Evaluation of the Regenerative Potential of Platelet-Lysate and Platelet-Poor Plasma Derived from the Cord Blood Units in Corneal Wound Healing Applications: An In Vitro Comparative Study on Corneal Epithelial Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Selection of Cord Blood Units
2.2. Cord Blood Units Testing and Eligibility
2.3. Cord Blood Platelet Lysate and Cord Blood Platelet Poor Plasma Production
2.4. Cord Blood Platelet Lysate and Cord Blood Platelet Poor Plasma Quality Characterization
2.5. Isolation of Corneal Epithelial Cells
2.6. Effect of Cord Blood Platelet Lysate and Cord Blood Platelet Poor Plasma on Corneal Epithelial Cells Characteristics
2.7. Scratch Wound Assay
2.8. Characterization of Corneal Epithelial Cells Using Immunoassays
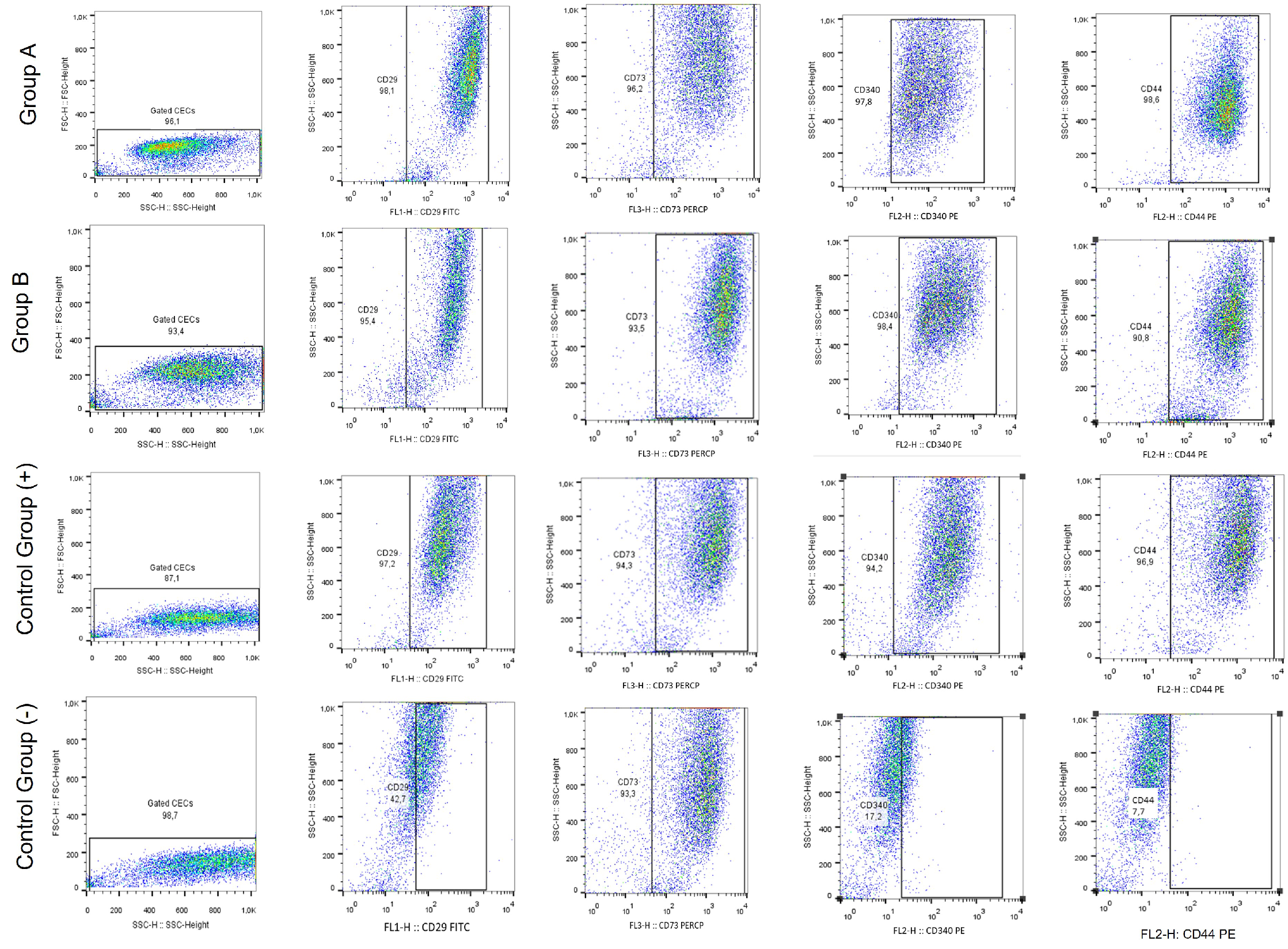
2.9. Flow Cytometric Analysis of Corneal Epithelial Cells
2.10. Gene Expression Analysis of Corneal Epithelial Cells
2.11. Functional Protein Analysis
2.12. Statistical Analysis
3. Results
3.1. Characterization of Cord Blood Derivatives
3.2. Quality Characteristics of Cord Blood Platelet Lysate and Cord Blood Platelet Poor Plasma
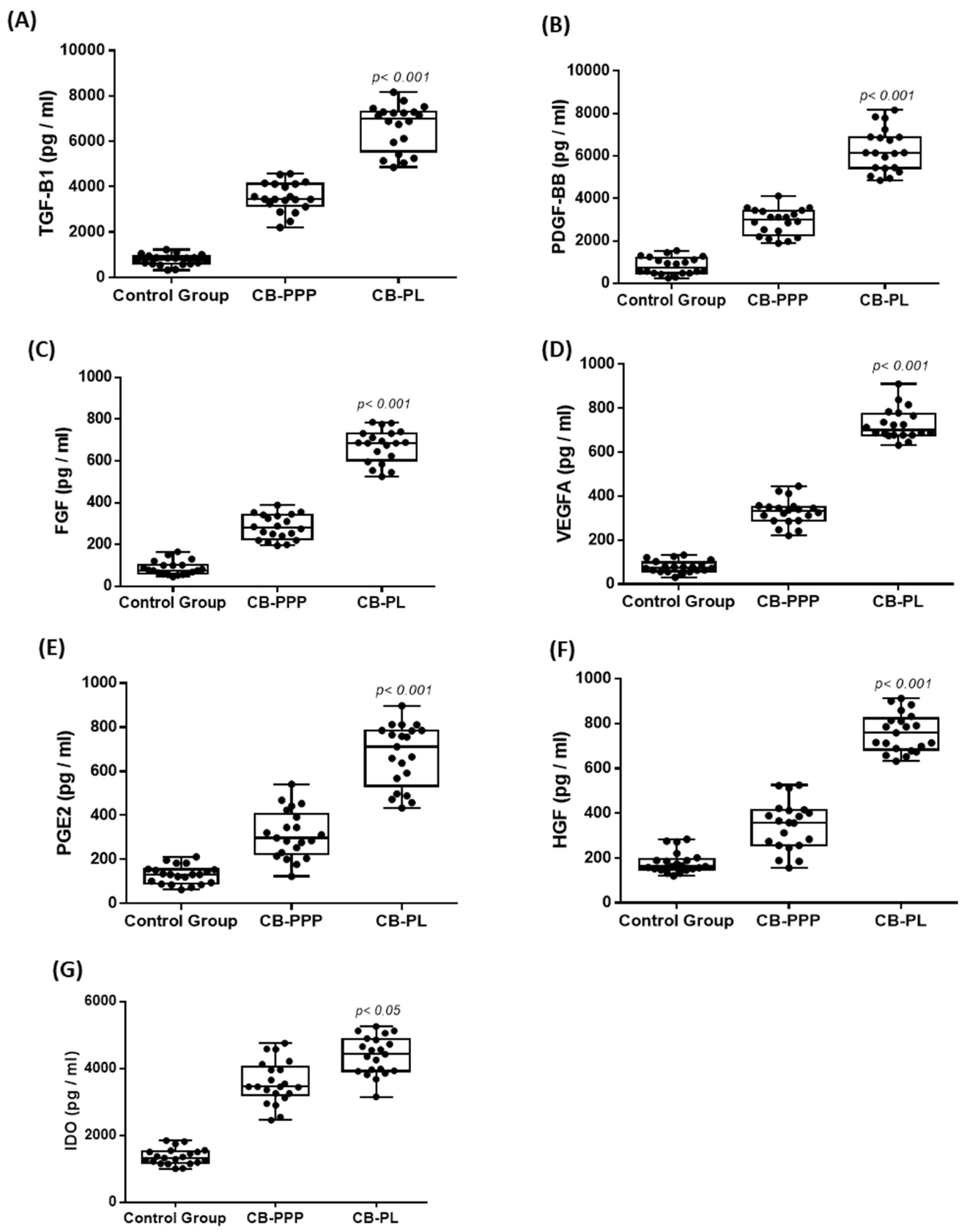
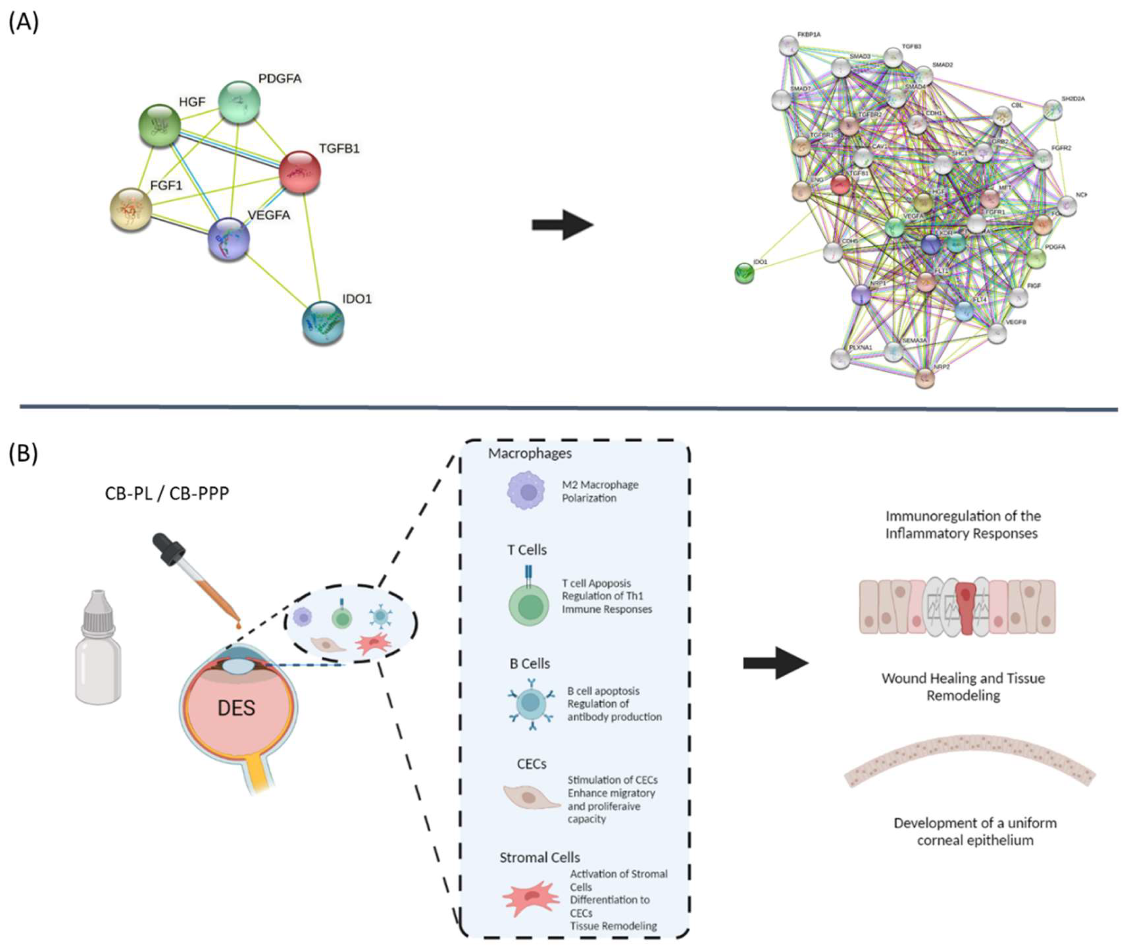
3.3. Biomolecule Quantification
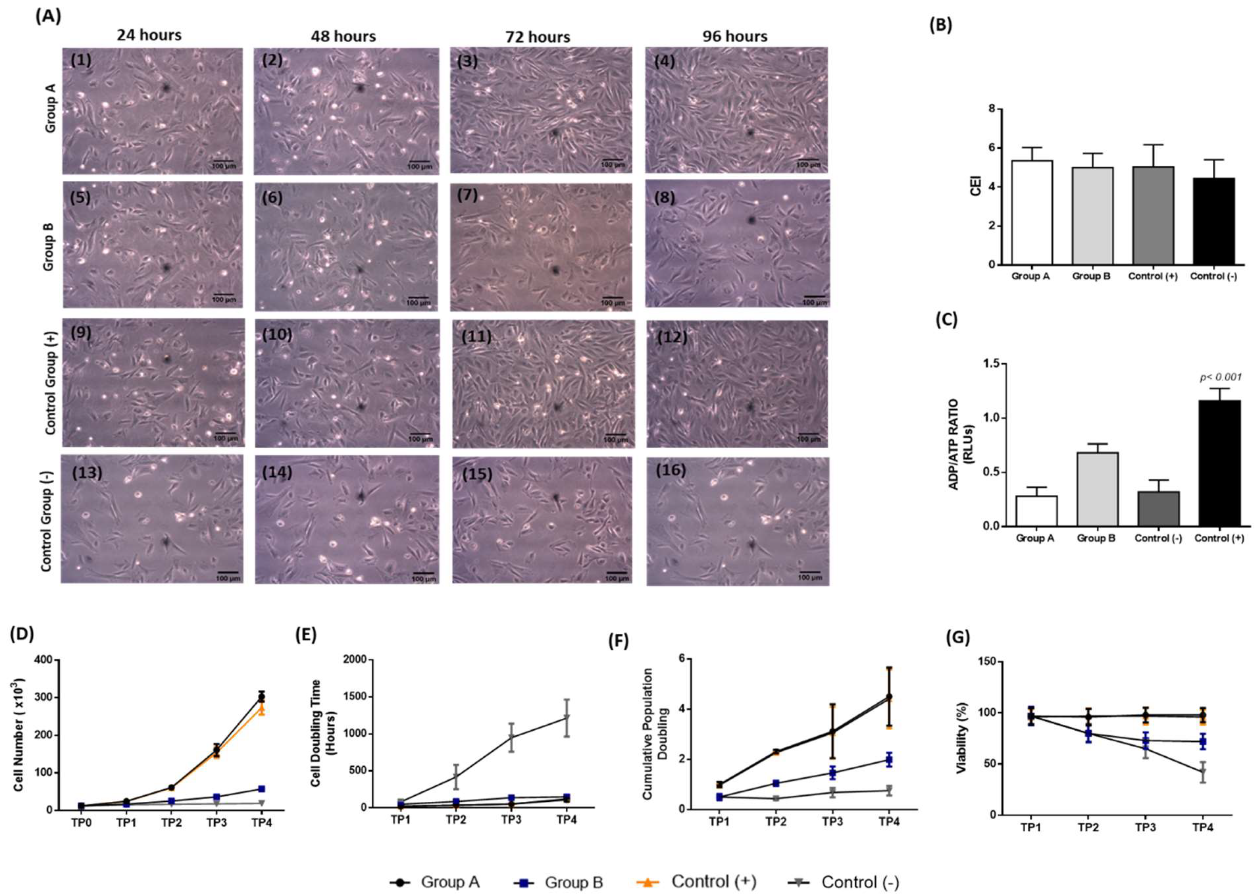
3.4. Isolation and Characterization of the Corneal Epithelial Cells
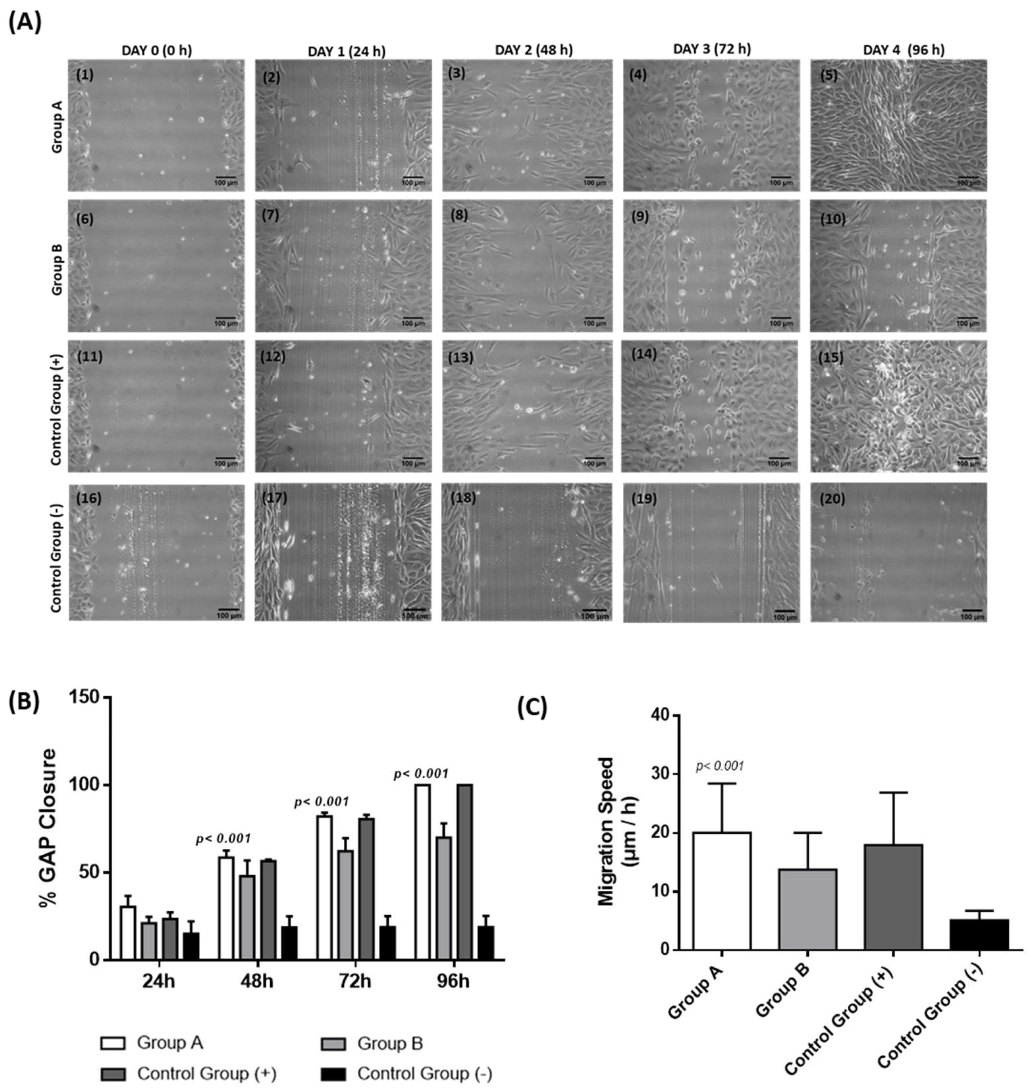
3.5. Scratch Wound Assay
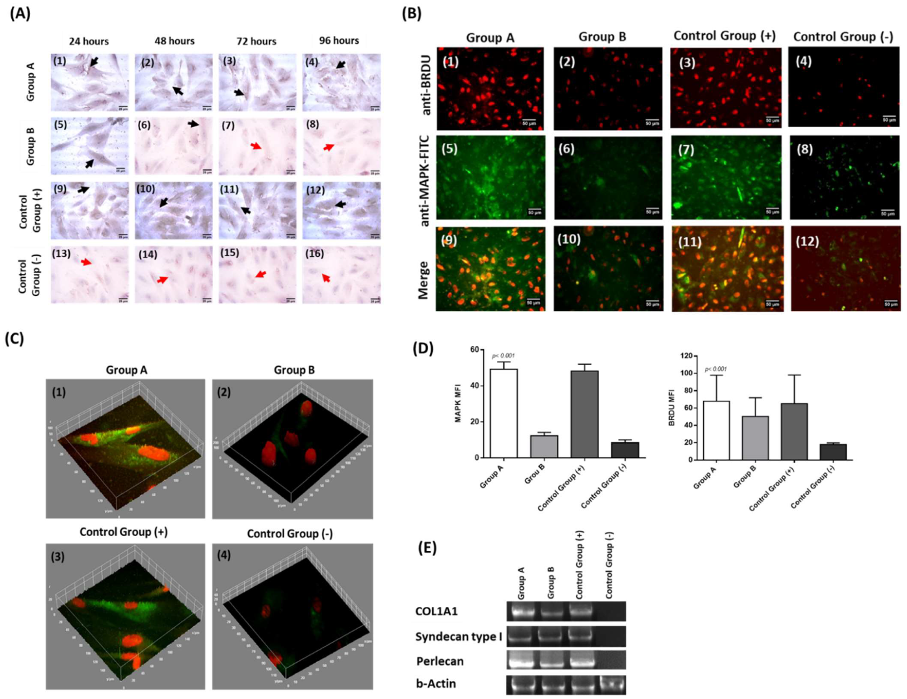
3.6. Immunostaining, Gene Expression Analysis and Immunophenotype of Corneal Epithelial Cells
3.7. Functional Protein Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schaumberg, D.A.; Dana, R.; Buring, J.E.; Sullivan, D.A. Prevalence of dry eye disease among US men: Estimates from the Physicians’ Health Studies. Arch. Ophthalmol. 2009, 127, 763–768. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, B.D.; Evans, J.E.; Dana, M.R.; Sullivan, D.A. Influence of aging on the polar and neutral lipid profiles in human meibomian gland secretions. Arch. Ophthalmol. 2006, 124, 1286–1292. [Google Scholar] [CrossRef] [PubMed]
- Drew, V.J.; Tseng, C.L.; Seghatchian, J.; Burnouf, T. Reflections on Dry Eye Syndrome Treatment: Therapeutic Role of Blood Products. Front. Med. 2018, 5, 33. [Google Scholar] [CrossRef]
- Schaumberg, D.A.; Sullivan, D.A.; Buring, J.E.; Dana, M.R. Prevalence of dry eye syndrome among US women. Am. J. Ophthalmol. 2003, 136, 318–326. [Google Scholar] [CrossRef]
- McDonald, M.; Patel, D.A.; Keith, M.S.; Snedecor, S.J. Economic and humanistic burden of dry eye disease in Europe, North America, and Asia: A systematic literature review. Ocul. Surf. 2016, 14, 144–167. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Asche, C.V.; Fairchild, C.J. The economic burden of dry eye disease in the United States: A decision tree analysis. Cornea 2011, 30, 379–387. [Google Scholar] [CrossRef]
- Pflugfelder, S.C.; Solomon, A.; Stern, M.E. The diagnosis and management of dry eye: A twenty-five-year review. Cornea 2000, 19, 644–649. [Google Scholar] [CrossRef]
- Messmer, E.M. The pathophysiology, diagnosis, and treatment of dry eye disease. Dtsch. Arztebl. Int. 2015, 112, 71. [Google Scholar] [CrossRef]
- Nebbioso, M.; Fameli, V.; Gharbiya, M.; Sacchetti, M.; Zicari, A.M.; Lambiase, A. Investigational drugs in dry eye disease. Expert Opin. Investig. Drugs 2016, 25, 1437–1446. [Google Scholar] [CrossRef]
- Shine, W.E.; McCulley, J.P. Polar lipids in human meibomian gland secretions. Curr Eye Res. 2003, 26, 89–94. [Google Scholar] [CrossRef]
- Barabino, S.; Labetoulle, M.; Rolando, M.; Messmer, E.M. Understanding symptoms and quality of life in patients with dry eye syndrome. Ocul. Surf. 2016, 14, 365–376. [Google Scholar] [CrossRef]
- Miljanović, B.; Dana, R.; Sullivan, D.A.; Schaumberg, D.A. Impact of dry eye syn-drome on vision-related quality of life. Am. J. Ophthalmol. 2007, 143, 409–415.e2. [Google Scholar] [CrossRef]
- Ayaki, M.; Kawashima, M.; Negishi, K.; Kishimoto, T.; Mimura, M.; Tsubota, K. Sleep and mood disorders in dry eye disease and allied irritating ocular diseases. Sci. Rep. 2016, 6, 22480. [Google Scholar] [CrossRef]
- Zheng, Y.; Wu, X.; Lin, X.; Lin, H. The prevalence of depression and depressive symptoms among eye disease patients: A systematic review and meta-analysis. Sci. Rep. 2017, 7, 46453. [Google Scholar] [CrossRef]
- Wan, K.H.; Chen, L.J.; Young, A.L. Depression and anxiety in dry eye disease: A systematic review and meta-analysis. Eye 2016, 30, 1558–1567. [Google Scholar] [CrossRef]
- McMonnies, C.W. Measurement of symptoms pre- and post-treatment of dry eye syndromes. Optom. Vis. Sci. 2016, 93, 1431–1437. [Google Scholar] [CrossRef]
- van der Meer, P.F.; Verbakel, S.K.; Honohan, Á.; Lorinser, J.; Thurlings, R.M.; Jacobs, J.F.M.; de Korte, D.; Eggink, C.A. Allogeneic and autologous serum eye drops: A pilot double-blind randomized crossover trial. Acta Ophthalmol. 2021, 99, 837–842. [Google Scholar] [CrossRef]
- Mallis, P.; Michalopoulos, E.; Balampanis, K.; Sarri, E.F.; Papadopoulou, E.; Theodoropoulou, V.; Georgiou, E.; Kountouri, A.; Lambadiari, V.; Stavropoulos-Giokas, C. Investigating the production of platelet lysate obtained from low volume Cord Blood Units: Focus on growth factor content and regenerative potential. Transfus. Apher. Sci. 2022, 20, 103465. [Google Scholar] [CrossRef]
- Mallis, P.; Gontika, I.; Dimou, Z.; Panagouli, E.; Zoidakis, J.; Makridakis, M.; Vlahou, A.; Georgiou, E.; Gkioka, V.; Stavropoulos-Giokas, C.; et al. Short Term Results of Fibrin Gel Obtained from Cord Blood Units: A Preliminary in Vitro Study. Bioengineering 2019, 6, 66. [Google Scholar] [CrossRef]
- Zhang, Y.; Alexander, P.B.; Wang, X.F. TGF-β Family Signaling in the Control of Cell Proliferation and Survival. Cold Spring Harb. Perspect. Biol. 2017, 9, a022145. [Google Scholar] [CrossRef]
- Holmes, D.I.; Zachary, I. The vascular endothelial growth factor (VEGF) family: Angiogenic factors in health and disease. Genome Biol. 2005, 6, 209. [Google Scholar] [CrossRef][Green Version]
- Mao, Y.; Liu, X.; Song, Y.; Zhai, C.; Zhang, L. VEGF-A/VEGFR-2 and FGF-2/FGFR-1 but not PDGF-BB/PDGFR-β play important roles in promoting immature and inflammatory intraplaque angiogenesis. PLoS ONE 2018, 13, e0201395. [Google Scholar] [CrossRef]
- Samarkanova, D.; Cox, S.; Hernandez, D.; Rodriguez, L.; Casaroli-Marano, R.P.; Madrigal, A.; Querol, S. Cord Blood Platelet Rich Plasma Derivatives for Clinical Applications in Non-transfusion Medicine. Front Immunol. 2020, 11, 942. [Google Scholar] [CrossRef]
- Valentini, C.G.; Nuzzolo, E.R.; Bianchi, M.; Orlando, N.; Iachininoto, M.G.; Pinci, P.; Teofili, L. Cord Blood Platelet Lysate: In Vitro Evaluation to Support the Use in Regenerative Medicine. Mediterr. J. Hematol. Infect Dis. 2019, 11, e2019021. [Google Scholar]
- Orlando, N.; Pellegrino, C.; Valentini, C.G.; Bianchi, M.; Barbagallo, O.; Sparnacci, S.; Forni, F.; Fontana, T.M.; Teofili, L. Umbilical cord blood: Current uses for transfusion and regenerative medicine. Transfus Apher. Sci. 2020, 59, 102952. [Google Scholar] [CrossRef]
- Jafar, H.; Hasan, M.; Al-Hattab, D.; Saleh, M.; Ameereh, L.A.; Khraisha, S.; Younes, N.; Awidi, A. Platelet lysate promotes the healing of long-standing diabetic foot ulcers: A report of two cases and in vitro study. Heliyon 2020, 6, e03929. [Google Scholar] [CrossRef]
- Volpe, P.; Marcuccio, D.; Stilo, G.; Alberti, A.; Foti, G.; Volpe, A.; Princi, D.; Surace, R.; Pucci, G.; Massara, M. Efficacy of cord blood platelet gel application for enhancing diabetic foot ulcer healing after lower limb revascularization. Semin. Vasc. Surg. 2017, 30, 106–112. [Google Scholar] [CrossRef]
- Mastrogiacomo, M.; Nardini, M.; Collina, M.C.; Di Campli, C.; Filaci, G.; Cancedda, R.; Odorisio, T. Innovative Cell and Platelet Rich Plasma Therapies for Diabetic Foot Ulcer Treatment: The Allogeneic Approach. Front Bioeng. Biotechnol. 2022, 10, 869408. [Google Scholar] [CrossRef]
- Barker, J.N.; Kurtzberg, J.; Ballen, K.; Boo, M.; Brunstein, C.; Cutler, C.; Horwitz, M.; Milano, F.; Olson, A.; Spellman, S.; et al. Optimal Practices in Unrelated Donor Cord Blood Transplantation for Hematologic Malignancies. Biol. Blood Marrow Transplant. 2017, 23, 882–896. [Google Scholar] [CrossRef]
- Pafumi, C.; Leanza, V.; Carbonaro, A.; Leanza, G.; Iemmola, A.; Abate, G.; Stracquadanio, M.G.; D’Agati, A. CD34(+) stem cells from umbilical cord blood. Clin. Pract. 2011, 1, e79. [Google Scholar] [CrossRef]
- Lemos, N.E.; Farias, M.G.; Kubaski, F.; Scotti, L.; Onsten, T.G.H.; Brondani, L.A.; Wagner, S.C.; Sekine, L. Quantification of peripheral blood CD34+ cells prior to stem cell harvesting by leukapheresis: A single center experience. Hematol. Transfus. Cell Ther. 2018, 40, 213–218. [Google Scholar] [CrossRef]
- Barker, J.N.; Mazis, C.M.; Devlin, S.M.; Davis, E.; Maloy, M.A.; Naputo, K.; Nhaissi, M.; Wells, D.; Scaradavou, A.; Politikos, I. Evaluation of Cord Blood Total Nucleated and CD34+ Cell Content, Cell Dose, and 8-Allele HLA Match by Patient Ancestry. Biol. Blood Marrow Transplant. 2020, 26, 734–744. [Google Scholar] [CrossRef]
- Bart, T.; Boo, M.; Balabanova, S.; Fischer, Y.; Nicoloso, G.; Foeken, L.; Oudshoorn, M.; Passweg, J.; Tichelli, A.; Kindler, V.; et al. Impact of selection of cord blood units from the United States and swiss registries on the cost of banking operations. Transfus. Med. Hemother. 2013, 40, 14–20. [Google Scholar] [CrossRef]
- Naing, M.W.; Gibson, D.A.; Hourd, P.; Gomez, S.G.; Horton, R.B.; Segal, J.; Williams, D.J. Improving umbilical cord blood processing to increase total nucleated cell count yield and reduce cord input wastage by managing the consequences of input variation. Cytotherapy 2015, 17, 58–67. [Google Scholar] [CrossRef]
- Meftahpour, V.; Malekghasemi, S.; Baghbanzadeh, A.; Aghebati-Maleki, A.; Pourakbari, R.; Fotouhi, A.; Aghebati-Maleki, L. Platelet lysate: A promising candidate in regenerative medicine. Regen. Med. 2021, 16, 71–85. [Google Scholar] [CrossRef]
- Amable, P.R.; Carias, R.B.; Teixeira, M.V.; da Cruz Pacheco, I.; Corrêa do Amaral, R.J.; Granjeiro, J.M.; Borojevic, R. Platelet-rich plasma preparation for regenerative medicine: Optimization and quantification of cytokines and growth factors. Stem. Cell Res. Ther. 2013, 4, 67. [Google Scholar] [CrossRef]
- Alio, J.L.; Rodriguez, A.E.; WróbelDudzińska, D. Eye platelet-rich plasma in the treatment of ocular surface disorders. Curr. Opin. Ophthalmol. 2015, 26, 325–332. [Google Scholar] [CrossRef]
- Mohamed, H.B.; Abd El-Hamid, B.N.; Fathalla, D.; Fouad, E.A. Current trends in pharmaceutical treatment of dry eye disease: A review. Eur. J. Pharm. Sci. 2022, 175, 106206. [Google Scholar] [CrossRef]
- Lee, Y.; Kim, M.; Galor, A. Beyond dry eye: How co-morbidities influence disease phenotype in dry eye disease. Clin. Exp. Optom. 2022, 105, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Lee, Y.; Mehra, D.; Sabater, A.L.; Galor, A. Dry eye: Why artificial tears are not always the answer. BMJ Open Ophthalmol. 2021, 6, e000697. [Google Scholar] [CrossRef]
- Parazzi, V.; Lazzari, L.; Rebulla, P. Platelet gel from cord blood: A novel tool for tissue engineering. Platelets 2010, 21, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Hübel, J.; Hieronymus, T. HGF/Met-Signaling Contributes to Immune Regulation by Modulating Tolerogenic and Motogenic Properties of Dendritic Cells. Biomedicines 2015, 3, 138–148. [Google Scholar] [CrossRef]
- Mbongue, J.C.; Nicholas, D.A.; Torrez, T.W.; Kim, N.S.; Firek, A.F.; Langridge, W.H. The Role of Indoleamine 2, 3-Dioxygenase in Immune Suppression and Autoimmunity. Vaccines 2015, 3, 703–729. [Google Scholar] [CrossRef]
- Kalinski, P. Regulation of immune responses by prostaglandin E2. J. Immunol. 2012, 188, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Rao, B.; Lou, J.; Li, J.; Liu, Z.; Li, A.; Cui, G.; Ren, Z.; Yu, Z. The Function of the HGF/c-Met Axis in Hepatocellular Carcinoma. Front Cell Dev Biol. 2020, 8, 55. [Google Scholar] [CrossRef] [PubMed]
- Omoto, M.; Suri, K.; Amouzegar, A.; Li, M.; Katikireddy, K.R.; Mittal, S.K.; Chauhan, S.K. Hepatocyte Growth Factor Suppresses Inflammation and Promotes Epithelium Repair in Corneal Injury. Mol. Ther. 2017, 25, 1881–1888. [Google Scholar] [CrossRef]
- Švajger, U.; Rožman, P. Induction of Tolerogenic Dendritic Cells by Endogenous Biomolecules: An Update. Front Immunol. 2018, 29, 2482. [Google Scholar] [CrossRef] [PubMed]
- Boks, M.A.; Kager-Groenland, J.R.; Haasjes, M.S.; Zwaginga, J.J.; van Ham, S.M.; ten Brinke, A. IL-10-generated tolerogenic dendritic cells are optimal for functional regulatory T cell induction–A comparative study of human clinical-applicable DC. Clin. Immunol. 2012, 142, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Ng, T.H.; Britton, G.J.; Hill, E.V.; Verhagen, J.; Burton, B.R.; Wraith, D.C. Regulation of adaptive immunity; the role of interleukin-10. Front Immunol. 2013, 4, 129. [Google Scholar] [CrossRef]
- Wu, H.; Gong, J.; Liu, Y. Indoleamine 2, 3-dioxygenase regulation of immune response (Review). Mol. Med. Rep. 2018, 17, 4867–4873. [Google Scholar] [CrossRef]
- Xu, H.; Zhang, G.X.; Ciric, B.; Rostami, A. IDO: A double-edged sword for T(H)1/T(H)2 regulation. Immunol. Lett. 2008, 121, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Novak, M.L.; Koh, T.J. Macrophage phenotypes during tissue repair. J. Leukoc. Biol. 2013, 93, 875–881. [Google Scholar] [CrossRef] [PubMed]
- Cowper, M.; Frazier, T.; Wu, X.; Curley, L.; Ma, M.H.; Mohiuddin, O.A.; Dietrich, M.; McCarthy, M.; Bukowska, J.; Gimble, J.M. Human Platelet Lysate as a Functional Substitute for Fetal Bovine Serum in the Culture of Human Adipose Derived Stromal/Stem Cells. Cells 2019, 8, 724. [Google Scholar] [CrossRef]
- Guiotto, M.; Raffoul, W.; Hart, A.M.; Riehle, M.O.; di Summa, P.G. Human platelet lysate to substitute fetal bovine serum in hMSC expansion for translational applications: A systematic review. J. Transl. Med. 2020, 18, 351. [Google Scholar] [CrossRef] [PubMed]
- Astori, G.; Amati, E.; Bambi, F.; Bernardi, M.; Chieregato, K.; Schäfer, R.; Sella, S.; Rodeghiero, F. Platelet lysate as a substitute for animal serum for the ex-vivo expansion of mesenchymal stem/stromal cells: Present and future. Stem. Cell Res. Ther. 2016, 7, 93. [Google Scholar] [CrossRef] [PubMed]
- Atashi, F.; Jaconi, M.E.; Pittet-Cuénod, B.; Modarressi, A. Autologous platelet-rich plasma: A biological supplement to enhance adipose-derived mesenchymal stem cell expansion. Tissue Eng. Part C Methods 2015, 21, 253–262. [Google Scholar] [CrossRef]
- Huang, C.J.; Sun, Y.C.; Christopher, K.; Pai, A.S.; Lu, C.J.; Hu, F.R.; Lin, S.Y.; Chen, W.L. Comparison of corneal epitheliotrophic capacities among human platelet lysates and other blood derivatives. PLoS ONE 2017, 12, e0171008. [Google Scholar] [CrossRef]
- Mishan, M.A.; Balagholi, S.; Chamani, T.; Feizi, S.; Soheili, Z.S.; Kanavi, M.R. Potential Effect of Human Platelet Lysate on in vitro Expansion of Human Corneal Endothelial Cells Compared with Y-27632 ROCK Inhibitor. J. Ophthalmic Vis. Res. 2021, 16, 349–356. [Google Scholar] [CrossRef]
- Seidelmann, N.; Duarte Campos, D.F.; Rohde, M.; Johnen, S.; Salla, S.; Yam, G.H.; Mehta, J.S.; Walter, P.; Fuest, M. Human platelet lysate as a replacement for fetal bovine serum in human corneal stromal keratocyte and fibroblast culture. J. Cell Mol. Med. 2021, 25, 9647–9659. [Google Scholar] [CrossRef]
- Mallis, P.; Alevrogianni, V.; Sarri, P.; Velentzas, A.D.; Stavropoulos-Giokas, C.; Michalopoulos, E. Effect of Cord Blood Platelet Gel on wound healing capacity of human Mesenchymal Stromal Cells. Transfus. Apher. Sci. 2020, 59, 102734. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, H.T. MAPK signal pathways in the regulation of cell proliferation in mammalian cells. Cell Res. 2002, 12, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Liu, W.Z.; Liu, T.; Feng, X.; Yang, N.; Zhou, H.F. Signaling pathway of MAPK/ERK in cell proliferation, differentiation, migration, senescence and apoptosis. J. Recept Signal Transduct. Res. 2015, 35, 600–604. [Google Scholar] [CrossRef] [PubMed]
- Zhao, P.; Fu, J.L.; Yao, B.Y.; Jia, Y.R.; Zhou, Z.C. S phase cell percentage normalized BrdU incorporation rate, a new parameter for determining S arrest. Biomed. Environ. Sci. 2014, 27, 215–219. [Google Scholar] [PubMed]
- Terai, K.; Call, M.K.; Liu, H.; Saika, S.; Liu, C.Y.; Hayashi, Y.; Chikama, T.; Zhang, J.; Terai, N.; Kao, C.W.; et al. Crosstalk between TGF-beta and MAPK signaling during corneal wound healing. Invest Ophthalmol. Vis. Sci. 2011, 52, 8208–8215. [Google Scholar] [CrossRef]
- Saika, S.; Okada, Y.; Miyamoto, T.; Yamanaka, O.; Ohnishi, Y.; Ooshima, A.; Liu, C.Y.; Weng, D.; Kao, W.W. Role of p38 MAP kinase in regulation of cell migration and proliferation in healing corneal epithelium. Invest Ophthalmol. Vis. Sci. 2004, 45, 100–109. [Google Scholar] [CrossRef]
- Maltseva, I.; Chan, M.; Kalus, I.; Dierks, T.; Rosen, S.D. The SULFs, extracellular sulfatases for heparan sulfate, promote the migration of corneal epithelial cells during wound repair. PLoS ONE 2013, 8, e69642. [Google Scholar] [CrossRef] [PubMed]
- Xue, M.; Jackson, C.J. Extracellular Matrix Reorganization During Wound Healing and Its Impact on Abnormal Scarring. Adv. Wound Care 2015, 4, 119–136. [Google Scholar] [CrossRef]
- Wang, L.; Wu, X.; Shi, T.; Lu, L. Epidermal growth factor (EGF)-induced corneal epithelial wound healing through nuclear factor κB subtype-regulated CCCTC binding factor (CTCF) activation. J. Biol. Chem. 2013, 288, 24363–24371. [Google Scholar] [CrossRef]
- Vadasz, Z.; Toubi, E. Semaphorins: Their dual role in regulating immune-mediated diseases. Clin. Rev. Allergy Immunol. 2014, 47, 17–25. [Google Scholar] [CrossRef]
- Takamatsu, H.; Okuno, T.; Kumanogoh, A. Regulation of immune cell responses by semaphorins and their receptors. Cell Mol. Immunol. 2010, 7, 83–88. [Google Scholar] [CrossRef]
- Available online: https://www.ema.europa.eu/en/documents/scientific-guideline/note-guidance-plasma-derived-medicinal-products (accessed on 20 September 2022).
- Samarkanova, D.; Martin, S.; Bisbe, L.; Puig, J.; Calatayud-Pinuaga, M.; Rodriguez, L.; Azqueta, C.; Coll, R.; Casaroli-Marano, R.; Madrigal, A.; et al. Barcelona CBED Study Group (Appendix I). Clinical evaluation of allogeneic eye drops from cord blood platelet lysate. Blood Transfus. 2021, 19, 347–356. [Google Scholar] [PubMed]
| Gene | Forward Primer | Reverse Primer | Size |
|---|---|---|---|
| Collagen 1 A1 (Col1A1) | GCTCCTCTTAGGGGCCACT | GCTCCTCTTAGGGGCCACT | 91 |
| Syndecan type-1 | TCAGGGGATGACGACTCGTTT | CTCCTGCTCGAAGTAGCCAGA | 81 |
| Perlecan | TTCCAGATGGTCTATTTCCGGG | CTTGGCACTTGCATCCTCC | 84 |
| b-actin | AGCTTCGGCACATATTTCATCTG | CGTTCACTCCCATGACAAACA | 89 |
| n = 25 | CBUs | CB-PC | CB-PPP | p-Value |
|---|---|---|---|---|
| Net Volume (mL) | 139.4 ± 5.8 | 10.4 ± 1.1 | 20.7 ± 2.6 | <0.001 |
| WBCs (×103/μL) | 9.2 ± 1.9 | 1.3 ± 0.6 | 0.23 ± 0.2 | <0.001 |
| RBCs (×106/μL) | 3.6 ± 1.1 | <0.1 | <0.1 | <0.001 |
| HgB (g/dL) | 11.3 ± 1.1 | <0.1 | <0.1 | <0.001 |
| PLTs (×103/μL) | 202.2 ± 27.9 | 1120.3 ± 70.4 | 16.1 ± 7.2 | <0.001 |
| PLTs (×109) | 28.1 ± 3.9 | 12.1 ± 1.5 | 0.3 ± 0.1 | <0.001 |
| PLTs Recovery (%) | - | 42.3 ± 8.2 | 1.1 ± 0.3 | <0.001 |
| (n = 25) | CBUs | CB-PL | CB-PPP | p-Value |
|---|---|---|---|---|
| pH | 7.0 ± 0.7 | 7.1 ± 0.4 | 7.2 ± 0.6 | NS |
| Lactate (mmol/l) | 2.0 ± 0.7 | 2.1 ± 0.5 | 2.1 ± 0.4 | NS |
| Glucose (mg/ dl) | 354.5 ± 76.3 | 369.5 ± 83.3 | 355.7 ± 79.8 | NS |
| PCO2 (mm Hg) | 30.1 ± 2.2 | 31.4 ± 2.3 | 32.3 ± 3.1 | NS |
| PO2 (mm Hg) | 32.2 ± 2.4 | 30.5 ± 1.2 | 31.8 ± 3.2 | NS |
| PT (sec) | 14.2 ± 1.4 | 13.7 ± 3.2 | 14.7 ± 2.4 | NS |
| APTT (sec) | 34.3 ± 2.4 | 32.1 ± 4.1 | 34.3 ± 3.9 | NS |
| Fibrinogen (mg/mL) | 150.2 ± 15.2 | 151.3 ± 13.3 | 151.4 ± 12.1 | NS |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mallis, P.; Michalopoulos, E.; Sarri, E.F.; Papadopoulou, E.; Theodoropoulou, V.; Katsimpoulas, M.; Stavropoulos-Giokas, C. Evaluation of the Regenerative Potential of Platelet-Lysate and Platelet-Poor Plasma Derived from the Cord Blood Units in Corneal Wound Healing Applications: An In Vitro Comparative Study on Corneal Epithelial Cells. Curr. Issues Mol. Biol. 2022, 44, 4415-4438. https://doi.org/10.3390/cimb44100303
Mallis P, Michalopoulos E, Sarri EF, Papadopoulou E, Theodoropoulou V, Katsimpoulas M, Stavropoulos-Giokas C. Evaluation of the Regenerative Potential of Platelet-Lysate and Platelet-Poor Plasma Derived from the Cord Blood Units in Corneal Wound Healing Applications: An In Vitro Comparative Study on Corneal Epithelial Cells. Current Issues in Molecular Biology. 2022; 44(10):4415-4438. https://doi.org/10.3390/cimb44100303
Chicago/Turabian StyleMallis, Panagiotis, Efstathios Michalopoulos, Eirini Faidra Sarri, Elena Papadopoulou, Vasiliki Theodoropoulou, Michalis Katsimpoulas, and Catherine Stavropoulos-Giokas. 2022. "Evaluation of the Regenerative Potential of Platelet-Lysate and Platelet-Poor Plasma Derived from the Cord Blood Units in Corneal Wound Healing Applications: An In Vitro Comparative Study on Corneal Epithelial Cells" Current Issues in Molecular Biology 44, no. 10: 4415-4438. https://doi.org/10.3390/cimb44100303
APA StyleMallis, P., Michalopoulos, E., Sarri, E. F., Papadopoulou, E., Theodoropoulou, V., Katsimpoulas, M., & Stavropoulos-Giokas, C. (2022). Evaluation of the Regenerative Potential of Platelet-Lysate and Platelet-Poor Plasma Derived from the Cord Blood Units in Corneal Wound Healing Applications: An In Vitro Comparative Study on Corneal Epithelial Cells. Current Issues in Molecular Biology, 44(10), 4415-4438. https://doi.org/10.3390/cimb44100303









