Transcriptomic and Non-Targeted Metabolomic Analyses Reveal the Flavonoid Biosynthesis Pathway in Auricularia cornea
Abstract
:1. Introduction
2. Results
2.1. Overview of Qualitative Metabolomics Analysis of A. cornea
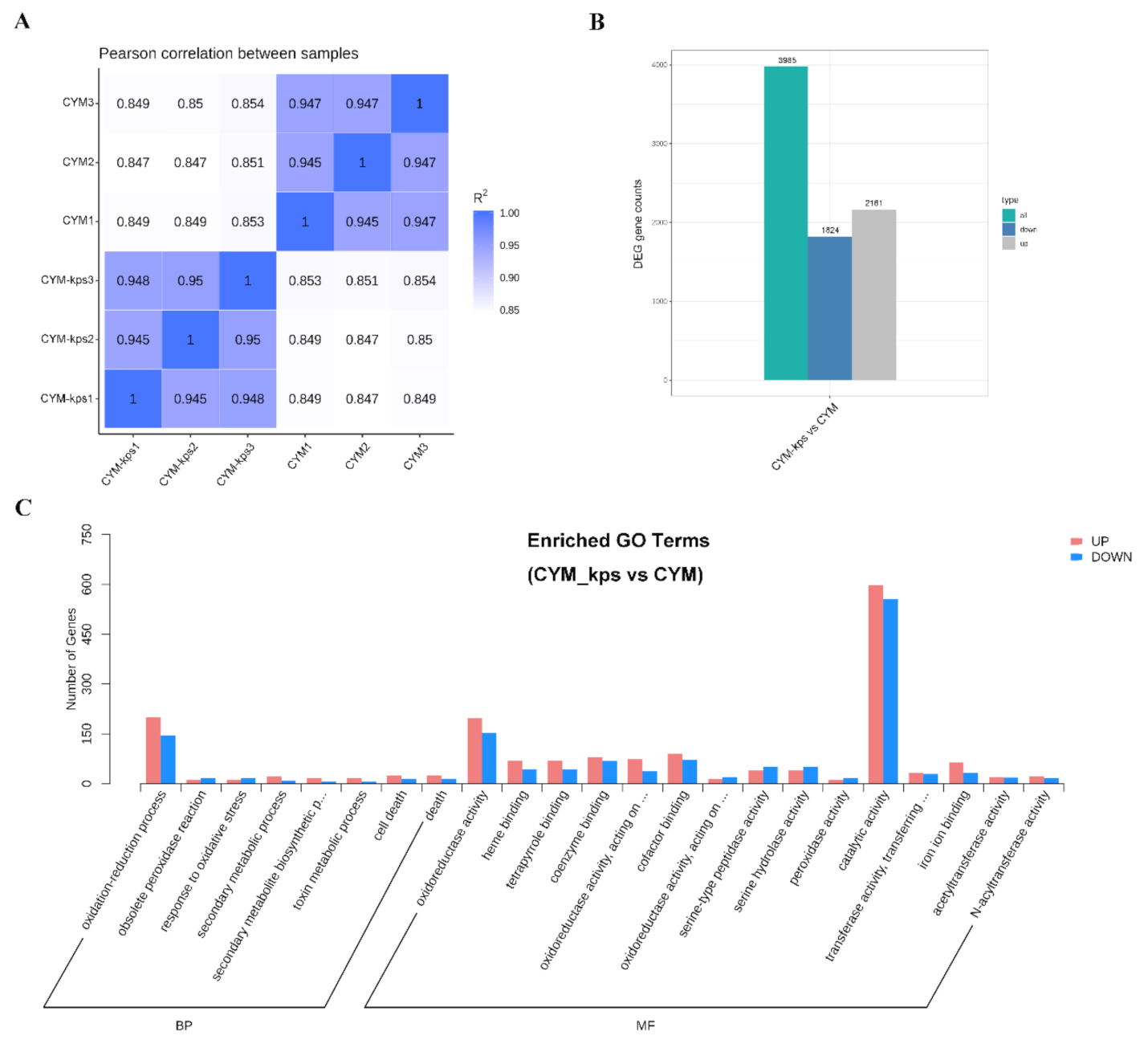
2.2. Overview of Qualitative Transcriptomic Analysis of A. cornea
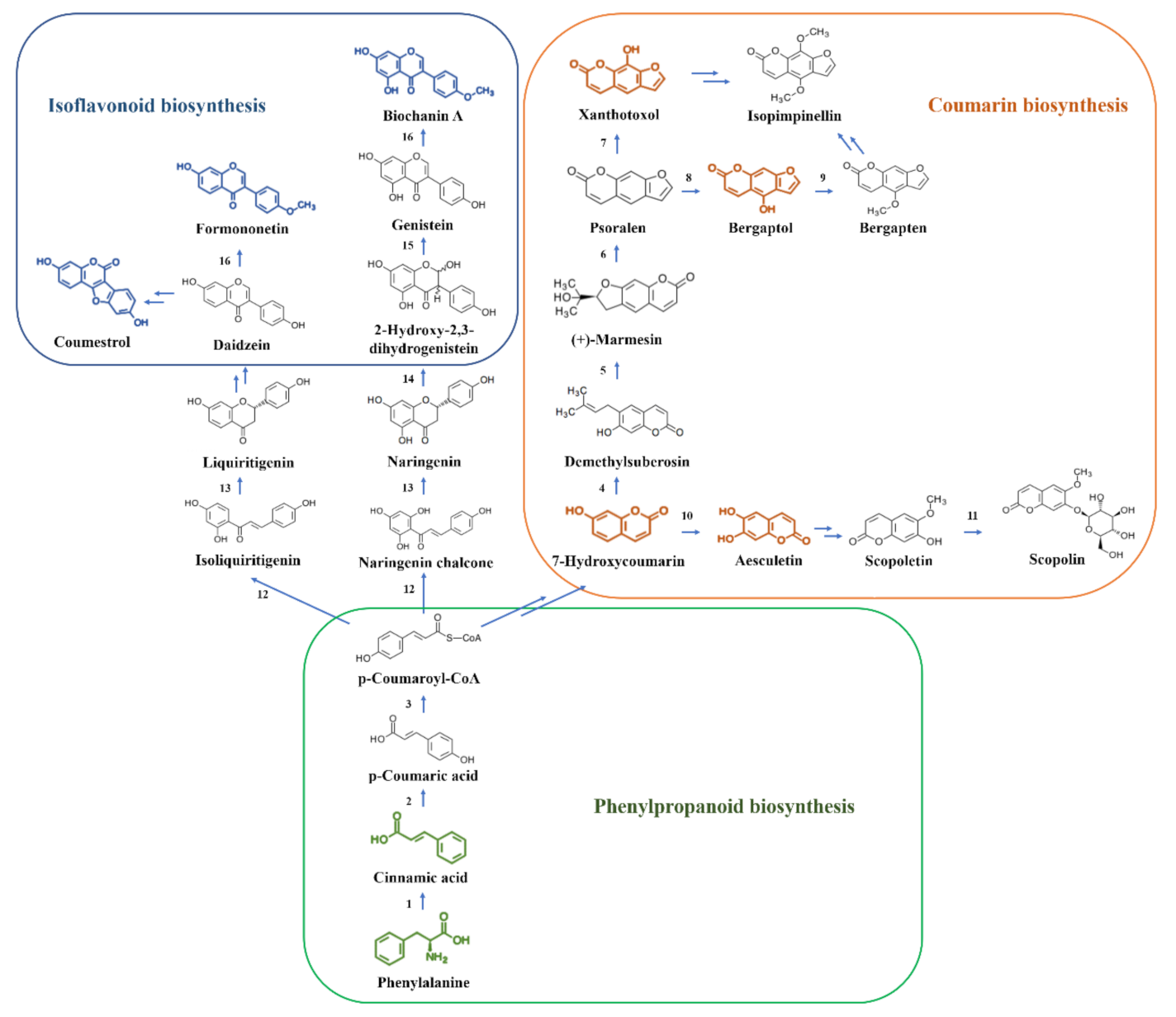
2.3. The Flavonoid Biosynthesis Pathway in A. cornea
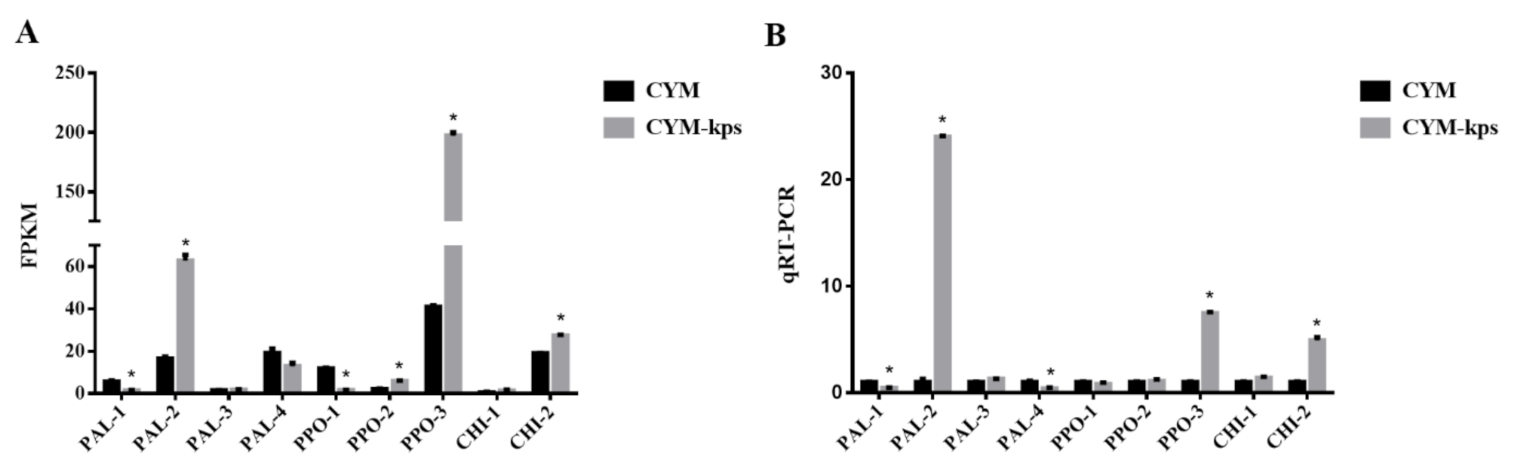
2.4. The Expression Levels of the Genes Encoding Key Enzymes Involved in the Flavonoid Biosynthesis Pathway
3. Discussion
4. Materials and Methods
4.1. Strains and Culture Conditions
4.2. Determination of Total Flavonoid Content
4.3. Sample Preparation and LC–MS Analysis
4.4. Metabolite Identification and Quantification
4.5. Transcriptome Sequencing Analysis
4.6. Gene Expression Analysis and Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Neuhouser, M.L. Dietary flavonoids and cancer risk: Evidence from human population studies. Nutr. Cancer 2004, 50, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Ruan, Y.; Li, Z.; Li, D. Flavonoid subclasses and type 2 diabetes mellitus risk: A meta-analysis of prospective cohort studies. Crit. Rev. Food Sci. Nutr. 2019, 59, 2850–2862. [Google Scholar] [CrossRef] [PubMed]
- Middleton, E. Effect of plant flavonoids on immune and inflammatory cell function. Adv. Exp. Med. Biol. 1998, 439, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Maleki, S.J.; Crespo, J.F.; Cabanillas, B. Anti-inflammatory effects of flavonoids. Food Chem. 2019, 299, 125124. [Google Scholar] [CrossRef] [PubMed]
- Erdman, J.W., Jr.; Balentine, D.; Arab, L.; Beecher, G.; Dwyer, J.T.; Folts, J.; Harnly, J.; Hollman, P.; Keen, C.L.; Mazza, G.; et al. Flavonoids and heart health: Proceedings of the ILSI North America Flavonoids Workshop, May 31-June 1, 2005, Washington, DC. J. Nutr. 2007, 137, 718S–737S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hou, Y.; Jiang, J.G. Origin and concept of medicine food homology and its application in modern functional foods. Food Funct. 2013, 4, 1727–1741. [Google Scholar] [CrossRef]
- Azieana, J.; Zainon, M.N.; Noriham, A.; Rohana, M.N. Total phenolic and flavonoid content and antioxidant activities of ten Malaysian wild mushrooms. Open Access Libr. J. 2017, 4, e3987. [Google Scholar] [CrossRef]
- Zheng, S.; Huang, H.; Zhang, R.; Cao, L. Removal of Cr (VI) from aqueous solutions by fruiting bodies of the jelly fungus (Auricularia polytricha). Appl. Microbiol. Biotechnol. 2014, 98, 8729–8736. [Google Scholar] [CrossRef]
- Jia, D.; Wang, B.; Li, X.; Peng, W.; Zhou, J.; Tan, H.; Tang, J.; Huang, Z.; Tan, W.; Gan, B.; et al. Proteomic Analysis Revealed the Fruiting-Body Protein Profile of Auricularia polytricha. Curr. Microbiol. 2017, 74, 943–951. [Google Scholar] [CrossRef]
- Bai, H.; Wang, Z.; Cui, J.; Yun, K.; Zhang, H.; Liu, R.H.; Fan, Z.; Cheng, C. Synergistic Radiation Protective Effect of Purified Auricularia auricular-judae Polysaccharide (AAP IV) with Grape Seed Procyanidins. Molecules 2014, 19, 20675–20694. [Google Scholar] [CrossRef]
- Fraser, C.M.; Chapple, C. The phenylpropanoid pathway in Arabidopsis. Arab. Book 2011, 9, e0152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Winkel, B.S.J. The Biosynthesis of Flavonoids. In The Science of Flavonoids; Grotewold, E., Ed.; Springer: New York, NY, USA, 2006; pp. 71–95. [Google Scholar]
- Lucheta, A.R.; Silva-Pinhati, A.C.O.; Basílio-Palmieri, A.C.; Berger, I.J.; Freitas-Astúa, J.; Cristofani, M. An in silico analysis of the key genes involved in flavonoid biosynthesis in Citrus sinensis. Genet. Mol. Biol. 2007, 30, 819–831. [Google Scholar] [CrossRef]
- Czemmel, S.; Heppel, S.C.; Bogs, J. R2R3 MYB transcription factors: Key regulators of the flavonoid biosynthetic pathway in grapevine. Protoplasma 2012, 249, S109–S118. [Google Scholar] [CrossRef] [PubMed]
- Zha, J.; Wu, X.; Gong, G.; Koffas, M.A.G. Pathway enzyme engineering for flavonoid production in recombinant microbes. Metab. Eng. Commun. 2019, 9, e00104. [Google Scholar] [CrossRef]
- Cheng, J.; Wang, J.; Cheng, J.; Luo, Z. Spatial-temporal variability of Caragana korshinskii vegetation growth in the Loess Plateau. Sci. Silvae Sin. 2013, 49, 14–20. [Google Scholar] [CrossRef]
- Du, F.; Qu, J.; Hu, Q.; Yuan, X.; Yin, G.; Wang, L.; Zou, Y. Maximizing the value of Korshinsk peashrub branches by the integration of Pleurotus tuoliensis cultivation and anaerobic digestion of spent mushroom substrate. Renew. Energy 2021, 179, 679–686. [Google Scholar] [CrossRef]
- Barros, L.; Dueñas, M.; Ferreira, I.C.F.R.; Baptista, P.; Santos-Buelga, C. Phenolic acids determination by HPLC-DAD-ESI/MS in sixteen different Portuguese wild mushrooms species. Food Chem. Toxicol. 2009, 47, 1076–1079. [Google Scholar] [CrossRef]
- Gil-Ramírez, A.; Pavo-Caballero, C.; Baeza, E.; Baenas, N.; Garcia-Viguera, C.; Marín, F.R.; Soler-Rivas, C. Mushrooms do not contain flavonoids. J. Funct. Foods 2016, 25, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Ribeiro, B.; Valentão, P.; Baptista, P.; Seabra, R.M.; Andrade, P.B. Phenolic compounds, organic acids profiles and antioxidative properties of beefsteak fungus (Fistulina hepatica). Food Chem. Toxicol. 2007, 45, 1805–1813. [Google Scholar] [CrossRef] [Green Version]
- Pasailiuk, M.V. Total flavonoid content, lipid peroxidation and total antioxidant activity of Hericium coralloides, Fomes fomentarius and Schizophyllum commune cultivated by the method of direct confrontation. Ital. J. Mycol. 2020, 49, 25–37. [Google Scholar] [CrossRef]
- Imran, M.M.; Mahroop, R.M.; Abdul, B.J.; Asarudeen, A. Determination of total phenol, flavonoid and antioxidant activity of edible mushrooms Pleurotus florida and Pleurotus eous. Int. Food Res. J. 2011, 18, 579–582. [Google Scholar]
- Ma, X.K.; Ma, H.; Chen, Q.; Ma, Y.; Daugulis, A.J.; Liang, J.; Zheng, P. The influence of monochromatic lights on flavonoid production by the fungus Sanghuangporus vaninii: Modeling of kinetic profiles and expression levels of important genes in flavonoid synthesis. Biochem. Eng. J. 2021, 166, 107876. [Google Scholar] [CrossRef]
- Mohanta, T.K. Fungi contain genes associated with flavonoid biosynthesis pathway. J. Funct. Foods 2020, 68, 103910. [Google Scholar] [CrossRef]
- Jiang, T.; Luo, Z.; Ying, T. Fumigation with essential oils improves sensory quality and enhanced antioxidant ability of shiitake mushroom (Lentinus edodes). Food Chem. 2015, 172, 692–698. [Google Scholar] [CrossRef]
- Butkhup, L.; Samappito, W.; Jorjong, S. Evaluation of bioactivities and phenolic contents of wild edible mushrooms from northeastern Thailand. Food Sci. Biotechnol. 2018, 27, 193–202. [Google Scholar] [CrossRef]
- Tel, G.; Apaydın, M.; Duru, M.E.; Öztürk, M. Antioxidant and Cholinesterase Inhibition Activities of Three Tricholoma Species with Total Phenolic and Flavonoid Contents: The Edible Mushrooms from Anatolia. Food Anal. Methods 2012, 5, 495–504. [Google Scholar] [CrossRef]
- Yıldız, S.; Yılmaz, A.; Can, Z.; Kılıç, C.; Yıldız, Ü.C. Total phenolic, flavonoid, tannin contents and antioxidant properties of Pleurotus ostreatus and Pleurotus citrinopileatus cultivated on various sawdust. GIDA 2017, 42, 315–323. [Google Scholar] [CrossRef]
- Hyun, M.W.; Yun, Y.H.; Kim, J.Y.; Kim, S.H. Fungal and Plant Phenylalanine Ammonia-lyase. Mycobiology 2011, 39, 257–265. [Google Scholar] [CrossRef] [Green Version]
- Shao, Y.; Guo, H.; Zhang, J.; Liu, H.; Wang, K.; Zuo, S.; Xu, P.; Xia, Z.; Zhou, Q.; Zhang, H.; et al. The genome of the medicinal macrofungus Sanghuang provides insights into the synthesis of diverse secondary metabolites. Front. Microbiol. 2020, 10, 3035. [Google Scholar] [CrossRef] [Green Version]
- Kouassi, K.A.; Kouadio, E.J.P.; Konan, K.H.; Dué, A.E.; Kouamé, L.P. Phenolic compounds, organic acid and antioxidant activity of Lactarius subsericatus, Cantharellus platyphyllus and Amanita rubescens, three edible ectomycorrhizal mushrooms from center of Côte d’Ivoire. Eurasian J. Anal. Chem. 2016, 11, 127–139. [Google Scholar] [CrossRef]
- Liaquat, A.; Saima, K.; Mamona, N.; Naheed, R.; Saima, N.; Gokhan, Z.; Mahreen, M.; Shehla, P.; Natasha, S.; Muhammad, S.; et al. Chemical profiling, in vitro biological activities and Pearson correlation between phenolic contents and antioxidant activities of Caragana brachyantha Rech.f. S. Afr. J. Bot. 2021, 140, 189–193. [Google Scholar] [CrossRef]
- Si, F.L.; Duan, S.; Wang, X.; Wang, L.L. Phenolic Antioxidants of Auricularia Species and Their Inhibitory Effects on α-Amylase, α-Glucosidase and Acetylcholine Esterase Activities. Curr. Top. Nutraceutical Res. 2020, 18, 132–140. [Google Scholar] [CrossRef]
- Hermann, A.; Hyacinthe, A.; Koffi, G.; Appolinaire, K.; Jean, K. Phenolic compound and antioxidant activity of two slightly consumed wild mushrooms (Lentinus squarrosulus and Auricularia politrich) in three regions from center Ivory Coast. Turk. J. Agric. Food Sci. Technol. 2021, 9, 1931–1937. [Google Scholar] [CrossRef]
- Ma, X.K.; Li, L.; Peterson, E.C.; Ruan, T.; Duan, X. The influence of naphthaleneacetic acid (NAA) and coumarin on flavonoid production by fungus Phellinus sp.: Modeling of production kinetic profiles. Appl. Microbiol. Biotechnol. 2015, 99, 9417–9426. [Google Scholar] [CrossRef]
- Dong, J.; Zhang, M.; Lu, L.; Sun, L.; Xu, M. Nitric oxide fumigation stimulates flavonoid and phenolic accumulation and enhances antioxidant activity of mushroom. Food Chem. 2012, 135, 1220–1225. [Google Scholar] [CrossRef]
- Meng, L.; Zhang, S.; Chen, B.; Bai, X.; Li, Y.; Yang, J.; Wang, W.; Li, C.; Li, Y.; Li, Z. The MADS-box transcription factor GlMADS1 regulates secondary metabolism in Ganoderma lucidum. Mycologia 2021, 113, 12–19. [Google Scholar] [CrossRef]
- Haug, K.; Cochrane, K.; Nainala, V.C.; Williams, M.; Chang, J.; Jayaseelan, K.V.; O’Donovan, C. MetaboLights: A resource evolving in response to the needs of its scientific community. Nucleic Acids Res. 2019, 48, D440–D444. [Google Scholar] [CrossRef] [Green Version]
- Meng, L.; Bai, X.; Zhang, S.; Zhang, M.; Zhou, S.; Mukhtar, I.; Wang, L.; Li, Z.; Wang, W. Enhanced ganoderic acids accumulation and transcriptional responses of biosynthetic genes in Ganoderma lucidum fruiting bodies by elicitation supplementation. Int. J. Mol. Sci. 2019, 20, 2830. [Google Scholar] [CrossRef] [Green Version]
- Jia, D.H.; Wang, B.; Li, X.L.; Tan, W.; Gan, B.C.; Peng, W.H. Validation of reference genes for quantitative gene expression analysis in Auricularia cornea. J. Microbiol. Methods 2019, 163, 105658. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]


| Genes | Forward Primer Sequence (5′-3′) | Reverse Primer Sequence (5′-3′) |
|---|---|---|
| UBQ | CGGATCTAACAGCGTGGACTCTTC | CCTCCTGAGCGATTGGCACTTG |
| PAL-1 | CGCCAATCAGGTCGCTAT | TTGCTGCTTCGTCGGC |
| PAL-2 | GCAGGCTCCTTCCATCC | AGGCACCTCCTCATCGTC |
| PAL-3 | AGTACGACGCACTGGGC | CGCATCTGCTCTATCACG |
| PAL-4 | CGCAGATGGAGGACA | AGCGAGACGAACGAAGC |
| PPO-1 | TCGTGGCTTCAGATTGG | GCGACGCTTGCTTT |
| PPO-2 | AGACCGATCAACTCAGGTT | CAGGACGGAGCGATACTT |
| PPO-3 | ACGCTTCTCAACACCCTC | TTCGTCGCCAAACACC |
| CHI-1 | GAGTCTGCCTCCACCTACC | TTGATCTCGGCCAGCA |
| CHI-2 | CAATGGCTTGGGCTCGTT | CGTGCCGCTTCAGATGGT |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meng, L.; Zhang, S.; Bai, X.; Li, X.; Wang, Q.; Wang, L.; Wang, W.; Li, Z. Transcriptomic and Non-Targeted Metabolomic Analyses Reveal the Flavonoid Biosynthesis Pathway in Auricularia cornea. Molecules 2022, 27, 2334. https://doi.org/10.3390/molecules27072334
Meng L, Zhang S, Bai X, Li X, Wang Q, Wang L, Wang W, Li Z. Transcriptomic and Non-Targeted Metabolomic Analyses Reveal the Flavonoid Biosynthesis Pathway in Auricularia cornea. Molecules. 2022; 27(7):2334. https://doi.org/10.3390/molecules27072334
Chicago/Turabian StyleMeng, Li, Shaoyan Zhang, Xiaoran Bai, Xiaobo Li, Qingji Wang, Li Wang, Wei Wang, and Zhuang Li. 2022. "Transcriptomic and Non-Targeted Metabolomic Analyses Reveal the Flavonoid Biosynthesis Pathway in Auricularia cornea" Molecules 27, no. 7: 2334. https://doi.org/10.3390/molecules27072334






