Tetrodotoxin Concentrations in Pleurobranchaea maculata: Temporal, Spatial and Individual Variability from New Zealand Populations
Abstract
:1. Introduction
2. Results
2.1. Spatial Variability in Tetrodotoxin Concentrations in Pleurobranchaea maculata from Around New Zealand
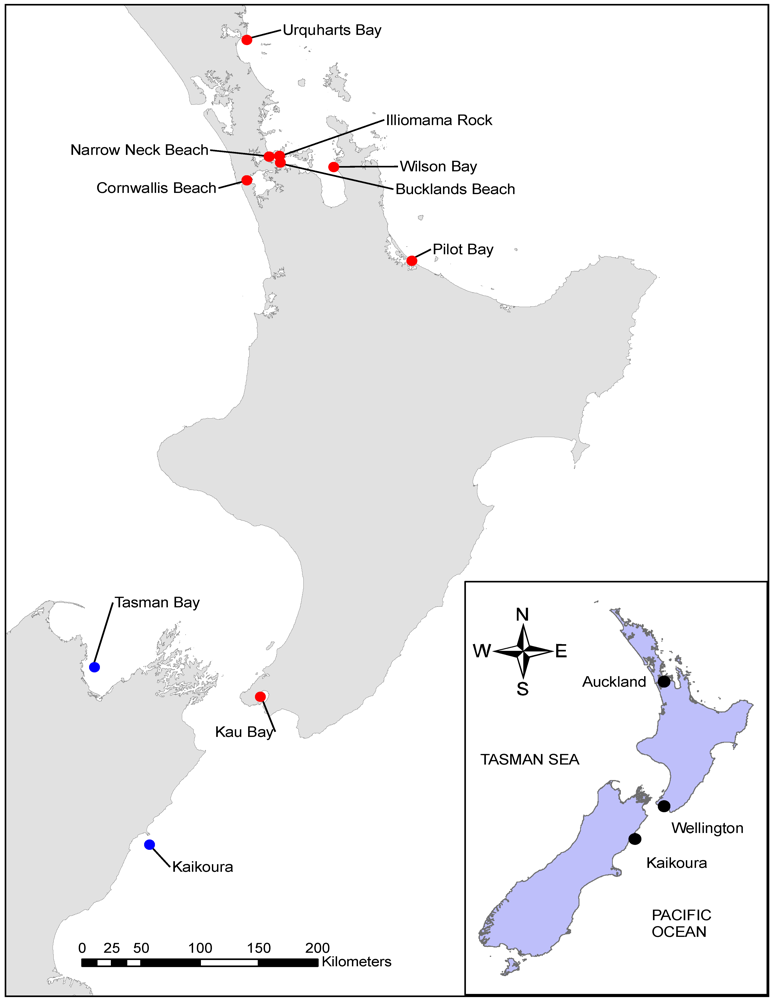
| Site | Depth (m) | Sampling Date | n | Tetrodotoxin (mg kg−1) | Pleurobranchaea maculata Mass (g) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ave | Min. | Max. | Std. Error | Ave | Min. | Max. | Std. Error | ||||
| Narrow Neck Beach | 3–6 | 14/06/2010 | 10 | 181.9 | 9.9 | 506.0 | 48.4 | 7.4 | 1.3 | 12.2 | 0.9 |
| 3–6 | 12/07/2010 | 10 | 85.7 | 15.2 | 150.2 | 15.3 | 14.3 | 5.6 | 20.8 | 1.5 | |
| 3–6 | 31/08/2010 | 9 | 52.6 | 10.3 | 120.5 | 12.1 | 20.7 | 14.0 | 42.7 | 2.9 | |
| 3–6 | 11/11/2010 | 9 | 21.7 | 1.7 | 83.1 | 8.9 | 20.7 | 12.9 | 33.4 | 2.4 | |
| 3–6 | 16/12/2010 | 10 | 17.5 | 1.1 | 48.4 | 5.4 | 15.5 | 7.8 | 25.7 | 1.7 | |
| Illiomama Rock | 6 | 14/06/2010 | 10 | 368.7 | 22.7 | 1414.0 | 146.6 | 6.3 | 3.6 | 9.7 | 0.6 |
| Bucklands Beach | 3–6 | 11/11/2010 | 10 | 28.8 | 1.5 | 39.4 | 12.5 | 30.8 | 20.4 | 39.5 | 2.1 |
| Tasman Bay | 20 | 05/11/2010 | 20 | 0.0 | 0.0 | 0.0 | - | 50.4 | 24.0 | 89.5 | 4.9 |
| Urquharts Bay | 3 | 28/08/2010 | 10 | 16.8 | 1.4 | 30.4 | 2.9 | 23.9 | 13.1 | 31.1 | 1.7 |
| Pilot Bay | 3–5 | 08/06/2011 | 8 | 89.6 | 29.7 | 205.6 | 19.7 | 11.6 | 8.7 | 16.3 | 1.1 |
| Kaikoura | 290 | 18/11/2011 | 1 | 0.0 | - | - | - | 17.5 | - | - | - |
| Cornwallis Beach | 3 | 02/09/2010 | 1 | 130.0 | - | - | - | 12.9 | - | - | - |
| Kau Bay | 14 | 01/08/2010 | 1 | 2.2 | - | - | - | 1.5 | - | - | - |
| Wilson Bay | 6 | 15/12/2010 | 1 | 0.1 | - | - | - | 7.2 | - | - | - |
| Urquharts Bay | Tasman Bay | Pilot Bay | Illiomama Rock | Bucklands Beach | Narrow Neck | |
|---|---|---|---|---|---|---|
| Urquharts Bay | 0.0001 | 0.0072 | 0.0001 | 1.0000 | 0.9981 | |
| Tasman Bay | 0.0001 | 0.0001 | 0.0001 | 0.0001 | 0.0001 | |
| Pilot Bay | 0.0072 | 0.0001 | 0.4334 | 0.0064 | 0.0022 | |
| Illiomama Rock | 0.0001 | 0.0001 | 0.4334 | 1.0000 | 0.0001 | |
| Bucklands Beach | 1.0000 | 0.0001 | 0.0064 | 0.0001 | 0.9981 | |
| Narrow Neck | 0.9981 | 0.0001 | 0.0022 | 0.0001 | 0.9981 |
2.2. Temporal Variation in Tetrodotoxin Concentrations in Pleurobranchaea maculata from Narrow Neck Beach
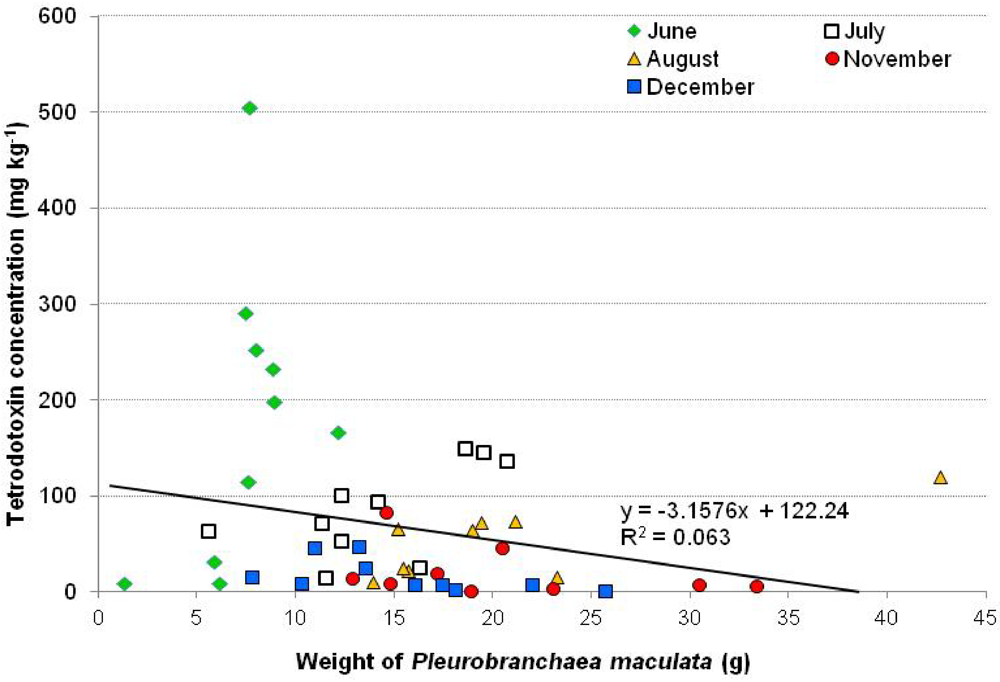
2.3. Rearing of a Second Generation Pleurobranchaea maculata in Captivity and Associated Tetrodotoxin
2.4. Cytochrome Oxidase Subunit 1 Gene Analysis of Narrow Neck Beach and Tasman Bay Pleurobranchaea maculata
3. Discussion
4. Conclusions
5. Experimental Section
5.1. Sampling Sites and Collection
5.2. Rearing of Pleurobranchaea maculata
5.3. Tetrodotoxin Extraction and Analysis
5.4. DNA Extraction and CO1 Analysis
5.5. Statistical Analysis
Acknowledgements
- Samples Availability: Available from the authors.
References
- Noguchi, T.; Ebesu, J.S.M. Puffer poisoning: Epidemiology and treatment. J. Toxicol. Toxin Rev. 2001, 20, 1–10. [Google Scholar]
- Mosher, H.S.; Fuhrman, G.J.; Fuhrman, F.A.; Fischer, H.G. Tarichatoxin-tetrodotoxin, a potent neurotoxin. Science 1965, 144, 1100–1110. [Google Scholar]
- Noguchi, T.; Arakawa, O. Tetrodotoxin—Distribution and accumulation in aquatic organisms, and cases of human intoxication. Mar. Drugs 2008, 6, 220–242. [Google Scholar]
- Wu, Z.; Xiea, L.; Xiab, G.; Zhanga, J.; Niea, Y.; Huc, J.; Wangc, S.; Zhanga, R. A new tetrodotoxin-producing actinomycete, Nocardiopsis dassonvillei, isolated from the ovaries of puffer fish Fugu rubripes. Toxicon 2005, 45, 851–859. [Google Scholar]
- Chau, R.; Kalaitzis, J.A.; Neilan, B.A. On the origins and biosynthesis of tetrodotoxin. Aquat. Toxicol. 2011, 104, 61–72. [Google Scholar]
- Matsumura, K. No ability to produce tetrodotoxin in bacteria. Appl. Environ. Microbiol. 2001, 67, 2393–2394. [Google Scholar]
- Matsumura, K. Re-examination of tetrodotoxin production by bacteria. Appl. Environ. Microbiol. 1995, 61, 3468–3470. [Google Scholar]
- Williams, B.L. Behavioral and chemical ecology of marine organisms with respect to tetrodotoxin. Mar. Drugs 2010, 8, 381–398. [Google Scholar]
- Shimizu, Y.; Kobayashi, M. Apparent lack of tetrodotoxin biosynthesis in captured Taricha torosa and Taricha granulosa. Chem. Pharm. Bull. (Tokyo) 1983, 31, 3625–3631. [Google Scholar]
- Daly, J.W.; Myers, C.W.; Whittaker, N. Further classification of skin alkaloids from neotropical poison frogs (Dendrobatidae), with a general survey of toxic/noxious substances in the amphibia. Toxicon 1987, 25, 1023–1095. [Google Scholar]
- Cardall, B.L.; Brodie, E.D.; Brodie, E.D.; Hanifin, C.T. Secretion and regeneration of tetrodotoxin in the rough-skin newt (Taricha garnulsoa). Toxicon 2004, 44, 933–938. [Google Scholar]
- Hanifin, C.T.; Brodie, E.D., III; Brodie, E.D., Jr. Tetrodotoxin levels of the rough-skin newt, Taricha granuslosa, increase in long term captivity. Toxicon 2002, 40, 1149–1153. [Google Scholar]
- Lehman, E.M.; Brodie, E.D.; Brodie, E.D. No evidence for an endosymbiotic bacterial origin of tetrodotoxin in the newt Taricha granulosa. Toxicon 2004, 44, 243–249. [Google Scholar]
- Ritson-Williams, R.R.; Yotsu-Yamashita, M.; Paul, V. Ecological functions of tetrodotoxin in a deadly polyclad flatworm. Proc. Natl. Acad. Sci. USA 2006, 103, 3176–3179. [Google Scholar]
- Hwang, P.A.; Noguchi, T.; Hwang, D.F. Neurotoxin tetrodotoxin as attractant for toxic snails. Fish. Sci. 2004, 70, 1106–1112. [Google Scholar]
- Matsumura, K. Tetrodotoxin as a pheromone. Nature 1995, 378, 563–564. [Google Scholar]
- Matsui, T.; Hamada, S.; Konosu, S. Difference in accumulation of puffer fish toxin and crystalline tetrodotoxin in the puffer fish, Fugu rubripes rubripes. Bull. Jpn. Soc. Sci. Fish. 1981, 47, 535–537. [Google Scholar]
- Hanifin, C.T.; Yotu-Yamashita, M.; Yasumoto, T.; Brodie, E.D., III; Brodie, E.D., Jr. Toxicity of dangerous prey: Variation of tetrodotoxin levels within and among populations of the newt Taricha granulosa. J. Chem. Ecol. 1999, 25, 2161–2175. [Google Scholar] [CrossRef]
- Brodie, E.D., Jr.; Ridenhour, B.J.; Brodie, E.D., III. The evolutionary response of predators to dangerous prey: Hotspots and coldspots in the geographic mosaic of coevolution between garter snakes and newts. Evolution 2002, 56, 2067–2082. [Google Scholar] [PubMed]
- Willan, R.C. New Zealand side-gilled sea slugs (Opisthobranchia, Notaspidea, Pleurobranchidae). Malacologia 1983, 23, 221–270. [Google Scholar]
- Gibson, G.D. Larval development and mtamorphosis in Pleurobranchaea maculata, with a review of development in the Notaspidea (Opisthobranchia). Biol. Bull. 2003, 205, 121–132. [Google Scholar]
- Ji, Y.; Liu, Y.; Gong, Q.-L.; Zhou, L.; Wang, Z.-P. Toxicity of cultured puffer fish and seasonal variations in China. Aquacul. Res. 2011, 42, 1186–1195. [Google Scholar]
- Miyazawa, K.; Noguchi, T. Distribution and origin of tetrodotoxin. Toxicol. Toxin Rev. 2001, 20, 11–33. [Google Scholar]
- Saitanu, K.; Piyakarnchana, T.; Sato, S.; Ogata, T.; Kodama, M. Toxicity of Two Species of Horseshoe Crab in Thailand. In Progress in Venom and Toxin Research; Gopalakrishnakone, P., Tan, C.K., Eds.; Faculty of Medicine, National University of Singapore: Singapore, 1988; pp. 493–498. [Google Scholar]
- Laymithuna, N.; Chun-Fai, Y.; Takatani, T.; Arakawa, O. Toxicity assessment for the horseshoe crab Carcinoscorpius rotundicauda collected from Cambodia. Toxicon 2007, 49, 843–847. [Google Scholar]
- Dao, H.V.; Yoshinobu, T.; Sato, S.; Fukuyo, Y.; Kodama, M. Frequent occurrence of the tetrodotoxin-bearing horseshoe crab Carcinoscorpius rotundicauda in Vietnam. Fish. Sci. 2009, 75, 435–438. [Google Scholar]
- Miyazawa, K.; Noguchi, T.; Maruyama, J.; Jeon, J.K.; Otsuka, M.; Hashimoto, K. Occurrence of tetrodotoxin in the starfishes Astropecten polyacanthus and A. scoparius in the Seto Inland Sea. Mar. Biol. 1985, 90, 61–64. [Google Scholar] [CrossRef]
- Noguchi, T.; Arakawa, O.; Takatani, T. Toxicity of pufferfish Takifugu rubripes cultured in net cages at sea or aquaria on land. Comp. Biochem. Phys. D 2006, 153–157. [Google Scholar]
- Taylor, D.I.; Wood, S.A.; McNabb, P. Population surveys of Pleurobranchaea maculata and tetrodotoxin in Waitemata Harbour. Prepared for Auckland Council, Cawthron Report No. 2006; 2011; pp. 1–11. [Google Scholar]
- Fuhrman, J.A.; Steele, J.A.; Hewson, I.; Schwalbach, M.S.; Brown, M.V.; Green, J.L.; Brown, J.H. A latitudinal diversity gradient in planktonic marine bacteria. Proc. Natl. Acad. Sci. USA 2008, 105, 7774–7778. [Google Scholar]
- Tuckey, B.J.; Gibbs, M.T.; Knight, B.R.; Gillespie, P.A. Tidal circulation in Tasman and Golden Bays: implications for river plume behaviour. N. Z. J. Mar. Freshwat. Res. 2006, 40, 305–324. [Google Scholar]
- Walters, R.; Gillibrand, P.; Bell, R.; Lane, E. A study of tides and currents in Cook Strait, New Zealand. Ocean Dynam. 2010, 60, 1559–1580. [Google Scholar]
- Hwang, D.E.; Lu, S.C.; Jeng, S.S. Occurrence of tetrodotoxin in the gastropods Rapana rapiformis and R. venosa venosa. Mar. Biol. 1991, 111, 65–69. [Google Scholar] [CrossRef]
- Brodie, E.D., Jr. Investigation on the skin toxin of the adult rough-skinned newt Taricha garnulsoa. Copeia 1968, 307–313. [Google Scholar]
- Brodie, E.D., Jr.; Hensel, J.L.; Johnson, J.A. Toxicity of the urodele amphibians Taricha, Notophthalmus, Cynops, and Paramesotriton (Salamandridae). Copeia 1974, 506–511. [Google Scholar]
- Asakawa, M.; Toyoshima, T.; Ito, K.; Bessho, K.; Yamaguchi, C.; Tsunetsugu, S.; Shida, Y.; Kajihara, H.; Mawatari, S.F.; Noguchi, T.; Miyazawa, K. Paralytic toxicity in the ribbon worm Cephalothrix species (Nemertea) in Hiroshima Bay, Hiroshima Prefecture, Japan and the isolation of tetrodotoxin as a main component of its toxins. Toxicon 2003, 41, 747–753. [Google Scholar]
- Chen, C.-Y.; Chou, H.-N. Detection of tetrodotoxin by high performance liquid chromatography in Lined-Moon shell and puffer fish. Acta Zool. Taiwanica 1998, 9, 41–48. [Google Scholar]
- Taniyama, S.; Isami, Y.; Matusmoto, T.; Nagashima, Y.; Takatani, T.; Arakawa, O. Toxicity and toxin profile of tetrodotoxin detected in the scavenging gastropod Nassarius (Alectrion) glans “kinshibai”. J. Food Hyg. Soc. Jpn. 2009, 50, 22–28. [Google Scholar] [CrossRef]
- Miyazawa, K.; Higashiyama, M.; Hori, K.; Noguchi, T.; Ito, K.; Hashimoto, K. Distribution of tetrodotoxin in various organs of the starfish Astropecten polyacanthus. Mar. Biol. 1987, 96, 385–390. [Google Scholar]
- Sabrah, M.M.; El-Ganainy, A.A.; Zaky, M.A. Biology and toxicity of the pufferfish Lagocephalus sceleratus (GMELIN, 1789) from the Gulf of Suez. Egypt. J. Aquat. Res. 2006, 32, 283–297. [Google Scholar]
- Ikeda, K.; Emoto, Y.; Tatsuno, R.; Wang, J.J.; Ngy, L.; Taniyama, S.; Takatani, T.; Arakawa, O. Maturation-associated changes in toxicity of the pufferfish Takifugu poecilonotus. Toxicon 2010, 55, 289–297. [Google Scholar]
- McNabb, P.; Selwood, A.I.; Munday, R.; Wood, S.A.; Taylor, D.I.; MacKenzie, A.L.; van Ginkel, R.; Rhodes, L.L.; Cornelisen, C.; Heasman, K.; et al. Detection of tetrodotoxin from the grey sided-gilled sea slug-Pleurobranchaea maculata, and associated dog neurotoxicosis on beaches adjacent to the Hauraki Gulf, Auckland, New Zealand. Toxicon 2010, 56, 466–473. [Google Scholar]
- Wood, S.A. Study showed decrease in TTX concentrations in sequentially laid egg masses of Pleurobranchaea maculata; Cawthron Institute: Nelson, New Zealand; p. 2011.
- Daly, J.W.; Padgett, W.L.; Saunders, R.L.; Cover, J.F., Jr. Absence of tetrodotoxins in a captive-raised riparian frog, Atelopus varius. Toxicon 1997, 35, 705–709. [Google Scholar]
- Gall, B.G.; Stokes, A.N.; French, S.S.; Schlepphorst, E.A.; Brodie, E.D., III; Brodie, E.D., Jr. Tetrodtoxin levels in larval and metamorphosed newts (Taricha granulosa) and palatability to predatory dragonflies. Toxicon 2011, 57, 978–983. [Google Scholar]
- Williams, B.L.; Hanifin, C.T.; Brodie, E.D., Jr.; Caldwell, R.L. Ontogenty of tetrodtoxin levels in blue-ringed octopuses: maternal investment and apparent independent production in offspring of Hapalochlaena lunulata. J. Chem. Ecol. 2011, 37, 10–17. [Google Scholar]
- Folmer, O.; Black, M.; Hoeh, W.; Lutz, R.; Vrijenhoek, R. DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol. Mar. Biol. Biotechnol. 1994, 3, 294–299. [Google Scholar]
- Benson, D.A.; Karsch-Mizrachi, I.; Lipman, D.J.; Ostell, J.; Wheeler, D.L. GenBank. Nucleic Acids Res. 2008, 36, D25–D30. [Google Scholar]
- Statistica, Version 8, StatSoft Inc.: Tulsa, OK, USA, 2008.
© 2012 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wood, S.A.; Taylor, D.I.; McNabb, P.; Walker, J.; Adamson, J.; Cary, S.C. Tetrodotoxin Concentrations in Pleurobranchaea maculata: Temporal, Spatial and Individual Variability from New Zealand Populations. Mar. Drugs 2012, 10, 163-176. https://doi.org/10.3390/md10010163
Wood SA, Taylor DI, McNabb P, Walker J, Adamson J, Cary SC. Tetrodotoxin Concentrations in Pleurobranchaea maculata: Temporal, Spatial and Individual Variability from New Zealand Populations. Marine Drugs. 2012; 10(1):163-176. https://doi.org/10.3390/md10010163
Chicago/Turabian StyleWood, Susanna A., David I. Taylor, Paul McNabb, Jarrod Walker, Janet Adamson, and Stephen Craig Cary. 2012. "Tetrodotoxin Concentrations in Pleurobranchaea maculata: Temporal, Spatial and Individual Variability from New Zealand Populations" Marine Drugs 10, no. 1: 163-176. https://doi.org/10.3390/md10010163
APA StyleWood, S. A., Taylor, D. I., McNabb, P., Walker, J., Adamson, J., & Cary, S. C. (2012). Tetrodotoxin Concentrations in Pleurobranchaea maculata: Temporal, Spatial and Individual Variability from New Zealand Populations. Marine Drugs, 10(1), 163-176. https://doi.org/10.3390/md10010163





