Alterations in B Cell and Follicular T-Helper Cell Subsets in Patients with Acute COVID-19 and COVID-19 Convalescents
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patient Characteristics
2.2. Sample Collection
2.3. Flow Cytometry B Cell Immunophenotyping
2.4. T Cell Immunophenotype by Flow Cytometry
2.5. Statistical Analysis
3. Results
3.1. Alterations in Peripheral Blood B Cell Subset Composition of COVID-19 Patients
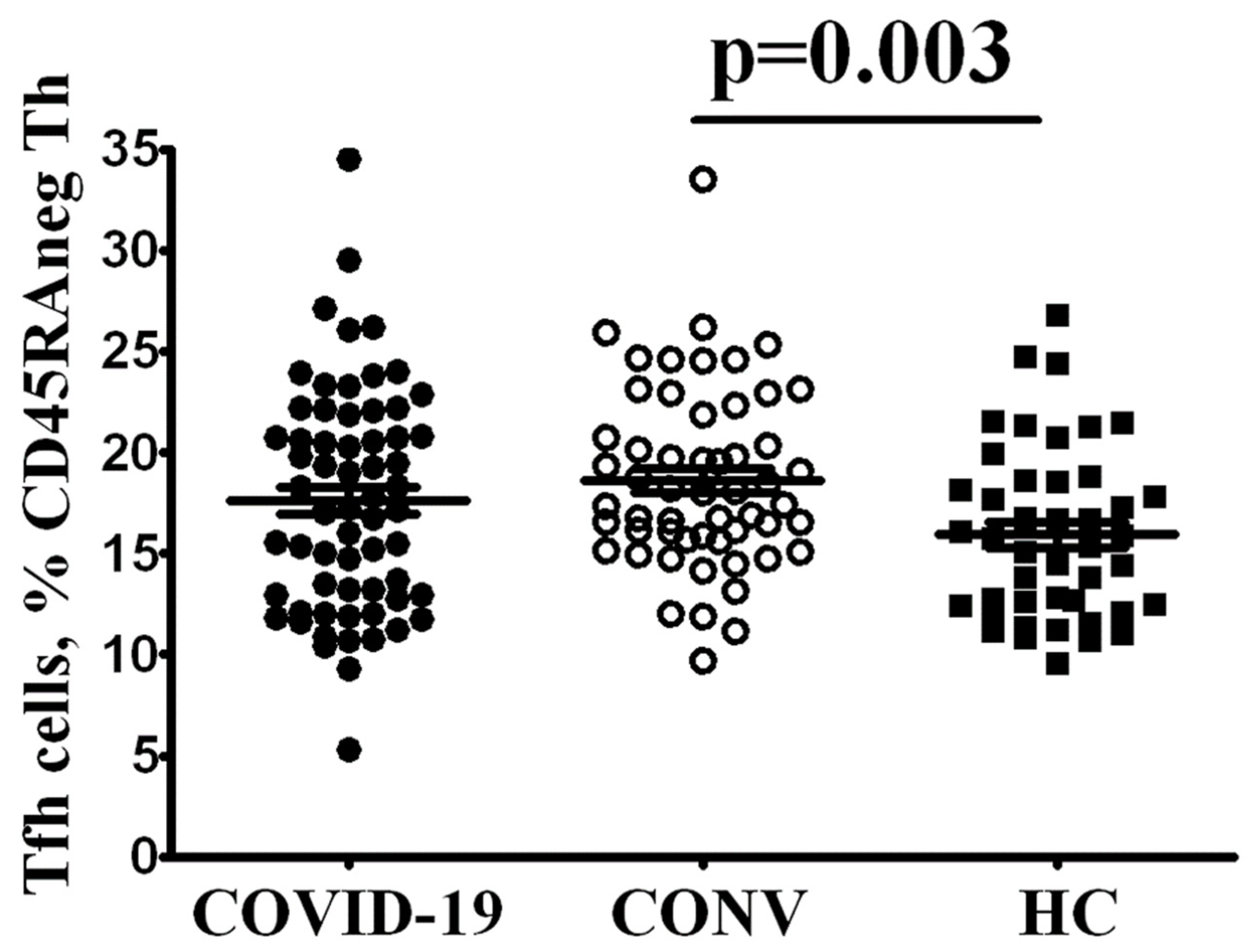
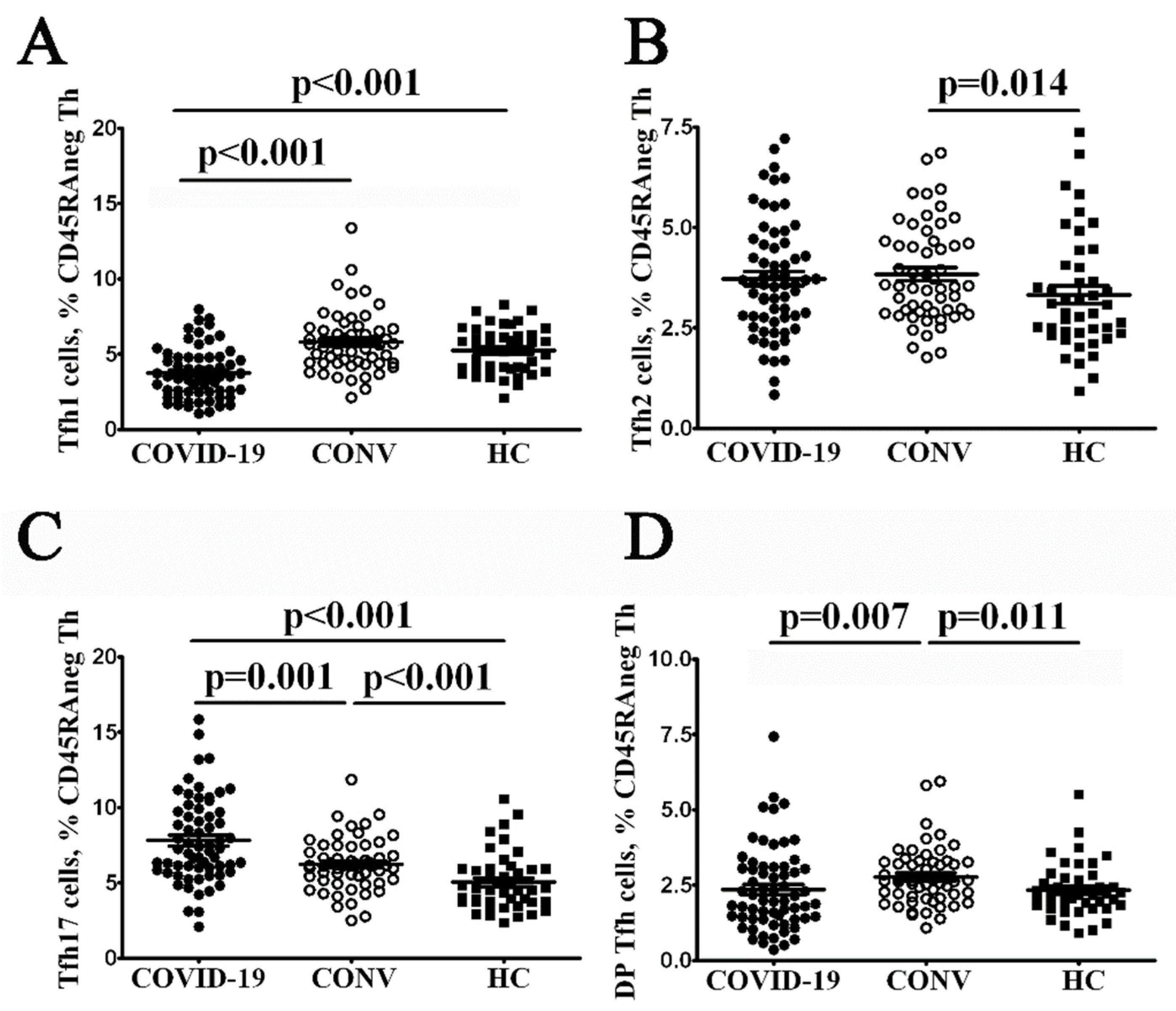
3.2. Tfh Subset Imbalance in COVID-19 Patients
4. Discussion
Author Contributions
Funding
Institution Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Azer, S. COVID-19: Pathophysiology, diagnosis, complications and investigational therapeutics. New Microbes New Infect. 2020, 37, 100738. [Google Scholar] [CrossRef]
- Parasher, A. COVID-19: Current understanding of its pathophysiology, clinical presentation and treatment. Postgrad. Med. J. 2020, 97, 312–320. [Google Scholar]
- Malkova, A.; Kudlay, D.; Kudryavtsev, I.; Starshinova, A.; Yablonskiy, P.; Shoenfeld, Y. Immunogenetic Predictors of Severe COVID-19. Vaccines 2021, 9, 211. [Google Scholar] [CrossRef] [PubMed]
- Giamarellos-Bourboulis, E.J.; Netea, M.G.; Rovina, N.; Akinosoglou, K.; Antoniadou, A.; Antonakos, N.; Damoraki, G.; Gkavogianni, T.; Adami, M.-E.; Katsaounou, P.; et al. Complex Immune Dysregulation in COVID-19 Patients with Severe Respiratory Failure. Cell Host Microbe. 2020, 27, 992–1000. [Google Scholar] [CrossRef]
- Spoerl, S.; Kremer, A.N.; Aigner, M.; Eisenhauer, N.; Koch, P.; Meretuk, L.; Löffler, P.; Tenbusch, M.; Maier, C.; Überla, K.; et al. Upregulation of CCR4 in activated CD8+ T cells indicates enhanced lung homing in patients with severe acute SARS-CoV-2 infection. Eur. J. Immunol. 2021, 51, 1436–1448. [Google Scholar] [CrossRef]
- Kudryavtsev, I.; Kalinina, O.; Bezrukikh, V.; Melnik, O.; Golovkin, A. The Significance of Phenotyping and Quantification of Plasma Extracellular Vesicles Levels Using High-Sensitivity Flow Cytometry during COVID-19 Treatment. Viruses 2021, 13, 767. [Google Scholar] [CrossRef]
- Iwamura, A.P.D.; Da Silva, M.R.T.; Hümmelgen, A.L.; Pereira, P.V.S.; Falcai, A.; Grumach, A.S.; Goudouris, E.; Neto, A.C.; Prando, C. Immunity and inflammatory biomarkers in COVID-19: A systematic review. Rev. Med. Virol. 2020, 31, e2199. [Google Scholar] [CrossRef] [PubMed]
- Arsentieva, N.A.; Liubimova, N.E.; Batsunov, O.K.; Korobova, Z.R.; Stanevich, O.V.; Lebedeva, A.A.; Vorobyov, E.A.; Vorobyova, S.V.; Kulikov, A.N.; Lioznov, D.A.; et al. Plasma cytokines in patients with COVID-19 during acute phase of the disease and following complete recovery. Med. Immunol. (Russia) 2021, 23, 311–326. [Google Scholar] [CrossRef]
- Crotty, S. T Follicular Helper Cell Differentiation, Function, and Roles in Disease. Immunity 2014, 41, 529–542. [Google Scholar] [CrossRef] [Green Version]
- Vinuesa, C.G.; Linterman, M.A.; Yu, D.; MacLennan, I.C. Follicular helper T cells. Annu. Rev. Immunol. 2016, 34, 335–368. [Google Scholar] [CrossRef] [PubMed]
- Bohnhorst, J.Ø.; Bjørgan, M.B.; Thoen, J.E.; Natvig, J.B.; Thompson, K.M. Bm1–Bm5 Classification of Peripheral Blood B Cells Reveals Circulating Germinal Center Founder Cells in Healthy Individuals and Disturbance in the B Cell Subpopulations in Patients with Primary Sjögren’s Syndrome. J. Immunol. 2001, 167, 3610–3618. [Google Scholar] [CrossRef] [PubMed]
- Hanley, P.; Sutter, J.A.; Goodman, N.G.; Du, Y.; Sekiguchi, D.R.; Meng, W.; Rickels, M.R.; Naji, A.; Prak, E.T.L. Circulating B cells in type 1 diabetics exhibit fewer maturation-associated phenotypes. Clin. Immunol. 2017, 183, 336–343. [Google Scholar] [CrossRef]
- Chevalier, N.; Jarrossay, D.; Ho, E.; Avery, D.T.; Ma, C.; Yu, D.; Sallusto, F.; Tangye, P.S.; Mackay, C. CXCR5 Expressing Human Central Memory CD4 T Cells and Their Relevance for Humoral Immune Responses. J. Immunol. 2011, 186, 5556–5568. [Google Scholar] [CrossRef] [PubMed]
- Morita, R.; Schmitt, N.; Bentebibel, S.-E.; Ranganathan, R.; Bourdery, L.; Zurawski, G.; Foucat, E.; Dullaers, M.; Oh, S.; Sabzghabaei, N.; et al. Human Blood CXCR5+CD4+ T Cells Are Counterparts of T Follicular Cells and Contain Specific Subsets that Differentially Support Antibody Secretion. Immunity 2011, 34, 108–121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sosa-Hernández, V.A.; Torres-Ruíz, J.; Cervantes-Díaz, R.; Romero-Ramírez, S.; Páez-Franco, J.C.; Meza-Sánchez, D.E.; Juárez-Vega, G.; Pérez-Fragoso, A.; Ortiz-Navarrete, V.; Ponce-De-León, A.; et al. B Cell Subsets as Severity-Associated Signatures in COVID-19 Patients. Front. Immunol. 2020, 11, 611004. [Google Scholar] [CrossRef] [PubMed]
- Lucas, C.; Wong, P.; Klein, J.; Castro, T.B.R.; Silva, J.; Sundaram, M.; Ellingson, M.K.; Mao, T.; Oh, J.E.; Israelow, B.; et al. Longitudinal analyses reveal immunological misfiring in severe COVID-19. Nature 2020, 584, 463–469. [Google Scholar] [CrossRef]
- He, R.; Lu, Z.; Zhang, L.; Fan, T.; Xiong, R.; Shen, X.; Feng, H.; Meng, H.; Lin, W.; Jiang, W.; et al. The clinical course and its correlated immune status in COVID-19 pneumonia. J. Clin. Virol. 2020, 127, 104361. [Google Scholar] [CrossRef]
- Mann, E.R.; Menon, M.; Knight, S.B.; Konkel, J.E.; Jagger, C.; Shaw, T.N.; Krishnan, S.; Rattray, M.; Ustianowski, A.; Bakerly, N.D.; et al. Longitudinal immune profiling reveals key myeloid signatures associated with COVID-19. Sci. Immunol. 2020, 5, 6197. [Google Scholar] [CrossRef]
- Golovkin, A.; Kalinina, O.; Bezrukikh, V.; Aquino, A.; Zaikova, E.; Karonova, T.; Melnik, O.; Vasilieva, E.; Kudryavtsev, I. Imbalanced Immune Response of T-Cell and B-Cell Subsets in Patients with Moderate and Severe COVID-19. Viruses 2021, 13, 1966. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, N.; Kuo, H.-H.; Boucau, J.; Farmer, J.R.; Allard-Chamard, H.; Mahajan, V.S.; Piechocka-Trocha, A.; Lefteri, K.; Osborn, M.; Bals, J.; et al. Loss of Bcl-6-Expressing T Follicular Helper Cells and Germinal Centers in COVID-19. Cell 2020, 183, 143–157.e13. [Google Scholar] [CrossRef]
- De Biasi, S.; Tartaro, D.L.; Meschiari, M.; Gibellini, L.; Bellinazzi, C.; Borella, R.; Fidanza, L.; Mattioli, M.; Paolini, A.; Gozzi, L.; et al. Expansion of plasmablasts and loss of memory B cells in peripheral blood from COVID-19 patients with pneumonia. Eur. J. Immunol. 2020, 50, 1283–1294. [Google Scholar] [CrossRef]
- Mathew, D.; Giles, J.R.; Baxter, A.E.; Oldridge, D.A.; Greenplate, A.R.; Wu, J.E.; Alanio, C.; Kuri-Cervantes, L.; Pampena, M.B.; D’Andrea, K.; et al. Deep immune profiling of COVID-19 patients reveals distinct immunotypes with therapeutic implications. Science 2020, 369, eabc8511. [Google Scholar] [CrossRef]
- Moratto, D.; Chiarini, M.; Giustini, V.; Serana, F.; Magro, P.; Roccaro, A.M.; Imberti, L.; Castelli, F.; Notarangelo, L.D.; Quiros-Roldan, E. Flow Cytometry Identifies Risk Factors and Dynamic Changes in Patients with COVID-19. J. Clin. Immunol. 2020, 40, 970–973. [Google Scholar] [CrossRef]
- Byazrova, M.; Yusubalieva, G.; Spiridonova, A.; Efimov, G.; Mazurov, D.; Baranov, K.; Baklaushev, V.; Filatov, A. Pattern of circulating SARS-CoV-2-specific antibody-secreting and memory B-cell generation in patients with acute COVID-19. Clin. Transl. Immunol. 2021, 10, e1245. [Google Scholar] [CrossRef]
- Gutiérrez-Bautista, J.F.; Rodriguez-Nicolas, A.; Rosales-Castillo, A.; Jiménez, P.; Garrido, F.; Anderson, P.; Ruiz-Cabello, F.; López-Ruz, M.Á. Negative Clinical Evolution in COVID-19 Patients Is Frequently Accompanied with an Increased Proportion of Undifferentiated Th Cells and a Strong Underrepresentation of the Th1 Subset. Front. Immunol. 2020, 11, 596553. [Google Scholar] [CrossRef]
- Kuri-Cervantes, L.; Pampena, M.B.; Meng, W.; Rosenfeld, A.M.; Ittner, C.A.G.; Weisman, A.R.; Agyekum, R.S.; Mathew, D.; Baxter, A.E.; Vella, L.A.; et al. Comprehensive mapping of immune perturbations associated with severe COVID-19. Sci. Immunol. 2020, 5, 7114. [Google Scholar] [CrossRef]
- Schultheiß, C.; Paschold, L.; Simnica, D.; Mohme, M.; Willscher, E.; von Wenserski, L.; Scholz, R.; Wieters, I.; Dahlke, C.; Tolosa, E.; et al. Next-Generation Sequencing of T and B Cell Receptor Repertoires from COVID-19 Patients Showed Signatures Associated with Severity of Disease. Immunity 2020, 53, 442–455.e4. [Google Scholar] [CrossRef]
- Gong, F.; Dai, Y.; Zheng, T.; Cheng, L.; Zhao, D.; Wang, H.; Liu, M.; Pei, H.; Jin, T.; Yu, D.; et al. Peripheral CD4+ T cell subsets and antibody response in COVID-19 convalescent individuals. J. Clin. Investig. 2020, 130, 6588–6599. [Google Scholar] [CrossRef] [PubMed]
- Juno, J.A.; Tan, H.X.; Lee, W.S.; Reynaldi, A.; Kelly, H.G.; Wragg, K.; Esterbauer, R.; Kent, H.E.; Batten, C.J.; Mordant, F.L.; et al. Humoral and circulating follicular helper T cell responses in recovered patients with COVID-19. Nat. Med. 2020, 26, 1428–1434. [Google Scholar] [CrossRef] [PubMed]
- Nalbandian, A.; Sehgal, K.; Gupta, A.; Madhavan, M.V.; McGroder, C.; Stevens, J.S.; Cook, J.R.; Nordvig, A.S.; Shalev, D.; Sehrawat, T.S.; et al. Post-Acute COVID-19 syndrome. Nat. Med. 2021, 27, 601–615. [Google Scholar] [CrossRef] [PubMed]
- Malkova, A.; Kudryavtsev, I.; Starshinova, A.; Kudlay, D.; Zinchenko, Y.; Glushkova, A.; Yablonskiy, P.; Shoenfeld, Y. Post COVID-19 Syndrome in Patients with Asymptomatic/Mild Form. Pathogens 2021, 10, 1408. [Google Scholar] [CrossRef]
- Kamal, M.; Omirah, M.A.; Hussein, A.; Saeed, H. Assessment and characterisation of post-COVID-19 manifestations. Int. J. Clin. Pr. 2021, 75, 13746. [Google Scholar] [CrossRef] [PubMed]
- Fink, K. Origin and Function of Circulating Plasmablasts during Acute Viral Infections. Front. Immunol. 2012, 3, 78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McElroy, A.; Akondy, R.; Davis, C.W.; Ellebedy, A.H.; Mehta, A.; Kraft, C.S.; Lyon, G.M.; Ribner, B.S.; Varkey, J.; Sidney, J.; et al. Human Ebola virus infection results in substantial immune activation. Proc. Natl. Acad. Sci. USA 2015, 112, 4719–4724. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pattanapanyasat, K.; Khowawisetsut, L.; Chuansumrit, A.; Chokephaibulkit, K.; Tangnararatchakit, K.; Apiwattanakul, N.; Techasaensiri, C.; Thitilertdecha, P.; Sae-Ung, T.; Onlamoon, N. B cell subset alteration and the expression of tissue homing molecules in dengue infected patients. J. Biomed. Sci. 2018, 25, 64. [Google Scholar] [CrossRef]
- Wijesinghe, A.; Gamage, J.; Goonewardena, H.; Gomes, L.; Jayathilaka, D.; Wijeratne, D.T.; de Alwis, R.; Jeewandara, C.; Wijewickrama, A.; Ogg, G.S.; et al. Phenotype and functionality of follicular helper T cells in patients with acute dengue infection. J. Biomed. Sci. 2020, 27, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abreu, R.B.; Kirchenbaum, G.A.; Sautto, G.A.; Clutter, E.F.; Ross, T.M. Impaired memory B-cell recall responses in the elderly following recurrent influenza vaccination. PLoS ONE 2021, 16, e0254421. [Google Scholar] [CrossRef]
- Tjiam, M.C.; Fernandez, S.; French, M.A. Characterising the Phenotypic Diversity of Antigen-Specific Memory B Cells before and after Vaccination. Front. Immunol. 2021, 12, 738123. [Google Scholar] [CrossRef] [PubMed]
- Niessl, J.; Sekine, T.; Buggert, M. T cell immunity to SARS-CoV-2. Semin. Immunol. 2021, 55, 101505. [Google Scholar] [CrossRef]
| B Cell Subset | Phenotype | COVID-19 | CONV | HC | Significant Differences |
|---|---|---|---|---|---|
| Bm1 | IgD+CD38− | 12.28 ± 1.23 | 15.83 ± 0.62 | 12.39 ± 0.73 | p1 < 0.001 p2 = 0.129 p3 = 0.004 |
| Bm2 | IgD+CD38+ | 57.79 ± 1.91 | 52.84 ± 1.06 | 56.83 ± 1.32 | p1 = 0.001 p2 = 0.165 p3 = 0.040 |
| Bm2′ | IgD+CD38++ | 9.98 ± 0.79 | 11.57 ± 0.77 | 8.97 ± 0.57 | p1 = 0.109 p2 = 0.919 p3 = 0.044 |
| Bm3+Bm4 | IgD−CD38+++ | 6.00 ± 0.68 | 2.16 ± 0.20 | 1.28 ± 0.16 | p1 < 0.001 p2 < 0.001 p3 < 0.001 |
| eBm5 | IgD−CD38+ | 7.47 ± 0.72 | 9.26 ± 0.44 | 10.93 ± 0.75 | p1 < 0.001 p2 < 0.001 p3 = 0.125 |
| Bm5 | IgD−CD38− | 6.49 ± 0.63 | 8.34 ± 0.48 | 9.60 ± 0.79 | p1 < 0.001 p2 < 0.001 p3 = 0.267 |
| B Cell Subset | Phenotype | COVID-19 | CONV | HC | Significant Differences |
|---|---|---|---|---|---|
| Naive mature | CD27−CD38+ | 12.28 ± 1.23 | 15.83 ± 0.62 | 12.39 ± 0.73 | p1 < 0.001 p2 = 0.129 p3 = 0.004 |
| Mature active | CD27+CD38+ | 57.79 ± 1.91 | 52.84 ± 1.06 | 56.83 ± 1.32 | p1 = 0.001 p2 = 0.165 p3 = 0.040 |
| DN cells | CD27−CD38− | 9.98 ± 0.79 | 11.57 ± 0.77 | 8.97 ± 0.57 | p1 = 0.109 p2 = 0.919 p3 = 0.044 |
| Memory | CD27+CD38− | 6.00 ± 0.68 | 2.16 ± 0.20 | 1.28 ± 0.16 | p1 < 0.001 p2 < 0.001 p3 < 0.001 |
| Plasmablasts | CD27++CD38+ | 7.47 ± 0.72 | 9.26 ± 0.44 | 10.93 ± 0.75 | p1 < 0.001 p2 < 0.001 p3 = 0.125 |
| Transitional cells | CD27−CD38++ | 6.49 ± 0.63 | 8.34 ± 0.48 | 9.60 ± 0.79 | p1 < 0.001 p2 < 0.001 p3 = 0.267 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kudryavtsev, I.V.; Arsentieva, N.A.; Batsunov, O.K.; Korobova, Z.R.; Khamitova, I.V.; Isakov, D.V.; Kuznetsova, R.N.; Rubinstein, A.A.; Stanevich, O.V.; Lebedeva, A.A.; et al. Alterations in B Cell and Follicular T-Helper Cell Subsets in Patients with Acute COVID-19 and COVID-19 Convalescents. Curr. Issues Mol. Biol. 2022, 44, 194-205. https://doi.org/10.3390/cimb44010014
Kudryavtsev IV, Arsentieva NA, Batsunov OK, Korobova ZR, Khamitova IV, Isakov DV, Kuznetsova RN, Rubinstein AA, Stanevich OV, Lebedeva AA, et al. Alterations in B Cell and Follicular T-Helper Cell Subsets in Patients with Acute COVID-19 and COVID-19 Convalescents. Current Issues in Molecular Biology. 2022; 44(1):194-205. https://doi.org/10.3390/cimb44010014
Chicago/Turabian StyleKudryavtsev, Igor V., Natalia A. Arsentieva, Oleg K. Batsunov, Zoia R. Korobova, Irina V. Khamitova, Dmitrii V. Isakov, Raisa N. Kuznetsova, Artem A. Rubinstein, Oksana V. Stanevich, Aleksandra A. Lebedeva, and et al. 2022. "Alterations in B Cell and Follicular T-Helper Cell Subsets in Patients with Acute COVID-19 and COVID-19 Convalescents" Current Issues in Molecular Biology 44, no. 1: 194-205. https://doi.org/10.3390/cimb44010014
APA StyleKudryavtsev, I. V., Arsentieva, N. A., Batsunov, O. K., Korobova, Z. R., Khamitova, I. V., Isakov, D. V., Kuznetsova, R. N., Rubinstein, A. A., Stanevich, O. V., Lebedeva, A. A., Vorobyov, E. A., Vorobyova, S. V., Kulikov, A. N., Sharapova, M. A., Pevtcov, D. E., & Totolian, A. A. (2022). Alterations in B Cell and Follicular T-Helper Cell Subsets in Patients with Acute COVID-19 and COVID-19 Convalescents. Current Issues in Molecular Biology, 44(1), 194-205. https://doi.org/10.3390/cimb44010014








