Hepatotoxic Seafood Poisoning (HSP) Due to Microcystins: A Threat from the Ocean?
Abstract
:1. Introduction
| Microcystis aeruginosa NIES-843 | Microcystis aeruginosa UV027 | Microcystis aeruginosa PCC7806 | Planktothrix agardhii | Planktothrix rubescens NIVA-KYA 98 | ||||||
| G | P | G | P | G | P | G | P | G | P | |
| mcyA | 71% (94%) | 68% (99%) | 71% (94%) | 68% (99%) | 71% (94%) | 68% (99%) | 67% (94%) | 66% (99%) | 67% (82%) | 66% (84%) |
| mcyB | 74% (99%) | 72% (100%) | 74% (99%) | 72% (100%) | 75% (99%) | 69% (100%) | 72% (99%) | 70% (100%) | 72% (99%) | 70% (100%) |
| mcyC | 76% (99%) | 74% (99%) | 75% (99%) | 74% (98%) | 75% (99%) | 74% (99%) | 81% (99%) | 80% (99%) | 81% (99%) | 80% (99%) |
| mcyD | 73% (99%) | 69% (99%) | 73% (99%) | 69% (99%) | 77% (99%) | 73% (99%) | 77% (99%) | 73% (99%) | ||
| mcyE | 76% (98%) | 75% (100%) | 75% (98%) | 75% (100%) | 79% (99%) | 77% (100%) | 79% (99%) | 77% (100%) | ||
| mcyF | 74% (90%) | 69% (91%) | 74% (90%) | 70% (91%) | ||||||
| mcyG | 76% (97%) | 71% (99%) | 75% (97%) | 71% (99%) | 79% (97%) | 75% (98%) | 79% (97%) | 75% (98%) | ||
| mcyH | 76% (94%) | 72% (96%) | 76% (93%) | 73% (95%) | 79% (94%) | 75% (99%) | 79% (94%) | 75% (99%) | ||
| mcyI | 75% (95%) | 74% (97%) | 75% (94%) | 72% (97%) | ||||||
| mcyJ | 81% (98%) | 83% (89%) | 81% (98%) | 82% (89%) | 81% (99%) | 83% (88%) | 81% (99%) | 83% (88%) | ||
2. Microcystins in the World Ocean
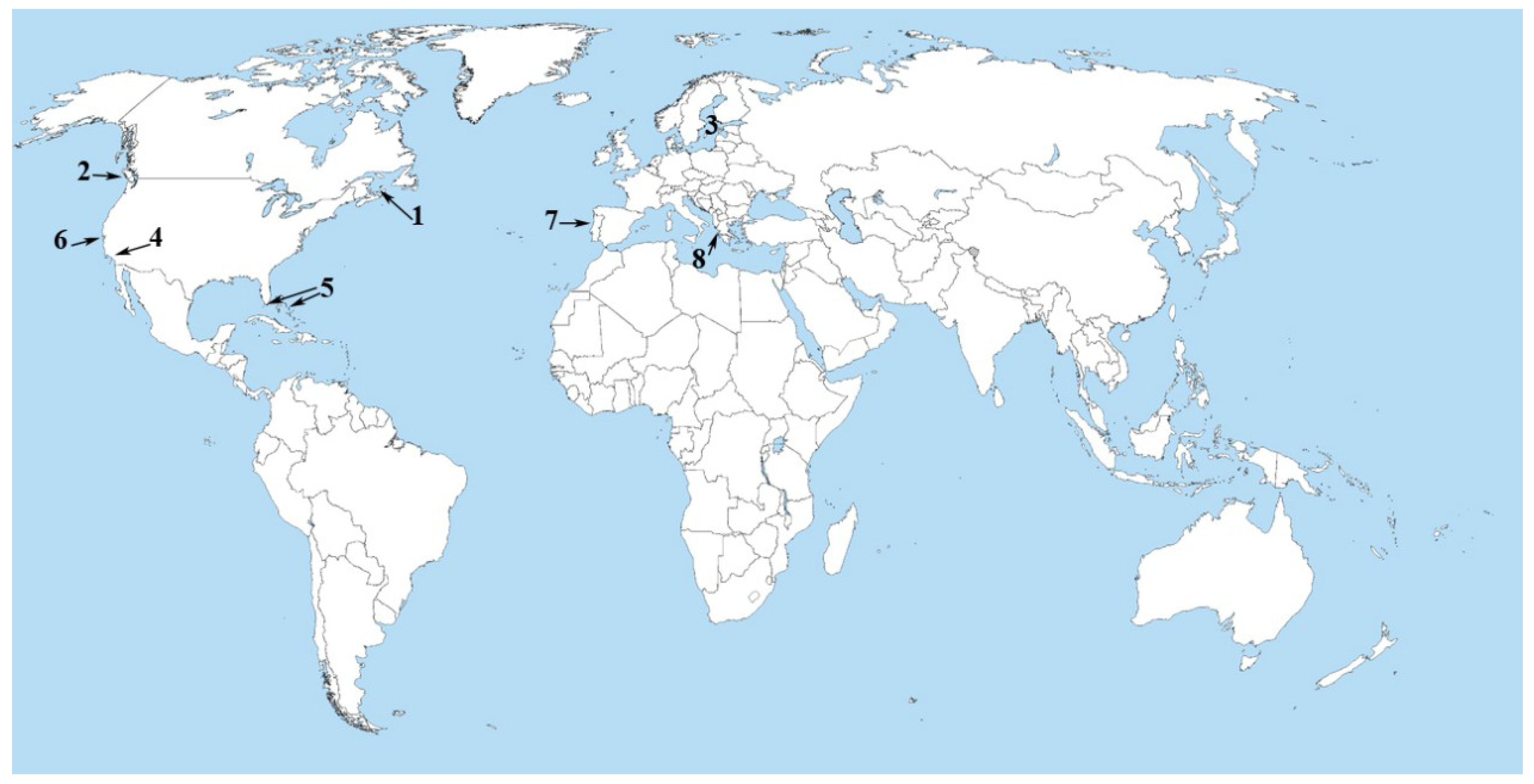
3. The Black Band Disease of Corals
4. Back to the Land—The Salton Sea
5. Atlantic Ocean (Portuguese Coastal Waters)
6. Mediterranean Sea (Amvrakikos Gulf, Greece)
7. Conclusions
Acknowledgments
References
- Carmichael, W.W. The toxins of cyanobacteria. Sci. Am. 1994, 270, 78–86. [Google Scholar] [CrossRef]
- Van Apeldoorn, M.E.; van Egmond, H.P.; Speijers, G.J.; Bakker, G.J. Toxins of cyanobacteria. Mol. Nutr. Food Res. 2007, 51, 7–60. [Google Scholar] [CrossRef]
- Martins, J.C.; Vasconcelos, V.M. Microcystin dynamics in aquatic organisms. J. Toxicol. Environ. Health B 2009, 12, 65–82. [Google Scholar] [CrossRef]
- Sainis, I.; Fokas, D.; Vareli, K.; Tzakos, A.G.; Kounnis, V.; Briasoulis, E. Cyanobacterial cyclopeptides as lead compounds to novel targeted cancer drugs. Mar. Drugs 2010, 8, 629–657. [Google Scholar] [CrossRef]
- Chorus, I.; Falconer, I.R.; Salas, H.J.; Bartram, J. Health risks caused by freshwater cyanobacteria in recreational waters. J. Toxicol. Environ. Health B 2000, 3, 323–347. [Google Scholar] [CrossRef]
- Mulvenna, V.; Dale, K.; Priestly, B.; Mueller, U.; Humpage, A.; Shaw, G.; Allinson, G.; Falconer, I. Health risk assessment for cyanobacterial toxins in seafood. Int. J. Environ. Res. Public Health 2012, 9, 807–820. [Google Scholar] [CrossRef]
- Falconer, I.R.; Choice, A.; Hosja, W. Toxicity of edible mussels (Mytilus edulis) growing naturally in an estuary during a water bloom of the blue-green alga Nodularia spumigena. Environ. Toxicol. Water Qual. 1992, 7, 119–123. [Google Scholar] [CrossRef]
- Moffitt, M.C.; Neilan, B.A. On the presence of peptide synthetase and polyketide synthase genes in the cyanobacterial genus Nodularia. FEMS Microbiol. Lett. 2001, 196, 207–214. [Google Scholar] [CrossRef]
- Sivonen, K.; Jones, G. Cyanobacterial Toxins. In Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring, and Management; Chorus, I., Bartram, J., Eds.; Spoon: London, UK, 1999; pp. 41–111. [Google Scholar]
- Zurawell, R.W.; Chen, H.; Burke, J.M.; Prepas, E.E. Hepatotoxic cyanobacteria: A review of the biological importance of microcystins in freshwater environments. J. Toxicol. Environ. Health B 2005, 8, 1–37. [Google Scholar] [CrossRef]
- Pearson, L.A.; Moffitt, M.C.; Ginn, H.P.; Neilan, B.A. The molecular genetics and regulation of cyanobacterial peptide hepatotoxin biosynthesis. Crit. Rev. Toxicol. 2008, 38, 847–856. [Google Scholar] [CrossRef]
- Tillett, D.; Dittmann, E.; Erhard, M.; von Döhren, H.; Börner, T.; Neilan, B.A. Structural organization of microcystin biosynthesis in Microcystis aeruginosa PCC7806: An integrated peptide–polyketide synthetase system. Chem. Biol. 2000, 7, 753–764. [Google Scholar] [CrossRef]
- Christiansen, G.; Fastner, J.; Erhard, M.; Börner, T.; Dittmann, E. Microcystin biosynthesis in Planktothrix: Genes, evolution, and manipulation. J. Bacteriol. 2003, 185, 564–572. [Google Scholar] [CrossRef]
- Rouhiainen, L.; Vakkilainen, T.; Siemer, B.L.; Buikema, W.; Haselkorn, R.; Sivonen, K. Genes coding for hepatotoxic heptapeptides (microcystins) in the cyanobacterium Anabaena strain 90. Appl. Environ. Microbiol. 2004, 70, 686–692. [Google Scholar] [CrossRef]
- Kaneko, T.; Narajima, N.; Okamoto, S.; Suzuki, I.; Tanabe, Y.; Tamaoki, M.; Nakamura, Y.; Kasai, F.; Watanabe, A.; Kawashima, K.; et al. Complete genomic structure of the bloom-forming toxic cyanobacterium Microcystis aeruginosa NIES-843. DNA Res. 2007, 14, 247–256. [Google Scholar]
- Frangeul, L.; Quillardet, P.; Castets, A.M.; Humbert, J.F.; Matthijs, H.C.; Cortez, D.; Tolonen, A.; Zhang, C.C.; Gribaldo, S.; Kehr, J.C.; et al. Highly plastic genome of Microcystis aeruginosa PCC 7806, a ubiquitous toxic freshwater cyanobacterium. BMC Genomics 2008, 9, 274. [Google Scholar] [CrossRef]
- Rounge, T.B.; Rohrlack, T.; Nederbragt, A.J.; Kristensen, T.; Jakobsen, K.S. A genome-wide analysis of nonribosomal peptide synthetase gene clusters and their peptides in a Planktothrix rubescens strain. BMC Genomics 2009, 10, 396. [Google Scholar]
- Rantala, A.; Rajaniemi-Wacklin, P.; Lyra, C.; Lepistö, L.; Rintala, J.; Mankiewicz-Boczek, J.; Sivonen, K. Detection of microcystin-producing cyanobacteria in Finnish lakes with genus-specific microcystin synthetase gene E (mcyE) PCR and associations with environmental factors. Appl. Environ. Microbiol. 2006, 72, 6101–6110. [Google Scholar] [CrossRef]
- Van der Westhuizen, A.J.; Eloff, J.N. Effect of temperature and light on the toxicity and growth of the blue-green alga Microcystis aeruginosa (UV-006). Planta 1985, 163, 55–59. [Google Scholar] [CrossRef]
- Sivonen, K. Effects of light, temperature, nitrate, orthophosphate, and bacteria on growth of and hepatotoxin production by Oscillatoria agardhii strains. Appl. Environ. Microbiol. 1990, 56, 2658–2666. [Google Scholar]
- Lukac, M.; Aegerter, R. Influence of trace metals on growth and toxin production of Microcystis aeruginosa. Toxicon 1993, 31, 293–305. [Google Scholar] [CrossRef]
- Song, L.; Sano, T.; Li, R.; Watanabe, M.; Liu, Y.; Kaya, K. Microcystin production of Microcystis viridis (cyanobacteria) under different culture conditions. Phycol. Res. 1998, 46, 19–23. [Google Scholar] [CrossRef]
- Kaebernick, M.; Neilan, B.A.; Börner, T.; Dittmann, E. Light and the transcriptional response of the microcystin biosynthesis gene cluster. Appl. Environ. Microbiol. 2000, 66, 3387–3392. [Google Scholar] [CrossRef]
- Ginn, P.H.; Pearson, A.L.; Neilan, A.B. NtcA from Microcystis aeruginosa PCC 7806 is autoregulatory and binds to the microcystin promoter. Appl. Environ. Microbiol. 2010, 76, 4362–4368. [Google Scholar] [CrossRef]
- Wei, T.F.; Ramasubramanian, T.S.; Pu, F.; Golden, J.W. Anabaena sp. strain PCC 7120 bifA gene encoding a sequence-specific DNA-binding protein cloned by in vivo transcriptional interference selection. J. Bacteriol. 1993, 175, 4025–4035. [Google Scholar]
- Ramasubramanian, T.S.; Wei, T.F.; Oldham, A.K.; Golden, J.W. Transcription of the Anabaena sp. strain PCC 7120 ntcA gene: multiple transcripts and NtcA binding. J. Bacteriol. 1996, 178, 922–926. [Google Scholar]
- Lindell, D.; Padan, E.; Post, A.F. Regulation of ntcA expression and nitrite uptake in the marine Synechococcus sp. strain WH 7803. J. Bacteriol. 1998, 180, 1878–1886. [Google Scholar]
- Muro-Pastor, A.M.; Valladares, A.; Flores, E.; Herrero, A. Mutual dependence of the expression of the cell differentiation regulatory protein HetR and the global nitrogen regulator NtcA during heterocyst development. Mol. Microbiol. 2002, 44, 1377–1385. [Google Scholar] [CrossRef]
- Codd, G.A.; Lindsay, J.; Young, F.M.; Morrison, L.F.; Metcalf, J.S. Cyanobacterial toxins. In Harmful Cyanobacteria; Huisman, J., Matthijs, H.C.P., Visser, P.M., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 1–23. [Google Scholar]
- Dietrich, D.; Hoeger, S. Guidance values for microcystins in water and cyanobacterial supplement products (blue-green algal supplements): A reasonable or misguided approach? Toxicol. Appl. Pharmacol. 2005, 203, 273–289. [Google Scholar] [CrossRef]
- Welker, M.; von Döhren, H. Cyanobacterial peptides—Nature’s own combinatorial synthesis. FEMS Microbiol. Rev. 2006, 30, 530–563. [Google Scholar] [CrossRef]
- Fischer, W.J.; Altheimer, S.; Cattori, V.; Meier, P.J.; Dietrich, D.R.; Hagenbuch, B. Organic anion transporting polypeptides expressed in liver and brain mediate uptake of microcystin. Toxicol. Appl. Pharmacol. 2005, 203, 257–263. [Google Scholar] [CrossRef]
- Hagenbuch, B.; Meier, P.J. Organic anion transporting polypeptides of the OATP/SLC21 family: Phylogenetic classification as OATP/SLCO superfamily, new nomenclature and molecular/functional properties. Pflugers Arch. 2004, 447, 653–665. [Google Scholar] [CrossRef]
- Feurstein, D.; Holst, K.; Fischer, A.; Dietrich, D.R. Oatp-associated uptake and toxicity of microcystins in primary murine whole brain cells. Toxicol. Appl. Pharmacol. 2009, 234, 247–255. [Google Scholar] [CrossRef]
- Runnegar, M.T.; Falconer, I.R.; Silver, J. Deformation of isolated rat hepatocytes by a peptide hepatotoxin from the blue-green alga Microcystis aeruginosa. Naunyn Schmiedebergs Arch. Pharmacol. 1981, 317, 268–272. [Google Scholar] [CrossRef]
- Eriksson, J.E.; Grönberg, L.; Nygård, S.; Slotte, J.P.; Meriluoto, J.A.O. Hepatocellular uptake of 3H-dihydromicrocystin-LR, a cyclic peptide toxin. Biochim. Biophys. Acta Biomembr. 1990, 1025, 60–66. [Google Scholar] [CrossRef]
- Bronger, H.; König, J.; Kopplow, K.; Steiner, H.H.; Ahmadi, R.; Herold-Mende, C.; Keppler, D.; Nies, A.T. ABCC drug efflux pumps and organic anion uptake transporters in human gliomas and the blood-tumor barrier. Cancer Res. 2005, 65, 11419–11428. [Google Scholar] [CrossRef]
- Feurstein, D.; Kleinteich, J.; Heussner, A.H; Stemmer, K.; Dietrich, D.R. Investigation of microcystin congener-dependent uptake into primary murine neurons. Environ. Health Perspect 2010, 118, 1370–1375. [Google Scholar] [CrossRef]
- Pereira, S.R.; Vasconcelos, V.M.; Antunes, A. The phosphoprotein phosphatase family of Ser/Thr phosphatases as principal targets of naturally occurring toxins. Crit. Rev. Toxicol. 2011, 41, 83–110. [Google Scholar]
- Nishiwaki-Matsushima, R.; Ohta, T.; Nishiwaki, S.; Suganuma, M.; Kohyama, K.; Ishikawa, T.; Carmichael, W.W.; Fujiki, H. Liver tumor promotion by the cyanobacterial cyclic peptide toxin microcystin-LR. J. Cancer Res. Clin. Oncol. 1992, 118, 420–424. [Google Scholar] [CrossRef]
- Janssens, V.; Goris, J. Protein phosphatase 2A: A highly regulated family of serine/threonine phosphatases implicated in cell growth and signalling. Biochem. J. 2001, 353, 417–439. [Google Scholar] [CrossRef]
- Gehringer, M.M. Microcystin-LR and okadaic acid-induced cellular effects: A dualistic response. FEBS Lett. 2004, 557, 1–8. [Google Scholar] [CrossRef]
- Janssens, V.; Goris, J.; Van Hoof, C. PP2A: The expected tumor suppressor. Curr. Opin. Genet. Dev. 2005, 15, 34–41. [Google Scholar] [CrossRef]
- Takumi, S.; Komatsu, M.; Furukawa, T.; Ikeda, R.; Sumizawa, T.; Akenaga, H.; Maeda, Y.; Aoyama, K.; Arizono, K.; Ando, S.; Takeuchi, T. p53 Plays an important role in cell fate determination after exposure to microcystin-LR. Environ. Health Perspect. 2010, 118, 1292–1298. [Google Scholar] [CrossRef]
- Falconer, I.R.; Yeung, D.S. Cytoskeletal changes in hepatocytes induced by Microcystis toxins and their relation to hyperphosphorylation of cell proteins. Chem. Biol. Interact. 1992, 81, 181–196. [Google Scholar] [CrossRef]
- Ding, W.-X.; Shen, H.-M.; Ong, C.-N. Microcystic cyanobacteria extract induces cytoskeletal disruption and intracellular glutathione alteration in hepatocytes. Environ. Health Perspect. 2000, 108, 605–609. [Google Scholar] [CrossRef]
- Lovell, R.A.; Schaeffer, D.J.; Hooser, S.B.; Haschek, W.M.; Dahlem, A.M.; Carmichael, W.W.; Beasley, V.R. Toxicity of intraperitoneal doses of microcystin-LR in two strains of male mice. J. Environ. Pathol. Toxicol. Oncol. 1989, 9, 221–237. [Google Scholar]
- Yoshida, T.; Makita, Y.; Nagata, S.; Tsutsumi, T.; Yoshida, F.; Sekijima, M.; Tamura, S.; Ueno, Y. Acute oral toxicity of microcystin-LR, a cyanobacterial hepatotoxin, in mice. Nat. Toxins 1997, 5, 91–95. [Google Scholar] [CrossRef]
- Billam, M.; Mukhi, S.; Tang, L.; Gao, W.; Wang, J.S. Toxic response indicators of microcystin-LR in F344 rats following a single-dose treatment. Toxicon 2008, 51, 1068–1080. [Google Scholar] [CrossRef]
- Moreno, I.; Pichardo, S.; Jos, A.; Gómez-Amores, L.; Mate, A.; Vazquez, C.M.; Cameán, A.M. Antioxidant enzyme activity and lipid peroxidation in liver and kidney of rats exposed to microcystin-LR administered intraperitoneally. Toxicon 2005, 45, 395–402. [Google Scholar] [CrossRef]
- Rao, P.V.; Bhattacharya, R. The cyanobacterial toxin microcystin-LR induced DNA damage in mouse liver in vivo. Toxicology 1996, 114, 29–36. [Google Scholar] [CrossRef]
- Žegura, B.; Sedmak, B.; Filipič, M. Microcystin-LR induces oxidative DNA damage in human hepatoma cell line HepG2. Toxicon 2003, 41, 41–48. [Google Scholar] [CrossRef]
- Zhan, L.; Sakamoto, H.; Sakuraba, M.; Wu, D.S.; Zhang, L.S.; Suzuki, T.; Hayashi, M.; Honma, M. Genotoxicity of microcystin-LR in human lymphoblastoid TK6 cells. Mutat. Res. 2004, 557, 1–6. [Google Scholar] [CrossRef]
- Žegura, B.; Zajc, I.; Lah, T.T.; Filipič, M. Patterns of microcystin-LR induced alteration of the expression of genes involved in response to DNA damage and apoptosis. Toxicon 2008, 51, 615–623. [Google Scholar] [CrossRef]
- Gaudin, J.; Le Hegarat, L.; Nesslany, F.; Marzin, D.; Fessard, V. In vivo genotoxic potential of microcystin-LR: A cyanobacterial toxin, investigated both by the unscheduled DNA synthesis (UDS) and the comet assays after intravenous administration. Environ. Toxicol. 2009, 24, 200–209. [Google Scholar] [CrossRef]
- Guzman, R.E.; Solter, P.F. Characterization of sublethal MC-LR exposure in mice. Vet. Pathol. 2002, 39, 17–26. [Google Scholar] [CrossRef]
- Xing, Y.; Xu, Y.; Chen, Y.; Jeffrey, P.D.; Chao, Y.; Lin, Z.; Li, Z.; Strack, S.; Stock, J.B.; Shi, Y. Structure of protein phosphatase 2A core enzyme bound to tumor-inducing toxins. Cell 2006, 127, 341–353. [Google Scholar] [CrossRef]
- Kondo, F.; Ikai, Y.; Oka, H.; Okumura, M.; Ishikawa, N.; Harada, K.; Matsuura, K.; Murata, H.; Suzuki, M. Formation, characterization, and toxicity of the glutathione and cysteine conjugates of toxic hepatopeptide microcystins. Chem. Res. Toxicol. 1992, 5, 591–596. [Google Scholar] [CrossRef]
- Kondo, F.; Matsumoto, H.; Yamada, S.; Ishikawa, N.; Ito, E.; Nagata, S.; Ueno, Y.; Suzuki, M.; Harada, K. Detection and identification of metabolites of microcystins formed in vivo in mouse and rat livers. Chem. Res. Toxicol. 1996, 9, 1355–1359. [Google Scholar] [CrossRef]
- Monserrat, J.M.; Pinho, G.L.L.; Yunes, J.S. Toxicological effects of hepatotoxins (microcystins) on aquatic organisms. Comments Toxicol. 2003, 9, 89–101. [Google Scholar] [CrossRef]
- Zhang, D.; Yang, Q.; Xie, P.; Deng, X.; Chen, J.; Dai, M. The role of cysteine conjugation in the detoxification of microcystin-LR in liver of bighead carp (Aristichthis mobilis): A field and laboratory study. Ecotoxicology 2012, 21, 244–252. [Google Scholar] [CrossRef]
- He, J.; Chen, J.; Xie, P.; Zhang, D.; Li, G.; Wu, L.; Zhang, W.; Guo, X.; Li, S. Quantitatively evaluating detoxification of the hepatotoxic microcystins through the glutathione and cysteine pathway in the cyanobacteria-eating bighead carp. Aquat. Toxicol. 2012, 116–117, 61–68. [Google Scholar]
- Žegura, B.; Straser, A.; Filipič, M. Genotoxicity and potential carcinogenicity of cyanobacterial toxins—A review. Mutat Res. 2011, 727, 16–41. [Google Scholar] [CrossRef]
- Rantala, A.; Fewer, D.P.; Hisbergues, M.; Rouhiainen, L.; Vaitomaa, J.; Börner, T.; Sivonen, K. Phylogenetic evidence for the early evolution of microcystin synthesis. Proc. Natl. Acad. Sci. USA 2004, 101, 568–573. [Google Scholar]
- Utkilen, H.; Gjolme, N. Iron-stimulated toxin production in Microcystis aeruginosa. Appl. Environ. Microbiol. 1995, 61, 797–800. [Google Scholar]
- Dittmann, E.; Erhard, M.; Kaebernick, M.; Scheler, C.; Neilan, B.A.; von Döhren, H.; Börner, T. Altered expression of two light-dependent genes in microcystin-lacking mutant of Microcystis aeruginosa PCC 7806. Microbiol. 2001, 147, 3113–3119. [Google Scholar]
- Armitage, A.R.; Fong, P. Upward cascading effects of nutrients: shifts in a benthic microalgal community and a negative herbivore response. Oecologia 2004, 139, 560–567. [Google Scholar] [CrossRef]
- Schatz, D.; Keren, Y.; Vardi, A.; Sukenik, A.; Carmeli, S.; Börner, T.; Dittmann, E.; Kaplan, A. Towards clarification of the biological role of microcystins, a family of cyanobacterial toxins. Environ. Microbiol. 2007, 9, 963–970. [Google Scholar]
- Jang, M.H.; Ha, K.; Takamura, N. Microcystin production by Microcystis aeruginosa exposed to different stages of herbivorous zooplankton. Toxicon 2008, 51, 882–889. [Google Scholar] [CrossRef]
- Zilliges, Y.; Kehr, J.C.; Meissner, S.; Ishida, K.; Mikkat, S.; Hagemann, M.; Kaplan, A.; Börner, T.; Duittmann, E. The cyanobacterial hepatotoxin microcystin binds to proteins and increases the fitness of Microcystis under oxidative stress conditions. PLoS One 2011, 6, e17615. [Google Scholar] [CrossRef]
- Gilroy, D.J.; Kauffman, K.W.; Hall, R.A.; Huang, X.; Chu, F.S. Assessing potential health risks from microcystin toxins in blue-green algae dietary supplements. Environ. Health Perspect. 2000, 108, 435–439. [Google Scholar] [CrossRef] [Green Version]
- O’Neil, J.M.; Davis, T.W.; Burford, M.A.; Gobler, C.J. The rise of harmful cyanobacteria blooms: The potential roles of eutrophication and climate change. Harmful Algae 2012, 14, 313–334. [Google Scholar] [CrossRef]
- Chen, D.X.Z.; Boland, M.P.; Smillie, M.A.; Klix, H.; Ptak, C.; Andersen, R.J.; Holmes, C.F.B. Identification of protein phosphatase inhibitors of the microcystin class in the marine environment. Toxicon 1993, 31, 1407–1414. [Google Scholar] [CrossRef]
- Andersen, R.J.; Luu, H.A.; Chen, D.Z.X.; Holmes, C.F.B.; Kent, M.; LeBlanc, M.; Taylor, F.J.R.; Williams, D.E. Chemical and biological evidence links microcystins to Salmon Netpen Liver Disease. Toxicon 1993, 31, 1315–1323. [Google Scholar] [CrossRef]
- Sivonen, K.; Kononen, K.; Carmichael, W.W.; Dahlem, A.M.; Rinehart, K.L.; Kiviranta, J.; Niemela, S.I. Occurrence of the hepatotoxic cyanobacterium Nodularia spumigena in the Baltic Sea and the structure of the toxin. Appl. Environ. Microbiol. 1989, 55, 1990–1995. [Google Scholar]
- Repka, S.; Meyerhöfer, M.; von Bröckel, K.; Sivonen, K. Associations of cyanobacterial toxin, nodularin, with environmental factors and zooplankton in the Baltic Sea. Microb. Ecol. 2004, 47, 350–358. [Google Scholar]
- Karlsson, K.; Kankaanpää, H.; Huttunen, M.; Meriluoto, J. First observation of microcystin-LR in pelagic cyanobacterial blooms in the northern Baltic Sea. Harmful Algae 2005, 4, 163–166. [Google Scholar] [CrossRef]
- Kankaanpää, H.T.; Sjövall, O.; Huttunen, M.; Olin, M.; Karlsson, K.; Hyvärinen, K.; Sneitz, L.; Härkönen, J.; Sipiä, V.O.; Meriluoto, J.A. Production and sedimentation of peptide toxins nodularin-R and microcystin-LR in the northern Baltic Sea. Environ. Pollut. 2009, 157, 1301–1309. [Google Scholar] [CrossRef]
- Samuelsson, M. Interannual salinity variations in the Baltic Sea during the period 1954–1990. Cont. Shelf. Res. 1996, 16, 1463–1477. [Google Scholar] [CrossRef]
- Herlemann, P.R.D.; Labrenz, M.; Jürgens, K.; Bertilsson, S.; Waniek, J.J.; Andersson, F.A. Transitions in bacterial communities along the 2000 km salinity gradient of the Baltic Sea. ISME J. 2011, 5, 1571–1579. [Google Scholar] [CrossRef]
- Johnston, R.B.; Jacoby, M.J. Cyanobacterial toxicity and migration in a mesotrophic lake in western Washington, USA. Hydrobiologia 2003, 495, 79–91. [Google Scholar] [CrossRef]
- Moisander, P.H.; Lehman, P.W.; Ochiai, M.; Corum, S. Diversity of Microcystis aeruginosa in the Klamath River and San Francisco Bay delta, California, USA. Aquat. Microb. Ecol. 2009, 57, 19–31. [Google Scholar] [CrossRef]
- Moisander, P.H.; Ochiai, M.; Lincoff, A. Nutrient limitation of Microcystis aeruginosa in northern California Klamath River reservoirs. Harmful Algae 2009, 8, 889–897. [Google Scholar] [CrossRef]
- Fetcho, K. Final 2007 Klamath River Blue-Green Algae Summary Report; Yurok Tribe Environmental Program: Klamath, CA, USA, 2008. The Yurok Tribe Home Page. Available online: http://www.yuroktribe.org/departments/ytep/documents/2007YurokFINALBGAReport071708.pdf (accessed on 14 April 2013).
- Lehman, P.W.; Boyer, G.; Hall, C.; Waller, S.; Gehrts, K. Distribution and toxicity of a new colonial Microcystis aeruginosa bloom in the San Francisco Bay Estuary, California. Hydrobiologia 2005, 541, 87–99. [Google Scholar] [CrossRef]
- Miller, M.A.; Kudela, R.M.; Mekebri, A.; Crane, D.; Oates, S.C.; Tinker, M.T.; Staedler, M.; Miller, W.A.; Toy-Choutka, S.; Dominik, C.; et al. Evidence for a novel marine harmful algal bloom: Cyanotoxin (microcystin) transfer from land to sea otters. PLoS One 2010, 5, e12576. [Google Scholar] [CrossRef]
- Dawson, R.M. The toxicology of microcystins. Toxicon 1998, 36, 953–962. [Google Scholar] [CrossRef]
- Fischer, W.J.; Dietrich, D.R. Pathological and biochemical characterization of microcystin-induced hepatopancreas and kidney damage in carp (Cyprinus carpio). Toxicol. Appl. Pharmacol. 2000, 164, 73–81. [Google Scholar] [CrossRef]
- Richardson, L.L.; Sekar, R.; Myers, J.L.; Gantar, M.; Voss, J.D.; Kaczmarsky, L.; Remily, E.R.; Boyer, G.L.; Zimba, P.V. The presence of the cyanobacterial toxin microcystin in black band disease of corals. FEMS Microbiol. Lett. 2007, 272, 182–187. [Google Scholar] [CrossRef]
- Kurmayer, R.; Christiansen, G. The genetic basis of toxin production in Cyanobacteria. Freshwater Rev. 2009, 2, 31–50. [Google Scholar]
- Tillett, D.; Parker, D.L.; Neilan, B.A. Detection of toxigenicity by a probe for the microcystin synthetase A gene (mcyA) of the cyanobacterial genus Microcystis: Comparison of toxicities with 16S rRNA and phycocyanin operon (phycocyanin intergenic spacer) phylogenies. Appl. Environ. Microbiol. 2001, 67, 2810–2818. [Google Scholar] [CrossRef]
- Hisbergues, M.; Christiansen, G.; Rouhiainen, L.; Sivonen, K.; Börner, T. PCR-based identification of microcystin producing genotypes of different cyanobacterial genera. Arch. Microbiol. 2003, 180, 402–410. [Google Scholar] [CrossRef]
- Stanić, D.; Oehrle, S.; Gantar, M.; Richardson, L.L. Microcystin production and ecological physiology of Caribbean black band disease cyanobacteria. Environ. Microbiol. 2011, 13, 900–910. [Google Scholar] [CrossRef]
- Carmichael, W.W.; Li, R.-H. Cyanobacteria toxins in the Salton Sea. Saline Syst. 2006, 2, 5. [Google Scholar] [CrossRef]
- Martins, R.; Pereira, P.; Welker, M.; Fastner, J.; Vasconcelos, V.M. Toxicity of culturable cyanobacteria strains isolated from the Portuguese coast. Toxicon 2005, 46, 454–464. [Google Scholar] [CrossRef]
- Carmichael, W.W. The cyanotoxins. In Advances in Botanical Research; Gallow, J., Ed.; Academic Press: London, UK, 1997; Volume 27, pp. 211–256. [Google Scholar]
- Teneva, I.; Asparuhova, D.; Dzhambazov, B.; Mladenov, R.; Schirmer, K. The freshwater cyanobacterium Lyngbya aerugineo-coerulea produces compounds toxic to mice and to mammalian and fish cells. Environ. Toxicol. 2003, 18, 9–20. [Google Scholar] [CrossRef]
- Teneva, I.; Dzhambazov, B.; Koleva, L.; Mladenov, R.; Schirmer, K. Toxic potential of five freshwater Phormidium species (Cyanoprokaryota). Toxicon 2005, 45, 711–725. [Google Scholar] [CrossRef]
- Frazão, B.; Martins, R.; Vasconcelos, V. Are known cyanotoxins involved in the toxicity of picoplanktonic and filamentous North Atlantic marine cyanobacteria? Mar. Drugs 2010, 8, 1906–1919. [Google Scholar]
- Kaebernick, M.; Rohrlack, T.; Christoffersen, K.; Neilan, B.A. A spontaneous mutant of microcystin biosynthesis: Genetic characterization and effect on Daphnia. Environ. Microbiol. 2001, 3, 669–679. [Google Scholar] [CrossRef]
- Vareli, K.; Zarali, E.; Zacharioudakis, G.S.A.; Vagenas, G.; Varelis, V.; Pilidis, G.; Briasoulis, E.; Sainis, I. Microcystin producing cyanobacterial communities in Amvrakikos Gulf (Mediterranean Sea, NW Greece) and toxin accumulation in mussels (Mytilus galloprovincialis). Harmful Algae 2012, 15, 109–118. [Google Scholar] [CrossRef]
- World Health Organization, Guidelines for Drinking-Water Quality, 2nd ed.; World Health Organization: Geneva, Switzerland, 1998; Volume 2.
- Nonneman, D.; Zimba, P. A PCR-based test to assess the potential for microcystin occurrence in channel catfish production ponds. J. Phycol. 2002, 38, 230–233. [Google Scholar] [CrossRef]
- Vareli, K.; Briasoulis, E.; Pilidis, G.; Sainis, I. Molecular confirmation of Planktothrix rubescens as the cause of intense, microcystin—Synthesizing cyanobacterial bloom in Lake Ziros, Greece. Harmful Algae 2009, 8, 447–453. [Google Scholar] [CrossRef]
- Vareli, K.; Pilidis, G.; Mavrogiorgou, M.-C.; Briasoulis, E.; Sainis, I. Molecular characterization of cyanobacterial diversity and yearly fluctuations of Microcystin loads in a suburban Mediterranean Lake (Lake Pamvotis, Greece). J. Environ. Monit. 2009, 11, 1506–1512. [Google Scholar] [CrossRef]
- Ahlgren, N.A.; Rocap, G. Culture isolation and culture independent clone libraries reveal new marine Synechococcus ecotypes with distinctive light and N physiologies. Appl. Environ. Microbiol. 2006, 72, 7193–7204. [Google Scholar] [CrossRef]
- Choi, D.H.; Noh, J.H. Phylogenetic diversity of Synechococcus strains isolated from the East China Sea and the East Sea. FEMS Microbiol. Ecol. 2009, 69, 439–448. [Google Scholar] [CrossRef]
- Korelusová, J.; Kaštovsky, J.; Komárek, J. Heterogeneity of the cyanobacterial genus Synechocystis and description of a new genus, Geminocystis. J. Phycol. 2009, 45, 928–937. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Vareli, K.; Jaeger, W.; Touka, A.; Frillingos, S.; Briasoulis, E.; Sainis, I. Hepatotoxic Seafood Poisoning (HSP) Due to Microcystins: A Threat from the Ocean? Mar. Drugs 2013, 11, 2751-2768. https://doi.org/10.3390/md11082751
Vareli K, Jaeger W, Touka A, Frillingos S, Briasoulis E, Sainis I. Hepatotoxic Seafood Poisoning (HSP) Due to Microcystins: A Threat from the Ocean? Marine Drugs. 2013; 11(8):2751-2768. https://doi.org/10.3390/md11082751
Chicago/Turabian StyleVareli, Katerina, Walter Jaeger, Anastasia Touka, Stathis Frillingos, Evangelos Briasoulis, and Ioannis Sainis. 2013. "Hepatotoxic Seafood Poisoning (HSP) Due to Microcystins: A Threat from the Ocean?" Marine Drugs 11, no. 8: 2751-2768. https://doi.org/10.3390/md11082751
APA StyleVareli, K., Jaeger, W., Touka, A., Frillingos, S., Briasoulis, E., & Sainis, I. (2013). Hepatotoxic Seafood Poisoning (HSP) Due to Microcystins: A Threat from the Ocean? Marine Drugs, 11(8), 2751-2768. https://doi.org/10.3390/md11082751





