Abstract
Pure compound screening has identified the dioxothiazino-quinoline-quinone ascidian metabolite ascidiathiazone A (2) to be a moderate growth inhibitor of Trypanosoma brucei rhodesiense (IC50 3.1 μM) and Plasmodium falciparum (K1 dual drug resistant strain) (IC50 3.3 μM) while exhibiting low levels of cytotoxicity (L6, IC50 167 μM). A series of C-7 amide and Δ2(3) analogues were prepared that explored the influence of lipophilicity and oxidation state on observed anti-protozoal activity and selectivity. Little variation in anti-malarial potency was observed (IC50 0.62–6.5 μM), and no correlation was apparent between anti-malarial and anti-T. brucei activity. Phenethylamide 7e and Δ2(3)-glycine analogue 8k exhibited similar anti-Pf activity to 2 but with slightly enhanced selectivity (SI 72 and 93, respectively), while Δ2(3)-phenethylamide 8e (IC50 0.67 μM, SI 78) exhibited improved potency and selectivity towards T. brucei rhodesiense compared to the natural product hit. A second series of analogues were prepared that replaced the quinoline ring of 2 with benzofuran or benzothiophene moieties. While esters 10a/10b and 15 were once again found to exhibit cytotoxicity, carboxylic acid analogues exhibited potent anti-Pf activity (IC50 0.34–0.035 μM) combined with excellent selectivity (SI 560–4000). In vivo evaluation of a furan carboxylic acid analogue against P. berghei was undertaken, demonstrating 85.7% and 47% reductions in parasitaemia with ip or oral dosing respectively.
1. Introduction
Natural products have historically played an important role in the discovery of new treatments for malaria [1]. From quinine and artemisinin [2,3] starting points, a diverse range of anti-malarials have been developed and have been the mainstay of frontline treatment for decades. Unfortunately with time has come loss of therapeutic efficacy due to the growing prevalence of drug resistant strains [4]. In the hunt for novel scaffolds from which to develop the next generation of anti-malarials, a structurally-diverse array of natural products, including those obtained from marine organisms, have been reported to exhibit activity towards Plasmodium falciparum [5,6,7].
As part of our own continuing search for new leads for the development of treatments for neglected human diseases [8,9,10,11,12] we have screened a library of synthesized and isolated marine natural products against a panel of four parasitic protozoa: Trypanosoma brucei rhodesiense, Trypanosoma cruzi, Leishmania donovani and Plasmodium falciparum K1 dual drug-resistant strain, with concurrent counter-screening for toxicity towards the non-malignant L6 rat myoblast cell line. We recently disclosed details of the first hit from this screen, the previously reported anti-inflammatory polyamine diamide ascidian metabolite orthidine F (1) [13,14,15] (Figure 1).
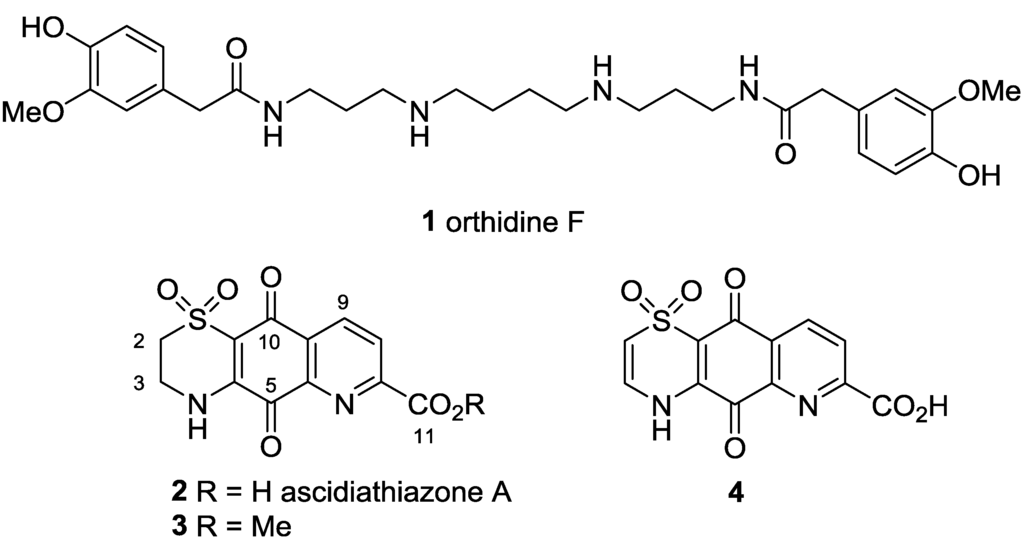
Figure 1.
Structures of orthidine F (1), ascidiathiazone A (2) and analogues 3 and 4.
A second series of hits identified in this screening program were ascidiathiazone A (2), also previously reported by us as an anti-inflammatory alkaloid from a New Zealand ascidian, and synthetic analogues 3 and 4 [16]. The anti-protozoal evaluation of 2 (Table 1, entry 1) established the natural product to be a moderately potent in vitro growth inhibitor of P. falciparum K1 strain (IC50 3.3 μM) and Trypanosoma brucei rhodesiense (IC50 3.1 μM) while being effectively inactive towards T. cruzi and Leishmania donovani and exhibiting low levels of cytotoxicity against a mammalian cell-line (L6, IC50 170 μM). Similar levels of potency and selectivity were observed for ester 3 (Table 1, entry 2), while Δ2(3) analogue 4 (Table 1, entry 3) exhibited more potent anti-malarial activity (IC50 0.6 μM) with enhanced selectivity (SI Pf 300). Herein we report the results of a preliminary structure-activity relationship study investigating the influence of C-2 amide functionalization and thiazine-Δ2(3) oxidation on the biological activity of 2. In addition, we report that novel furan and thiophene analogues of 2 exhibit potent in vitro anti-malarial activity and that one analogue exhibits in vivo activity towards P. berghei.

Table 1.
Anti-protozoal, cytotoxic activities and clogP values of 2–4, 7a–h, j, 8a–k.
| Entry | Compound | IC50 (μM) a | SI Pf g | clogP h | ||||
|---|---|---|---|---|---|---|---|---|
| T. b. rhod. b | T. cruzi c | L. don. d | P. falc. K1 e | L6 f | ||||
| 1 | 2 | 3.1 | >290 | 270 | 3.3 | 170 | 50 | −1.1 ± 1.1 |
| 2 | 3 | 6.6 | 180 | 31 | 2.5 | 140 | 56 | −0.5 ± 0.9 |
| 3 | 4 | 4.0 | >290 | 190 | 0.60 | 180 | 300 | −1.1 ± 1.0 |
| 4 | 7a | 5.5 | 63 | >280 | 0.94 | 24 | 26 | 0.3 ± 0.5 |
| 5 | 7b | 1.8 | 15 | 29 | 0.62 | 23 | 37 | 0.8 ± 0.6 |
| 6 | 7c | 3.9 | 15 | 48 | 1.1 | 12 | 10 | 2.3 ± 0.6 |
| 7 | 7d | 1.9 | 43 | 21 | 1.1 | 15 | 14 | 0.6 ± 0.5 |
| 8 | 7e | 2.4 | 140 | 160 | 1.5 | 110 | 72 | 0.9 ± 0.5 |
| 9 | 7f | 2.4 | 27 | 47 | 1.4 | 13 | 10 | 1.4 ± 0.4 |
| 10 | 7g | 3.4 | 41 | 83 | 1.6 | 24 | 15 | 2.1 ± 0.6 |
| 11 | 7h | >150 | 53 | 170 | 2.4 | 41 | 17 | −0.6 ± 0.7 |
| 12 | 7j | 120 | 250 | >260 | 3.4 | 110 | 31 | −0.9 ± 0.5 |
| 13 | 8a | 3.7 | 63 | >280 | 0.70 | 23 | 33 | 0.3 ± 0.8 |
| 14 | 8b | 3.6 | 48 | >270 | 1.5 | 4.8 | 3 | 0.8 ± 0.8 |
| 15 | 8c | 2.4 | 42 | 53 | 0.98 | 6.5 | 6 | 2.2 ± 0.8 |
| 16 | 8d | 4.2 | 160 | >250 | 4.7 | 34 | 7 | 0.8 ± 0.6 |
| 17 | 8e | 0.67 | 140 | 160 | 6.5 | 52 | 8 | 0.9 ± 0.7 |
| 18 | 8f | 5.9 | 59 | >240 | 1.2 | 6.5 | 5 | 1.3 ± 0.7 |
| 19 | 8g | 2.5 | 42 | 150 | 1.1 | 4.9 | 4 | 2.1 ± 0.7 |
| 20 | 8h | 10 | 150 | 230 | 1.7 | 50 | 29 | −0.6 ± 0.8 |
| 21 | 8i | 13 | 140 | 150 | 1.5 | 99 | 67 | −1.0 ± 0.9 |
| 22 | 8j | 35 | 160 | 220 | 1.8 | 100 | 57 | −1.1 ± 1.0 |
| 23 | 8k | 42 | 160 | >280 | 1.2 | 110 | 93 | −1.2 ± 0.7 |
| Melarsoprol i | 0.005 | |||||||
| Benznidazole i | 1.8 | |||||||
| Miltefosine i | 0.53 | |||||||
| Chloroquine i | 0.28 | |||||||
| Podophyllotoxin i | 0.019 | |||||||
a IC50 values reported are the average of two independent assays. Assay protocols are described in [5]; b Trypanosoma brucei rhodesiense, STIB 900 strain, trypomastigotes stage; c Trypanosoma cruzi, Tulahuen C4 strain, amastigotes stage; d Leishmania donovani, MHOM-ET-67/L82 strain, amastigote/axenic stage; e Plasmodium falciparum, K1 strain, IEF stage; f L6 rat skeletal myoblast cell line; g Selectivity index for P. falciparum = IC50 L6/IC50 Pf; h cLogP calculated using ALOGPS 2.1, as described in [17,18]; i Melarsoprol, benznidazole, miltefosine, chloroquine and podophyllotoxin were used as positive controls.
2. Results and Discussion
2.1. Chemistry
We undertook a preliminary structure-activity relationship study to explore the effect of carboxylic acid functionalization and thiazine ring oxidation state towards the observed anti-protozoal activity of 2. Efforts to directly prepare amide derivatives of 2 by reaction of the synthesized natural product [16] with various amines in the presence of peptide coupling reagents, led to the formation of complex product mixtures and low yields (data not shown). Instead we made use of a longer four step reaction sequence (Scheme 1). Commercially available 8-hydroxyquinoline-2-carboxylic acid was converted to amides 5a–5j by reaction with the appropriate amine using PyBOP as the coupling agent in DMF. Subsequent oxidation using PIFA (phenyliodine bis(trifluoroacetate)) in MeCN/H2O yielded unstable quinones 6a–6j.
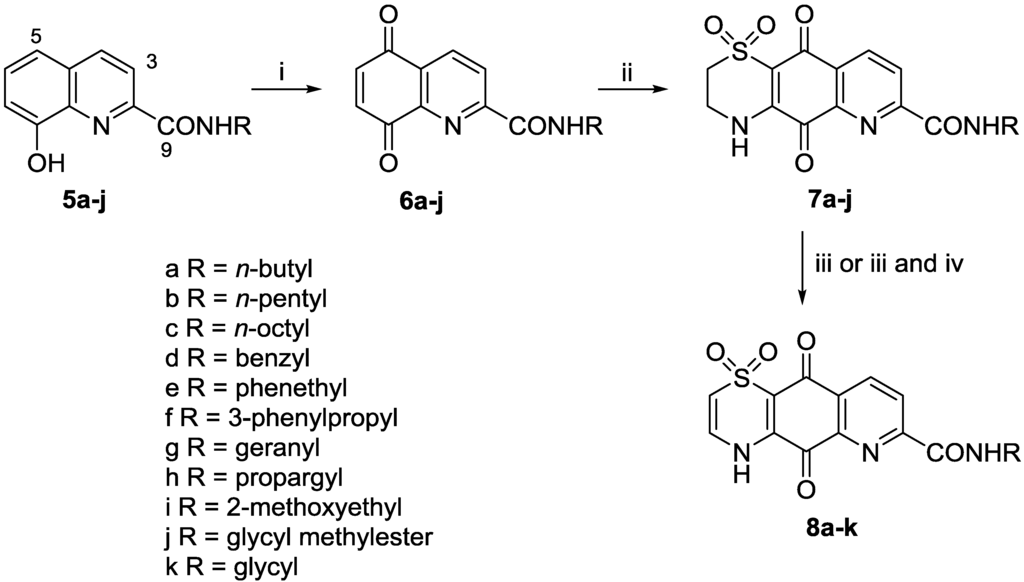
Scheme 1.
General reaction sequence for the preparation of analogues 7a–j and 8a–k. Reagents and conditions: (i) PIFA (2–3 equiv.), MeCN/H2O, 0 °C, 20 min; (ii) Hypotaurine (0.8 equiv.), CeCl3·7H2O, MeCN/EtOH, rt, 2 days; (iii) 2 N NaOH, DMF, rt, 2 h; (iv) SOCl2, MeOH, 0 °C then rt, then 65 °C, 2 h, 93% yield.
Previous studies by ourselves [16,19] and others [20,21] have found that hypotaurine addition to quinones typically yields a mixture of regio-isomeric thiazine adducts. In the present study, we found that slow addition of a dilute solution of hypotaurine in MeCN/EtOH at room temperature afforded, after filtration and washing, analogues 7a–7j in yields of 14%, 27%, 57%, 17%, 49%, 57%, 29%, 29%, 26% and 20%, respectively. The regio-isomeric identity of the product in each case was established by 2D-NMR data, particularly HMBC experiments optimized for 8.3 and 2.0 Hz, which showed correlations from H-9 to quinone C-10 (8.3 Hz) and from NH-4 to quinone C-5 (2.0 Hz) [16]. Reaction of each of 7a–7j with 2 N NaOH in DMF for 2 h [16] afforded the desired hydrolysed and autoxidised Δ2(3)-thiazine analogues 8a–8i and 8k in variable yield (30%–83%). In the specific case of the glycine methylester 7j the product of this reaction was the Δ2(3)-thiazino carboxylic acid 8k, methylation of which (SOCl2, MeOH, 93% yield) afforded ester 8j.
Thiophene analogues of ascidiathiazone A were prepared (Scheme 2) starting from the literature quinone 9 [22]. Reaction with hypotaurine yielded two isomeric products 10a and 10b in a ratio of 1:0.3, as determined by NMR. Despite extensive attempts using chromatography, the isomers could not be separated and so were used as a mixture in the following steps. The regio-isomeric identity of 10a and 10b could not be established, as no relevant long range 1H-13C correlations were observed in HMBC data. Acid-mediated ester hydrolysis afforded carboxylic acids 11a and 11b, again characterized as an inseparable 1:0.3 mixture. HMBC data obtained for this isomeric mixture however was able to establish that the major regio-isomer was 11a as shown. Thus correlations observed between the major isomer H-8 resonance (δH 7.84) to quinonoid resonance δC 171.7 (C-9) and from the thiazine NH (δH 9.31) to a second quinonoid resonance δC 173.1 (C-5) confirmed the identity of 11a. In the case of base hydrolysis/autoxidation, reaction of the isomeric mixture 10a/10b with 1N NaOH in a biphasic reaction in EtOAc, yielded the expected Δ2(3) product 12.
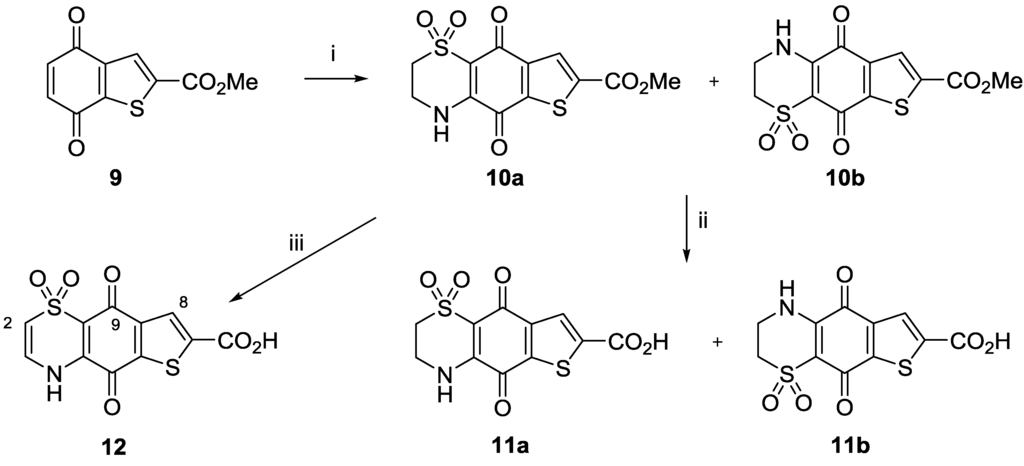
Scheme 2.
Preparation of thiophene analogues 10a/10b, 11a/11b and 12. Reagents and conditions: (i) Hypotaurine (1 equiv.), CeCl3·7H2O (1 equiv.), MeCN/EtOH, rt, 2 days, 18% yield (10a + 10b); (ii) conc. HCl, rt, 5 h, 57% yield (11a + 11b); (iii) 1 N NaOH, EtOAc, rt, 1 h, 78% yield.
Column chromatography in this case was successful in affording the major regio-isomeric product in pure form. HMBC data analysis, in particular the observation of correlations from H-2 (δH 6.57) and H-8 (δH 7.82) to the same quinonoid carbon resonance at δC 175.2 (C-9) established the dioxothiazine ring regiochemistry of 12 as shown.
A series of furan analogues were prepared in analogous fashion, this time starting from commercially available 7-methoxy-benzofuran-2-carboxylic acid ethyl ester 13 (Scheme 3). Oxidation using acidified ceric ammonium sulfate afforded quinone 14 in 85% yield. Slow addition of hypotaurine to the quinone afforded a single regio-isomer 15 of the expected dioxothiazine product (43% yield). As demonstrated earlier, acidic hydrolysis of ester 15 yielded the carboxylic acid 16 (63% yield), while biphasic 1N NaOH/EtOAc hydrolysis and autoxidation afforded the Δ2(3) carboxylic acid 17 in 47% yield.
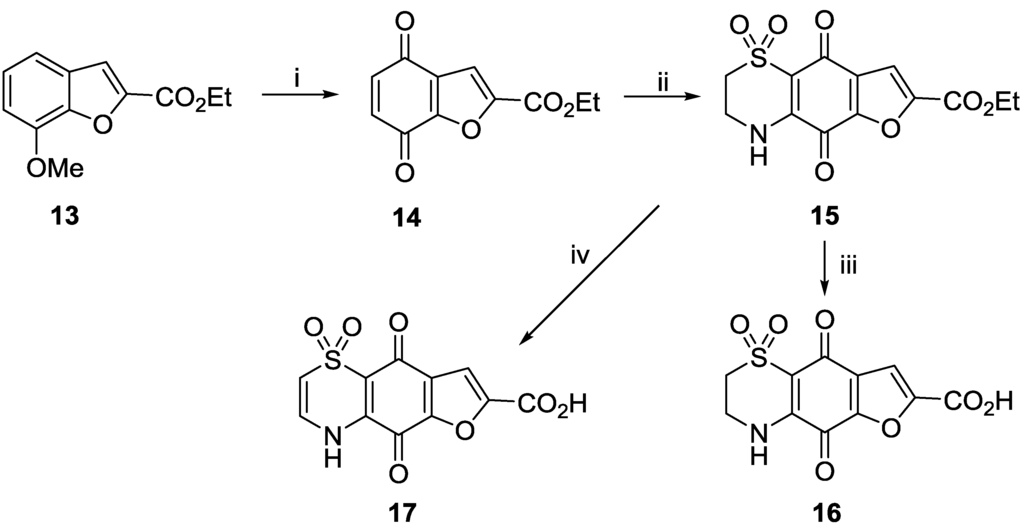
Scheme 3.
Preparation of furan analogues 15–17. Reagents and conditions: (i) (NH4)4Ce(SO4)4·2H2O, MeCN/H2SO4, 60 °C, 90 min, 85% yield; (ii) Hypotaurine (1 equiv.), CeCl3·7H2O (0.5 equiv.), MeCN/EtOH, rt, 1 days, 43% yield; (iii) conc. HCl, 100 °C, 2 h, 63% yield; (iv) 1 N NaOH, EtOAc, rt, 2 h, 47% yield.
2.2. Biological Activities
2.2.1. In Vitro Biological Evaluation
The library of target analogues were screened against a set of four protozoa and for cytotoxicity towards the rat skeletal myoblast cell line L6 and the results are summarized in Table 1 (amide and oxidized analogues of 2) and Table 2 (thiophene and ester analogues). All of the amide analogues 7a–h,j evaluated were either equipotent or slightly more active against P. falciparum than the natural product 2. A similar observation was made for activities towards T. brucei rhodesiense, except for propargyl 7h (Table 1, entry 11) and glycyl ester 7j (Table 1, entry 12) both of which were significantly less active than 2. Notable in this series, unfortunately, was the lack of selectivity with most analogues exhibiting selectivity indices (SI) of 40 or less. Of this sub-set, only phenethyl amide 7e (Table 1, entry 8) exhibited anti-protozoal activity and cytotoxic selectivity similar to those observed for 2. The corresponding Δ2(3) analogues 8a–k while being typically equipotent or slightly more active against P. falciparum, were on the whole more cytotoxic with low selectivity. Significant amongst the series were the Δ2(3) phenethyl amide 8e (Table 1, entry 17), which was the most active anti-T. brucei rhodesiense analogue, and ether 8i, ester 8j and carboxylic acid 8k (Table 1, entries 21–23) which maintained the anti-Pf activity of 2 but with modestly enhanced selectivity. There was no apparent correlation between calculated logP and observed biological activity (Table 1).

Table 2.
Anti-protozoal and cytotoxic activities of 10a/10b, 11a/11b, 12, 15–17.
| Entry | Compound | IC50 (μM) a | SI Pf g | clogP h | ||||
|---|---|---|---|---|---|---|---|---|
| T. b. rhod.b | T. cruzic | L. don. d | P. falc. K1 e | L6 f | ||||
| 1 | 10a/10b | 0.39 | 0.51 | 6.3 | 0.028 | 0.52 | 18 | 0.1 ± 0.8 |
| 2 | 11a/11b | 5.4 | >290 | 260 | 0.086 | 230 | 2700 | −0.2 ± 0.7 |
| 3 | 12 | 2.2 | 210 | >290 | 0.035 | 140 | 4000 | −0.2 ± 0.7 |
| 4 | 15 | 1.1 | 4.7 | 40 | 0.11 | 5.1 | 46 | −0.1 ± 0.8 |
| 5 | 16 | 7.5 | >300 | >300 | 0.12 | 290 | 2400 | −0.8 ± 0.7 |
| 6 | 17 | 2.7 | >300 | 120 | 0.34 | 190 | 560 | −0.8 ± 0.9 |
| Melarsoprol i | 0.01 | |||||||
| Benznidazole i | 1.4 | |||||||
| Miltefosine i | 0.53 | |||||||
| Chloroquine i | 0.28 | |||||||
| Podophyllotoxin i | 0.019 | |||||||
a IC50 values reported are the average of two independent assays. Assay protocols are described in [5]; b Trypanosoma brucei rhodesiense, STIB 900 strain, trypomastigotes stage; c Trypanosoma cruzi, Tulahuen C4 strain, amastigotes stage; d Leishmania donovani, MHOM-ET-67/L82 strain, amastigote/axenic stage; e Plasmodium falciparum, K1 strain, IEF stage; f L6 rat skeletal myoblast cell line; g Selectivity index for P. falciparum respectively = IC50 L6/IC50 Pf; h cLogP calculated using ALOGPS 2.1, as described in [17,18]; i Melarsoprol, benznidazole, miltefosine, chloroquine and podophyllotoxin were used as positive controls.
Thiophene and furan analogues 10a/10b, 11a/11b, 12, and 15–17 were evaluated against the same selection of protozoa and for cytotoxicity (Table 2). Potent anti-Pf activity was observed for the thiophene examples, with the isomerically pure carboxylic acid 12 (Table 2, entry 3) showing a desirable combination of nanomolar potency (Pf IC50 35 nM) and excellent selectivity (SI Pf 4000). The furan analogues 15–17 (Table 2, entries 4–6) were slightly less active towards P. falciparum, exhibiting IC50′s in the 110–340 nm range, with carboxylic acid 16 (Table 2, entry 5) exhibiting the best selectivity (SI Pf 2400). It is interesting to note the broad-range activities of esters 10a/10b (Table 2, entry 1) and 15 (Table 2, entry 4): such pan-panel activity suggests the presence of an underlying general cytotoxic mechanism for these analogues. Once again, there was no apparent correlation between biological activity and calculated logP values.
2.2.2. In Vivo Anti-Malarial Evaluation
Furan carboxylic acid analogue 16 was selected for preliminary proof-of-principle in vivo evaluation in Plasmodium berghei infected mice. Preliminary ip acute toxicity of 16 showed no toxicity up to the highest test dose of 150 mg/kg. Using a standard test protocol [23], a repeated ip dose of 50 (mg/kg)/day for four days led to an 85.7% reduction in parasitaemia, and an increase in mean survival time from 4–6 days (untreated control) to 9.6 days. Switching to an oral dosing experiment (100 mg/kg once per day for 4 days) yielded a 47% reduction of parasitaemia. Although not considered significant, these levels of activity for both ip and po dosing clearly identifies heterocyclic dioxothiazinoquinone carboxylic acids to be a novel anti-malarial drug scaffold warranting further structure-activity relationship studies.
3. Experimental Section
3.1. General
HRMS data were acquired on a Bruker micrOTOF-QII mass spectrometer. Infrared spectra were recorded on a Perkin-Elmer Spectrum 100 Fourier-transform IR spectrometer equipped with a universal ATR accessory. Melting points were obtained on an Electrothermal melting point apparatus and are uncorrected. NMR spectra were recorded using either a Bruker Avance DRX 300 or 400 spectrometer operating at 300 MHz or 400 MHz for 1H nuclei and 75 MHz or 100 MHz for 13C nuclei. Resonance assignments were made by interpretation of 2D data. NMR assignments marked by an asterisk are interchangeable. Proto-deutero solvent signals were used as internal references (DMSO-d6: δH 2.50, δC 39.52; CDCl3: δH 7.25, δC 77.0; CD3OD: δH 3.30, δC 49.05). Flash column chromatography was performed using reversed-phase Merck Lichroprep RP-18, or Kieselgel 60 PF silica gel. Thin layer chromatography used 0.2 mm thick plates of Kiesegel F254 (Merck, Manakau, New Zealand). The syntheses of 2–4 [16] and 9 [22] have been reported previously.
3.2. Synthetic Procedures
3.2.1. General Procedure for the Preparation of 8-Hydroxyquinoline-2-carboxamides 5a–5j
To a solution of 8-hydroxyquinoline-2-carboxylic acid and PyBOP (1.25 equiv.) in dry DMF (3–6 mL), amine (1–2 equiv.) and triethylamine (1.25 equiv.) were added under N2. The reaction mixture was then stirred under N2 at rt for 12 h, after which time the mixture was dried in vacuo. The residue was purified by reversed-phase C18 flash column chromatography (0%–80% MeOH in H2O (0.05% TFA)) and silica gel column chromatography (0%–1% MeOH in CH2Cl2).
3.2.1.1. N-n-Butyl-8-hydroxyquinoline-2-carboxamide (5a)
From 8-hydroxyquinoline-2-carboxylic acid (100 mg, 0.529 mmol), PyBOP (330 mg, 0.64 mmol), n-butylamine (104 µL, 1.05 mmol) and triethylamine (88 µL, 0.632 mmol) in DMF (6 mL) to give 5a as a yellow oil (108 mg, 84% yield).
Rf = 0.68 (1% MeOH/CH2Cl2); IR νmax (ATR) 3291, 1650, 1539, 1502, 1465, 1159 cm−1; 1H NMR (CDCl3, 400 MHz) δH 8.34 (1H, d, J = 8.4 Hz, H-3), 8.29 (1H, d, J = 8.4 Hz, H-4), 8.01 (1H, br s, NH-2′), 7.86 (1H, br s, OH), 7.53 (1H, t, J = 7.8 Hz, H-6), 7.40 (1H, d, J = 8.4 Hz, H-5), 7.23 (1H, d, J = 8.0 Hz, H-7), 3.53 (2H, dt, J = 7.2, 7.2 Hz, H2-3′), 1.63 (2H, p, J = 7.3 Hz, H2-4′), 1.40 (2H, sex., J = 7.6 Hz, H2-5′), 0.92 (3H, t, J = 7.6 Hz, H3-6′); 13C NMR (CDCl3, 100 MHz) δC 164.1 (C-1′), 152.2 (C-8), 148.2 (C-2), 137.8 (C-4), 136.1 (C-8a), 129.7 (C-4a), 129.2 (C-6), 119.9 (C-3), 118.3 (C-5), 111.2 (C-7), 39.5 (C-3′), 31.8 (C-4′), 20.2 (C-5′), 13.8 (C-6′); (+)-ESIMS m/z 245 [M + H]+; (+)-HRESIMS m/z 245.1287 [M + H]+ (calcd. for C14H17N2O2, 245.1285).
3.2.1.2. N-n-Pentyl-8-hydroxyquinoline-2-carboxamide (5b)
From 8-hydroxyquinoline-2-carboxylic acid (50 mg, 0.26 mmol), PyBOP (165 mg, 0.32 mmol), n-pentylamine (61 µL, 0.53 mmol) and triethylamine (44 µL, 0.32 mmol) in DMF (3 mL) to give 5b as a colorless oil (65 mg, 97% yield).
Rf = 0.65 (5% MeOH/CH2Cl2); IR νmax (ATR) 3266, 2929, 1647, 1500 cm−1; 1H NMR (CDCl3, 400 MHz) δH 8.46 (1H, t, J = 5.8 Hz, NH-2′), 8.33 (1H, d, J = 8.6 Hz, H-3), 8.24 (1H, d, J = 8.6 Hz, H-4), 7.49 (1H, t, J = 8.2 Hz, H-6), 7.34 (1H, d, J = 8.2 Hz, H-5), 7.20 (1H, d, J = 8.2 Hz, H-7), 3.46 (2H, dt, J = 7.4, 5.8 Hz, H2-3′), 1.58 (2H, p, J = 7.4 Hz, H2-4′), 1.30–1.18 (4H, m, H2-5′/H2-6′), 0.81 (3H, t, J = 7.4 Hz, H3-7′); 13C NMR (CDCl3, 100 MHz) δC 164.4 (C-1′), 152.4 (C-8), 148.0 (C-2), 137.6 (C-4), 136.6 (C-8a), 129.7 (C-4a), 129.2 (C-6), 119.7 (C-3), 118.1 (C-5), 111.2 (C-7), 39.8 (C-3′), 29.4 (C-4′), 29.1 (C-5′), 22.3 (C-6′), 13.8 (C-7′); (+)-ESIMS m/z 281 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 281.1259 (calcd. for C15H18N2NaO2, 281.1260).
3.2.1.3. N-n-Octyl-8-hydroxyquinoline-2-carboxamide (5c)
From 8-hydroxyquinoline-2-carboxylic acid (50 mg, 0.26 mmol), PyBOP (165 mg, 0.32 mmol), n-octylamine (87 µL, 0.527 mmol) and triethylamine (44 µL, 0.32 mmol) in DMF (3 mL) to give 5c as a colorless oil (73 mg, 94% yield).
Rf = 0.80 (5% MeOH/CH2Cl2); IR νmax (ATR) 3297, 2924, 1648, 1501 cm−1; 1H NMR (CDCl3, 400 MHz) δH 8.69 (1H, t, J = 5.7 Hz, NH-2′), 8.33 (1H, d, J = 8.6 Hz, H-3), 8.22 (1H, d, J = 8.6 Hz, H-4), 7.48 (1H, t, J = 8.0 Hz, H-6), 7.32 (1H, d, J = 8.0 Hz, H-5), 7.18 (1H, d, J = 8.0 Hz, H-7), 3.45 (2H, dt, J = 7.2, 5.7 Hz, H2-3′), 1.55 (2H, p, J = 7.2 Hz, H2-4′), 1.27–1.09 (10H, m, H2-5′/H2-6′/H2-7′/H2-8′/H2-9′), 0.80 (3H, t, J = 7.2 Hz, H3-10′); 13C NMR (CDCl3, 100 MHz) δC 164.6 (C-1′), 152.5 (C-8), 147.8 (C-2), 137.6 (C-4), 136.6 (C-8a), 129.6 (C-4a), 129.2 (C-6), 119.6 (C-3), 118.1 (C-5), 111.2 (C-7), 39.9 (C-3′), 31.7 (C-6′*), 29.6 (C-4′), 29.2 (C-7′*), 29.1 (C-8′*), 27.0 (C-5′), 22.5 (C-9′*), 14.0 (C-10′); (+)-ESIMS m/z 323 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 323.1740 (calcd. for C18H24N2NaO2, 323.1730).
3.2.1.4. N-Benzyl-8-hydroxyquinoline-2-carboxamide (5d)
From 8-hydroxyquinoline-2-carboxylic acid (50 mg, 0.26 mmol), PyBOP (165 mg, 0.32 mmol), benzylamine (58 µL, 0.53 mmol) and triethylamine (44 µL, 0.32 mmol) in DMF (3 mL) to give 5d as a colorless oil (57 mg, 79% yield).
Rf = 0.72 (5% MeOH/CH2Cl2); IR νmax (ATR) 3251, 3062, 1642, 1501 cm−1; 1H NMR (CDCl3, 400 MHz) δH 8.90 (1H, br s, NH-2′), 8.29 (1H, d, J = 8.4 Hz, H-3), 8.20 (1H, d, J = 8.4 Hz, H-4), 7.48 (1H, t, J = 7.9 Hz, H-6), 7.33 (1H, d, J = 7.9 Hz, H-5), 7.25–7.13 (6H, m, H-7/2H-5′/2H-6′/H-7′), 4.61 (2H, br s, H2-3′); 13C NMR (CDCl3, 100 MHz) δC 164.6 (C-1′), 152.4 (C-8), 147.5 (C-2), 137.9 (C-4′), 137.6 (C-4), 136.5 (C-8a), 129.7 (C-4a), 129.3 (C-6), 128.5 (C-5′), 127.7 (C-6′), 127.3 (C-7′), 119.7 (C-3), 118.1 (C-5), 111.3 (C-7), 43.6 (C-3′); (+)-ESIMS m/z 301 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 301.0949 (calcd. for C17H14N2NaO2, 301.0947).
3.2.1.5. N-Phenethyl-8-hydroxyquinoline-2-carboxamide (5e)
From 8-hydroxyquinoline-2-carboxylic acid (50 mg, 0.26 mmol), PyBOP (165 mg, 0.32 mmol), phenethylamine (66 µL, 0.53 mmol) and triethylamine (44 µL, 0.32 mmol) in DMF (3 mL) to give 5e as a colorless oil (65 mg, 86% yield).
Rf = 0.65 (5% MeOH/CH2Cl2); IR νmax (ATR) 3288, 3073, 1643, 1501 cm−1; 1H NMR (CDCl3, 300 MHz) δH 8.33 (1H, d, J = 8.5 Hz, H-3), 8.28 (1H, d, J = 8.5 Hz, H-4), 7.53 (1H, t, J = 7.8 Hz, H-6), 7.40–7.20 (7H, m, H-5/H-7/2H-6′/2H-7′/H-8′), 3.77 (2H, dt, J = 7.0, 6.8 Hz, H2-3′), 2.96 (2H, t, J = 7.0 Hz, H2-4′); 13C NMR (CDCl3, 75 MHz) δC 164.1 (C-1′), 152.3 (C-8), 147.9 (C-2), 138.8 (C-5′), 137.8 (C-4), 136.5 (C-8a), 129.7 (C-4a), 129.3 (C-6), 128.8 (C-6′), 128.7 (C-7′), 126.7 (C-8′), 119.7 (C-3), 118.1 (C-5), 111.2 (C-7), 40.7 (C-3′), 35.8 (C-4′); (+)-ESIMS m/z 293 [M + H]+; (+)-HRESIMS m/z [M + H]+ 293.1292 (calcd. for C18H17N2O2, 293.1285).
3.2.1.6. N-(3-Phenylpropyl)-8-hydroxyquinoline-2-carboxamide (5f)
From 8-hydroxyquinoline-2-carboxylic acid (50 mg, 0.26 mmol), PyBOP (165 mg, 0.32 mmol), 3-phenylpropylamine (66 µL, 0.46 mmol) and triethylamine (44 µL, 0.32 mmol) in DMF (3 mL) to give 5f as a colorless oil (67 mg, 84% yield).
Rf = 0.65 (5% MeOH/CH2Cl2); IR νmax (ATR) 3257, 2929, 1647, 1500 cm−1; 1H NMR (CDCl3, 400 MHz) δH 8.64 (1H, br s, NH-2′), 8.32 (1H, d, J = 8.5 Hz, H-3), 8.20 (1H, d, J = 8.5 Hz, H-4), 7.49 (1H, t, J = 7.9 Hz, H-6), 7.33 (1H, d, J = 7.9 Hz, H-5), 7.22–7.00 (6H, m, H-7/2H-7′/2H-8′/H-9′), 3.53–3.46 (2H, m, H2-3′), 2.57 (2H, t, J = 7.2 Hz, H2-5′), 1.89 (2H, p, J = 7.2 Hz, H2-4′); 13C NMR (CDCl3, 100 MHz) δC 164.6 (C-1′), 152.5 (C-8), 147.7 (C-2), 141.2 (C-6′), 137.6 (C-4), 136.5 (C-8a), 129.6 (C-4a), 129.2 (C-6), 128.2 (C-7′), 128.1 (C-8′), 125.8 (C-9′), 119.5 (C-3), 118.1 (C-5), 111.2 (C-7), 39.4 (C-3′), 33.2 (C-5′), 31.0 (C-4′); (+)-ESIMS m/z 329 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 329.1267 (calcd. for C19H18N2NaO2, 329.1260).
3.2.1.7. N-Geranyl-8-hydroxyquinoline-2-carboxamide (5g)
From 8-hydroxyquinoline-2-carboxylic acid (50 mg, 0.26 mmol), PyBOP (165 mg, 0.32 mmol), geranylamine (98 µL, 0.53 mmol) and triethylamine (44 µL, 0.32 mmol) in DMF (3 mL) to give 5g as a colorless oil (85 mg, 100% yield).
Rf = 0.66 (5% MeOH/CH2Cl2); IR νmax (ATR) 3276, 2914, 1646, 1501 cm−1; 1H NMR (CDCl3, 300 MHz) δH 8.38 (1H, t, J = 5.6 Hz, NH-2′), 8.33 (1H, d, J = 8.5 Hz, H-3), 8.23 (1H, d, J = 8.5 Hz, H-4), 7.49 (1H, t, J = 6.1 Hz, H-6), 7.34 (1H, dd, J = 6.1, 1.1 Hz, H-5), 7.19 (1H, dd, J = 6.1, 1.1 Hz, H-7), 5.27 (1H, t, J = 6.9 Hz, H-4′), 4.99 (1H, t, J = 6.9 Hz, H-8′), 4.12 (2H, dd, J = 6.3, 6.3 Hz, H2-3′), 2.04–1.95 (2H, m, H2-7′), 1.93–1.86 (2H, m, H2-6′), 1.62 (6H, s, H3-11′/H3-12′), 1.53 (3H, s, H3-10′); 13C NMR (CDCl3, 75 MHz) δC 164.2 (C-1′), 152.4 (C-8), 148.0 (C-2), 139.7 (C-5′), 137.6 (C-4), 136.5 (C-8a), 131.6 (C-9′), 129.6 (C-4a), 129.2 (C-6), 123.7 (C-8′), 119.7 (C-4′), 119.6 (C-3), 118.1 (C-5), 111.2 (C-7), 39.4 (C-6′), 37.7 (C-3′), 26.3 (C-7′), 25.6 (C-12′), 17.6 (C-10′), 16.3 (C-11′); (+)-ESIMS m/z 325 [M + H]+; (+)-HRESIMS m/z [M + H]+ 325.1903 (calcd. for C20H25N2O2, 325.1911).
3.2.1.8. N-Propargyl-8-hydroxyquinoline-2-carboxamide (5h)
From 8-hydroxyquinoline-2-carboxylic acid (50 mg, 0.26 mmol), PyBOP (165 mg, 0.32 mmol), propargylamine (29 mg, 0.53 mmol) and triethylamine (44 µL, 0.32 mmol) in DMF (3mL) to give 5h as a colorless oil (47 mg, 80% yield).
Rf = 0.56 (10% MeOH/CH2Cl2); IR νmax (ATR) 3303, 3273, 1646, 1504 cm−1; 1H NMR (DMSO-d6, 400 MHz) δH 10.17 (1H, br s, OH-9), 9.99 (1H, t, J = 5.8 Hz, NH-2′), 8.49 (1H, d, J = 8.4 Hz, H-4), 8.14 (1H, d, J = 8.4 Hz, H-3), 7.56 (1H, t, J = 7.8 Hz, H-6), 7.46 (1H, d, J = 7.8 Hz, H-5), 7.18 (1H, d, J = 7.8, Hz, H-7), 4.23 (2H, dd, J = 5.8, 2.4 Hz, H2-3′), 3.22 (1H, t, J = 2.4 Hz, H2-5′); 13C NMR (DMSO-d6, 100 MHz) δC 163.5 (C-1′), 153.7 (C-8), 147.0 (C-2), 137.8 (C-4), 136.4 (C-8a), 129.6 (C-4a and C-6), 118.8 (C-3), 117.5 (C-5), 111.6 (C-7), 81.1 (C-4′), 73.3 (C-5′), 28.1 (C-3′); (+)-ESIMS m/z 227 [M + H]+; (+)-HRESIMS m/z [M + H]+ 227.0814 (calcd. for C13H11N2O2, 227.0815).
3.2.1.9. N-(2-Methoxyethyl)-8-hydroxyquinoline-2-carboxamide (5i)
From 8-hydroxyquinoline-2-carboxylic acid (100 mg, 0.529 mmol), PyBOP (330 mg, 0.64 mmol), 2-methoxyethylamine (92.6 µL, 1.07 mmol) and triethylamine (88 µL, 0.632 mmol) in DMF (6 mL) to give 5i as a colorless oil (106 mg, 82% yield).
Rf = 0.83 (10% MeOH/CH2Cl2); IR νmax (ATR) 3315, 1648, 1542, 1501, 1120 cm−1; 1H NMR (CDCl3, 400 MHz) δH 8.68 (1H, br s, NH-2′), 8.38 (1H, br s, OH), 8.32 (1H, d, J = 8.4 Hz, H-3), 8.26 (1H, d, J = 8.4 Hz, H-4), 7.50 (1H, t, J = 8.0 Hz, H-6), 7.36 (1H, d, J = 8.4 Hz, H-5), 7.21 (1H, d, J = 7.6 Hz, H-7), 3.73 (2H, dt, J = 5.6, 5.2 Hz, H2-3′), 3.61 (2H, t, J = 5.2 Hz, H2-4′), 3.36 (3H, s, H3-5′); 13C NMR (CDCl3, 100 MHz) δC 164.6 (C-1′), 152.6 (C-8), 147.9 (C-2), 137.6 (C-4), 136.6 (C-8a), 129.7 (C-4a), 129.3 (C-6), 119.7 (C-3), 118.1 (C-5), 111.3 (C-7), 71.3 (C-4′), 58.8 (C-5′), 39.5 (C-3′); (+)-ESIMS m/z 247 [M + H]+; (+)-HRESIMS m/z 247.1076 [M + H]+ (calcd. for C13H15N2O3, 247.1077).
3.2.1.10. N-Glycine(methylester)-8-hydroxyquinoline-2-carboxamide (5j)
From 8-hydroxyquinoline-2-carboxylic acid (100 mg, 0.529 mmol), PyBOP (330 mg, 0.64 mmol), glycine methyl ester hydrochloride (94.4 mg, 0.76 mmol) and triethylamine (220 µL, 1.58 mmol) in DMF (6 mL) to give 5j as a colorless oil (128 mg, 93% yield).
Rf = 0.86 (10% MeOH/CH2Cl2); 1H NMR (CDCl3, 400 MHz) δH 9.08 (1H, t, J = 6.1 Hz, NH-2′), 8.38 (1H, br s, OH), 8.03 (2H, s, H-3 and H-4), 7.43 (1H, t, J = 8.0 Hz, H-6), 7.23 (1H, dd, J = 8.4, 1.0 Hz, H-5), 7.13 (1H, dd, J = 7.6, 1.0 Hz, H-7), 4.27 (2H, d, J = 6.1 Hz, H2-3′), 3.72 (3H, s, H3-5′); 13C NMR (CDCl3, 100 MHz) δC 171.2 (C-4′), 165.0 (C-1′), 152.7 (C-8), 146.8 (C-2), 137.2 (C-4), 136.4 (C-8a), 129.6 (C-4a), 129.5 (C-6), 119.3 (C-3), 117.9 (C-5), 111.3 (C-7), 52.5 (C-5′), 41.3 (C-3′); (+)-ESIMS m/z 261 [M + H]+; (+)-HRESIMS m/z 261.0863 [M + H]+ (calcd. for C13H13N2O4, 261.0870).
3.2.2. General Procedure for Preparation of Quinones 6a–6j
A solution of PIFA (2–3 equiv.) in MeCN/H2O (2:1 mL) was cooled to 0 °C, followed by the addition of the appropriate 8-hydroxyquinoline-2-carboxamide in CH2Cl2 (1 mL). The dark brown suspension was stirred for 20 min. at 0 °C before being poured into a mixture of CH2Cl2 (20 mL) and H2O (30 mL). The organic phase was dried in vacuo and the crude product used in the subsequent reaction without further purification.
3.2.2.1. N-n-Butyl-5,8-dioxo-5,8-dihydroquinoline-2-carboxamide (6a)
From N-n-butyl-8-hydroxyquinoline-2-carboxamide (5a) (48 mg, 0.20 mmol), PIFA (254 mg, 0.59 mmol) to give 6a (44 mg, 85% yield) as a brown oil.
1H NMR (CDCl3, 400 MHz) δH 8.59 (1H, d, J = 8.0 Hz, H-3*), 8.56 (1H, d, J = 8.0 Hz, H-4*), 8.28 (1H, br s, NH-2′), 7.19 (1H, d, J = 10.4 Hz, H-7), 7.11 (1H, d, J = 10.4 Hz, H-6), 3.50 (2H, dt, J = 6.4, 6.4 Hz, H2-3′), 1.64 (2H, p, J = 7.3 Hz, H2-4′), 1.41 (2H, sex., J = 7.2 Hz, H2-5′), 0.94 (3H, t, J = 7.6 Hz, H3-6′); 13C NMR (CDCl3, 100 MHz) δC 183.8 (C-5), 182.6 (C-8), 162.4 (C-1′), 153.9 (C-2), 145.8 (C-8a), 139.3 (C-7*), 138.3 (C-6*), 136.5 (C-3*), 130.2 (C-4a), 126.2 (C-4*), 39.6 (C-3′), 31.6 (C-4′), 20.1 (C-5′), 13.8 (C-6′); (+)-ESIMS m/z 281 [M + Na]+; (+)-HRESIMS m/z 281.0893 [M + Na]+ (calcd. for C14H14N2NaO3, 281.0897).
3.2.2.2. N-n-Pentyl-5,8-dioxo-5,8-dihydroquinoline-2-carboxamide (6b)
From N-n-pentyl-8-hydroxyquinoline-2-carboxamide (5b) (38 mg, 0.15 mmol), PIFA (127 mg, 0.30 mmol) to give 6b (31 mg, 76% yield) as a brown oil.
1H NMR (CDCl3, 300 MHz) δH 8.57 (2H, s, H-3/H-4), 8.30 (1H, br s, NH-2′), 7.20 (1H, d, J = 10.3 Hz, H-7), 7.12 (1H, d, J = 10.3 Hz, H-6), 3.53–3.44 (2H, m, H2-3′), 1.71–1.61 (2H, m, H2-4′), 1.39–1.33 (4H, m, H2-5′/H2-6′), 0.90 (3H, t, J = 7.3 Hz, H3-7′); 13C NMR (CDCl3, 75 MHz) δC 183.7 (C-5*), 182.4 (C-8*), 162.4 (C-1′), 154.0 (C-2), 145.7 (C-8a), 139.2 (C-7), 138.2 (C-6), 136.3 (C-3*), 130.2 (C-4a), 126.1 (C-4*), 39.7 (C-3′), 29.2 (C-4′), 29.0 (C-5′), 22.2 (C-6′), 13.9 (C-7′); (+)-ESIMS m/z 295 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 295.1061 (calcd. for C15H16NaN2O3, 295.1053).
3.2.2.3. N-n-Octyl-5,8-dioxo-5,8-dihydroquinoline-2-carboxamide (6c)
From N-n-octyl-8-hydroxyquinoline-2-carboxamide (5c) (73 mg, 0.24 mmol), PIFA (127 mg, 0.30 mmol) to give 6c (64 mg, 85% yield) as a brown oil.
1H NMR (CDCl3, 300 MHz) δH 8.56 (2H, s, H-3/H-4), 8.23 (1H, br s, NH-2′), 7.18 (1H, d, J = 10.5 Hz, H-7), 7.10 (1H, d, J = 10.5 Hz, H-6), 3.48 (2H, dt, J = 6.7, 6.0 Hz, H2-3′), 1.65 (2H, p, J = 7.2 Hz, H2-4′), 1.42–1.20 (10H, br s, H2-5′/H2-6′/H2-7′/H2-8′/H2-9′), 0.86 (3H, t, J = 7.1 Hz, H3-10′); 13C NMR (CDCl3, 75 MHz) δC 183.8 (C-5), 182.5 (C-8), 162.4 (C-1′), 154.1 (C-2), 145.0 (C-8a), 139.3 (C-7), 138.2 (C-6), 136.4 (C-3*), 130.3 (C-4a), 126.2 (C-4*), 39.9 (C-3′), 31.8 (C-6′*), 29.6 (C-4′), 29.2 (C-7′*), 29.1 (C-8′*), 27.0 (C-5′), 22.6 (C-9′*), 14.0 (C-10′); (+)-ESIMS m/z 337 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 337.1531 (calcd. for C18H22N2NaO3, 337.1523).
3.2.2.4. N-Benzyl-5,8-dioxo-5,8-dihydroquinoline-2-carboxamide (6d)
From N-benzyl-8-hydroxyquinoline-2-carboxamide (5d) (51 mg, 0.18 mmol), PIFA (127 mg, 0.30 mmol) to give 6d (44 mg, 84% yield) as a brown oil.
1H NMR (CDCl3, 400 MHz) δH 8.67 (1H, br s, NH-2′), 8.60 (1H, d, J = 8.1 Hz, H-3*), 8.55 (1H, d, J = 8.1 Hz, H-4*), 7.36–7.26 (5H, m, 2H-5′/2H-6′/H-7′), 7.14 (1H, d, J = 10.6 Hz, H-7), 7.09 (1H, d, J = 10.6 Hz, H-6), 4.69 (2H, d, J = 6.3 Hz, H2-3′); 13C NMR (CDCl3, 100 MHz) δC 183.7 (C-5), 182.4 (C-8), 162.5 (C-1′), 153.8 (C-2), 145.8 (C-8a), 139.2 (C-7), 138.2 (C-6), 137.4 (C-4′), 136.4 (C-3*), 130.2 (C-4a), 128.7 (C-5′), 127.8 (C-6′), 127.5 (C-7′), 126.4 (C-4*), 43.6 (C-3′); (+)-ESIMS m/z 315 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 315.0748 (calcd. for C17H12N2NaO3, 315.0740).
3.2.2.5. N-Phenethyl-5,8-dioxo-5,8-dihydroquinoline-2-carboxamide (6e)
From N-phenethyl-8-hydroxyquinoline-2-carboxamide (5e) (58 mg, 0.20 mmol), PIFA (127 mg, 0.30 mmol) to give 6e (42 mg, 69% yield) as a brown oil.
1H NMR (CDCl3, 300 MHz) δH 8.57 (2H, s, H-3/H-4), 8.32 (1H, br s, NH-2′), 7.35–7.22 (5H, m, 2H-6′/2H-7′/H-8′), 7.17 (1H, d, J = 10.5 Hz, H-7), 7.10 (1H, d, J = 10.5 Hz, H-6), 3.75 (2H, dt, J = 7.7, 6.9 Hz, H2-3′), 2.98 (2H, t, J = 7.7 Hz, H2-4′); 13C NMR (CDCl3, 100 MHz) δC 183.7 (C-5), 182.3 (C-8), 162.4 (C-1′), 153.9 (C-2), 145.8 (C-8a), 139.3 (C-7), 138.2 (C-6), 137.4 (C-5′), 136.4 (C-3*), 130.2 (C-4a), 128.7 (C-6′*), 128.6 (C-7′*), 126.5 (C-8′), 126.1 (C-4*), 41.2 (C-3′), 35.8 (C-4′); (+)-ESIMS m/z 329 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 329.0904 (calcd. for C18H14N2NaO3, 329.0897).
3.2.2.6. N-(3-Phenpropyl)-5,8-dioxo-5,8-dihydroquinoline-2-carboxamide (6f)
From N-(3-phenylpropyl)-8-hydroxyquinoline-2-carboxamide (5f) (65 mg, 0.21 mmol), PIFA (127 mg, 0.30 mmol) to give 6f (45 mg, 67% yield) as a brown oil.
1H NMR (CDCl3, 400 MHz) δH 8.57 (2H, s, H-3/H-4), 8.34 (1H, br s, NH-2′), 7.33–7.19 (5H, m, 2H-7′/2H-8′/H-9′), 7.17 (1H, d, J = 10.4 Hz, H-7), 7.10 (1H, d, J = 10.4 Hz, H-6), 3.54 (2H, dt, J = 7.5, 6.8 Hz, H2-3′), 2.72 (2H, t, J = 7.5 Hz, H2-5′), 2.01 (2H, p, J = 7.5 Hz, H2-4′); 13C NMR (CDCl3, 100 MHz) δC 183.7 (C-5), 182.4 (C-8), 162.4 (C-1′), 153.9 (C-2), 145.7 (C-8a), 139.2 (C-7), 138.2 (C-6), 137.4 (C-6′), 136.4 (C-3*), 130.2 (C-4a), 128.4 (C-7′), 128.3 (C-8′), 126.1 (C-9′), 125.9 (C-4*), 39.3 (C-3′), 33.2 (C-5′), 31.0 (C-4′); (+)-ESIMS m/z 343 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 343.1063 (calcd. for C19H16N2NaO3, 343.1053).
3.2.2.7. N-Geranyl-5,8-dioxo-5,8-dihydroquinoline-2-carboxamide (6g)
From N-geranyl-8-hydroxyquinoline-2-carboxamide (5g) (48 mg, 0.15 mmol), PIFA (127 mg, 0.30 mmol) to give 6g (35 mg, 69% yield) as a brown oil.
1H NMR (CDCl3, 400 MHz) δH 8.57 (1H, d, J = 8.1 Hz, H-3), 8.54 (1H, d, J = 8.1 Hz, H-4), 8.18 (1H, br s, NH-2′), 7.19 (1H, d, J = 10.5 Hz, H-7), 7.09 (1H, d, J = 10.5 Hz, H-6), 5.28 (1H, t, J = 5.6 Hz, H-4′), 5.06 (1H, t, J = 6.8 Hz, H-8′), 4.10 (2H, dd, J = 6.4, 5.6 Hz, H2-3′), 2.11–2.05 (2H, m, H2-7′), 2.04–1.99 (2H, m, H2-6′), 1.72 (3H, s, H3-11′), 1.65 (3H, s, H3-12′), 1.58 (3H, s, H3-10′); 13C NMR (CDCl3, 100 MHz) δC 183.8 (C-5), 182.5 (C-8), 162.2 (C-1′), 154.1 (C-2), 145.8 (C-8a), 140.0 (C-5′), 139.3 (C-7), 138.2 (C-6), 136.3 (C-3*), 131.7 (C-9′), 130.3 (C-4a), 126.2 (C-4*), 123.8 (C-8′), 119.4 (C-4′), 39.5 (C-6′), 37.7 (C-3′), 26.3 (C-7′), 25.6 (C-12′), 17.7 (C-10′), 16.4 (C-11′); (+)-ESIMS m/z 361 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 361.1526 (calcd. for C20H22N2NaO3, 361.1523).
3.2.2.8. N-Propargyl-5,8-dioxo-5,8-dihydroquinoline-2-carboxamide (6h)
From N-propargyl-8-hydroxyquinoline-2-carboxamide (5h) (45 mg, 0.20 mmol), PIFA (229 mg, 0.53 mmol) to give 6h (40 mg, 83% yield) as a brown oil.
1H NMR (CDCl3, 400 MHz) δH 8.57 (2H, br s, H-3/H-4), 7.19 (1H, d, J = 10.5 Hz, H-7), 7.12 (1H, d, J = 10.5 Hz, H-6), 4.30 (2H, dd, J = 5.6, 2.5 Hz, H2-3′), 2.28 (1H, t, J = 2.5 Hz, H-5′); 13C NMR (CDCl3, 100 MHz) δC 183.6 (C-5), 182.4 (C-8), 162.3 (C-1′), 153.3 (C-2), 145.8 (C-8a), 139.3 (C-6*), 138.3 (C-7*), 136.5 (C-3*), 130.2 (C-4a), 126.4 (C-4*), 78.9 (C-4′), 71.8 (C-5′), 29.3 (C-3′); (+)-ESIMS m/z 263 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 263.0431 (calcd. for C13H8N2NaO3, 263.0427).
3.2.2.9. N-(2-Methoxyethyl)-5,8-dioxo-5,8-dihydroquinoline-2-carboxamide (6i)
From N-(2-methoxyethyl)-8-hydroxyquinoline-2-carboxamide (5i) (86 mg, 0.35 mmol), PIFA (300 mg, 0.70 mmol) to give 6i (78 mg, 86% yield) as a brown oil.
1H NMR (CDCl3, 400 MHz) δH 8.58 (2H, s, H-3/H-4), 8.49 (1H, br s, NH-2′), 7.19 (1H, d, J = 10.4 Hz, H-7), 7.10 (1H, d, J = 10.4 Hz, H-6), 3.72 (2H, dt, J = 5.6, 5.2 Hz, H2-3′), 3.61 (2H, t, J = 5.2 Hz, H2-4′), 3.40 (3H, s, H3-5′); 13C NMR (CDCl3, 100 MHz) δC 183.8 (C-5), 182.3 (C-8), 162.7 (C-1′), 153.9 (C-2), 145.9 (C-8a), 139.3 (C-7*), 138.2 (C-6*), 136.4 (C-3*), 130.2 (C-4a), 126.2 (C-4*), 70.9 (C-4′), 58.9 (C-5′), 39.6 (C-3′); (+)-ESIMS m/z 283 [M + Na]+; (+)-HRESIMS m/z 283.0696 [M + Na]+ (calcd. for C13H12N2NaO4, 283.0689).
3.2.2.10. N-Glycine(methylester)-5,8-dioxo-5,8-dihydroquinoline-2-carboxamide (6j)
From N-glycine(methylester)-8-hydroxyquinoline-2-carboxamide (5j) (50 mg, 0.19 mmol), PIFA (132 mg, 0.31 mmol) to give 6j (40 mg, 77% yield) as a brown oil.
1H NMR (CDCl3, 400 MHz) δH 8.77 (2H, br t, J = 5.2 Hz, NH-2′), 8.59 (1H, d, J = 8.3 Hz, H-3), 8.55 (1H, d, J = 8.3 Hz, H-4), 7.20 (1H, d, J = 10.5 Hz, H-7), 7.13 (1H, d, J = 10.5 Hz, H-6), 4.31 (2H, d, J = 6.1 Hz, H2-3′), 3.79 (3H, s, H3-5′); 13C NMR (CDCl3, 100 MHz) δC 183.7 (C-5), 182.4 (C-8), 169.7 (C-4′), 163.1 (C-1′), 153.2 (C-2), 145.9 (C-8a), 139.4 (C-7), 138.3 (C-6), 136.5 (C-4), 130.2 (C-4a), 126.4 (C-3), 52.5 (C-5′), 41.4 (C-3′); (+)-ESIMS m/z 297 [M + Na]+; (+)-HRESIMS m/z 297.0477 [M + Na]+ (calcd. for C13H10N2NaO5, 297.0482).
3.2.3. General Procedure for the Preparation of Carboxamide Analogues 7a–7j
A solution of 5,8-dioxo-5,8-dihydroquinoline-2-carboxamide (6a–6j) was dissolved in MeCN/EtOH (1:1) before being cooled to 0 °C. In some cases, CeCl3·7H2O (1 equiv.) was also added to the reaction. Hypotaurine (0.8 equiv.) in H2O was added dropwise over 3.5 h. The reaction mixture changed color from dark brown to dark orange, and was stirred at rt for 2 days. The product was purified either by filtration and washing with H2O (3 × 20 mL) and MeOH (3 × 20 mL), or by reversed-phase C18 flash column chromatography (0%–80% MeOH in H2O (0.05% TFA)).
3.2.3.1. N-n-Butyl-5,10-dioxo-3,4,5,10-tetrahydro-2H-[1,4]thiazino[2,3-g]quinoline-7-carboxamide 1,1-Dioxide (7a)
From 6a (54 mg, 0.21 mmol) in MeCN/EtOH (1:1, 20 mL) and hypotaurine (16.0 mg, 0.15 mmol) in H2O (3 mL). Filtration gave 7a as an orange powder (11.0 mg, 14% yield).
Mp 200 °C (decomp.); Rf = 0.49 (10% MeOH/CH2Cl2); IR νmax (ATR) 3300, 3237, 1682, 1594, 1580, 1508, 1336, 1280, 1170, 1107 cm−1; 1H NMR (DMSO-d6, 400 MHz) δH 9.35 (1H, br s, NH-4), 8.70 (1H, t, J = 6.4 Hz, NH-2′), 8.53 (1H, d, J = 8.2 Hz, H-9), 8.40 (1H, d, J = 8.2 Hz, H-8), 3.92–3.87 (2H, m, H2-3), 3.43–3.36 (obscured by solvent, H2-2 and H2-3′), 1.55 (2H, p, J = 7.2 Hz, H2-4′), 1.33 (2H, sex., J = 7.6 Hz, H2-5′), 0.91 (3H, t, J = 7.6 Hz, H3-6′); 13C NMR (DMSO-d6, 100 MHz) δC 176.2 (C-5), 173.4 (C-10), 162.5 (C-1′), 152.6 (C-7), 147.7 (C-4a), 145.3 (C-5a), 136.0 (C-9), 131.4 (C-9a), 126.5 (C-8), 110.7 (C-10a), 48.2 (C-2), 39.3 (obscured by solvent, C-3 and C-3′), 31.3 (C-4′), 19.6 (C-5′), 13.7 (C-6′); (+)-ESIMS m/z 386 [M + Na]+; (+)-HRESIMS m/z 386.0791 [M + Na]+ (calcd. for C16H17N3NaO5S, 386.0781).
3.2.3.2. N-n-Pentyl-5,10-dioxo-3,4,5,10-tetrahydro-2H-[1,4]thiazino[2,3-g]quinoline-7-carboxamide 1,1-Dioxide (7b)
From 6b (31 mg, 0.11 mmol), CeCl3.7H2O (37 mg, 98 µmol) in MeCN/EtOH (1:1, 14 mL) and hypotaurine (8.2 mg, 0.075 mmol) in H2O (2 mL). Filtration gave 7b as a red-brown powder (11.0 mg, 27% yield).
Mp 200 °C (decomp.); Rf = 0.44 (10% MeOH/CH2Cl2); IR νmax (ATR) 3234, 2933, 1686, 1508 cm−1; 1H NMR (DMSO-d6, 400 MHz) δH 9.36 (1H, br s, NH-4), 8.72 (1H, t, J = 5.9 Hz, NH-2′), 8.53 (1H, d, J = 8.2 Hz, H-9), 8.40 (1H, d, J = 8.2 Hz, H-8), 3.90 (2H, br s, H2-3), 3.43–3.33 (4H, obscured by H2O, H2-2, H2-3′), 1.57 (2H, p, J = 6.8 Hz, H2-4′), 1.34–1.27 (4H, m, H2-5′/H2-6′), 0.88 (3H, t, J = 6.8 Hz, H3-7′); 13C NMR (DMSO-d6, 100 MHz) δC 176.2 (C-5), 173.5 (C-10), 162.5 (C-1′), 152.6 (C-7), 147.7 (C-4a), 145.4 (C-5a), 136.0 (C-9), 131.4 (C-9a), 126.5 (C-8), 110.7 (C-10a), 48.2 (C-2), 40.8 (C-3), 38.8 (C-3′), 28.8 (C-4′), 28.7 (C-5′*), 21.9 (C-6′*), 13.9 (C-7′); (+)-ESIMS m/z 378 [M + H]+; (+)-HRESIMS m/z [M + H]+ 378.1107 (calcd. for C17H20N3O5S, 378.1118).
3.2.3.3. N-n-Octyl-5,10-dioxo-3,4,5,10-tetrahydro-2H-[1,4]thiazino[2,3-g]quinoline-7-carboxamide 1,1-Dioxide (7c)
From 6c (32 mg, 0.10 mmol), CeCl3·7H2O (39 mg, 0.10 mmol) in MeCN/EtOH (1:1, 14 mL) and hypotaurine (9.7 mg, 0.089 mmol) in H2O (2 mL). Filtration and solvent wash gave 7c as a red-brown powder (24.0 mg, 57% yield).
Mp 200 °C (decomp.); Rf = 0.44 (10% MeOH/CH2Cl2); IR νmax (ATR) 3240, 2925, 1669, 1521 cm−1; 1H NMR (DMSO-d6, 400 MHz) δH 9.36 (1H, br s, NH-4), 8.71 (1H, t, J = 6.1 Hz, NH-2′), 8.53 (1H, d, J = 8.0 Hz, H-9), 8.39 (1H, d, J = 8.0 Hz, H-8), 3.90 (2H, br s, H2-3), 3.41 (2H, br t, J = 6.1 Hz, H2-2), 3.32 (2H, obscured by H2O, H2-3′), 1.61–1.53 (2H, m, H2-4′), 1.37–1.20 (10H, m, H2-5′/H2-6′/H2-7′/H2-8′/H2-9′), 0.85 (3H, t, J = 6.7 Hz, H3-10′); 13C NMR (DMSO-d6, 100 MHz) δC 176.1 (C-5), 173.4 (C-10), 162.4 (C-1′), 152.5 (C-7), 147.6 (C-4a), 145.2 (C-5a), 135.9 (C-9), 131.3 (C-9a), 126.4 (C-8), 110.6 (C-10a), 48.1 (C-2), 39.5 (C-3/C-3′), 31.1 (C-8′), 29.0 (C-4′), 28.6 (C-6′), 26.4 (C-5′), 22.0 (C-9′), 13.9 (C-10′); (+)-ESIMS m/z 420 [M + H]+; (+)-HRESIMS m/z [M + H]+ 420.1581 (calcd. for C20H26N3O5S, 420.1588).
3.2.3.4. N-Benzyl-5,10-dioxo-3,4,5,10-tetrahydro-2H-[1,4]thiazino[2,3-g]quinoline-7-carboxamide 1,1-Dioxide (7d)
From 6d (44 mg, 0.15 mmol), CeCl3.7H2O (55 mg, 0.15 mmol) in MeCN/EtOH (1:1, 14 mL) and hypotaurine (13.0 mg, 0.12 mmol) in H2O (2 mL). Filtration and solvent wash gave 7d as a red-brown powder (10.0 mg, 17% yield).
Mp 200 °C (decomp.); Rf = 0.44 (10% MeOH/CH2Cl2); IR νmax (ATR) 3267, 1676, 1595, 1513 cm−1; 1H NMR (DMSO-d6, 300 MHz) δH 9.37 (1H, br s, NH-4), 9.28 (1H, t, J = 6.2 Hz, NH-2′), 8.55 (1H, d, J = 8.2 Hz, H-9), 8.43 (1H, d, J = 8.2 Hz, H-8), 7.37–7.23 (5H, m, 2H-5′/2H-6′/H-7′), 4.57 (2H, d, J = 6.4 Hz, H2-3′), 3.90 (2H, br s, H2-3), 3.41 (2H, t, J = 6.2 Hz, H2-2); 13C NMR (DMSO-d6, 75 MHz) δC 176.2 (C-5), 173.4 (C-10), 162.8 (C-1′), 152.5 (C-7), 147.7 (C-4a), 145.5 (C-5a), 139.3 (C-4′), 136.0 (C-9), 131.5 (C-9a), 128.4 (C-5′), 127.5 (C-6′), 126.9 (C-8), 126.7 (C-7′), 110.7 (C-10a), 48.2 (C-2), 42.7 (C-3′), 39.4 (C-3); (+)-ESIMS m/z 420 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 420.0618 (calcd. for C19H15N3NaO5S, 420.0625).
3.2.3.5. N-Phenethyl-5,10-dioxo-3,4,5,10-tetrahydro-2H-[1,4]thiazino[2,3-g]quinoline-7-carboxamide 1,1-Dioxide (7e)
From 6e (22.8 mg, 0.075 mmol), CeCl3.7H2O (51 mg, 0.14 mmol) in MeCN/EtOH (1:1, 14 mL) and hypotaurine (12.0 mg, 0.11 mmol) in H2O (2 mL). Filtration and solvent wash gave 7e as a red-brown powder (15.0 mg, 49% yield).
Mp 240 °C (decomp.); Rf = 0.48 (10% MeOH/CH2Cl2); IR νmax (ATR) 3230, 1678, 1580, 1555 cm−1; 1H NMR (DMSO-d6, 300 MHz) δH 9.37 (1H, br s, NH-4), 8.77 (1H, t, J = 5.9 Hz, NH-2′), 8.54 (1H, d, J = 8.1 Hz, H-9), 8.40 (1H, d, J = 8.1 Hz, H-8), 7.33–7.19 (5H, m, 2H-6′/2H-7′/H-8′), 3.93–3.87 (2H, m, H2-3), 3.60 (2H, dt, J = 7.7, 5.9 Hz, H2-3′), 3.40 (2H, br t, J = 5.4 Hz, H2-2), 2.90 (2H, t, J = 7.7 Hz, H2-4′); 13C NMR (DMSO-d6, 75 MHz) δC 176.2 (C-5), 173.4 (C-10), 162.5 (C-1′), 152.4 (C-7), 147.7 (C-4a), 145.3 (C-5a), 139.2 (C-5′), 136.0 (C-9), 131.4 (C-9a), 128.6 (C-6′), 128.4 (C-7′), 126.5 (C-8′), 126.2 (C-8), 110.7 (C-10a), 48.2 (C-2), 40.7 (C-3′), 38.6 (C-3), 35.1 (C-4′); (+)-ESIMS m/z 434 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 434.0768 (calcd. for C20H17N3NaO5S, 434.0781).
3.2.3.6. N-(3-Phenylpropyl)-5,10-dioxo-3,4,5,10-tetrahydro-2H-[1,4]thiazino[2,3-g] quinoline-7-carboxamide 1,1-Dioxide (7f)
From 6f (39.0 mg, 0.12 mmol), CeCl3.7H2O (51.0 mg, 0.14 mmol) in MeCN and EtOH (1:1, 14 mL) and hypotaurine (12.0 mg, 0.11 mmol) in H2O (2 mL). Filtration and solvent wash gave 7f as a red-brown powder (29.0 mg, 57% yield).
Mp 204 °C (decomp.); Rf = 0.52 (10% MeOH/CH2Cl2); IR νmax (ATR) 3247, 2922, 1670, 1528 cm−1; 1H NMR (DMSO-d6, 300 MHz) δH 9.36 (1H, br s, NH-4), 8.77 (1H, t, J = 6.0 Hz, NH-2′), 8.54 (1H, d, J = 8.1 Hz, H-9), 8.40 (1H, d, J = 8.1 Hz, H-8), 7.31–7.15 (5H, m, H-7′/H-8′/H-9′), 3.90 (2H, t, J = 5.5 Hz, H2-3), 3.44–3.35 (4H, m, H2-2/H2-3′), 2.64 (2H, t, J = 7.8 Hz, H2-5′), 1.89 (2H, p, J = 7.8 Hz, H2-4′); 13C NMR (DMSO-d6, 75 MHz) δC 176.2 (C-5), 173.5 (C-10), 162.6 (C-1′), 152.6 (C-7), 147.7 (C-4a), 145.4 (C-5a), 141.6 (C-6′), 136.0 (C-9), 131.4 (C-9a), 128.3 (C-7′/C-8′), 126.6 (C-9′), 125.8 (C-8), 110.7 (C-10a), 48.2 (C-2), 39.1 (C-3), 38.8 (C-3′), 32.6 (C-5′), 30.8 (C-4′); (+)-ESIMS m/z 426 [M + H]+; (+)-HRESIMS m/z [M + H]+ 426.1119 (calcd. for C21H20N3O5S, 426.1118).
3.2.3.7. (E)-N-(3,7-Dimethylocta-2,6-dien-1-yl)-5,10-dioxo-3,4,5,10-tetrahydro-2H-[1,4]thiazino [2,3-g]quinoline-7-carboxamide 1,1-Dioxide (7g)
From 6g (26.6 mg, 0.079 mmol), CeCl3.7H2O (31 mg, 0.083 mmol) in MeCN/EtOH (1:1, 14 mL) and hypotaurine (7.2 mg, 0.066 mmol) in H2O (2 mL). Filtration and solvent wash gave 7g as a dark orange powder (10.0 mg, 29% yield).
Mp 200 °C (decomp.); Rf = 0.45 (10% MeOH/CH2Cl2); IR νmax (ATR) 3230, 3076, 1693, 1561 cm−1; 1H NMR (DMSO-d6, 300 MHz) δH 9.35 (1H, br s, NH-4), 8.73 (1H, t, J = 5.7 Hz, NH-2′), 8.53 (1H, d, J = 8.1 Hz, H-9), 8.41 (1H, d, J = 8.1 Hz, H-8), 5.27 (1H, t, J = 6.2 Hz, H-4′), 5.07 (1H, t, J = 6.2 Hz, H-8′), 3.97 (2H, dd, J = 6.2, 5.7 Hz, H2-3′), 3.90 (2H, br t, J = 5.5 Hz, H2-3), 3.41 (2H, t, J = 5.5 Hz, H2-2), 2.09–2.02 (2H, m, H2-7′), 2.01–1.95 (2H, m, H2-6′), 1.71 (3H, s, H3-11′), 1.62 (3H, s, H3-12′), 1.56 (3H, s, H3-10′); 13C NMR (DMSO-d6, 75 MHz) δC 176.2 (C-5), 173.4 (C-10), 162.3 (C-1′), 152.6 (C-7), 147.7 (C-4a), 145.3 (C-5a), 137.5 (C-5′), 136.0 (C-9), 131.4 (C-9a), 130.9 (C-9′), 126.5 (C-8), 123.9 (C-8′), 121.0 (C-4′), 110.7 (C-10a), 48.2 (C-2), 40.3 (C-3), 38.6 (C-6′), 37.1 (C-3′), 25.9 (C-7′), 25.5 (C-12′), 17.5 (C-10′), 16.1 (C-11′); (+)-ESIMS m/z 466 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 466.1395 (calcd. for C22H25N3NaO5S, 466.1407).
3.2.3.8. N-(Prop-2-yn-1-yl)-5,10-dioxo-3,4,5,10-tetrahydro-2H-[1,4]thiazino[2,3-g]quinoline-7-carboxamide 1,1-Dioxide (7h)
From 6h (35 mg, 0.15 mmol), CeCl3.7H2O (46.5 mg, 0.12 mmol) in MeCN/EtOH (1:1, 14 mL) and hypotaurine (9.5 mg, 0.087 mmol) in H2O (2 mL). The crude reaction mixture was purified by reversed-phase C18 flash column chromatography to give 7h as a bright yellow powder (15.0 mg, 29% yield).
Mp 280 °C (decomp.); Rf = 0.52 (10% MeOH/CH2Cl2); IR νmax (ATR) 3369, 3255, 2936, 1667, 1595 cm−1; 1H NMR (DMSO-d6, 300 MHz) δH 9.38 (1H, t, J = 5.4 Hz, NH-4), 9.09 (1H, t, J = 6.0 Hz, NH-2′), 8.55 (1H, d, J = 8.1 Hz, H-9), 8.41 (1H, d, J = 8.1 Hz, H-8), 4.13 (2H, dd, J = 6.0, 2.4 Hz, H2-3′), 3.93–3.87 (2H, m, H2-3), 3.40 (2H, obscured by water, H2-2), 3.12 (1H, t, J = 2.4 Hz, H-5′); 13C NMR (DMSO-d6, 75 MHz) δC 176.1 (C-5), 173.3 (C-10), 162.6 (C-1′), 152.0 (C-7), 147.7 (C-4a), 145.5 (C-5a), 136.0 (C-9), 131.5 (C-9a), 126.7 (C-8), 110.7 (C-10a), 80.9 (C-4′), 72.8 (C-5′), 48.2 (C-2), 39.4 (C-3), 28.7 (C-3′); (+)-ESIMS m/z 368 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 368.0294 (calcd. for C15H11N3NaO5S, 368.0312).
3.2.3.9. N-(2-Methoxyethyl)-5,10-dioxo-3,4,5,10-tetrahydro-2H-[1,4]thiazino[2,3-g] quinoline-7-carboxamide 1,1-Dioxide (7i)
From 6i (31 mg, 0.12 mmol) in MeCN/EtOH (1:1, 20 mL) and hypotaurine (7.8 mg, 0.072 mmol) in H2O (3 mL). The crude reaction mixture was purified by reversed-phase C18 flash column chromatography to give 7i as an orange powder (11.2 mg, 26% yield).
Mp 200 °C (decomp.); Rf = 0.54 (10% MeOH/CH2Cl2); IR νmax (ATR) 3546, 3251, 1673, 1594, 1581, 1556, 1339, 1122 cm−1; 1H NMR (DMSO-d6, 400 MHz) δH 9.37 (1H, br s, NH-4), 8.64 (1H, t, J = 5.4 Hz, NH-2′), 8.54 (1H, d, J = 8.0 Hz, H-9), 8.41 (1H, d, J = 8.0 Hz, H-8), 3.92–3.88 (2H, m, H2-3), 3.56–3.51 (4H, m, H2-3′ and H2-4′), 3.43–3.39 (2H, m, H2-2), 3.29 (3H, s, H3-5′); 13C NMR (DMSO-d6, 100 MHz) δC 176.2 (C-5), 173.4 (C-10), 162.5 (C-1′), 152.2 (C-7), 147.7 (C-4a), 145.4 (C-5a), 136.1 (C-9), 131.4 (C-9a), 126.5 (C-8), 110.7 (C-10a), 70.3 (C-4′), 57.9 (C-5′), 48.2 (C-2), 39.2 (C-3), 38.7 (C-3′); (+)-ESIMS m/z 388 [M + Na]+; (+)-HRESIMS m/z 388.0566 [M + Na]+ (calcd. for C15H15N3NaO6S, 388.0574).
3.2.3.10. Methyl 2-(1,1-Dioxido-5,10-dioxo-3,4,5,10-tetrahydro-2H-[1,4]thiazino[2,3-g] quinoline-7-carboxamido)acetate (7j)
From 6j (50 mg, 0.18 mmol) in MeCN/EtOH (1:1, 20 mL) and hypotaurine (11.9 mg, 0.11 mmol) in H2O (3 mL). Reversed-phase C18 flash column chromatography gave 7j as a bright red powder (13.8 mg, 20% yield).
Mp 200 °C (decomp.); Rf = 0.46 (10% MeOH/CH2Cl2); IR νmax (ATR) 3576, 3335, 1748, 1666, 1594, 1581, 1557, 1346, 1271, 1212, 1164, 1115 cm−1; 1H NMR (DMSO-d6, 400 MHz) δH 9.40 (1H, br t, J = 3.4 Hz, NH-4), 9.08 (1H, t, J = 6.1 Hz, NH-2′), 8.56 (1H, d, J = 8.0 Hz, H-9), 8.42 (1H, d, J = 8.0 Hz, H-8), 4.15 (2H, d, J = 6.1 Hz, H-3′), 3.92–3.88 (2H, m, H2-3), 3.67 (3H, s, H3-5′), 3.43–3.40 (2H, m, H2-2); 13C (DMSO-d6, 100 MHz) δC 176.2 (C-5), 173.4 (C-10), 170.0 (C-4′), 163.0 (C-1′), 151.7 (C-7), 147.7 (C-4a), 145.6 (C-5a), 136.1 (C-9), 131.6 (C-9a), 126.6 (C-8), 110.8 (C-10a), 51.9 (C-5′), 48.2 (C-2), 41.2 (C-3′), 39.2 (C-3); (+)-ESIMS m/z 380 [M + H]+; (+)-HRESIMS m/z 380.0538 [M + H]+ (calcd. for C15H14N3O7S, 380.0547).
3.2.4. General Procedure for Preparation of Δ2(3) Analogues 8a–8i, 8k
Thiazine-quinoline-carboxamide (7a–7j) in DMF (1–3 mL) was stirred in 2 N NaOH (3 mL) at rt for 2 h. HCl (10% vol) was added dropwise until the reaction mixture was pH 5 and the mixture was then purified by reversed-phase C18 flash column chromatography (0%–10% MeOH (0.05% TFA)) to give the desired product.
3.2.4.1. N-n-Butyl-5,10-dioxo-5,10-dihydro-4H-[1,4]thiazino[2,3-g]quinoline-7-carboxamide 1,1-Dioxide (8a)
From 7a (34.0 mg, 0.094 mmol) using the general procedure to give 8a as a yellow solid (13.0 mg, 38% yield).
Mp 200 °C (decomp.); Rf = 0.53 (10% MeOH/CH2Cl2); IR νmax (ATR) 3402, 3058, 1714, 1653, 1632, 1527, 1503, 1318, 1125 cm−1; 1H NMR (DMSO-d6, 400 MHz) δH 11.42 (1H, br s, NH-4), 8.76 (1H, t, J = 6.0 Hz, NH-2′), 8.58 (1H, d, J = 8.0 Hz, H-9), 8.43 (1H, d, J = 8.0 Hz, H-8), 7.17 (1H, d, J = 9.0 Hz, H-3), 6.62 (1H, d, J = 9.0 Hz, H-2), 3.38 (2H, dt, J = 6.9, 6.9 Hz, H2-3′), 1.56 (2H, p, J = 7.0 Hz, H2-4′), 1.33 (2H, sex., J = 7.5 Hz, H2-5′), 0.91 (3H, t, J = 7.5 Hz, H3-6′); 13C NMR (DMSO-d6, 100 MHz) δC 177.7 (C-10), 175.5 (C-5), 162.4 (C-1′), 153.3 (C-7), 145.5 (C-5a), 141.4 (C-4a), 136.1 (C-9), 130.6 (C-9a), 130.5 (C-3), 126.4 (C-8), 115.2 (C-10a), 112.0 (C-2), 38.6 (C-3′), 31.3 (C-4′), 19.6 (C-5′), 13.7 (C-6′); (+)-ESIMS m/z 384 [M + Na]+; (+)-HRESIMS m/z 384.0632 [M + Na]+ (calcd. for C16H15N3NaO5S, 384.0625).
3.2.4.2. N-n-Pentyl-5,10-dioxo-5,10-dihydro-4H-[1,4]thiazino[2,3-g]quinoline-7-carboxamide 1,1-Dioxide (8b)
From 7b (10.0 mg, 0.027 mmol) using the general procedure to give 8b as a yellow solid (4.0 mg, 40% yield).
Mp 280 °C (decomp.); Rf = 0.44 (10% MeOH/CH2Cl2); IR νmax (ATR) 3319, 3057, 1710, 1635, 1528 cm−1; 1H NMR (DMSO-d6, 300 MHz) δH 11.44 (1H, br s, NH-4), 8.78 (1H, t, J = 6.1 Hz, NH-2′), 8.58 (1H, d, J = 8.1 Hz, H-9), 8.43 (1H, d, J = 8.1 Hz, H-8), 7.17 (1H, d, J = 8.9 Hz, H-3), 6.62 (1H, d, J = 8.9 Hz, H-2), 3.40–3.34 (2H, m, H2-3′), 1.58 (2H, p, J = 7.5 Hz, H2-4′), 1.34–1.27 (4H, m, H2-5′/H2-6′), 0.88 (3H, t, J = 7.5 Hz, H3-7′); 13C NMR (DMSO-d6, 75 MHz) δC 177.7 (C-10), 175.5 (C-5), 162.4 (C-1′), 153.3 (C-7), 145.6 (C-5a), 141.4 (C-4a), 136.1 (C-9), 130.6 (C-9a), 130.5 (C-3), 126.4 (C-8), 115.2 (C-10a), 112.0 (C-2), 38.8 (C-3′), 28.9 (C-4′), 28.7 (C-5′), 21.9 (C-6′), 14.0 (C-7′); (+)-ESIMS m/z 398 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 398.0776 (calcd. for C17H17N3NaO5S, 3798.0781).
3.2.4.3. N-n-Octyl-5,10-dioxo-5,10-dihydro-4H-[1,4]thiazino[2,3-g]quinoline-7-carboxamide 1,1-Dioxide (8c)
From 7c (12.0 mg, 0.029 mmol) using the general procedure to give 8c as a yellow solid (6.0 mg, 50% yield).
Mp 280 °C (decomp.); Rf = 0.41 (10% MeOH/CH2Cl2); IR νmax (ATR) 3289, 2924, 1635, 1527 cm−1; 1H NMR (DMSO-d6, 300 MHz) δH 11.44 (1H, d, J = 5.3 Hz, NH-4), 8.79 (1H, t, J = 5.8 Hz, NH-2′), 8.58 (1H, d, J = 8.0 Hz, H-9), 8.43 (1H, d, J = 8.0 Hz, H-8), 7.17 (1H, dd, J = 8.8, 5.3 Hz, H-3), 6.63 (1H, d, J = 8.8 Hz, H-2), 3.37 (obscured by solvent, H2-3′), 1.60–1.52 (2H, m, H2-4′), 1.31–1.23 (10H, m, H2-5′/H2-6′/H2-7′/H2-8′/H2-9′), 0.85 (3H, t, J = 6.8 Hz, H3-10′); 13C NMR (DMSO-d6, 75 MHz) δC 177.7 (C-10), 175.5 (C-5), 162.4 (C-1′), 153.3 (C-7), 145.6 (C-5a), 141.4 (C-4a), 136.1 (C-9), 130.6 (C-9a), 130.5 (C-3), 126.4 (C-8), 115.2 (C-10a), 112.0 (C-2), 38.9 (C-3′), 31.3 (C-8′), 29.2 (C-4′), 28.8 (C-6′), 28.7 (C-7′), 26.5 (C-5′), 22.1 (C-9′), 14.0 (C-10′); (+)-ESIMS m/z 440 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 440.1232 (calcd. for C20H23N3NaO5S, 440.1251).
3.2.4.4. N-Benzyl-5,10-dioxo-5,10-dihydro-4H-[1,4]thiazino[2,3-g]quinoline-7-carboxamide 1,1-Dioxide (8d)
From 7d (10.0 mg, 0.025 mmol) using the general procedure to give 8d as a yellow solid (3.0 mg, 30% yield).
Mp 280 °C (decomp.); Rf = 0.47 (10% MeOH/CH2Cl2); IR νmax (ATR) 3213, 1706, 1634, 1513 cm−1; 1H NMR (DMSO-d6, 400 MHz) δH 11.41 (1H, d, J = 5.6 Hz, NH-4), 9.33 (1H, t, J = 6.3 Hz, NH-2′), 8.59 (1H, d, J = 8.2 Hz, H-9), 8.46 (1H, d, J = 8.2 Hz, H-8), 7.38–7.30 (4H, m, 2H-5′/2H-6′), 7.27–7.23 (1H, m, H-7′), 7.17 (1H, dd, J = 8.9, 5.6 Hz, H-3), 6.61 (1H, d, J = 8.9 Hz, H-2), 4.58 (2H, d, J = 6.3 Hz, H2-3′); 13C NMR (DMSO-d6, 100 MHz) δC 177.6 (C-10), 175.4 (C-5), 162.7 (C-1′), 153.1 (C-7), 145.6 (C-5a), 141.3 (C-4a), 139.2 (C-4′), 136.1 (C-9), 130.7 (C-9a), 130.5 (C-3), 128.3 (C-5′), 127.5 (C-6′), 126.9 (C-8), 126.6 (C-7′), 115.2 (C-10a), 112.0 (C-2), 42.7 (C-3′); (+)-ESIMS m/z 418 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 418.0470 (calcd. for C19H13N3NaO5S, 418.0468).
3.2.4.5. N-Phenethyl-5,10-dioxo-5,10-dihydro-4H-[1,4]thiazino[2,3-g]quinoline-7-carboxamide 1,1-Dioxide (8e)
From 7e (11.0 mg, 0.027 mmol) using the general procedure to give 8e as a yellow solid (4.0 mg, 36% yield).
Mp 290 °C (decomp.); Rf = 0.47 (10% MeOH/CH2Cl2); IR νmax (ATR) 3103, 3067, 1714, 1678, 1512 cm−1; 1H NMR (DMSO-d6, 300 MHz) δH 11.45 (1H, d, J = 5.6, NH-4), 8.85 (1H, t, J = 6.1 Hz, NH-2′), 8.58 (1H, d, J = 8.1 Hz, H-9), 8.44 (1H, d, J = 8.1 Hz, H-8), 7.34–7.22 (5H, m, 2H-6′/2H-7′/H-8′), 7.17 (1H, dd, J = 8.7, 5.6 Hz, H-3), 6.62 (1H, d, J = 8.7 Hz, H-2), 3.61 (2H, dt, J = 6.9, 6.1 Hz, H2-3′), 2.90 (2H, t, J = 6.9 Hz, H2-4′); 13C NMR (DMSO-d6, 100 MHz) δC 177.7 (C-10), 175.5 (C-5), 162.4 (C-1′), 153.0 (C-7), 145.6 (C-5a), 141.4 (C-4a), 139.3 (C-5′), 136.2 (C-9), 130.7 (C-9a), 130.5 (C-3), 128.7 (C-6′), 128.5 (C-7′), 126.4 (C-7′), 126.2 (C-8), 115.2 (C-10a), 112.0 (C-2), 40.8 (C-3′), 35.1 (C-4′); (+)-ESIMS m/z 432 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 432.0618 (calcd. for C20H15N3NaO5S, 432.0625).
3.2.4.6. N-(3-Phenylpropyl)-5,10-dioxo-5,10-dihydro-4H-[1,4]thiazino[2,3-g]quinoline-7-carboxamide 1,1-Dioxide (8f)
From 7f (20.0 mg, 0.047 mmol) using the general procedure to give 8f as a yellow solid (6.0 mg, 30% yield).
Mp 230 °C (decomp.); Rf = 0.41 (10% MeOH/CH2Cl2); IR νmax (ATR) 3059, 2930, 1653, 1511 cm−1; 1H NMR (DMSO-d6, 300 MHz) δH 11.45 (1H, br s, NH-4), 8.86 (1H, t, J = 6.1 Hz, NH-2′), 8.58 (1H, d, J = 8.2 Hz, H-9), 8.44 (1H, d, J = 8.2 Hz, H-8), 7.31–7.15 (6H, m, H-3/2H-7′/2H-8′/H-9′), 6.63 (1H, d, J = 9.1 Hz, H-2), 3.40 (2H, dt, J = 7.5, 6.1 Hz, H2-3′), 2.64 (2H, t, J = 7.5 Hz, H2-5′), 1.89 (2H, p, J = 7.5 Hz, H2-4′); 13C NMR (DMSO-d6, 100 MHz) δC 177.7 (C-10), 175.5 (C-5), 162.5 (C-1′), 153.3 (C-7), 145.6 (C-5a), 141.6 (C-6′), 141.4 (C-4a), 136.1 (C-9), 130.6 (C-9a), 130.5 (C-3), 128.3 (C-7′/C-8′), 126.5 (C-9′), 125.8 (C-8), 115.2 (C-10a), 112.0 (C-2), 38.9 (C-3′), 32.7 (C-5′), 30.8 (C-4′); (+)-ESIMS m/z 446 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 446.0790 (calcd. for C21H17N3NaO5S, 446.0781).
3.2.4.7. (E)-N-(3,7-Dimethylocta-2,6-dien-1-yl)-5,10-dioxo-5,10-dihydro-4H-[1,4]thiazino[2,3-g] quinoline-7-carboxamide 1,1-Dioxide (8g)
From 7g (10.0 mg, 0.023 mmol) using the general procedure to give 8g a yellow solid (5.0 mg, 50% yield).
Mp 280 °C (decomp.); Rf = 0.44 (10% MeOH/CH2Cl2); 1H NMR (DMSO-d6, 400 MHz) δH 11.44 (1H, d, J = 5.7 Hz, NH-4), 8.81 (1H, t, J = 6.2 Hz, NH-2′), 8.58 (1H, d, J = 7.8 Hz, H-9), 8.44 (1H, d, J = 7.9 Hz, H-8), 7.17 (1H, dd, J = 8.6, 5.7 Hz, H-3), 6.62 (1H, d, J = 8.6 Hz, H-2), 5.27 (1H, t, J = 6.6 Hz, H-4′), 5.07 (1H, t, J = 7.0 Hz, H-8′), 3.98 (2H, dd, J = 6.2, 5.8 Hz, H2-3′), 2.07–2.03 (2H, m, H2-6′), 2.01–1.96 (2H, m, H2-7′), 1.71 (3H, s, H3-11′), 1.62 (3H, s, H3-12′), 1.56 (3H, s, H3-10′); 13C NMR (DMSO-d6, 100 MHz) δC 177.6 (C-10), 175.5 (C-5), 162.2 (C-1′), 153.2 (C-7), 145.6 (C-5a), 141.4 (C-4a), 137.6 (C-5′), 136.1 (C-9), 131.0 (C-9a), 130.6 (C-9′), 130.5 (C-3), 126.4 (C-8), 123.9 (C-8′), 121.1 (C-4′), 115.2 (C-10a), 112.0 (C-2), 38.9 (C-6′), 37.1 (C-3′), 26.0 (C-7′), 25.5 (C-12′), 17.6 (C-10′), 16.2 (C-11′); (+)-ESIMS m/z 464 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 464.1254 (calcd. for C22H23N3NaO5S, 464.1251).
3.2.4.8. N-(Prop-2-yn-1-yl)-5,10-dioxo-5,10-dihydro-4H-[1,4]thiazino[2,3-g]quinoline-7-carboxamide 1,1-Dioxide (8h)
From 7h (8.0 mg, 0.023 mmol) using the general procedure to give 8h as a yellow solid (6.0 mg, 76% yield).
Mp 230 °C (decomp.); Rf = 0.46 (10% MeOH/CH2Cl2); IR νmax (ATR) 3310, 3058, 1636, 1509 cm−1; 1H NMR (DMSO-d6, 300 MHz) δH 11.45 (1H, d, J = 5.5 Hz, NH-4), 9.16 (1H, t, J = 6.2 Hz, NH-2′), 8.60 (1H, d, J = 8.2 Hz, H-9), 8.45 (1H, d, J = 8.2 Hz, H-8), 7.17 (1H, dd, J = 8.8, 5.5 Hz, H-3), 6.62 (1H, d, J = 8.8 Hz, H-2), 4.15 (2H, dd, J = 5.9, 2.5 Hz, H2-3′), 3.13 (1H, t, J = 2.5 Hz, H-5′); 13C NMR (CDCl3, 100 MHz) δC 177.6 (C-10), 175.4 (C-5), 162.5 (C-1′), 152.7 (C-7), 145.8 (C-5a), 141.4 (C-4a), 136.1 (C-9), 130.8 (C-9a), 130.5 (C-3), 126.6 (C-8), 115.3 (C-10a), 112.0 (C-2), 80.9 (C-4′), 72.9 (C-5′), 28.7 (C-3′); (+)-ESIMS m/z 366 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 366.0151 (calcd. for C15H9N3NaO5S, 366.0155).
3.2.4.9. N-(2-Methoxyethyl)-5,10-dioxo-5,10-dihydro-4H-[1,4]thiazino[2,3-g] quinoline-7-carboxamide 1,1-Dioxide (8i)
From 7i (18.8 mg, 0.052 mmol) using the general procedure to give 8i as a yellow solid (15.6 mg, 83% yield).
Mp 200 °C (decomp.); Rf = 0.54 (10% MeOH/CH2Cl2); IR νmax (ATR) 3250, 3057, 1633, 1508, 1278, 1097 cm−1; 1H NMR (DMSO-d6, 400 MHz) δH 11.44 (1H, s, NH-4), 8.70 (1H, t, J = 5.6 Hz, H-2′), 8.59 (1H, d, J = 8.0 Hz, H-9), 8.45 (1H, d, J = 8.0 Hz, H-8), 7.17 (1H, d, J = 9.0 Hz, H-3), 6.62 (1H, d, J = 9.0 Hz, H-2), 3.59–3.50 (4H, m, H2-3′ and H2-4′), 3.29 (3H, s, H3-5′); 13C NMR (DMSO-d6, 100 MHz) δC 177.6 (C-10), 175.4 (C-5), 162.4 (C-1′), 152.8 (C-7), 145.5 (C-5a), 141.3 (C-4a), 136.2 (C-9), 130.7 (C-9a), 130.5 (C-3), 126.3 (C-8), 115.3 (C-10a), 112.0 (C-2), 70.2 (C-4′), 57.9 (C-5′), 38.4 (C-3′); (+)-ESIMS m/z 364 [M + H]+; (+)-HRESIMS m/z 364.0606 [M + H]+ (calcd. for C15H14N3O6S, 364.0598).
3.2.4.10. 2-(1,1-Dioxido-5,10-dioxo-5,10-dihydro-4H-[1,4]thiazino[2,3-g]quinoline-7-carboxamido)acetic Acid (8k)
From 7j (13.8 mg, 0.036 mmol) using the general procedure to give carboxylic acid 8k as a yellow oil (8.2 mg, 62% yield).
Rf = 0.20 (10% MeOH/CH2Cl2); IR νmax (ATR) 3582, 3250, 3057, 1748, 1634, 1508, 1279, 1127 cm−1; 1H NMR (DMSO-d6, 400 MHz) δH 11.95 (1H, br s, NH-4), 9.00 (1H, t, J = 6.0 Hz, NH-2′), 8.60 (1H, d, J = 8.0 Hz, H-9), 8.45 (1H, d, J = 8.0 Hz, H-8), 7.18 (1H, d, J = 8.8 Hz, H-3), 6.62 (1H, d, J = 8.8 Hz, H-2), 4.07 (2H, d, J = 6.0 Hz, H2-3′); 13C NMR (DMSO-d6, 100 MHz) δC 177.6 (C-10), 175.5 (C-5), 170.9 (C-4′), 162.7 (C-1′), 152.4 (C-7), 145.7 (C-5a), 141.6 (C-4a), 136.2 (C-9), 130.8 (C-9a), 130.6 (C-3), 126.4 (C-8), 115.2 (C-10a), 112.0 (C-2), 41.3 (C-3′); (−)-ESIMS m/z 362 [M − H]−; (−)-HRESIMS m/z 362.0083 [M − H]− (calcd. for C14H8N3O7S, 362.0088).
3.2.5. Methyl 2-(1,1-dioxido-5,10-dioxo-5,10-dihydro-4H-[1,4]thiazino[2,3-g] quinoline-7-carboxamido)acetate (8j)
Thionyl chloride (8.4 µL, 0.116 mmol) was added to a solution of 8k (7.0 mg, 0.019 mmol) in dry MeOH (3 mL) at 0 °C. The reaction mixture was stirred at that temperature for 20 min, then heated to 65 °C and stirred for an additional 2 h. The solution was then cooled to rt and loaded directly onto a C18 reversed-phase chromatography column. The crude material was washed with two column volumes of H2O and the product eluted with 100% MeOH (+0.05% TFA) to afford 8j as a yellow oil (6.8 mg, 93% yield).
Rf = 0.49 (10% MeOH/CH2Cl2); IR νmax (ATR) 3247, 3056, 1753, 1635, 1508, 1273, 1128 cm−1; 1H NMR (DMSO-d6, 400 MHz) δH 11.46 (1H, br d, J = 5.2 Hz, NH-4), 9.14 (1H, t, J = 6.2 Hz, NH-2′), 8.61 (1H, d, J = 8.0 Hz, H-9), 8.45 (1H, d, J = 8.0 Hz, H-8), 7.17 (1H, dd, J = 8.8, 5.2 Hz, H-3), 6.63 (1H, d, J = 8.8 Hz, H-2), 4.16 (2H, d, J = 6.2 Hz, H2-3′), 3.68 (3H, s, H3-5′); 13C NMR (DMSO-d6, 100 MHz) δC 177.6 (C-10), 175.4 (C-5), 170.0 (C-4′), 162.9 (C-1′), 152.3 (C-7), 145.7 (C-5a), 141.4 (C-4a), 136.2 (C-9), 130.9 (C-9a), 130.4 (C-3), 126.4 (C-8), 115.3 (C-10a), 112.0 (C-2), 51.9 (C-5′), 41.3 (C-3′); (+)-ESIMS m/z 400 [M + Na]+; (+)-ESIMS m/z 400.0222 [M + Na]+ (calcd. for C15H11N3NaO7S, 400.0210).
3.2.6. Methyl 5,9-Dioxo-3,4,5,9-tetrahydro-2H-thieno[2′,3′:4,5]benzo[1,2-b][1,4]thiazine-7-carboxylate 1,1-Dioxide (10a) and Methyl 5,9-Dioxo-2,3,5,9-tetrahydro-1H-thieno[3′,2′:4,5]benzo [1,2-b][1,4]thiazine-7-carboxylate 4,4-Dioxide (10b)
A solution of methyl 4,7-dioxo-4,7-dihydrobenzo[b]thiophene-2-carboxylate (9) [22] (74 mg, 0.33 mmol) and CeCl3·7H2O (124 mg, 0.33 mmol) in MeCN (10 mL) and EtOH (10 mL) was cooled to 0 °C. Hypotaurine (36 mg, 0.33 mmol) in H2O (2 mL) was added dropwise to the mixture leading to a color change from yellow to orange. The reaction was stirred at rt for 2 days. The residue was filtered and washed with H2O (3 × 20 mL) and MeOH (3 × 20 mL), to give a mixture of regioisomers (10a/10b, 1:0.3 ratio determined by NMR)) as an orange solid (20 mg, 18% yield).
Mp 280 °C (decomp.); Rf = 0.36 (10% MeOH/CH2Cl2); IR νmax (ATR) 3222, 3003, 1726, 1683, 1579 cm−1; 1H NMR (DMSO-d6, 400 MHz) δH 9.36 (1H, br s, NH), 8.03 (1H, s, H-8 minor isomer), 7.93 (1H, s, H-8), 3.91 (3H, s, H3-2′), 3.89 (3H, s, H3-2′ minor isomer), 3.88–3.84 (2H, m, H2-3), 3.37 (2H, obscured by water, H2-2); 13C NMR (DMSO-d6, 75 MHz) 10a δC 173.1 (C-5), 171.5 (C-9), 160.8 (C-1′), 148.0 (C-4a), 142.6 (C-5a*), 141.8 (C-8a*), 141.1 (C-7), 130.2 (C-8), 109.6 (C-9a), 53.3 (C-2′), 48.1 (C-2), 39.2 (C-3); (+)-FABMS m/z 328 [M + H]+; (+)-HRFABMS m/z [M + H]+ 327.9950 (calcd. for C12H10NO6S2, 327.9950).
3.2.7. 5,9-Dioxo-3,4,5,9-tetrahydro-2H-thieno[2′,3′:4,5]benzo[1,2-b][1,4]thiazine-7-carboxylic Acid 1,1-Dioxide (11a) and 5,9-Dioxo-2,3,5,9-tetrahydro-1H-thieno[3′,2′:4,5]benzo[1,2-b][1,4] thiazine-7-carboxylic Acid 4,4-Dioxide (11b)
Methyl ester (as a mixture of regioisomers) 10a/10b (20.0 mg, 0.061 mmol) was dissolved in conc. HCl (3 mL), and stirred at rt for 5 h, after which time, the mixture was heated to 100 °C and stirred for a further 2 h. The crude reaction mixture was subjected to reversed-phase C18 column chromatography (0%–10% MeOH/H2O (0.05% TFA)) to give 11a/11b as a mixture of regioisomers (1:0.3, 11.0 mg, 57% yield) as a bright orange solid.
Mp 200 °C (decomp.); Rf = 0.25 (10% MeOH/CH2Cl2); IR νmax (ATR) 3357, 3230, 1674, 1577; 1H NMR (DMSO-d6, 300 MHz) δH 9.42 (br s, NH minor isomer), 9.31 (1H, br s, NH), 7.93 (s, H-8 minor isomer), (7.84 (1H, s, H-8), 3.86 (2H, br s, H2-3), 3.40–3.34 (2H, br m, H2-2); 13C NMR (DMSO-d6, 75 MHz) 11a δC 173.1 (C-5), 171.7 (C-9), 161.8 (C-1′), 147.9 (C-4a), 144.2 (C-7), 142.8 (C-8a), 141.2 (C-5a), 129.6 (C-8), 109.6 (C-9a), 48.1 (C-2), 39.2 (C-3); (+)-ESIMS m/z 336 [M + Na]+; (+)-HRESIMS m/z [M + Na]+ 335.9605 (calcd. for C11H7NNaO6S2, 335.9607).
3.2.8. 5,9-Dioxo-5,9-dihydro-4H-thieno[2′,3′:4,5]benzo[1,2-b][1,4]thiazine-7-carboxylic Acid 1,1-Dioxide (12)
Thiophene methyl ester (10a/10b) (20.0 mg, 0.061 mmol) was dissolved in hot EtOAc (2 mL), followed by the addition of 1 N NaOH (1 mL). The biphasic mixture was stirred at rt for 1.5 h. HCl (10% vol) was added dropwise until the reaction mixture turned acidic. The crude mixture was subjected to reversed-phase C18 column chromatography (0%–10% MeOH/H2O (0.05% TFA)) to give 12 (single regio-isomer) (15 mg, 78% yield) as a bright orange solid.
Mp 290 °C (decomp.); Rf = 0.27 (10% MeOH/CH2Cl2); IR νmax (ATR) 3227, 3068, 1689, 1637, 1510; 1H NMR (DMSO-d6, 300 MHz) δH 11.41 (1H, br s, NH), 7.82 (1H, s, H-8), 7.13 (1H, d, J = 8.9 Hz, H-3), 6.57 (1H, d, J = 8.9 Hz, H-2); 13C NMR (DMSO-d6, 75 MHz) δC 175.2 (C-9), 172.5 (C-5), 161.7 (C-1′), 147.4 (C-7), 141.5 (C-5a*), 141.4 (C-4a*), 141.2 (C-8a*), 130.3 (C-3), 128.1 (C-8), 114.5 (C-9a), 112.1 (C-2); (+)-FABMS m/z 312 [M + H]+; (+)-HRFABMS m/z [M + H]+ 311.9642 (calcd. for C11H6NO6S2, 311.9637).
3.2.9. Methyl 4,7-Dihydroxybenzo[b]thiophene-2-carboxylate (14)
Commercially available 7-methoxy-benzofuran-2-carboxylic acid ethyl ester (13) (105 mg, 0.477 mmol) in MeCN/4 N H2SO4 (20 mL/5 mL) was stirred at rt, before addition of (NH4)4Ce(SO4)4·2H2O (1.80 g, 3.02 mol) in 4 N H2SO4 (25 mL). The reaction mixture was heated to 60 °C for 90 min. changing the colour from orange to yellow as well as inducing the formation of a white precipitate. The reaction was cooled, filtered, and the filtrate was extracted repeatedly with CH2Cl2 (5 × 50 mL). The combined organic phases were then dried (MgSO4) and the solvent removed in vacuo to give 14 as a yellow solid (89 mg, 85% yield). The product was used immediately in the next step without further purification.
IR νmax (ATR) 3570, 2955, 1752, 1726, 1534, 1475, 1367, 1187, 1160, 1139 cm−1; 1H NMR (DMSO-d6, 75 MHz) δH 7.49 (1H, s, H-3), 6.82 (2H, s, H-5/H-6), 4.45 (2H, q, J = 7.2 Hz, H2-3′), 1.42 (3H, t, J = 7.2 Hz, H3-4′); EIMS m/z 220 [M]+; (+)-HREIMS m/z [M]+ 220.0369 (calcd. for C11H8O5, 220.0372).
3.2.10. Ethyl 5,9-Dioxo-3,4,5,9-tetrahydro-2H-benzofuro[5,6-b][1,4]thiazine-7-carboxylate 1,1-Dioxide (15)
A solution of quinone 14 (100 mg, 0.45 mmol) and CeCl3.7H2O (78 mg, 0.21 mmol) in MeCN (10 mL) and EtOH (10 mL) was cooled to 0 °C. Hypotaurine (49 mg, 0.45 mmol) in H2O (1 mL) was added dropwise to the reaction mixture, changing the colour from yellow to orange. The reaction was stirred at rt for 24 h. The residue was filtered and washed with H2O (3 × 20 mL) and MeOH (3 × 20 mL), to give 15 (62 mg, 43% yield) as a red solid.
Mp 277 °C; Rf = 0.36 (10% MeOH/CH2Cl2); IR νmax (ATR) 3225, 1733, 1695, 1566 cm−1; 1H NMR (DMSO-d6, 300 MHz) δH 9.38 (1H, br s, NH), 7.58 (1H, s, H-8), 4.37 (2H, q, J = 7.1 Hz, H2-3′), 3.85 (2H, dt, J = 5.7, 5.7 Hz, H2-3), 3.34 (2H, t, J = 5.7 Hz, H2-2), 1.33 (3H, t, J = 7.1 Hz, H-4′); 13C NMR (DMSO-d6, 75 MHz) δC 172.2 (C-9), 167.8 (C-5), 157.1 (C-1′), 149.2 (C-5a), 148.7 (C-7), 147.4 (C-4a), 130.1 (C-8a), 114.2 (C-8), 108.9 (C-9a), 62.0 (C-3′), 48.1 (C-2), 39.2 (C-3), 14.0 (C-4′); (+)-FABMS m/z 326 [M + H]+; (+)-HRFABMS m/z [M + H]+ 326.0341 (calcd. for C13H12NO7S, 326.0335).
3.2.11. 5,9-Dioxo-3,4,5,9-tetrahydro-2H-benzofuro[5,6-b][1,4]thiazine-7-carboxylic Acid 1,1-Dioxide (16)
Ethyl ester 15 (64 mg, 0.20 mmol) was dissolved in conc. HCl (3 mL), and the mixture was heated to 100 °C and stirred for 2 h. The crude reaction mixture was purified by reversed-phase C18 column chromatography (0%–10% MeOH/H2O (0.05% TFA)), to give 16 (37 mg, 63% yield) as a bright red solid.
Mp 210 °C (decomp.); Rf = 0.23 (10% MeOH/CH2Cl2); IR νmax (ATR) 3234, 3093, 1635, 1694, 1561 cm−1; 1H NMR (DMSO-d6, 300 MHz) δH 9.35 (1H, br s, NH-4), 7.47 (1H, s, H-8), 3.87–3.82 (2H, m, H2-3), 3.37–3.31 (2H, m, H2-2); 13C NMR (DMSO-d6, 75 MHz) δC 172.5 (C-9), 167.8 (C-5), 158.5 (C-1′), 150.3 (C-5a*), 149.0 (C-7*), 147.4 (C-4a), 130.3 (C-8a), 113.5 (C-8), 108.9 (C-9a), 48.1 (C-2), 39.5 (C-3); (+)-ESIMS m/z 298 [M + H]+; (+)-HRESIMS m/z [M + H]+ 298.0009 (calcd. for C11H8NO7S, 298.0016).
3.2.12. 5,9-Dioxo-5,9-dihydro-4H-benzofuro[5,6-b][1,4]thiazine-7-carboxylic Acid 1,1-Dioxide (17)
Ethyl ester 15 (15.0 mg, 0.046 mmol) was dissolved in hot EtOAc (2 mL), followed by the addition of 1 N NaOH (1 mL). The biphasic mixture was stirred at rt for 2 h. HCl (10% vol) was added dropwise until the reaction mixture turned acidic. The crude product was purified by reversed-phase C18 column chromatography (0%–10% MeOH/H2O (0.05% TFA)), to give 17 (6.4 mg, 47% yield) as a red solid.
Mp 280 °C (decomp.); Rf = 0.36 (10% MeOH/CH2Cl2); IR νmax (ATR) 3223, 3072, 1677, 1577, 1516 cm−1; 1H NMR (DMSO-d6, 400 MHz) δH 11.42 (1H, br s, NH-4), 7.47 (1H, s, H-8), 7.12 (1H, d, J = 8.8 Hz, H-3), 6.58 (1H, d, J = 8.8 Hz, H-2); 13C NMR (DMSO-d6, 75 MHz) δC 176.1 (C-9), 167.4 (C-5), 158.5 (C-1′), 152.0 (C-7), 149.2 (C-5a), 140.4 (C-4a), 130.2 (C-3), 129.1 (C-8a), 113.9 (C-9a), 112.3 (C-2 and C-8); (+)-FABMS m/z 296 [M + H]+; (+)-HRFABMS m/z [M + H]+ 295.9861 (calcd. for C11H6NO7S, 295.9865).
3.3. Biological Assays
3.3.1. In Vitro Anti-Protozoal Activity
The in vitro activities against the protozoan parasites T.b. rhodesiense, T. cruzi, L. donovani, and P. falciparum and cytotoxicity assessment against L6 cells were determined as reported elsewhere [5]. The following strains, parasite forms and positive controls were used: T.b. rhodesiense, STIB900, trypomastigote forms, melarsoprol, IC50 of 0.01 μM (4 ng/mL); T. cruzi, Tulahuen C2C4, amastigote forms in L6 rat myoblasts, benznidazole, IC50 of 1.4 μM (0.352 μg/mL); L. donovani, MHOM/ET/67/L82, axenic amastigote forms, miltefosine, IC50 of 0.5 μM (0.213 μg/mL); P. falciparum, K1 (chloroquine and pyrimethamine resistant), erythrocytic stages, chloroquine, IC50 of 0.20 μM (0.065 μg/mL) and L6 cells, rat skeletal myoblasts, podophyllotoxin, IC50 of 0.01 μM (0.004 μg/mL).
3.3.2. In Vivo Anti-Malarial Efficacy Studies
In vivo anti-malarial activity was assessed as previously described [23]. Groups of three female NMRI mice (20–22 g) were intravenously infected with 2 × 107 parasitized erythrocytes on day 0 with GFP-transfected P. berghei strain ANKA [24]. Compounds were formulated in 100% DMSO, diluted 10-fold in distilled water and administered intraperitoneally in a volume of 10 ml kg−1 on four consecutive days (4, 24, 48 and 72 h post infection). Control experiments used DMSO-H2O vehicle alone. Parasitemia was determined on day 4 post infection (24 h after last treatment) by FACS analysis. Activity was calculated as the difference between the mean per cent parasitaemia for the control (n = 5 mice) and treated groups expressed as a per cent relative to the control group. The survival of the animals was usually monitored up to 30 days: a compound was considered curative if the animal survived to day 30 after infection with no detectable parasites. In vivo efficacy studies in mice were conducted according to the rules and regulations for the protection of animal rights (“Tierschutzverordnung”) of the Swiss “Bundesamt für Veterinärwesen”. They were approved by the veterinary office of Canton Basel-Stadt, Switzerland.
4. Conclusions
The dioxothiazinoquinone marine natural product ascidiathiazone A (2) has been identified as a moderate in vitro growth inhibitor of Trypanosoma brucei rhodesiense and Plasmodium falciparum. A series of C-7 amide and Δ2(3) analogues were prepared that explored the influence of lipophilicity and oxidation state on observed anti-protozoal activity and selectivity. Little variation in anti-malarial potency was observed (IC50 0.62–6.5 μM), and no correlation was apparent between anti-malarial and anti-T. brucei activity. Changing the quinoline-based structure of 2 to incorporate benzofuran or benzothiophene moieties yielded particularly potent anti-malarials. The finding of ip and oral dosing anti-malarial activity for benzofuran carboxylic acid 16 is highly encouraging, suggesting that future studies should be directed at exploring this novel antiprotozoal pharmacophore.
Acknowledgments
We acknowledge funding from AMRF (81547) and the University of Auckland. We thank M. Cal, C. Braghiroli and G.Riccio (Swiss TPH) for parasite assay results, M. Schmitz for assistance with NMR data acquisition, and Ms. R. Imatdieva and N. Lloyd for MS data.
Conflict of Interest
The authors declare no conflict of interest.
References
- Guantai, E.; Chibale, K. How can natural products serve as a viable source of lead compounds for the development of new/novel anti-malarials? Malar. J. 2011, 10 (Suppl. 1), S2. [Google Scholar] [CrossRef]
- Kumar, V.; Mahajan, A.; Chibale, K. Synthetic medicinal chemistry of selected antimalarial natural products. Bioorg. Med. Chem. 2009, 17, 2236–2275. [Google Scholar] [CrossRef]
- Hsu, E. Reflections on the “discovery” of the antimalarial qinghao. Br. J. Clin. Pharmacol. 2006, 61, 666–670. [Google Scholar] [CrossRef]
- Dondorp, A.M.; Yeung, S.; White, L.; Nguon, C.; Day, N.P.J.; Socheat, D.; von Seidlein, L. Artemisinin resistance: Current status and scenarios for containment. Nat. Rev. Microbiol. 2010, 8, 272–280. [Google Scholar]
- Orhan, I.; Şener, B.; Kaiser, M.; Brun, R.; Tasdemir, D. Inhibitory activity of marine sponge-derived natural products against parasitic protozoa. Mar. Drugs 2010, 8, 47–58. [Google Scholar] [CrossRef]
- Watts, K.R.; Tenney, K.; Crews, P. The structural diversity and promise of antiparasitic invertebrate-derived small molecules. Curr. Opin. Biotechnol. 2010, 21, 808–818. [Google Scholar] [CrossRef]
- Fattorusso, E.; Taglialatela-Scafarti, O. Marine antimalarials. Mar. Drugs 2009, 7, 130–152. [Google Scholar] [CrossRef]
- McCracken, S.T.; Kaiser, M.; Boshoff, H.I.; Boyd, P.D.W.; Copp, B.R. Synthesis and antimalarial and antituberculosis activities of a series of natural and unnatural 4-methoxy-6-styryl-pyran-2-ones, Dihydro analogues and photo-dimers. Bioorg. Med. Chem. 2012, 20, 1482–1493. [Google Scholar] [CrossRef]
- Chan, S.T.S.; Pearce, A.N.; Januario, A.H.; Page, M.J.; Kaiser, M.; McLaughlin, R.J.; Harper, J.L.; Webb, V.L.; Barker, D.; Copp, B.R. Anti-inflammatory and antimalarial Meroterpenoids from the New Zealand ascidian Aplidium scabellum. J. Org. Chem. 2011, 76, 9151–9156. [Google Scholar] [CrossRef]
- Chan, S.T.S.; Pearce, A.N.; Page, M.J.; Kaiser, M.; Copp, B.R. Antimalarial β-carbolines from the New Zealand ascidian Pseudodistoma opacum. J. Nat. Prod. 2011, 74, 1972–1979. [Google Scholar] [CrossRef]
- Finlayson, R.; Pearce, A.N.; Page, M.J.; Kaiser, M.; Bourguet-Kondracki, M.-L.; Harper, J.L.; Webb, V.L.; Copp, B.R. Didemnidines A and B, indole spermidine alkaloids from the New Zealand ascidian Didemnum sp. J. Nat. Prod. 2011, 74, 888–892. [Google Scholar] [CrossRef]
- Wang, J.; Bourguet-Kondracki, M.-L.; Longeon, A.; Dubois, J.; Valentin, A.; Copp, B.R. Chemical and biological explorations of the electrophilic reactivity of the bioactive marine natural product halenaquinone with biomimetic nucleophiles. Bioorg. Med. Chem. Lett. 2011, 21, 1261–1264. [Google Scholar] [CrossRef]
- Pearce, A.N.; Chia, E.W.; Berridge, M.V.; Maas, E.W.; Page, M.J.; Harper, J.L.; Webb, V.L.; Copp, B.R. Orthidines A–E, tubastrine, 3,4-dimethoxyphenethyl-β-guanidine, and 1,14-sperminedihomovanillamide: potential anti-inflammatory alkaloids isolated from the New Zealand ascidian Aplidium orthium that act as inhibitors of neutrophil respiratory burst. Tetrahedron 2008, 64, 5748–5755. [Google Scholar] [CrossRef]
- Liew, L.P.P.; Kaiser, M.; Copp, B.R. Discovery and preliminary structure-activity relationship analysis of 1,14-sperminediphenylacetamides as potent and selective antimalarial lead compounds. Bioorg. Med. Chem. Lett. 2013, 23, 452–454. [Google Scholar] [CrossRef]
- Liew, L.P.P.; Pearce, A.N.; Kaiser, M.; Copp, B.R. Synthesis and in vitro and in vivo evaluation of antimalarial polyamines. Eur. J. Med. Chem. 2013, 69, 22–31. [Google Scholar] [CrossRef]
- Pearce, A.N.; Chia, E.W.; Berridge, M.V.; Clark, G.R.; Harper, J.L.; Larsen, L.; Maas, E.W.; Page, M.J.; Perry, N.B.; Webb, V.L.; et al. Anti-inflammatory thiazine alkaloids isolated from the New Zealand ascidian Aplidium sp.: Inhibitors of the neutrophils respiratory burst in a model of gouty arthritis. J. Nat. Prod. 2007, 70, 936–940. [Google Scholar] [CrossRef]
- Tetko, I.V.; Gasteiger, J.; Todeschini, R.; Mauri, A.; Livingstone, D.; Ertl, P.; Palyulin, V.A.; Radchenko, E.V.; Zefirov, N.S.; Makarenko, A.S.; et al. Virtual computational chemistry laboratory—design and description. J. Comput. Aided Mol. Des. 2005, 19, 453–463. [Google Scholar] [CrossRef]
- ALOGPS 2.1; Virtual Computational Chemistry Laboratory: Neuherberg, Germany, 2005.
- Khalil, I.M.; Barker, D.; Copp, B.R. Biomimetic synthesis of thiaplidiaquinones A and B. J. Nat. Prod. 2012, 75, 2256–2260. [Google Scholar] [CrossRef]
- Carbone, A.; Lucas, C.L.; Moody, C.J. Biomimetic synthesis of the apoptosis-inducing thiazinoquinone thiaplidiaquinone A. J. Org. Chem. 2012, 77, 9179–9189. [Google Scholar] [CrossRef]
- Aiello, A.; Fattorusso, E.; Luciano, P.; Menna, M.; Calzado, M.A.; Munoz, E.; Bonadies, F.; Guiso, M.; Sanasi, M.F.; Cocco, G.; et al. Synthesis of structurally simplified analogues of aplidinone A, a pro-apoptotic marine thiazinoquinone. Bioorg. Med. Chem. 2010, 18, 719–727. [Google Scholar] [CrossRef]
- Ruiz, V.M.; Tapia, R.; Valderrama, J.; Vega, J.C. Studies on quinones. VII. Synthesis of some benzo[b]thiophene-4,7-diones. Heterocyclic Chem. 1981, 18, 1161–1164. [Google Scholar] [CrossRef]
- Peters, W. Chemotherapy, and Drug Resistance in Malaria; Academic Press, Inc.: New York, NY, USA, 1987; pp. 145–273. [Google Scholar]
- Franke-Fayard, B.; Trueman, H.; Ramesar, J.; Mendoza, J.; van der Keur, M.; van der Linden, R.; Sinden, R.E.; Waters, A.P.; Janse, C.J. A Plasmodium berghei reference line that constitutively expresses GFP at a high level throughout the complete life cycle. Mol. Biochem. Parasitol. 2004, 137, 23–33. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).