Analysis of Immune Checkpoints on Peripheral Blood Mononuclear Cells Can Predict Clinical Outcome and Reveal Potential of HVEM-BTLA Axis in Epithelial Ovarian Cancers
Abstract
1. Introduction
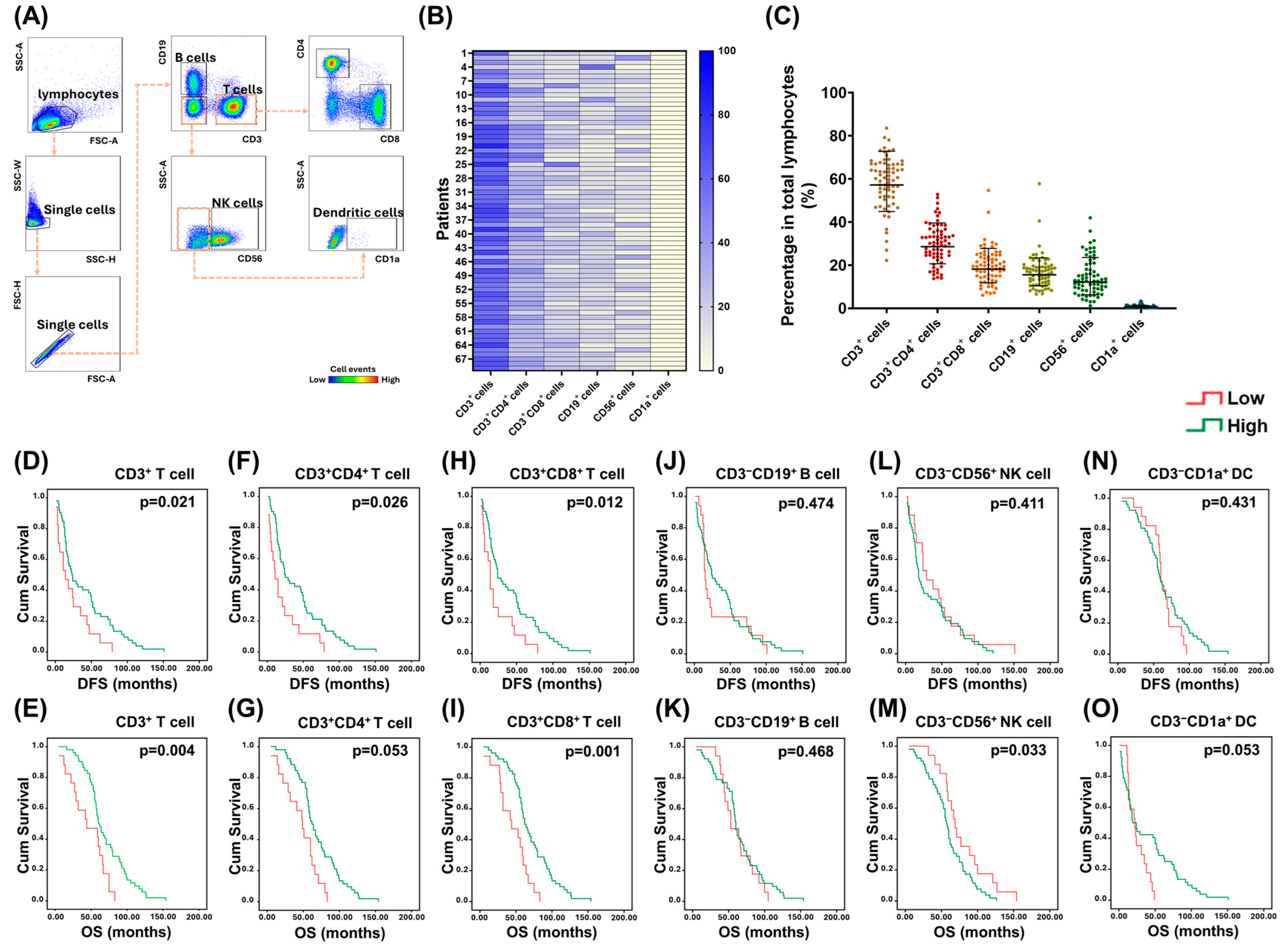
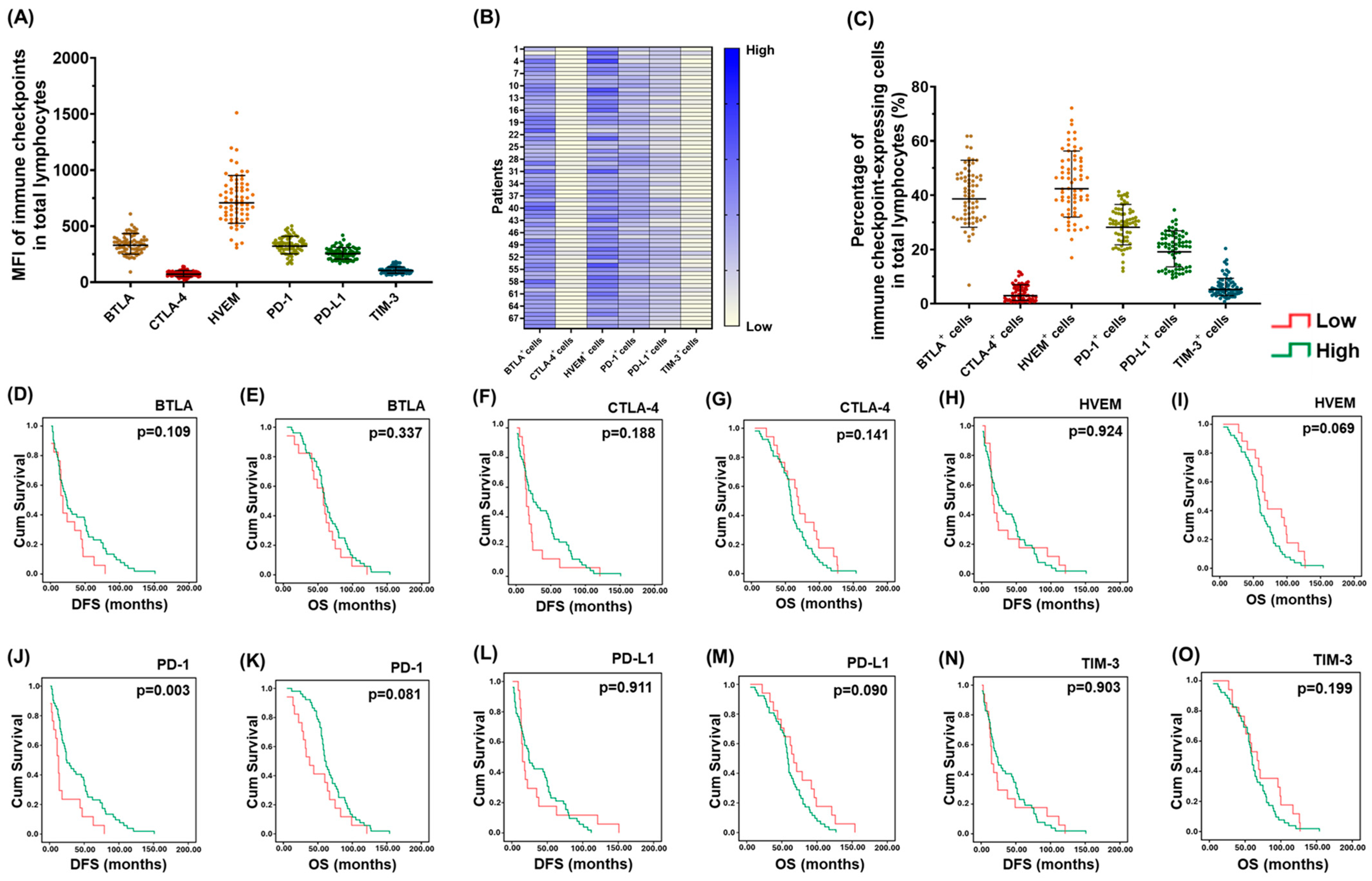
2. Results
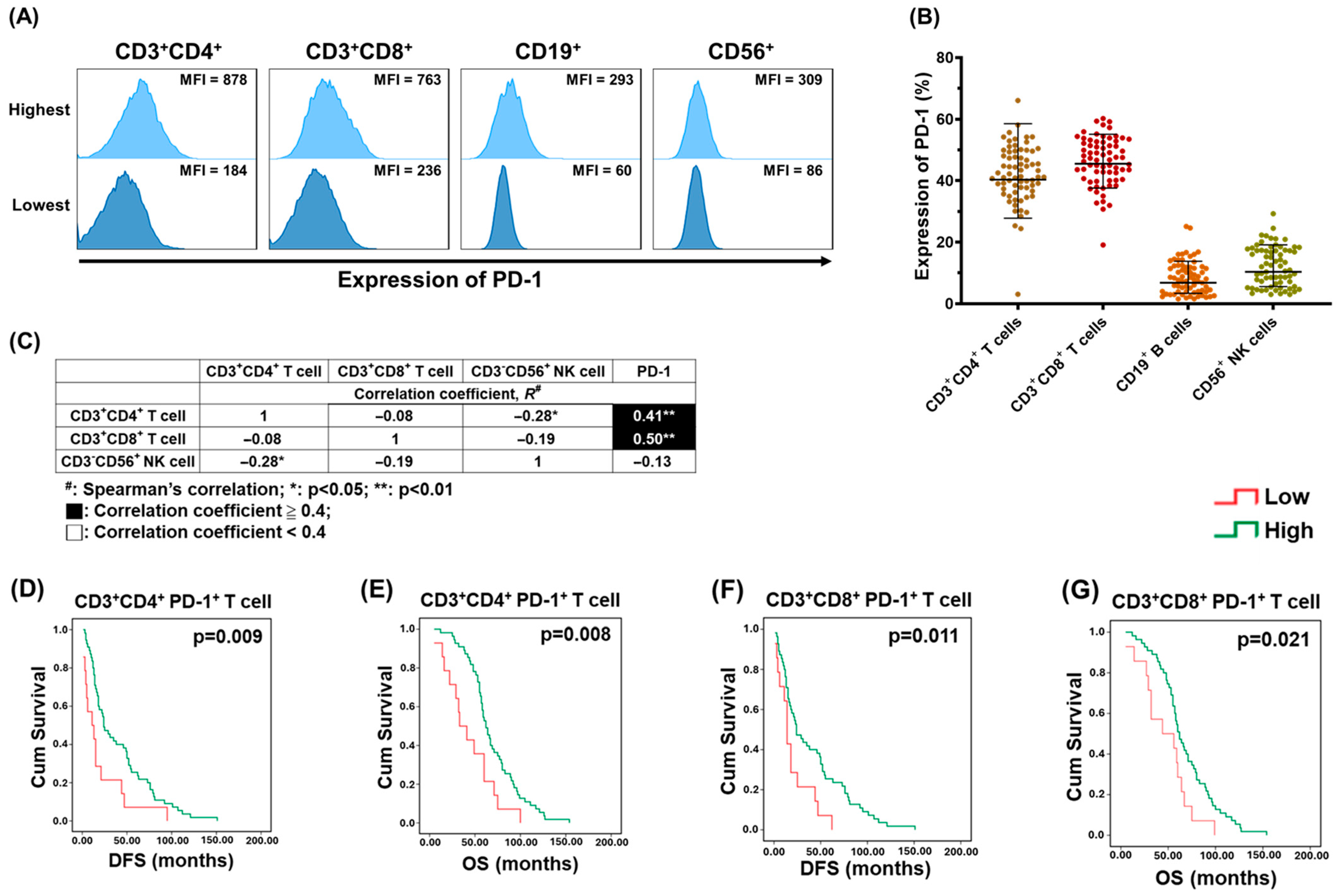
2.1. PD-1+ T lymphocytes in PBMCs Were Associated with the Survival of EOC Patients
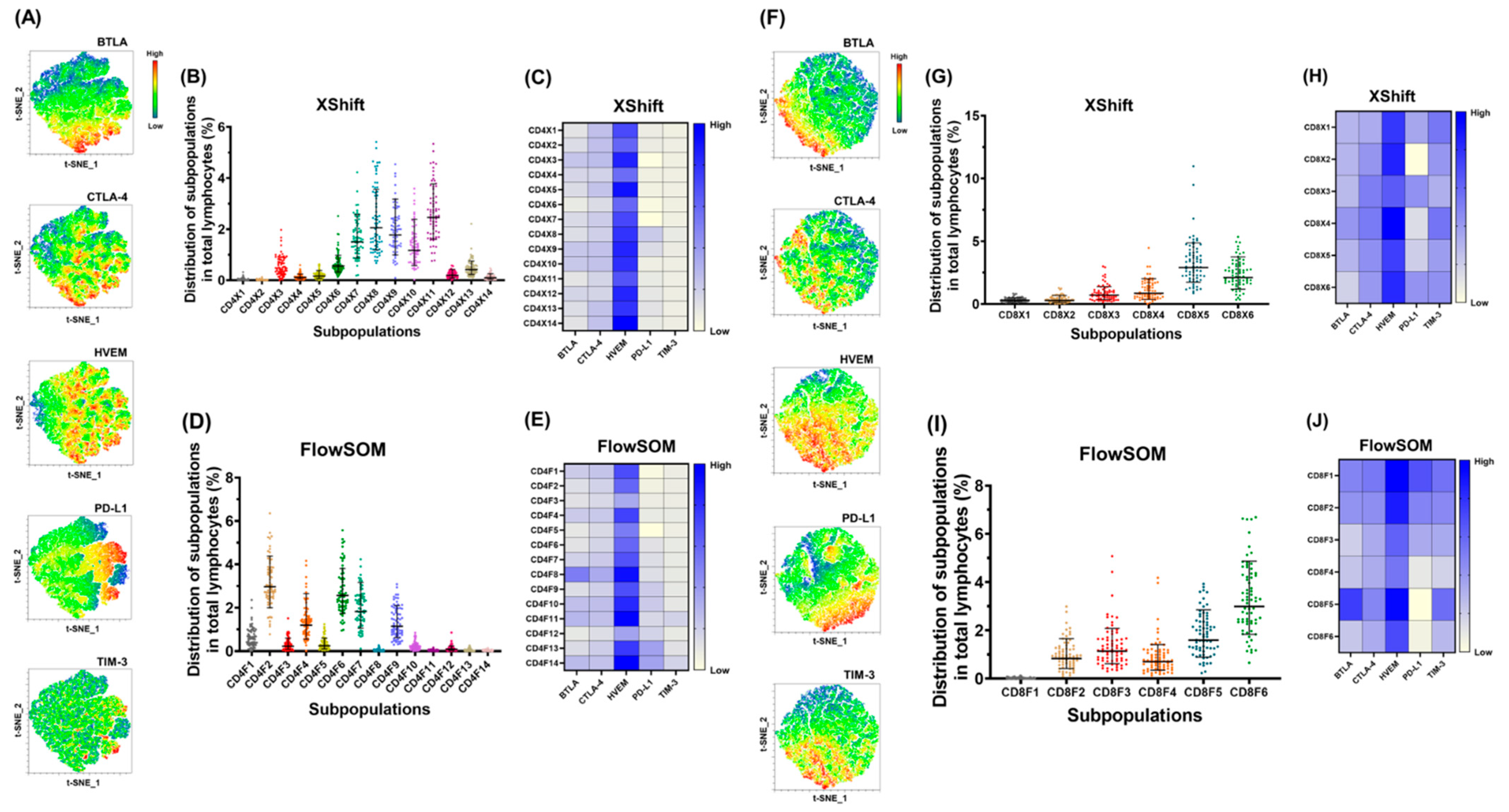
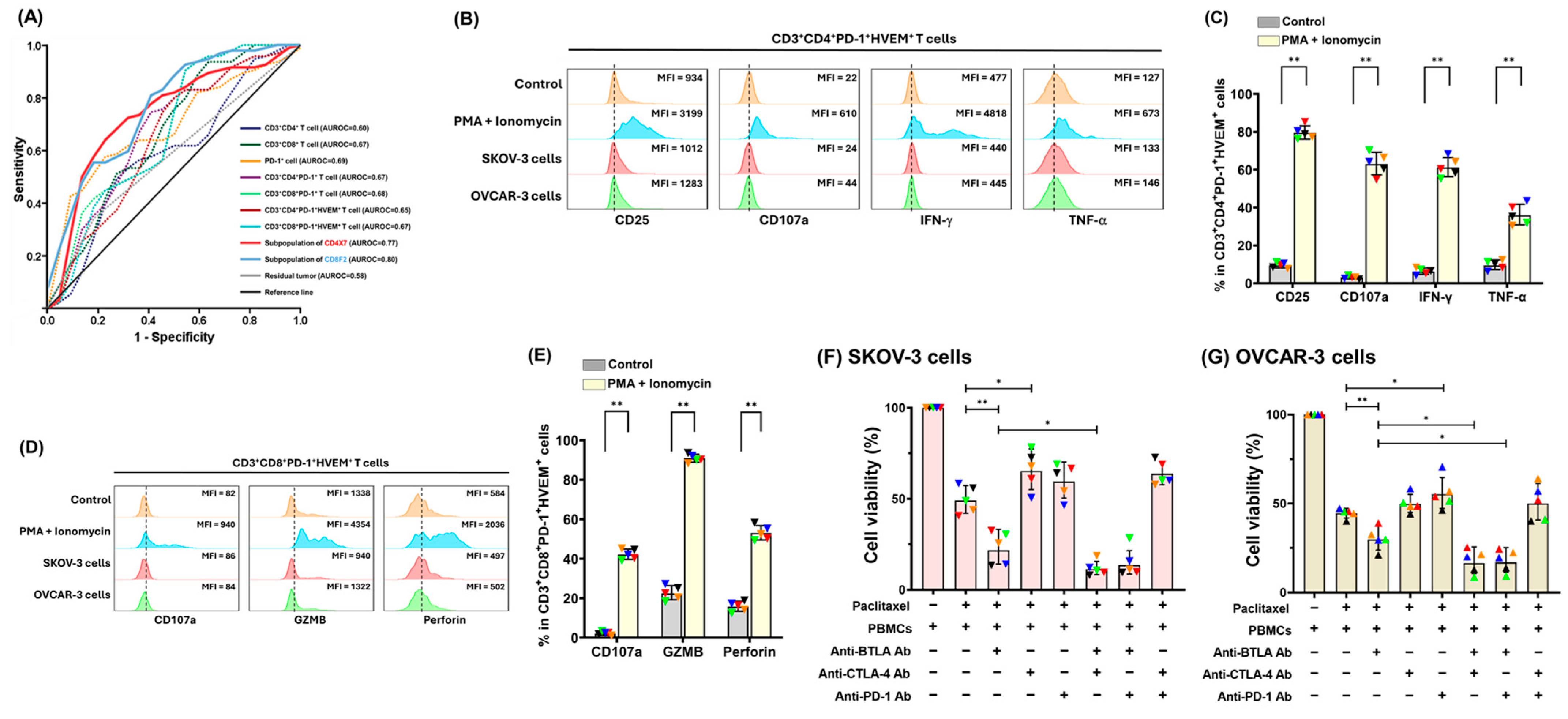
2.2. Co-Expression of HVEM on PD-1+ T lymphocytes Was Highly Detected
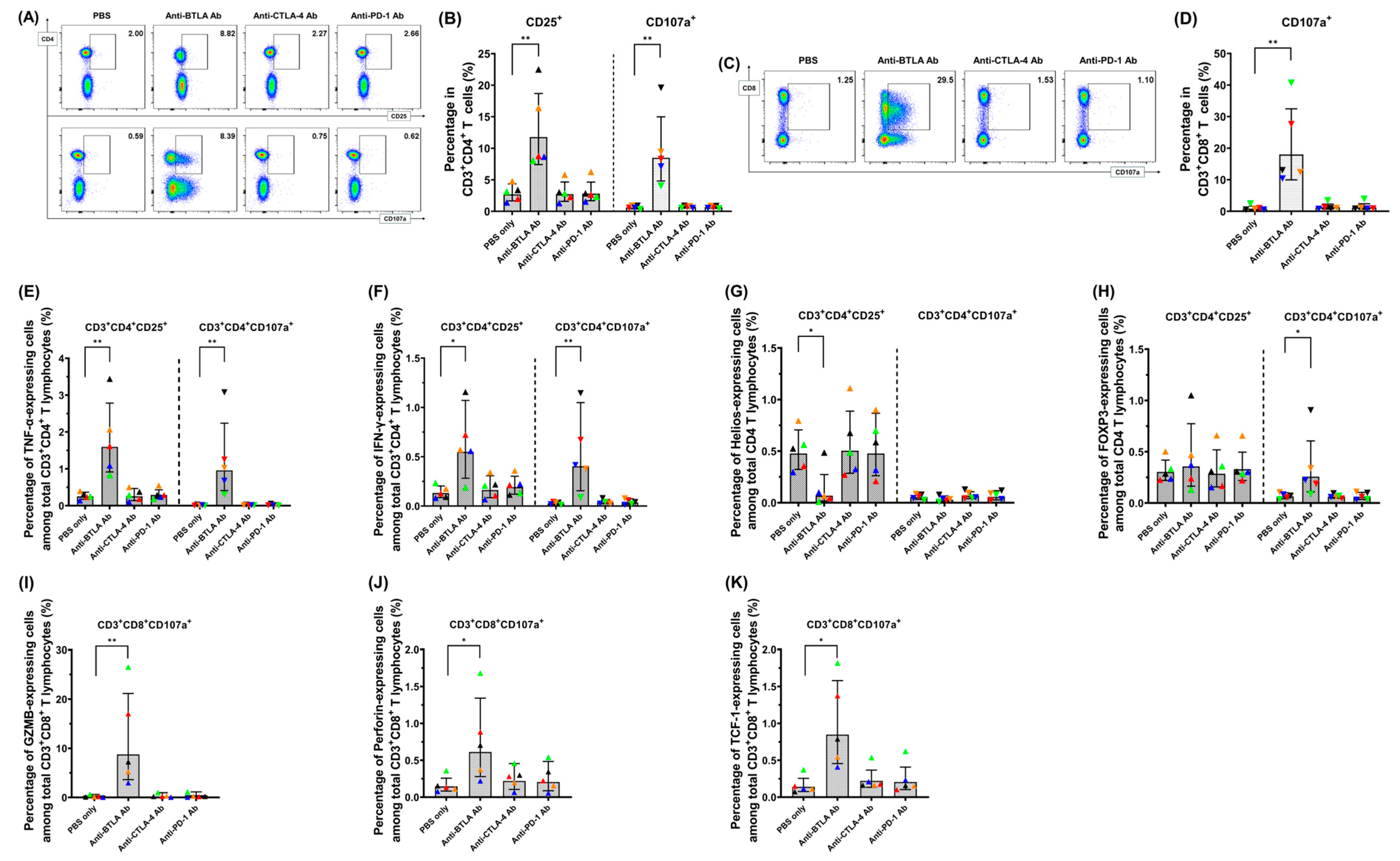
2.3. Addition of Anti-BTLA Ab to Other ICIs Can Generate More Potent Tumoricidal Effects
3. Discussion
4. Materials and Methods
4.1. Patient and PBMC Preparation
4.2. Surface and Intracellular Staining of PBMCs Analyzed by Flow Cytometry, t-SNE, XShift, and FlowSOM
4.3. Preparation of Cell Lines
4.4. Preparation of PTX and Anti-PD-1, CTLA-4, and BTLA Abs
4.5. Activation Potential of PD-1+HVEM+ T Cells in PBMCs from Healthy Donors
4.6. Tumoricidal Abilities of PBMCs from the Healthy Donors Treated with Various ICI Strategies
4.7. Impact of Respective ICIs on Activation of T Cells in PMBCs from Healthy Donors
4.8. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| EOC | epithelial ovarian cancer |
| PTX | paclitaxel |
| PARP | poly-ADP ribose polymerase |
| ICIs | immune checkpoint inhibitors |
| CTLA-4 | cytotoxic T lymphocyte antigen 4 |
| PD-1 | programmed cell death 1 |
| PD-L1 | programmed death-ligand 1 |
| ICs | immune checkpoints |
| TME | tumor microenvironment |
| PBMCs | peripheral blood mononuclear cells |
| t-SNE | t-distributed stochastic neighbor embedding |
| Abs | antibodies |
| DFS | disease-free survival |
| OS | overall survival |
| FBS | fetal bovine serum |
| BTLA | B and T lymphocyte attenuator |
| HVEM | herpes virus entry mediator |
| PMA | phorbol myristate acetate |
| Tregs | regulatory T cells |
| TCF-1 | T-cell factor 1 |
| ROC curve | receiver operating characteristic curve |
| AUROC | area under the ROC curve |
| TIM-3 | T-cell immunoglobulin domain and mucin domain 3 |
| GZMB | granzyme B |
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Bowtell, D.D.; Böhm, S.; Ahmed, A.A.; Aspuria, P.-J.; Bast, R.C.; Beral, V.; Berek, J.S.; Birrer, M.J.; Blagden, S.; Bookman, M.A.; et al. Rethinking Ovarian Cancer II: Reducing Mortality from High-Grade Serous Ovarian Cancer. Nat. Rev. Cancer 2015, 15, 668–679. [Google Scholar] [CrossRef]
- Armstrong, D.K.; Alvarez, R.D.; Bakkum-Gamez, J.N.; Barroilhet, L.; Behbakht, K.; Berchuck, A.; Chen, L.-M.; Cristea, M.; DeRosa, M.; Eisenhauer, E.L.; et al. Ovarian Cancer, Version 2.2020, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Canc Netw. 2021, 19, 191–226. [Google Scholar] [CrossRef]
- Moore, K.; Colombo, N.; Scambia, G.; Kim, B.-G.; Oaknin, A.; Friedlander, M.; Lisyanskaya, A.; Floquet, A.; Leary, A.; Sonke, G.S.; et al. Maintenance Olaparib in Patients with Newly Diagnosed Advanced Ovarian Cancer. N. Engl. J. Med. 2018, 379, 2495–2505. [Google Scholar] [CrossRef]
- González-Martín, A.; Pothuri, B.; Vergote, I.; DePont Christensen, R.; Graybill, W.; Mirza, M.R.; McCormick, C.; Lorusso, D.; Hoskins, P.; Freyer, G.; et al. Niraparib in Patients with Newly Diagnosed Advanced Ovarian Cancer. N. Engl. J. Med. 2019, 381, 2391–2402. [Google Scholar] [CrossRef]
- Ledermann, J.; Harter, P.; Gourley, C.; Friedlander, M.; Vergote, I.; Rustin, G.; Scott, C.L.; Meier, W.; Shapira-Frommer, R.; Safra, T.; et al. Olaparib Maintenance Therapy in Patients with Platinum-Sensitive Relapsed Serous Ovarian Cancer: A Preplanned Retrospective Analysis of Outcomes by BRCA Status in a Randomised Phase 2 Trial. Lancet Oncol. 2014, 15, 852–861. [Google Scholar] [CrossRef] [PubMed]
- Poveda, A.M.; Selle, F.; Hilpert, F.; Reuss, A.; Savarese, A.; Vergote, I.; Witteveen, P.; Bamias, A.; Scotto, N.; Mitchell, L.; et al. Bevacizumab Combined with Weekly Paclitaxel, Pegylated Liposomal Doxorubicin, or Topotecan in Platinum-Resistant Recurrent Ovarian Cancer: Analysis by Chemotherapy Cohort of the Randomized Phase III AURELIA Trial. J. Clin. Oncol. 2015, 33, 3836–3838. [Google Scholar] [CrossRef] [PubMed]
- Pardoll, D.M. The Blockade of Immune Checkpoints in Cancer Immunotherapy. Nat. Rev. Cancer 2012, 12, 252–264. [Google Scholar] [CrossRef] [PubMed]
- Topalian, S.L.; Hodi, F.S.; Brahmer, J.R.; Gettinger, S.N.; Smith, D.C.; McDermott, D.F.; Powderly, J.D.; Carvajal, R.D.; Sosman, J.A.; Atkins, M.B.; et al. Safety, Activity, and Immune Correlates of Anti-PD-1 Antibody in Cancer. N. Engl. J. Med. 2012, 366, 2443–2454. [Google Scholar] [CrossRef]
- Hodi, F.S.; O’Day, S.J.; McDermott, D.F.; Weber, R.W.; Sosman, J.A.; Haanen, J.B.; Gonzalez, R.; Robert, C.; Schadendorf, D.; Hassel, J.C.; et al. Improved Survival with Ipilimumab in Patients with Metastatic Melanoma. N. Engl. J. Med. 2010, 363, 711–723. [Google Scholar] [CrossRef]
- Hamid, O.; Robert, C.; Daud, A.; Hodi, F.S.; Hwu, W.-J.; Kefford, R.; Wolchok, J.D.; Hersey, P.; Joseph, R.W.; Weber, J.S.; et al. Safety and Tumor Responses with Lambrolizumab (Anti-PD-1) in Melanoma. N. Engl. J. Med. 2013, 369, 134–144. [Google Scholar] [CrossRef]
- Hamanishi, J.; Mandai, M.; Iwasaki, M.; Okazaki, T.; Tanaka, Y.; Yamaguchi, K.; Higuchi, T.; Yagi, H.; Takakura, K.; Minato, N.; et al. Programmed Cell Death 1 Ligand 1 and Tumor-Infiltrating CD8+ T lymphocytes Are Prognostic Factors of Human Ovarian Cancer. Proc. Natl. Acad. Sci. USA 2007, 104, 3360–3365. [Google Scholar] [CrossRef] [PubMed]
- Ma, V.T.; Milhem, M.M.; Hidalgo, M.; Shields, A.F.; Segar, J.M.; Khalil, M.; Davis, A.A.; Verschraegen, C.F.; Wong, D.J.L.; Schilder, R.J.; et al. Phase I Dose-Escalation and Cohort Expansion Study of the Anti-BTLA Antibody, Tifcemalimab, in Combination with Toripalimab (Anti-PD-1) in Heavily Pretreated Patients (Pts) with Advanced Malignancies. J. Clin. Oncol. 2024, 42, 2596. [Google Scholar] [CrossRef]
- Xu, C.; Cai, Q.; Ma, J.; Yang, K.; Wang, S.; Shen, L.; Qu, S.; Huang, J.; Huang, X.; Tang, L.-L.; et al. Combination of Tim-3 Blockade TQB2618 with Penpulimab and Chemotherapy in the First-Line Treatment of Recurrent/Metastatic Nasopharyngeal Carcinoma (R/M NPC): A Multicenter, Single-Arm, Two-Cohort, Phase 2 Study. J. Clin. Oncol. 2025, 43, 6031. [Google Scholar] [CrossRef]
- Sun, Q.; Hong, Z.; Zhang, C.; Wang, L.; Han, Z.; Ma, D. Immune Checkpoint Therapy for Solid Tumours: Clinical Dilemmas and Future Trends. Sig Transduct. Target. Ther. 2023, 8, 320. [Google Scholar] [CrossRef] [PubMed]
- Sordo-Bahamonde, C.; Lorenzo-Herrero, S.; Granda-Díaz, R.; Martínez-Pérez, A.; Aguilar-García, C.; Rodrigo, J.P.; García-Pedrero, J.M.; Gonzalez, S. Beyond the Anti-PD-1/PD-L1 Era: Promising Role of the BTLA/HVEM Axis as a Future Target for Cancer Immunotherapy. Mol. Cancer 2023, 22, 142. [Google Scholar] [CrossRef] [PubMed]
- Cheung, T.C.; Oborne, L.M.; Steinberg, M.W.; Macauley, M.G.; Fukuyama, S.; Sanjo, H.; D’Souza, C.; Norris, P.S.; Pfeffer, K.; Murphy, K.M.; et al. T Cell Intrinsic Heterodimeric Complexes between HVEM and BTLA Determine Receptivity to the Surrounding Microenvironment. J. Immunol. 2009, 183, 7286–7296. [Google Scholar] [CrossRef]
- Peng, H.; He, X.; Wang, Q. Immune Checkpoint Blockades in Gynecological Cancers: A Review of Clinical Trials. Acta Obstet. Gynecol. Scand. 2022, 101, 941–951. [Google Scholar] [CrossRef]
- Thommen, D.S.; Schreiner, J.; Müller, P.; Herzig, P.; Roller, A.; Belousov, A.; Umana, P.; Pisa, P.; Klein, C.; Bacac, M.; et al. Progression of Lung Cancer Is Associated with Increased Dysfunction of T Cells Defined by Coexpression of Multiple Inhibitory Receptors. Cancer Immunol. Res. 2015, 3, 1344–1355. [Google Scholar] [CrossRef]
- Demerlé, C.; Gorvel, L.; Olive, D. BTLA-HVEM Couple in Health and Diseases: Insights for Immunotherapy in Lung Cancer. Front. Oncol. 2021, 11, 682007. [Google Scholar] [CrossRef]
- Wherry, E.J.; Kurachi, M. Molecular and Cellular Insights into T Cell Exhaustion. Nat. Rev. Immunol. 2015, 15, 486–499. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.-Z.; Lin, H.-W.; Chen, W.-Y.; Chien, C.-L.; Lai, Y.-L.; Chen, J.; Chen, Y.-L.; Cheng, W.-F. Dual Inhibition of BTLA and PD-1 Can Enhance Therapeutic Efficacy of Paclitaxel on Intraperitoneally Disseminated Tumors. J. Immunother. Cancer 2023, 11, e006694. [Google Scholar] [CrossRef]
- Curiel, T.J.; Coukos, G.; Zou, L.; Alvarez, X.; Cheng, P.; Mottram, P.; Evdemon-Hogan, M.; Conejo-Garcia, J.R.; Zhang, L.; Burow, M.; et al. Specific Recruitment of Regulatory T Cells in Ovarian Carcinoma Fosters Immune Privilege and Predicts Reduced Survival. Nat. Med. 2004, 10, 942–949. [Google Scholar] [CrossRef]
- Palucka, A.K.; Coussens, L.M. The Basis of Oncoimmunology. Cell 2016, 164, 1233–1247. [Google Scholar] [CrossRef]
- Zhang, L.; Conejo-Garcia, J.R.; Katsaros, D.; Gimotty, P.A.; Massobrio, M.; Regnani, G.; Makrigiannakis, A.; Gray, H.; Schlienger, K.; Liebman, M.N.; et al. Intratumoral T Cells, Recurrence, and Survival in Epithelial Ovarian Cancer. N. Engl. J. Med. 2003, 348, 203–213. [Google Scholar] [CrossRef]
- Schreiber, R.D.; Old, L.J.; Smyth, M.J. Cancer Immunoediting: Integrating Immunity’s Roles in Cancer Suppression and Promotion. Science 2011, 331, 1565–1570. [Google Scholar] [CrossRef]
- Chatterjee, J.; Dai, W.; Aziz, N.H.A.; Teo, P.Y.; Wahba, J.; Phelps, D.L.; Maine, C.J.; Whilding, L.M.; Dina, R.; Trevisan, G.; et al. Clinical Use of Programmed Cell Death-1 and Its Ligand Expression as Discriminatory and Predictive Markers in Ovarian Cancer. Clin. Cancer Res. 2017, 23, 3453–3460. [Google Scholar] [CrossRef]
- Greer, A.; Gockley, A.; Manning-Geist, B.; Melamed, A.; Sisodia, R.C.; Berkowitz, R.; Horowitz, N.; Del Carmen, M.; Growdon, W.B.; Worley, M. Impact of Residual Disease at Interval Debulking Surgery on Platinum Resistance and Patterns of Recurrence for Advanced-Stage Ovarian Cancer. Int. J. Gynecol. Cancer 2021, 31, 1341–1347. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, S.M.; Schnack, T.H.; Høgdall, C. Impact of Residual Disease on Overall Survival in Women with Federation of Gynecology and Obstetrics Stage IIIB-IIIC vs Stage IV Epithelial Ovarian Cancer after Primary Surgery. Acta Obstet. Gynecol. Scand. 2019, 98, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Dabi, Y.; Huchon, C.; Ouldamer, L.; Bendifallah, S.; Collinet, P.; Bricou, A.; Daraï, E.; Ballester, M.; Lavoue, V.; Haddad, B.; et al. Patients with Stage IV Epithelial Ovarian Cancer: Understanding the Determinants of Survival. J. Transl. Med. 2020, 18, 134. [Google Scholar] [CrossRef] [PubMed]
- MacFarlane, A.W.; Jillab, M.; Plimack, E.R.; Hudes, G.R.; Uzzo, R.G.; Litwin, S.; Dulaimi, E.; Al-Saleem, T.; Campbell, K.S. PD-1 Expression on Peripheral Blood Cells Increases with Stage in Renal Cell Carcinoma Patients and Is Rapidly Reduced after Surgical Tumor Resection. Cancer Immunol. Res. 2014, 2, 320–331. [Google Scholar] [CrossRef]
- Galon, J.; Bruni, D. Approaches to Treat Immune Hot, Altered and Cold Tumours with Combination Immunotherapies. Nat. Rev. Drug Discov. 2019, 18, 197–218. [Google Scholar] [CrossRef] [PubMed]
- Blanc-Durand, F.; Clemence Wei Xian, L.; Tan, D.S.P. Targeting the Immune Microenvironment for Ovarian Cancer Therapy. Front. Immunol. 2023, 14, 1328651. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-L.; Lin, H.-W.; Chien, C.-L.; Lai, Y.-L.; Sun, W.-Z.; Chen, C.-A.; Cheng, W.-F. BTLA Blockade Enhances Cancer Therapy by Inhibiting IL-6/IL-10-Induced CD19high B lymphocytes. J. Immunother. Cancer 2019, 7, 313. [Google Scholar] [CrossRef] [PubMed]
- Wilky, B.A. Immune Checkpoint Inhibitors: The Linchpins of Modern Immunotherapy. Immunol. Rev. 2019, 290, 6–23. [Google Scholar] [CrossRef]
- Emoto, M.; Kaufmann, S.H.E. Liver NKT Cells: An Account of Heterogeneity. Trends Immunol. 2003, 24, 364–369. [Google Scholar] [CrossRef]
- Chen, Y.-L.; Chang, M.-C.; Huang, C.-Y.; Chiang, Y.-C.; Lin, H.-W.; Chen, C.-A.; Hsieh, C.-Y.; Cheng, W.-F. Serous Ovarian Carcinoma Patients with High Alpha-Folate Receptor Had Reducing Survival and Cytotoxic Chemo-Response. Mol. Oncol. 2012, 6, 360–369. [Google Scholar] [CrossRef]
- Han, P.; Goularte, O.D.; Rufner, K.; Wilkinson, B.; Kaye, J. An Inhibitory Ig Superfamily Protein Expressed by lymphocytes and APCs Is Also an Early Marker of Thymocyte Positive Selection. J. Immunol. 2004, 172, 5931–5939. [Google Scholar] [CrossRef]
- Chang, M.-C.; Chen, Y.-L.; Lin, H.-W.; Chiang, Y.-C.; Chang, C.-F.; Hsieh, S.-F.; Chen, C.-A.; Sun, W.-Z.; Cheng, W.-F. Irradiation Enhances Abscopal Anti-Tumor Effects of Antigen-Specific Immunotherapy through Regulating Tumor Microenvironment. Mol. Ther. 2018, 26, 404–419. [Google Scholar] [CrossRef]
- Compaan, D.M.; Gonzalez, L.C.; Tom, I.; Loyet, K.M.; Eaton, D.; Hymowitz, S.G. Attenuating Lymphocyte Activity: The Crystal Structure of the BTLA-HVEM Complex. J. Biol. Chem. 2005, 280, 39553–39561. [Google Scholar] [CrossRef]
- Kim, H.-J.; Barnitz, R.A.; Kreslavsky, T.; Brown, F.D.; Moffett, H.; Lemieux, M.E.; Kaygusuz, Y.; Meissner, T.; Holderried, T.A.W.; Chan, S.; et al. Stable Inhibitory Activity of Regulatory T Cells Requires the Transcription Factor Helios. Science 2015, 350, 334–339. [Google Scholar] [CrossRef]
- Wu, T.; Ji, Y.; Moseman, E.A.; Xu, H.C.; Manglani, M.; Kirby, M.; Anderson, S.M.; Handon, R.; Kenyon, E.; Elkahloun, A.; et al. The TCF1-Bcl6 Axis Counteracts Type I Interferon to Repress Exhaustion and Maintain T Cell Stemness. Sci. Immunol. 2016, 1, eaai8593. [Google Scholar] [CrossRef] [PubMed]
- Schober, P.; Boer, C.; Schwarte, L.A. Correlation Coefficients: Appropriate Use and Interpretation. Anesth. Analg. 2018, 126, 1763. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-L.; Cheng, W.-F.; Chang, M.-C.; Lin, H.-W.; Huang, C.-T.; Chien, C.-L.; Chen, C.-A. Interferon-Gamma in Ascites Could Be a Predictive Biomarker of Outcome in Ovarian Carcinoma. Gynecol. Oncol. 2013, 131, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-L.; Chou, C.-Y.; Chang, M.-C.; Lin, H.-W.; Huang, C.-T.; Hsieh, S.-F.; Chen, C.-A.; Cheng, W.-F. IL17a and IL21 Combined with Surgical Status Predict the Outcome of Ovarian Cancer Patients. Endocr. Relat. Cancer 2015, 22, 703–711. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lai, Y.-L.; Lin, H.-W.; Huang, Y.-W.; Chen, J.; Tai, M.-C.; Wu, C.-Y.; Yang, T.-S.; Oblin, V.; Shea, K.; Chen, Y.-L. Analysis of Immune Checkpoints on Peripheral Blood Mononuclear Cells Can Predict Clinical Outcome and Reveal Potential of HVEM-BTLA Axis in Epithelial Ovarian Cancers. Pharmaceuticals 2025, 18, 1295. https://doi.org/10.3390/ph18091295
Lai Y-L, Lin H-W, Huang Y-W, Chen J, Tai M-C, Wu C-Y, Yang T-S, Oblin V, Shea K, Chen Y-L. Analysis of Immune Checkpoints on Peripheral Blood Mononuclear Cells Can Predict Clinical Outcome and Reveal Potential of HVEM-BTLA Axis in Epithelial Ovarian Cancers. Pharmaceuticals. 2025; 18(9):1295. https://doi.org/10.3390/ph18091295
Chicago/Turabian StyleLai, Yen-Ling, Han-Wei Lin, Yu-Wen Huang, Jung Chen, Ming-Chien Tai, Chia-Ying Wu, Tyan-Shin Yang, Valentina Oblin, Kristin Shea, and Yu-Li Chen. 2025. "Analysis of Immune Checkpoints on Peripheral Blood Mononuclear Cells Can Predict Clinical Outcome and Reveal Potential of HVEM-BTLA Axis in Epithelial Ovarian Cancers" Pharmaceuticals 18, no. 9: 1295. https://doi.org/10.3390/ph18091295
APA StyleLai, Y.-L., Lin, H.-W., Huang, Y.-W., Chen, J., Tai, M.-C., Wu, C.-Y., Yang, T.-S., Oblin, V., Shea, K., & Chen, Y.-L. (2025). Analysis of Immune Checkpoints on Peripheral Blood Mononuclear Cells Can Predict Clinical Outcome and Reveal Potential of HVEM-BTLA Axis in Epithelial Ovarian Cancers. Pharmaceuticals, 18(9), 1295. https://doi.org/10.3390/ph18091295





