Lower Urinary Tract Disorders as Adverse Drug Reactions—A Literature Review
Abstract
1. Introduction
2. Urinary Retention or Urinary Incontinence
3. Urinary Tract Infections
4. Urolithiasis
5. Erectile Dysfunction
6. Retroperitoneal Fibrosis
7. Clinical Examples of Drug-Induced Urological Disorders
8. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADR | adverse drug reaction |
| CC | chemical cystitis |
| CIOMS | Council for International Organizations of Medical Sciences |
| CNS | central nervous system |
| DU | detrusor underactivity |
| ED | erectile dysfunction |
| HC | hemorrhagic cystitis |
| KIC | ketamine-induced cystitis |
| LUTS | lower urinary tract symptoms |
| OAB | overactive bladder |
| PAG | periaqueductal gray |
| PMC | pontine micturition centre |
| RPF | retroperitoneal fibrosis |
| UAB | underactive bladder |
| UI | urinary incontinence |
| UR | urinary retention |
| UTI | urinary tract infection |
| WHO | World Health Organization |
References
- World Health Organization. Geneva 2002. Safety of Medicines. A Guide to Detecting and Reporting Adverse Drug Reactions. Available online: https://www.who.int/publications/i/item/WHO-EDM-QSM-2002-2 (accessed on 29 April 2023).
- U.S. Food and Drug Administration. IND Application Reporting: Safety Reports. Available online: https://www.fda.gov/drugs/investigational-new-drug-ind-application/ind-application-reporting-safety-reports (accessed on 29 April 2023).
- Council for International Organizations of Medical Sciences. Cumulative Glossary with a Focus on Pharmacovigilance. Version 2.0. Geneva, Switzerland. 2022. Available online: https://cioms.ch/publications/product/cioms-cumulative-pharmacovigilance-glossary/ (accessed on 30 April 2023).
- EudraVigilance—European Database of Suspected Adverse Drug Reaction Reports. Available online: https://www.adrreports.eu/en/glossary.html (accessed on 29 April 2023).
- Edwards, I.R.; Aronson, J.K. Adverse drug reactions: Definitions, diagnosis, and management. Lancet 2000, 356, 1255–1259. [Google Scholar] [CrossRef] [PubMed]
- Schatz, S.N.; Weber, R.J. Adverse drug reactions. PSAP CNS/Pharm. Pract. 2015, 5–21. Available online: https://www.accp.com/docs/bookstore/psap/2015B2.SampleChapter.pdf (accessed on 29 April 2023).
- Smith, W. Adverse drug reactions. Aust Fam. Physician 2013, 42, 12–16. [Google Scholar] [PubMed]
- Olawale, O.O.; Kezeli, T.; Nikoleishvili, E.; Sulashvili, N. Adverse drug reaction. Cauc. J. Health Sci. Public Health 2018, 2 (Suppl. S3), 1–4. [Google Scholar]
- Kumar, R.; Singh, S.; Arora, S.; Bhati, S. Adverse drug reactions: A comprehensive review. J. Drug Deliv. Ther. 2018, 8, 103–107. [Google Scholar] [CrossRef]
- Coleman, J.J.; Pontefract, S.K. Adverse drug reactions. Clin. Med. (Lond.) 2016, 16, 481–485. [Google Scholar] [CrossRef] [PubMed]
- Woo, S.D.; Yoon, J.; Doo, G.E.; Park, Y.; Lee, Y.; Lee, S.H.; Lee, Y.H.; Ye, Y.M. Common causes and characteristics of adverse drug reactions in older adults: A retrospective study. BMC Pharmacol. Toxicol. 2020, 21, 87. [Google Scholar] [CrossRef] [PubMed]
- Riedl, M.A.; Casillas, A.M. Adverse drug reactions: Types and treatment options. Am. Fam. Physician 2003, 68, 1781–1791. [Google Scholar]
- Khalil, H.; Huang, C. Adverse drug reactions in primary care: A scoping review. BMC Health Serv. Res. 2020, 20, 5. [Google Scholar] [CrossRef]
- Reporting adverse drug reactions. Definitions of terms and criteria for their use. In Council for International Organizations of Medical Sciences (CIOMS); Geneva, Switzerland. 2000; Available online: https://cioms.ch/publications/product/reporting-adverse-drug-reactions-definitions-of-terms-and-criteria-for-their-use/ (accessed on 30 April 2023).
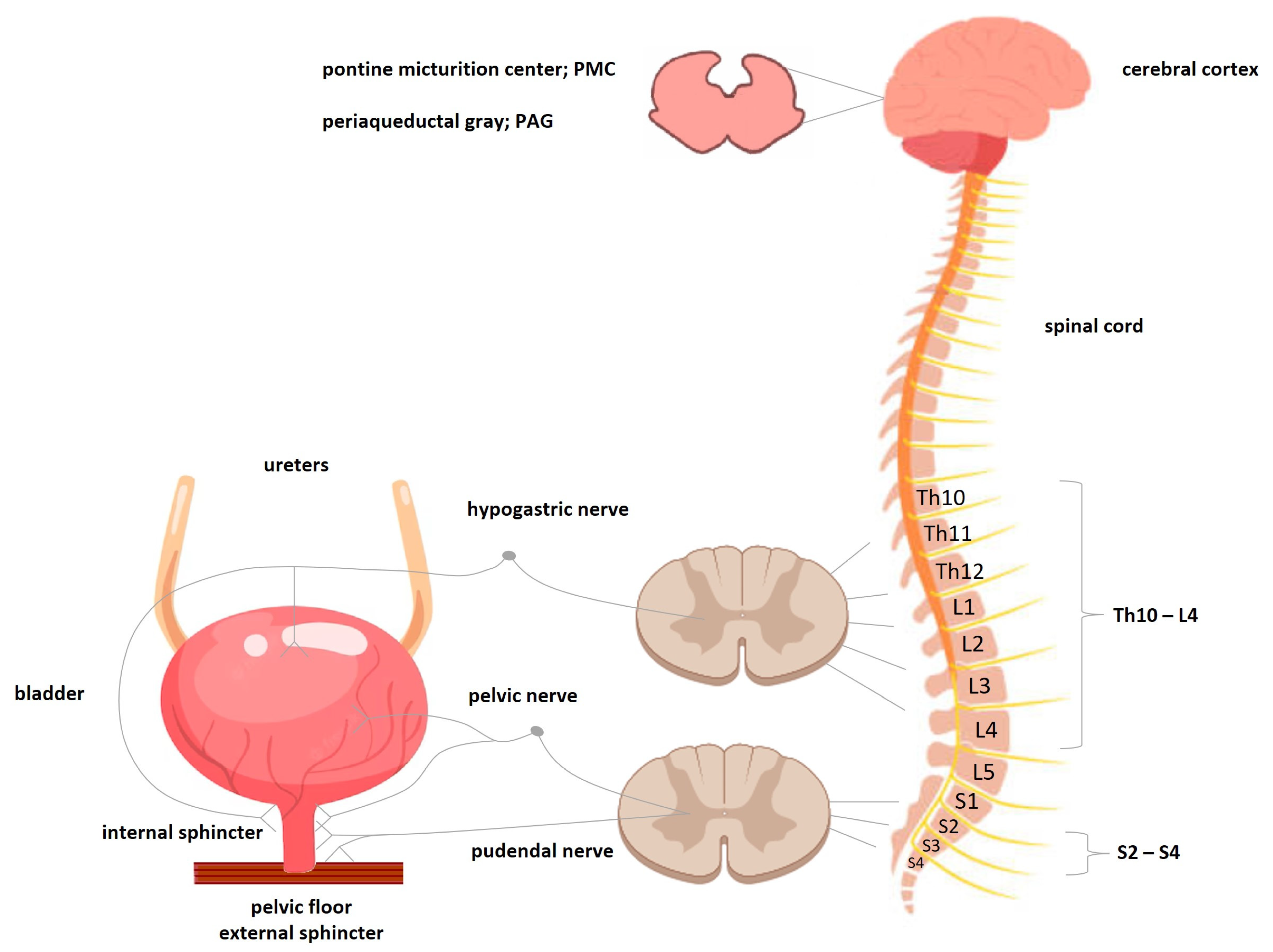
- Beckel, J.M.; Holstege, G. Neurophysiology of the lower urinary tract. Handb. Exp. Pharmacol. 2011, 202, 149–169. [Google Scholar]
- Cortes, G.A.; Flores, J.L. Physiology, Urination. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK562181/ (accessed on 30 April 2023).
- Zare, A.; Jahanshahi, A.; Rahnama’I, M.S.; Schipper, S.; van Koeveringe, G.A. The Role of the periaqueductal gray matter in lower urinary tract function. Mol. Neurobiol. 2019, 56, 920–934. [Google Scholar] [CrossRef] [PubMed]
- Verhamme, K.M.; Sturkenboom, M.C.; Stricker, B.H.; Bosch, R. Drug-induced urinary retention: Incidence, management and prevention. Drug Saf. 2008, 31, 373–388. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.; Rahman, M.; Siddik, A.B. Neuroanatomy, Pontine Micturition Center. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023; Available online: https://www.statpearls.com/ArticleLibrary/viewarticle/27426# (accessed on 30 April 2023).
- Dobrek, Ł.; Juszczak, K.; Wyczółkowski, M.; Thor, P.J. Overactive bladder—Current definition and basic pathophysiology concepts. Adv. Clin. Exp. Med. 2011, 20, 119–129. [Google Scholar]
- Abrams, P. New words for old: Lower urinary tract symptoms for “prostatism”. BMJ 1994, 308, 929–930. [Google Scholar] [CrossRef]
- Chapple, C.R.; Wein, A.J.; Abrams, P.; Dmochowski, R.R.; Giuliano, F.; Kaplan, S.A.; McVary, K.T.; Roehrborn, C.G. Lower urinary tract symptoms revisited: A broader clinical perspective. Eur. Urol. 2008, 54, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Halbgewachs, C.; Domes, T. Postobstructive diuresis. Can. Fam. Physician 2015, 61, 137–142. [Google Scholar]
- Osman, N.; Mangera, A.; Hillary, C.; Inman, R.; Chapple, C. The underactive bladder: Detection and diagnosis. F1000Research 2016, 5, 102. [Google Scholar] [CrossRef]
- Amarenco, G.; Sheikh Ismael, S.; Chesnel, C.; Charlanes, A.; Le Breton, F. Diagnosis and clinical evaluation of neurogenic bladder. Eur. J. Phys. Rehabil. Med. 2017, 53, 975–980. [Google Scholar] [CrossRef]
- Gani, J.; Hennessey, D. The underactive bladder: Diagnosis and surgical treatment options. Transl. Androl. Urol. 2017, 6 (Suppl. S2), S186–S195. [Google Scholar] [CrossRef]
- Drake, M.J.; Nixon, P.M.; Crew, J.P. Drug-induced bladder and urinary disorders. Incidence, prevention and management. Drug Saf. 1998, 19, 45–55. [Google Scholar] [CrossRef]
- Baydal, K.; Anton, C.; Ferner, R. Adverse drug reactions and urological disease. Trends Urol. Men’s Health 2017, 8, 21–25. [Google Scholar] [CrossRef]
- Živković, D.; Franić, D.; Kojović, V. Urinary incontinence—From childhood onwards. Acta Clin. Croat. 2022, 61, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Denisenko, A.A.; Clark, C.B.; D’Amico, M.; Murphy, A.M. Evaluation and management of female urinary incontinence. Can. J. Urol. 2021, 28, 27–32. [Google Scholar] [PubMed]
- Das, A.K.; Kucherov, V.; Glick, L.; Chung, P. Male urinary incontinence after prostate disease treatment. Can. J. Urol. 2020, 27, 36–43. [Google Scholar] [PubMed]
- Tsakiris, P.; Oelke, M.; Michel, M.C. Drug-induced urinary incontinence. Drugs Aging 2008, 25, 541–549. [Google Scholar] [CrossRef]
- Panesar, K. Drug-induced urinary incontinence. US Pharm. 2014, 39, 24–29. [Google Scholar]
- Gandi, C.; Sacco, E. Pharmacological management of urinary incontinence: Current and emerging treatment. Clin. Pharmacol. 2021, 13, 209–223. [Google Scholar] [CrossRef]
- Hu, J.S.; Pierre, E.F. Urinary Incontinence in women: Evaluation and management. Am. Fam. Physician 2019, 100, 339–348. [Google Scholar]
- Bono, M.J.; Leslie, S.W.; Reygaert, W.C. Urinary tract infection. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK470195/ (accessed on 17 March 2023).
- Flores-Mireles, A.L.; Walker, J.N.; Caparon, M.; Hultgren, S.J. Urinary tract infections: Epidemiology, mechanisms of infection and treatment options. Nat. Rev. Microbiol. 2015, 13, 269–284. [Google Scholar] [CrossRef]
- Kamei, J.; Fujimura, T. Urinary tract infection in patients with lower urinary tract dysfunction. J. Infect. Chemother. 2023, 29, 744–748. [Google Scholar] [CrossRef]
- Wagenlehner, F.M.E.; Bjerklund Johansen, T.E.; Cai, T.; Koves, B.; Kranz, J.; Pilatz, A.; Tandogdu, Z. Epidemiology, definition and treatment of complicated urinary tract infections. Nat. Rev. Urol. 2020, 17, 586–600. [Google Scholar] [CrossRef] [PubMed]
- Mancuso, G.; Midiri, A.; Gerace, E.; Marra, M.; Zummo, S.; Biondo, C. Urinary tract infections: The current scenario and future prospects. Pathogens 2023, 12, 623. [Google Scholar] [CrossRef] [PubMed]
- Dobrek, Ł. Drug-related urinary tract infections. Wiad. Lek. 2021, 74, 1728–1736. [Google Scholar] [CrossRef] [PubMed]
- Petca, R.C.; Popescu, R.I.; Toma, C.; Dumitrascu, M.C.; Petca, A.; Sandru, F.; Chibelean, C.B. Chemical hemorrhagic cystitis: Diagnostic and therapeutic pitfalls (Review). Exp. Ther. Med. 2021, 21, 624. [Google Scholar] [CrossRef]
- Matz, E.L.; Hsieh, M.H. Review of advances in uroprotective agents for cyclophosphamide- and ifosfamide-induced hemorrhagic cystitis. Urology 2017, 100, 16–19. [Google Scholar] [CrossRef]
- Sherif, I. Uroprotective mechanisms of natural products against cyclophosphamide-induced urinary bladder toxicity: A comprehensive review. Acta Sci. Pol. Technol. Aliment. 2020, 19, 333–346. [Google Scholar] [CrossRef]
- Payne, H.; Adamson, A.; Bahl, A.; Borwell, J.; Dodds, D.; Heath, C.; Huddart, R.; McMenemin, R.; Patel, P.; Peters, J.L.; et al. Chemical- and radiation-induced haemorrhagic cystitis: Current treatments and challenges. BJU Int. 2013, 112, 885–897. [Google Scholar] [CrossRef]
- Dobrek, Ł.; Thor, P.J. Bladder urotoxicity pathophysiology induced by the oxazaphosphorine alkylating agents and its chemoprevention. Postep. Hig. Med. Dosw. (Online) 2012, 66, 592–602. [Google Scholar] [CrossRef]
- Zhou, J.; Scott, C.; Miab, Z.R.; Lehmann, C. Current approaches for the treatment of ketamine-induced cystitis. Neurourol. Urodyn. 2023, 42, 680–689. [Google Scholar] [CrossRef]
- Chen, C.L.; Wu, S.T.; Cha, T.L.; Sun, G.H.; Meng, E. Molecular pathophysiology and potential therapeutic strategies of ketamine-related cystitis. Biology (Basel) 2022, 11, 502. [Google Scholar] [CrossRef]
- Jhang, J.F.; Hsu, Y.H.; Kuo, H.C. Possible pathophysiology of ketamine-related cystitis and associated treatment strategies. Int. J. Urol. 2015, 22, 816–825. [Google Scholar] [CrossRef] [PubMed]
- Ramello, A.; Vitale, C.; Marangella, M. Epidemiology of nephrolithiasis. J. Nephrol. 2000, 13 (Suppl. S3), S45–S50. [Google Scholar] [PubMed]
- Jayaraman, U.C.; Gurusamy, A. Review on uro-lithiasis pathophysiology and aesculapian discussion. IOSR J. Pharm. 2018, 8, 30–42. [Google Scholar]
- Dobrek, Ł. Kidney stone disease with special regard to drug-induced kidney stones—A contemporary synopsis. Wiad. Lek. 2020, 73, 2031–2039. [Google Scholar] [CrossRef] [PubMed]
- Bishop, K.; Momah, T.; Ricks, J. Nephrolithiasis. Prim. Care 2020, 47, 661–671. [Google Scholar] [CrossRef]
- Viljoen, A.; Chaudhry, R.; Bycroft, J. Renal stones. Ann. Clin. Biochem. 2019, 56, 15–27. [Google Scholar] [CrossRef]
- Alelign, T.; Petros, B. Kidney stone disease: An update on current concepts. Adv. Urol. 2018, 2018, 3068365. [Google Scholar] [CrossRef]
- Begum, S.; Madani, M.; Hassan, M. Nephrolithiasis: An update on current concepts. IAJPS 2019, 6, 14073–14087. [Google Scholar]
- European Associaton of Urology Guidelines. Urolithiasis. Available online: https://uroweb.org/guidelines/urolithiasis (accessed on 17 March 2023).
- Osborne, C.A.; Lulich, L.P.; Swanson, L.L.; Albasan, H. Drug-induced urolithiasis. Vet. Clin. Small Anim. 2008, 39, 55–63. [Google Scholar] [CrossRef]
- Sighinolfi, M.C.; Eissa, A.; Bevilacqua, L.; Zoeir, A.; Ciarlariello, S.; Morini, E.; Puliatti, S.; Durante, V.; Ceccarelli, P.L.; Micali, S.; et al. Drug-induced urolithiasis in pediatric patients. Paediatr. Drugs 2019, 21, 323–344. [Google Scholar] [CrossRef]
- Dobrek, L. A synopsis of current theories on drug-induced nephrotoxicity. Life 2023, 13, 325. [Google Scholar] [CrossRef] [PubMed]
- Matlaga, B.R.; Shah, O.D.; Assimos, D.G. Drug-induced urinary calculi. Rev. Urol. 2003, 5, 227–231. [Google Scholar] [PubMed]
- Daudon, M.; Frochot, V.; Bazin, D.; Jungers, P. Drug-induced kidney stones and crystalline nephropathy: Pathophysiology, prevention and treatment. Drugs 2018, 78, 163–201. [Google Scholar] [CrossRef] [PubMed]
- Daudon, M.; Jungers, P. Drug-induced renal calculi: Epidemiology, prevention and management. Drugs 2004, 64, 245–275. [Google Scholar] [CrossRef] [PubMed]
- Gordijn, R.; Teichert, M.; Nicolai, M.P.J.; Elzevier, H.W.; Guchelaar, H.J. Adverse drug reactions on sexual functioning: A systematic overview. Drug Discov. Today 2019, 24, 890–897. [Google Scholar] [CrossRef]
- Conaglen, H.M.; Conaglen, J.V. Drug-induced sexual dysfunction in men and woman. Aust. Prescr. 2013, 36, 42–46. [Google Scholar] [CrossRef]
- Razdan, S.; Greer, A.B.; Patel, A.; Alameddine, M.; Jue, J.S.; Ramasamy, R. Effect of prescription medications on erectile dysfunction. Postgrad. Med. J. 2018, 94, 171–178. [Google Scholar] [CrossRef]
- Fecik, S.E. Drug-induced sexual dysfunction. Med. Update Psychiatr. 1998, 3, 176–181. [Google Scholar] [CrossRef]
- Engelsgjerd, J.S.; LaGrange, C.A. Retroperitoneal Fibrosis. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK482409/ (accessed on 18 March 2023).
- Tanaka, T.; Masumori, N. Current approach to diagnosis and management of retroperitoneal fibrosis. Int. J. Urol. 2020, 27, 387–394. [Google Scholar] [CrossRef]
- Runowska, M.; Majewski, D.; Puszczewicz, M. Retroperitoneal fibrosis—The state-of-the-art. Reumatologia 2016, 54, 256–263. [Google Scholar] [CrossRef]
- Alberti, C. Revisiting the retroperitoneal fibroses: Are there any significant news about their aetiopathogenesis and diagnostic approaches? Eur. Rev. Med. Pharmacol. Sci. 2012, 16, 1078–1094. [Google Scholar] [PubMed]
- Alberti, C. Drug-induced retroperitoneal fibrosis: Short aetiopathogenetic note, from the past times of ergot-derivatives large use to currently applied bio-pharmacology. G Chir. 2015, 36, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Brasselet, D.; Chouchana, L.; Vial, T.; Damin-Pernik, M.; Lebrun-Vignes, B. Drug-induced retroperitoneal fibrosis: A case/non-case study in the French PharmacoVigilance Database. Expert Opin. Drug Saf. 2020, 19, 903–914. [Google Scholar] [CrossRef]
- Trinchieri, M.; Perletti, G.; Magri, V.; Stamatiou, K.; Montanari, E.; Trinchieri, A. Urinary side effects of psychotropic drugs: A systematic review and metanalysis. Neurourol. Urodyn. 2021, 40, 1333–1348. [Google Scholar] [CrossRef]
- Winkler, D.; Grohmann, R.; Friedrich, M.E.; Toto, S.; Bleich, S.; Seifert, J.; Konstantinidis, A.; Shariat, S.F.; Kasper, S.; Pjrek, E. Urological adverse drug reactions of psychotropic medication in psychiatric inpatients—A drug surveillance report from German-speaking countries. J. Psychiatr. Res. 2021, 144, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Burke, H.; Hernandez, M.; Ruth, L. Sertraline-induced urinary retention reversed by mirtazapine in an adolescent. Prim. Care Companion CNS Disord. 2022, 24, 22cr03254. [Google Scholar] [CrossRef] [PubMed]
- Ozdemir, P.G.; Aydin, A.; Gulec, M.; Cim, E.F.A. Switching to fluoxetine in a case of sertraline-induced urinary incontinence: A case report. Düşünen Adam J. Psychiatry Neurol. Sci. 2023, 26, 117–118. [Google Scholar] [CrossRef]
- Pease, E.; Singh, B.; Ziegelmann, M.; Betcher, H. Dose-dependent urinary incontinence with sertraline use in obsessive-compulsive disorder. Psychopharmacol. Bull. 2021, 51, 117–121. [Google Scholar]
- Howard, R. Amitryptyline in BPH: Increased risk of PVR and urinary retention. Prescriber 2008, 19, 42–45. [Google Scholar]
- Stamatiou, K.; Alevizos, A.; Petrakos, G.; Lentzas, I.; Mariolis, A.; Panagopoulos, P. Tricyclic antidepressant-induced acute urine retention in a young woman with postpartum depression--case report. Clin. Exp. Obstet. Gynecol. 2007, 34, 60. [Google Scholar]
- Mirzakhani, H.; Rahim, M.; Mathew, J., Jr. Expanding our understanding of atypical antipsychotics: Acute urinary retention secondary to olanzapine. Case Rep. Psychiatry 2020, 2020, 6157548. [Google Scholar] [CrossRef] [PubMed]
- Purohith, A.N.; Ramesh, P.; Vaidya, B.P.; Shenoy, S.; Sharma, P.S.V.N. Olanzapine-associated chronic urinary retention and ciliochoroidal effusion: Rare adverse effects of a commonly prescribed antipsychotic. Indian J. Psychol. Med. 2023, 45, 96–97. [Google Scholar] [CrossRef] [PubMed]
- Panicker, J.N.; Game, X.; Khan, S.; Kessler, T.M.; Gonzales, G.; Elneil, S.; Fowler, C.J. The possible role of opiates in women with chronic urinary retention: Observations from a prospective clinical study. J. Urol. 2012, 188, 480–484. [Google Scholar] [CrossRef] [PubMed]
- Edwards, R.T.; McCormick-Deaton, C.; Hosanagar, A. Acute urinary retention secondary to buprenorphine administration. Am. J. Emerg. Med. 2014, 32, 109.e1–109.e2. [Google Scholar] [CrossRef]
- Landi, F.; Cesari, M.; Russo, A.; Onder, G.; Sgadari, A.; Bernabei, R.; Silvernet-HC Study Group. Benzodiazepines and the risk of urinary incontinence in frail older persons living in the community. Clin. Pharmacol. Ther. 2002, 72, 729–734. [Google Scholar] [CrossRef]
- Aykut, D.S.; Uysal, R.A.E. Acute urinary retention after alprazolam use: A case report. Psychiatry Clin. Psychopharmacol. 2018, 28, 220–221. [Google Scholar] [CrossRef]
- Hall, S.A.; Yang, M.; Gates, M.A.; Steers, W.D.; Tennstedt, S.L.; McKinlay, J.B. Associations of commonly used medications with urinary incontinence in a community based sample. J. Urol. 2012, 188, 183–189. [Google Scholar] [CrossRef]
- Elhebir, E.S.; Hughes, J.D.; Hilmi, S.C. Calcium antagonists use and its association with lower urinary tract symptoms: A cross-sectional study. PLoS ONE 2013, 8, e66708. [Google Scholar] [CrossRef]
- Salman, M.; Khan, A.H.; Syed Sulaiman, S.A.; Khan, J.H.; Hussain, K.; Shehzadi, N. Effect of calcium channel blockers on lower urinary tract symptoms: A systematic review. Biomed. Res. Int. 2017, 2017, 4269875. [Google Scholar] [CrossRef]
- Uitrakul, S.; Aksonnam, K.; Srivichai, P.; Wicheannarat, S.; Incomenoy, S. The incidence and risk factors of urinary tract infection in patients with type 2 diabetes mellitus using SGLT2 inhibitors: A real-world observational study. Medicines 2022, 9, 59. [Google Scholar] [CrossRef]
- Figueiredo, I.R.; Rose, S.C.P.; Freire, N.B.; Patrocínio, M.S.; Pierdoná, N.; Bittencourt, R.J. Use of sodium-glucose cotransporter-2 inhibitors and urinary tract infections in type 2 diabetes patients: A systematic review. Rev. Assoc. Med. Bras. 2019, 65, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Hashmi, M.S.; Rana, M.A.; Zafar, G.M.; Asif, S.; Farooq, M.T.; Zahoor, S. Frequency of urinary tract infections in type 2 diabetic patients taking dapagliflozin. Cureus 2022, 14, e21720. [Google Scholar] [CrossRef] [PubMed]
- Tasian, G.E.; Jemielita, T.; Goldfarb, D.S.; Copelovitch, L.; Gerber, J.S.; Wu, Q.; Denburg, M.R. Oral antibiotic exposure and kidney stone disease. J. Am. Soc. Nephrol. 2018, 29, 1731–1740. [Google Scholar] [CrossRef] [PubMed]
- Samajdar, S.S.; Mukherjee, S.; Saha, D.; Jumani, D.; Tripathi, S.K. Drug-induced erectile dysfunction: Two interesting cases. J. Pharmacol. Pharmacother. 2021, 12, 177–179. [Google Scholar]
- Cichon, G.J.; Qadri, S.F. Risperidone-induced sexual dysfunction case report. Cureus 2023, 15, e35357. [Google Scholar] [CrossRef]
- Maehara, M.; Sugiyama, M. A community pharmacist’s intervention in antipsychotic drug-induced sexual dysfunction in a patient with schizophrenia. Clin. Case Rep. 2021, 9, 2074–2076. [Google Scholar] [CrossRef]
- Waters, V.V. Hydralazine, hydrochlorothiazide and ampicillin associated with retroperitoneal fibrosis: Case report. J. Urol. 1989, 141, 936–937. [Google Scholar] [CrossRef]
- Bilici, A.; Karadag, B.; Doventas, A.; Erdincler, D.S.; Cetinkaya, S.; Ogut, G.; Tezcan, V.; Beger, T. Retroperitoneal fibrosis caused by pergolide in a patient with Parkinson’s disease. Neth. J. Med. 2004, 62, 389–392. [Google Scholar]
- Graham, J.R.; Suby, H.I.; LeCompte, P.R.; Sadowsky, N.L. Fibrotic disorders associated with methysergide therapy for headache. N. Engl. J. Med. 1966, 274, 359–368. [Google Scholar] [CrossRef]
- Fujiu, K.; Utsumi, Y.; Omi, F.; Suzuki, M.; Suzuki, H. Two cases of retroperitoneal fibrosis during anti-programmed cell death 1 antibody treatment. JJLC 2022, 62, 1044–1047. [Google Scholar] [CrossRef]
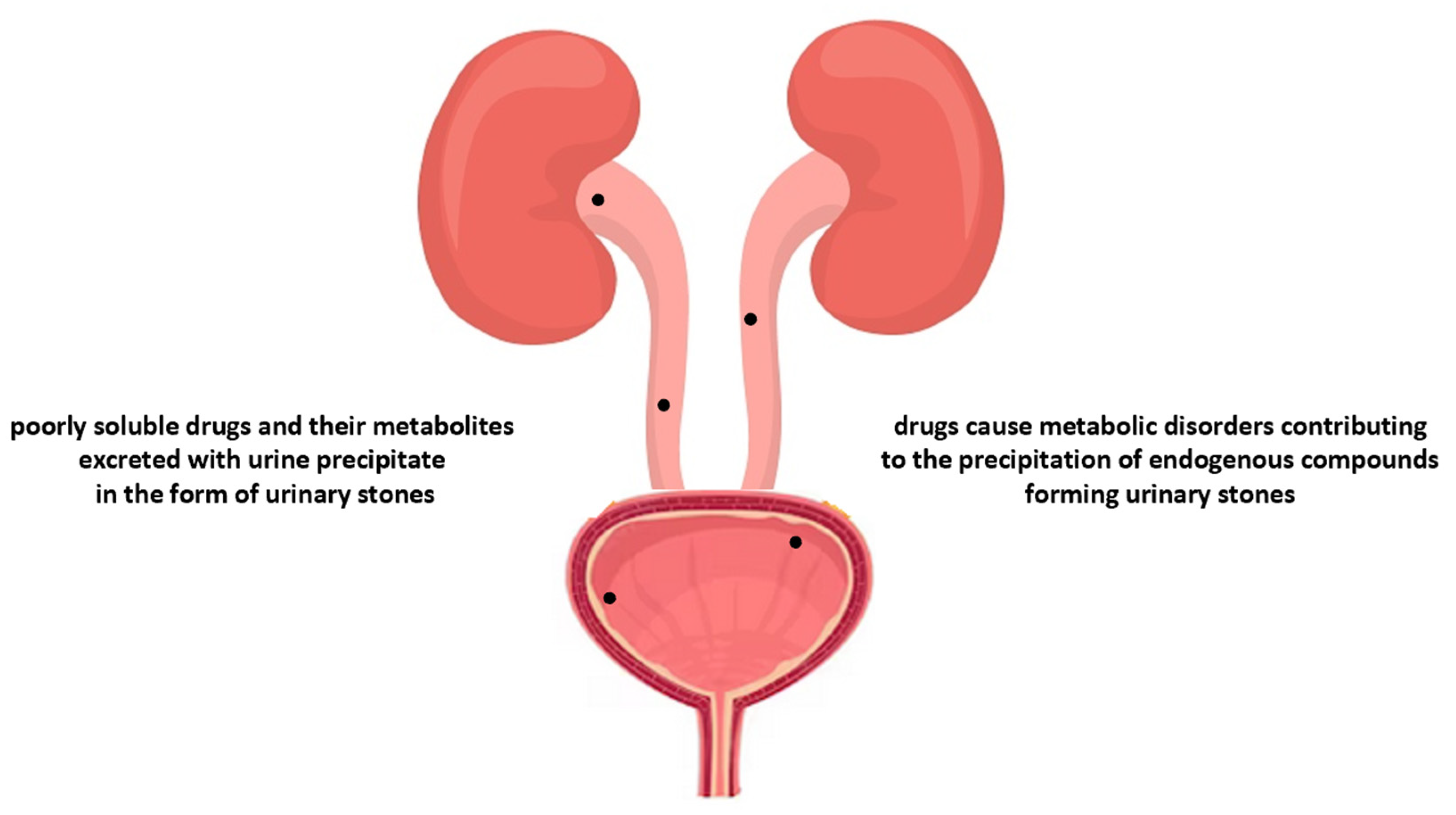
| Drug Class | Examples | Postulated Mechanism Contributing to Urinary Retention | |
|---|---|---|---|
| Drugs with anticholinergic effects–muscarinic receptor antagonists (MRA) | Tricyclic antidepressants | Amitriptyline, Imipramine, Clomipramine, Nortriptyline | Drugs with antimuscarinic effect can cause or exacerbate urinary retention due to the failure of cholinergic-mediated bladder contraction (especially in patients with a pre-existing bladder outlet obstruction, e.g., benign prostate hyperplasia). |
| Classic antipsychotics | Thioridazine, Chlorpromazine, Chlorprothixene, Risperidone, Clozapine | ||
| H1-antihistamines (1st generation) | Chlorpheniramine, Triprolidine Cyproheptadine, Clemastine, Dimenhydrinate, Diphenhydramine, Doxylamine | ||
| Antispasmodics | Dicyclomine, Propantheline, Oxybutynin | ||
| Antiparkinsonian agents | Benzatropine, Orphenadrine, Procyclidine | ||
| Antiarrhythmic drugs class I | Disopyramide, Flecainide | ||
| Antimuscarinic bronchodilators | Ipratropium, Tiotropium, Glycopyrronium | ||
| Anticholinergic drugs used to treat overactive bladder | Oxybutynin, Fesoterodine, Solifenacin, Trospium, Tolterodine | ||
| Premedication drugs (cholinolytics) | Atropine, Hyoscine | ||
| Anaesthetics | Bupivacaine, Propofol, Ketamine, General anaesthetics of duration greater than 60 min | Inhibitory effects on the contraction of the bladder (inhibition of micturition reflex). | |
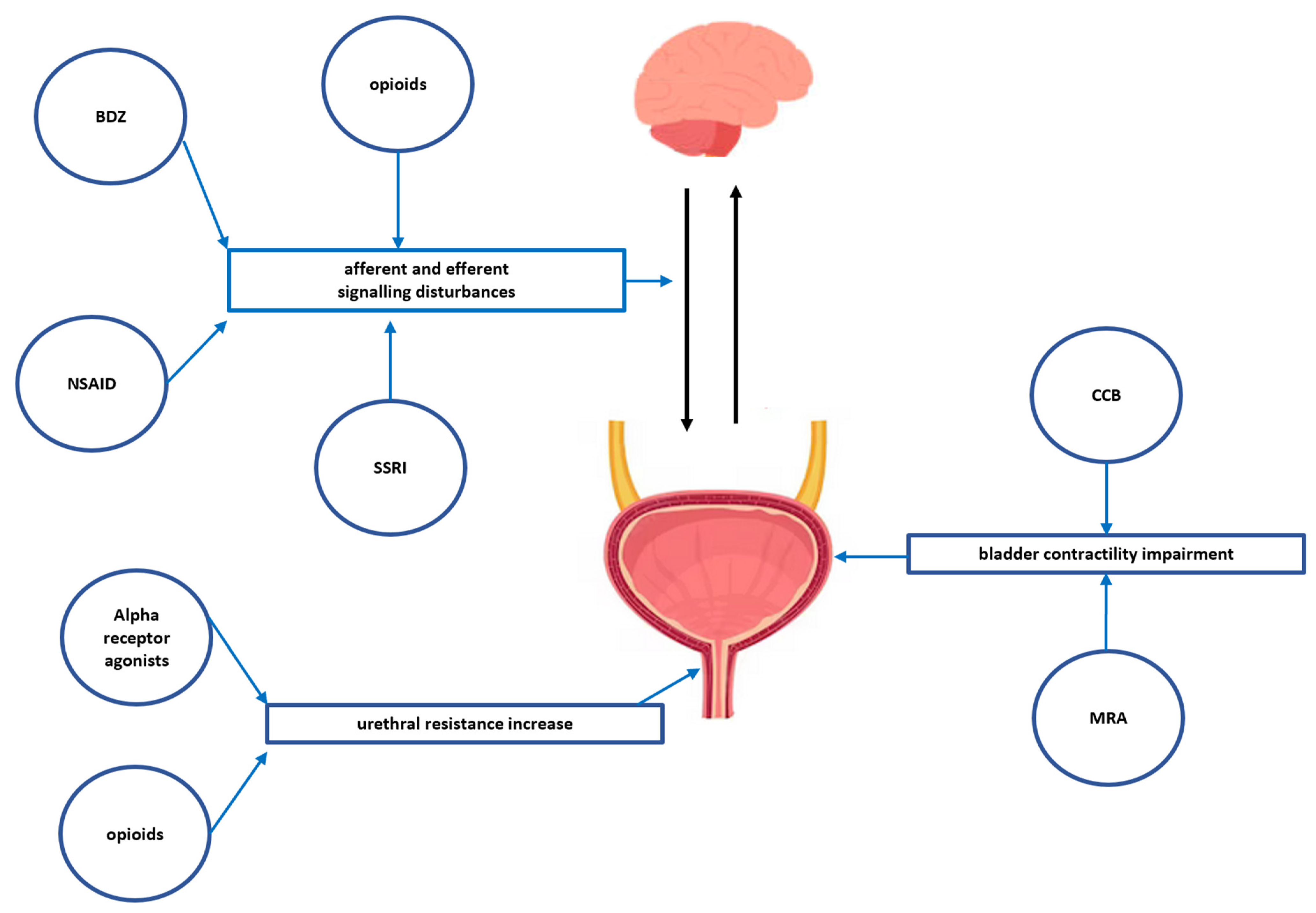
| Opioids | Morphine, Pethidine, Pentazocine, Fentanyl | Urinary retention occurs due to the mu-receptor-mediated blockade of the sensory input from the bladder and the volume-evoked micturition reflex that results in decreasing the usual sensation of bladder fullness and bladder overdistention, and partially the anticholinergic effect of opioids. In addition, opioids can increase bladder sphincter tone due to excessive sympathetic stimulation, resulting in a bladder outlet restraint. | |
| Non-steroidal anti-inflammatory drugs | Diclofenac, Ketoprofen | Impairment of the direct prostaglandin-induced bladder contractility and by the impairment of tachykinins stimulating neurokinin receptors on afferent nerves and detrusor smooth muscle. | |
| Benzodiazepines | Diazepam, Clonazepam | Probably caused by detrusor muscle relaxation | |
| Selective serotonin reuptake inhibitors (SSRI) | Fluoxetine, Sertraline, Citalopram | Serotonin is an important neurotransmitter which facilitates storage by increasing sympathetic activity and inhibiting parasympathetic control over voiding; thus, SSRI may cause an increase in the post-voiding residual volume (the volume of urine left in the bladder at the end of micturition). | |
| Calcium channel blockers (CCB) | NDHP blockers: Diltiazem and Verapamil DHP blockers: Amlodipine and Nifedipine (the highly vascular selective DHP Felodipine and Lercanidipine seem to have less effect on micturition | Reduce bladder smooth muscle contractility via the inhibition of the calcium influx. | |
| Alpha-1-agonists | Methoxamine, Phenylephrine, Ethylephrine | Binding to the alpha-1A/D adrenergic receptors of the internal urethral sphincter resulting in a higher tone of the internal sphincter. | |
| Non-selective alpha-agonists | Pseudoephedrine (e.g., found in many over-the-counter cold remedies) | ||
| Drug Class | Examples | Postulated Mechanism Contributing to Urinary Incontinence |
|---|---|---|
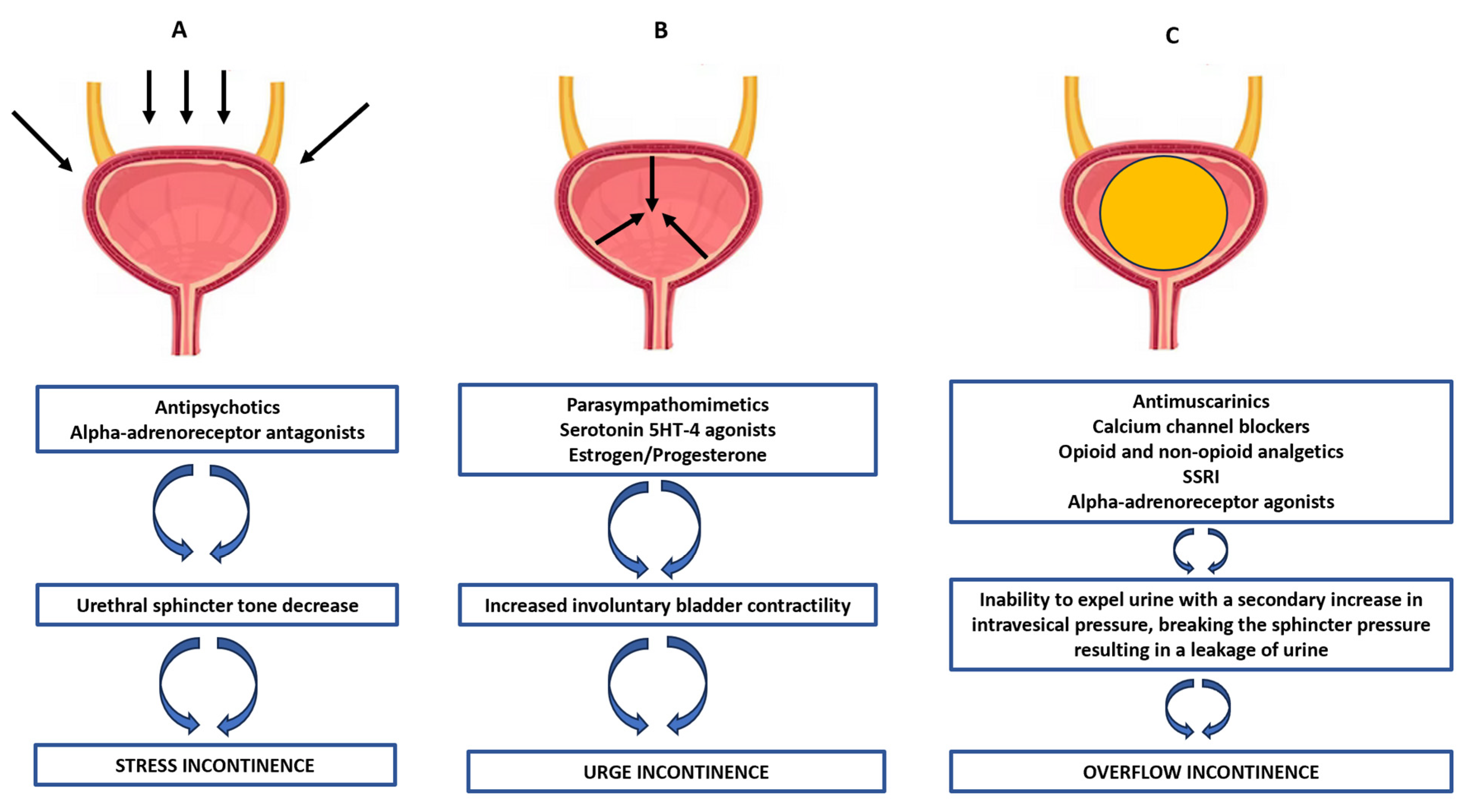
| Antipsychotics | Chlorpromazine, Thioridazine, Chlorprothixene, Trifluoperazine, Fluphenazine, Haloperidol | Stress urinary incontinence: These drugs decrease urethral sphincter tone (e.g., via the inhibition of alpha-adrenoreceptors or as a result of a complex action on the central and peripheral serotonin or muscarinic or GABA receptors); thus, situations related to increased intra-abdominal pressure may result in a breakdown of the sphincter pressure and the possibility of urine leakage. |
| Alpha-adrenoreceptor antagonists | Prazosin, Doxazosin, Terazosin, Phenoxybenzamine, Phentolamine | |
| Benzodiazepines and non-benzodiazepine sedatives | Diazepam, Zopiclone, Zolpidem | |
| Other drugs | Reserpine, Misoprostol, Urapidil | |
| Parasympathomimetic (direct and indirect) | Acetylcholine esterase inhibitors | Urge urinary incontinence: These drugs may increase involuntary detrusor contractility during the storage phase of the bladder, resulting from the stimulation of muscarinic receptors or as a result of a complex action on other central and peripheral receptors (e.g., serotonin ones) or estrogen/progesterone receptors located in the urethra, vesical triangle, sacrouterine ligament, levator ani muscles and pubocervical fascia. |
| Antidepressants | Amitriptyline, Nortriptyline, Desimipramine | |
| Serotonin 5-HT4 receptor agonists | Cisapride, Tegaserod, Prucalopride | |
| Hormone replacement therapy | Oestrogen/Progesterone preparations | |
| Drugs listed in Table 1 (e.g., antimuscarinics, antihistamines, antiparkinsonian agents, beta-adrenergic agonists, calcium channels blockers, opioids, sedatives) | Overflow urinary incontinence: These drugs contribute to an inability to expel urine and consequently, a secondary increase in intravesical pressure, breaking the sphincter pressure and causing a leakage of urine. Angiotensin receptor blockade also decreases both detrusor contractility and urethral sphincter tone, leading to increased stress urinary incontinence, which may be worsened by a cough related to the use of these drugs. | |
| Angiotensin-converting enzyme inhibitors/Angiotensin receptor antagonists | Benazepril, Captopril, Enalapril, Perindopril, Quinapril | |
| Miscellaneous | Alcohol, Caffeine, Diuretics | These compounds contribute to excessive urine production by inhibiting the reabsorption of electrolytes and water from primary urine or by enhancing glomerular filtration. Thus, they cause the bladder to fill faster and increase the frequency of urination. |
| Drug Class | Examples | Postulated Mechanism Contributing to Urinary Tract Inections |
|---|---|---|
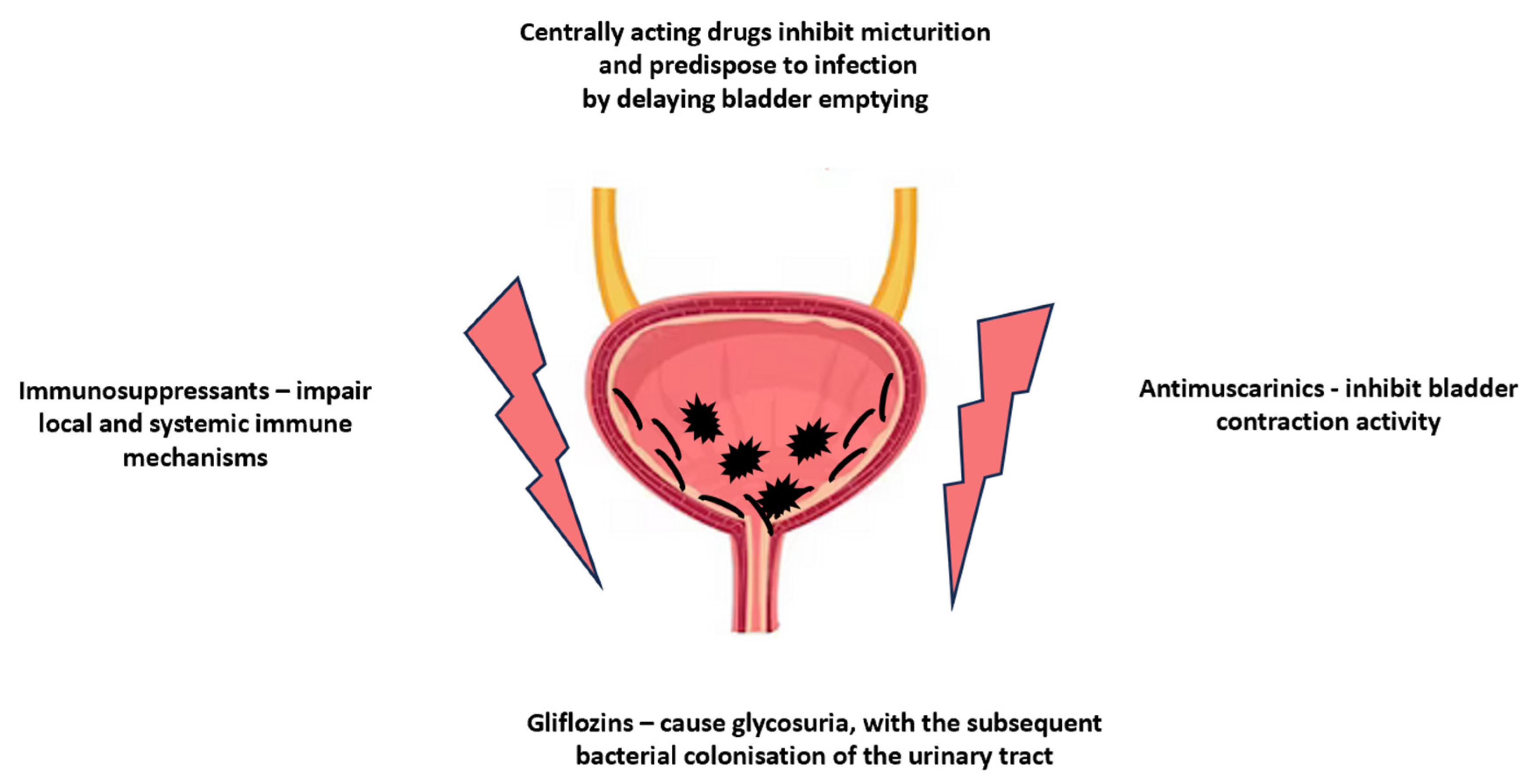
| Gliflozins–inhibitors of kidney sodium-glucose transport proteins 2 (SGLT2) | Canagliflozin, Dapagliflozin, Empagliflozin | Gliflozins cause glycosuria, with subsequent bacterial colonisation of the urinary tract and genital area. |
| Immunosuppressive agents | Glucocorticoids, Azathioprine, Cyclosporin A, Sirolimus–rapamycin, Mycophenolate mofetil | These drugs cause the impairment of local and systemic immune mechanisms, thereby facilitating bacterial colonisation. |
| Drugs with anticholinergic effects listed in Table 1 (e.g., antimuscarinics, antihistamines, antiparkinsonian agents, beta-adrenergic agonists, calcium channels blockers, opioids, sedatives) | These drugs affect the micturition by inhibiting the contractile activity of the bladder, dependent on the activation of muscarinic receptors, resulting in urinary retention in the bladder, which facilitates the bacterial colonisation of the bladder and the reduced evacuation of bacteria along with the urine stream. | |
| Benzodiazepines, Calcium channels antagonists, Opioids, and Non-steroidal anti-inflammatory drugs (NSAIDs) are listed in Table 2 | These drugs may impair micturition through detrusor muscle relaxation and contribute to voiding reflex disturbances, either on a spinal or supraspinal level, resulting in reducing and delaying the emptying of the bladder, which provides better conditions for bacterial colonisation. | |
| Miscellaneous | Antibacterial agents (Sulphonamides, Ciprofloxacin, Ampicillin), Antacids, Calcium, Vitamin D and/or C, Laxatives, Loop diuretics (Furosemide), Anhydrase inhibitors (Acetazolamide), Xanthine oxidase inhibitors (Allopurinol) | These drugs or their metabolites may precipitate in the urinary tract and/or these drugs change the pH of the urine, contributing to the crystallisation of endogenous lithogenic substances in the urinary tract, thereby enabling the formation of urinary stones. |
| Drug Class | Examples | Postulated Mechanism Contributing to Kidney Stone Development |
|---|---|---|
| Drug-containing stones | ||
| Sulfonamides | Sulfadiazine, Sulfaguanidine, Sulfamethoxazole, Sulfasalazine | These drugs and their metabolites are poorly soluble in urine; in the case of long-term treatment they may precipitate in urine. |
| Aminopenicillins Cephalosporins | Ampicillin, Amoxicillin, Ceftriaxone | |
| Quinolones | Pipemidic acid, Ciprofloxacin, Norfloxacin | |
| Other antibacterial drugs | Nitrofurantoin | |
| Protease inhibitors | Indinavir, Nelfinavir, Atazanavir | |
| Antacids aluminum hydroxide | Magnesium trisilicate, Aluminum hydroxide | |
| Miscellaneous | Triamterene, Allopurinol, Ephedrine, Acyclovir, Methotrexate | |
| Drug-induced “metabolic stones” | ||
| Calcium-containing supplements | Many preparations are commercially available, also as dietary supplements | These drugs enhance intestinal calcium absorption, leading to hypercalcemia and the hypercalciuric state. |
| Vitamin D-containing supplements | ||
| Loop diuretics | Furosemide | |
| Anhydrase inhibitors | Acetazolamide, Zonisamide, Topiramate | These drugs inhibit bicarbonate reabsorption and hydrogen ion excretion in proximal tubules, leading to systemic metabolic acidosis, an increase in urinary pH and a decrease in urinary citrates. |
| Laxative drugs (when abused) | Hyperosmotic or stimulant agents | These drugs, when abused, cause increased gastrointestinal fluid and potassium loss and low urinary output. The potassium depletion contributes to intracellular acidosis, compensated by ammonia genesis enhancement in kidney proximal tubules and increased citrate reabsorption, potentiated by increased expression of the H+/K+ activity in the distal tubules. |
| Corticosteroids | Cortisol | These drugs promote the release of calcium from the bones and lead to the hypercalciuria and hyperphosphaturia state. |
| Ascorbic acid (vitamin C) | Many preparations are commercially available, also as dietary supplements | The excess of vitamin C is metabolised to form oxalates and it increases the urinary oxalates excretion. Moreover, high doses of vitamin C cause acidification of the urine. |
| Xanthine oxidase inhibitors | Allopurinol | The drug inhibits the biotransformation of hypoxanthine into xanthine and the final synthesis of uric acid, leading to the formation of xanthine-containing purine stones. |
| Uricosuric drugs | Benzbromarone, Probenecid | These drugs reduce hyperuricemia by enhancing urinary uric acid excretion, leading to the formation of uric acid-containing purine stones. |
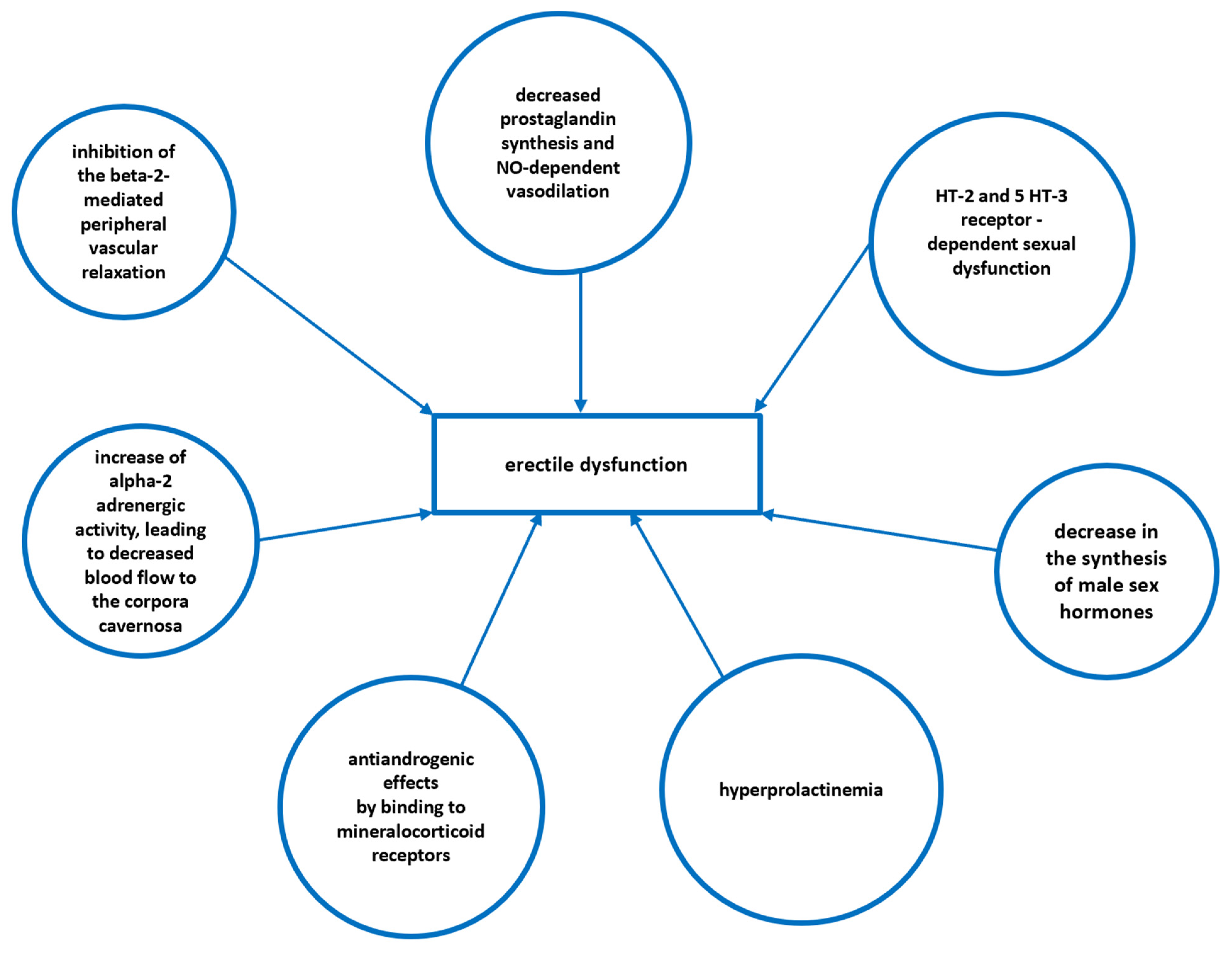
| Drug Class | Examples | Postulated Mechanism Contributing to Erectile Dysfunction |
|---|---|---|
| Beta-adrenergic antagonists | Non-selective (Propranolol, Sotalol, Oxyprenolol) | Beta-blockers suppress the sympathetic outflow and inhibit the beta-2-mediated peripheral relaxation, indirectly predisposing patients to the stimulation of alpha-adrenergic receptors that promote vasoconstriction. Non-selective beta-antagonists have a higher potential to cause ED resulting from the vasoconstriction within the corpora cavernosa. |
| Beta1-selective agents (Metoprolol, Atenolol) | ||
| Thiazide diuretics | Chlorthalidone Hydrochlorothiazide | Thiazides can cause sexual dysfunction through direct effects on vascular smooth muscles or by interfering with catecholamine responsiveness. Moreover, the thiazide-induced sodium loss and lower blood pressure may contribute to increased alpha-2 adrenergic activity, leading to decreased blood flow to the corpora cavernosa. |
| Potassium-sparing diuretics (aldosterone inhibitors) | Spironolactone, Eplerenone | These agents exhibit strong antiandrogenic effects by binding to mineralocorticoid receptors, causing gynecomastia, mastodynia and ED. |
| Statins | Simvastatin | The statin-induced decrease in cholesterol synthesis may negatively impact the male steroidal hormone level (androgens–testosterone). Moreover, statins inhibit not only HMG-CoA, but also 17-ketosteroid-oxidoreductase that catalyses the synthesis of androstenedione and testosterone. Thus, a decrease in serum testosterone level is observed in males with hypercholesterolemia treated with statins. |
| Tricyclic antidepressants | Imipramine, Clomipramine | The increased availability of serotonin due to the inhibition of its reuptake evoked by SSRI/SNRI leads to the increased binding of this neurotransmitter to the 5-HT2 and 5-HT3 receptors which disturbs sexual functions. Moreover, the complex, multi-receptor effect of tricyclic antidepressants also contributes to peripheral circulatory disturbances. In general, antidepressants with lower affinity to dopamine and noradrenaline receptors have a lower influence on ED. |
| Selective serotonin or noradrenalin reuptake inhibitors (SSRI/SNRI) | Sertraline, Citalopram, Paroxetine, Escitalopram, Duloxetine, Venlafaxine | |
| First generation antipsychotics | Haloperidol, Clozapine, Thioridazine | First-generation antipsychotics block central D2 receptors, decreasing the inhibition of prolactin production in the anterior pituitary gland, resulting in secondary disturbances in testosterone synthesis. Moreover, antipsychotics may also impair peripheral vasodilatation via cholinergic antagonism |
| Antiepileptics | Phenytoin, Phenobarbital, Carbamazepine | Both antiepileptic drugs and seizures interfere with the hypothalamic-pituitary-gonadal axis and alter sex steroid hormone production. Moreover, some antiepileptics induce hepatic enzymes resulting in increases of sex hormone binding globulin, which leads to lower free testosterone level. |
| Non-steroidal anti-inflammatory drugs | Naproxen, Diclofenac, Ibuprofen | Cyclooxygenase inhibition decreases the production of prostanoids (prostaglandins and thromboxanes) with a subsequent decrease of vasodilatory, and the erectile effect of nitric oxide (NO). |
| Muscle relaxants | Baclofen | The inhibition of afferent stimulation from the penis, somatic motor efferents mediating perineal muscle contraction and visceral motor efferents controlling the penile vasculature. |
| H2 receptor antagonists | Cimetidine, Ranitidine, Famotidine | H2 receptors are strongly associated with the relaxation of the corpus cavernosum; thus, H2 receptor antagonism results in decreased relaxation, increased contraction and a decreased potential for erection. Moreover, cimetidine exhibits anti-androgen, which affects and causes hyperprolactinemia |
| 5-alpha reductase inhibitors | Finasteride, Dutasteride | These drugs inhibit type II 5-alpha reductase–the enzyme that catalyses the conversion of testosterone into more biologically potent dihydrotestosterone (DHT), with the subsequent decrease in nitric oxide synthase activity |
| Cancer drugs | Cisplatin, Vincristine, Vinblastine | Chemotherapy is known to cause ED by causing neural and vasculature damage. Moreover, long-acting gonadotrophin-releasing hormone agonists used for prostate and breast cancer cause hypogonadism, leading to a reduction in sexual desire and ED. |
| Miscellaneous | Antiandrogens (cyproterone), Digoxin, Steroids, Immunosuppressants (Sirolimus, Everolimus), Protease inhibitors in HIV | Various, complex mechanisms: the blocking of the androgen receptors, the lowering of serum testosterone and the impairment of gonadal functions. |
| Drug Class | Examples | Postulated Mechanism Contributing to Retroperitoneal Fibrosis |
|---|---|---|
| Non-opioid and opioid analgetics | Aspirin, Paracetamol, Codeine | Drugs may initiate an immune response in the retroperitoneum followed by a progression to a chronic inflammatory process in the form of the proliferation of fibroblasts and the deposition of collagen, fibronectin, tenascin and glycosaminoglycans. The process of fibrosis involves the adjacent organs, including the ureters, causing their compression and possible symptoms of obstructive uropathy. |
| Antihypertensives | Hydralazine, Methyldopa, Reserpine, Hydrochlorothiazide | |
| Beta-adrenergic antagonists | Non-selective (Propranolol, Sotalol, Oxyprenolol) | |
| Beta1-selective agents (Metoprolol, Atenolol) | ||
| Cytostatics | Carboplatin, Methotrexate | |
| Biological drugs | Etanercept (TNF-alpha receptor blocker), Infliximab (anti-TNF-alpha monoclonal antibody) | |
| Miscellaneous | Egrot derivatives, Haloperidol, Ampicillin, Glibenclamide, |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dobrek, L. Lower Urinary Tract Disorders as Adverse Drug Reactions—A Literature Review. Pharmaceuticals 2023, 16, 1031. https://doi.org/10.3390/ph16071031
Dobrek L. Lower Urinary Tract Disorders as Adverse Drug Reactions—A Literature Review. Pharmaceuticals. 2023; 16(7):1031. https://doi.org/10.3390/ph16071031
Chicago/Turabian StyleDobrek, Lukasz. 2023. "Lower Urinary Tract Disorders as Adverse Drug Reactions—A Literature Review" Pharmaceuticals 16, no. 7: 1031. https://doi.org/10.3390/ph16071031
APA StyleDobrek, L. (2023). Lower Urinary Tract Disorders as Adverse Drug Reactions—A Literature Review. Pharmaceuticals, 16(7), 1031. https://doi.org/10.3390/ph16071031






