Diversity of Nonribosomal Peptide Synthetase Genes in the Microbial Metagenomes of Marine Sponges
Abstract
:1. Introduction
2. Results and Discussion
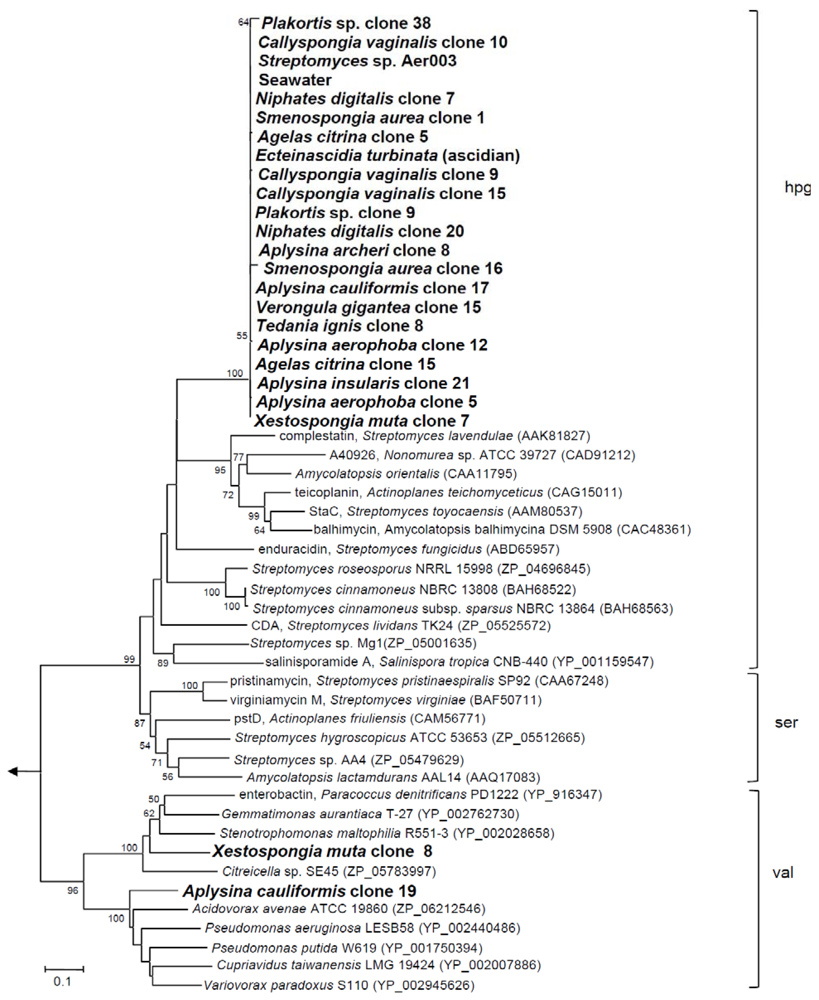
2.1. NRPS Gene Diversity
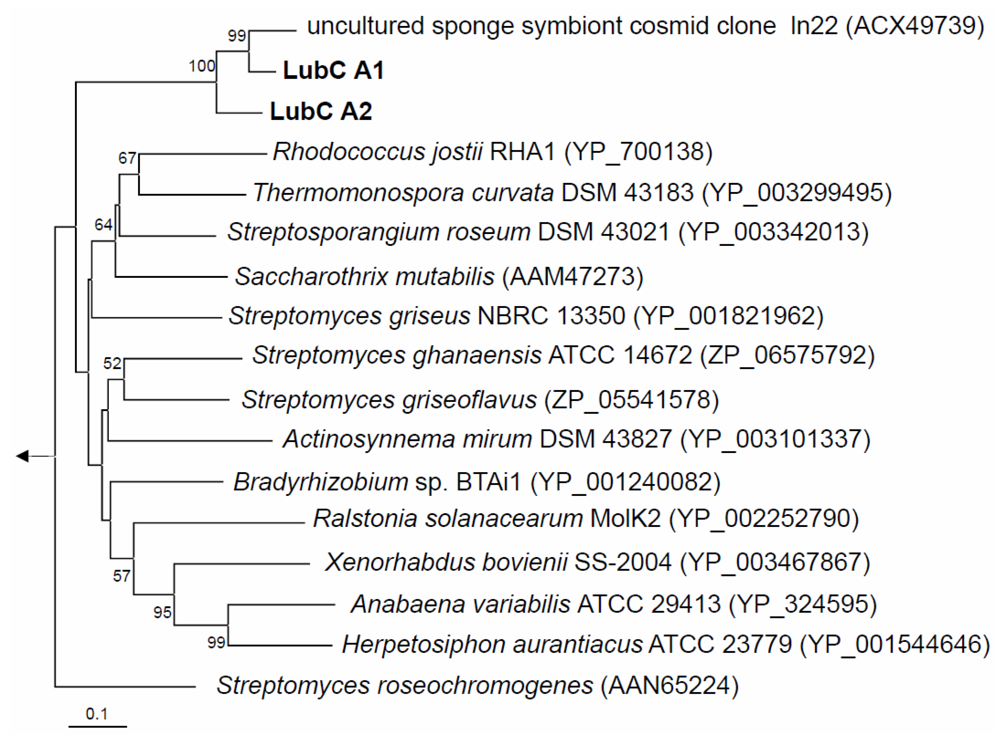
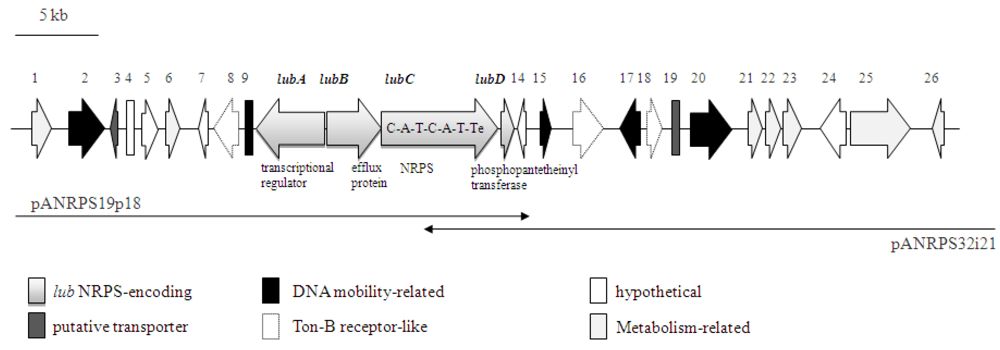
2.2. NRPS-Containing Metagenomic Cosmid Clone
| CDs | Position (nd) | Putative Function | Most Similar Homolog, (Acc#), origin | Identity/Similarity (%) | No. of Amino Acids | ||||
|---|---|---|---|---|---|---|---|---|---|
| ORF1 | 3626-4807 | Serine-threonine phosphatase | PrpA (NP_487771), Nostoc sp. PCC 7120 | 53/69 | 399 | ||||
| ORF2 | 5909-8170 | Helicase RecD/TraA | BAL199_00820, (ZP_02192076), Alpha proteobacterium BAL199 | 72/82 | 753 | ||||
| ORF3 | 8441-8953 | ABC transporter | Cagg_3718, (YP_002464990), Chloroflexus aggregans DSM 9485 | 42/51 | 170 | ||||
| ORF4 | 9894-10364 | Hypothetical protein | MC7420_1635, (ZP_05030609), Microcoleus chthonoplastes PCC 7420 | 40/51 | 156 | ||||
| ORF5 | 10853-11857 | Hypothetical protein | AM1_4437, (YP_001518731), Acaryochloris marina MBIC11017 | 34/52 | 334 | ||||
| ORF6 | 11903-12802 | Hydrolase | VEx25_1601, (ZP_04922735), Vibrio sp. Ex25 | 29/41 | 299 | ||||
| ORF7 | 13907-14512 | Hypothetical protein | BACCOPRO_03255, (ZP_03644864), Bacteroides coprophilus DSM 18228 | 26/40 | 201 | ||||
| ORF8 | 14867-16432 | TonB-dependent receptor | MXAN_6044, (YP_634179), Myxococcus xanthus DK 1622 | 35/54 | 521 | ||||
| ORF9 | 16943-17452 | Exonuclease | RLO149_22990, (ZP_02142576), Roseobacter litoralis Och 149 | 39/51 | 169 | ||||
| lubA | 17745-22022 | LuxR transcriptional regulator | HNE_2502, (YP_761196), Hyphomonas neptunium ATCC 15444 | 26/44 | 1429 | ||||
| lubB | 21850-25236 | Resistance protein | Sputw3181_3288, (YP_964656), Shewanella sp. W3-18-1 | 43/63 | 1128 | ||||
| lubC | 25260-32465 | NRPS (C-A-T-C-A-T-Te) | Siderophore, (ACX49739), uncultured marine bacterium 1n22 | 81/87 | 2401 | ||||
| lubD | 32655-33497 | Phosphopantetheinyl transferase | Mnod_1716, (YP_002497009), Methylobacterium nodulans ORS 2060 | 36/46 | 280 | ||||
| ORF14 | 33681-34217 | Hypothetical protein | MldDRAFT_3697, (ZP_01290808), Delta proteobacterium MLMS-1 | 63/77 | 178 | ||||
| ORF15 | 35067-35822 | Transposase | EbA6749, (YP_160886), Aromatoleum aromaticum EbN1 | 58/74 | 251 | ||||
| ORF16 | 37148-38974 | TonB-dependent receptor | Sama_2896, (YP_928768), Shewanella amazonensis SB2B | 65/82 | 608 | ||||
| ORF17 | 40041-41315 | Transposase | BAL199_06759, (ZP_02191799), Alpha proteobacterium BAL199 | 45/56 | 424 | ||||
| ORF18 | 41701-42630 | TonB-dependent receptor | GPB2148_3348, (ZP_05093557), marine Gamma proteobacterium HTCC2148 | 44/59 | 309 | ||||
| ORF19 | 43924-44364 | Membrane transport protein | Ykris0001_15620, (ZP_04625017), Yersinia kristensenii ATCC 33638 | 43/56 | 146 | ||||
| ORF20 | 44366-46975 | DNA invertase | NB231_12409, (ZP_01126794), Nitrococcus mobilis Nb-231 | 66/78 | 869 | ||||
| ORF21 | 47982-48890 | Methyltransferase | MaviaA2_010100001311, (ZP_05214826), Mycobacterium avium ATCC 25291 | 40/49 | 302 | ||||
| ORF22 | 49061-50023 | NADH-quinone oxidoreductase | Psta_3148, (YP_003371672), Pirellula staleyi DSM 6068 | 25/40 | 320 | ||||
| ORF23 | 50133-51254 | 2,3-Dihydroxybenzoic acid decarboxylase | PJE062_2683, (ZP_05084178), Pseudovibrio sp. JE062 | 65/75 | 373 | ||||
| ORF24 | 52396-54060 | Hypothetical protein | ZP_05710821, (ZP_05710821), Desulfurivibrio alkaliphilus AHT2 | 49/68 | 554 | ||||
| ORF25 | 54272-58006 | Cyclopropane-fatty-acyl-phospholipid synthase | ADG881_908, (ZP_05041385), Alcanivorax sp. DG881 | 51/66 | 1244 | ||||
| ORF26 | 59373-60101 | Nucleoside 2-deoxyribosyltransferase | P9211_14861, (YP_001551371), Prochlorococcus marinus str. MIT 9211 | 63/76 | 242 | ||||
3. Experimental Section
3.1. Sponge Collection
3.2. Cultivation and Identification of Sponge-Associated Bacteria
3.3. DNA Extraction, PCR Amplification and Sequencing of A Domains of NRPS Genes
3.4. Metagenomic Library Construction and Screening for NRPS-Encoding Clones
3.5. Sequence Analysis
4. Conclusions
Acknowledgements
References
- Blunt, J.W.; Copp, B.R.; Munro, M.H.; Northcote, P.T.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2010, 27, 165–237. [Google Scholar] [CrossRef]
- Taylor, M.W.; Radax, R.; Steger, D.; Wagner, M. Sponge-associated microorganisms: Evolution, ecology, and biotechnological potential. Microbiol. Mol. Biol. Rev. 2007, 71, 295–347. [Google Scholar] [CrossRef]
- Piel, J. Metabolites from symbiotic bacteria. Nat. Prod. Rep. 2009, 26, 338–362. [Google Scholar] [CrossRef]
- Piel, J. Approaches to capturing and designing biologically active small molecules produced by uncultured microbes. Ann. Rev. Microbiol. 2011, 65, 431–453. [Google Scholar] [CrossRef]
- Mootz, H.D.; Schwarzer, D.; Marahiel, M.A. Ways of assembling complex natural products on modular nonribosomal peptide synthetases. ChemBioChem 2001, 3, 490–504. [Google Scholar]
- Schwarzer, D.; Finking, R.; Marahiel, M.A. Nonribosomal peptides: From genes to products. Nat. Prod. Rep. 2003, 20, 275–287. [Google Scholar] [CrossRef]
- Cane, D.E.; Walsh, C.T.; Khosla, C. Harnessing the biosynthetic code: Combinations, permutations, and mutations. Science 1998, 282, 63–68. [Google Scholar] [CrossRef]
- Mansson, M.; Gram, L.; Larsen, T.O. Production of bioactive secondary metabolites by marine vibrionaceae. Mar. Drugs 2011, 9, 1440–1468. [Google Scholar] [CrossRef]
- Crosa, J.H.; Walsh, C.T. Genetics and assembly line enzymology of siderophore biosynthesis in bacteria. Microbiol. Mol. Biol. Rev. 2002, 66, 223–249. [Google Scholar] [CrossRef]
- Cane, D.E.; Walsh, C.T. The parallel and convergent universes of polyketide synthases and nonribosomal peptide synthetases. Chem. Biol. 1999, 6, 319–325. [Google Scholar] [CrossRef]
- Janso, J.E.; Carter, G.T. Biosynthetic potential of phylogenetically unique endophytic actinomycetes from tropical plants. Appl. Environ. Microbiol. 2010, 76, 4377–4386. [Google Scholar] [CrossRef]
- Johnson, R.; Voisey, C.; Johnson, L.; Pratt, J.; Fleetwood, D.; Khan, A.; Bryan, G. Distribution of NRPS gene families within the Neotyphodium/Epichloe complex. Fungal Genet. Biol. 2007, 44, 1180–1190. [Google Scholar] [CrossRef]
- Schneemann, I.; Nagel, K.; Kajahn, I.; Labes, A.; Wiese, J.; Imhoff, J.F. Comprehensive investigation of marine Actinobacteria associated with the sponge Halichondria panicea. Appl. Environ. Microbiol. 2010, 76, 3702–3714. [Google Scholar] [CrossRef]
- Jiang, S.; Sun, W.; Chen, M.; Dai, S.; Zhang, L.; Liu, Y.; Lee, K.J.; Li, X. Diversity of culturable actinobacteria isolated from marine sponge Haliclona sp. Antonie Van Leeuwenhoek 2007, 92, 405–416. [Google Scholar] [CrossRef]
- Zhang, W.; Li, Z.; Miao, X.; Zhang, F. The screening of antimicrobial bacteria with diverse novel nonribosomal peptide synthetase (NRPS) genes from South China Sea sponges. Mar. Biotechnol. 2009, 11, 346–355. [Google Scholar] [CrossRef]
- Zhou, K.; Zhang, X.; Zhang, F.; Li, Z. Phylogenetically diverse cultivable fungal community and polyketide synthase (PKS), non-ribosomal peptide synthase (NRPS) genes associated with the South China Sea sponges. Microb. Ecol. 2011, 62, 644–654. [Google Scholar] [CrossRef]
- Ehrenreich, I.M.; Waterbury, J.B.; Webb, E.A. Distribution and diversity of natural product genes in marine and freshwater cyanobacterial cultures and genomes. Appl. Environ. Microbiol. 2005, 71, 7401–7413. [Google Scholar] [CrossRef]
- Gontang, E.A.; Gaudêncio, S.P.; Fenical, W.; Jensen, P.R. Sequence-based analysis of secondary-metabolite biosynthesis in marine actinobacteria. Appl. Environ. Microbiol. 2010, 76, 2487–2499. [Google Scholar] [CrossRef]
- Hodges, T.W.; Slattery, M.; Olson, J.B. Unique actinomycetes from marine caves and coral reef sediments provide novel PKS and NRPS biosynthetic gene clusters. Mar. Biotechnol. 2012, 14, 270–280. [Google Scholar] [CrossRef]
- Schirmer, A.; Gadkari, R.; Reeves, C.D.; Ibrahim, F.; DeLong, E.F.; Hutchinson, C.R. Metagenomic analysis reveals diverse polyketide synthase gene clusters in microorganisms associated with the marine sponge Discodermia dissoluta. Appl. Environ. Microbiol. 2005, 71, 4840–4849. [Google Scholar]
- Siegl, A.M.; Hentschel, U. PKS and NRPS gene clusters from microbial symbiont cells of marine sponges by whole genome amplification. Environ. Microbiol. Rep. 2010, 2, 507–513. [Google Scholar]
- Fieseler, L.; Hentschel, U.; Grozdanov, L.; Schirmer, A.; Wen, G.; Platzer, M.; Hrvatin, S.; Butzke, D.; Zimmermann, K.; Piel, J. Widespread occurrence and genomic context of unusually small polyketide synthase genes in microbial consortia associated with marine sponges. Appl. Environ. Microbiol. 2007, 73, 2144–2155. [Google Scholar]
- Hochmuth, T.; Niederkrüger, H.; Gernert, C.; Siegl, A.; Taudien, S.; Platzer, M.; Crews, P.; Hentschel, U.; Piel, J. Linking chemical and microbial diversity in marine sponges: Possible role for Poribacteria as producers of methyl-branched fatty acids. ChemBioChem. 2010, 11, 2572–2578. [Google Scholar] [CrossRef]
- Bayer, K.; Scheuermayer, M.; Fieseler, L.; Hentschel, U. Genomic mining for novel FADH2-dependent halogenases in marine sponge-associated microbial consortia. Mar. Biotechnol. 2012. [Google Scholar]
- Ayuso-Sacido, A.; Genilloud, O. New PCR primers for the screening of NRPS and PKS-I systems in actinomycetes: Detection and distribution of these biosynthetic gene sequences in major taxonomic groups. Microb. Ecol. 2005, 49, 10–24. [Google Scholar] [CrossRef]
- Ansari, M.Z.; Yadav, G.; Gokhale, R.S.; Mohanty, D. NRPS-PKS: A knowledge-based resource for analysis of NRPS/PKS megasynthases. Nucleic Acids Res. 2004, 1, 405–413. [Google Scholar]
- Tamura, K.; Dudley, J.; Nei, M.; Kumar, S. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol. Biol. Evol. 2007, 24, 1596–1599. [Google Scholar] [CrossRef]
- Röttig, M.; Medema, M.H.; Blin, K.; Weber, T.; Rausch, C.; Kohlbacher, O. NRPSpredictor2-a web server for predicting NRPS adenylation domain specificity. Nucleic Acids Res. 2011, 39, 362–367. [Google Scholar]
- Hopkinson, B.M.; Barbeau, K.A. Iron transporters in marine prokaryotic genomes and metagenomes. Environ. Microbiol. 2012, 14, 114–128. [Google Scholar] [CrossRef]
- Mincer, T.J.; Jensen, P.R.; Kauffman, C.A.; Fenical, W. Widespread and persistent populations of a major new marine actinomycete taxon in ocean sediments. Appl. Environ. Microbiol. 2002, 68, 5005–5011. [Google Scholar]
- Hentschel, U.; Schmid, M.; Wagner, M.; Fieseler, L.; Gernert, C.; Hacker, J. Isolation and phylogenetic analysis of bacteria with antimicrobial activities from the Mediterranean sponges Aplysina aerophoba and Aplysina cavernicola. FEMS Microbiol. Ecol. 2001, 35, 305–312. [Google Scholar] [CrossRef]
- Li, A.; Piel, J. A gene cluster from a marine Streptomyces encoding the biosynthesis of the aromatic spiroketal polyketide griseorhodin A. Chem. Biol. 2002, 9, 1017–1026. [Google Scholar] [CrossRef]
- Piel, J.; Hui, D.; Wen, G.; Butzke, D.; Platzer, M.; Fusetani, N.; Matsunaga, S. Antitumor polyketide biosynthesis by an uncultivated bacterial symbiont of the marine sponge Theonella swinhoei. Proc. Natl. Acad. Sci. USA 2004, 101, 16222–16227. [Google Scholar]
- Altschul, S.F.; Madden, T.L.; Schaffer, A.A.; Zhang, J.; Zhang, Z.; Miller, W.; Lipman, D.J. Gapped BLAST and PSI-BLAST: A new generation of protein database search programs. Nucleic Acids. Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef]
- Samples Availability: Available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pimentel-Elardo, S.M.; Grozdanov, L.; Proksch, S.; Hentschel, U. Diversity of Nonribosomal Peptide Synthetase Genes in the Microbial Metagenomes of Marine Sponges. Mar. Drugs 2012, 10, 1192-1202. https://doi.org/10.3390/md10061192
Pimentel-Elardo SM, Grozdanov L, Proksch S, Hentschel U. Diversity of Nonribosomal Peptide Synthetase Genes in the Microbial Metagenomes of Marine Sponges. Marine Drugs. 2012; 10(6):1192-1202. https://doi.org/10.3390/md10061192
Chicago/Turabian StylePimentel-Elardo, Sheila Marie, Lubomir Grozdanov, Sebastian Proksch, and Ute Hentschel. 2012. "Diversity of Nonribosomal Peptide Synthetase Genes in the Microbial Metagenomes of Marine Sponges" Marine Drugs 10, no. 6: 1192-1202. https://doi.org/10.3390/md10061192
APA StylePimentel-Elardo, S. M., Grozdanov, L., Proksch, S., & Hentschel, U. (2012). Diversity of Nonribosomal Peptide Synthetase Genes in the Microbial Metagenomes of Marine Sponges. Marine Drugs, 10(6), 1192-1202. https://doi.org/10.3390/md10061192




