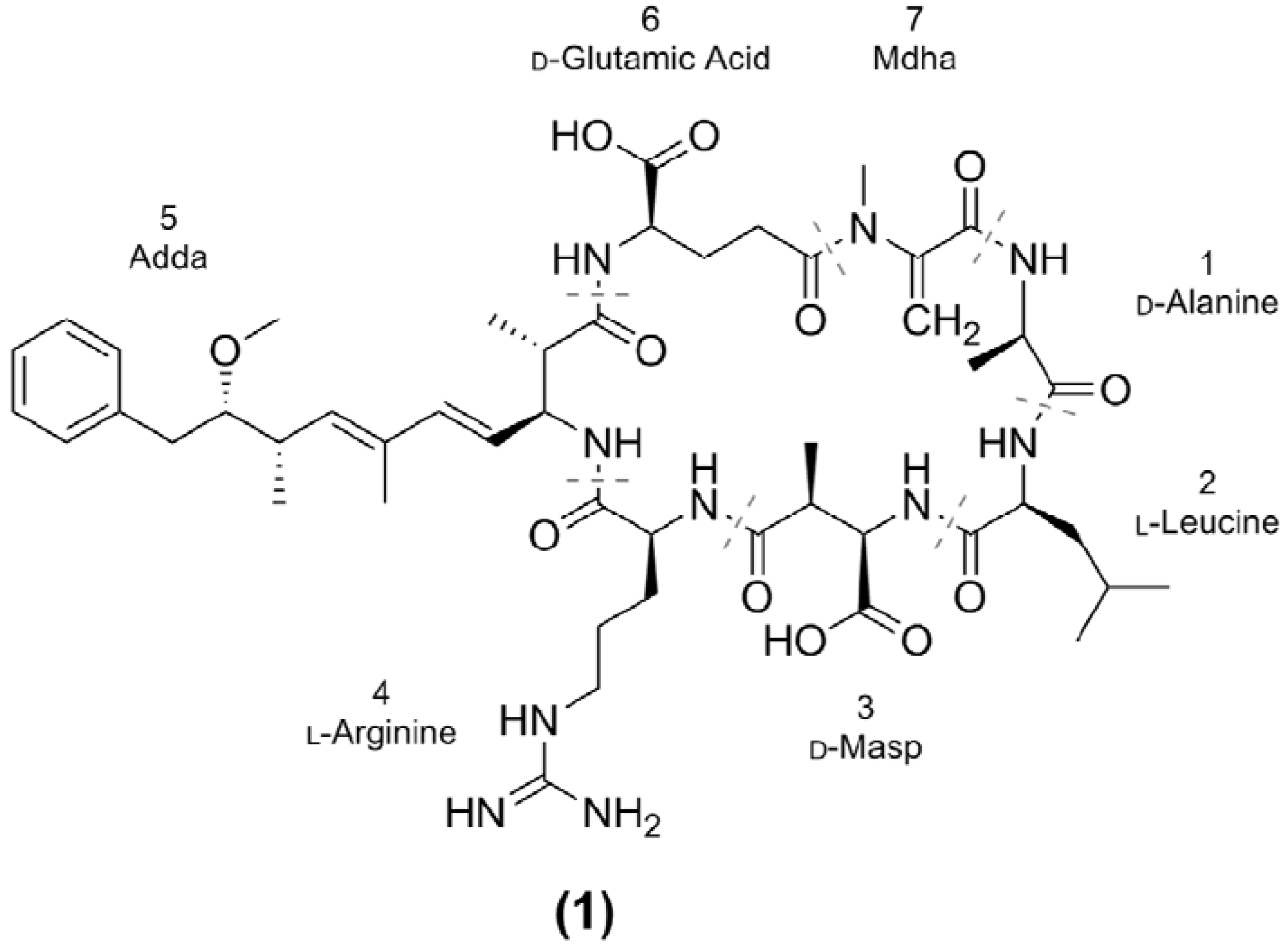
2. Results and Discussion
2.1. Oligopeptide Diversity of Microcystis CAWBG11
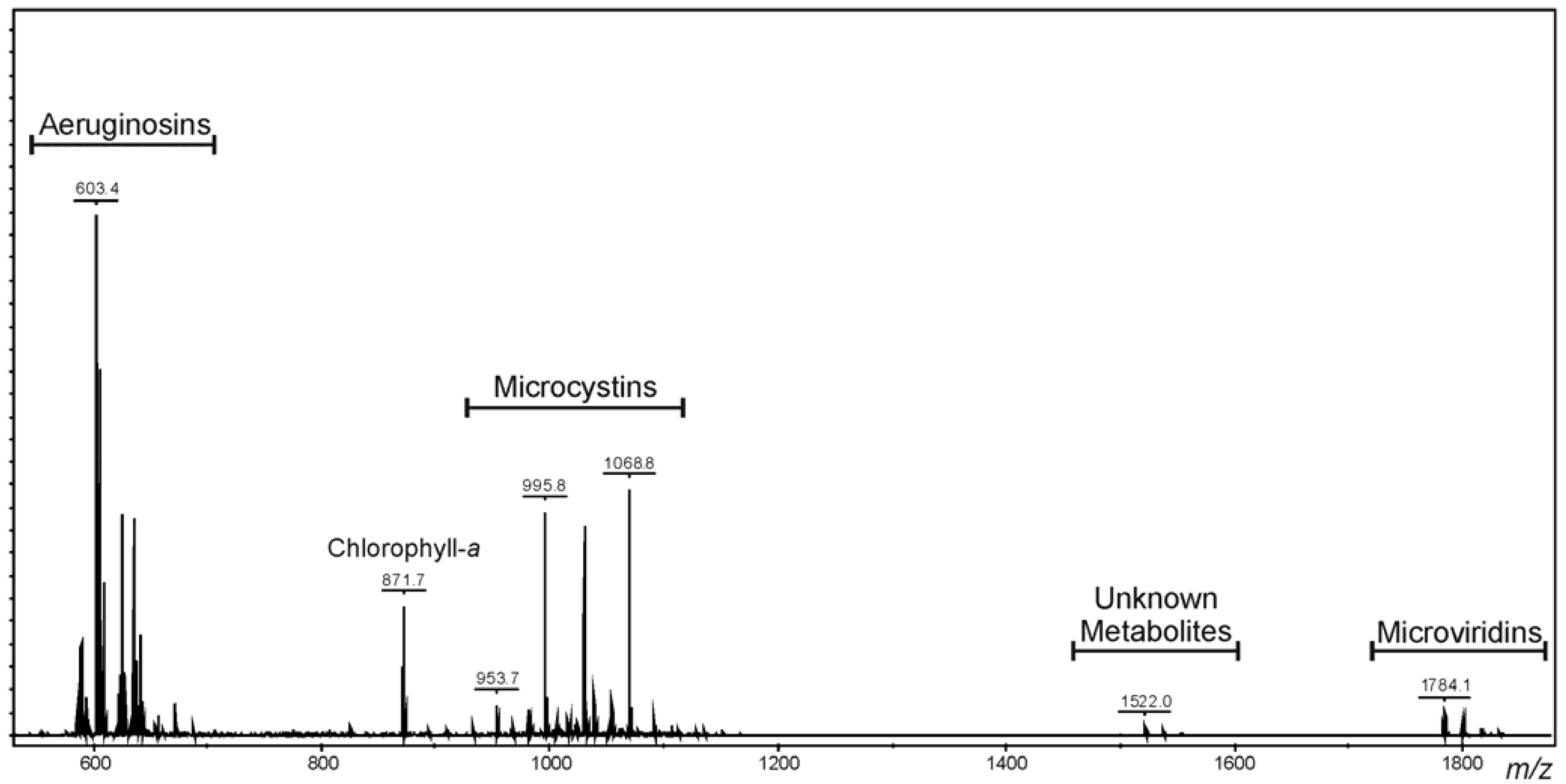
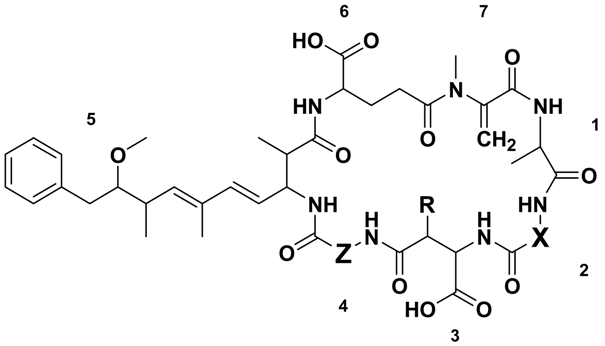
| Microcystin | Mr a (Da) | RT b (min) | X | Z | R | Reference(s) | |
|---|---|---|---|---|---|---|---|
| MC-RRs | [Asp3] MC-RR (2) | 1023.6 | 6.32 | Arg | Arg | H | [29] |
| MC-RR (3) | 1037.6 | 6.45 | Arg | Arg | CH3 | [30,31,32] | |
| MC-XRs | MC-YR (4) | 1044.5 | 7.20 | Tyr | Arg | CH3 | [33] |
| [Asp3] MC-LR (5) | 980.5 | 7.32 | Leu | Arg | H | [34,35] | |
| MC-LR (1) | 994.5 | 7.35 | Leu | Arg | CH3 | [16,33] | |
| [Asp3] MC-FR (6) | 1014.5 | 7.52 | Phe | Arg | H | [36] | |
| MC-FR (7) | 1028.5 | 7.54 | Phe | Arg | CH3 | [37] | |
| [Asp3] MC-WR (8) | 1053.5 | 7.56 | Trp | Arg | H | [36] | |
| MC-WR (9) | 1067.5 | 7.60 | Trp | Arg | CH3 | [37] | |
| MC-RZs | [Asp3] MC-RA (10) | 938.5 | 7.80 | Arg | Ala | H | This Study |
| MC-RA (11) | 952.5 | 7.84 | Arg | Ala | CH3 | [38,39] | |
| [Asp3] MC-RAba (12) | 952.5 | 8.16 | Arg | Aba | H | This Study | |
| MC-RAba (13) | 966.5 | 8.18 | Arg | Aba | CH3 | [39] | |
| MC-RL (14) | 994.5 | 8.35 | Arg | Leu | CH3 | [39] | |
| MC-XAs | MC-YA (15) | 959.5 | 9.01 | Tyr | Ala | CH3 | [33] |
| [Asp3] MC-LA (16) | 895.5 | 9.12 | Leu | Ala | H | [40] | |
| MC-LA (17) | 909.5 | 9.29 | Leu | Ala | CH3 | [41] | |
| [Asp3] MC-FA (18) | 929.5 | 9.46 | Phe | Ala | H | This Study | |
| MC-FA (19) | 943.5 | 9.52 | Phe | Ala | CH3 | [42] | |
| [Asp3] MC-WA (20) | 968.5 | 9.48 | Trp | Ala | H | This Study | |
| MC-WA (21) | 982.5 | 9.54 | Trp | Ala | CH3 | [42] | |
| MC-XAbas | MC-LAba (22) | 923.5 | 9.69 | Leu | Aba | CH3 | [40,43,44] |
| MC-FAba (23) | 957.5 | 9.97 | Phe | Aba | CH3 | This Study | |
| MC-WAba (24) | 996.5 | 10.24 | Trp | Aba | CH3 | [28] | |
| MC-XLs | MC-LL (25) | 951.5 | 10.50 | Leu | Leu | CH3 | [45] |
| MC-FL (26) | 985.5 | 10.71 | Phe | Leu | CH3 | This Study | |
| MC-WL (27) | 1024.5 | 10.77 | Trp | Leu | CH3 | [28] | |
2.2. Characterization of Microcystins from Microcystis CAWBG11
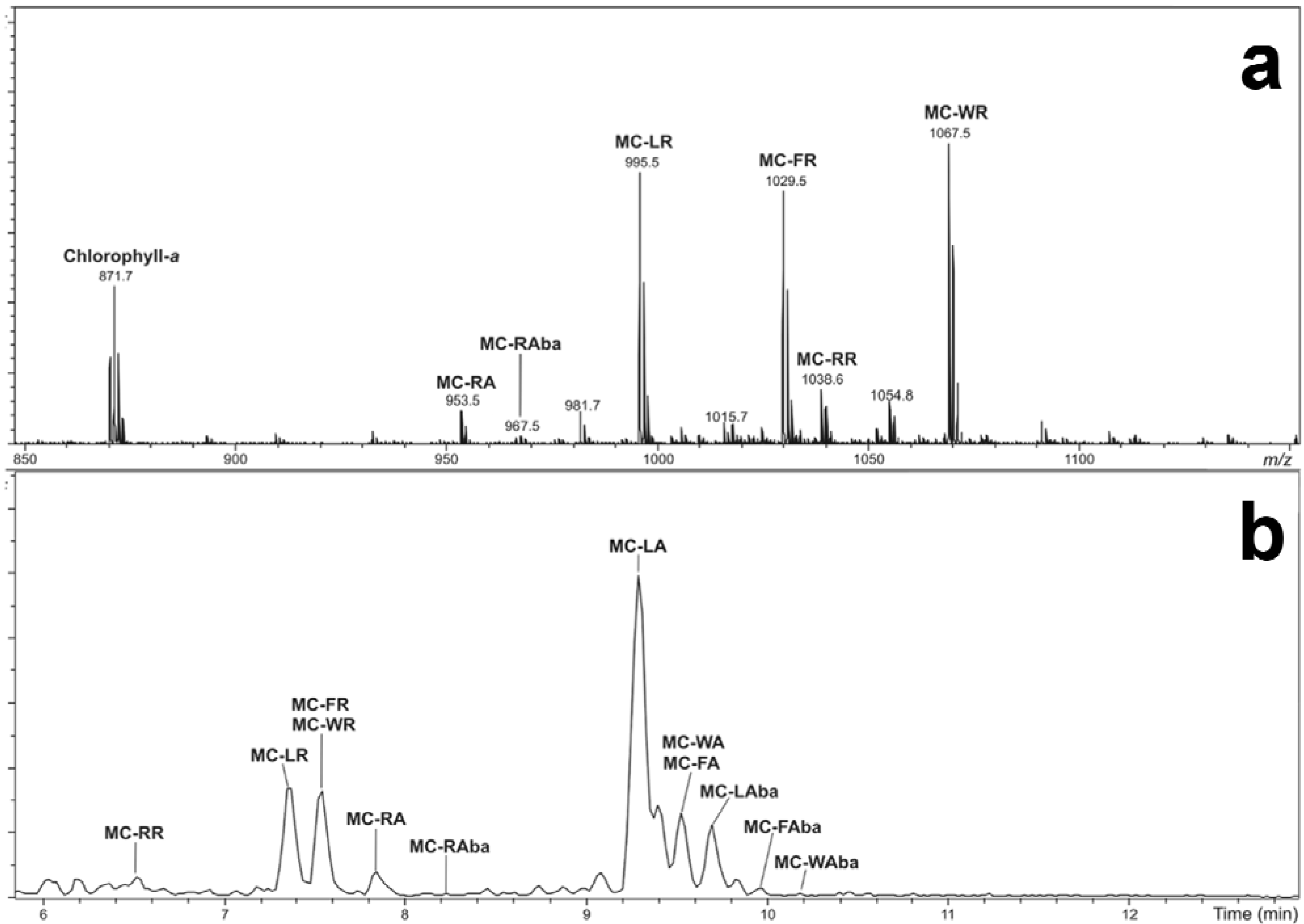
2.3. Tandem Mass Spectrometry Characterization of New Microcystins from Microcystis CAWBG11
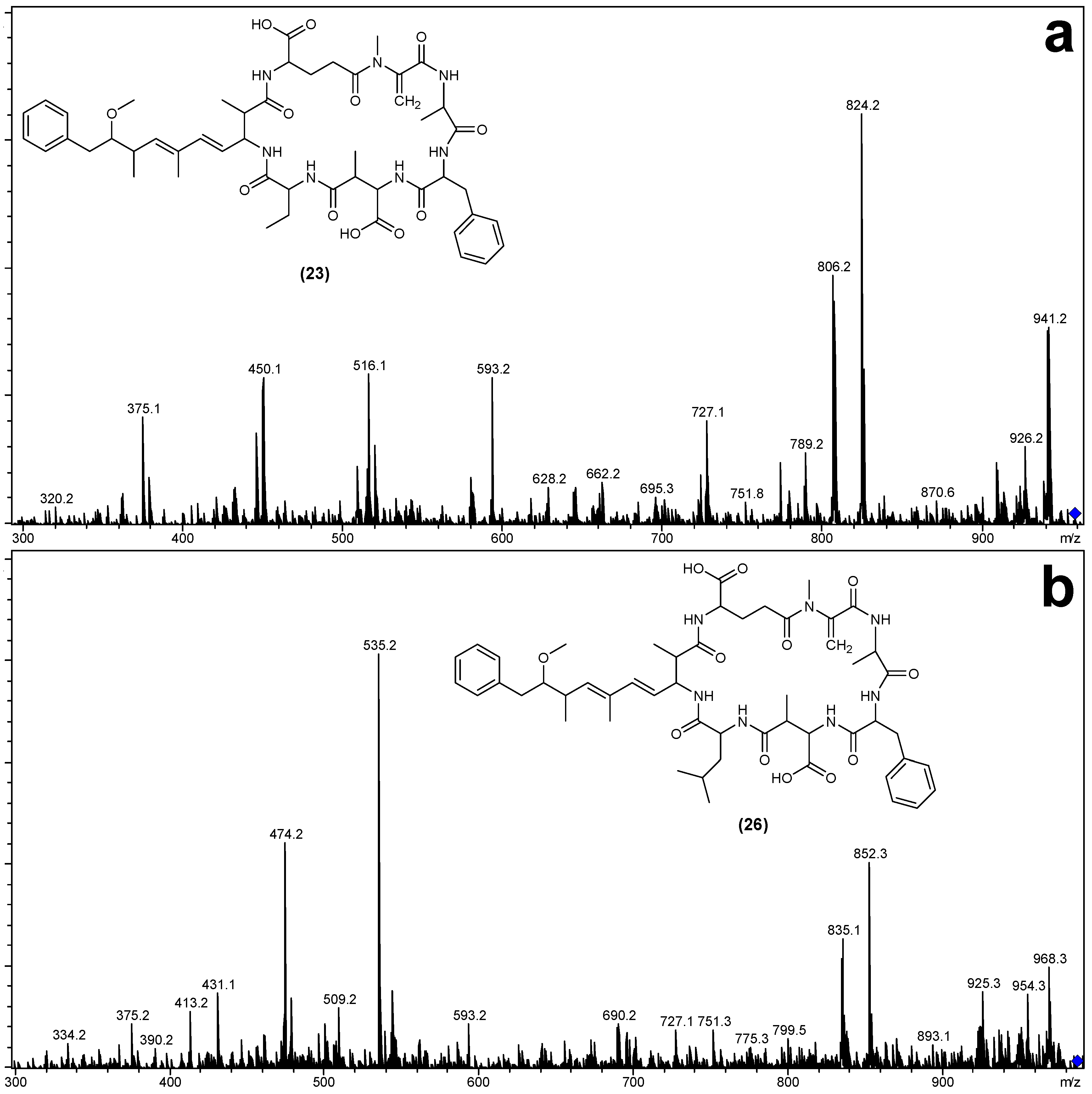
| Fragment Assignment a | MC-FA (19) | MC-FAba (23) | MC-FL (26) |
|---|---|---|---|
| M + H | 944 | 958 | 986 |
| M − H2O + H | 926 | 940 | 968 |
| M − Mdha − H2O + H | 843 | 857 | 885 |
| M − Adda sidechain + H | 810 | 824 | 852 |
| M − Adda sidechain − H2O + H | 792 | 806 | 834 |
| M − Adda + H | 631 | 645 | 673 |
| M − Adda − H2O + H | 613 | 627 | 655 |
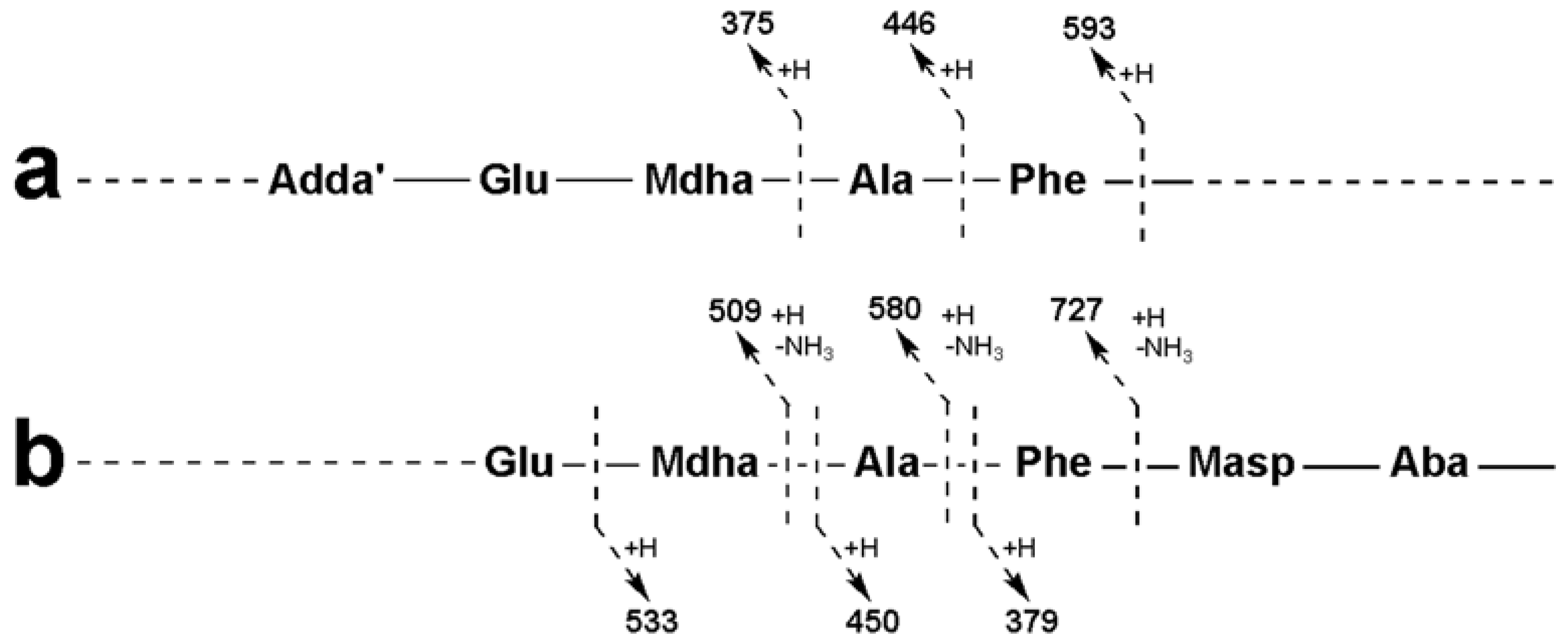
| Adda-Glu-Mdha-Ala-Phe-Masp − NH3 + H | 856 | 856 | 856 |
| Adda-Glu-Mdha-Ala-Phe − NH3 + H | 727 | 727 | 727 |
| Adda-Glu-Mdha-Ala − NH3 + H | 580 | 580 | 580 |
| Adda-Glu-Mdha − NH3 + H | 509 | 509 | 509 |
| Adda′-Glu-Mdha-Ala-Phe + H | 593 | 593 | 593 |
| Adda′-Glu-Mdha-Ala + H | 446 | 446 | 446 |
| Adda′-Glu-Mdha + H | 375 | 375 | 375 |
| Mdha-Ala-Phe-Masp-Z + NH4 | 471 | 533 | 561 |
| Ala-Phe-Masp-Z + NH4 | 388 | 450 | 478 |
| Phe-Masp-Z + NH4 | 317 | 379 | 407 |
| Mdha-Ala-Phe-Masp-Z + H | 454 | 516 | 544 |
| Ala-Phe-Masp-Z + H | 371 | 433 | 461 |
| Phe-Masp-Z + H | 300 | 362 | 390 |
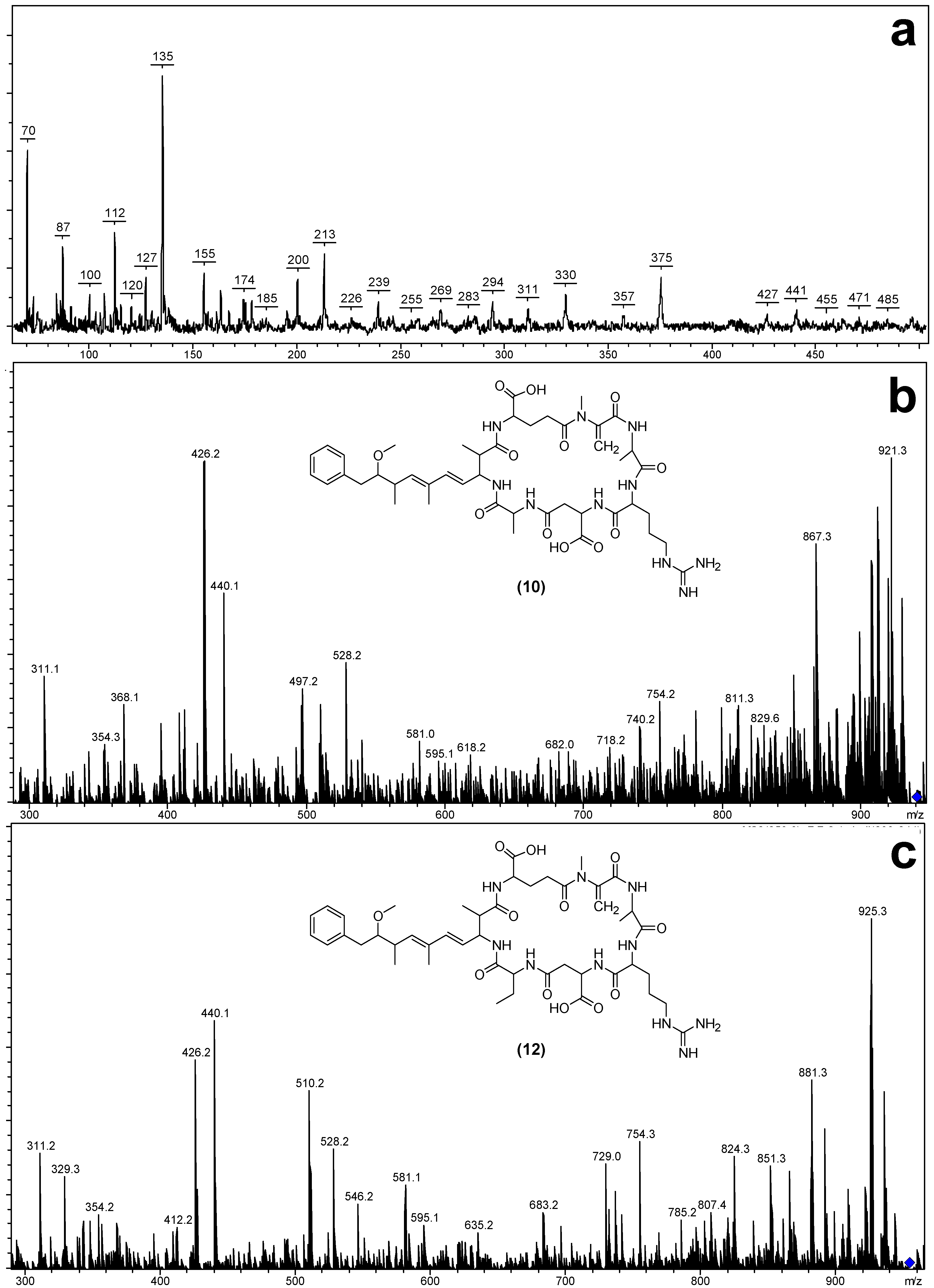
| Fragment Assignment a | [Asp3] MC-RA (10) | MC-RA (11) | [Asp3] MC-RAba (12) | MC-Raba (13) |
|---|---|---|---|---|
| M + H b,c | 939 | 953 | 953 | 967 |
| M − H2O + H b,c | 921 | 935 | 935 | 949 |
| M − COOH + H c | 894 | 908 | 908 | 922 |
| M − Z + H c | 868 | 882 | 868 | 882 |
| M − CH2NHCN2H3 + H c | 867 | 881 | 881 | 895 |
| M − Glu + H c | 810 | 824 | 824 | 838 |
| M − (Me)Asp + H c | 824 | 824 | 838 | 838 |
| M − Adda sidechain + H c | 787 | 819 | 819 | 833 |
| Mdha-Ala-Arg-(Me)Asp-Z + NH4 c | 514 | 528 | 528 | 542 |
| Mdha-Ala-Arg-(Me)Asp-Z − H2O + NH4 c | 496 | 510 | 510 | 524 |
| Mdha-Ala-Arg-(Me)Asp-Z + H c | 497 | 511 | 511 | 525 |
| Mdha-Ala-Arg-(Me)Asp − CH2NHCN2H3 + H c | 354 | 368 | 354 | 368 |
| Mdha-Ala-Arg-(Me)Asp + H c | 426 | 440 | 426 | 440 |
| Mdha-Ala-Arg + H b,c | 311 | 311 | 311 | 311 |
| Mdha-Ala + H b | 155 | 155 | - | 155 |
| Arg-(Me)Asp-Z + H c | 343 | 357 | 357 | |
| Glu-Mdha-Ala-Arg − COOH + H c | 395 | 395 | 395 | 395 |
| Glu-Mdha-Ala-Arg − CH2NHCN2H3 + H c | 368 | 368 | 368 | 368 |
| Glu-Mdha-Ala-Arg + H c | 440 | 440 | 440 | 440 |
| Glu-Mdha + H b | 213 | - | 213 | |
| Adda′-Glu-Mdha + H b,c | 375 | 375 | 375 | 375 |
| Adda′ + H b | 163 | 163 | - | 163 |
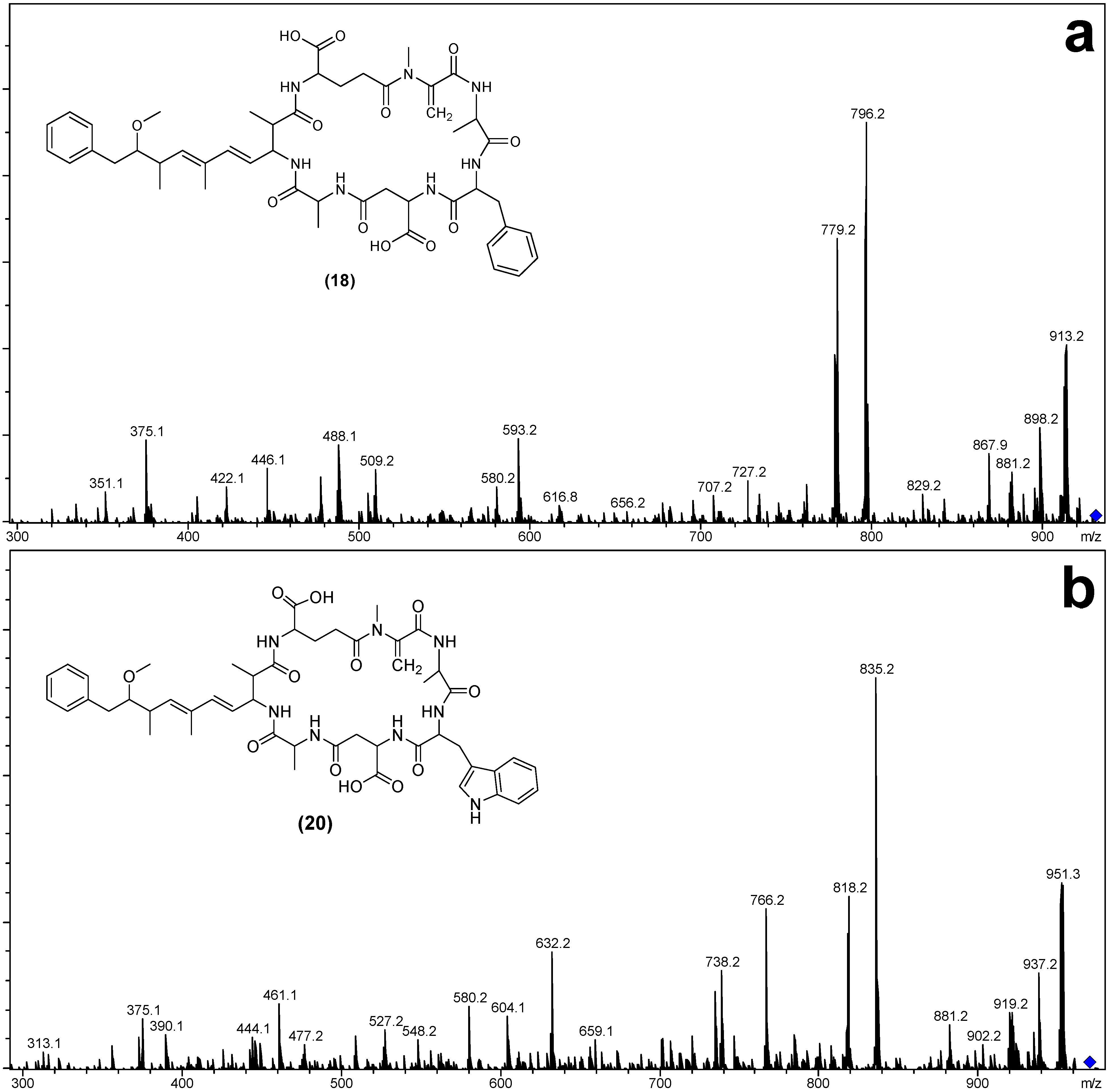
| Fragment Assignment a | [Asp3] MC-FA (18) | MC-FA (19) | [Asp3] MC-WA (20) | MC-WA (21) |
|---|---|---|---|---|
| M + H | 930 | 944 | 969 | 983 |
| M − H2O + H | 912 | 926 | 951 | 965 |
| M − Mdha − H2O + H | 829 | 843 | 868 | 882 |
| M − Adda sidechain + H | 796 | 810 | 835 | 849 |
| M − Adda sidechain − H2O + H | 778 | 792 | 817 | 831 |
| M − Adda + H | 617 | 631 | 656 | 670 |
| M − Adda − H2O + H | 599 | 613 | 638 | 652 |
| Adda-Glu-Mdha-Ala-X-(Me)Asp − NH3 + H | 842 | 856 | 881 | 895 |
| Adda-Glu-Mdha-Ala-X − NH3 + H | 727 | 727 | 766 | 766 |
| Adda-Glu-Mdha-Ala − NH3 + H | 580 | 580 | 580 | 580 |
| Adda-Glu-Mdha − NH3 + H | 509 | 509 | 509 | 509 |
| Adda′-Glu-Mdha-Ala-X + H | 593 | 593 | 632 | 632 |
| Adda′-Glu-Mdha-Ala + H | 446 | 446 | 446 | 446 |
| Adda′-Glu-Mdha + H | 375 | 375 | 375 | 375 |
| Mdha-Ala-X-(Me)Asp-Ala + NH4 | 505 | 519 | 558 | |
| Ala-X-(Me)Asp-Ala + NH4 | 422 | 436 | 461 | 475 |
| X-(Me)Asp-Ala + NH4 | 351 | 365 | 390 | 404 |
| Mdha-Ala-X-(Me)Asp-Ala + H | 334 | 502 | 527 | 541 |
| Ala-X-(Me)Asp-Ala + H | 405 | 419 | 444 | 458 |
| X-(Me)Asp-Ala + H | 488 | 348 | 373 | 387 |
2.4. The Microcystin Diversity of Microcystis CAWBG11
3. Experimental Section
3.1. Microcystis CAWBG11
3.2. General Experimental Procedures
3.3. Matrix-Assisted Laser Desorption/Ionization-Time of Flight Mass Spectrometry Analysis
3.4. Liquid Chromatography-Mass Spectrometry Analysis
3.5. β-mercaptoethanol Derivatization for Mdha/Dhb Determination
3.6. Isolation of MC-RA and MC-RAba
3.7. Advanced Marfey’s Amino Acid Analysis
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Holt, J.G.; Krieg, N.R.; Sneath, P.H.A.; Staley, J.T.; Williams, S.T. Oxygenic photosynthetic bacteria. In Bergey’s Manual of Determinative Bacteriology, 9th ed.; Lippincott Williams and Wilkins: Philadelphia, PA, USA, 2000; pp. 377–426. [Google Scholar]
- Whitton, B.A.; Potts, M. The Ecology of Cyanobacteria, Their Diversity in Time and Space; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2000. [Google Scholar]
- Jones, G.J.; Korth, W. In situ production of volatile odour compounds by river and reservoir phytoplankton populations in Australia. Water Sci. Technol. 1995, 31, 145–151. [Google Scholar] [CrossRef]
- Sivonen, K.; Jones, G. Cyanobacterial toxins. In Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management; Chorus, I., Bartram, J., Eds.; E & FN Spon: London, UK, 1999; pp. 55–124. [Google Scholar]
- Van Apeldoorn, M.E.; van Egmond, H.P.; Speijers, G.J.A.; Bakker, G.J.I. Toxins of cyanobacteria. Mol. Nutr. Food Res. 2007, 51, 7–60. [Google Scholar] [CrossRef] [PubMed]
- Niedermeyer, T. Microcystin Congeners Described in the Literature. 2013. Available online: http://dx.doi.org/10.6084/m9.figshare.880756 (accessed on 21 October 2014).
- Rinehart, K.; Namikoshi, M.; Choi, B. Structure and biosynthesis of toxins from blue-green algae (cyanobacteria). J. Appl. Phycol. 1994, 6, 159–176. [Google Scholar] [CrossRef]
- Tillett, D.; Dittmann, E.; Erhard, M.; von Döhren, H.; Börner, T.; Neilan, B.A. Structural organization of microcystin biosynthesis in Microcystis aeruginosa PCC7806: An integrated peptide-polyketide synthetase system. Chem. Biol. 2000, 7, 753–764. [Google Scholar] [CrossRef] [PubMed]
- Moore, R.E.; Chen, J.L.; Moore, B.S.; Patterson, G.M.L.; Carmichael, W.W. Biosynthesis of microcystin-LR: Origin of the carbons in the Adda and Masp units. J. Am. Chem. Soc. 1991, 113, 5083–5084. [Google Scholar] [CrossRef]
- Nishizawa, T.; Asayama, M.; Fujii, K.; Harada, K.-I.; Shirai, M. Genetic analysis of the peptide synthetase genes for a cyclic heptapeptide microcystin in Microcystis spp. J. Biochem. 1999, 126, 520–529. [Google Scholar] [CrossRef]
- Nishizawa, T.; Ueda, A.; Asayama, M.; Fujii, K.; Harada, K.-I.; Ochi, K.; Shirai, M. Polyketide synthase gene coupled to the peptide synthetase module involved in the biosynthesis of the cyclic heptapeptide microcystin. J. Biochem. 2000, 127, 779–789. [Google Scholar] [CrossRef] [PubMed]
- Börner, T.; Dittmann, E. Molecular biology of cyanobacterial toxins. In Harmful Cyanobacteria; Huisman, J., Matthijs, H.C.P., Visser, P.M., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 25–40. [Google Scholar]
- Mikalsen, B.; Boison, G.; Skulberg, O.M.; Fastner, J.; Davies, W.; Gabrielsen, T.M.; Rudi, K.; Jakobsen, K.S. Natural variation in the microcystin synthetase operon mcyABC and impact on microcystin production in Microcystis strains. J. Bacteriol. 2003, 185, 2774–2785. [Google Scholar] [CrossRef] [PubMed]
- Fewer, D.; Rouhiainen, L.; Jokela, J.; Wahlsten, M.; Laakso, K.; Wang, H.; Sivonen, K. Recurrent adenylation domain replacement in the microcystin synthetase gene cluster. BMC Evol. Biol. 2007, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Fischer, A.; Hoeger, S.J.; Stemmer, K.; Feurstein, D.J.; Knobeloch, D.; Nussler, A.; Dietrich, D.R. The role of organic anion transporting polypeptides (OATPs/SLCOs) in the toxicity of different microcystin congeners in vitro: A comparison of primary human hepatocytes and OATP-transfected HEK293 cells. Toxicol. Appl. Pharmacol. 2010, 245, 9–20. [Google Scholar] [CrossRef]
- An, J.; Carmichael, W.W. Use of a colorimetric protein phosphatase inhibition assay and enzyme linked immunosorbent assay for the study of microcystins and nodularins. Toxicon 1994, 32, 1495–1507. [Google Scholar] [CrossRef] [PubMed]
- Runnegar, M.T.; Falconer, I.R.; Silver, J. Deformation of isolated rat hepatocytes by a peptide hepatotoxin from the blue-green alga Microcystis aeruginosa. Naunyn-Schmiedeberg’s Arch. Pharmacol. 1981, 317, 268–272. [Google Scholar] [CrossRef]
- Falconer, I.R.; Yeung, D.S.K. Cytoskeletal changes in hepatocytes induced by Microcystis toxins and their relation to hyperphosphorylation of cell proteins. Chem. Biol. Interact. 1992, 81, 181–196. [Google Scholar] [CrossRef] [PubMed]
- Fujiki, H.; Suganuma, M. Tumor promotion by inhibitors of protein phosphatases 1 and 2A: The okadaic acid class of compounds. Adv. Cancer Res. 1993, 61, 143–194. [Google Scholar] [PubMed]
- Sainis, I.; Fokas, D.; Vareli, K.; Tzakos, A.; Kounnis, V.; Briasoulis, E. Cyanobacterial cyclopeptides as lead compounds to novel targeted cancer drugs. Mar. Drugs 2010, 8, 629–657. [Google Scholar] [CrossRef] [PubMed]
- Niedermeyer, T.H.J.; Daily, A.; Swiatecka-Hagenbruch, M.; Moscow, J.A. Selectivity and potency of microcystin congeners against OATP1B1 and OATP1B3 expressing cancer cells. PLoS One 2014, 9, e91476. [Google Scholar] [CrossRef] [PubMed]
- Rueckert, A.; Wood, S.A.; Cary, S.C. Development and field assessment of a quantitative PCR for the detection and enumeration of the noxious bloom-former Anabaena planktonica. Limnol. Oceanogr. Methods 2007, 5, 474–483. [Google Scholar] [CrossRef]
- Ishida, K.; Okita, Y.; Matsuda, H.; Okino, T.; Murakami, M. Aeruginosins, protease inhibitors from the cyanobacterium Microcystis aeruginosa. Tetrahedron 1999, 55, 10971–10988. [Google Scholar] [CrossRef]
- Ploutno, A.; Carmeli, S. Modified peptides from a water bloom of the cyanobacterium Nostoc sp. Tetrahedron 2002, 58, 9949–9957. [Google Scholar] [CrossRef]
- Welker, M.; Marsálek, B.; Sejnohová, L.; von Döhren, H. Detection and identification of oligopeptides in Microcystis (cyanobacteria) colonies: Toward an understanding of metabolic diversity. Peptides 2006, 27, 2090–2103. [Google Scholar] [CrossRef] [PubMed]
- Murakami, M.; Okita, Y.; Matsuda, H.; Okino, T.; Yamaguchi, K. Aeruginosin 298-A, a thrombin and trypsin inhibitor from the blue-green alga Microcystis aeruginosa (NIES-298). Tetrahedron Lett. 1994, 35, 3129–3132. [Google Scholar] [CrossRef]
- Howard, K.L.; Boyer, G.L. Quantitative analysis of cyanobacterial toxins by matrix-assisted laser desorption ionization mass spectrometry. Anal. Chem. 2007, 79, 5980–5986. [Google Scholar] [CrossRef] [PubMed]
- Puddick, J.; Prinsep, M.R.; Wood, S.A.; Miles, C.O.; Rise, F.; Cary, S.C.; Hamilton, D.P.; Wilkins, A.L. Structural characterization of new microcystins containing tryptophan and oxidized tryptophan residues. Mar. Drugs 2013, 11, 3025–3045. [Google Scholar] [CrossRef] [PubMed]
- Meriluoto, J.A.O.; Sandström, A.; Eriksson, J.E.; Remaud, G.; Grey Graig, A.; Chattopadhyaya, J. Structure and toxicity of a peptide hepatotoxin from the cyanobacterium Oscillatoria agardhii. Toxicon 1989, 27, 1021–1034. [Google Scholar] [CrossRef] [PubMed]
- Kusumi, T.; Ooi, T.; Watanabe, M.M.; Takahashi, H.; Kakisawa, H. Cyanoviridin RR, a toxin from the cyanobacterium (blue-green alga) Microcystis viridis. Tetrahedron Lett. 1987, 28, 4695–4698. [Google Scholar] [CrossRef]
- Painuly, P.; Perez, R.; Fukai, T.; Shimizu, Y. The structure of a cyclic peptide toxin, cyanogenosin-RR from Microcystis aeruginosa. Tetrahedron Lett. 1988, 29, 11–14. [Google Scholar] [CrossRef]
- Watanabe, M.F.; Oishi, S.; Harada, K.-I.; Matsuura, K.; Kawai, H.; Suzuki, M. Toxins contained in Microcystis species of cyanobacteria (blue-green algae). Toxicon 1988, 26, 1017–1025. [Google Scholar] [CrossRef] [PubMed]
- Botes, D.P.; Wessels, P.L.; Kruger, H.; Runnegar, M.T.C.; Santikarn, S.; Smith, R.J.; Barna, J.C.J.; Williams, D.H. Structural studies on cyanoginosins-LR, -YR, -YA, and -YM, peptide toxins from Microcystis aeruginosa. Org. Bioorg. Chem. 1985, 12, 2747–2748. [Google Scholar]
- Krishnamurthy, T.; Szafraniec, L.; Hunt, D.F.; Shabanowitz, J.; Yates, J.R.; Hauer, C.R.; Carmichael, W.W.; Skulberg, O.; Codd, G.A.; Missler, S. Structural characterization of toxic cyclic peptides from blue-green algae by tandem mass spectrometry. Proc. Natl. Acad. Sci. USA 1989, 86, 770–774. [Google Scholar] [CrossRef] [PubMed]
- Harada, K.-I.; Ogawa, K.; Kimura, Y.; Murata, H.; Suzuki, M.; Thorn, P.M.; Evans, W.R.; Carmichael, W.W. Microcystins from Anabaena flos-aquae NRC 525-17. Chem. Res. Toxicol. 1991, 4, 535–540. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.-H.; Chou, H.-N. Isolation and identification of seven microcystins from a cultured M.TN-2 strain of Microcystis aeruginosa. Bot. Bull. Academ. Sinica 2000, 41, 197–202. [Google Scholar]
- Namikoshi, M.; Rinehart, K.L.; Sakai, R.; Stotts, R.R.; Dahlem, A.M.; Beasley, V.R.; Carmichael, W.W.; Evans, W.R. Identification of 12 hepatotoxins from a Homer Lake bloom of the cyanobacteria Microcystis aeruginosa, Microcystis viridis, and Microcystis wesenbergii: Nine new microcystins. J. Org. Chem. 1992, 57, 866–872. [Google Scholar] [CrossRef]
- Lee, T.-H.; Chen, Y.-M.; Chou, H.-N. First report of microcystins in Taiwan. Toxicon 1998, 36, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Miles, C.O.; Sandvik, M.; Nonga, H.E.; Rundberget, T.; Wilkins, A.L.; Rise, F.; Ballot, A. Thiol derivatization for LC-MS identification of microcystins in complex matrices. Environ. Toxicol. 2012, 46, 8937–8944. [Google Scholar]
- Diehnelt, C.W.; Dugan, N.R.; Peterman, S.M.; Budde, W.L. Identification of microcystin toxins from a strain of Microcystis aeruginosa by liquid chromatography introduction into a hybrid linear ion trap-fourier transform ion cyclotron resonance mass spectrometer. Anal. Chem. 2006, 78, 501–512. [Google Scholar] [CrossRef] [PubMed]
- Botes, D.P.; Tuinman, A.A.; Wessels, P.L.; Viljoen, C.C.; Kruger, H.; Williams, D.H.; Santikarn, S.; Smith, R.J.; Hammond, S.J. The structure of cyanoginosin-LA, a cyclic heptapeptide toxin from the cyanobacterium Microcystis aeruginosa. Org. Bioorg. Chem. 1984, 10, 2311–2318. [Google Scholar]
- Puddick, J.; Prinsep, M.R.; Wood, S.A.; Cary, S.C.; Hamilton, D.P.; Wilkins, A.L. Isolation and structure determination of two new hydrophobic microcystins from Microcystis sp. (CAWBG11). Phytochem. Lett. 2013, 6, 575–581. [Google Scholar] [CrossRef]
- Del Campo, F.F.; Ouahid, Y. Identification of microcystins from three collection strains of Microcystis aeruginosa. Environ. Pollut. 2010, 158, 2906–2914. [Google Scholar]
- Gathercole, P.S.; Thiel, P.G. Liquid chromatographic determination of the cyanoginosins, toxins produced by the cyanobacterium Microcystis aeruginosa. J. Chromatogr. A 1987, 408, 435–440. [Google Scholar] [CrossRef]
- Craig, M.; McCready, T.L.; Luu, H.A.; Smillie, M.A.; Dubord, P.; Holmes, C.F.B. Identification and characterization of hydrophobic microcystins in Canadian freshwater cyanobacteria. Toxicon 1993, 31, 1541–1549. [Google Scholar] [CrossRef] [PubMed]
- Beattie, K.A.; Kaya, K.; Sano, T.; Codd, G.A. Three dehydrobutyrine-containing microcystins from Nostoc. Phytochemistry 1998, 47, 1289–1292. [Google Scholar] [CrossRef]
- Sano, T.; Beattie, K.A.; Codd, G.A.; Kaya, K. Two (Z)-dehydrobutyrine-containing microcystins from a hepatotoxic bloom of Oscillatoria agardhii from Soulseat Loch, Scotland. J. Nat. Prod. 1998, 61, 851–853. [Google Scholar] [CrossRef] [PubMed]
- Sano, T.; Kaya, K. Two new (E)-2-amino-2-butenoic acid (Dhb)-containing microcystins isolated from Oscillatoria agardhii. Tetrahedron 1998, 54, 463–470. [Google Scholar] [CrossRef]
- Miles, C.O.; Sandvik, M.; Haande, S.; Nonga, H.; Ballot, A. First use of LC-MS analysis with thiol derivatization to differentiate [Dhb7]- from [Mdha7]-microcystins: Analysis of cyanobacterial blooms, Planktothrix cultures and European crayfish from Lake Steinsfjorden, Norway. Environ. Sci. Technol. 2013, 47, 4080–4087. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.L.; Boyer, G.L. Standardization of microcystin extraction from fish tissues: A novel internal standard as a surrogate for polar and non-polar variants. Toxicon 2009, 53, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Frias, H.V.; Mendes, M.A.; Cardozo, K.H.M.; Carvalho, V.M.; Tomazela, D.; Colepicolo, P.; Pinto, E. Use of electrospray tandem mass spectrometry for identification of microcystins during a cyanobacterial bloom event. Biochem. Biophys. Res. Commun. 2006, 344, 741–746. [Google Scholar] [CrossRef] [PubMed]
- Okello, W.; Portmann, C.; Erhard, M.; Gademann, K.; Kurmayer, R. Occurrence of microcystin-producing cyanobacteria in Ugandan freshwater habitats. Environ. Toxicol. 2010, 25, 367–380. [Google Scholar] [CrossRef] [PubMed]
- Fujii, K.; Ikai, Y.; Oka, H.; Suzuki, M.; Harada, K.-I. A nonempirical method using LC/MS for determination of the absolute configuration of constituent amino acids in a peptide: Combination of Marfey’s method with mass spectrometry and its practical application. Anal. Chem. 1997, 69, 5146–5151. [Google Scholar] [CrossRef]
- Mahakhant, A.; Sano, T.; Ratanachot, P.; Tong-a-ram, T.; Srivastava, V.C.; Watanabe, M.M.; Kaya, K. Detection of microcystins from cyanobacterial water blooms in Thailand fresh water. Phycol. Res. 1998, 46, 25–29. [Google Scholar] [CrossRef]
- Okello, W.; Ostermaier, V.; Portmann, C.; Gademann, K.; Kurmayer, R. Spatial isolation favours the divergence in microcystin net production by Microcystis in Ugandan freshwater lakes. Water Res. 2010, 44, 2803–2814. [Google Scholar] [CrossRef] [PubMed]
- Gademann, K.; Portmann, C.; Blom, J.F.; Zeder, M.; Jüttner, F. Multiple toxin production in the cyanobacterium Microcystis: Isolation of the toxic protease inhibitor cyanopeptolin 1020. J. Nat. Prod. 2010, 73, 980–984. [Google Scholar] [CrossRef] [PubMed]
- Namikoshi, M.; Rinehart, K.L.; Sakai, R.; Sivonen, K.; Carmichael, W.W. Structures of three new cyclic heptapeptide hepatotoxins produced by the cyanobacterium (blue-green alga) Nostoc sp. strain 152. J. Org. Chem. 1990, 55, 6135–6139. [Google Scholar] [CrossRef]
- Sivonen, K.; Namikoshi, M.; Evans, W.R.; Fardig, M.; Carmichael, W.W.; Rinehart, K.L. Three new microcystins, cyclic heptapeptide hepatotoxins, from Nostoc sp. strain 152. Chem. Res. Toxicol. 1992, 5, 464–469. [Google Scholar] [CrossRef] [PubMed]
- Sivonen, K.; Carmichael, W.W.; Namikoshi, M.; Rinehart, K.L.; Dahlem, A.M.; Niemela, S.I. Isolation and characterization of hepatotoxic microcystin homologs from the filamentous freshwater cyanobacterium Nostoc sp. strain 152. Appl. Environ. Microbiol. 1990, 56, 2650–2657. [Google Scholar] [PubMed]
- Oksanen, I.; Jokela, J.; Fewer, D.P.; Wahlsten, M.; Rikkinen, J.; Sivonen, K. Discovery of rare and highly toxic microcystins from lichen-associated cyanobacterium Nostoc sp. strain IO-102-I. Appl. Environ. Microbiol. 2004, 70, 5756–5763. [Google Scholar] [CrossRef] [PubMed]
- Bolch, C.; Blackburn, S. Isolation and purification of Australian isolates of the toxic cyanobacterium; Microcystis aeruginosa; Kütz. J. Appl. Phycol. 1996, 8, 5–13. [Google Scholar] [CrossRef]
- Fujii, K.; Ikai, Y.; Mayumi, T.; Oka, H.; Suzuki, M.; Harada, K.-I. A nonempirical method using LC/MS for determination of the absolute configuration of constituent amino acids in a peptide: Elucidation of limitations of Marfey’s method and of its separation mechanism. Anal. Chem. 1997, 69, 3346–3352. [Google Scholar] [CrossRef]
- Marfey, P. Determination of d-amino acids. II. Use of a bifunctional reagent, 1,5-difluoro-2,4-dinitrobenzene. Carlsberg Res. Commun. 1984, 49, 591–596. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Puddick, J.; Prinsep, M.R.; Wood, S.A.; Kaufononga, S.A.F.; Cary, S.C.; Hamilton, D.P. High Levels of Structural Diversity Observed in Microcystins from Microcystis CAWBG11 and Characterization of Six New Microcystin Congeners. Mar. Drugs 2014, 12, 5372-5395. https://doi.org/10.3390/md12115372
Puddick J, Prinsep MR, Wood SA, Kaufononga SAF, Cary SC, Hamilton DP. High Levels of Structural Diversity Observed in Microcystins from Microcystis CAWBG11 and Characterization of Six New Microcystin Congeners. Marine Drugs. 2014; 12(11):5372-5395. https://doi.org/10.3390/md12115372
Chicago/Turabian StylePuddick, Jonathan, Michèle R. Prinsep, Susanna A. Wood, Sangata A. F. Kaufononga, Stephen Craig Cary, and David P. Hamilton. 2014. "High Levels of Structural Diversity Observed in Microcystins from Microcystis CAWBG11 and Characterization of Six New Microcystin Congeners" Marine Drugs 12, no. 11: 5372-5395. https://doi.org/10.3390/md12115372
APA StylePuddick, J., Prinsep, M. R., Wood, S. A., Kaufononga, S. A. F., Cary, S. C., & Hamilton, D. P. (2014). High Levels of Structural Diversity Observed in Microcystins from Microcystis CAWBG11 and Characterization of Six New Microcystin Congeners. Marine Drugs, 12(11), 5372-5395. https://doi.org/10.3390/md12115372







