Artemisinin-Naphthoquine Combination (ARCO®): An Overview of the Progress
Abstract
:1. Introduction
2. A Literature Overview
2.1. General Aspects
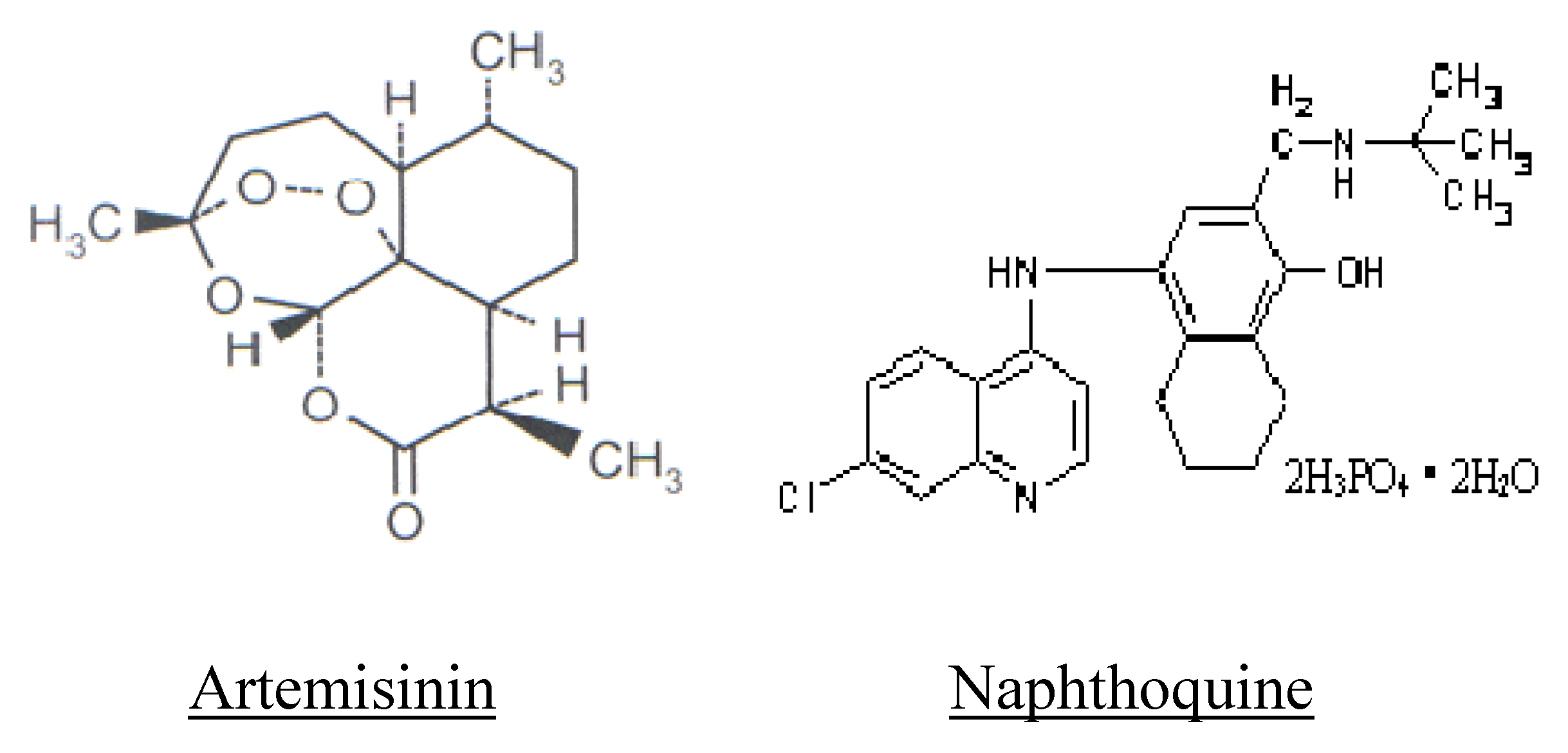
2.2. Co-Formulation
2.3. Therapeutic Dosage Regimen of ARCO®
3. Clinical and Therapeutic Assessment of ARCO®
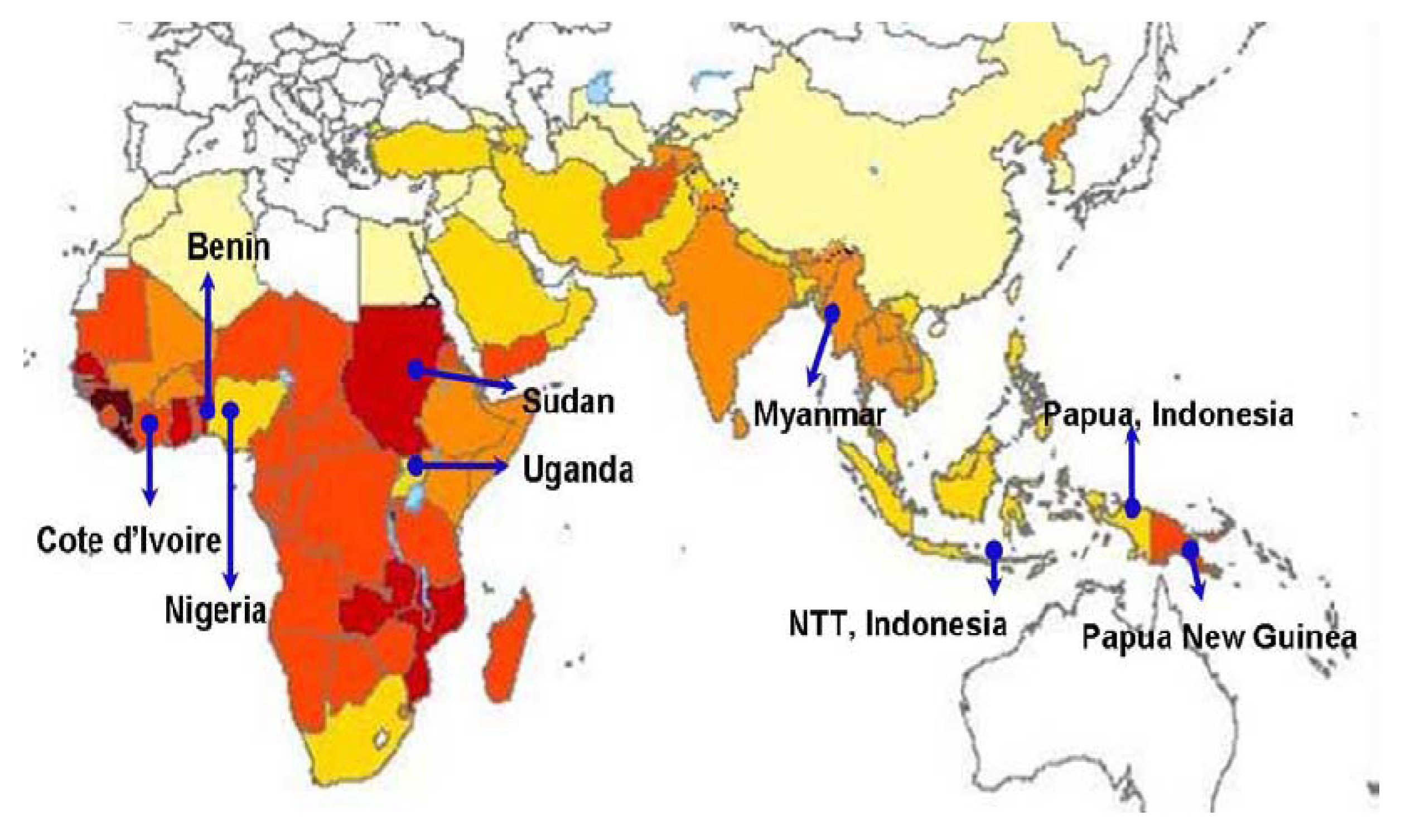
3.1. General Overview
| Countries | Study design (ARCO® vs. others) | Sample size | Measured outcomes | ||
|---|---|---|---|---|---|
| CR/ACPR (%) | PCT (h) | FCT (h) | |||
| Papua New Guinea (adults) | ARCO® | 51 | 100 | 24 | 19.8 |
| CQ + SP | 49 | 98 | 40 | 32.0 | |
| Indonesia (adults) | ARCO® | 78 | 99 | 28 | 13 |
| DHA + PPQ | 74 | 97 | 26 | 11 | |
| Myanmar (adults) | ARCO® | 53 | 98 | 21 | 18 |
| No comparator | __ | 21 | 18.2 | ||
| Sudan (adults) | ARCO® | 129 | 98 | 28 | 12 |
| Art/SP | 71 | 97 | __ | 14 | |
| Art | 65 | 95 | __ | 11 | |
| Uganda (adults) | ARCO® | 118 | 100 | 27 | 24 |
| AL | 116 | 95 | __ | __ | |
| Uganda (children) | ARCO® | 113 | 98 | __ | __ |
| AL | 112 | 98 | __ | __ | |
| Nigeria (adults | ARCO® | 74 | 92 | 24 | 19.5 |
| No comparator | __ | __ | __ | ||
| Nigeria (children) | ARCO® | 50 | 100 | 32 | 24.7 |
| AL | 46 | 96 | __ | __ | |
| Nigeria (mixed) | ARCO® | 62 | 100 | 24 | __ |
| AL | 58 | 96 | 48 | __ | |
| Art/AQ | 54 | 100 | 24 | __ | |
| Benin (children) | ARCO® (1-dose) | 25 | 96 | 24 | 36 |
| ARCO® (2÷doses) | 25 | 96 | __ | 24 | |
| Ivory Coast (mixed) | ARCO® | 60 | 100 | 25 | 24 |
| AL | 60 | 98 | __ | 24 | |
3.2. General Review Findings
3.2.1. Therapeutic efficacy
3.2.2. Safety profile
3.3. Discussion
4. Conclusions
5. Identified Research Gaps
5.1. Children
5.2. Pregnancy
5.3. Therapeutic Dosing Regimen and Pharmacokinetics
Acknowledgements and Contributions
References
- Hastings, I.M.; Watkins, W.M.; White, N.J. The evolution of drug-resistant malaria: the role of drug elimination half-life. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2002, 357, 505–519. [Google Scholar]
- Arav-Boger, R.; Shapiro, T.A. Molecular mechanisms of resistance in antimalarial chemotherapy: the unmet challenge. Annu. Rev. Pharmacol. Toxicol. 2005, 45, 565–585. [Google Scholar]
- Hastings, I.M.; Watkins, W.M. Tolerance is the key to understanding antimalarial drug resistance. Trends Parasitol. 2006, 22, 71–77. [Google Scholar]
- Na Bangchang, K.; Ruengweerayut, R.; Mahamad, P.; Ruengweerayut, K.; Chaijaroenkul, W. Declining efficacy of a three-day combination regimen of mefloquine-artesunate in a multi-drug resistance area along Thai-Myanmar border. Malar. J. 2010, 9, 273–282. [Google Scholar]
- Choi, S.R.; Mukherjee, P.; Avery, M.A. The fight against drug-resistant malaria: novel plasmodial targets and antimalarial drugs. Curr. Med. Chem. 2008, 15, 161–171. [Google Scholar]
- White, N.J.; Ho, M. The pathophysiology of malaria. Adv. Parasitol. 1992, 31, 83–173. [Google Scholar]
- Maitland, K.; Marsh, K. Pathophysiology of severe malaria in children. Acta Tropica 2003, 90, 131–140. [Google Scholar]
- Dondorp, A.M. Pathophysiology, clinical presentation and treatment of cerebral malaria. Neurol. Asia. 2005, 10, 67–77. [Google Scholar]
- White, N.J. Assessment of the pharmacodynamic properties of antimalarial drug in-vivo. Antimicrobial. Agents Chemother. 1997, 41, 1413–1422. [Google Scholar]
- Qinghaosu: Antimalarial Coordinating Research Group. Antimalarial studies on Qinghaosu. Chin. Med. J. 1979, 92, 811–816.
- Jiang, J.B.; Li, G.Q.; Guo, X.B.; Kong, Y.C.; Arnold, K. Antimalarial activity of mefloquine and Qinghaosu. Lancet 1982, 320, 285–288. [Google Scholar]
- White, N.J. Clinical pharmacokinetics and pharmacodynamics of artemisinin and its derivatives. Trans. R. Soc. Trop. Med. Hyg. 1994, 88 Suppl. 1, S41–S43. [Google Scholar]
- Li, G.Q.; Guo, X.B.; Fu, L.C.; Jian, H.X.; Wang, X.H. Clinical trials of artemisinin and its derivatives in the treatment of malaria in China. Trans. R. Soc. Trop. Med. Hyg. 1994, 88 Suppl. 1, S5–S6. [Google Scholar]
- Wang, J.Y.; Shan, C.Q.; Fu, D.D.; Sun, Z.W.; Ding, D.B. Efficacy of naphthoquine, artemisinine and a combination of the two drugs in the treatment of f alciparum malaria. Zhongguo Ji Sheng Chong Xue Yu Ji Sheng Chong Bing Za Zhi 2003, 21, 131–133. [Google Scholar]
- Wang, J.Y.; Cao, W.C.; Shan, C.Q.; Zhang, M.; Li, G.F.; Ding, D.B.; Shi, Y.L.; Wu, B.A. Naphthoquine phosphate and its combination with artemisinin. Acta Tropica 2004, 89, 375–381. [Google Scholar]
- Wang, J.Y.; Ding, D.B.; Li, G.F.; Zhao, J.H. Therapeutic efficacy of naphthoquine phosphate combined with artemisinine against Plasmodium knowlesi. Zhongguo Ji Sheng Chong Xue Yu Ji Sheng Chong Bing Za Zhi 2008, 26, 442–444. [Google Scholar]
- Tun, T.; Tint, H.S.; Lin, K.; Kyaw, T.T.; Myunt, M.K.; Khaing, W.; Tun, Z.U. Efficacy of oral single dose therapy with artemisinin-naphthoquine phosphate in uncomplicated falciparum malaria. Acta Tropica 2009, 111, 275–278. [Google Scholar]
- Toure, O.A.; Penali, L.K.; Yapi, J.; Ako, B.A.; Toure, W.; Djerea, K.; Gomez, G.O.; Makaila, O.A. Comparative, randomized clinical trial of artemisinin/naphthoquine twice daily one day versus artemether/lumefantrine six doses regimen in children and adults with uncomplicated falciparum malaria in Cote d’Ivoire. Malar. J. 2009, 8, 148–158. [Google Scholar]
- Hombhanje, F.W.; Linge, D.; Saweri, A.; Kuanch, C.; Jones, R.; Toraso, S.; Geita, J.; Masta, A.; Kevau, I.; Hiawalyer, G.; Sapuri, M. Artemisinin-naphthoquine combination (ARCO™) therapy for uncomplicated falciparum malaria in adults of Papua New Guinea: a preliminary report on safety and efficacy. Malar. J. 2009, 8, 196–304. [Google Scholar]
- Klayman, D.L. Qinghaosu (artemisinin): an antimalarial drug from China. Science 1985, 228, 1049–1055. [Google Scholar]
- Ye, L.; Xu, G.; Wang, R.; Li, J.; Zhu, C.; Tang, S.; Sun, Y. Hundred and one cases of Plasmodium falciparum patients treated with naphthoquine phosphate (in Chinese). Chin. J. Parasitol. Parasitic. Dis. 1999, 17, 20–23. [Google Scholar]
- Kuhn, T.; Wang, Y. Artemisinin-an innovative cornerstone for anti-malaria therapy. Prog. Drug. Res. 2008, 66, 385–422. [Google Scholar]
- Guo, W.; Guo, X.; Zheng, Q.; Tan, B.; Chen, R.; Ou, F.; Fu, L. A randomized-control study of naphthoquine and artesunate in the treatment of falciparum malaria. J. Guangzhou Uni. Trad. Chin. Med. 2000, 17, 235–237. [Google Scholar]
- Cui, L.; Su, X.Z. Discovery, mechanisms of action and combination therapy of artemisinin. Expert Rev. Anti-Infect. Ther. 2009, 7, 999–1013. [Google Scholar]
- Medhi, B.; Patyar, S.; Rao, R.S.; Byrav, P.D.S.; Prakash, A. Pharmacokinetic and toxicological profile of artemisinin compounds: an update. Pharmacology 2009, 84, 323–332. [Google Scholar]
- Qu, H.Y.; Gao, H.Z.; Hao, G.T.; Li, Y.Y.; Li, H.Y.; Hu, J.C.; Wang, X.F.; Liu, W.L.; Liu, Z.Y. Single-Dose safety, pharmacokinetics, and food effects studies of compound naphthoquine phosphate tablets in healthy volunteers. J. Clin. Pharmacol. 2010. [Google Scholar]
- Wang, J.; Huang, L.; Li, J.; Fan, Q.; Long, Y.; Li, Y.; Zhou, B. Artemisinin directly targets malarial mitochondria through its specific mitochondrial activation. PLoS One 2010, 5, e9582. [Google Scholar]
- Giao, P.T.; de Vries, P.J. Pharmacokinetic interactions of antimalarial agents. Clin. Pharmacokinet. 2001, 40, 343–373. [Google Scholar]
- Martinelli, A.; Moreira, R.; Ravo, P.V. Malaria combination therapies: advantages and shortcomings. Mini Rev. Med. Chem. 2008, 8, 201–212. [Google Scholar]
- World Health Organization, Assessment and Monitoring of Antimalarial Drug Efficacy for the Treatment of Uncomplicated Falciparum Malaria; WHO: Geneva, Switzerland, 2003.
- WHO. Guidelines for the Treatment of Malaria; 2nd. WHO: Geneva, Switzerland, 2003. Available online: Whqlibdoc.who.int/publications 2010/9789241547925_eng.pdf/ accessed on 14 December 2010.
- World Health Organization, Antimalarial drug combination therapy. In Report of a WHO Technical Consultation (WHO/CDS/RBM/2001.35); WHO: Geneva, Switzerland, 2001.
- Lefevre, G.; Looareesuwan, S.; Treeprasertsuk, S.; Krudsood, S.; Silachamroon, U.; Gathmann, I.; Mull, R.; Bakshi, R. A clinical and pharmacokinetic trial of six doses of artemether-lumefantrine for multidrug resistant Plasmodium falciparum malaria in Thailand. Am. J. Trop. Med. Hyg. 2001, 64, 247–256. [Google Scholar]
- Krudsood, S.; Chalermrut, K.; Pengruksa, C.; Srivilairit, S.; Silachamroon, U.; Treeprasetsuk, S.; Kano, S.; et al. Comparative clinical trial of two-fixed combinations of dihydroartemisinin-naphthoquine-trimethoprim (DNP) and artemether-lumefantrine (Coartem/Riamet) in the treatment of acute uncomplicated falciparum malaria in Thailand. Southeast Asian J. Trop. Med. Public. Health 2003, 34, 316–321. [Google Scholar]
- van den Broek, I.; Kitx, C.; Al Atas, S.; Libama, F.; Balasegaram, M.; Guthmann, J.-P. Efficacy of three artemisinin combination therapies for the treatment of uncomplicated Plasmodium falciparum malaria in the Republic of Congo. Malar. J. 2006, 5, 113–116. [Google Scholar]
- Kokwaro, G.; Mwai, L.; Nzila, A. Artemether/lumefantrine in the treatment of uncomplicated falciparum malaria. Exp. Opi. Pharmacother. 2007, 8, 75–94. [Google Scholar]
- Falade, C.; Makanga, M.; Premji, Z.; Ortmann, C.E.; Stockmeyer, M.; de Palacios, P.I. Efficacy and safety of artemether-lumefantrine (Coartem®) tablets (six-dose regimen) in African infants and children with acute, uncomplicated falciparum malaria. Trans. R. Soc. Trop. Med. Hyg. 2005, 99, 459–467. [Google Scholar]
- Mulenga, M.; VangGeertruyden, J.P.; Mwananyanda, L.; Chalwe, V.; Moerman, F.; Chilengi, R.; Van Overmeir, C.; Dujardin, J.C.; D'Alessandro, U. Safety and efficacy of lumefantrine-artemether (Coartem®) for the treatment of uncomplicated Plasmodium falciparum malaria in Zambian adults. Malar. J. 2006, 5, 73–80. [Google Scholar]
- Jackson, Y.; Chappuis, F.; Loutan, L.; Taylor, W. Malaria treatment failures after artemisinin-based therapy in three expatriates: could improved manufacturer information help to decrease the risk of treatment failure? Malar. J. 2006, 5, 81–85. [Google Scholar]
- Wasunna, W.; Zurovac, D.; Goodman, C.A.; Robert, W.; Snow, R.W. Why don't health workers prescribe ACT? A qualitative study of factors affecting the prescription of artemether-lumefantrine. Malar. J. 2008, 7, 29–37. [Google Scholar]
- Price, R.N.; Nosten, F.; Luxemburger, C.; ter Kuile, F.O.; Paiphun, L.; Price, R.N.; Chongsuphajaisiddhi, T.; White, N.J. Effects of artemisinin derivatives on malaria transmissibility. Lancet 1996, 347, 1654–1658. [Google Scholar]
- Okell, L.C.; Drakeley, C.J.; Ghani, A.C.; Bousema, T.; Sutherland, C.J. Reduction of transmission from malaria patients by artemisinin combination therapies: a pooled analysis of six randomized trials. Malar. J. 2008, 7, 125–137. [Google Scholar]
- Barnes, K.I.; Chanda, P.; Barnabas, G. Impact of large-scale deployment of artemether-lumefantrine on the malaria disease burden in Africa: case study of South Africa, Zambia and Ethiopia. Malar. J. 2009, 8 suppl. 1, S8. [Google Scholar]
- Qigui, L.; Weina, P.J. Severe Embryotoxicity of artemisinin derivatives in experimental animals, but possibly safe in pregnant women. Molecules 2010, 15, 40–57. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hombhanje, F.W.; Huang, Q. Artemisinin-Naphthoquine Combination (ARCO®): An Overview of the Progress. Pharmaceuticals 2010, 3, 3581-3593. https://doi.org/10.3390/ph3123581
Hombhanje FW, Huang Q. Artemisinin-Naphthoquine Combination (ARCO®): An Overview of the Progress. Pharmaceuticals. 2010; 3(12):3581-3593. https://doi.org/10.3390/ph3123581
Chicago/Turabian StyleHombhanje, Francis W., and Qingyun Huang. 2010. "Artemisinin-Naphthoquine Combination (ARCO®): An Overview of the Progress" Pharmaceuticals 3, no. 12: 3581-3593. https://doi.org/10.3390/ph3123581
APA StyleHombhanje, F. W., & Huang, Q. (2010). Artemisinin-Naphthoquine Combination (ARCO®): An Overview of the Progress. Pharmaceuticals, 3(12), 3581-3593. https://doi.org/10.3390/ph3123581




