2.1. Antiviral Activity of N. aculeata Extracts and Fractions
The methanol (MeOH) extract of
N. aculeata was suspended in distilled water and partitioned successively with hexane, ethyl acetate (EtOAc), and
n-butanol. Each organic fraction was subjected to cytotoxicity and antiviral activity assays (
Table 1). Among these, the EtOAc-soluble fraction (ESF) exhibited inhibitory effect against HRV2 and HRV3, with IC
50 values of 15.50 ± 4.17 μg/mL and 16.50 ± 4.17 μg/mL, respectively. The water soluble fraction (WSF) showed antiviral activity against HRV2, but not against HRV3. The hexane-soluble (HSF) and butanol-soluble (BSF) fractions showed no antiviral activity against either HRV2 or HRV3 (
Table 1).
Table 1.
Antiviral activity of Neorhodomela aculeata extracts against HRV2 and HRV3 in HeLa cells.
Table 1.
Antiviral activity of Neorhodomela aculeata extracts against HRV2 and HRV3 in HeLa cells.
| Test material | | HRV2 (Type B) | HRV3 (Type A) |
| CC50a | IC50b | TI c | IC50b | TI c |
| Methanolic extract | >20 | 17.58 ± 0.59 | 1.14 | 18.27 ± 2.22 | 1.09 |
| HSF | >20 | ND d | - | ND d | - |
| ESF | >20 | 15.50 ± 4.17 | 1.29 | 16.50 ± 4.17 | 1.21 |
| BSF | >20 | ND d | - | ND d | - |
| WSF | >20 | 16.50 ± 2.76 | 1.21 | ND d | - |
The most active fraction, ESF, was fractionated by silica gel column chromatography. Seven fractions (F1–F7) were analyzed by reversed-phase HPLC, monitoring at 280 nm (
Figure 1), and tested on HeLa cells for antiviral activity (
Table 2).
Figure 1.
RP-HPLC profile of sub-fractions of EtOAc-soluble fraction (ESF) of N. aculeata. Performed on an Agilent 1300 HPLC system fitted with a Phenomenex Luna C18 (2) column (150 × 4.6 mm, 5 μm). The elution solvent system was binary gradient of solvent A (0.02% trifluoroacetic acid (TFA) in water); solvent B (0.02% TFA in acetonitrile). The gradient flow program was, as follows: 0 min, 10% B; 30 min. The flow rate was 0.7 mL/min and detection wavelength was set at 280 nm and column temperature was 25 °C. The chromatogram of F1 was not shown.
Figure 1.
RP-HPLC profile of sub-fractions of EtOAc-soluble fraction (ESF) of N. aculeata. Performed on an Agilent 1300 HPLC system fitted with a Phenomenex Luna C18 (2) column (150 × 4.6 mm, 5 μm). The elution solvent system was binary gradient of solvent A (0.02% trifluoroacetic acid (TFA) in water); solvent B (0.02% TFA in acetonitrile). The gradient flow program was, as follows: 0 min, 10% B; 30 min. The flow rate was 0.7 mL/min and detection wavelength was set at 280 nm and column temperature was 25 °C. The chromatogram of F1 was not shown.
Fraction 4 (F4) was investigated for its antiviral activity and was demonstrated to have an IC
50 value of 18.55 ± 0.51 μg/mL against HRV2 and 18.52 ± 0.49 μg/mL against HRV3 (
Table 2). Fractions F1, F2, and F7 showed antiviral activity against only HRV2, with IC
50 values of 11.38 ± 3.01 μg/mL, 8.36 ± 2.42 μg/mL, and 10.69 ± 1.34 μg/mL, respectively. F3 only showed antiviral activity against HRV3, with a IC
50 value of 7.69 ± 0.45 μg/mL (
Table 2).
Table 2.
Antiviral activity of sub-fractions of ESF against HRV2 and HRV3 in HeLa cells.
Table 2.
Antiviral activity of sub-fractions of ESF against HRV2 and HRV3 in HeLa cells.
| Test material | HRV2 | HRV3 |
| CC50a | IC50b | TI c | CC50a | IC50b | TI c |
| F1 | >20 | 11.38 ± 3.01 | 1.76 | >20 | ND d | - |
| F2 | >20 | 8.36 ± 2.42 | 2.39 | >20 | ND d | - |
| F3 | 22.93 | ND d | - | 25.80 | 7.69 ± 0.45 | 2.60 |
| F4 | >20 | 18.55 ± 0.51 | 1.08 | >20 | 18.52 ± 0.49 | 1.08 |
| F5 | 24.29 | ND d | - | 18.98 | ND d | - |
| F6 | >20 | ND d | - | >20 | ND d | - |
| F7 | >20 | 10.69 ± 1.34 | 1.87 | >20 | ND d | - |
| Ribavirin | >20 | 17.14 ± 1.48 | 1.17 | >20 | 14.25 ± 2.20 | 1.40 |
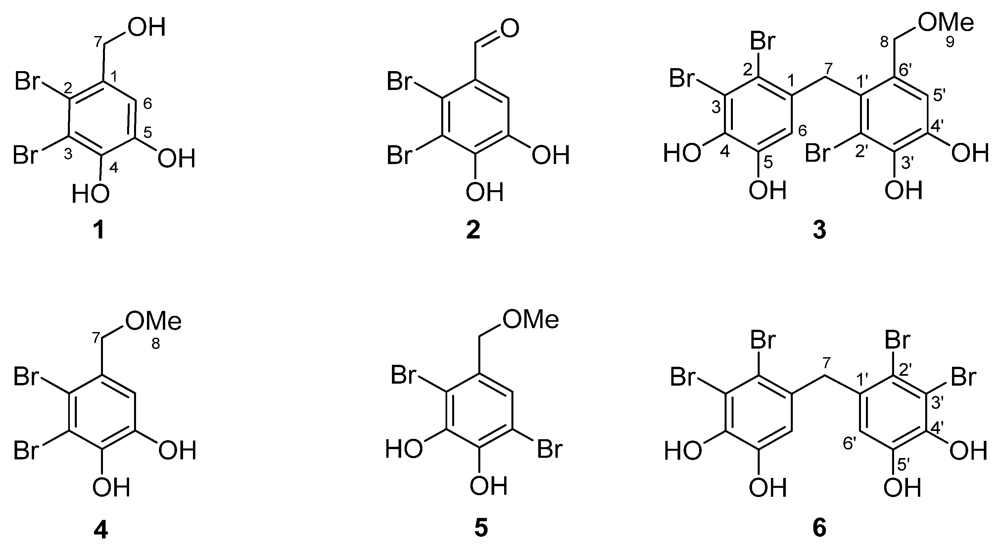
Fraction F4 was purified by preparative HPLC and semi-preparative HPLC at 280 nm, affording six pure compounds (
1–
6), as shown in
Figure 2. Their structures were elucidated by spectroscopic methods, including 1D and 2D NMR techniques and MS.
Figure 2.
Structures of compounds 1–6.
Figure 2.
Structures of compounds 1–6.
Compound
1 was a yellowish-brown needle-shaped crystal, with a molecular formula of C
7H
579Br
81BrO
3, as determined from
13C data (
Table 3) and ESIMS
m/z 296.9 [M − H]
−. The
1H NMR spectrum of compound
1 in acetone-
d6 showed the presence of a single aromatic proton at δ
H 7.20 (1H, s, H-6) and a methylene group at δ
H 4.62 (2H, s, H-7) on a catechol. The
13C signals at δ
C 144.8, 143.1, 134.1, 113.7, 112.6, 112.4, and 64.2 corresponded to carbons C-5, C-4, C-1, C-6, C-2, C-3, and C-7, respectively. Accordingly, the structure of
1 was determined to be 2,3-dibromo-4,5-dihydroxybenzyl alcohol (lanosol) (
Table 3) [
18].
Compound
2 was a colorless solid with a molecular formula of C
7H
579Br
81BrO
3, as determined by ESIMS
m/z 296.9 [M + H]
+. The
1H NMR spectrum of compound
2 in acetone-
d6 exhibited an aldehyde proton signal at δ
H 10.22 (1H, s, H-7) and an aromatic proton signal at δ
H 7.41 (1H, s, H-6). Six aromatic carbon signals were observed at δ
C 157.0 (C-4), 150.3 (C-5), 144.4 (C-4), 127.4 (C-3), 120.7 (C-2), and 113.1 (C-6), and an aldehyde carbon signal was seen at δ
C 190.4 (C-7). Therefore, the structure of
2 was determined to be 2,3-dibromo-4,5-dihydroxybenzaldehyde (
Table 3) [
19].
Compound
3 was also obtained as a colorless solid having a molecular formula of C
15H
1379Br
281BrO
5Na, as determined by ESIMS
m/z 534.8 [M + Na]
+. In addition to a singlet exchangeable signal that integrated for four protons at δ
H 8.73 (s, 1H, OH-4), 8.71 (s, 1H, OH-3′), 8.17 (s, 1H, OH-4′), and 8.08 (s, 1H, OH-5); the
1H NMR spectrum of the compound in acetone-
d6 demonstrated two singlets attributable to aromatic protons at δ
H 7.00 (s, 1H, H-5′) and 6.08 (s, 1H, H-6), two methylene groups at δ
H 4.21 (s, 2H, H-8) and 4.12 (s, 2H, H-7) and a methoxy group at δ
H 3.24 (s, 3H, H-9). The
13C NMR spectra of compound
3 showed 15 carbons, assignable to two benzene rings at δ
C 115.4 (C-5′), and 114.0 (C-6); two methylenes at δ
C 72.3 (C-8) and 38.4 (C-7); and a methoxy at δ
C 57.3 (C-9) with four oxygenated carbons at δ
C 144.5 (C-4′), 144.0 (C-5), and 142.6 (C-4, C-3′). The above spectral data indicated that compound
3 possessed a tribrominated diphenylmethane structure with four hydroxyl groups and one methoxy group. Thus, the structure of
3 was determined to be 2,2′,3-tribromo-3′,4,4′,5-tetrahydroxy-6′-methoxymethyldiphenylmethane (
Table 3) [
20].
Compound
4 was also isolated as a colorless solid, but with a molecular formula of C
8H
779Br
81BrO
3, as determined by ESIMS
m/z 310.9 [M − H]. The
1H NMR spectrum of compound
4 in acetone-
d6 showed one singlet aromatic proton at δ
H 7.07 (s, 1H, H-6); a methylene group at δ
H 4.41 (s, 2H, H-7) of a catechol; and an attached methoxy at δ
H 3.39 (s, 3H, H-8). It was also determined to have five aromatic carbon signals at δ
C 144.9 (C-5), 143.7 (C-4), 131.6 (C-1), 114.8 (C-2, 6), and 114.4 (C-3); one methylene carbon signal at δ
C 75.0 (C-7); and one methoxy carbon signal at δ
C 58.4 (C-8). Therefore, the structure of
4 was determined to be 2,3-dibromo-4,5-dihydroxybenzyl methyl ether (
Table 3) [
18].
Compound
5 was another colorless solid, with an ESIMS
m/z 310.9 [M − H]-determined molecular formula of C
8H
779Br
81BrO
3. The
1H NMR spectrum of the compound in acetone-
d6 revealed the presence of one aromatic hydrogen at δ
H 6.71 (s, 1H, H-6); a methylene group at δ
H 4.43 (s, 2H, H-7) of a catechol; and an attached methoxy at δ
H 3.34 (s, 3H, H-8). The compound also showed six aromatic carbon signals at δ
C 152.2 (C-4), 144.9 (C-3), 129.1 (C-1), 112.0 (C-2), 111.9 (C-6), 109.3 (C-5); one methylene carbon signal at δ
C 75.2 (C-7); and one methoxy carbon signal at δ
C 57.9 (C-8). Thus, the structure of
5 was established as 2,5-dibromo-3,4-dihydroxy-benzyl methyl ether (
Table 3) [
18].
Compound
6 was purified as a white amorphous solid with a molecular formula of C
13H
779Br
281Br
2O
4, determined using ESIMS
m/z 546.7 [M − H]. Its
1H NMR spectrum revealed one aromatic hydrogen at δ
H 6.58 (s, 2H, H-6, 6′) and a methylene group at δ
H 4.04 (s, 2H, H-7). The
13C NMR spectra showed seven carbons assignable to one benzene ring at δ
C 145.4 (C-5, 5′), 143.8 (C-4, 4′), 132.0 (C-1, 1′), 116.5 (C-6, 6′), 116.4 (C-2, 2′), and 113.6 (C-3, 3′), and to one methylene group at δ
C 44.5 (C-7). Consequently, the assigned structure was 2,2′,3,3′-tetrabromo-4,4′,5,5′-tetrahydroxydiphenyl methane (
Table 3) [
20].
Table 3.
The 1H and 13C NMR spectroscopy data of compounds 1–6a.
Table 3.
The 1H and 13C NMR spectroscopy data of compounds 1–6a.
| No. | | 1 | | | 2 | | | 3 | | | 4 | | | 5 | | | 6 | |
|---|
| | δH | δC | | δH | δC | | δH | δC | | δH | δC | | δH | δC | | δH | δC |
|---|
| 1 | | | 134.1 | | | 144.4 | | | 131.5 | | | 131.6 | | | 129.1 | | | 132.0 |
| 2 | | | 112.6 | | | 127.4 | | | 115.4 | | | 114.8 | | | 112.0 | | | 116.4 |
| 3 | | | 112.4 | | | 120.7 | | | 112.8 | | | 114.4 | | | 144.9 | | | 113.6 |
| 4 | | | 143.1 | | | 157.0 | | 8.73 (s, 1H, OH) | 142.6 | | | 143.7 | | | 152.2 | | | 143.8 |
| 5 | | | 144.8 | | | 150.3 | | 8.08 (s, 1H, OH) | 144.0 | | | 144.9 | | | 109.3 | | | 145.4 |
| 6 | | 7.20 (s, 1H) | 113.7 | | 7.41 (s, 1H) | 113.1 | | 6.08 (s, 1H) | 114.0 | | 7.07 (s, 1H) | 114.8 | | 6.71 (s, 1H) | 111.9 | | 6.58 (s, 1H) | 116.5 |
| 7 | | 4.62 (s, 2H) | 64.2 | | 10.22 (s, 1H) | 190.4 | | 4.12 (s, 2H) | 38.4 | | 4.41 (s, 2H) | 75.0 | | 4.43 (s, 2H) | 75.2 | | 4.04 (s, 2H) | 44.5 |
| 8 | | | | | | | | 4.21 (s, 2H) | 72.3 | | 3.39 (s, 3H) | 58.4 | | 3.34 (s, 3H) | 57.9 | | | |
| 9 | | | | | | | | 3.24 (s, 3H) | 57.9 | | | | | | | | | |
| 1′ | | | | | | | | | 128.6 | | | | | | | | | 132.0 |
| 2′ | | | | | | | | | 114.0 | | | | | | | | | 116.4 |
| 3′ | | | | | | | | 8.71 (s, 1H, OH) | 142.6 | | | | | | | | | 113.6 |
| 4′ | | | | | | | | 8.17 (s, 1H, OH) | 144.5 | | | | | | | | | 143.8 |
| 5′ | | | | | | | | 7.00 (s, 1H) | 115.4 | | | | | | | | | 145.4 |
| 6′ | | | | | | | | | 129.8 | | | | | | | | 6.58 (s, 1H) | 116.5 |
Table 4.
Antiviral activity of compound 1 and compound 3 isolated from N. aculeata against HRV 2 and HRV3.
Table 4.
Antiviral activity of compound 1 and compound 3 isolated from N. aculeata against HRV 2 and HRV3.
| Test material | | HRV2 | HRV3 |
| CC50a | IC50b | TI c | IC50b | TI c |
| Compound 1 | >20 | 2.50 ± 0.66 | 8.00 | ND d | - |
| Compound 3 | >20 | 7.11 ± 0.64 | 2.81 | 4.69 ± 0.44 | 4.26 |
| Ribavirin | >20 | 2.15 ± 0.51 | 9.30 | 5.09 ± 0.60 | 3.93 |
2.2. Antiviral Activity and Cytotoxicity of Compound 1 and Compound 3 against HRV2 and HRV3
The antiviral activity of the six isolated compounds (
1–
6) was tested; compounds
2,
4,
5, and
6 showed no antiviral effect (data not shown). However, the antiviral assays demonstrated that compound
1 showed anti-HRV2 activity with a 50% inhibitory concentration (IC
50) value of 2.50 μg/mL and a 50% cytotoxic concentration (CC
50) value of more than 20 μg/mL, although it did not show anti-HRV3 activity (
Table 4). Compound
3 also possessed strong antiviral activity with IC
50 values of 7.11 μg/mL against HRV2 and 4.69 μg/mL against HRV3, and a CC
50 value of more than 20 μg/mL (
Table 4). Ribavirin, tested as a positive control, also showed antiviral activity in HeLa cells infected with HRV2 and HRV3 with IC
50 values of 2.15 μg/mL and 5.09 μg/mL, respectively, and exhibited a CC
50 value of more than 20 μg/mL (
Table 4).
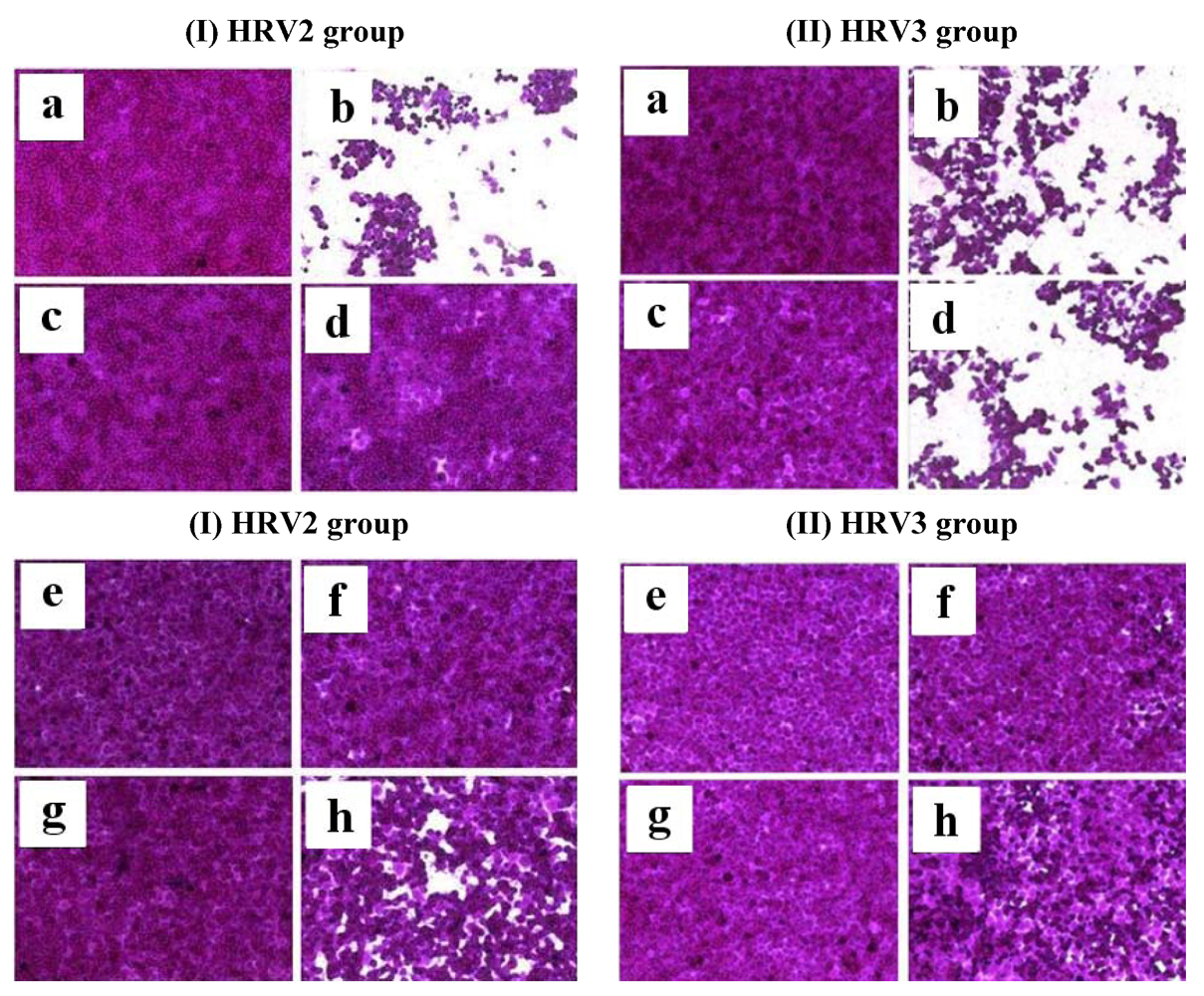
After a 2-day infection of HeLa cells with HRV2 and HRV3, the effect of compound
1 and compound
3 on HRV-induced cytopathic effect (CPE) investigated. As the results, uninfected cells (
Figure 3-I-a) or cells treated with compound
1 (
Figure 3-I-c), compound
3 (
Figure 3-I-e), and ribavirin (
Figure 3-I-g) showed typical spread-out shapes and normal morphology. No signs of cytotoxicity were observed for compounds
1,
3, or ribavirin at exposure concentrations of 20 μg/mL. Infection with HRV2 and HRV3 in the absence of compounds
1 and
3 resulted in a severe CPE (
Figure 3-I-b and 3-II-b). The addition of compound
1 or
3 to cultures of infected HeLa cells inhibited the formation of a visible CPE (
Figure 3-I-d, 3-I-f, and 3-II-f). Incubation of HRV2- or HRV3-infected HeLa cells with ribavirin also prevented a CPE (
Figure 3-I-h and 3-II-h).
Figure 3.
The effect of compound 1 or compound 3 on HRV2 (left group; I) and HRV 3 (right group; II)-induced CPE. The virus-infected cells were treated with compound 1 or compound 3 of 20 μg/mL. After incubation at 32 °C in 5% of CO2 for 2 days, the cell morphology was photographed under a microscope. (a) Non-infected cells; (I-b) HRV2 or (II-b) HRV3-infected cells without compound; (c) non-infected cells treated with compound 1; (I-d) HRV2 or (II-d) HRV3-infected cells with compound 1; (e) non-infected cells treated with compound 3; (I-f) HRV2 or (II-f) HRV3-infected cells with compound 3; (g) non-infected cells treated with ribavirin; (I-h) HRV2 or (II-h) HRV3-infected cells with ribavirin.
Figure 3.
The effect of compound 1 or compound 3 on HRV2 (left group; I) and HRV 3 (right group; II)-induced CPE. The virus-infected cells were treated with compound 1 or compound 3 of 20 μg/mL. After incubation at 32 °C in 5% of CO2 for 2 days, the cell morphology was photographed under a microscope. (a) Non-infected cells; (I-b) HRV2 or (II-b) HRV3-infected cells without compound; (c) non-infected cells treated with compound 1; (I-d) HRV2 or (II-d) HRV3-infected cells with compound 1; (e) non-infected cells treated with compound 3; (I-f) HRV2 or (II-f) HRV3-infected cells with compound 3; (g) non-infected cells treated with ribavirin; (I-h) HRV2 or (II-h) HRV3-infected cells with ribavirin.