Screening Tests for the Rapid Detection of Diarrhetic Shellfish Toxins in Washington State
Abstract
:1. Introduction
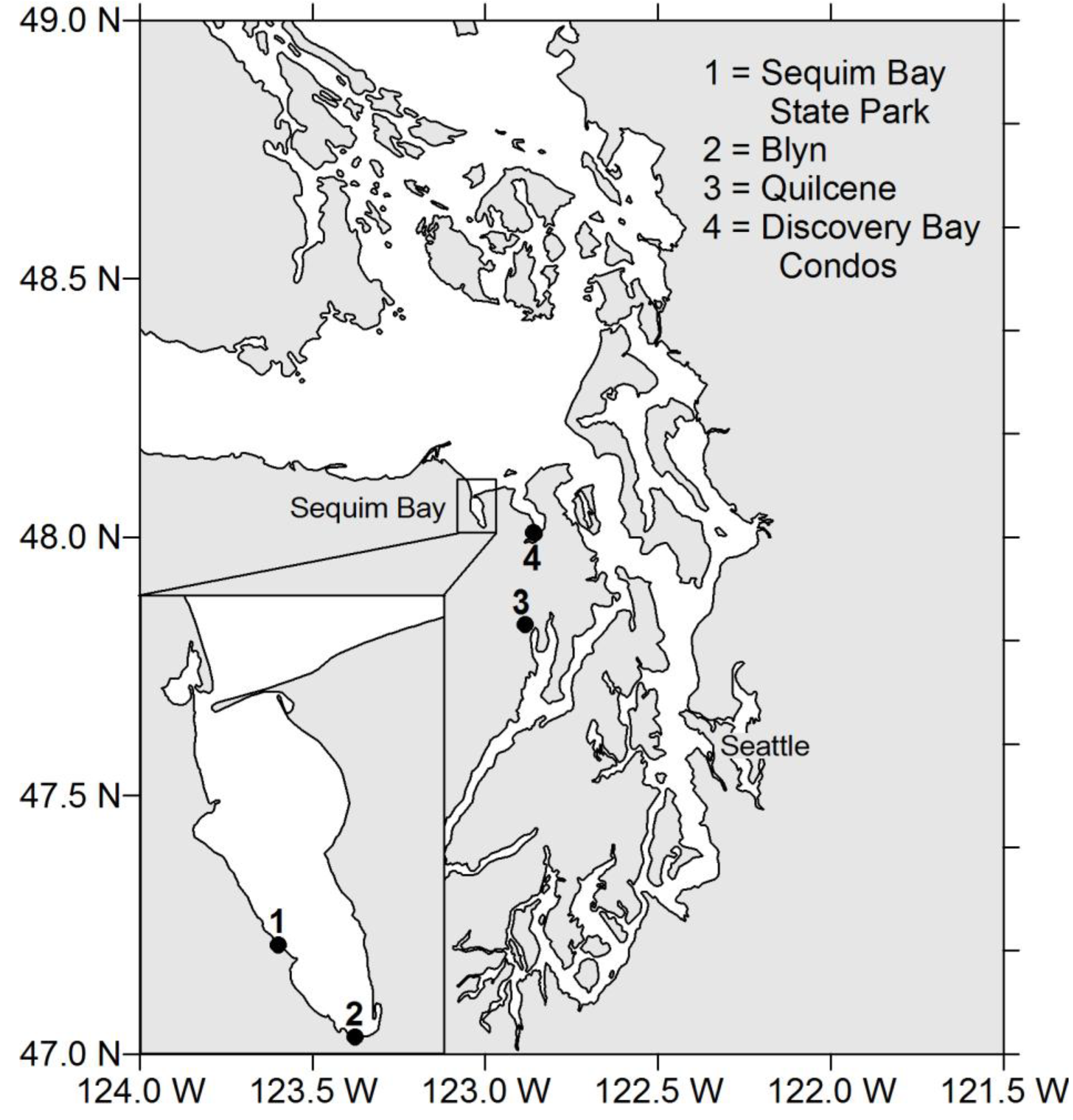
2. Results
2.1. Analysis of DSTs by LC-MS/MS
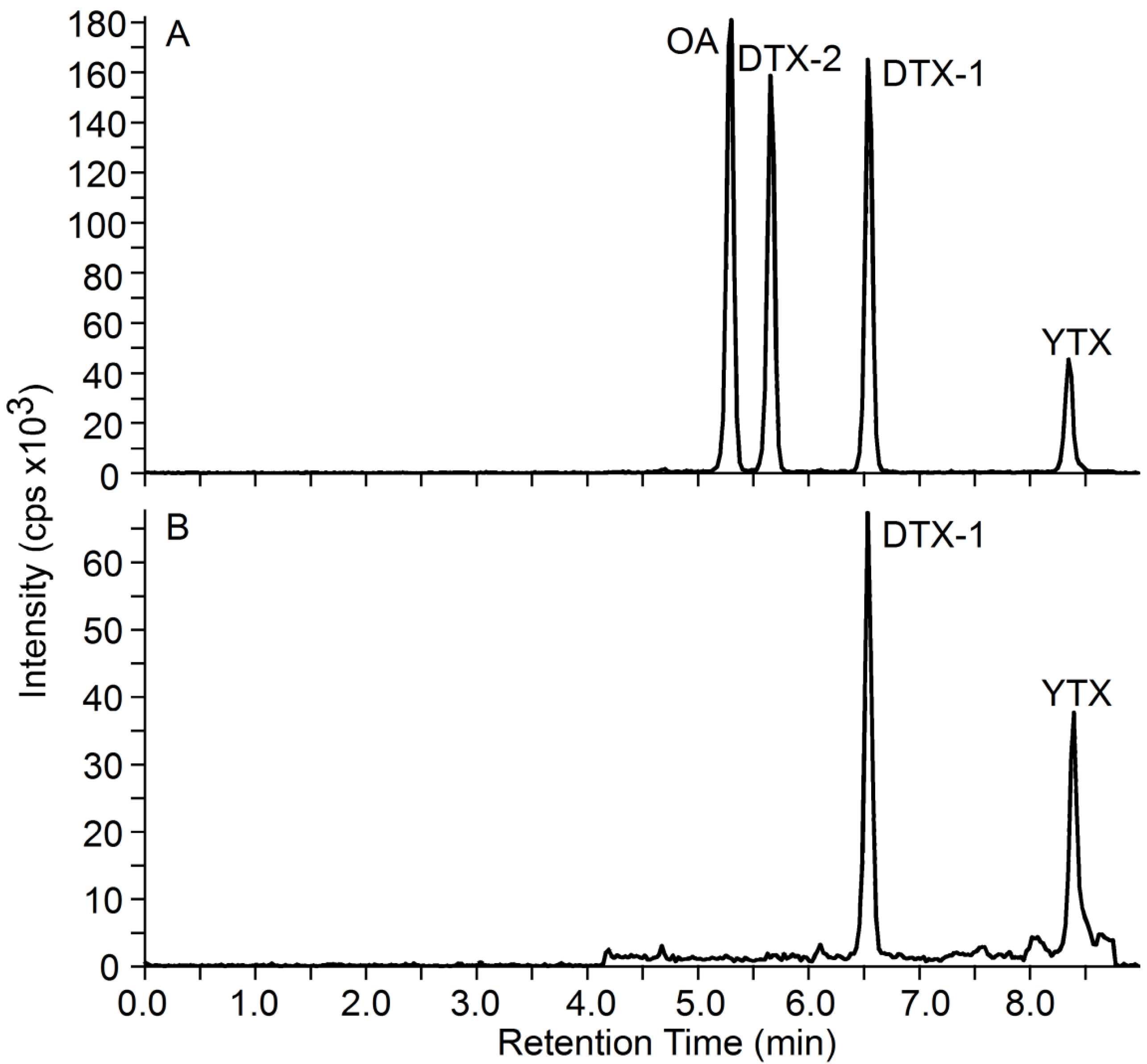
2.2. Tissue Matrix Effects
| Hydrolyzed | DST concentration (μg total OA equiv./100 g shellfish) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Site | OA | DTX-2 | DTX-1 | OA | DTX-2 | DTX-1 | OA | DTX-2 | DTX-1 |
| (% extract) a | 100 | 100 | 100 | 50 | 50 | 50 | 10 | 10 | 10 |
| Quilcene | bd | bd | 27.92 | bd | bd | 30.99 | na | na | na |
| Discovery Bay Condos | bd | bd | 25.64 | bd | bd | 25.61 | na | na | na |
| Discovery Bay Condos | bd | bd | 21.84 | bd | bd | 22.23 | na | na | na |
| Sequim Bay State Park | bd | bd | 50.31 | bd | bd | 46.32 | na | na | na |
| Sequim Bay State Park | bd | bd | 19.09 | bd | bd | 16.91 | na | na | na |
| Sequim Bay State Park | bd | bd | 82.87 | bd | bd | 82.48 | na | na | na |
| Sequim Bay Blyn | bd | bd | 37.08 | bd | bd | 33.62 | na | na | na |
| Sequim Bay Blyn | bd | bd | 31.53 | bd | bd | 29.87 | na | na | na |
| Non-hydrolyzed | DST concentration (μg OA equiv./100 g shellfish) | ||||||||
| Site | OA | DTX-2 | DTX-1 | OA | DTX-2 | DTX-1 | OA | DTX-2 | DTX-1 |
| (% extract) a | 100 | 100 | 100 | 50 | 50 | 50 | 10 | 10 | 10 |
| Quilcene | 1.33 | bd | 14.18 | bd | bd | 13.72 | bd | bd | 12.86 |
| Discovery Bay Condos | bd | bd | 13.53 | bd | bd | 11.40 | bd | bd | 10.72 |
| Discovery Bay Condos | bd | bd | 11.90 | bd | bd | 10.40 | bd | bd | 10.01 |
| Sequim Bay State Park | bd | bd | 31.65 | bd | bd | 25.86 | bd | bd | 16.84 |
| Sequim Bay State Park | bd | bd | 26.56 | bd | bd | 25.92 | bd | bd | 17.66 |
| Sequim Bay State Park | bd | bd | 44.46 | bd | bd | 20.60 | bd | bd | 25.42 |
| Sequim Bay Blyn | bd | bd | 20.98 | bd | bd | 18.70 | bd | bd | 14.23 |
| Sequim Bay Blyn | bd | bd | 20.21 | bd | bd | 18.53 | bd | bd | 14.52 |
2.3. Comparison of Screening Methods to LC-MS/MS
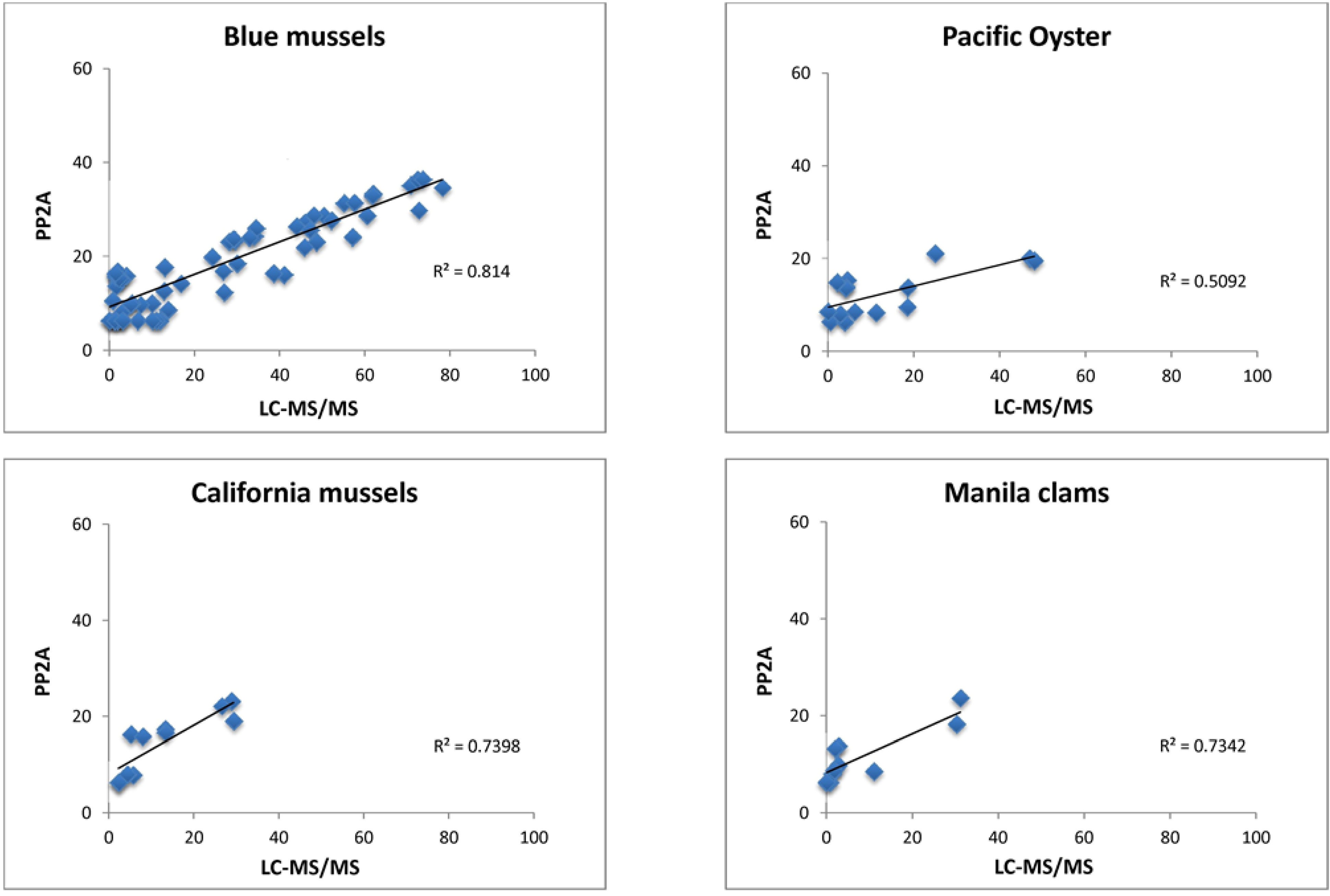
| EU Extracts | ||||
|---|---|---|---|---|
| LC-MS/MS | PP2A | ELISA | Jellett | LC-MS/MS |
| 96.5 | 48.9 | 51.8 | − | 39.8 |
| 63.6 | 38.2 | 52.3 | − | 31.7 |
| 60.6 | 38.1 | 58.8 | − | 32.4 |
| 38.7 | 35.7 | 35.7 | − | 18.3 |
| 30.3 | 28.7 | 22.6 | − | 17.1 |
| 36.9 | 36.0 | 63.0 | − | 21.1 |
| 26.2 | 31.2 | 31.8 | − | 12.9 |
| 32.7 | 33.5 | 33.8 | − | 15.1 |
| 26.5 | 34.1 | 28.8 | − | 13.9 |
| 37.7 | 35.1 | 51.3 | − | 16.9 |
| 60.5 | 29.4 | 49.3 | − | 26.0 |
| 28.1 | 33.6 | 25.3 | − | 12.7 |
| 31.5 | 34.5 | 20.5 | − | 18.4 |
| <LoQ | <LoQ | 10.9 | − | <LoQ |
| <LoQ | <LoQ | 20.5 | − | 2.0 |
| 36.1 | 16.9 | 28.8 | − | 16.2 |
| 21.9 | 12.8 | 20.3 | − | 13.1 |
| 25.3 | 16.5 | 67.8 | − | 14.5 |
| 6.3 | 14.7 | 27.2 | − | 3.0 |
| 3.7 | <LoQ | 23.6 | − | 2.2 |
| 7.9 | 13.0 | 31.7 | − | 2.9 |
| 37.7 | 13.1 | 61.3 | − | 14.4 |
| 14.2 | 26.6 | 32.7 | − | 6.5 |
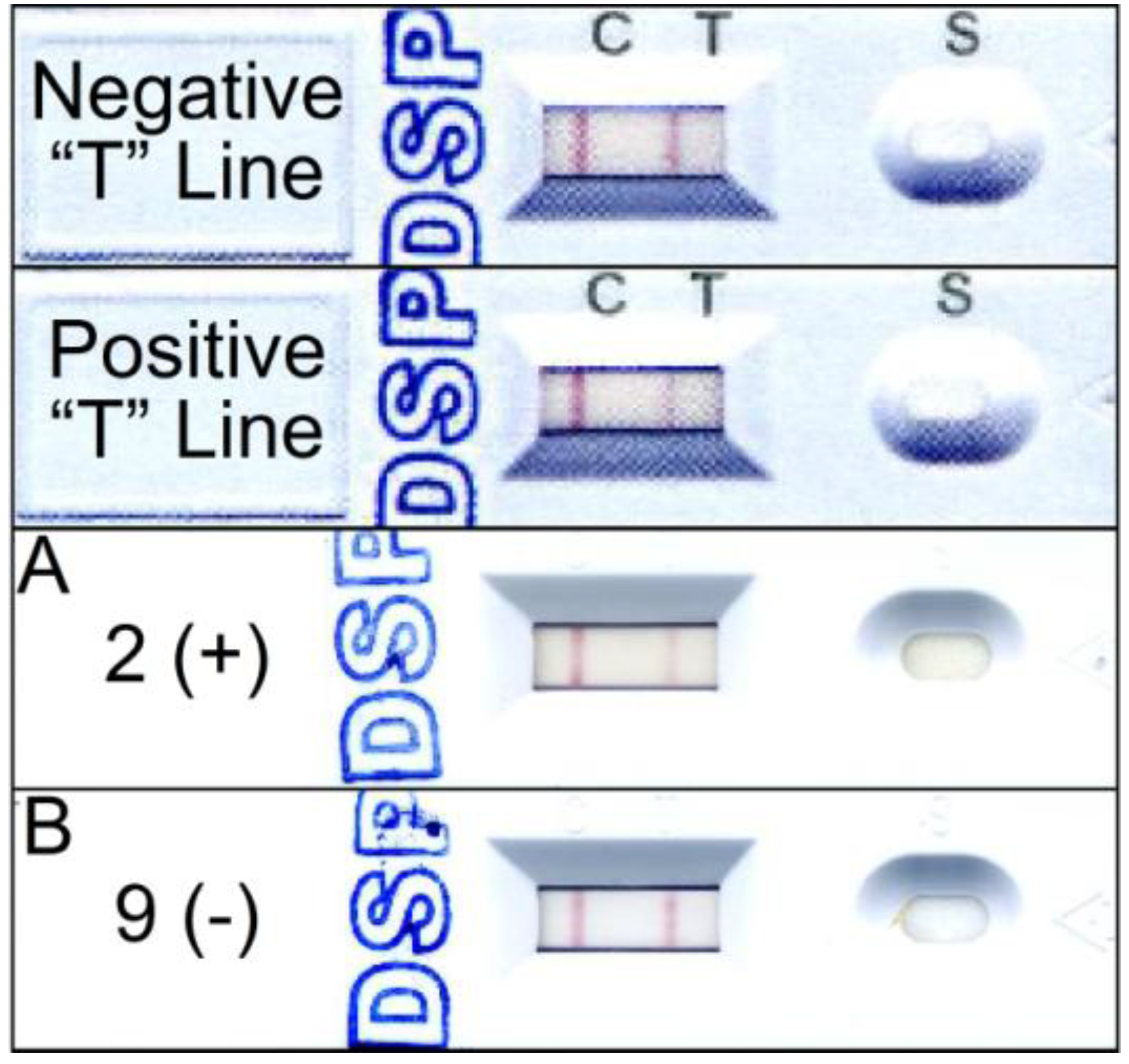
3. Discussion
4. Experimental Section
4.1. Shellfish Collection
4.2. Sample Preparation
4.3. Tissue Extraction
4.4. Tissue Extract Hydrolysis
4.5. LC-MS/MS
4.6. Enzyme-Linked Immunosorbent Assay (ELISA)
4.7. Protein Phosphatase 2A (PP2A)
4.8. Jellett Rapid Test Strip
5. Conclusions
Acknowledgments
Conflict of Interest
References
- Van Egmond, H.P.; Van Apeldoorn, M.E.; Speijers, G.J.A. Marine Biotoxins. In Food and Nutrition; FAO: Rome, Italy, 2004; pp. 53–96. [Google Scholar]
- Control of Communicable Diseases Manual, 19th ed.Heymann, D.L. (Ed.) American Public Health Association Press: Washington, DC, USA, 2008.
- Cembella, A.D. Occurrence of okadaic acid, a major diarrheic shellfish toxin, in natural populations of Dinophysis spp. from the eastern coast of North America. J. Appl. Phycol. 1989, 1, 307–310. [Google Scholar] [CrossRef]
- Takai, A.; Bialojan, C.; Troschka, M.; Ruegg, J.C. Smooth-muscle myosin phosphate inhibition and force enhancement by black sponge toxin. FEBS Lett. 1987, 217, 81–84. [Google Scholar] [CrossRef]
- Fujiki, H.; Suganuma, M. Tumor Promotion by Inhibitors of Protein Phosphatase-1 and Phosphatase-2A—The Okadaic Acid Class of Compounds. Adv. Cancer Res. 1993, 61, 143–194. [Google Scholar] [CrossRef]
- Bialojan, C.; Takai, A. Inhibitory Effect of a Marine-Sponge Toxin, Okadaic Acid, on Protein Phosphatases-Specificity and Kinetics. Biochem. J. 1988, 256, 283–290. [Google Scholar]
- Burgess, V.; Shaw, G. Pectenotoxins—an issue for public health—A review of their comparative toxicology and metabolism. Environ. Int. 2001, 27, 275–283. [Google Scholar] [CrossRef]
- Barceloux, D.G. Diarrhetic shellfish poisoning and okadaic acid. In Medical Toxicology of Natural Substances: Foods, Fungi, Medicinal Herbs, Plants, and Venomous Animals; John Wiley & Sons: Hoboken, NJ, USA, 2008; pp. 222–226. [Google Scholar]
- Stamman, E.; Segar, D.A.; Davis, P.G. A Preliminary Epidemiological Assessment of the Potential for Diarrhetic Shellfish Poisoning in the Northeast United States. In NOAA Technical Memorandum NOS OMA 34; NOAA: Rockville, MD, USA, 1987. [Google Scholar]
- Manerio, E.; Rodas, V.L.; Costas, E.; Hernandez, J.M. Shellfish consumption: A major risk factor for colorectal cancer. Med. Hypotheses 2008, 70, 409–412. [Google Scholar] [CrossRef]
- Cordier, S.; Monfort, C.; Miossec, L.; Richardson, S.; Belin, C. Ecological analysis of digestive cancer mortality related to contamination by diarrhetic shellfish poisoning toxins along the coasts of France. Environ. Res. 2000, 84, 145–150. [Google Scholar] [CrossRef]
- Lloyd, J.; Duchin, J.; Borchert, J.; Flores-Quintana, H.; Robertson, A. Diarrhetic shellfish poisoning in Washington State, USA, 2011. Emerg. Infect. Dis. 2013, in press. [Google Scholar]
- Taylor, M.; McIntyre, L.; Ritson, M.; Stone, J.; Bronson, R.; Bitzikos, O.; Rourke, W.; Galanis, E.; Team, O. Outbreak of Diarrhetic Shellfish Poisoning Associated with Mussels, British Columbia, Canada. Mar. Drugs 2013, 11, 1669–1676. [Google Scholar] [CrossRef]
- Horner, R.A.; Garrison, D.L.; Plumley, F.G. Harmful algal blooms and red tide problems on the U. S. west coast. Limnol. Oceanogr. 1997, 42, 1076–1088. [Google Scholar] [CrossRef]
- Trainer, V.; Moore, L.; Bill, B.; Adams, N.; Harrington, N.; Borchert, J.; da Silva, D.; Eberhart, B.-T. Diarrhetic Shellfish Toxins and Other Lipophilic Toxins of Human Health Concern in Washington State. Mar. Drugs 2013, 11, 1815–1835. [Google Scholar] [CrossRef]
- Commission Regulation (EU) No 15/2011 of 10 January 2011 amending Regulation (EC) No 2074/2005 as regards recognised testing methods for detecting marine biotoxins in live bivalve molluscs. Off. J. Eur. Union 2011, 54, 3–6.
- Anderson, D.M.; Andersen, P.; Bricelj, V.M.; Cullen, J.J.; Rensel, J.E.J. Monitoring and Management Strategies for Harmful Algal Blooms in Coastal Waters; Technical Report No. APEC #201-MR-01.1; Asia Pacific Economic Program, Singapore, and Intergovernmental Oceanographic Commission: Paris, France, 2001. [Google Scholar]
- Usagawa, T.; Nishimura, M.; Itoh, Y.; Uda, T.; Yasumoto, T. Preparation of monoclonal-antibodies against okadaic acid prepared from the sponge Halichondria okadai. Toxicon 1989, 27, 1323–1330. [Google Scholar] [CrossRef]
- Vale, P.; Sampayo, M.A.D. Comparison between HPLC and a commercial immunoassay kit for detection of okadaic acid and esters in Portuguese bivalves. Toxicon 1999, 37, 1565–1577. [Google Scholar] [CrossRef]
- Laycock, M.V.; Jellett, J.F.; Easy, D.J.; Donovan, M.A. First report of a new rapid assay for diarrhetic shellfish poisoning toxins. Harmful Algae 2006, 5, 74–78. [Google Scholar] [CrossRef]
- Tubaro, A.; Florio, C.; Luxich, E.; Sosa, S.; DellaLoggia, R.; Yasumoto, T. A protein phosphatase 2A inhibition assay for a fast and sensitive assessment of okadaic acid contamination in mussels. Toxicon 1996, 34, 743–752. [Google Scholar] [CrossRef]
- Honakanen, R.E.; Moody, D.E.; Dickey, R.W. Detection of DSP-toxins, okadaic acid, and dinophysis toxin-1 in shellfish by serine/threonine protein phosphatase assay. J. AOAC Int. 1996, 79, 1336–1343. [Google Scholar]
- Mountfort, D.O.; Suzuki, T.; Truman, P. Protein phosphatase inhibition assay adapted for determination of total DSP in contaminated mussels. Toxicon 2001, 39, 383–390. [Google Scholar] [CrossRef]
- Ramstad, H.; Shen, J.L.; Larsen, S.; Aune, T. The validity of two HPLC methods and a colorimetric PP2A assay related to the mouse bioassay in quantification of diarrhetic toxins in blue mussels (Mytilus edulis). Toxicon 2001, 39, 1387–1391. [Google Scholar] [CrossRef]
- Simon, J.F.; Vemoux, J.-P. Highly sensitive assay of okadaic acid using protein phosphatase and paranitrophenyl phosphate. Nat. Toxins 1994, 2, 293–301. [Google Scholar] [CrossRef]
- Vieytes, M.R.; Fontal, O.I.; Leira, F.; deSousa, J.; Botana, L.M. A fluorescent microplate assay for diarrheic shellfish toxins. Anal. Biochem. 1997, 248, 258–264. [Google Scholar] [CrossRef]
- Gonzalez, J.C.; Fontal, O.I.; Vieytes, M.R.; Vieytes, J.M.; Botana, L.M. Basis for a new procedure to eliminate diarrheic shellfish toxins from a contaminated matrix. J. Agric. Food Chem. 2002, 50, 400–405. [Google Scholar] [CrossRef]
- Prassopoulou, E.; Katikou, P.; Georgantelis, D.; Kyritsakis, A. Detection of okadaic acid and related esters in mussels during diarrhetic shellfish poisoning (DSP) episodes in Greece using the mouse bioassay, the PP2A inhibition assay and HPLC with fluorimetric detection. Toxicon 2009, 53, 214–227. [Google Scholar] [CrossRef]
- Smienk, H.G.F.; Calvo, D.; Razquin, P.; Dominguez, E.; Mata, L. Single Laboratory Validation of A Ready-to-Use Phosphatase Inhibition Assay for Detection of Okadaic Acid Toxins. Toxins 2012, 4, 339–352. [Google Scholar] [CrossRef]
- Sassolas, A.; Catanante, G.; Hayat, A.; Marty, J.L. Development of an efficient protein phosphatase-based colorimetric test for okadaic acid detection. Anal. Chim. Acta 2011, 702, 262–268. [Google Scholar] [CrossRef]
- Smienk, H.; Dominguez, E.; Rodriguez-Velasco, M.L.; Clarke, D.; Kapp, K.; Katikou, P.; Cabado, A.G.; Otero, A.; Vieites, J.M.; Razquin, P.; et al. Quantitative Determination of the Okadaic Acid Toxins Group by a Colorimetric Phosphatase Inhibition Assay: Interlaboratory Study. J. AOAC Int. 2013, 96, 77–85. [Google Scholar] [CrossRef]
- Garibo, D.; Damaso, E.; Eixarch, H.; de la Iglesia, P.; Fernandez-Tejedor, M.; Diogene, J.; Pazos, Y.; Campas, M. Protein phosphatase inhibition assays for okadaic acid detection in shellfish: Matrix effects, applicability and comparison with LC-MS/MS analysis. Harmful Algae 2012, 19, 68–75. [Google Scholar] [CrossRef]
- Annual Report 2011: Commercial and Recreational Shellfish Areas in Washington State; Office of Shellfish and Water Protection: Olympia, WA, USA, 2012.
- DeGrasse, S.; Conrad, S.; DiStefano, P.; Vanegas, C.; Wallace, D.; Jensen, P.; Hickey, J.M.; Cenci, F.; Pitt, J.; Deardorff, D.; et al. Onboard screening dockside testing as a new means of managing paralytic shellfish poisoning risks in federally closed waters. Deep Sea Res. Part II: Top. Stud. Oceanogr. 2013, in press. [Google Scholar]
- EU-Harmonized Standard Operation Procedure for Determination of Lipophilic Marine Biotoxins in Molluscs by LC-MS/MS, Version 4; European Union Reference Laboratory for Marine Biotoxins (EU-RL-MB), Agencia Esponola de Seguridad Alimentaria y Nutricion (AESAN): Vigo, Spain, 2011. Available online: http://www.aesan.msps.es/CRLMB/docs/docs/metodos_analiticos_de_desarrollo/EU-Harmonised-SOP-LIPO-LCMSMS_Version4.pdf (accessed on 10 May 2013).
- McCarron, P.; Giddings, S.D.; Quilliam, M.A. A mussel tissue certified reference material for multiple phycotoxins. Part 2: Liquid chromatography-mass spectrometry, sample extraction and quantitation procedures. Anal. Bioanal. Chem. 2011, 400, 835–846. [Google Scholar] [CrossRef]
- User’s Guide. Algal Toxins. Available online: http://www.abraxiskits.com/productByCat.php?catId=36 (accessed on 9 July 2011).
- Cohen, P. The Structure and Regulation of Protein Phosphatases. Annu. Rev. Biochem. 1989, 58, 453–508. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Eberhart, B.-T.L.; Moore, L.K.; Harrington, N.; Adams, N.G.; Borchert, J.; Trainer, V.L. Screening Tests for the Rapid Detection of Diarrhetic Shellfish Toxins in Washington State. Mar. Drugs 2013, 11, 3718-3734. https://doi.org/10.3390/md11103718
Eberhart B-TL, Moore LK, Harrington N, Adams NG, Borchert J, Trainer VL. Screening Tests for the Rapid Detection of Diarrhetic Shellfish Toxins in Washington State. Marine Drugs. 2013; 11(10):3718-3734. https://doi.org/10.3390/md11103718
Chicago/Turabian StyleEberhart, Bich-Thuy L., Leslie K. Moore, Neil Harrington, Nicolaus G. Adams, Jerry Borchert, and Vera L. Trainer. 2013. "Screening Tests for the Rapid Detection of Diarrhetic Shellfish Toxins in Washington State" Marine Drugs 11, no. 10: 3718-3734. https://doi.org/10.3390/md11103718
APA StyleEberhart, B.-T. L., Moore, L. K., Harrington, N., Adams, N. G., Borchert, J., & Trainer, V. L. (2013). Screening Tests for the Rapid Detection of Diarrhetic Shellfish Toxins in Washington State. Marine Drugs, 11(10), 3718-3734. https://doi.org/10.3390/md11103718





