Protective Effect of Melatonin Administration against SARS-CoV-2 Infection: A Systematic Review
Abstract
:1. Introduction
2. Material and Methods
- Trip Medical Database.
- Cochrane Library.
- PubMed.
- MedlinePlus.
- BVS.
- Cinahl.
- Cuiden.
- Dialnet.
- Scholar Google.
- Patient: people of any age and sex infected by the SARS-CoV 2 virus.
- Intervention: intervention program through the administration of melatonin to prevent infection by SARS-CoV 2 and the development of COVID-19.
- Comparation: compare the currently available treatment protocols of etiological active ingredients and vaccines with the administration of melatonin as a preventive of COVID-19.
- Outcomes: raise the results that are to be obtained through the proposed intervention to plan the objectives of the work, such as reducing the incidence of COVID-19 infection and the variability of the results according to parameters by population groups (age, sex and previous pathologies).
- (1)
- Full text documents.
- (2)
- Language: Spanish and English.
- (3)
- Less than 10 years old, except for those documents that present relevant information and that are not obsolete.
- (4)
- Works focused on the administration of substances with preventive power of the COVID-19 infection, especially those that refer to melatonin.
- (5)
- Case or group studies, books, articles published in prestigious scientific journals, conference proceedings and communications from official bodies in the health field.
- (1)
- Documents without scientific validity.
- (2)
- Documents with high risk of bias.
- (3)
- Opinion articles.
- (4)
- Studies with a follow-up time of less than 1 month.
3. Results
4. Discussion
5. Conclusions
- (1)
- Melatonin is a simple molecule, with well-documented pathophysiological functions, such as: the anti-inflammatory, antioxidant, and immunomodulatory action, as well as its inhibitory capacity of the Mpro protease and the MMP9 protein, which would make it a therapeutic alternative to consider against various infectious diseases.
- (2)
- It has been known for years and it has been demonstrated once again with the administration of vaccines that the administration of melatonin can enhance the immune response, with a response in the rate of specific antibodies much higher than when the vaccine components are administered without melatonin.
- (3)
- Before the onset of this pandemic, it had been shown in various viral infections that it could inhibit and/or mitigate the pathogenic action of these microbial agents in experimental animals.
- (4)
- When melatonin is used in the laboratory (animal experimentation) and in the human clinic, a very wide safety margin has been demonstrated, well above most of the drugs used in ICUs against SARS-CoV-2 infection.
- (5)
- We believe that there is a sufficient level of scientific evidence to authorize its use as a preventive drug against COVID-19 infection, due to its proven physiological actions, although it must be said that the exact dose to achieve this preventive effect has still not been determined.
- (6)
- In infected patients with progressive disease, the scientific evidence is clear, and its administration is recommended for several reasons: (a) because it has been able to significantly reduce the consequences of the disease; (b) because there are no studies that say otherwise; (c) because its security profile is very broad.
- (7)
- Although several administration guidelines have already been published in patients infected with SARS-CoV-2, it would be advisable to launch new clinical trials to define the best administration protocol, especially regarding dose and times when it should be administered, to respect its circadian rhythmicity.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Trivedi, N.; Verma, A.; Kumar, D. Possible treatment and strategies for COVID-19: Review and assessment. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 12593–12608. [Google Scholar] [CrossRef] [PubMed]
- Johns Hopkins University. COVID-19 Dashboard by the Center for Systems Science and Engineering (CSSE). Available online: https://coronavirus.jhu.edu/map.html (accessed on 1 December 2021).
- Dong, E.; Du, H.; Gardner, L. An Interactive Web-Based Dashboard to Track COVID-19 in Real Time. Lancet Infect. Dis. 2020, 20, 533–534. [Google Scholar] [CrossRef]
- World Health Organization. Coronavirus Disease (COVID-19)-Situation Report. 2021. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports (accessed on 6 February 2021).
- Jeyanathan, M.; Afkhami, S.; Smaill, F.; Miller, M.S.; Lichty, B.D.; Xing, Z. Immunological considerations for COVID-19 vaccine strategies. Nat. Rev. Immunol. 2020, 20, 615–632. [Google Scholar] [CrossRef] [PubMed]
- Oran, D.P.; Topol, E.J. Prevalence of asymptomatic SARS-CoV-2 infection: A narrative review. Ann. Intern. Med. 2020, 173, 362–367. [Google Scholar] [CrossRef]
- World Health Organization. What Happens to People Who Get Seriously Ill? Available online: https://www.who.int/news-room/q-a-detail/q-a-coronaviruses (accessed on 15 October 2020).
- Gibbons, C.L.; Mangen, M.-J.J.; Plass, D.; Havelaar, A.H.; Brooke, R.J.; Kramarz, P.; Peterson, K.L.; Stuurman, A.L.; Cassini, A.; Fèvre, E.M.; et al. Measuring underreporting and under-ascertainment in infectious disease datasets: A comparison of methods. BMC Public Health 2014, 14, 147. [Google Scholar] [CrossRef] [Green Version]
- Russell, T.W.; Golding, N.; Hellewell, J.; Abbott, S.; Wright, L.; Pearson, C.A.B.; Van Zandvoort, K.; Jarvis, C.I.; Gibbs, H.; Liu, Y.; et al. Reconstructing the early global dynamics of under-ascertained COVID-19 cases and infections. BMC Med. 2020, 18, 332. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Immune Responses and Immunity to SARS-CoV-2. Available online: https://www.ecdc.europa.eu/en/covid-19/latest-evidence/immune-responses (accessed on 15 October 2020).
- Anand, S.; Montez-Rath, M.; Han, J.; Bozeman, J.; Kerschmann, R.; Beyer, P.; Parsonnet, J.; Chertow, G.M. Prevalence of SARS-CoV-2 antibodies in a large nationwide sample of patients on dialysis in the USA: A cross-sectional study. Lancet 2020, 396, 1335–1344. [Google Scholar] [CrossRef]
- Iwasaki, A. What reinfections mean for COVID-19. Lancet Infect. Dis. 2021, 21, 3–5. [Google Scholar] [CrossRef]
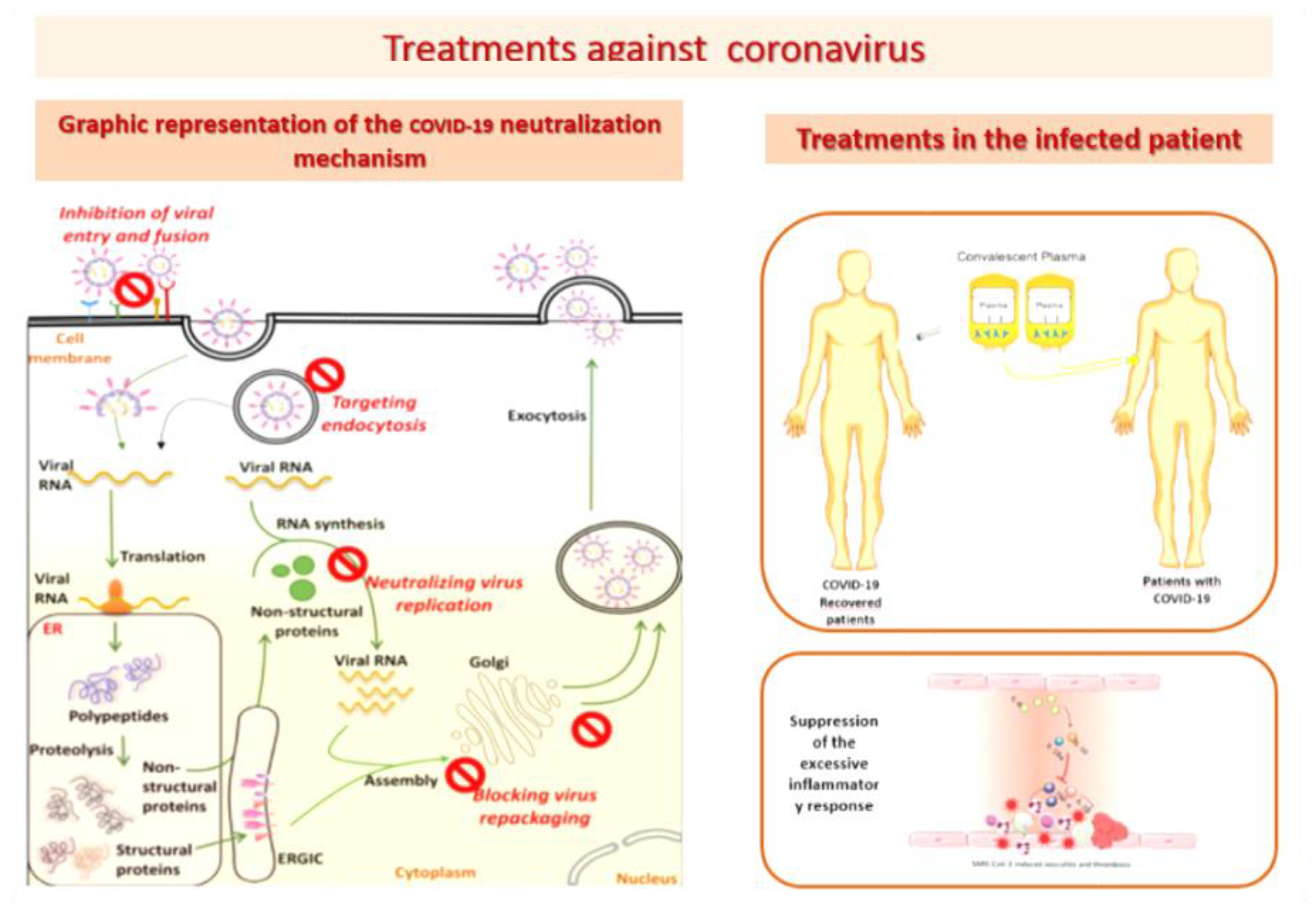
- Liu, S.T.H.; Lin, H.-M.; Baine, I.; Wajnberg, A.; Gumprecht, J.P.; Rahman, F.; Rodriguez, D.; Tandon, P.; Bassily-Marcus, A.; Bander, J.; et al. Convalescent plasma treatment of severe COVID-19: A propensity score-matched control study. Nat. Med. 2020, 26, 1708–1713. [Google Scholar] [CrossRef]
- Watanabe, Y.; Allen, J.D.; Wrapp, D.; McLellan, J.S.; Crispin, M. Site-specific glycan analysis of the SARS-CoV-2 spike. Science 2020, 369, 330–333. [Google Scholar] [CrossRef]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.-H.; Nitsche, A.; et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 2020, 181, 271–280. [Google Scholar] [CrossRef]
- Chi, X.; Yan, R.; Zhang, J.; Zhang, G.; Zhang, Y.; Hao, M.; Zhang, Z.; Fan, P.; Dong, Y.; Yang, Y.; et al. A neutralizing human antibody binds to the N-terminal domain of the Spike protein of SARS-CoV-2. Science 2020, 369, 650–655. [Google Scholar] [CrossRef]
- Liu, L.; Wang, P.; Nair, M.S.; Yu, J.; Rapp, M.; Wang, Q.; Luo, Y.; Chan, J.F.; Sahi, V.; Figueroa, A.; et al. Potent neutralizing antibodies against multiple epitopes on SARS-CoV-2 spike. Nature 2020, 584, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Wrapp, D.; Wang, N.; Corbett, K.S.; Goldsmith, J.A.; Hsieh, C.-L.; Abiona, O.; Graham, B.S.; McLellan, J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science 2020, 367, 1260–1263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walls, A.C.; Park, Y.-J.; Tortorici, M.A.; Wall, A.; McGuire, A.T.; Veesler, D. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell 2020, 181, 281–292. [Google Scholar] [CrossRef]
- World Health Organization. Draft Landscape of COVID-19 Candidate Vaccines. Available online: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines (accessed on 29 January 2021).
- Wise, J. COVID-19: European countries suspend use of Oxford-AstraZeneca vaccine after reports of blood clots. BMJ 2021, 372, 699. [Google Scholar] [CrossRef]
- Joint CDC and FDA Statement on Johnson & Johnson COVID-19 Vaccine. Available online: https://www.fda.gov/news-events/press-announcements/joint-cdc-and-fda-statement-johnson-johnson-covid-19-vaccine (accessed on 14 April 2021).
- Greinacher, A.; Thiele, T.; Warkentin, T.E.; Weisser, K.; Kyrle, P.A.; Eichinger, S. Thrombotic thrombocytopenia after ChAdOx1 nCov-19 vaccination. N. Engl. J. Med. 2021, 384, 2092–2101. [Google Scholar] [CrossRef]
- Schultz, N.H.; Sorvoll, I.H.; Michelsen, A.E.; Munthe, L.A.; Lund-Johansen, F.; Ahlen, M.T. Thrombosis and thrombocytopenia after ChAdOx1 nCoV-19 vaccination. N. Engl. J. Med. 2021, 384, 2124–2130. [Google Scholar] [CrossRef]
- Connors, M.; Graham, B.S.; Lane, H.C.; Fauci, A.S. SARS-CoV-2 vaccines: Much accomplished, much to learn. Ann. Intern. Med. 2021. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.P.; Gupta, V. COVID-19 vaccine: A comprehensive status report. Virus Res. 2020, 288, 198114. [Google Scholar] [CrossRef]
- Bakhiet, M.; Taurin, S. SARS-CoV-2: Targeted managements and vaccine development. Cytokine Growth Factor Rev. 2021, 58, 16–29. [Google Scholar] [CrossRef] [PubMed]
- Addetia, A.; Crawford, K.H.D.; Dingens, A.; Zhu, H.; Roychoudhury, P.; Huang, M.L. Neutralizing antibodies correlate with protection from SARS-CoV-2 in humans during a fishery vessel outbreak with a high attack rate. J. Clin. Microbiol. 2020, 58, e02107-20. [Google Scholar] [CrossRef]
- Watanabe, Y.; Mendonça, L.; Allen, E.R.; Howe, A.; Lee, M.; Allen, J.D.; Chawla, H.; Pulido, D.; Donnellan, F.; Davies, H.; et al. Native-like SARS-CoV-2 spike glycoprotein expressed by ChAdOx1 nCoV-19/AZD1222 vaccine. ACS Cent. Sci. 2021, 7, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Collignon, C.; Bol, V.; Chalon, A.; Surendran, N.; Morel, S.; van den Berg, R.A. Innate immune responses to chimpanzee adenovirus vector 155 vaccination in mice and mon-keys. Front. Immunol. 2020, 11, 579872. [Google Scholar] [CrossRef] [PubMed]
- Beeraka, N.M.; Sadhu, S.P.; Madhunapantula, S.V.; Pragada, R.R.; Svistunov, A.A.; Nikolenko, V.N.; Mikhaleva, L.M.; Aliev, G. Strategies for Targeting SARS-CoV-2: Small Molecule Inhibitors-The Current Status. Front. Immunol. 2020, 11, 552925. [Google Scholar] [CrossRef]
- Anand, U.; Jakhmola, S.; Indari, O.; Jha, H.C.; Chen, Z.S.; Tripathi, V.; Pérez de la Lastra, J.M. Potential Therapeutic Targets and Vaccine Development for SARS-CoV-2/COVID-19 Pandemic Management: A Review on the Recent Update. Front. Immunol. 2021, 12, 658519. [Google Scholar] [CrossRef]
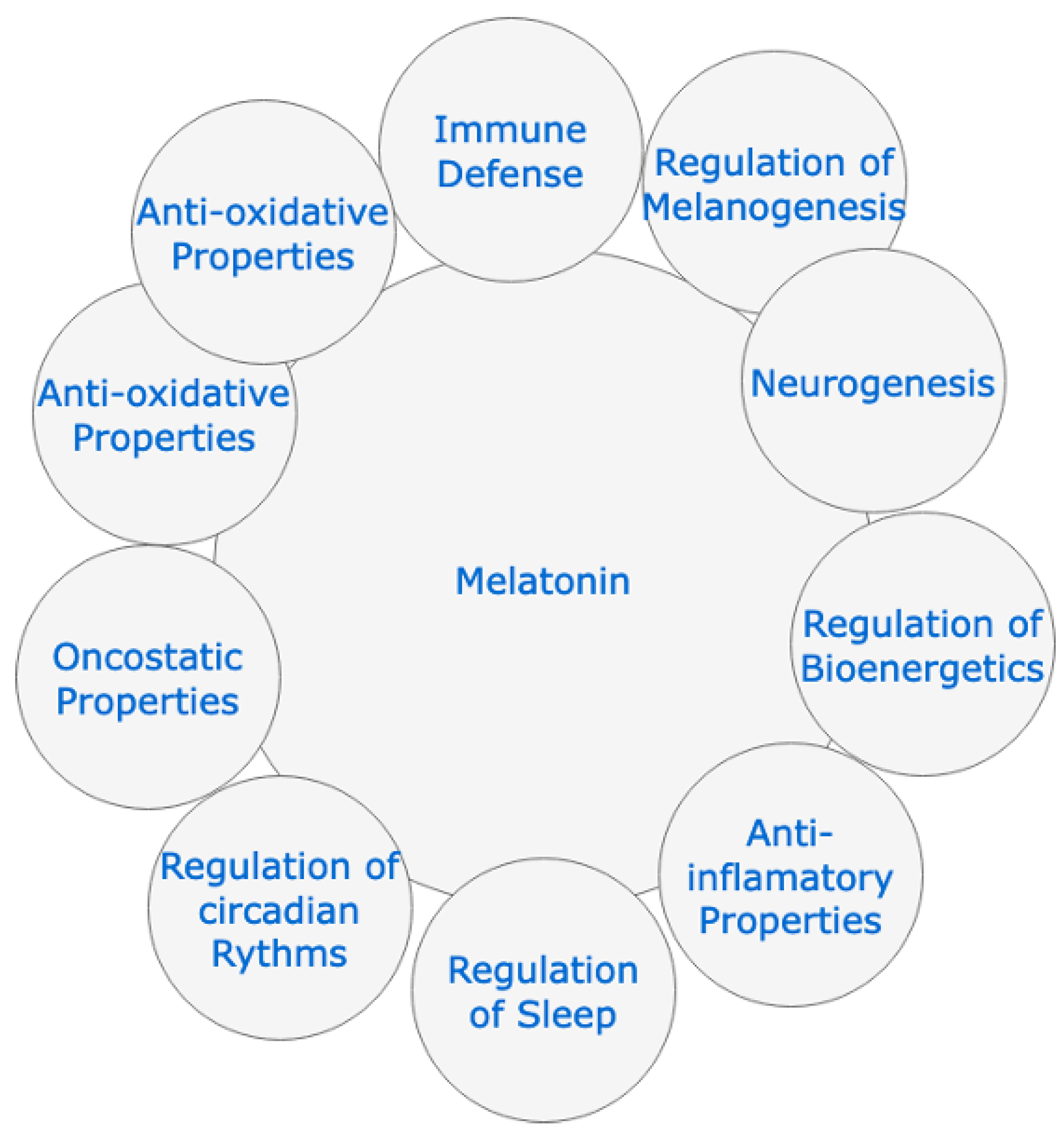
- Anderson, G.; Reiter, R. Melatonin: Roles in influenza, COVID-19, and other viral infections. Rev. Med. Virol. 2020, 30, 1–10. [Google Scholar] [CrossRef]
- Kleszczyński, K.; Slominski, A.T.; Steinbrink, K.; Reiter, R.J. Clinical Trials for Use of Melatonin to Fight against COVID-19 Are Urgently Needed. Nutrients 2020, 12, 2561. [Google Scholar] [CrossRef]
- Karamitri, A.; Jockers, R. Melatonin in type 2 diabetes mellitus and obesity. Nat. Rev. Endocrinol. 2019, 15, 105–125. [Google Scholar] [CrossRef]
- Alghamdi, B.S. The neuroprotective role of melatonin in neurological disorders. J. Neurosci. Res. 2018, 96, 1136–1149. [Google Scholar] [CrossRef]
- Sehirli, A.O.; Sayiner, S.; Serakinci, N. Role of melatonin in the treatment of COVID-19; as an adjuvant through cluster differentiation 147 (CD147). Mol. Biol. Rep. 2020, 47, 8229–8233. [Google Scholar] [CrossRef] [PubMed]
- Feitosa, E.L.; Júnior, F.T.D.S.S.; Nery Neto, J.A.O.; Matos, L.F.L.; Moura, M.H.S.; Rosales, T.O.; De Freitas, G.B.L. COVID-19: Rational discovery of the therapeutic potential of Melatonin as a SARS-CoV-2 main Protease Inhibitor. Int. J. Med. Sci. 2020, 17, 2133–2146. [Google Scholar] [CrossRef] [PubMed]
- Hazra, S.; Chaudhuri, A.G.; Tiwary, B.K.; Chakrabarti, N. Matrix metallopeptidase 9 as a host protein target of chloroquine and melatonin for immunoregulation in COVID-19: A network-based meta-analysis. Life Sci. 2020, 257, 118096. [Google Scholar] [CrossRef] [PubMed]
- Martín Giménez, V.M.; Prado, N.; Diez, E.; Manucha, W.; Reiter, R.J. New proposal involving nanoformulated melatonin targeted to the mitochondria as a potential COVID-19 treatment. Nanomedicine 2020, 15, 2819–2821. [Google Scholar] [CrossRef]
- Zhang, R.; Wang, X.; Ni, L.; Di, X.; Ma, B.; Niu, S.; Liu, C.; Reiter, R.J. COVID-19Melatonin as a potential adjuvant treatment. Life Sci. 2020, 250, 117583. [Google Scholar] [CrossRef]
- Brusco, L.; Cruz, P.; Cangas, A.; Gonzalez Rojas, C.; Vigo, D.E.; Cardinali, D.P. Efficacy of melatonin in non-intensivecare unit patients with COVID-19 pneumonia and sleep dysregulation. Melatonin Res. 2021, 4, 173–188. [Google Scholar] [CrossRef]
- Cardinali, D.; Brown, G.; Pandi-Perumal, S.R. An urgent proposal for the immediate use of melatonin as an adjuvant to anti-SARS-CoV-2 vaccination. Melatonin Res. 2021, 4, 206–212. [Google Scholar] [CrossRef]
- Bahrampour Juybari, K.; Pourhanifeh, M.H.; Hosseinzadeh, A.; Hemati, K.; Mehrzadi, S. Melatonin potentials against viral infections including COVID-19: Current evidence and new findings. Virus Res. 2020, 287, 198108. [Google Scholar] [CrossRef]
- Acuña-Castroviejo, D.; Escames, G.; Figueira, J.C.; de la Oliva, P.; Borobia, A.M.; Acuña-Fernández, C. Clinical trial to test the efficacy of melatonin in COVID-19. J. Pineal. Res. 2020, 69, e12683. [Google Scholar] [CrossRef]
- Urrútia, G.; Bonfill, X. La declaración PRISMA: Un paso adelante en la mejora de las publicaciones de la Revista Española de Salud Pública. Rev. Esp. Salud. Pública 2013, 87, 99–102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pardal-Refoyo, J.L.; Pardal-Peláez, B. Anotaciones para estructurar una revisión sistemática. Rev. ORL 2020, 11, 155–160. [Google Scholar]
- Kim, M.S.; An, M.H.; Kim, W.J.; Hwang, T.H. Comparative efficacy and safety of pharmacological interventions for the treatment of COVID-19: A systematic review and network meta-analysis. PLoS Med. 2020, 17, e1003501. [Google Scholar] [CrossRef]
- Tesarik, J.M.D.; Reiter, R.J. Melatonin as an Adjuvant in COVID-19 Vaccination. J. SARS-CoV-2 Coronavirus Dis. 2021, 1, 39. [Google Scholar]
- Cardinali, D.P.; Brown, G.M.; Pandi-Perumal, S.R. Can Melatonin Be a Potential “Silver Bullet” in Treating COVID-19 Patients? Diseases 2020, 8, 44. [Google Scholar] [CrossRef] [PubMed]
- Maestroni, G. Exogenous melatonin as potential adjuvant in anti-SarsCov2 vaccines. J. Neuroimmune. Pharm. 2020, 15, 572–573. [Google Scholar] [CrossRef]
- Muñoz-Hoyos, A.; Bonillo-Perales, A.; Avila-Villegas, R.; González-Ripoll, M.; Uberos, J.; Florido-Navío, J.; Molina-Carballo, A. Melatonin levels during the first week of life and their relation with the antioxidant response in the perinatal period. Neonatology 2007, 92, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Zhang, M.; Yin, L.; Wang, K.; Zhou, Y.; Zhou, M.; Lu, Y. COVID-19 treatment: Close to a cure? A rapid review of pharmacotherapies for the novel coronavirus (SARS-CoV-2). Int. J. Antimicrob. Agents 2020, 56, 106080. [Google Scholar] [CrossRef]
- Becker, R.C. COVID-19 treatment update: Follow the scientific evidence. J. Thromb. Thrombolysis 2020, 50, 43–53. [Google Scholar] [CrossRef] [Green Version]
- Boozari, M.; Hosseinzadeh, H. Natural products for COVID-19 prevention and treatment regarding to previous coronavirus infections and novel studies. Phytoth. Res. 2021, 35, 864–876. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Han, M.; Li, T.; Sun, W.; Wang, D.; Fu, B.; Zhou, Y.; Zheng, X.; Yang, Y.; Li, X.; et al. Effective treatment of severe COVID-19 patients with tocilizumab. Proc. Natl. Acad. Sci. USA 2020, 117, 10970–10975. [Google Scholar] [CrossRef]
- Reiter, R.J.; Abreu-González, P.; Marik, P.E.; Dominguez-Rodriguez, A. Therapeutic Algorithm for Use of Melatonin in Patients With COVID-19. Front Med. 2020, 7, 226. [Google Scholar] [CrossRef]
- Zúñiga-Blanco, B.L.; Pruneda-Álvarez, L.; Enríquez-Macías, M.L.; Fyda, J. Nuevas perspectivas para el tratamiento ambulatorio o en etapas tempranas de COVID-19. Med. Int. Méx. 2020, 36, 323–331. [Google Scholar]
- Ben-Nathan, D.; Maestroni, G.J.; Lustig, S.; Conti, A. Protective effects of melatonin in mice infected with encephalitis viruses. Arch. Virol. 1995, 140, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Öztürk, G.; Akbulut, K.G.; Güney, Ş. Melatonin, aging, and COVID-19: Could melatonin be beneficial for COVID-19 treatment in the elderly? Turk. J. Med. Sci. 2020, 50, 1504–1512. [Google Scholar] [CrossRef] [PubMed]
- Artigas, L.; Coma, M.; Matos-Filipe, P.; Aguirre-Plans, J.; Farrés, J.; Valls, R.; Fernández-Fuentes, N.; de la Haba-Rodríguez, J.; Olvera, A.; Barbera, J.; et al. In-silico drug repurposing study predicts the combination of pirfenidone and melatonin as a promising candidate therapy to reduce SARS-CoV-2 infection progression and respiratory distress caused by cytokine storm. PLoS ONE 2020, 15, e0240149. [Google Scholar]
- Zeng, X.; Song, X.; Ma, T.; Pan, X.; Zhou, Y.; Hou, Y.; Cheng, F. Repurpose open data to discover therapeutics for COVID-19 using deep learning. J. Proteome Res. 2020, 19, 4624–4636. [Google Scholar] [CrossRef] [PubMed]
- Montalvan, V.; Lee, J.; Bueso, T.; De Toledo, J.; Rivas, K. Neurological manifestations of COVID-19 and other coronavirus infections: A systematic review. Clin. Neurol. Neurosurg. 2020, 194, 105921. [Google Scholar] [CrossRef]
- Lin, J.E.; Asfour, A.; Sewell, T.B.; Hooe, B.; Pryce, P.; Earley, C.; Shen, M.Y.; Kerner-Rossi, M.; Thakur, K.T.; Vargas, W.S.; et al. Neurological issues in children with COVID-19. Neurosci. Lett. 2021, 743, 135567. [Google Scholar] [CrossRef]
- Verstrepen, K.; Baisier, L.; De Cauwer, H. Neurological manifestations of COVID-19, SARS and MERS. Acta Neurol. Belg. 2020, 120, 1051–1060. [Google Scholar] [CrossRef]
- Orsucci, D.; Ienco, E.C.; Nocita, G.; Napolitano, A.; Vista, M. Neurological features of COVID-19 and their treatment: A review. Drugs Context. 2020, 9, 1–12. [Google Scholar] [CrossRef]
- Molina-Carballo, A.; Muñoz-Hoyos, A.; Reiter, R.J.; Sánchez-Forte, M.; Moreno-Madrid, F.; Rufo-Campos, M.; Molina-Font, J.A.; Acuña-Castroviejo, D. Utility of high doses of melatonin as adjunctive anticonvulsant therapy in a child with severe myoclonic epilepsy: Two years’ experience. J. Pineal Res. 1997, 23, 97–105. [Google Scholar] [CrossRef]
- Muñoz-Hoyos, A.; Sánchez-Forte, M.; Molina-Carballo, A.; Escames, G.; Martin-Medina, E.; Reiter, R.J.; Molina-Font, J.A.; Acuña-Castroviejo, D. Melatonin’s role as an anticonvulsant and neuronal protector: Experimental and clinical evidence. J. Child Neurol. 1998, 13, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.; Tang, L.; Garcia, J.J.; Muñoz-Hoyos, A. Pharmacological actions of melatonin in oxygen radical pathophysiology. Life Sci. 1997, 60, 2255–2271. [Google Scholar] [CrossRef]
- Checa-Ros, A.; Muñoz-Hoyos, A.; Molina-Carballo, A.; Muñoz-Gallego, A.; Narbona-Galdó, S.; Jerez-Calero, A.; Augustín-Morales, M.D.C. Analysis of Different Melatonin Secretion Patterns in Children with Sleep Disorders: Melatonin Secretion Patterns in Children. J. Child Neurol. 2017, 32, 1000–1008. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, A.; Palomo, J.P.; Jerez-Calero, A. Use of an ANN to Value MTF and Melatonin Effect on ADHD Affected Children. IEEE Acces. 2019, 7, 127254–127264. [Google Scholar] [CrossRef]
- Acuña-Castroviejo, D.; Escames, G.; Macías, M.; Muñóz Hoyos, A.; Molina Carballo, A.; Arauzo, M.; Montes, R. Cell protective role of melatonin in the brain. J. Pineal Res. 1995, 19, 57–63. [Google Scholar] [CrossRef] [PubMed]
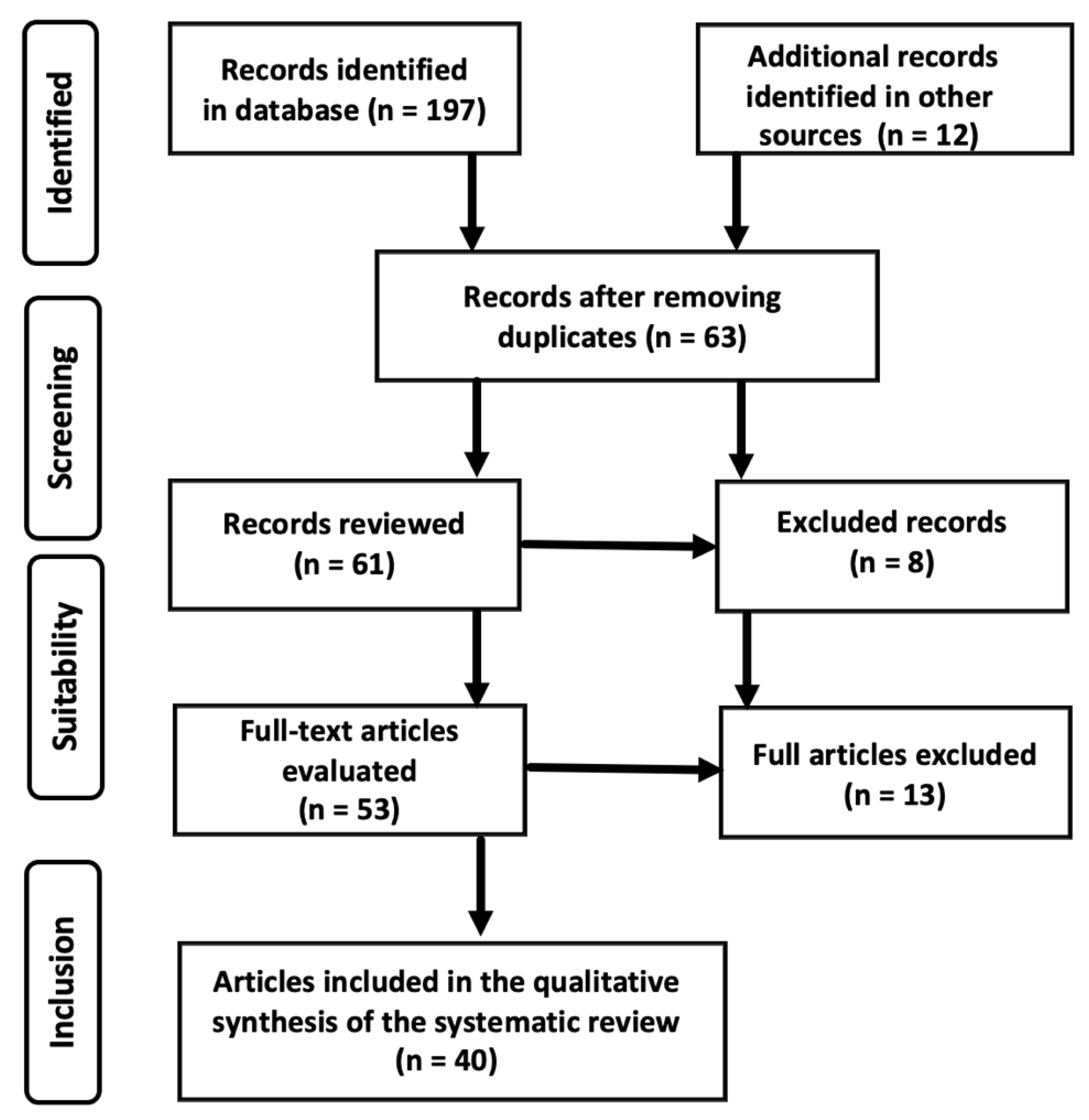
| Database | Articles Found | Articles Rejected for Content | Nº of Duplicate Items | Nº of Items Used |
|---|---|---|---|---|
| PUB MED | 83 | 53 | 9 | 21 |
| SCOPUS | 18 | 9 | 4 | 5 |
| DIALNET | 42 | 30 | 5 | 7 |
| SCIELO | 27 | 19 | 2 | 6 |
| GOOGLE SCHOLAR | 27 | 23 | 3 | 1 |
| Total | 197 | 134 | 23 | 40 |
| Author/Journal | Objective | Type of Study | Q | Results | Conclusions |
|---|---|---|---|---|---|
| Brusco L et al. Melatonin Res. [42] | To analyze the therapeutic potential of melatonin to counteract the consequences of COVID-19 infection. | Review of clinical trials | A | The efficacy of melatonin at a dose of 9 mg/day is suggested to reduce the ICU stay of patients with pneumonia associated with COVID-19. | The importance of maintaining normal sleep and circadian rhythm in patients is confirmedInfected by COVID-19 in ICU. |
| Cardinali D, et al. Melatonin Res. [43] | Check for antigen reduction in individuals who experience partial or total loss of sleep prior to vaccination. | Longitudinal and prospective analytical observational. | A | The use of exogenous melatonin increases the potency of the immune response induced by the vaccine by increasing peripheral blood CD4 + T cells and B cells expressing Ig G | The administration of melatonin between 2 days before vaccination and 4 weeks after it may be an ideal complement to improve the effectiveness of vaccination against SARS-CoV-2. |
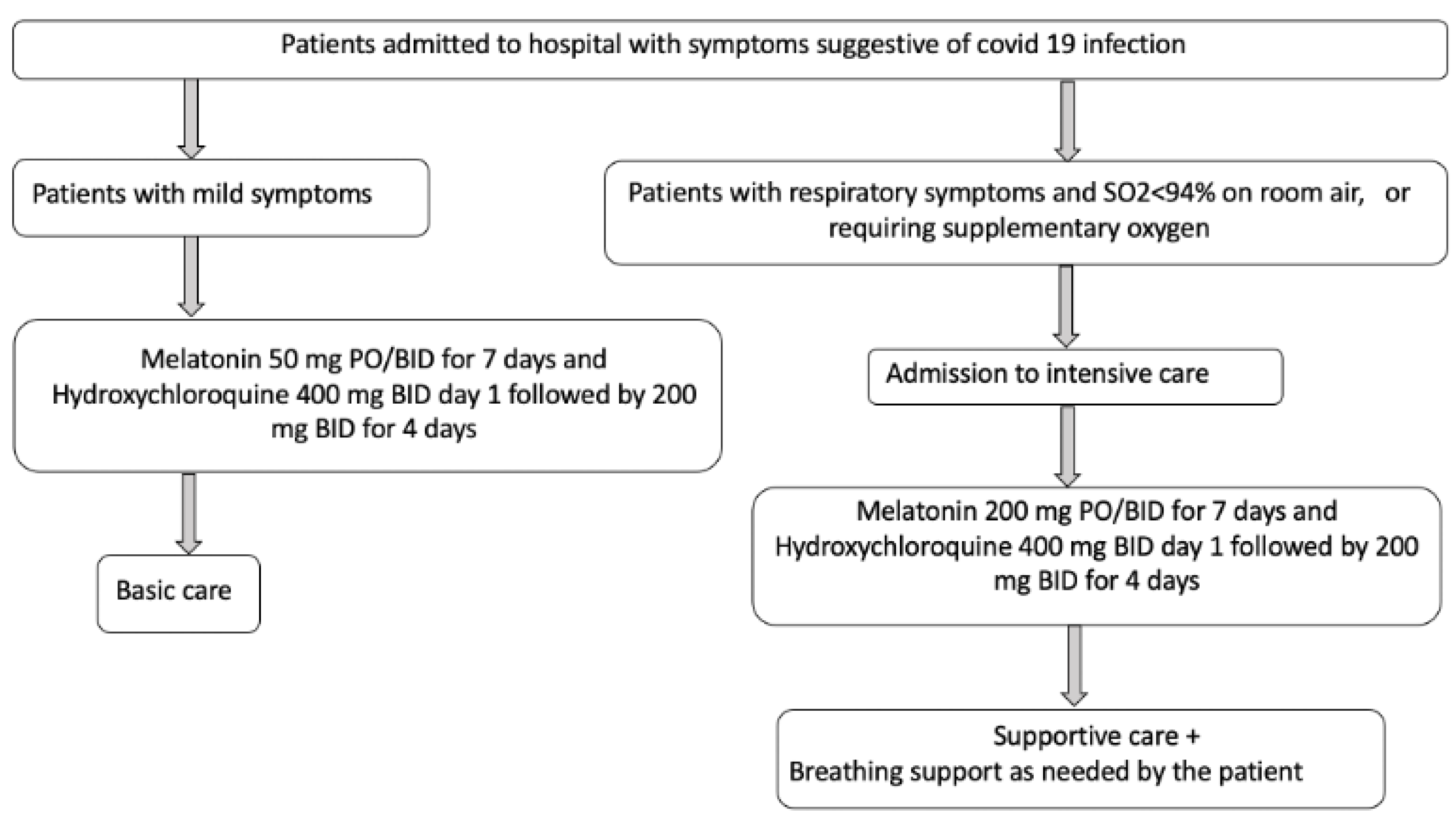
| Reiter RJ et al. Front Med (Lausanne). [48] | Identify safe and currently available molecules that can be used to slow or treat COVID-19 disease. | Longitudinal and prospective analytical observational. | A | Development of a therapeutic algorithm for the use of melatonin in patients with COVID-19 | Melatonin should be considered for prophylactic use or treatment alone, or in combination with other drugs, and they propose a therapeutic algorithm for use in patients. In addition to its easy availability, it can be easily synthesized in large quantities, it is cheap, it has a very high safety profile, and it can be self-administered. |
| Zúñiga-Blanco BL, et al. Med Int Méx. [49] | Develop new guidelines to treat symptomatic patients and reduce viral load in asymptomatic carrier patients | Individual experimental study with sick subjects: clinical trial | M | Until effective antiviral treatment is available, early indication of prophylaxis with hydroxychloroquine is recommended. | In addition to cardiovascular support treatment, it is suggested to assess the indication of melatonin and tocilizumab to reduce clinical symptoms and the deterioration of patients with severe COVID-19 |
| Ben-Nathan D, et al. Arch Virol. [50] | To study the effect of the pineal neurohormone melatonin (MLT) on protection against viral encephalitis | Experimental with mice | M | Injections of MLT to mice (10 PFU) reduced mortality in mice from 100% to 44%. By increasing the dose (100PFU) mortality decreased by 20%. | MLT’s Efficacy to Protect Against Lethal Viral Infections Warrants Further Investigation of Its Mechanisms of Action |
| Bahrampour Juybari K, et al. Virus Res [44] | To analyze the current evidence on the treatment of melatonin in viral infections | Longitudinal analytical observational | L | Melatonin promotes both humoral and cell-mediated immunity. Motivates synthesis of macrophage and granulocyte progenitor cells, NK cells and T helper cells specifically CD4 + cells | It is suggested that the use of melatonin in an outbreak of COVID-19 is beneficial, in the absence of a specific and effective treatment. |
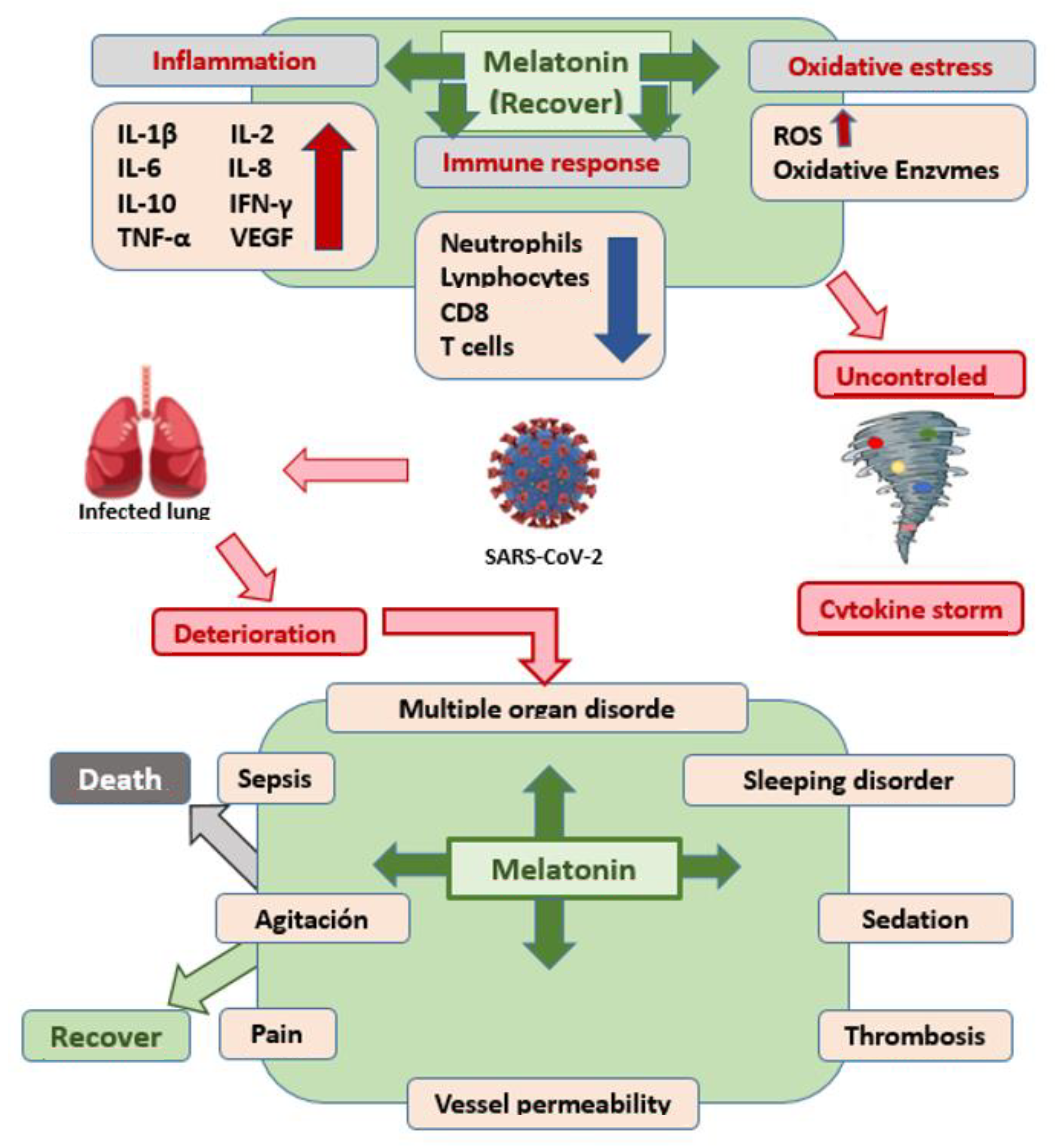
| Sehirli AO, et al. Mol. Biol. Rep. [37] | To evaluate the importance of the CD147 protein and the possible protective effect of melatonin mediated by this protein. | Longitudinal analytical observational | M | CD147 is a glycoprotein responsible for the formation of the cytokine storm in the lungs through mediation of viral invasion. Melatonin reduces heart damage by blocking the activity of CD 147. | Melatonin is a safe drug that can prevent severe symptoms, reduce the severity of symptoms, and reduce the adverse effects of other antiviral drugs in COVID-19 patients. |
| Feitosa EL, et al. Int J Med Sci. [38] | To rationally identify new inhibitors of the main protease (Mpro) of SARS-CoV-2 using silico-tools, which show additional pharmacological properties against COVID-19 | Molecular docking studies of binding sites and interaction energies of 74 Mpro-ligand complexes | A | 59 impact compounds are identified, with melatonin standing out for its immunomodulatory and anti-inflammatory activity. The results do not confirm the antiviral activity of melatonin, but are the basis for clinical trials | The use of melatonin may have a response potential in early stages due to its possible effects on ACE-2 and Mpro, as well as in severe stages due to its action against hyperinflammation |
| Hazra S, et al. Life Sci. [39] | Explore the host protein (s) targeted by potent reused in COVID-19 | Clinical trial with patient blood microarray data | A | Chloroquine and melatonin showed functions associated with neutrophil-mediated immunoinflammation | The present study reveals that between chloroquine and melatonin, melatonin appears to be a more promising repurposed drug against MMP9 for better immunocompromise in COVID-19. |
| Martín Giménez VM, et al. Nanomedicine (Lond). [40] | Carry out a new proposal based on the use of nanoformulated melatonin targeting mitochondria as a possible treatment for COVID-19 | Descriptive basic research | L | Nanoformulation of melatonin offers advantages over conventional pharmaceutical preparations due to the kinetics of drug release, greater protection against early oxidation, and improves cellular absorption and bioavailability. | Based on the available technology, the administration of melatonin in nanoparticles should be analyzed as a new therapeutic alternative for the treatment of COVID-19 and/or other viral infections. |
| Öztürk G, et al. Turk J Med Sci. [51] | To analyze the benefits of melatonin as a treatment for COVID-19 in the elderly | Comparative descriptive study | L | Melatonin plays a fundamental role in the prevention of oxidative stress and mitochondrial dysfunction caused by free radical reactions initiated by mitochondria in the aging process | With age, melatonin levels decrease, so supplementation in the elderly could be beneficial in the treatment of COVID-19. More studies are required along this line. |
| Artigas L, et al. PLoS One. [52] | Combining Pirfenidone and Melatonin Medications to Identify Appropriate COVID-19 Treatment | Clinical trial in symptomatic patients of COVID-19 with different severity | A | Pirfenidone and melatonin are safe drugs that can be combined with the current standard of care treatments for COVID-19 | The combination of pirfenidone and melatonin with standard COVID-19 treatment is considered of interest in patients at risk of developing serious pulmonary complications |
| Acuña-CastroViejo D, et al. J Pineal Res [45] | Determine the doses to be used and the efficacy of melatonin in SARS-CoV-2 infection | Phase II, double-blind, randomized, placebo-contrasted trial | M | Intravenous administration of 60 mg/d of a proprietary formulation of melatonin improved septic patients, reduced their mortality to zero and their hospital stay by 40% | Melatonin may be useful in the treatment of COVID-19 for the following reasons: (1) Its antioxidant action to reduce the effects of free radicals, restore mitochondrial metabolism, and prevent lung damage. (2) Anti-inflammatory function, caused by SARS-CoV-2 infection. (3) Inhibitor of p65, preventing the transcriptional capacity and the inflammatory response. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molina-Carballo, A.; Palacios-López, R.; Jerez-Calero, A.; Augustín-Morales, M.C.; Agil, A.; Muñoz-Hoyos, A.; Muñoz-Gallego, A. Protective Effect of Melatonin Administration against SARS-CoV-2 Infection: A Systematic Review. Curr. Issues Mol. Biol. 2022, 44, 31-45. https://doi.org/10.3390/cimb44010003
Molina-Carballo A, Palacios-López R, Jerez-Calero A, Augustín-Morales MC, Agil A, Muñoz-Hoyos A, Muñoz-Gallego A. Protective Effect of Melatonin Administration against SARS-CoV-2 Infection: A Systematic Review. Current Issues in Molecular Biology. 2022; 44(1):31-45. https://doi.org/10.3390/cimb44010003
Chicago/Turabian StyleMolina-Carballo, Antonio, Rafael Palacios-López, Antonio Jerez-Calero, María Carmen Augustín-Morales, Ahmed Agil, Antonio Muñoz-Hoyos, and Antonio Muñoz-Gallego. 2022. "Protective Effect of Melatonin Administration against SARS-CoV-2 Infection: A Systematic Review" Current Issues in Molecular Biology 44, no. 1: 31-45. https://doi.org/10.3390/cimb44010003
APA StyleMolina-Carballo, A., Palacios-López, R., Jerez-Calero, A., Augustín-Morales, M. C., Agil, A., Muñoz-Hoyos, A., & Muñoz-Gallego, A. (2022). Protective Effect of Melatonin Administration against SARS-CoV-2 Infection: A Systematic Review. Current Issues in Molecular Biology, 44(1), 31-45. https://doi.org/10.3390/cimb44010003








