A Pilot Study of a Panel of Ocular Inflammation Biomarkers in Patients with Primary Sjögren’s Syndrome
Abstract
1. Introduction
2. Materials and Methods
2.1. Tear Samples
2.2. Antibody Microarray Analysis of the Samples
2.3. Statistical Analysis
3. Results
3.1. Tear Samples
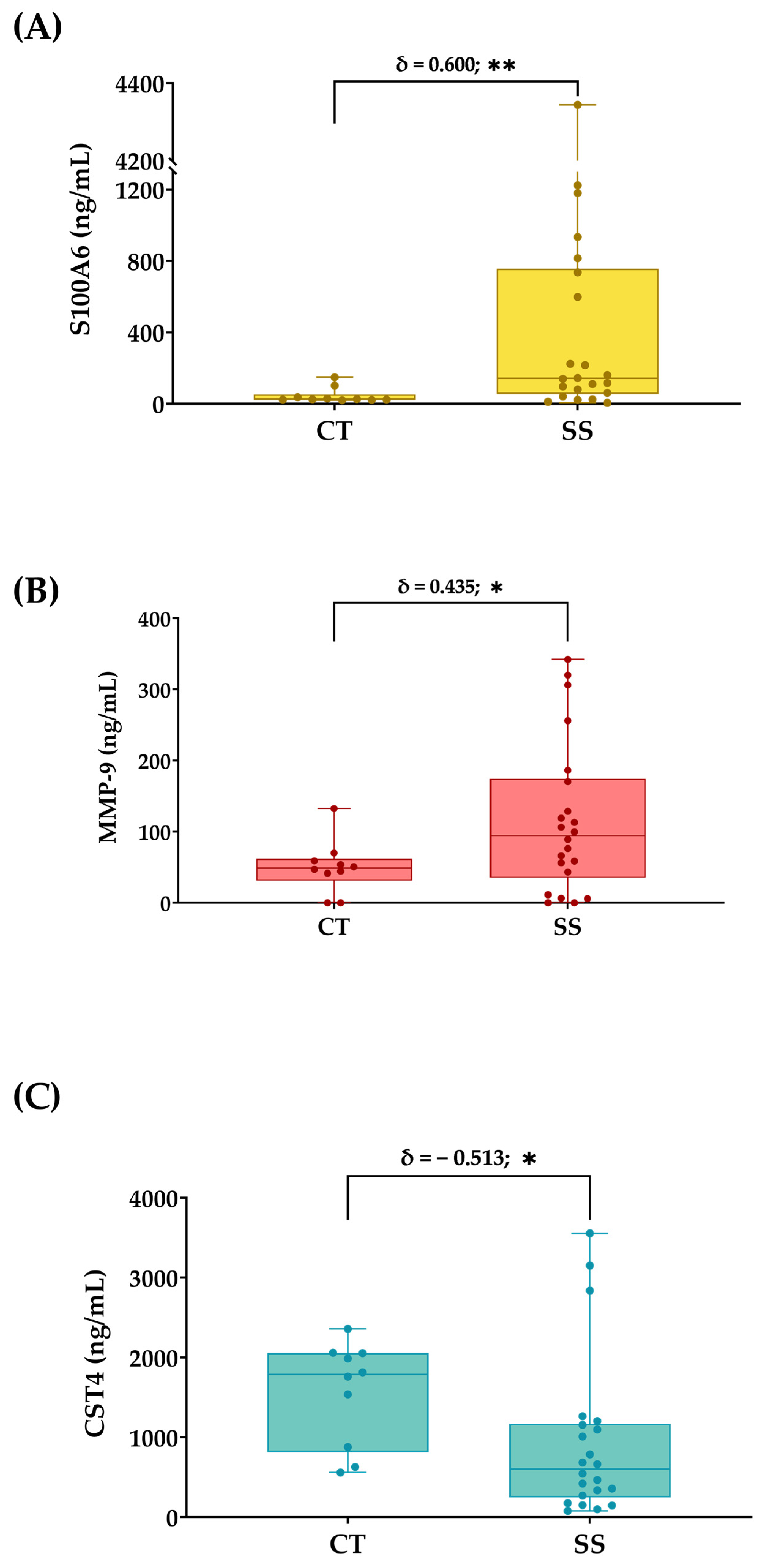
3.2. Antibody Microarray Analysis of Pathological Samples
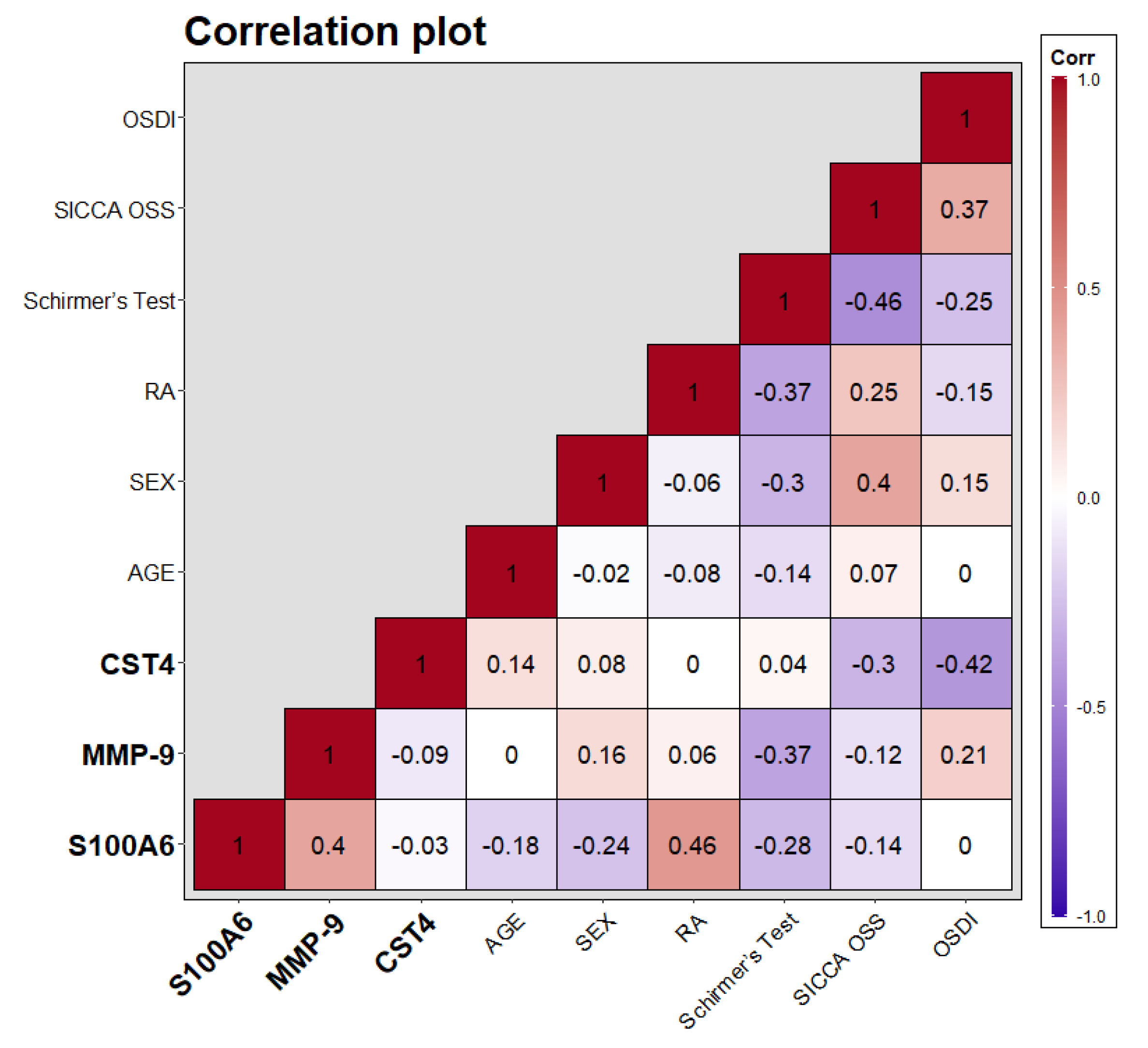
3.3. Relationship between Rheumatoid Arthritis and the Presence of the Biomarkers
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sisto, M.; Ribatti, D.; Lisi, S. Sjögren’s Syndrome-Related Organs Fibrosis: Hypotheses and Realities. J. Clin. Med. 2022, 11, 3551. [Google Scholar] [CrossRef]
- Wallace, D.J.; Daniel, J. The Sjogren’s Book; Oxford University Press: Oxford, UK, 2012; p. 393. [Google Scholar]
- Narváez, J.; Sánchez-Fernández, S.Á.; Seoane-Mato, D.; Díaz-González, F.; Bustabad, S. Prevalence of Sjögren’s Syndrome in the General Adult Population in Spain: Estimating the Proportion of Undiagnosed Cases. Sci. Rep. 2020, 10, 10627. [Google Scholar] [CrossRef] [PubMed]
- Cortes, J.B.; López-Rodríguez, J.A.; Gascón, T.G.; Del Cura-González, M.I.; Domínguez-Berjón, M.F.; Esteban-Vasallo, M.D.; Chalco-Orrego, J.P.; Rabaneda, E.V.; Baldini, C.; Seghieri, C.; et al. Prevalence and Comorbidities of Sjogren’s Syndrome Patients in the Community of Madrid: A Population-Based Cross-Sectional Study. Jt. Bone Spine 2023, 90, 105544. [Google Scholar] [CrossRef] [PubMed]
- Vitali, C.; Minniti, A.; Pignataro, F.; Maglione, W.; Del Papa, N. Management of Sjögren’s Syndrome: Present Issues and Future Perspectives. Front. Med. 2021, 8, 676885. [Google Scholar] [CrossRef]
- Akpek, E.K.; Bunya, V.Y.; Saldanha, I.J. Sjögren’s Syndrome: More Than Just Dry Eye. Cornea 2019, 38, 658–661. [Google Scholar] [CrossRef]
- Wu, K.Y.; Kulbay, M.; Tanasescu, C.; Jiao, B.; Nguyen, B.H.; Tran, S.D. An Overview of the Dry Eye Disease in SjöGren’s Syndrome Using Our Current Molecular Understanding. Int. J. Mol. Sci. 2023, 24, 1580. [Google Scholar] [CrossRef] [PubMed]
- Caban, M.; Omulecki, W.; Latecka-Krajewska, B. Dry Eye in Sjögren’s Syndrome—Characteristics and Therapy. Eur. J. Ophthalmol. 2022, 32, 3174–3184. [Google Scholar] [CrossRef] [PubMed]
- Swaminathan, R. Clinical Enzymology. In Handbook of Clinical Biochemistry; World Scientific: Singapore, 2011; pp. 325–347. ISBN 978-981-283-737-0. [Google Scholar]
- Califf, R.M. Biomarker Definitions and Their Applications. Exp. Biol. Med. 2018, 243, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Martin-Gutierrez, L.; Wilson, R.; Castelino, M.; Jury, E.C.; Ciurtin, C. Multi-Omic Biomarkers for Patient Stratification in Sjogren’s Syndrome-A Review of the Literature. Biomedicines 2022, 10, 1773. [Google Scholar] [CrossRef]
- Jung, J.-Y.; Kim, J.-W.; Kim, H.-A.; Suh, C.-H. Salivary Biomarkers in Patients with Sjögren’s Syndrome-A Systematic Review. Int. J. Mol. Sci. 2021, 22, 12903. [Google Scholar] [CrossRef]
- Lee, H.S.; Jun, J.-H.; Jung, E.-H.; Koo, B.A.; Kim, Y.S. Epigalloccatechin-3-Gallate Inhibits Ocular Neovascularization and Vascular Permeability in Human Retinal Pigment Epithelial and Human Retinal Microvascular Endothelial Cells via Suppression of MMP-9 and VEGF Activation. Molecules 2014, 19, 12150–12172. [Google Scholar] [CrossRef]
- Shetty, R.; Ghosh, A.; Lim, R.R.; Subramani, M.; Mihir, K.; Reshma, A.R.; Ranganath, A.; Nagaraj, S.; Nuijts, R.M.M.A.; Beuerman, R.; et al. Elevated Expression of Matrix Metalloproteinase-9 and Inflammatory Cytokines in Keratoconus Patients Is Inhibited by Cyclosporine A. Investig. Ophthalmol. Vis. Sci. 2015, 56, 738–750. [Google Scholar] [CrossRef] [PubMed]
- Lima, T.B.; Ribeiro, A.P.; da Conceição, L.F.; Bandarra, M.; Manrique, W.G.; Laus, J.L. Ketorolac Eye Drops Reduce Inflammation and Delay Re-Epithelization in Response to Corneal Alkali Burn in Rabbits, without Affecting INOS or MMP-9. Arq. Bras. Oftalmol. 2015, 78, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Trujillo Piso, D.Y.; Ribeiro, A.P.; Silva, M.L.; Guimarães, P.J.; Morales, A.; Martins, B.C.; Padua, I.M.; Renzo, R.; Andrade, A.L.; Uscátegui, R.R.; et al. Effects of Antiproteolytic Agents on Corneal Epithelial Viability and Matrix Metalloproteinase-2 and Metalloproteinase-9 Activity in Alkali-Burned Corneas of Rats. Vet. Ophthalmol. 2014, 17, 23–31. [Google Scholar] [CrossRef]
- Laihia, J.; Kaarniranta, K. Trehalose for Ocular Surface Health. Biomolecules 2020, 10, 809. [Google Scholar] [CrossRef]
- de Paiva, C.S.; Corrales, R.M.; Villarreal, A.L.; Farley, W.J.; Li, D.Q.; Stern, M.E.; Pflugfelder, S.C. Corticosteroid and Doxycycline Suppress MMP-9 and Inflammatory Cytokine Expression, MAPK Activation in the Corneal Epithelium in Experimental Dry Eye. Exp. Eye Res. 2006, 83, 526–535. [Google Scholar] [CrossRef]
- Silva, M.L.; Ribeiro, A.P.; Silva, G.A.; Sanchez, I.X.B.; Renzo, R.; Uscategui, R.; Lima, T.B.; Aldrovani, M.; Laus, J.L. Expressions of Matrix Metalloproteinases-1 and -9 and Opioid Growth Factor in Rabbit Cornea after Lamellar Keratectomy and Treatment with 1% Nalbuphine. Arq. Bras. Oftalmol. 2015, 78, 141–145. [Google Scholar] [CrossRef] [PubMed]
- Dursun, D.; Kim, M.C.; Solomon, A.; Pflugfelder, S.C. Treatment of Recalcitrant Recurrent Corneal Erosions With Inhibitors of Matrix Metalloproteinase-9, Doxycycline and Corticosteroids. Am. J. Ophthalmol. 2001, 132, 8–13. [Google Scholar] [CrossRef]
- Pinto-Fraga, J.; Enríquez-de-Salamanca, A.; Calonge, M.; González-García, M.J.; López-Miguel, A.; López-de la Rosa, A.; García-Vázquez, C.; Calder, V.; Stern, M.E.; Fernández, I. Severity, Therapeutic, and Activity Tear Biomarkers in Dry Eye Disease: An Analysis from a Phase III Clinical Trial. Ocul. Surf. 2018, 16, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, H.; Yaraee, R.; Faghihzadeh, S.; Ghassemi-Broumand, M.; Mahmoudi, M.; Babaei, M.; Naderi, M.; Safavi, M.; Ghazanfari, Z.; Rastin, M.; et al. Tear and Serum MMP-9 and Serum TIMPs Levels in the Severe Sulfur Mustard Eye Injured Exposed Patients. Int. Immunopharmacol. 2019, 77, 105812. [Google Scholar] [CrossRef]
- Gomathinayagam, R.; Ponnalagu, M.; Prajna, N.; Lalitha, P.; Muthukkaruppan, V. Matrix Metalloproteinases (MMP-8, MMP-9) and the Tissue Inhibitors of Metalloproteinases (TIMP-1, TIMP-2) in Patients With Fungal Keratitis. Cornea 2007, 26, 207–211. [Google Scholar] [CrossRef]
- Singh, A.; Maurya, O.P.S.; Jagannadhan, M.v.; Patel, A. Matrix Metalloproteinases (MMP-2 and MMP-9) Activity in Corneal Ulcer and Ocular Surface Disorders Determined by Gelatin Zymography. J. Ocul. Biol. Dis. Inform. 2012, 5, 31–35. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, S.; He, Y.; Yao, B.; Zhang, Y. Regulation of Matrix Metalloproteinases 2 and 9 in Corneal Neovascularization. Chem. Biol. Drug Des. 2020, 95, 485–492. [Google Scholar] [CrossRef]
- Ranganath, A.; Shetty, R.; D’Souza, S.; Wadia, K.; Das, D.; Ghosh, A. Matrix Metalloproteinase-9 drives disease progression of Keratoconus. Investig. Ophthalmol. Vis. Sci. 2013, 54, 4070. [Google Scholar]
- Acera, A.; Rocha, G.; Vecino, E.; Lema, I.; Durán, J.A. Inflammatory Markers in the Tears of Patients with Ocular Surface Disease. Ophthalmic Res. 2008, 40, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Kook, K.Y.; Jin, R.; Li, L.; Yoon, J.; Yoon, K.C. Tear Osmolarity and Matrix Metallopeptidase-9 in Dry Eye Associated with Sjögren’s Syndrome. Korean J. Ophthalmol. 2020, 34, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Jamerson, E.C.; Elhusseiny, A.M.; ElSheikh, R.H.; Eleiwa, T.K.; el Sayed, Y.M. Role of Matrix Metalloproteinase 9 in Ocular Surface Disorders. Eye Contact Lens 2020, 46 (Suppl. 2), S57–S63. [Google Scholar] [CrossRef] [PubMed]
- Soria, J.; Acera, A.; Merayo-Lloves, J.; Durán, J.A.; González, N.; Rodriguez, S.; Bistolas, N.; Schumacher, S.; Bier, F.F.; Peter, H.; et al. Tear Proteome Analysis in Ocular Surface Diseases Using Label-Free LC-MS/MS and Multiplexed-Microarray Biomarker Validation. Sci. Rep. 2017, 7, 17478. [Google Scholar] [CrossRef]
- Donato, R.; Sorci, G.; Giambanco, I. S100A6 Protein: Functional Roles. Cell. Mol. Life Sci. 2017, 74, 2749–2760. [Google Scholar] [CrossRef]
- Turk, V.; Stoka, V.; Turk, D.; Institute, J.S. Cystatins: Biochemical and Structural Properties, and Medical Relevance. Front. Biosci. Landmark 2008, 13, 5406–5420. [Google Scholar] [CrossRef]
- Tamhane, M.; Cabrera-Ghayouri, S.; Abelian, G.; Viswanath, V. Review of Biomarkers in Ocular Matrices: Challenges and Opportunities. Pharm. Res. 2019, 36, 40. [Google Scholar] [CrossRef] [PubMed]
- Shiboski, S.C.; Shiboski, C.H.; Criswell, L.A.; Baer, A.N.; Challacombe, S.; Lanfranchi, H.; Schiødt, M.; Umehara, H.; Vivino, F.; Zhao, Y.; et al. American College of Rheumatology Classification Criteria for Sjögren’s Syndrome: A Data-Driven, Expert Consensus Approach in the Sjögren’s International Collaborative Clinical Alliance Cohort. Arthritis Care Res. 2012, 64, 475–487. [Google Scholar] [CrossRef] [PubMed]
- Acera, A.; Vecino, E.; Duran, J.A. Tear MMP-9 Levels as a Marker of Ocular Surface Inflammation in Conjunctivochalasis. Investig. Ophthalmol. Vis. Sci. 2013, 54, 8285–8291. [Google Scholar] [CrossRef]
- Acera, A.; Suárez, T.; Rodríguez-Agirretxe, I.; Vecino, E.; Durán, J.A. Changes in Tear Protein Profile in Patients with Conjunctivochalasis. Cornea 2011, 30, 42–49. [Google Scholar] [CrossRef] [PubMed]
- de la Fuente, M.; Rodríguez-Agirretxe, I.; Vecino, E.; Astigarraga, E.; Acera, A.; Barreda-Gómez, G. Elevation of Tear MMP-9 Concentration as a Biomarker of Inflammation in Ocular Pathology by Antibody Microarray Immunodetection Assays. Int. J. Mol. Sci. 2022, 23, 5639. [Google Scholar] [CrossRef] [PubMed]
- Manuel Benitez-Del-Castillo, J.; Soria, J.; Acera, A.; María Muñoz, A.; Rodríguez, S.; Suárez, T. Quantification of a Panel for Dry-Eye Protein Biomarkers in Tears: A Comparative Pilot Study Using Standard ELISA and Customized Microarrays. Mol. Vis. 2021, 27, 243. [Google Scholar]
- Whitcher, J.P.; Shiboski, C.H.; Shiboski, S.C.; Heidenreich, A.M.; Kitagawa, K.; Zhang, S.; Hamann, S.; Larkin, G.; McNamara, N.A.; Greenspan, J.S.; et al. A Simplified Quantitative Method for Assessing Keratoconjunctivitis Sicca From the Sjögren’s Syndrome International Registry. Am. J. Ophthalmol. 2010, 149, 405–415. [Google Scholar] [CrossRef]
- Grubbs, J.R.; Tolleson-Rinehart, S.; Huynh, K.; Davis, R.M. A Review of Quality of Life Measures in Dry Eye Questionnaires. Cornea 2014, 33, 215–218. [Google Scholar] [CrossRef]
- Brandt, J.E.; Priori, R.; Valesini, G.; Fairweather, D. Sex Differences in Sjögren’s Syndrome: A Comprehensive Review of Immune Mechanisms. Biol. Sex Differ. 2015, 6, 19. [Google Scholar] [CrossRef]
- Korpole, N.; Kurada, P.; Korpole, M. Gender Difference in Ocular Diseases, Risk Factors and Management with Specific Reference to Role of Sex Steroid Hormones. J. Midlife Health 2022, 13, 20–25. [Google Scholar] [CrossRef]
- Patel, R.; Shahane, A. The Epidemiology of Sjögren’s Syndrome. Clin. Epidemiol. 2014, 6, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Soria, J.; Durán, J.A.; Etxebarria, J.; Merayo, J.; González, N.; Reigada, R.; García, I.; Acera, A.; Suárez, T. Tear Proteome and Protein Network Analyses Reveal a Novel Pentamarker Panel for Tear Film Characterization in Dry Eye and Meibomian Gland Dysfunction. J. Proteom. 2013, 78, 94–112. [Google Scholar] [CrossRef] [PubMed]
- Tsai, P.S.; Evans, J.E.; Green, K.M.; Sullivan, R.M.; Schaumberg, D.A.; Richards, S.M.; Dana, M.R.; Sullivan, D.A. Proteomic Analysis of Human Meibomian Gland Secretions. Br. J. Ophthalmol. 2006, 90, 372–377. [Google Scholar] [CrossRef]
- Liu, S.; Richards, S.M.; Lo, K.; Hatton, M.; Fay, A.; Sullivan, D.A. Changes in Gene Expression in Human Meibomian Gland Dysfunction. Investig. Ophthalmol. Vis. Sci. 2011, 52, 2727–2740. [Google Scholar] [CrossRef]
- Fábián, T.K.; Hermann, P.; Beck, A.; Fejérdy, P.; Fábián, G. Salivary Defense Proteins: Their Network and Role in Innate and Acquired Oral Immunity. Int. J. Mol. Sci. 2012, 13, 4295–4320. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, S.; Thangavelu, M.; Zhang, L.; Green, K.B.; Nichols, K.K. ITRAQ Quantitative Proteomics in the Analysis of Tears in Dry Eye Patients. Investig. Ophthalmol. Vis. Sci. 2012, 53, 5052–5059. [Google Scholar] [CrossRef]
- Koo, B.S.; Lee, D.Y.; Ha, H.S.; Kim, J.C.; Kim, C.W. Comparative Analysis of the Tear Protein Expression in Blepharitis Patients Using Two-Dimensional Electrophoresis. J. Proteome Res. 2005, 4, 719–724. [Google Scholar] [CrossRef]
- Willcox, M.D.P.; Argüeso, P.; Georgiev, G.A.; Holopainen, J.M.; Laurie, G.W.; Millar, T.J.; Papas, E.B.; Rolland, J.P.; Schmidt, T.A.; Stahl, U.; et al. TFOS DEWS II Tear Film Report. Ocul. Surf. 2017, 15, 366–403. [Google Scholar] [CrossRef]
- Li, B.; Sheng, M.; Li, J.; Yan, G.; Lin, A.; Li, M.; Wang, W.; Chen, Y. Tear Proteomic Analysis of Sjögren Syndrome Patients with Dry Eye Syndrome by Two-Dimensional-Nano-Liquid Chromatography Coupled with Tandem Mass Spectrometry. Sci. Rep. 2014, 4, srep05772. [Google Scholar] [CrossRef]
- Macbeth, G.; Razumiejczyk, E.; Ledesma, R.D. Cliff’s Delta Calculator: A Non-Parametric Effect Size Program for Two Groups of Observations. Univ. Psychol. 2011, 10, 545–555. [Google Scholar] [CrossRef]
- Beckman, K.A.; Luchs, J.; Milner, M.S. Making the Diagnosis of Sjögren’s Syndrome in Patients with Dry Eye. Clin. Ophthalmol. 2016, 10, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Aletaha, D.; Neogi, T.; Silman, A.J.; Funovits, J.; Felson, D.T.; Bingham, C.O.; Birnbaum, N.S.; Burmester, G.R.; Bykerk, V.P.; Cohen, M.D.; et al. 2010 Rheumatoid Arthritis Classification Criteria: An American College of Rheumatology/European League Against Rheumatism Collaborative Initiative. Arthritis Rheum. 2010, 62, 2569–2581. [Google Scholar] [CrossRef] [PubMed]
- Lazarus, M.N.; Isenberg, D.A. Development of Additional Autoimmune Diseases in a Population of Patients with Primary Sjögren’s Syndrome. Ann. Rheum. Dis. 2005, 64, 1062–1064. [Google Scholar] [CrossRef]
- Bell, D.A. Can We Rely on Anti-Citrulline Antibody Determination for the Diagnosis of Early Rheumatoid Arthritis? J. Rheumatol. 2006, 33, 2369–2371. [Google Scholar] [PubMed]
- Theander, E.; Jonsson, R.; Sjöström, B.; Brokstad, K.; Olsson, P.; Henriksson, G. Prediction of Sjögren’s Syndrome Years Before Diagnosis and Identification of Patients With Early Onset and Severe Disease Course by Autoantibody Profiling. Arthritis Rheumatol. 2015, 67, 2427–2436. [Google Scholar] [CrossRef]
- Jonsson, R.; Theander, E.; Sjöström, B.; Brokstad, K.; Henriksson, G. Autoantibodies Present before Symptom Onset in Primary Sjögren Syndrome. JAMA 2013, 310, 1854–1855. [Google Scholar] [CrossRef]
- Martinez-Prat, L.; Nissen, M.J.; Lamacchia, C.; Bentow, C.; Cesana, L.; Roux-Lombard, P.; Gabay, C.; Mahler, M. Comparison of Serological Biomarkers in Rheumatoid Arthritis and Their Combination to Improve Diagnostic Performance. Front. Immunol. 2018, 9, 1113. [Google Scholar] [CrossRef]
- Aggarwal, R.; Liao, K.; Nair, R.; Ringold, S.; Costenbader, K.H. Anti-Citrullinated Peptide Antibody (ACPA) Assays and Their Role in the Diagnosis of Rheumatoid Arthritis. Arthritis Rheum. 2009, 61, 1472–1483. [Google Scholar] [CrossRef]
- Matsui, T.; Shimada, K.; Ozawa, N.; Hayakawa, H.; Hagiwara, F.; Nakayama, H.; Sugii, S.; Ozawa, Y.; Tohma, S. Diagnostic Utility of Anti-Cyclic Citrullinated Peptide Antibodies for Very Early Rheumatoid Arthritis. J. Rheumatol. 2006, 33, 2390–2397. [Google Scholar]
- Huttenlocher, A.; Smith, J.A. Neutrophils in Pediatric Autoimmune Disease. Curr. Opin. Rheumatol. 2015, 27, 500–504. [Google Scholar] [CrossRef]
- Khandpur, R.; Carmona-Rivera, C.; Vivekanandan-Giri, A.; Gizinski, A.; Yalavarthi, S.; Knight, J.S.; Friday, S.; Li, S.; Patel, R.M.; Subramanian, V.; et al. NETs Are a Source of Citrullinated Autoantigens and Stimulate Inflammatory Responses in Rheumatoid Arthritis. Sci. Transl. Med. 2013, 5, 178ra40. [Google Scholar] [CrossRef] [PubMed]
- Foell, D.; Wittkowski, H.; Hammerschmidt, I.; Wulffraat, N.; Schmeling, H.; Frosch, M.; Horneff, G.; Kuis, W.; Sorg, C.; Roth, J. Monitoring Neutrophil Activation in Juvenile Rheumatoid Arthritis by S100A12 Serum Concentrations. Arthritis Rheum. 2004, 50, 1286–1295. [Google Scholar] [CrossRef] [PubMed]
- Gerss, J.; Roth, J.; Holzinger, D.; Ruperto, N.; Wittkowski, H.; Frosch, M.; Wulffraat, N.; Wedderburn, L.; Stanevicha, V.; Mihaylova, D.; et al. Phagocyte-Specific S100 Proteins and High-Sensitivity C Reactive Protein as Biomarkers for a Risk-Adapted Treatment to Maintain Remission in Juvenile Idiopathic Arthritis: A Comparative Study. Ann. Rheum. Dis. 2012, 71, 1991–1997. [Google Scholar] [CrossRef] [PubMed]
- Leśniak, W.; Filipek, A. S100A6 Protein-Expression and Function in Norm and Pathology. Int. J. Mol. Sci. 2023, 24, 1341. [Google Scholar] [CrossRef] [PubMed]
- Ometto, F.; Friso, L.; Astorri, D.; Botsios, C.; Raffeiner, B.; Punzi, L.; Doria, A. Calprotectin in Rheumatic Diseases. Exp. Biol. Med. 2017, 242, 859–873. [Google Scholar] [CrossRef] [PubMed]
- Bettner, L.F.; Peterson, R.A.; Bergstedt, D.T.; Kelmenson, L.B.; Demoruelle, M.K.; Mikuls, T.R.; Edison, J.D.; Parish, M.C.; Feser, M.L.; Frazer-Abel, A.A.; et al. Combinations of Anticyclic Citrullinated Protein Antibody, Rheumatoid Factor, and Serum Calprotectin Positivity Are Associated With the Diagnosis of Rheumatoid Arthritis Within 3 Years. ACR Open Rheumatol. 2021, 3, 684–689. [Google Scholar] [CrossRef]
- Ananthi, S.; Venkatesh Prajna, N.; Lalitha, P.; Valarnila, M.; Dharmalingam, K. Pathogen Induced Changes in the Protein Profile of Human Tears from Fusarium Keratitis Patients. PLoS ONE 2013, 8, e53018. [Google Scholar] [CrossRef]
- Martini, D.; Gallo, A.; Vella, S.; Sernissi, F.; Cecchettini, A.; Luciano, N.; Polizzi, E.; Conaldi, P.G.; Mosca, M.; Baldini, C. Cystatin S-a Candidate Biomarker for Severity of Submandibular Gland Involvement in Sjögren’s Syndrome. Rheumatology 2017, 56, 1031–1038. [Google Scholar] [CrossRef]
- Zaleska-Żmijewska, A.; Strzemecka, E.; Wawrzyniak, Z.M.; Szaflik, J.P. Extracellular MMP-9-Based Assessment of Ocular Surface Inflammation in Patients with Primary Open-Angle Glaucoma. J. Ophthalmol. 2019, 2019, 1240537. [Google Scholar] [CrossRef]
- Barabino, S. A Narrative Review of Current Understanding and Classification of Dry Eye Disease with New Insights on the Impact of Dry Eye during the COVID-19 Pandemic. Ophthalmol. Ther. 2021, 10, 495–507. [Google Scholar] [CrossRef]
- Assi, L.; Chamseddine, F.; Ibrahim, P.; Sabbagh, H.; Rosman, L.; Congdon, N.; Evans, J.; Ramke, J.; Kuper, H.; Burton, M.J.; et al. A Global Assessment of Eye Health and Quality of Life a Systematic Review of Systematic Reviews. JAMA Ophthalmol. 2021, 139, 526–541. [Google Scholar] [CrossRef] [PubMed]
- Knudtson, M.D.; Klein, B.E.K.; Klein, R.; Cruickshanks, K.J.; Lee, K.E. Age-Related Eye Disease, Quality of Life, and Functional Activity. Arch. Ophthalmol. 2005, 123, 807–814. [Google Scholar] [CrossRef] [PubMed]
- Desmet, C.; Blum, L.J.; Marquette, C.A. Multiplex Microarray ELISA versus Classical ELISA, a Comparison Study of Pollutant Sensing for Environmental Analysis. Environ. Sci. Process. Impacts 2013, 15, 1876–1882. [Google Scholar] [CrossRef] [PubMed]
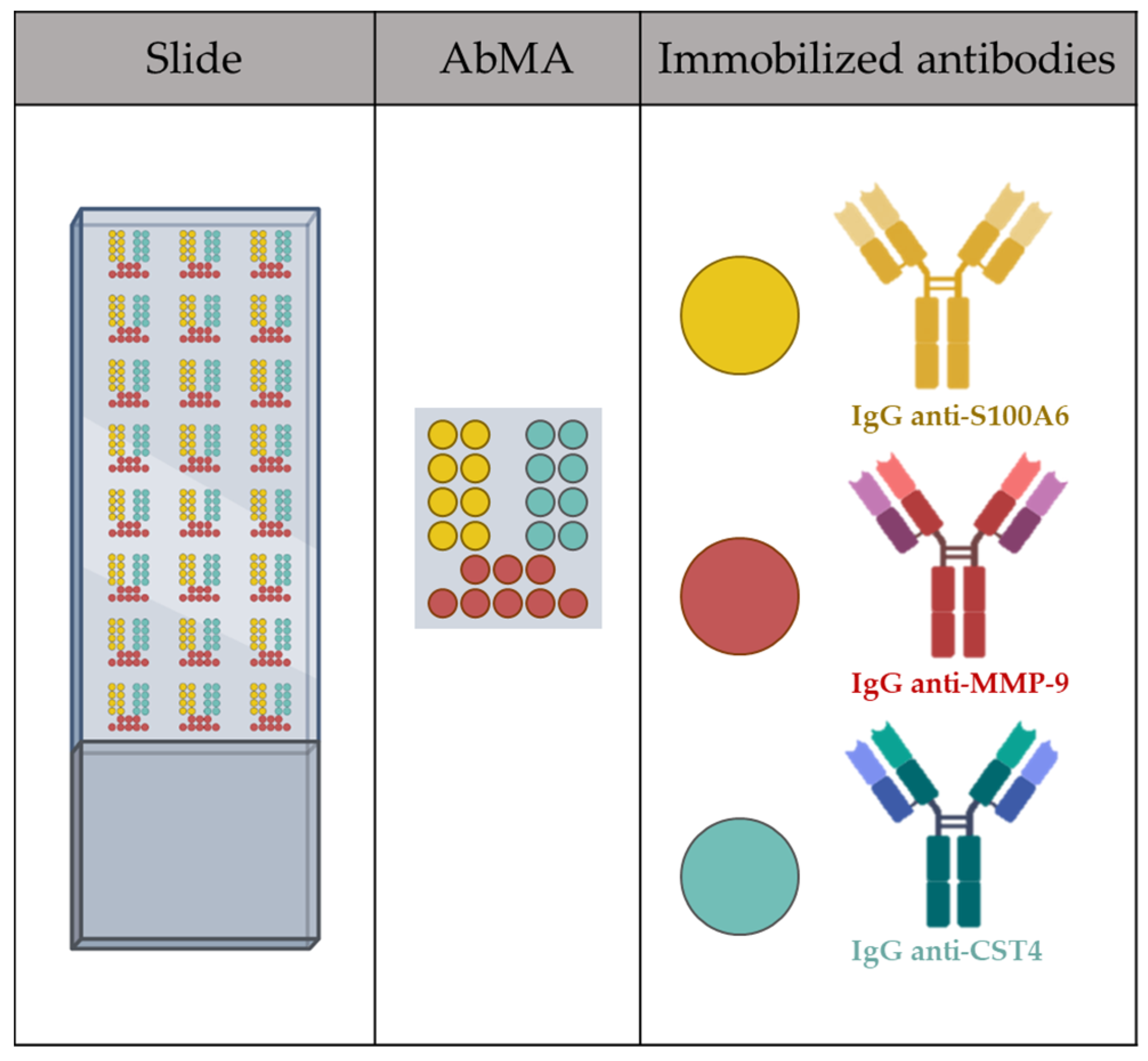
| Variables, Units | SS | CT |
|---|---|---|
| n | 22 | 10 |
| Age, years | 59.2 (13.4) | 54.2 (10.5) |
| Sex (F/M), % | 90.9/9.1 | 60.0/40.0 |
| RA (Y/N), % | 18.2/81.8 | 0.0/100.0 |
| Schirmer’s Test, mm | 4.5 (4.6) | 11.5 (3.3) |
| SICCA OSS, score | 5.9 (3.4) | 0.0 (0.0) |
| OSDI, score | 37.8 (33.2) | 2.4 (2.9) |
| S100A6, ng/mL | 513.0 (941.7) | 45.4 (43.9) |
| MMP-9, ng/mL | 116.6 (106.0) | 47.3 (37.4) |
| CST4, ng/mL | 930.3 (995.8) | 1564.1 (645.3) |
| Variables | p-Value | Fold-Change |
|---|---|---|
| S100A6 | 0.0071 | 10.31 |
| MMP-9 | 0.0462 | 1.46 |
| CST4 | 0.0155 | −0.41 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boto de los Bueis, A.; de la Fuente, M.; Montejano-Milner, R.; del Hierro Zarzuelo, A.; Vecino, E.; Acera, A. A Pilot Study of a Panel of Ocular Inflammation Biomarkers in Patients with Primary Sjögren’s Syndrome. Curr. Issues Mol. Biol. 2023, 45, 2881-2894. https://doi.org/10.3390/cimb45040188
Boto de los Bueis A, de la Fuente M, Montejano-Milner R, del Hierro Zarzuelo A, Vecino E, Acera A. A Pilot Study of a Panel of Ocular Inflammation Biomarkers in Patients with Primary Sjögren’s Syndrome. Current Issues in Molecular Biology. 2023; 45(4):2881-2894. https://doi.org/10.3390/cimb45040188
Chicago/Turabian StyleBoto de los Bueis, Ana, Miguel de la Fuente, Rafael Montejano-Milner, Almudena del Hierro Zarzuelo, Elena Vecino, and Arantxa Acera. 2023. "A Pilot Study of a Panel of Ocular Inflammation Biomarkers in Patients with Primary Sjögren’s Syndrome" Current Issues in Molecular Biology 45, no. 4: 2881-2894. https://doi.org/10.3390/cimb45040188
APA StyleBoto de los Bueis, A., de la Fuente, M., Montejano-Milner, R., del Hierro Zarzuelo, A., Vecino, E., & Acera, A. (2023). A Pilot Study of a Panel of Ocular Inflammation Biomarkers in Patients with Primary Sjögren’s Syndrome. Current Issues in Molecular Biology, 45(4), 2881-2894. https://doi.org/10.3390/cimb45040188










