Construction of an Integration Vector with a Chimeric Signal Peptide for the Expression of Monoclonal Antibodies in Mammalian Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Plasmid Vectors and Cell Cultures
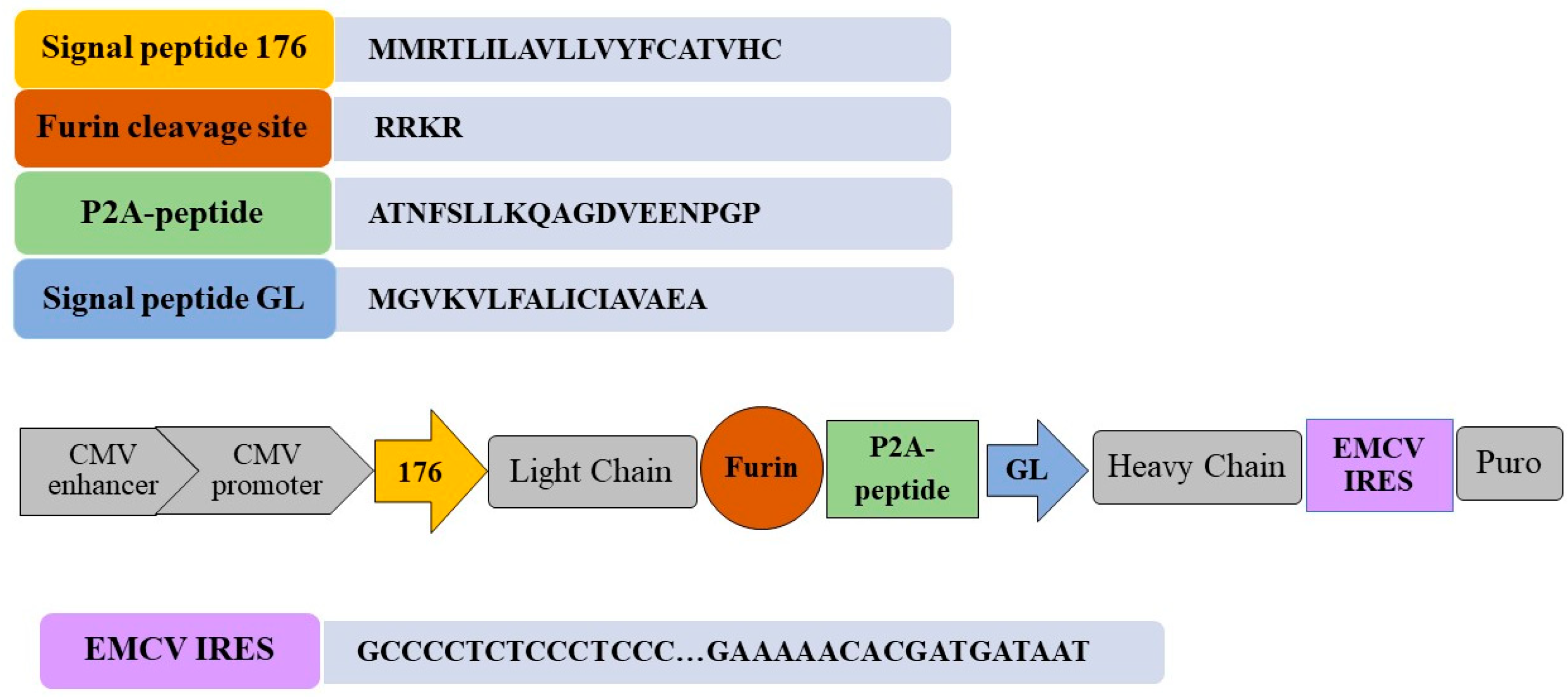
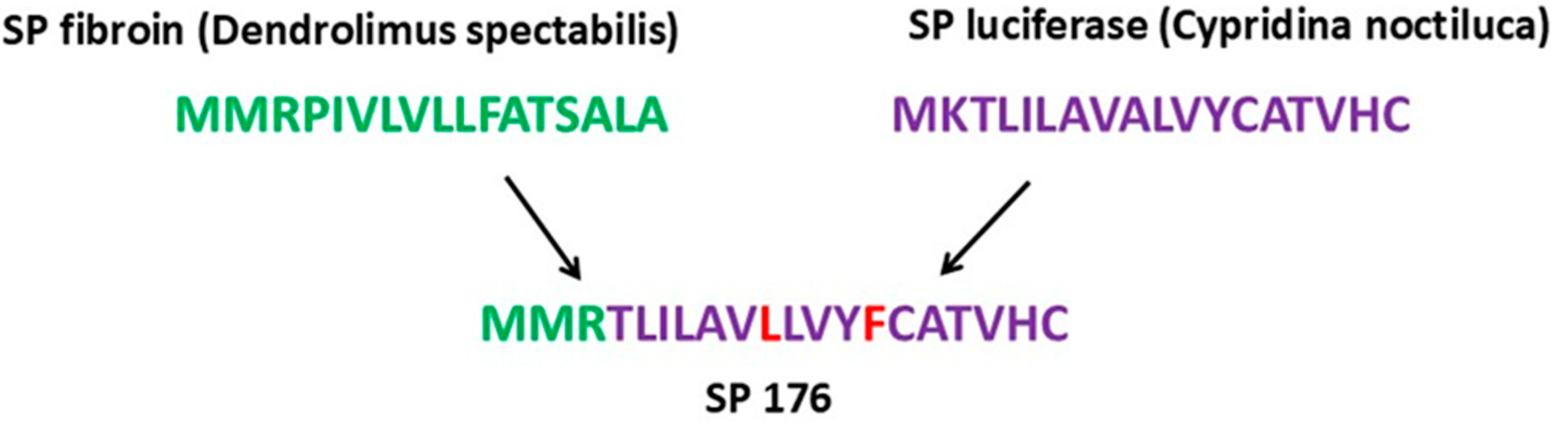
2.2. Construction of the Integration Vector pVEAL3-10H10ch
2.3. Generation of the Producer Strain CHO-K1-10H10ch
2.4. Roller Cultivation of the CHO-K1-10H10ch Producer Strain
2.5. Affinity Purification of Chimeric Full-Length Antibody 10H10ch
2.6. Purification of Mouse 10H10 Antibody with Caprylic Acid from Ascites Fluid
2.7. Enzyme-Linked Immunosorbent Assay (ELISA)
2.8. Statistical Analysis
3. Results and Discussion
3.1. Design and Construction of the Integration Vector pVEAL3-10H10ch
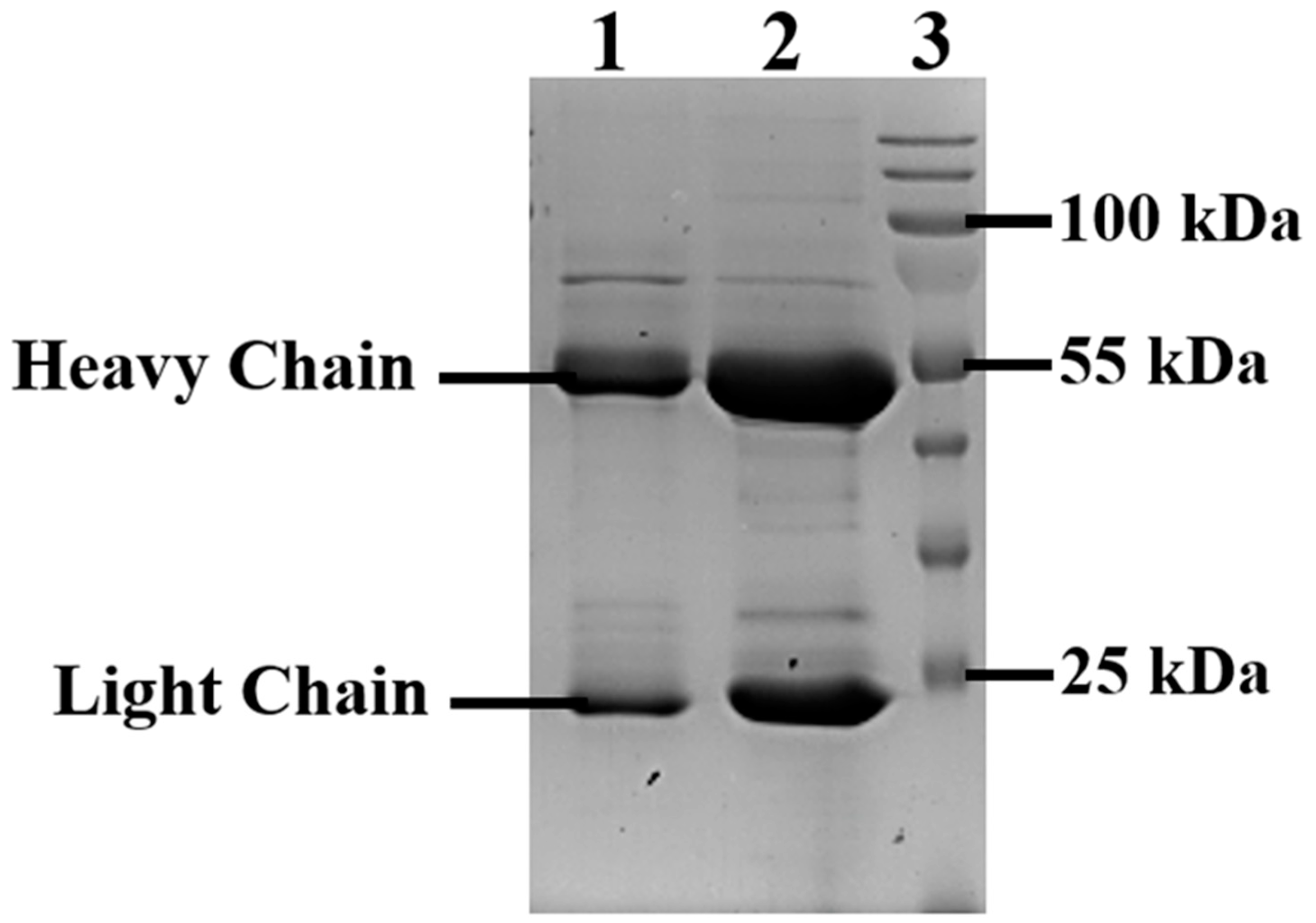
3.2. Obtaining a Stable Producer Strain CHO-K1-10H10ch
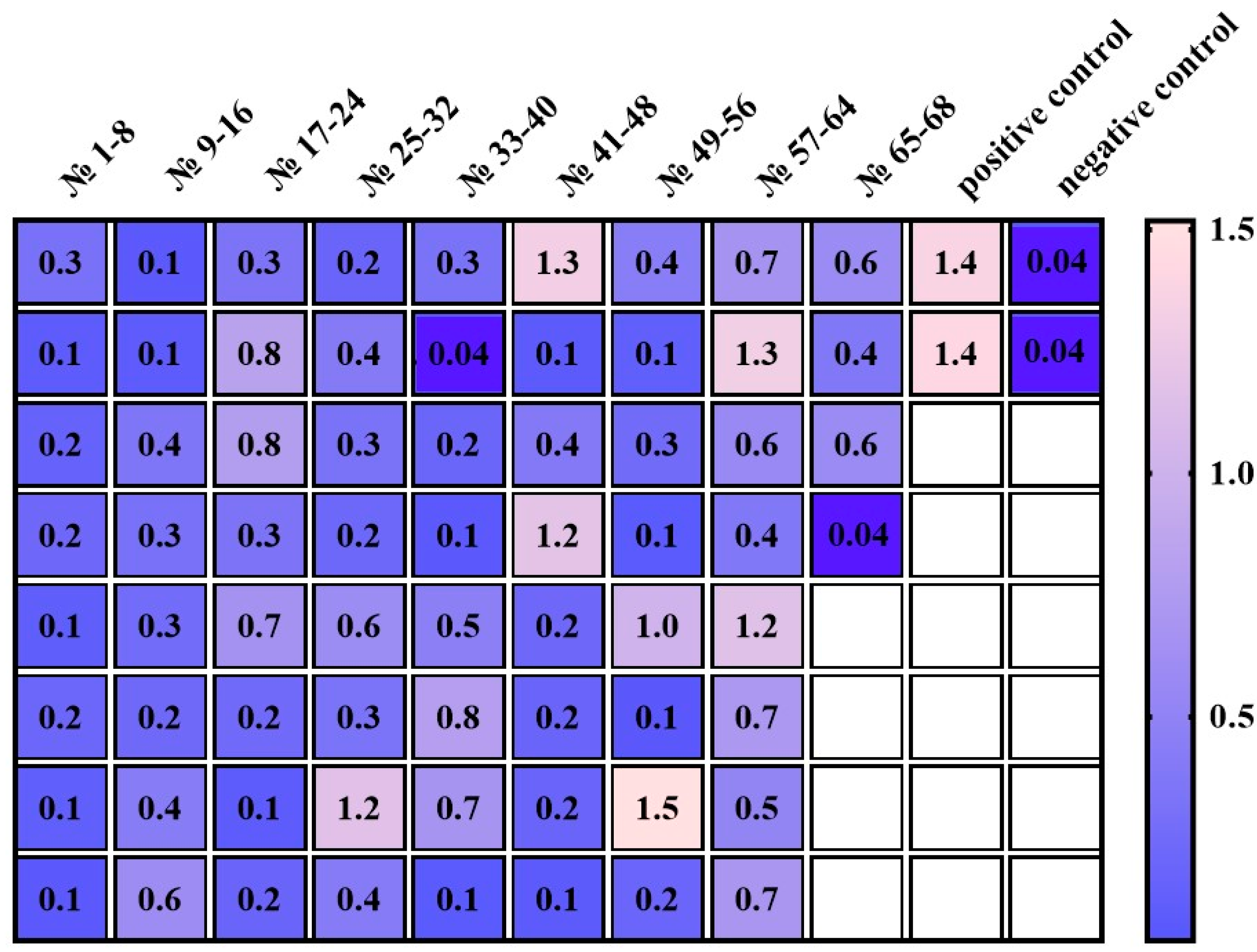
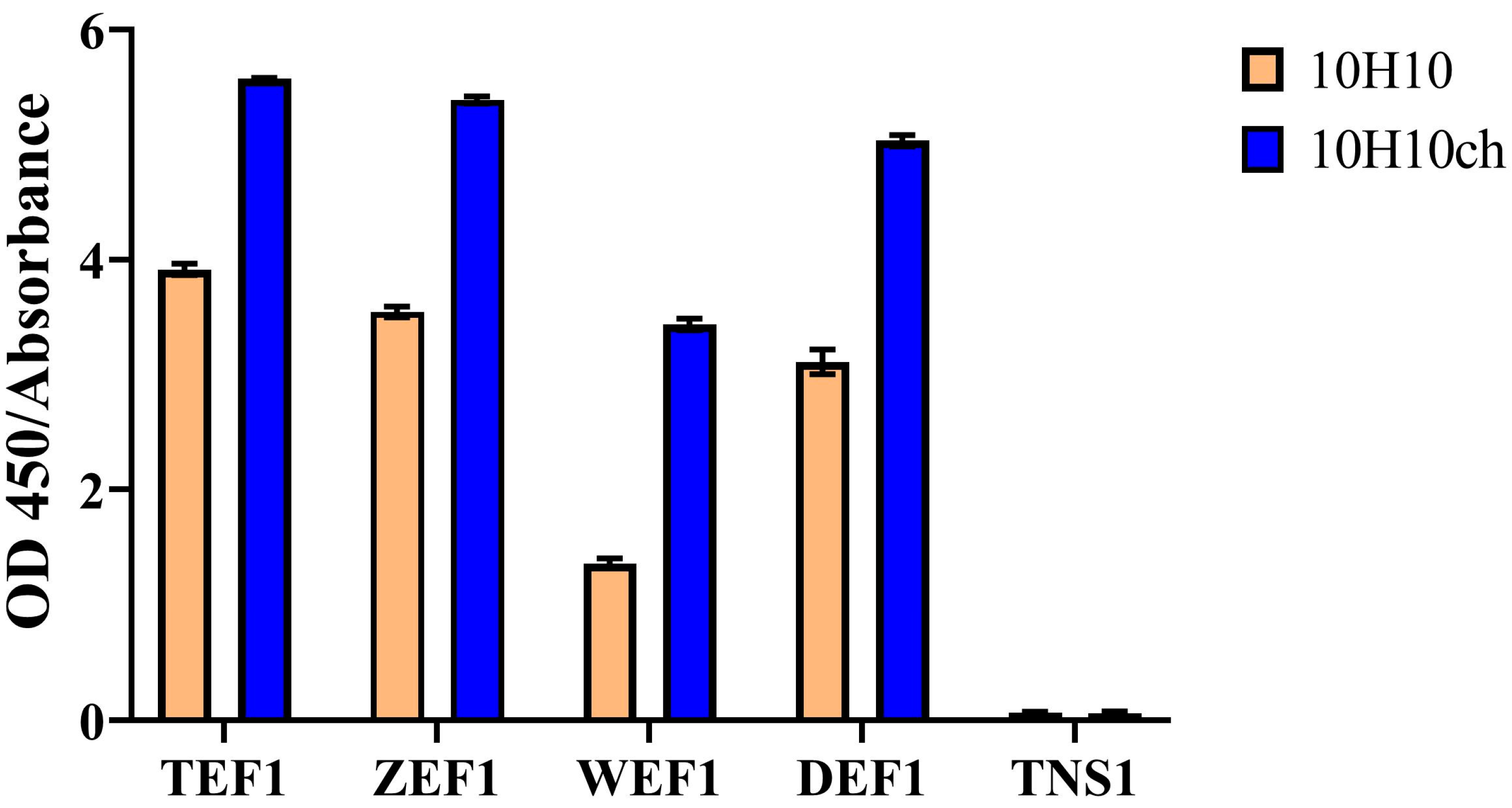
3.3. Evaluation of the Immunochemical Properties of the Full-Length 10H10ch Antibody
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hunter, M.; Yuan, P.; Vavilala, D.; Fox, M. Optimization of Protein Expression in Mammalian Cells. Curr. Protoc. Protein Sci. 2019, 95, e77. [Google Scholar] [CrossRef]
- Zhang, J.H.; Shan, L.L.; Liang, F.; Du, C.Y.; Li, J.J. Strategies and Considerations for Improving Recombinant Antibody Production and Quality in Chinese Hamster Ovary Cells. Front. Bioeng. Biotechnol. 2022, 10, 856049. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.M.; Fan, Z.L.; Wang, X.Y.; Wang, T.Y. Factors Affecting the Expression of Recombinant Protein and Improvement Strategies in Chinese Hamster Ovary Cells. Front. Bioeng. Biotechnol. 2022, 10, 880155. [Google Scholar] [CrossRef]
- Carrara, S.C.; Fiebig, D.; Bogen, J.P.; Grzeschik, J.; Hock, B.; Kolmar, H. Recombinant antibody production using a dual-promoter single plasmid system. Antibodies 2021, 10, 18. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, M.; Mahboudi, F.; Akbari Eidgahi, M.R.; Nasr, R.; Davami, F. Evaluating the efficiency of phiC31 integrase-mediated monoclonal antibody expression in CHO cells. Biotechnol. Prog. 2016, 32, 1570–1576. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.M.; Wang, M.; Wang, T.Y.; Wei, Y.G.; Dou, Y.Y. Effects of different 2A peptides on transgene expression mediated by tricistronic vectors in transfected CHO cells. Mol. Biol. Rep. 2020, 47, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Ebadat, S.; Ahmadi, S.; Ahmadi, M.; Nematpour, F.; Mahboudi, F. Evaluating the efficiency of CHEF and CMV promoter with IRES and Furin/2A linker sequences for monoclonal antibody expression in CHO cells. PLoS ONE 2017, 12, e0185967. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Hung, C.Y.; Bhattacharya, C.; Nichols, S.; Xie, J. An effective way of producing fully assembled antibody in transgenic tobacco plants by linking heavy and light chains via a self-cleaving 2a peptide. Front. Plant Sci. 2018, 9, 1379. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Ma, B.; Ma, J.; Zhu, R. RB1 5′UTR contains an IRES related to cell cycle control and cancer progression. Gene 2023, 887, 147724. [Google Scholar] [CrossRef]
- Fitzgerald, K.D.; Semler, B.L. Bridging IRES elements in mRNAs to the eukaryotic translation apparatus. Biochim. Biophys. Acta Gene Regul. Mech. 2009, 1789, 518–528. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Chen, Z.; Zhang, M.; Zou, L.; He, S.; Liu, J.; Wu, J. DeepIRES: A hybrid deep learning model for accurate identification of internal ribosome entry sites in cellular and viral mRNAs. Brief. Bioinform. 2024, 25, bbae439. [Google Scholar] [CrossRef]
- López-Ulloa, B.; Fuentes, Y.; Pizarro-Ortega, M.S.; López-Lastra, M. RNA-Binding Proteins as Regulators of Internal Initiation of Viral mRNA Translation. Viruses 2022, 14, 188. [Google Scholar] [CrossRef]
- Andreev, D.E.; Niepmann, M.; Shatsky, I.N. Elusive Trans-Acting Factors Which Operate with Type I (Poliovirus-like) IRES Elements. Int. J. Mol. Sci. 2022, 23, 15497. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Liu, J.; Sun, L.; Zhang, J.; Hou, J. Developing polycistronic expression tool in Yarrowia lipolytica. Synth. Syst. Biotechnol. 2025, 10, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Lin, Q.; Berro, J. 2A peptide from ERBV-1 efficiently separates endogenous protein domains in the fission yeast Schizosaccharomyces pombe. Micropubl. Biol. 2023, 1, 2–7. [Google Scholar] [CrossRef]
- de Lima, J.G.S.; Lanza, D.C.F. 2a and 2a-like sequences: Distribution in different virus species and applications in biotechnology. Viruses 2021, 13, 2160. [Google Scholar] [CrossRef]
- Luke, G.A.; Ryan, M.D. Using the 2A protein coexpression system: Multicistronic 2A vectors expressing gene(s) of interest and reporter proteins. Methods Mol. Biol. 2018, 1755, 31–48. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Yan, F.; Doronina, V.A.; Escuin-Ordinas, H.; Ryan, M.D.; Brown, J.D. 2A peptides provide distinct solutions to driving stop-carry on translational recoding. Nucleic Acids Res. 2012, 40, 3143–3151. [Google Scholar] [CrossRef] [PubMed]
- Luke, G.A.; Ross, L.S.; Lo, Y.; Wu, H.; Ryan, M.D. Picornavirus Evolution: Genomes Encoding Multiple 2ANPGP Sequences—Biomedical and Biotechnological Utility. Viruses 2024, 16, 1587. [Google Scholar] [CrossRef]
- Toporova, V.A.; Argentova, V.V.; Aliev, T.K.; Panina, A.A.; Dolgikh, D.A.; Kirpichnikov, M.P. Optimization of recombinant antibody production based on the vector design and the level of metabolites for generation of Ig- producing stable cell lines. Genet. Eng. Biotechnol. 2023, 21, 23. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Menzel, C.; Meier, D.; Zhang, C.; Dübel, S.; Jostock, T. A comparative study of different vector designs for the mammalian expression of recombinant IgG antibodies. Immunol. Methods 2007, 318, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Fallahee, I.; Hawiger, D. Episomal Vectors for Stable Production of Recombinant Proteins and Engineered Antibodies. Antibodies 2024, 13, 18. [Google Scholar] [CrossRef]
- Kangro, K.; Roose, E.; Vandenbulcke, A.; Dekimpe, C.; Vanhoorelbeke, K. Improvement of recombinant ADAMTS13 production through a more optimal signal peptide or an N-terminal fusion protein. Thromb. Haemost. 2022, 20, 2379–2385. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Han, Z.; Cheng, W.; Niu, S.; Wang, T.; Wang, X. Improvement strategies for transient gene expression in mammalian cells. Appl. Microbiol. Biotechnol. 2024, 108, 480. [Google Scholar] [CrossRef]
- Yang, C.H.; Li, H.C.; Lo, S.Y. Enhancing recombinant antibody yield in Chinese hamster ovary cells. Tzu Chi Med. 2024, 36, 240–250. [Google Scholar] [CrossRef]
- Шaньшин, Д.B.; Щepбaкoв, Д.H.; Hecмeянoвa, B.C.; Иcaeвa, А.А.; Лoктeв, B.Б.; Пpoтoпoпoвa, Е.B. Плaзмиднaя гeнeтичecкaя кoнcтpyкция pVEAL3-10H10ch, штaмм peкoмбинaнтнoй клeтoчнoй линии CHO-K1-10H10ch и химepнoe aнтитeлo 10H10ch пpoтив виpyca клeщeвoгo энцeфaлитa, пpoдyциpyeмoe yкaзaнным штaммoм клeтoчнoй линии CHO-K1-10H10ch. Пaтeнт нa изoбpeтeниe RU 2800471 C1, 21.07.2023. Заявка № 2022126714 oт 13.10.2022. Available online: https://elibrary.ru/item.asp?id=54232012 (accessed on 25 November 2024).
- Li, Y.; Tian, Z.; Xu, D.; Wang, X.; Wang, T. Construction strategies for developing expression vectors for recombinant monoclonal antibody production in CHO cells. Mol. Biol. Rep. 2018, 45, 2907–2912. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.Y.; Guo, X. Expression vector cassette engineering for recombinant therapeutic production in mammalian cell systems. Appl. Microbiol. Biotechnol. 2020, 104, 5673–5688. [Google Scholar] [CrossRef] [PubMed]
- Ho, S.C.; Bardor, M.; Feng, H.; Tong, Y.W.; Song, Z.; Yap, M.G.; Yang, Y. IRES-mediated Tricistronic vectors for enhancing generation of high monoclonal antibody expressing CHO cell lines. Biotechnology 2012, 157, 130–139. [Google Scholar] [CrossRef]
- Shanshin, D.V.; Nesmeyanova, V.S.; Protopopova, E.V.; Shelemba, A.A.; Loktev, V.B.; Shcherbakov, D.N. Preparation and Construction of Chimeric Humanized Broadly Reactive Antibody 10H10 to Protein E of Tick-Borne Encephalitis Virus. Bull. Exp. Biol. Med. 2024, 177, 770–773. [Google Scholar] [CrossRef] [PubMed]
- Haбepeжнoв, Д.C. Cpaвнитeльный aнaлиз пpoмoтopoв, иcпoльзyeмых для экcпpeccии тpaнcгeнoв в клeткaх млeкoпитaющих. Hayкocфepa 2021, 9, 15–21. [Google Scholar] [CrossRef]
- Csató-Kovács, E.; Salamon, P.; Fikó-Lászlo, S.; Kovács, K.; Koka, A.; András-Korodi, M.; Albert, B. Development of a Mammalian Cell Line for Stable Production of Anti-PD-1. Antibodies 2024, 13, 82. [Google Scholar] [CrossRef] [PubMed]
- Bochkov, Y.A.; Palmenberg, A.C. Translational efficiency of EMCV IRES in bicistronic vectors is dependent upon IRES sequence and gene location. Biotechniques 2006, 41, 283–292. [Google Scholar] [CrossRef]
- Wang, X.; Marchisio, M.A. Synthetic polycistronic sequences in eukaryotes. Synth. Syst. Biotechnol. 2021, 6, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Van der Weken, H.; Cox, E.; Devriendt, B. Rapid production of a chimeric antibody-antigen fusion protein based on 2A-peptide cleavage and green fluorescent protein expression in CHO cells. MAbs 2019, 11, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.; Buck, L.; Daris, K.; Welborn, B.; Luo, Q.; Wypych, J. Investigation of the free heavy chain homodimers of a monoclonal antibody. Biotechnol. Prog. 2018, 34, 738–745. [Google Scholar] [CrossRef]
- Borgoyakova, M.B.; Karpenko, L.I.; Rudometov, A.P.; Shanshin, D.V.; Ilyichev, A.A. Immunogenic properties of the DNA construct encoding the receptor-binding domain of the SARS-CoV-2 spike protein. Mol. Biol. 2021, 55, 987–998. [Google Scholar] [CrossRef] [PubMed]
- Ha, T.K.; Kim, D.; Kim, C.L.; Grav, L.M.; Lee, G.M. Factors affecting the quality of therapeutic proteins in recombinant Chinese hamster ovary cell culture. Biotechnol. Adv. 2022, 54, 107831. [Google Scholar] [CrossRef] [PubMed]
- Hussain, H.; Patel, T.; Ozanne, A.M.; Vito, D.; Ellis, M.; Smales, C.M. A comparative analysis of recombinant Fab and full-length antibody production in Chinese hamster ovary cells. Biotechnol. Bioeng. 2021, 118, 4815–4828. [Google Scholar] [CrossRef]
- Choa JB, D.; Sasaki, T.; Kajiura, H.; Ikuta, K.; Fujiyama, K.; Misaki, R. Effects of various disaccharide adaptations on recombinant IgA1 production in CHO-K1 suspension cells. Cytotechnology 2023, 75, 219–229. [Google Scholar] [CrossRef]
- Pirkalkhoran, S.; Grabowska, W.R.; Kashkoli, H.H.; Mirhassani, R.; Guiliano, D.; Dolphin, C.; Khalili, H. Bioengineering of Antibody Fragments: Challenges and Opportunities. Bioengineering 2023, 10, 122. [Google Scholar] [CrossRef]
- Ochmann, M.T.; Ivics, Z. Jumping ahead with sleeping beauty: Mechanistic insights into cut-and-paste transposition. Viruses 2021, 13, 76. [Google Scholar] [CrossRef]
- Ruf, S.; Symmons, O.; Uslu, V.V.; Dolle, D.; Hot, C.; Ettwiller, L.; Spitz, F. Large-scale analysis of the regulatory architecture of the mouse genome with a transposon-associated sensor. Nat. Genet. 2011, 43, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Ivics, Z.; Izsvák, Z.; Medrano, G.; Chapman, K.M.; Hamra, F.K. Sleeping Beauty transposon mutagenesis in rat spermatogonial stem cells. Nat. Protoc. 2011, 6, 1521–1535. [Google Scholar] [CrossRef] [PubMed]
- Protopopova, Y.V.; Khusainova, A.D.; Konovalova, S.N.; Loktev, V.B. Preparation and study of properties of anti-idiotypic antibodies, carrying hemagglutinating paratopes of tick-borne encephalitis virus on their surface. Vopr. Virusol. 1996, 41, 50–53. Available online: https://europepmc.org/article/med/8686271 (accessed on 23 May 2024). [PubMed]
- Frenzel, A.; Hust, M.; Schirrmann, T. Expression of recombinant antibodies. Front. Immunol. 2013, 4, 217. [Google Scholar] [CrossRef] [PubMed]
- Shanshin, D.V.; Borisevich, S.S.; Bondar, A.A.; Porozov, Y.B.; Shcherbakov, D.N. Can Modern Molecular Modeling Methods Help Find the Area of Potential Vulnerability of Flaviviruses? Int. J. Mol. Sci. 2022, 23, 7721. [Google Scholar] [CrossRef]
- Ormundo, L.F.; Barreto, C.T.; Tsuruta, L.R. Development of Therapeutic Monoclonal Antibodies for Emerging Arbovirus Infections. Viruses 2023, 15, 2177. [Google Scholar] [CrossRef] [PubMed]
- Kandari, D.; Bhatnagar, R. Antibody engineering and its therapeutic applications. Int. Rev. Immunol. 2023, 42, 156–183. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nesmeyanova, V.S.; Shanshin, D.V.; Murashkin, D.E.; Shcherbakov, D.N. Construction of an Integration Vector with a Chimeric Signal Peptide for the Expression of Monoclonal Antibodies in Mammalian Cells. Curr. Issues Mol. Biol. 2024, 46, 14464-14475. https://doi.org/10.3390/cimb46120868
Nesmeyanova VS, Shanshin DV, Murashkin DE, Shcherbakov DN. Construction of an Integration Vector with a Chimeric Signal Peptide for the Expression of Monoclonal Antibodies in Mammalian Cells. Current Issues in Molecular Biology. 2024; 46(12):14464-14475. https://doi.org/10.3390/cimb46120868
Chicago/Turabian StyleNesmeyanova, Valentina S., Daniil V. Shanshin, Denis E. Murashkin, and Dmitriy N. Shcherbakov. 2024. "Construction of an Integration Vector with a Chimeric Signal Peptide for the Expression of Monoclonal Antibodies in Mammalian Cells" Current Issues in Molecular Biology 46, no. 12: 14464-14475. https://doi.org/10.3390/cimb46120868
APA StyleNesmeyanova, V. S., Shanshin, D. V., Murashkin, D. E., & Shcherbakov, D. N. (2024). Construction of an Integration Vector with a Chimeric Signal Peptide for the Expression of Monoclonal Antibodies in Mammalian Cells. Current Issues in Molecular Biology, 46(12), 14464-14475. https://doi.org/10.3390/cimb46120868





