Assessment of Bioavailability after In Vitro Digestion and First Pass Metabolism of Bioactive Peptides from Collagen Hydrolysates
Abstract
:1. Introduction
2. Materials and Methods
2.1. Peptide Standards
2.2. Cells
2.3. Treatments
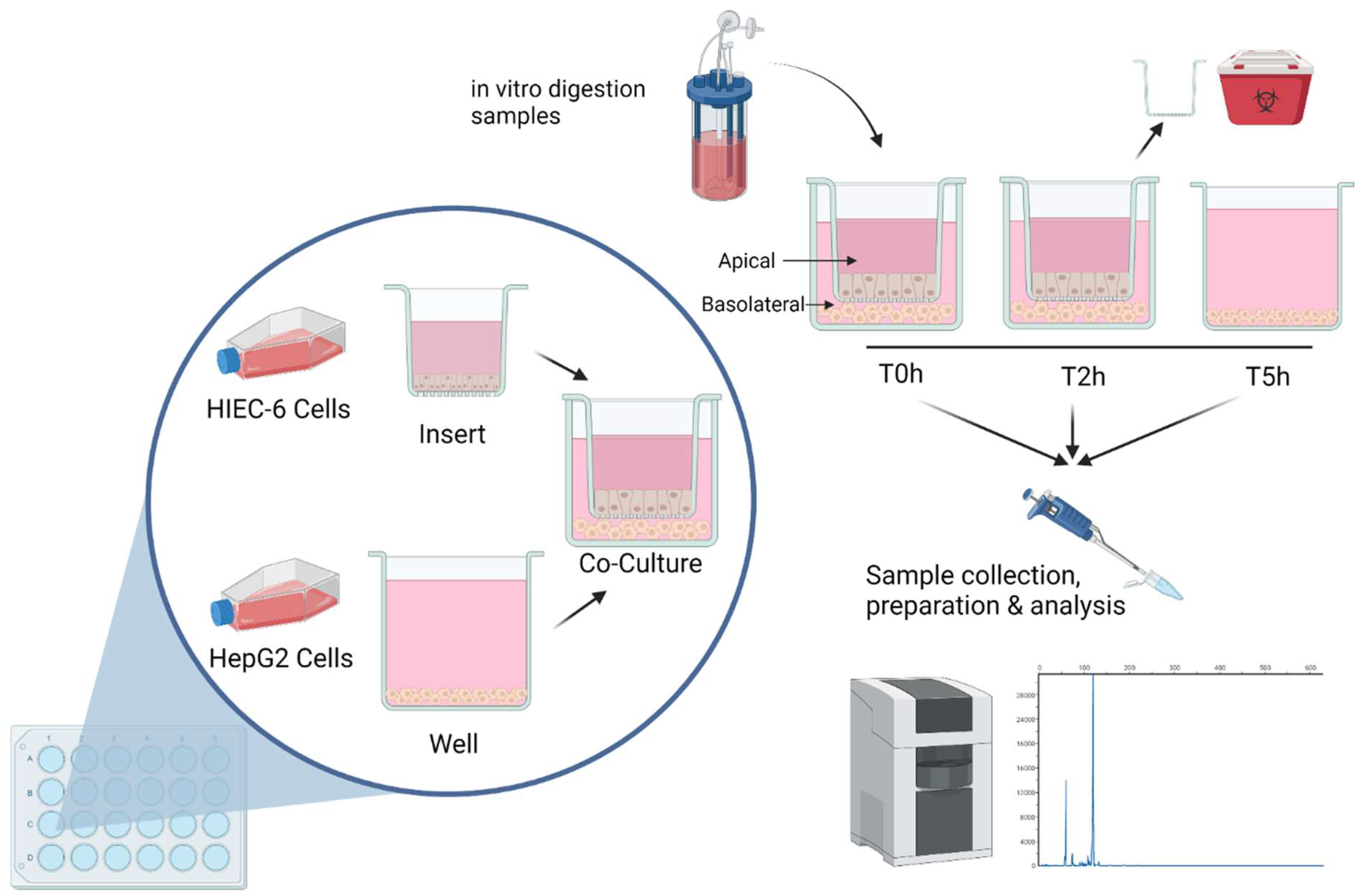
2.4. Simulated Digestion
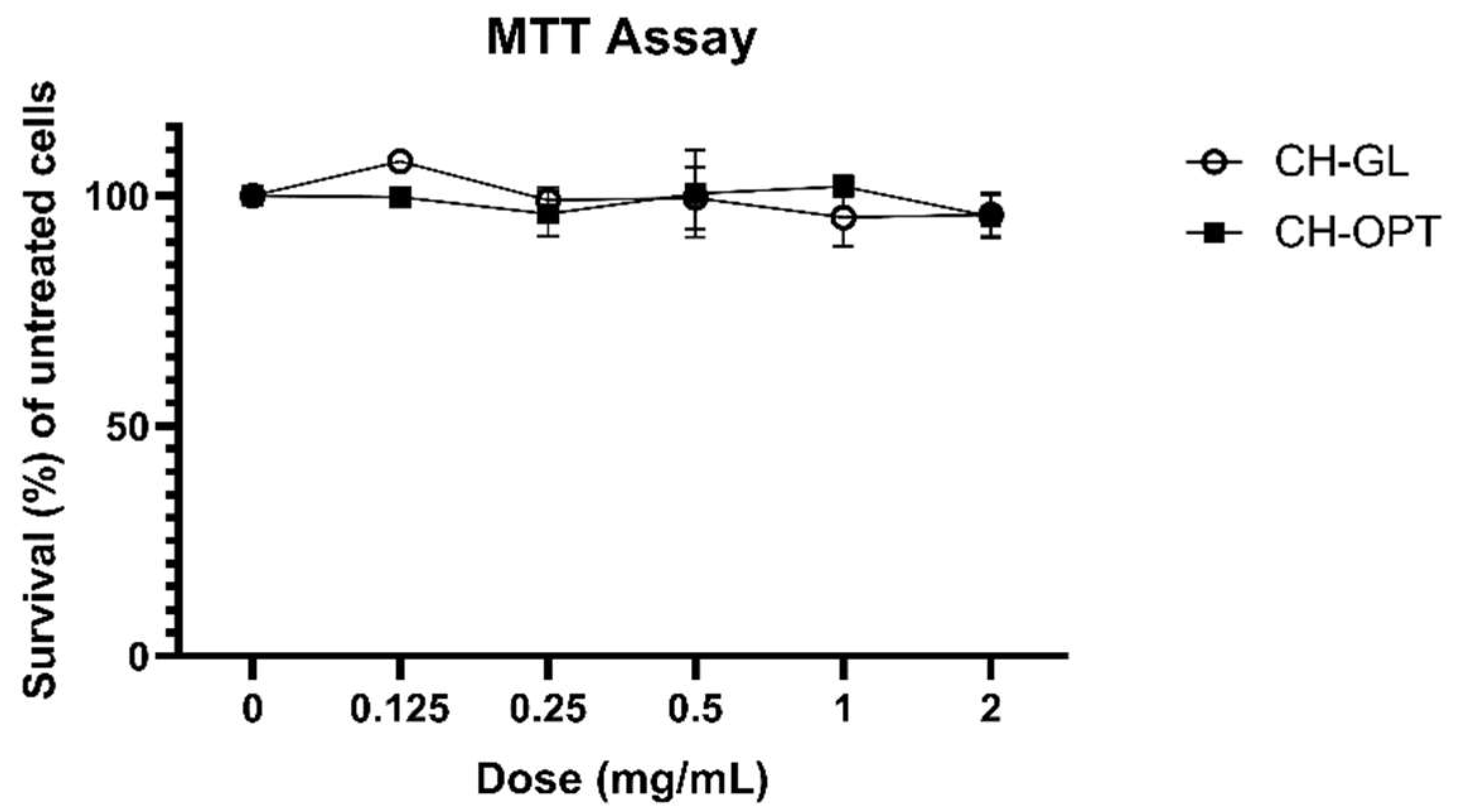
2.5. 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl Tetrazolium Bromide (MTT) Assay
2.6. Co-Culture
2.7. Targeted Peptide Quantification Using Capillary Electrophoresis (CE)
2.8. Statistical Analysis
3. Results
3.1. MTT Assay
3.2. Peptide Transport
3.3. Hepatic First Pass Effects
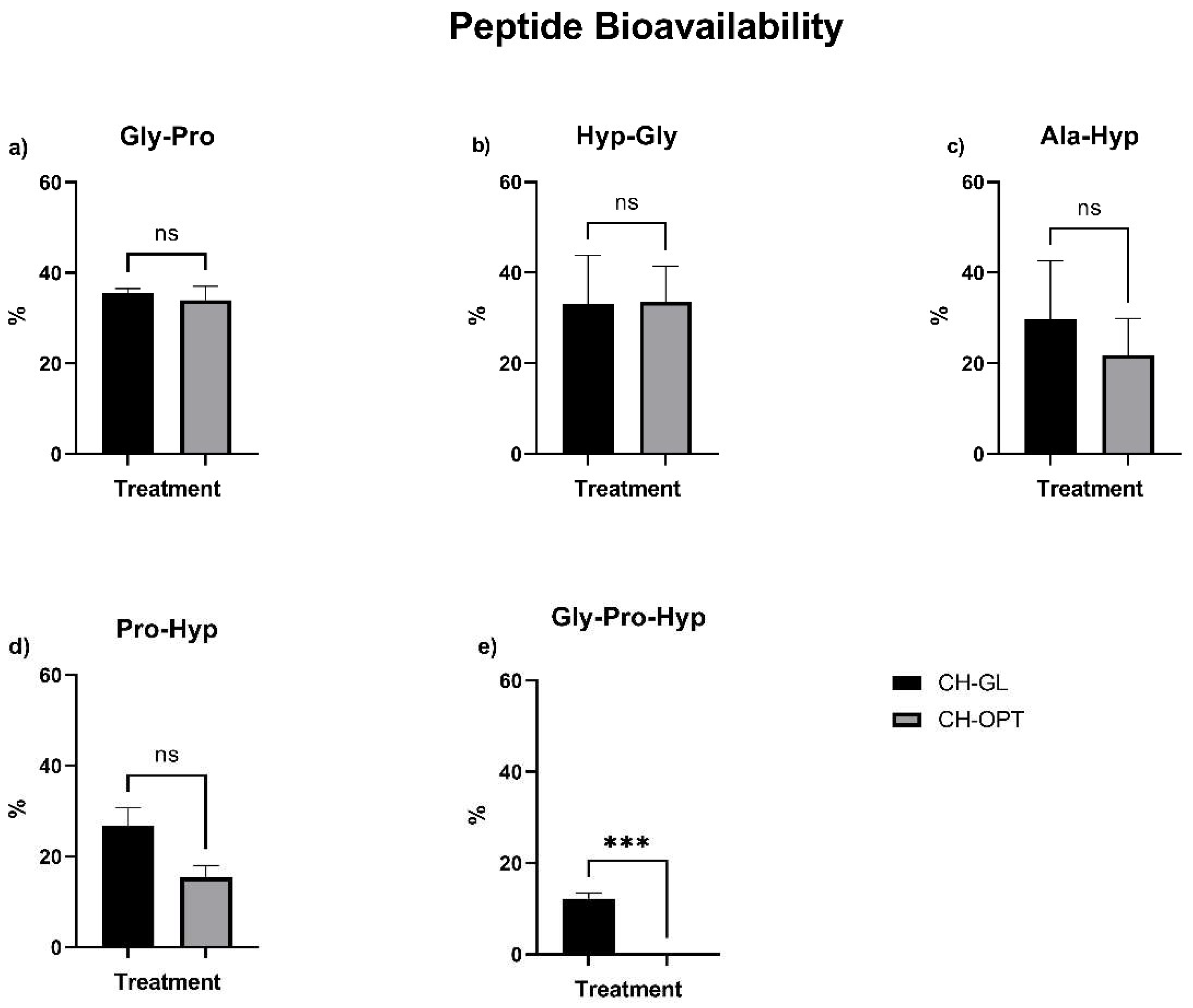
3.4. Peptide Bioavailability
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zdzieblik, D.; Oesser, S.; Gollhofer, A.; König, D. Improvement of activity-related knee joint discomfort following supplementation of specific collagen peptides. Appl. Physiol. Nutr. Metab. 2017, 42, 588–595. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amigo, L.; Hernández-Ledesma, B. Current evidence on the bioavailability of food bioactive peptides. Molecules 2020, 25, 4479. [Google Scholar] [CrossRef] [PubMed]
- Pal, G.K.; Suresh, P.V. Sustainable valorisation of seafood by-products: Recovery of collagen and development of collagen-based novel functional food ingredients. Innov. Food Sci. Emerg. Technol. 2016, 37, 201–215. [Google Scholar] [CrossRef]
- Skov, K.; Oxfeldt, M.; Thøgersen, R.; Hansen, M.; Bertram, H.C. Enzymatic hydrolysis of a collagen hydrolysate enhances postprandial absorption rate-a randomized controlled trial. Nutrients 2019, 11, 1064. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Larder, C.E.; Iskandar, M.M.; Kubow, S. Gastrointestinal digestion model assessment of peptide diversity and microbial fermentation products of collagen hydrolysates. Nutrients 2021, 13, 2720. [Google Scholar] [CrossRef]
- Iwai, K.; Hasegawa, T.; Taguchi, Y.; Morimatsu, F.; Sato, K.; Nakamura, Y.; Higashi, A.; Kido, Y.; Nakabo, Y.; Ohtsuki, K. Identification of food-derived collagen peptides in human blood after oral ingestion of gelatin hydrolysates. J. Agric. Food Chem. 2005, 53, 6531–6536. [Google Scholar] [CrossRef]
- Song, H.; Tian, Q.; Li, B. Novel Hyp-Gly-containing antiplatelet peptides from collagen hydrolysate after simulated gastrointestinal digestion and intestinal absorption. Food Funct. 2020, 11, 5553–5564. [Google Scholar] [CrossRef]
- Sadeghi Ekbatan, S.; Iskandar, M.M.; Sleno, L.; Sabally, K.; Khairallah, J.; Prakash, S.; Kubow, S. Absorption and metabolism of phenolics from digests of polyphenol-rich potato extracts using the Caco-2/HepG2 co-culture system. Foods 2018, 7, 8. [Google Scholar] [CrossRef] [Green Version]
- Brayden, D.J. Evolving peptides for oral intake. Nat. Biomed. Eng. 2020, 4, 487–488. [Google Scholar] [CrossRef]
- Shigemura, Y.; Suzuki, A.; Kurokawa, M.; Sato, Y.; Sato, K. Changes in composition and content of food-derived peptide in human blood after daily ingestion of collagen hydrolysate for 4 weeks. J. Sci. Food Agric. 2018, 98, 1944–1950. [Google Scholar] [CrossRef]
- Yamamoto, S.; Deguchi, K.; Onuma, M.; Numata, N.; Sakai, Y. Absorption and urinary excretion of peptides after collagen tripeptide ingestion in humans. Biol. Pharm. Bull. 2016, 39, 428–434. [Google Scholar] [CrossRef] [Green Version]
- Shigemura, Y.; Nakaba, M.; Shiratsuchi, E.; Suyama, M.; Yamada, M.; Kiyono, T.; Fukamizu, K.; Park, E.Y.; Nakamura, Y.; Sato, K. Identification of food-derived elastin peptide, prolyl-glycine (Pro-Gly), in human blood after ingestion of elastin hydrolysate. J. Agric. Food Chem. 2012, 60, 5128–5133. [Google Scholar] [CrossRef] [PubMed]
- Bruyère, O.; Zegels, B.; Leonori, L.; Rabenda, V.; Janssen, A.; Bourges, C.; Reginster, J.Y. Effect of collagen hydrolysate in articular pain: A 6-month randomized, double-blind, placebo controlled study. Complement. Ther. Med. 2012, 20, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, Q.; Liang, Q.; He, Y.; Wang, Z.; He, S.; Xu, J.; Ma, H. Determination of bioavailability and identification of collagen peptide in blood after oral ingestion of gelatin. J. Sci. Food Agric. 2015, 95, 2712–2717. [Google Scholar] [CrossRef] [PubMed]
- Sontakke, S.B.; Jung, J.H.; Piao, Z.; Chung, H.J. Orally available collagen tripeptide: Enzymatic stability, intestinal permeability, and absorption of Gly-Pro-Hyp and Pro-Hyp. J. Agric. Food Chem. 2016, 64, 7127–7133. [Google Scholar] [CrossRef] [PubMed]
- Taga, Y.; Kusubata, M.; Ogawa-Goto, K.; Hattori, S. Identification of collagen-derived hydroxyproline (Hyp)-containing cyclic dipeptides with high oral bioavailability: Efficient formation of cyclo(X-Hyp) from X-Hyp-Gly-type tripeptides by heating. J. Agric. Food Chem. 2017, 65, 9514–9521. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, Q.; Qian, J.; Liang, Q.; Wang, Z.; Xu, J.; He, S.; Ma, H. Bioavailability and bioavailable forms of collagen after oral administration to rats. J. Agric. Food Chem. 2015, 63, 3752–3756. [Google Scholar] [CrossRef] [PubMed]
- Punt, A.; Peijnenburg, A.; Hoogenboom, R.; Bouwmeester, H. Non-animal approaches for toxicokinetics in risk evaluations of food chemicals. ALTEX 2017, 34, 501–514. [Google Scholar] [CrossRef] [Green Version]
- Feng, M.; Betti, M. Transepithelial transport efficiency of bovine collagen hydrolysates in a human Caco-2 cell line model. Food Chem. 2017, 224, 242–250. [Google Scholar] [CrossRef]
- Larregieu, C.A.; Benet, L.Z. Drug discovery and regulatory considerations for improving in silico and in vitro predictions that use Caco-2 as a surrogate for human intestinal permeability measurements. Am. Assoc. Pharm. Sci. J. 2013, 15, 483–497. [Google Scholar] [CrossRef] [Green Version]
- Takenaka, T.; Harada, N.; Kuze, J.; Chiba, M.; Iwao, T.; Matsunaga, T. Human small intestinal epithelial cells differentiated from adult intestinal stem cells as a novel system for predicting oral drug absorption in humans. Drug Metab. Dispos. 2014, 42, 1947–1954. [Google Scholar] [CrossRef] [Green Version]
- Takenaka, T.; Harada, N.; Kuze, J.; Chiba, M.; Iwao, T.; Matsunaga, T. Application of a human intestinal epithelial cell monolayer to the prediction of oral drug absorption in humans as a superior alternative to the Caco-2 cell monolayer. J. Pharm. Sci. 2016, 105, 915–924. [Google Scholar] [CrossRef] [Green Version]
- Pászti-Gere, E.; Pomothy, J.; Jerzsele, Á.; Pilgram, O.; Steinmetzer, T. Exposure of human intestinal epithelial cells and primary human hepatocytes to trypsin-like serine protease inhibitors with potential antiviral effect. J. Enzym. Inhib. Med. Chem. 2021, 36, 659–668. [Google Scholar] [CrossRef]
- Zhao, X.; Xu, X.-X.; Liu, Y.; Xi, E.-Z.; An, J.-J.; Tabys, D.; Liu, N. The in vitro protective role of bovine lactoferrin on intestinal epithelial barrier. Molecules 2019, 24, 148. [Google Scholar] [CrossRef] [PubMed]
- Framroze, B.; Havaldar, F.; Misal, S. An in vitro study on the regulation of oxidative protective genes in human gingival and intestinal epithelial cells after treatment with salmon protein hydrolysate peptide. Funct. Foods Health Dis. 2018, 8, 398–411. [Google Scholar] [CrossRef]
- Larder, C.E.; Iskandar, M.M.; Sabally, K.; Kubow, S. Capillary electrophoresis: A fast, cost effective and efficient method replacement for LC-MS when identifying and quantifying small peptides from simulated gastrointestinal digestion of collagen hydrolysate. In Proceedings of the Presentation at the BenefiQ Conference, Quebec City Convention Centre, Québec, QC, Canada, 2–4 October 2018. [Google Scholar] [CrossRef]
- Larder, C.E.; Iskandar, M.M.; Sabally, K.; Kubow, S. Complementary and efficient methods for di- and tri- peptide analysis and amino acid quantification from simulated gastrointestinal digestion of collagen hydrolysate. 2021, Manuscript submitted for publication.
- Louisa, M.; Suyatna, F.D.; Wanandi, S.I.; Asih, P.B.S.; Syafruddin, D. Differential expression of several drug transporter genes in HepG2 and Huh-7 cell lines. Adv. Biomed. Res. 2016, 5, 104. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Xu, J.; Wang, T.; Wan, X.; Zhang, F.; Wang, L.; Zhu, X.; Gao, P.; Shu, G.; Jiang, Q.; et al. The dipeptide Pro-Gly promotes IGF-1 expression and secretion in HepG2 and female mice via PepT1-JAK2/STAT5 pathway. Front. Endocrinol. 2018, 9, 424. [Google Scholar] [CrossRef] [Green Version]
- Cheng, K.C.; Li, C.; Hsieh, Y.; Montgomery, D.; Liu, T.; White, R. Development of a high-throughput in vitro assay using a novel Caco-2/rat hepatocyte system for the prediction of oral plasma area under the concentration versus time curve (AUC) in rats. J. Pharmacol. Toxicol. Methods 2006, 53, 215–218. [Google Scholar] [CrossRef]
- Lau, Y.; Chen, Y.; Liu, T.; Li, C.; Cui, X.; White, R.; Cheng, K. Evaluation of a novel in vitro Caco-2 hepatocyte hybrid system for predicting in vivo oral bioavailability. Drug Metab. Dispos. 2004, 32, 937–942. [Google Scholar]
- Alemán, A.; Gómez-Guillén, M.C.; Montero, P. Identification of ace-inhibitory peptides from squid skin collagen after in vitro gastrointestinal digestion. Food Res. Int. 2013, 54, 790–795. [Google Scholar] [CrossRef]
- Miranda, L.; Deusser, H.; Evers, D. The impact of in vitro digestion on bioaccessibility of polyphenols from potatoes and sweet potatoes and their influence on iron absorption by human intestinal cells. Food Funct. 2013, 4, 1595–1601. [Google Scholar] [CrossRef] [PubMed]
- Iskandar, M.; Lands, L.; Sabally, K.; Azadi, B.; Meehan, B.; Mawji, N.; Skinner, C.; Kubow, S. High hydrostatic pressure pretreatment of whey protein isolates improves their digestibility and antioxidant capacity. Foods 2015, 4, 184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Mikulíková, K.; Eckhardt, A.; Pataridis, S.; Mikšík, I.; Paris, F. Study of posttranslational non-enzymatic modifications of collagen using capillary electrophoresis/mass spectrometry and high performance liquid chromatography/mass spectrometry. J. Chromatogr. 2007, 1155, 125–133. [Google Scholar] [CrossRef]
- Hsiao, Y.M.; Ko, J.L.; Lo, C.C. Determination of tetracycline and streptomycin in mixed fungicide products by capillary zone electrophoresis. J. Agric. Food Chem. 2001, 49, 1669–1674. [Google Scholar] [CrossRef]
- Yee, S. In vitro permeability across Caco-2 cells (colonic) can predict in vivo (small intestinal) absorption in man--fact or myth. Pharm. Res. 1997, 14, 763–766. [Google Scholar] [CrossRef]
- Hatanaka, T.; Kawakami, K.; Uraji, M. Inhibitory effect of collagen-derived tripeptides on dipeptidylpeptidase-iv activity. J. Enzym. Inhib. Med. Chem. 2014, 29, 823–828. [Google Scholar] [CrossRef] [Green Version]
- Feng, M.; Betti, M. Both PepT1 and GLUT intestinal transporters are utilized by a novel glycopeptide Pro-Hyp-CONH-GlcN. J. Agric. Food Chem. 2017, 65, 3295–3304. [Google Scholar] [CrossRef]
- Nakatani, S.; Mano, H.; Sampei, C.; Shimizu, J.; Wada, M. Chondroprotective effect of the bioactive peptide prolyl-hydroxyproline in mouse articular cartilage in vitro and in vivo. Osteoarthr. Cartil. 2009, 17, 1620–1627. [Google Scholar] [CrossRef] [Green Version]
- Ting, Y.; Zhao, Q.; Xia, C.; Huang, Q. Using in vitro and in vivo models to evaluate the oral bioavailability of nutraceuticals. J. Agric. Food Chem. 2015, 63, 1332–1338. [Google Scholar] [CrossRef]
- Li, C.; Liu, T.; Cui, X.; Annette, U.; Cheng, K.C. Development of in vitro pharmacokinetic screens using Caco-2, human hepatocyte, and Caco-2/human hepatocyte hybrid systems for the prediction of oral bioavailability in humans. J. Biomol. Screen 2007, 12, 1084–1091. [Google Scholar] [PubMed]
- Amidon, G.L.; Lee, H.J. Absorption of peptide and peptidomimetic drugs. Annu. Rev. Pharmacol. Toxicol. 1994, 34, 321–341. [Google Scholar] [CrossRef] [PubMed]
- König, D.; Oesser, S.; Scharla, S.; Zdzieblik, D.; Gollhofer, A. Specific collagen peptides improve bone mineral density and bone markers in postmenopausal women-a randomized controlled study. Nutrients 2018, 10, 97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Peptide | Gly-Pro | Hyp-Gly | Ala-Hyp | Pro-Hyp | Gly-Pro-Hyp | |
|---|---|---|---|---|---|---|
| Treatment | ||||||
| CH-GL | 33.11 ± 3.08 | 62.41 ± 11.11 | 9.27 ± 2.49 | 19.18 ± 4.81 | 59.44 ± 11.32 | |
| CH-OPT | 40.35 ± 2.85 | 82.53 ± 36.53 | 26.4 ± 5.78 | 24.15 ± 1.42 | nd | |
| Peptide | Gly-Pro | Hyp-Gly | Ala-Hyp | Pro-Hyp | Gly-Pro-Hyp | |
|---|---|---|---|---|---|---|
| Treatment | ||||||
| CH-GL | 109.2 ± 9.600 | 55.16 ± 16.01 | 304.9 ± 57.2 | 151.4 ± 24.3 * | 22.32 ± 5.09 | |
| CH-OPT | 86.12 ± 14.09 | 28.23 ± 6.55 | 198.0 ± 107.6 | 63.63 ± 8.63 | nd | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Larder, C.E.; Iskandar, M.M.; Kubow, S. Assessment of Bioavailability after In Vitro Digestion and First Pass Metabolism of Bioactive Peptides from Collagen Hydrolysates. Curr. Issues Mol. Biol. 2021, 43, 1592-1605. https://doi.org/10.3390/cimb43030113
Larder CE, Iskandar MM, Kubow S. Assessment of Bioavailability after In Vitro Digestion and First Pass Metabolism of Bioactive Peptides from Collagen Hydrolysates. Current Issues in Molecular Biology. 2021; 43(3):1592-1605. https://doi.org/10.3390/cimb43030113
Chicago/Turabian StyleLarder, Christina E., Michèle M. Iskandar, and Stan Kubow. 2021. "Assessment of Bioavailability after In Vitro Digestion and First Pass Metabolism of Bioactive Peptides from Collagen Hydrolysates" Current Issues in Molecular Biology 43, no. 3: 1592-1605. https://doi.org/10.3390/cimb43030113
APA StyleLarder, C. E., Iskandar, M. M., & Kubow, S. (2021). Assessment of Bioavailability after In Vitro Digestion and First Pass Metabolism of Bioactive Peptides from Collagen Hydrolysates. Current Issues in Molecular Biology, 43(3), 1592-1605. https://doi.org/10.3390/cimb43030113







