The Effect of a Diet Enriched with Jerusalem artichoke, Inulin, and Fluoxetine on Cognitive Functions, Neurogenesis, and the Composition of the Intestinal Microbiota in Mice
Abstract
1. Introduction
2. Materials and Methods
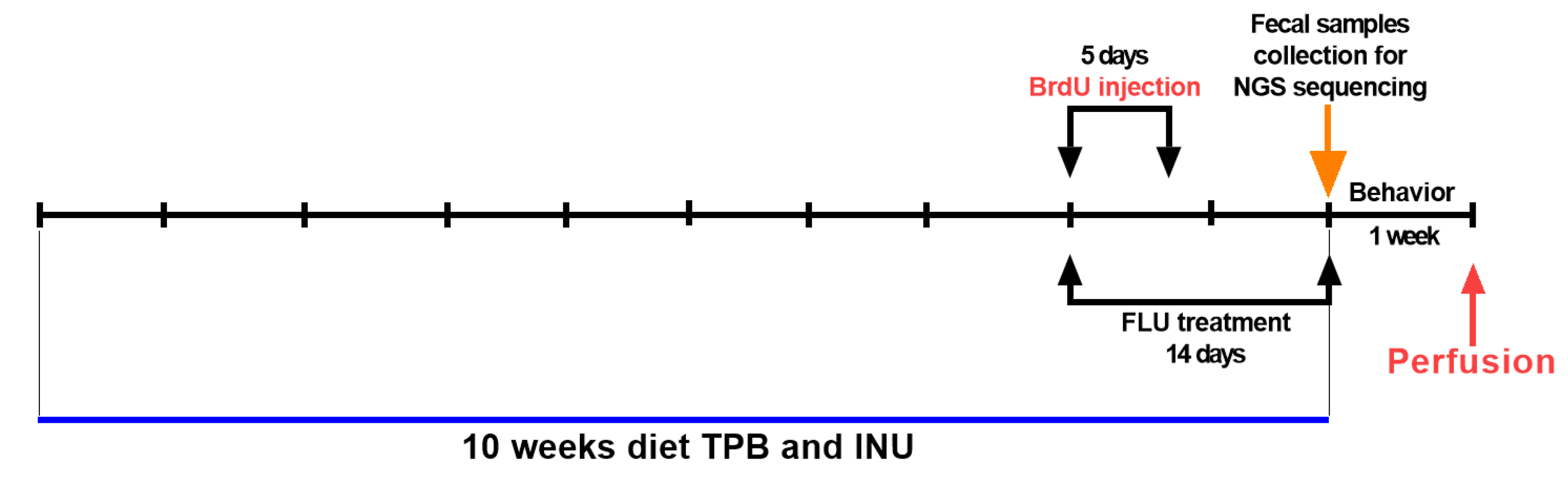
2.1. Animals and Experimental Conditions
2.2. Drugs
2.3. Drugs Administration
- TPB
- INU
- FLU
- control group (water for injection)
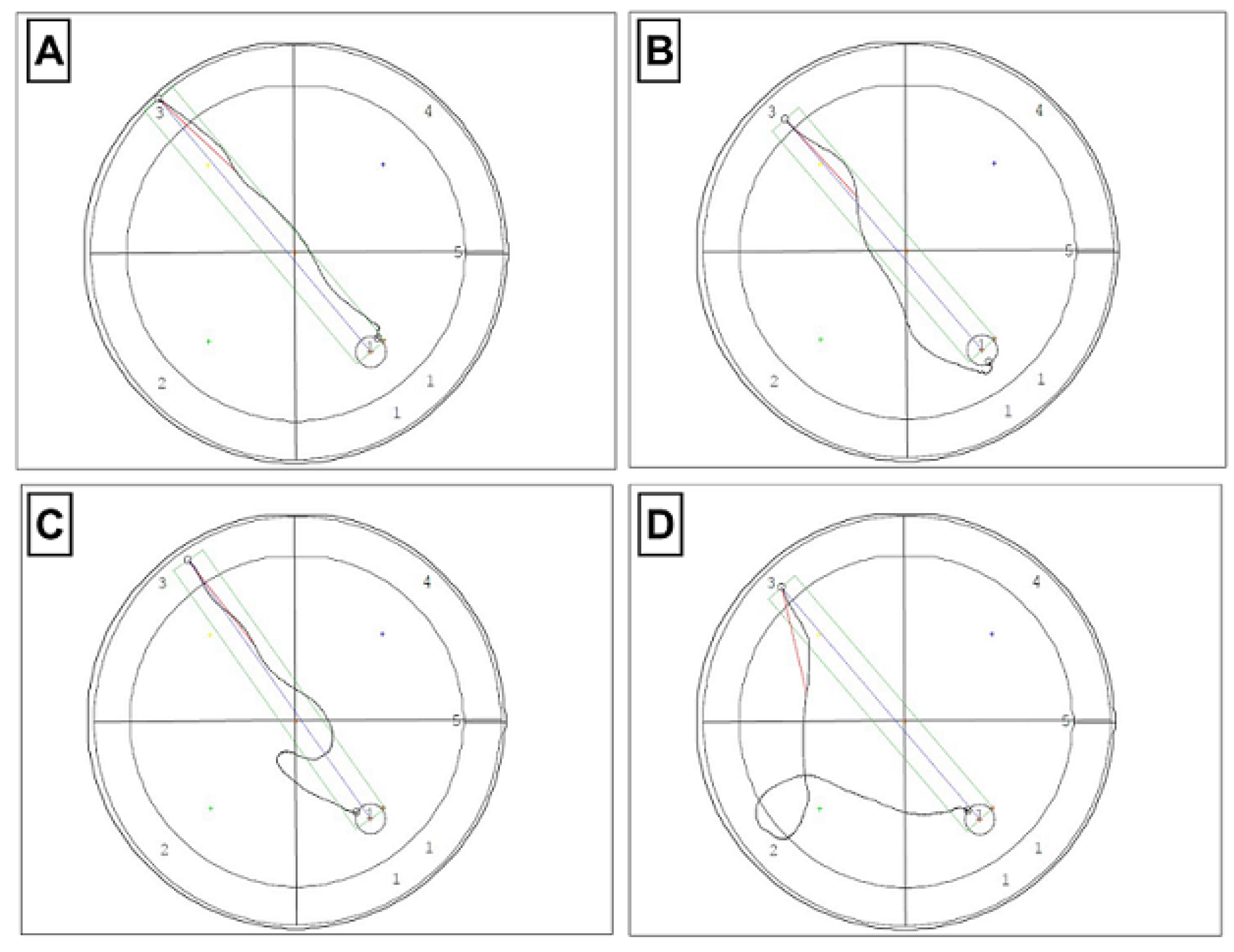
2.4. Morris Water Maze (MWM) Test
2.5. Fecal Collection, DNA Extraction, and NGS Sequencing
Biographical Analysis of Microorganisms
2.6. Transcardial Perfusion and Brain Slice Preparation
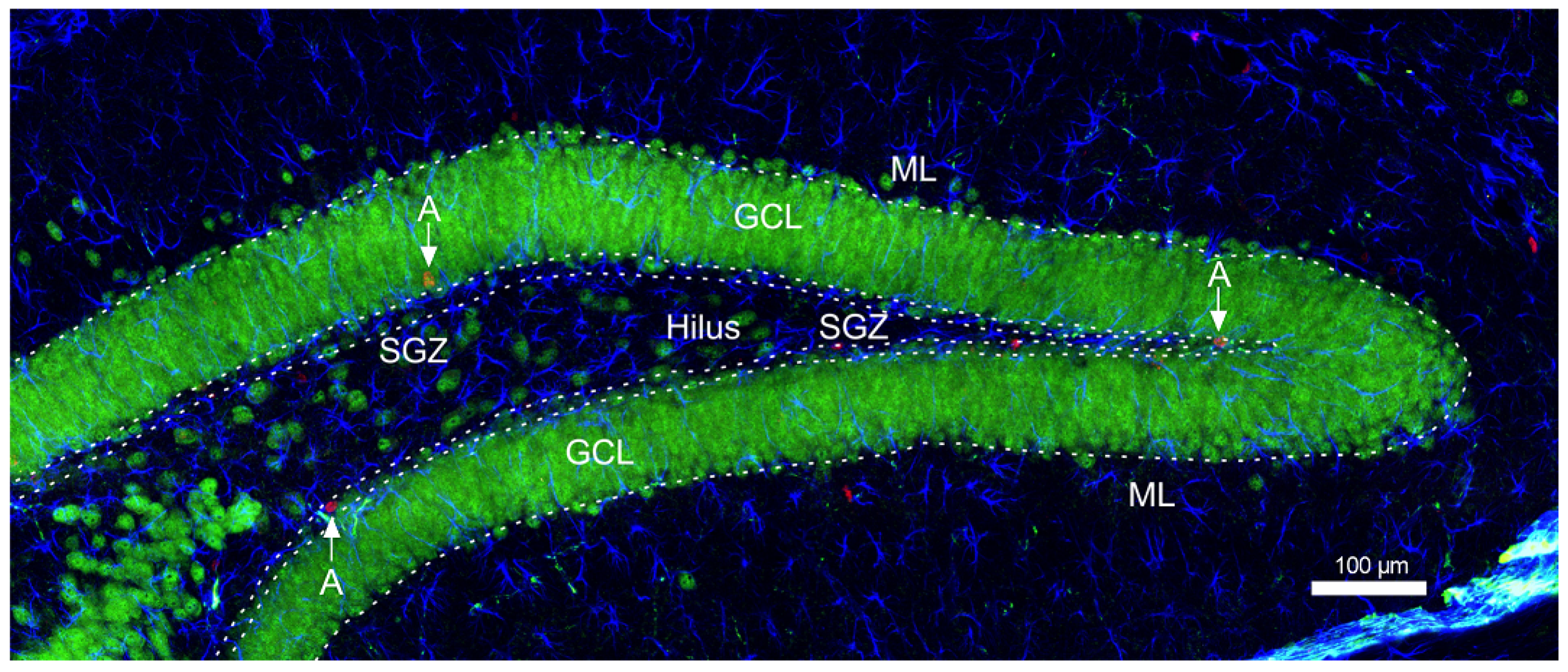
2.7. Immunohistochemical Staining-Neurogenesis
2.8. Confocal Microscopy and Cell Counting
2.9. Statistical Analysis of the Results
2.9.1. Statistical Analysis of MWM and Neurogenesis Results
2.9.2. Principal Coordinate Analysis (PCoA)
2.9.3. ANOSIM and PERMANOVA Analysis
2.9.4. SIMPER Analysis
2.9.5. LEfSe Analysis
3. Results
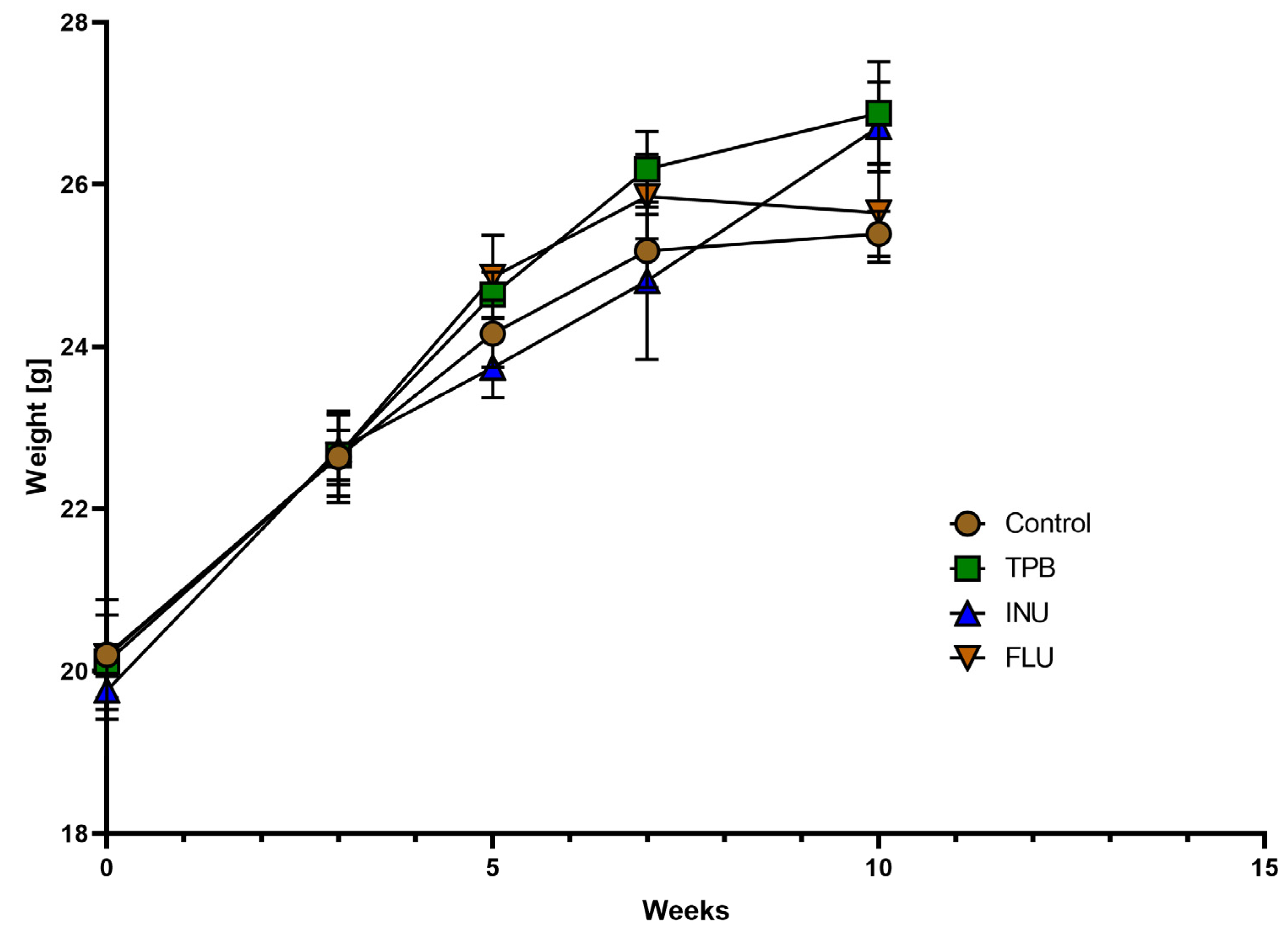
3.1. Effect of TPB, INU, and FLU Administration on the Body Weight of Healthy Mice
3.2. Effect of TPB, INU, and FLU Administration on Mouse Spatial Learning and Memory
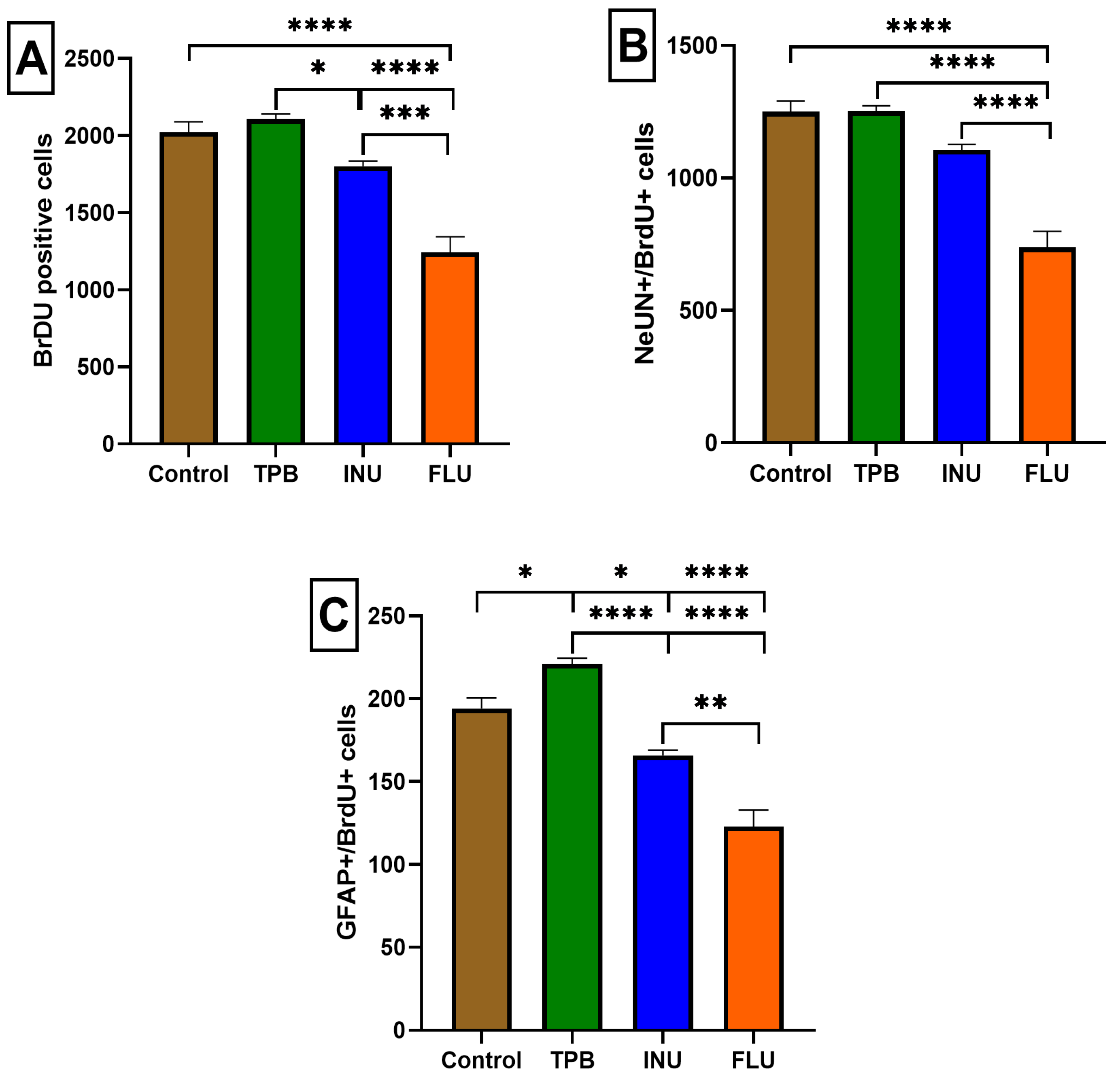
3.3. The Impact of Long-Term Treatment with TPB, INU and FLU on the Neurogenesis in the SGZ and GCL in Mice
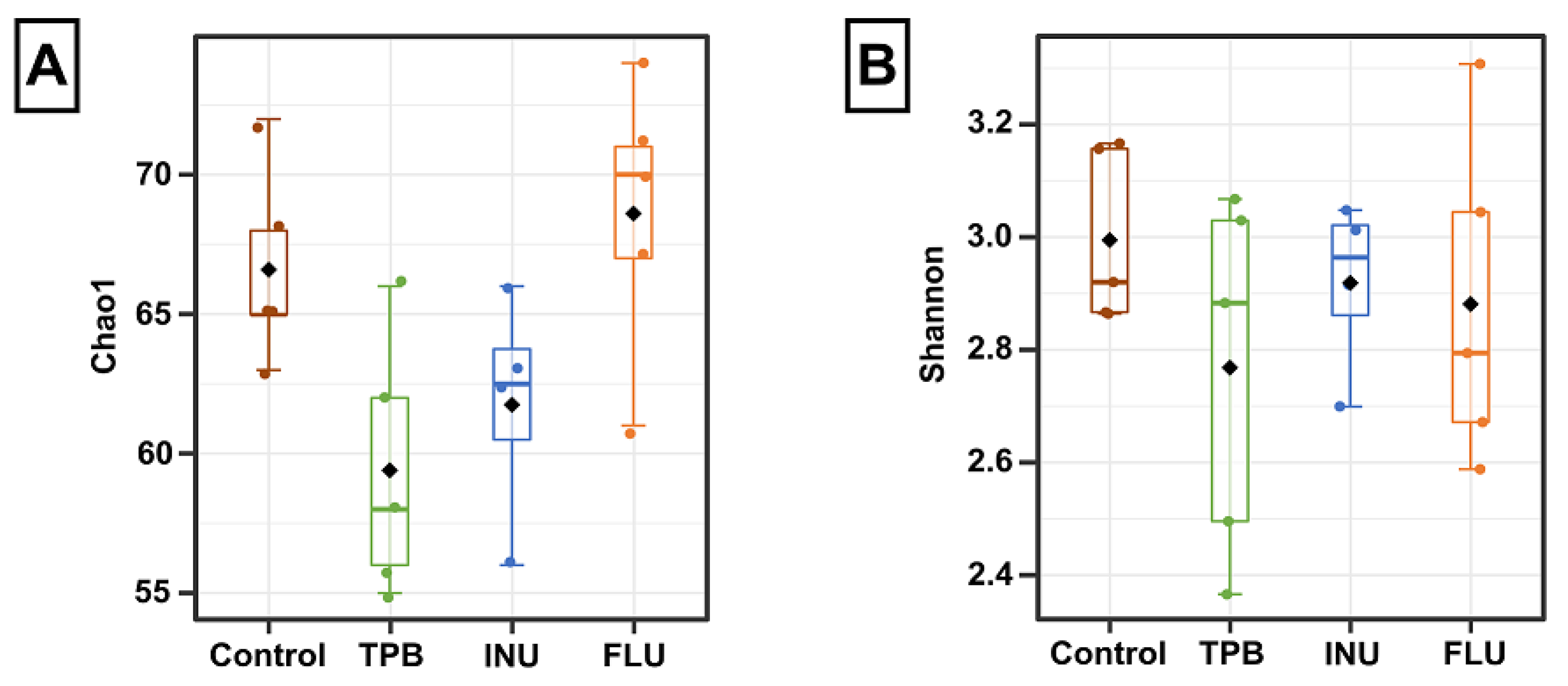
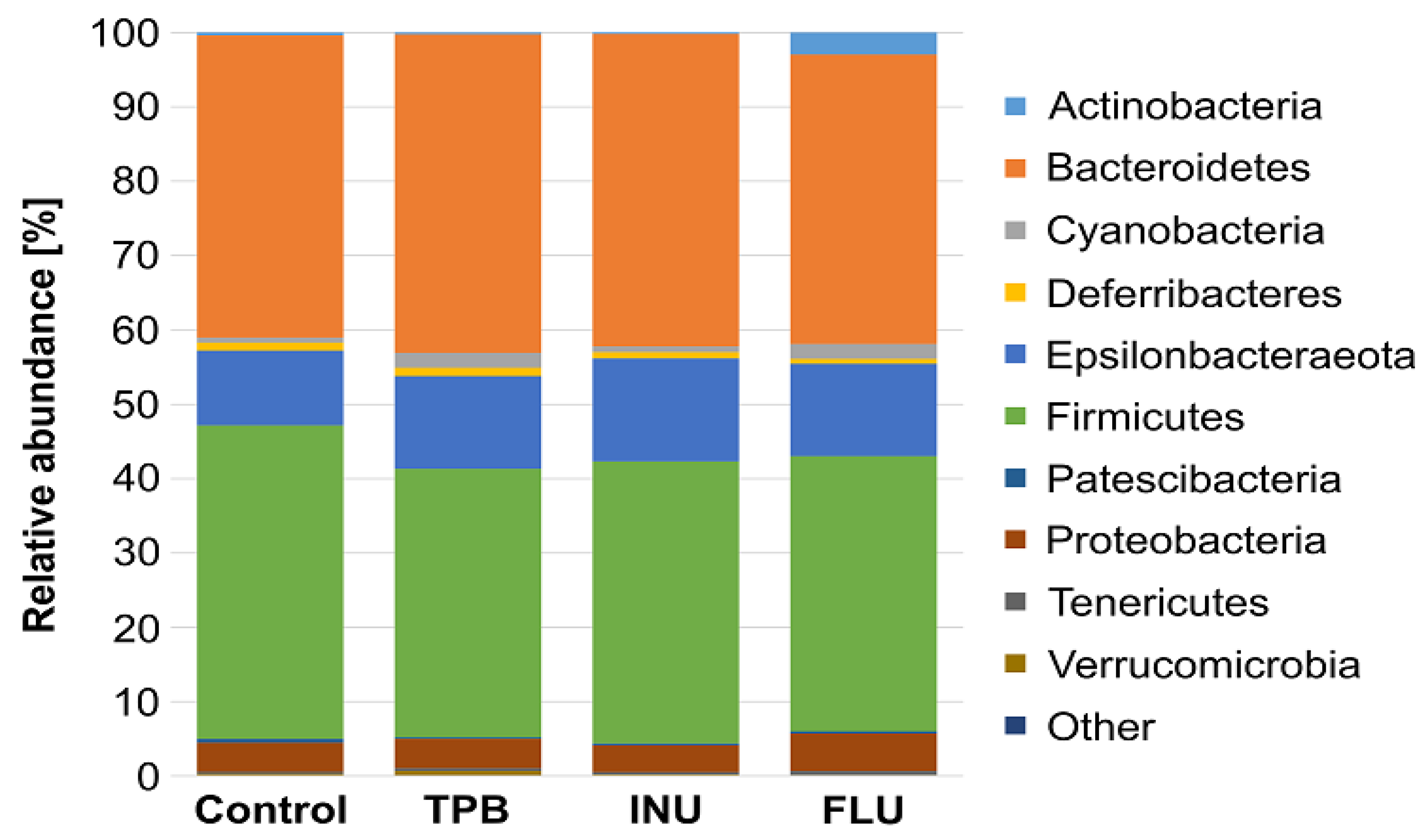
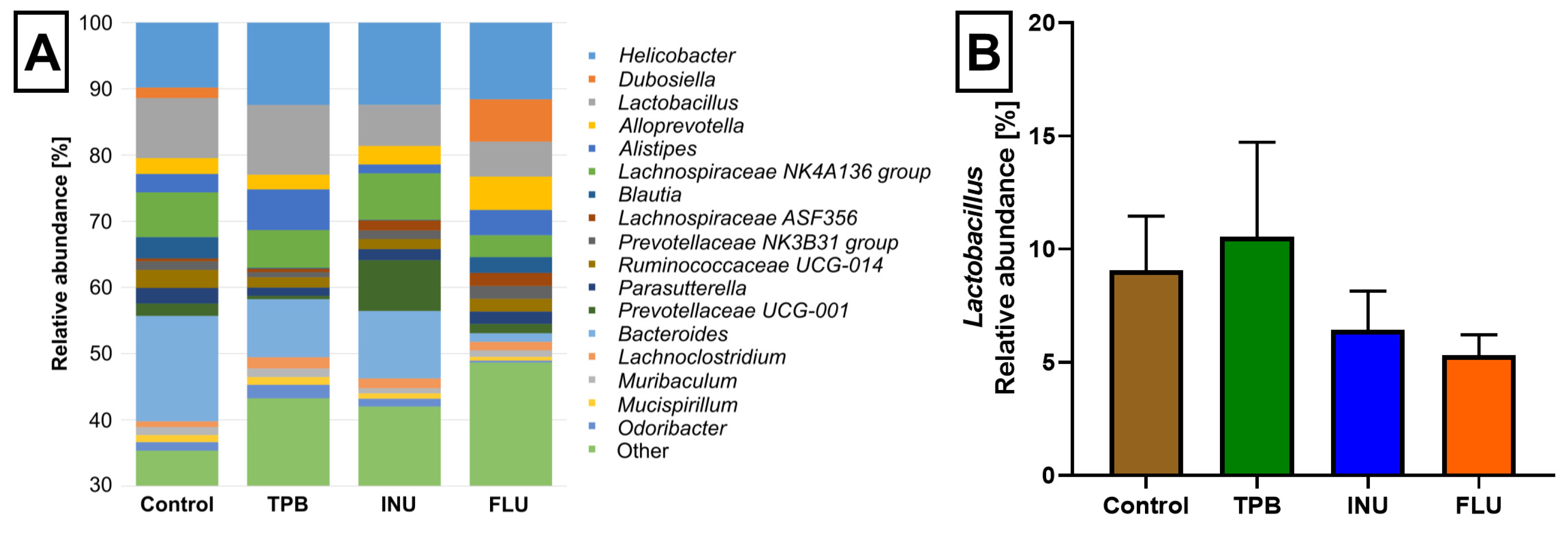
3.4. Effect of Long-Term Administration of TPB, INU, and FLU on the Composition of the Intestinal Microbiota in Mice
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sender, R.; Fuchs, S.; Milo, R. Revised Estimates for the Number of Human and Bacteria Cells in the Body. PLoS Biol. 2016, 14, e1002533. [Google Scholar] [CrossRef] [PubMed]
- Cryan, J.F.; Dinan, T.G. Mind-altering microorganisms: The impact of the gut microbiota on brain and behaviour. Nat. Rev. Neurosci. 2012, 13, 701–712. [Google Scholar] [CrossRef]
- Sekirov, I.; Russell, S.L.; Antunes, L.C.M.; Finlay, B.B.; Galla, S.; Chakraborty, S.; Cheng, X.; Yeo, J.; Mell, B.; Zhang, H.; et al. Gut Microbiota in Health and Disease. Physiol. Rev. 2010, 90, 859–904. [Google Scholar] [CrossRef]
- Murphy, E.F.; Cotter, P.D.; Healy, S.; Marques, T.M.; O’Sullivan, O.; Fouhy, F.; Clarke, S.F.; O’Toole, P.W.; Quigley, E.M.; Stanton, C.; et al. Composition and energy harvesting capacity of the gut microbiota: Relationship to diet, obesity and time in mouse models. Gut 2010, 59, 1635–1642. [Google Scholar] [CrossRef] [PubMed]
- Cong, X.; Henderson, W.A.; Graf, J.; McGrath, J.M. Early Life Experience and Gut Microbiome: The Brain-Gut-Microbiota Signaling System. Adv. Neonatal Care 2015, 15, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Rowland, I.; Gibson, G.; Heinken, A.; Scott, K.; Swann, J.; Thiele, I.; Tuohy, K. Gut microbiota functions: Metabolism of nutrients and other food components. Eur. J. Nutr. 2018, 57, 1–24. [Google Scholar] [CrossRef]
- Patterson, E.; Cryan, J.F.; Fitzgerald, G.F.; Ross, R.P.; Dinan, T.G.; Stanton, C. Gut microbiota, the pharmabiotics they produce and host health. Proc. Nutr. Soc. 2014, 73, 477–489. [Google Scholar] [CrossRef]
- Rhee, S.H.; Pothoulakis, C.; Mayer, E.A. Principles and clinical implications of the brain–Gut–Enteric microbiota axis. Nat. Rev. Gastroenterol. Hepatol. 2009, 6, 306–314. [Google Scholar] [CrossRef]
- Wood, J.D.; Alpers, D.H.; Andrews, P.L.R. Fundamentals of neurogastroenterology. Gut 1999, 45, II6–II16. [Google Scholar] [CrossRef]
- Bercik, P.; Park, A.J.; Sinclair, D.; Khoshdel, A.; Lu, J.; Huang, X.; Deng, Y.; Blennerhassett, P.A.; Fahnestock, M.; Moine, D.; et al. The Anxiolytic effect of bifidobacterium longum NCC3001 involves vagal pathways for gut-brain communication. Neurogastroenterol. Motil. 2011, 23, 1132–1139. [Google Scholar] [CrossRef]
- Bravo, J.A.; Forsythe, P.; Chew, M.V.; Escaravage, E.; Savignac, H.M.; Dinan, T.G.; Bienenstock, J.; Cryan, J.F. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc. Natl. Acad. Sci. USA 2011, 108, 16050–16055. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Yang, X.; Yang, J.; Lai, G.; Yong, T.; Tang, X.; Shuai, O.; Zhou, G.; Xie, Y.; Wu, Q. Prebiotic Effect of Fructooligosaccharides from Morinda officinalis on Alzheimer’s Disease in Rodent Models by Targeting the Microbiota-Gut-Brain Axis. Front. Aging Neurosci. 2017, 9, 403. [Google Scholar] [CrossRef] [PubMed]
- Bercik, P.; Collins, S.M. The effects of inflammation, infection and antibiotics on the microbiota-gut-brain axis. In Micro-Bial Endocrinology: The Microbiota-Gut-Brain Axis in Health and Disease; Lyte, M., Cryan, J.F., Eds.; Springer: New York, NY, USA, 2014; pp. 279–289. ISBN 978-1-4939-0897-4. [Google Scholar]
- Sudo, N.; Chida, Y.; Aiba, Y.; Sonoda, J.; Oyama, N.; Yu, X.-N.; Kubo, C.; Koga, Y. Postnatal microbial colonization programs the hypothalamic-pituitary-adrenal system for stress response in mice. J. Physiol. 2004, 558, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Dinan, T.G.; Cryan, J.F. Brain–Gut–microbiota axis—Mood, metabolism and behaviour. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 69–70. [Google Scholar] [CrossRef] [PubMed]
- Gareau, M.G. Microbiota-Gut-Brain Axis and Cognitive Function. Adv. Exp. Med. Biol. 2014, 817, 357–371. [Google Scholar] [CrossRef]
- Chen, Y.; Xu, J.; Chen, Y. Regulation of Neurotransmitters by the Gut Microbiota and Effects on Cognition in Neurological Disorders. Nutrients 2021, 13, 2099. [Google Scholar] [CrossRef]
- Wang, X.-Q.; Li, H.; Li, X.-N.; Yuan, C.-H.; Zhao, H. Gut-Brain Axis: Possible Role of Gut Microbiota in Perioperative Neurocognitive Disorders. Front. Aging Neurosci. 2021, 13, 745774. [Google Scholar] [CrossRef]
- Escobar, Y.-N.H.; O’Piela, D.; Wold, L.E.; Mackos, A.R. Influence of the Microbiota-Gut-Brain Axis on Cognition in Alzheimer’s Disease. J. Alzheimer’s Dis. 2022, 87, 17–31. [Google Scholar] [CrossRef]
- Ogbonnaya, E.S.; Clarke, G.; Shanahan, F.; Dinan, T.G.; Cryan, J.F.; O’Leary, O.F. Adult Hippocampal Neurogenesis Is Regulated by the Microbiome. Biol. Psychiatry 2015, 78, e7–e9. [Google Scholar] [CrossRef]
- Kundu, P.; Lee, H.U.; Garcia-Perez, I.; Tay, E.X.Y.; Kim, H.; Faylon, L.E.; Martin, K.A.; Purbojati, R.; Drautz-Moses, D.I.; Ghosh, S.; et al. Neurogenesis and prolongevity signaling in young germ-free mice transplanted with the gut microbiota of old mice. Sci. Transl. Med. 2019, 11, 4760. [Google Scholar] [CrossRef]
- Scott, G.A.; Terstege, D.J.; Vu, A.P.; Law, S.; Evans, A.; Epp, J.R. Disrupted Neurogenesis in Germ-Free Mice: Effects of Age and Sex. Front. Cell Dev. Biol. 2020, 8, 407. [Google Scholar] [CrossRef] [PubMed]
- Celorrio, M.; Abellanas, M.A.; Rhodes, J.; Goodwin, V.; Moritz, J.; Vadivelu, S.; Wang, L.; Rodgers, R.; Xiao, S.; Anabayan, I.; et al. Gut microbial dysbiosis after traumatic brain injury modulates the immune response and impairs neurogenesis. Acta Neuropathol. Commun. 2021, 9, 40. [Google Scholar] [CrossRef]
- Cameron, H.A.; Glover, L.R. Adult Neurogenesis: Beyond Learning and Memory. Annu. Rev. Psychol. 2015, 66, 53–81. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J.R.; Keane, V.O.; Cryan, J.F.; Clarke, G.; Dinan, T.G. Mood and Microbes. Gastroenterol. Clin. N. Am. 2019, 48, 389–405. [Google Scholar] [CrossRef]
- Pusceddu, M.M.; Barboza, M.; Keogh, C.E.; Schneider, M.; Stokes, P.; Sladek, J.A.; Kim, H.J.D.; Torres-Fuentes, C.; Goldfild, L.R.; Gillis, S.E.; et al. Nod-like receptors are critical for gut–brain axis signalling in mice. J. Physiol. 2019, 597, 5777–5797. [Google Scholar] [CrossRef]
- Ribeiro, M.F.; Santos, A.A.; Afonso, M.B.; Rodrigues, P.M.; Sá Santos, S.; Castro, R.E.; Rodrigues, C.M.P.; Solá, S. Di-et-dependent gut microbiota impacts on adult neurogenesis through mitochondrial stress modulation. Brain Commun. 2020, 2, fcaa165. [Google Scholar] [CrossRef] [PubMed]
- Cryan, J.F.; O’Mahony, S.M. The microbiome-gut-brain axis: From bowel to behavior. Neurogastroenterol. Motil. 2011, 23, 187–192. [Google Scholar] [CrossRef]
- Grenham, S.; Clarke, G.; Cryan, J.F.; Dinan, T.G. Brain-Gut-Microbe Communication in Health and Disease. Front. Physiol. 2011, 2, 94. [Google Scholar] [CrossRef]
- O’Mahony, S.M.; Hyland, N.P.; Dinan, T.G.; Cryan, J.F. Maternal separation as a model of brain-gut axis dysfunction. Psychopharmacology 2011, 214, 71–88. [Google Scholar] [CrossRef]
- Vich Vila, A.; Collij, V.; Sanna, S.; Sinha, T.; Imhann, F.; Bourgonje, A.; Mujagic, Z.; Jonkers, D.M.A.E.; Masclee, A.A.M.; Fu, J.; et al. Impact of commonly used drugs on the composition and metabolic function of the gut microbiota. Nat. Commun. 2020, 11, 362. [Google Scholar] [CrossRef]
- Zhang, W.; Qu, W.; Wang, H.; Yan, H. Antidepressants fluoxetine and amitriptyline induce alterations in intestinal microbiota and gut microbiome function in rats exposed to chronic unpredictable mild stress. Transl. Psychiatry 2021, 11, 131. [Google Scholar] [CrossRef] [PubMed]
- Sommi, R.W.; Crismon, M.L.; Bowden, C.L. Fluoxetine: A Serotonin-specific, Second-generation Antidepressant. Pharmacother. J. Hum. Pharmacol. Drug Ther. 1987, 7, 3496. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.J.; Martins, M.C.; Pereira, A.M.; Meisel, L.M.; Gonzalez-Rey, M.; Bebianno, M.J.; Lino, C.M.; Pena, A. Uptake, accumulation and metabolization of the antidepressant fluoxetine by Mytilus galloprovincialis. Environ. Pollut. 2016, 213, 432–437. [Google Scholar] [CrossRef] [PubMed]
- Cussotto, S.; Strain, C.R.; Fouhy, F.; Strain, R.G.; Peterson, V.L.; Clarke, G.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Differential effects of psychotropic drugs on microbiome composition and gastrointestinal function. Psychopharmacology 2018, 236, 1671–1685. [Google Scholar] [CrossRef]
- Lyte, M.; Daniels, K.M.; Schmitz-Esser, S. Fluoxetine-induced alteration of murine gut microbial community structure: Evidence for a microbial endocrinology-based mechanism of action responsible for fluoxetine-induced side effects. PeerJ 2019, 7, e6199. [Google Scholar] [CrossRef]
- Quigley, E.M. Prebiotics and Probiotics in Digestive Health. Clin. Gastroenterol. Hepatol. 2018, 17, 333–344. [Google Scholar] [CrossRef]
- Markowiak, P.; Śliżewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef]
- Saulnier, D.M.; Ringel, Y.; Heyman, M.B.; Foster, J.A.; Bercik, P.; Shulman, R.J.; Versalovic, J.; Verdu, E.F.; Dinan, T.G.; Hecht, G.; et al. The intestinal microbiome, probiotics and prebiotics in neurogastroenterology. Gut Microbes 2013, 4, 17–27. [Google Scholar] [CrossRef]
- Moshfegh, A.J.; Friday, J.E.; Goldman, J.P.; Ahuja, J.K.C. Presence of Inulin and Oligofructose in the Diets of Americans. J. Nutr. 1999, 129, S1407–S1411. [Google Scholar] [CrossRef]
- Szewczyk, A.; Zagaja, M.; Bryda, J.; Kosikowska, U.; Stępień-Pyśniak, D.; Winiarczyk, S.; Andres-Mach, M. Topinambur–new possibilities for use in a supplementation diet. Ann. Agric. Environ. Med. 2019, 26, 24–28. [Google Scholar] [CrossRef]
- Zaky, E.A. Physiological Response to Diets Fortified with Jerusalem Artichoke Tubers (Helianthus tuberosus L.) Powder by Diabetic Rats. Environ. Sci. 2009, 5, 682–688. [Google Scholar]
- Samal, L.; Chaturvedi, V.; Pattanaik, A. Effects of dietary supplementation with Jerusalem artichoke (Helianthus tuberosus L.) tubers on growth performance, nutrient digestibility as well as activity and composition of large intestinal microbiota in rats. J. Anim. Feed. Sci. 2017, 26, 50–58. [Google Scholar] [CrossRef]
- Sun, L.; Zhang, H.; Cao, Y.; Wang, C.; Zhao, C.; Wang, H.; Cui, G.; Wang, M.; Pan, Y.; Shi, Y.; et al. Fluoxetine ameliorates dysbiosis in a depression model induced by chronic unpredicted mild stress in mice. Int. J. Med. Sci. 2019, 16, 1260–1270. [Google Scholar] [CrossRef] [PubMed]
- Andres-Mach, M.; Szewczyk, A.; Zagaja, M.; Luszczki, J.; Maj, M.; Rola, R.; Abram, M.; Kaminski, K. Evaluation of the impact of compound C11 a new anticonvulsant candidate on cognitive functions and hippocampal neurogenesis in mouse brain. Neuropharmacology 2020, 163, 107849. [Google Scholar] [CrossRef] [PubMed]
- Andres-Mach, M.; Szewczyk, A.; Zagaja, M.; Szala-Rycaj, J.; Lemieszek, M.K.; Maj, M.; Abram, M.; Kaminski, K. Preclinical Assessment of a New Hybrid Compound C11 Efficacy on Neurogenesis and Cognitive Functions after Pilocarpine Induced Status Epilepticus in Mice. Int. J. Mol. Sci. 2021, 22, 3240. [Google Scholar] [CrossRef]
- Szewczyk, A.; Zagaja, M.; Szala-Rycaj, J.; Maj, M.; Andres-Mach, M. Effect of Lacosamide and Ethosuximide Chronic Treatment on Neural Precursor Cells and Cognitive Functions after Pilocarpine Induced Status Epilepticus in Mice. Brain Sci. 2021, 11, 1014. [Google Scholar] [CrossRef]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glöckner, F.O. Evaluation of General 16S Ribosomal RNA Gene PCR Primers for Classical and Next-Generation Sequencing-Based Diversity Studies. Nucleic Acids Res. 2013, 41, e1. [Google Scholar] [CrossRef]
- Kaczmarczyk-Ziemba, A.; Zagaja, M.; Wagner, G.K.; Pietrykowska-Tudruj, E.; Staniec, B. First Insight into Microbiome Profiles of Myrmecophilous Beetles and Their Host, Red Wood Ant Formica polyctena (Hymenoptera: Formicidae)—A Case Study. Insects 2020, 11, 134. [Google Scholar] [CrossRef]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, Interactive, Scalable and Extensible Microbiome Data Science using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef]
- Callahan, B.J.; Mcmurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA Ribosomal RNA Gene Database Project: Improved Data Processing and Web-Based Tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef]
- Chong, J.; Liu, P.; Zhou, G.; Xia, J. Using MicrobiomeAnalyst for comprehensive statistical, functional, and meta-analysis of microbiome data. Nat. Protoc. 2020, 15, 799–821. [Google Scholar] [CrossRef]
- Dhariwal, A.; Chong, J.; Habib, S.; King, I.L.; Agellon, L.B.; Xia, J. MicrobiomeAnalyst: A web-based tool for comprehensive statistical, visual and meta-analysis of microbiome data. Nucleic Acids Res. 2017, 45, W180–W188. [Google Scholar] [CrossRef] [PubMed]
- Andres-Mach, M.; Haratym-Maj, A.; Zagaja, M.; Rola, R.; Maj, M.; Chrościńska-Krawczyk, M.; Luszczki, J.J. ACEA (a highly selective cannabinoid CB1 receptor agonist) stimulates hippocampal neurogenesis in mice treated with antiepileptic drugs. Brain Res. 2015, 1624, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Andres-Mach, M.; Zagaja, M.; Haratym-Maj, A.; Rola, R.; Maj, M.; Haratym, J.; Dudra-Jastrzębska, M.; Łuszczki, J.J. A Long-Term Treatment with Arachidonyl-2′-Chloroethylamide Combined with Valproate Increases Neurogenesis in a Mouse Pilocarpine Model of Epilepsy. Int. J. Mol. Sci. 2017, 18, 900. [Google Scholar] [CrossRef] [PubMed]
- Clarke, K.R.; Gorley, R.N. PRIMER v7: User Manual/Tutorial; PRIMER-E: Devon, UK, 2015; Volume 29, pp. 1060–1065. [Google Scholar]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontol. Electronica. 2001, 4, 9. [Google Scholar]
- Segata, N.; Izard, J.; Waldron, L.; Gevers, D.; Miropolsky, L.; Garrett, W.S.; Huttenhower, C. Metagenomic biomarker discovery and explanation. Genome Biol. 2011, 12, R60. [Google Scholar] [CrossRef]
- Douglas, G.M.; Maffei, V.J.; Zaneveld, J.R.; Yurgel, S.N.; Brown, J.R.; Taylor, C.M.; Huttenhower, C.; Langille, M.G.I. PICRUSt2 for prediction of metagenome functions. Nat. Biotechnol. 2020, 38, 685–688. [Google Scholar] [CrossRef] [PubMed]
- Koch, F.; Derno, M.; Langhammer, M.; Tuchscherer, A.; Hammon, H.M.; Mielenz, M.; Metges, C.C.; Kuhla, B. A high-protein diet containing inulin/oligofructose supports body weight gain associated with lower energy expenditure and carbohydrate oxidation, and alters faecal microbiota in C57BL/6 mice. J. Nutr. Sci. 2021, 10, 42. [Google Scholar] [CrossRef]
- Petersen, A.; Heegaard, P.M.; Pedersen, A.L.; Andersen, J.B.; Sørensen, R.B.; Frøkiær, H.; Lahtinen, S.J.; Ouwehand, A.C.; Poulsen, M.; Licht, T.R. Some putative prebiotics increase the severity of Salmonella entericaserovar Typhimurium infection in mice. BMC Microbiol. 2009, 9, 245. [Google Scholar] [CrossRef]
- Serralde-Zuñiga, A.E.; González-Garay, A.G.; Rodríguez-Carmona, Y.; Meléndez-Mier, G. Use of Fluoxetine to Reduce Weight in Adults with Overweight or Obesity: Abridged Republication of the Cochrane Systematic Review. Obes. Facts 2022, 15, 473–486. [Google Scholar] [CrossRef]
- Majlessi, N.; Naghdi, N. Impaired spatial learning in the Morris water maze induced by serotonin reuptake inhibitors in rats. Behav. Pharmacol. 2002, 13, 237–242. [Google Scholar] [CrossRef]
- Golub, M.S.; Hackett, E.P.; Hogrefe, C.E.; Leranth, C.; Elsworth, J.D.; Roth, R.H. Cognitive performance of juvenile monkeys after chronic fluoxetine treatment. Dev. Cogn. Neurosci. 2017, 26, 52–61. [Google Scholar] [CrossRef]
- Messaoudi, M.; Rozan, P.; Nejdi, A.; Hidalgo, S.; Desor, D. Behavioural and cognitive effects of oligofructose-enriched inulin in rats. Br. J. Nutr. 2005, 93, S27–S30. [Google Scholar] [CrossRef]
- Romo-Araiza, A.; Gutiérrez-Salmeán, G.; Galván, E.J.; Hernández-Frausto, M.; Herrera-López, G.; Romo-Parra, H.; García-Contreras, V.; Fernández-Presas, A.M.; Jasso-Chávez, R.; Borlongan, C.V.; et al. Probiotics and Prebiotics as a Therapeutic Strategy to Improve Memory in a Model of Middle-Aged Rats. Front. Aging Neurosci. 2018, 10, 416. [Google Scholar] [CrossRef]
- Schmidt, K.; Cowen, P.; Harmer, C.; Tzortzis, G.; Errington, S.; Burnet, P.W.J. Prebiotic intake reduces the waking cortisol response and alters emotional bias in healthy volunteers. Psychopharmacology 2014, 232, 1793–1801. [Google Scholar] [CrossRef]
- Smith, A.P. The concept of well-being: Relevance to nutrition research. Br. J. Nutr. 2005, 93, S1–S5. [Google Scholar] [CrossRef]
- Talbott, S.; Talbott, J. Effect of BETA 1, 3/1, 6 GLUCAN on Upper Respiratory Tract Infection Symptoms and Mood State in Marathon Athletes. J. Sports Sci. Med. 2009, 8, 509–515. [Google Scholar]
- Klomp, A.; Václavů, L.; Meerhoff, G.F.; Reneman, L.; Lucassen, P.J. Effects of Chronic Fluoxetine Treatment on Neurogenesis and Tryptophan Hydroxylase Expression in Adolescent and Adult Rats. PLoS ONE 2014, 9, e97603. [Google Scholar] [CrossRef]
- Cowen, D.S.; Takase, L.F.; Fornal, C.A.; Jacobs, B.L. Age-dependent decline in hippocampal neurogenesis is not altered by chronic treatment with fluoxetine. Brain Res. 2008, 1228, 14–19. [Google Scholar] [CrossRef]
- Navailles, S.; Hof, P.R.; Schmauss, C. Antidepressant drug-induced stimulation of mouse hippocampal neurogenesis is age-dependent and altered by early life stress. J. Comp. Neurol. 2008, 509, 372–381. [Google Scholar] [CrossRef]
- Marcussen, A.B.; Flagstad, P.; Kristjansen, P.E.G.; Johansen, F.F.; Englund, U. Increase in neurogenesis and behavioural benefit after chronic fluoxetine treatment in Wistar rats. Acta Neurol. Scand. 2007, 117, 94–100. [Google Scholar] [CrossRef]
- Hovorka, M.; Ewing, D.; Middlemas, D.S. Chronic SSRI Treatment, but Not Norepinephrine Reuptake Inhibitor Treatment, Increases Neurogenesis in Juvenile Rats. Int. J. Mol. Sci. 2022, 23, 6919. [Google Scholar] [CrossRef]
- Hodes, G.E.; Hill-Smith, T.E.; Lucki, I. Fluoxetine treatment induces dose dependent alterations in depression associated behavior and neural plasticity in female mice. Neurosci. Lett. 2010, 484, 12–16. [Google Scholar] [CrossRef]
- Umu, Ö.C.O.; Rudi, K.; Diep, D.B. Modulation of the gut microbiota by prebiotic fibres and bacteriocins. Microb. Ecol. Health Dis. 2017, 28, 1348886. [Google Scholar] [CrossRef]
- Ayaz, M.; Subhan, F.; Ahmed, J.; Khan, A.-U.; Ullah, F.; Ullah, I.; Ali, G.; Syed, N.-I.; Hussain, S. Sertraline enhances the activity of antimicrobial agents against pathogens of clinical relevance. J. Biol. Res. 2015, 22, 4. [Google Scholar] [CrossRef]
- Coban, A.Y.; Cayci, Y.T.; Uludağ, S.K.; Durupinar, B. Investigation of antibacterial activity of sertraline. Mikrobiyoloji Bul. 2009, 43, 651–656. [Google Scholar]
- Sun, X.; Wang, D.; Wei, L.; Ding, L.; Guo, Y.; Wang, Z.; Kong, Y.; Yang, J.; Sun, L.; Sun, L. Gut Microbiota and SCFAs Play Key Roles in QingFei Yin Recipe Anti-Streptococcal Pneumonia Effects. Front. Cell. Infect. Microbiol. 2021, 11, 791466. [Google Scholar] [CrossRef]
- Zhong, Q.; Chen, J.-J.; Wang, Y.; Shao, W.-H.; Zhou, C.-J.; Xie, P. Differential Gut Microbiota Compositions Related With the Severity of Major Depressive Disorder. Front. Cell. Infect. Microbiol. 2022, 12, 907239. [Google Scholar] [CrossRef]
- Zhou, Y.; Chen, C.; Yu, H.; Yang, Z. Fecal Microbiota Changes in Patients With Postpartum Depressive Disorder. Front. Cell. Infect. Microbiol. 2020, 10, 567268. [Google Scholar] [CrossRef]
- Zafar, H.; Saier, M.H., Jr. Gut Bacteroides species in health and disease. Gut Microbes 2021, 13, 8158. [Google Scholar] [CrossRef]
- Song, X.; Zhong, L.; Lyu, N.; Liu, F.; Li, B.; Hao, Y.; Xue, Y.; Li, J.; Feng, Y.; Ma, Y.; et al. Inulin Can Alleviate Me-tabolism Disorders in ob/ob Mice by Partially Restoring Leptin-related Pathways Mediated by Gut Microbiota. Genom. Proteom. Bioinform. 2019, 17, 64–75. [Google Scholar] [CrossRef]
- Nakayama, J.; Yamamoto, A.; Palermo-Conde, L.A.; Higashi, K.; Sonomoto, K.; Tan, J.; Lee, Y.-K. Impact of Westernized Diet on Gut Microbiota in Children on Leyte Island. Front. Microbiol. 2017, 8, 197. [Google Scholar] [CrossRef]
- De Filippo, C.; Cavalieri, D.; Di Paola, M.; Ramazzotti, M.; Poullet, J.B.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl. Acad. Sci. USA 2010, 107, 14691–14696. [Google Scholar] [CrossRef]
- Stilling, R.M.; Dinan, T.G.; Cryan, J.F. Microbial genes, brain &behaviour—Epigenetic regulation of the gut-brain axis. Genes Brain Behav. 2014, 13, 69–86. [Google Scholar] [CrossRef]
- Donia, M.S.; Fischbach, M.A. Small molecules from the human microbiota. Science 2015, 349, 1254766. [Google Scholar] [CrossRef]
- Liang, S.; Wang, T.; Hu, X.; Luo, J.; Li, W.; Wu, X.; Duan, Y.; Jin, F. Administration of Lactobacillus helveticus NS8 improves behavioral, cognitive, and biochemical aberrations caused by chronic restraint stress. Neuroscience 2015, 310, 561–577. [Google Scholar] [CrossRef]

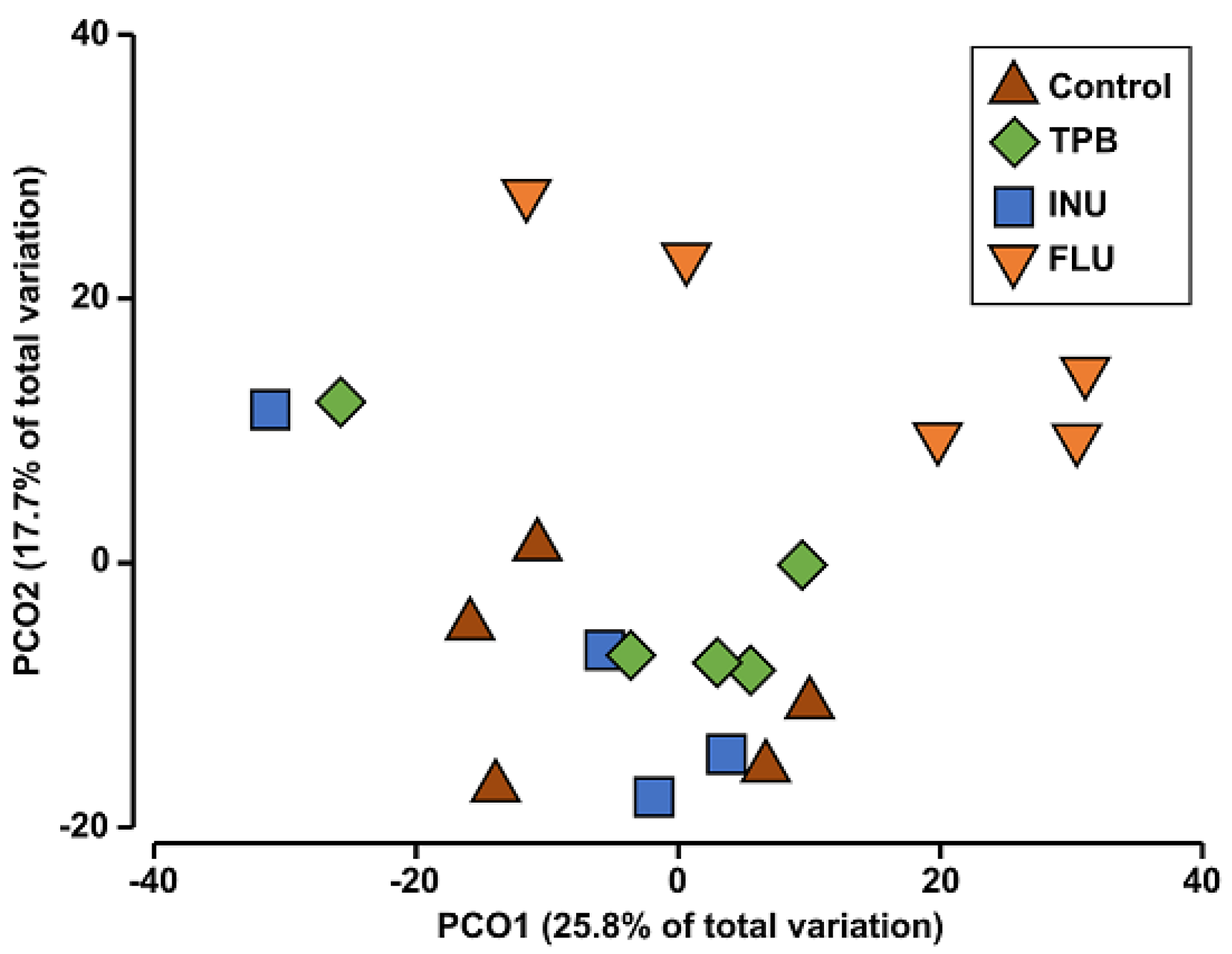

| Compared Groups | Pseudo-F | p |
|---|---|---|
| control—FLU | 1.9887 | 0.007 * |
| control—INU | 1.2850 | 0.103 |
| control—TPB | 1.2337 | 0.152 |
| FLU—INU | 1.7685 | 0.010 * |
| FLU—TPB | 1.5593 | 0.007 * |
| INU—TPB | 1.1114 | 0.260 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szewczyk, A.; Andres-Mach, M.; Zagaja, M.; Kaczmarczyk-Ziemba, A.; Maj, M.; Szala-Rycaj, J. The Effect of a Diet Enriched with Jerusalem artichoke, Inulin, and Fluoxetine on Cognitive Functions, Neurogenesis, and the Composition of the Intestinal Microbiota in Mice. Curr. Issues Mol. Biol. 2023, 45, 2561-2579. https://doi.org/10.3390/cimb45030168
Szewczyk A, Andres-Mach M, Zagaja M, Kaczmarczyk-Ziemba A, Maj M, Szala-Rycaj J. The Effect of a Diet Enriched with Jerusalem artichoke, Inulin, and Fluoxetine on Cognitive Functions, Neurogenesis, and the Composition of the Intestinal Microbiota in Mice. Current Issues in Molecular Biology. 2023; 45(3):2561-2579. https://doi.org/10.3390/cimb45030168
Chicago/Turabian StyleSzewczyk, Aleksandra, Marta Andres-Mach, Mirosław Zagaja, Agnieszka Kaczmarczyk-Ziemba, Maciej Maj, and Joanna Szala-Rycaj. 2023. "The Effect of a Diet Enriched with Jerusalem artichoke, Inulin, and Fluoxetine on Cognitive Functions, Neurogenesis, and the Composition of the Intestinal Microbiota in Mice" Current Issues in Molecular Biology 45, no. 3: 2561-2579. https://doi.org/10.3390/cimb45030168
APA StyleSzewczyk, A., Andres-Mach, M., Zagaja, M., Kaczmarczyk-Ziemba, A., Maj, M., & Szala-Rycaj, J. (2023). The Effect of a Diet Enriched with Jerusalem artichoke, Inulin, and Fluoxetine on Cognitive Functions, Neurogenesis, and the Composition of the Intestinal Microbiota in Mice. Current Issues in Molecular Biology, 45(3), 2561-2579. https://doi.org/10.3390/cimb45030168






