The Novel Tetra-Specific Drug C-192, Conjugated Using UniStac, Alleviates Non-Alcoholic Steatohepatitis in an MCD Diet-Induced Mouse Model
Abstract
1. Introduction
2. Results
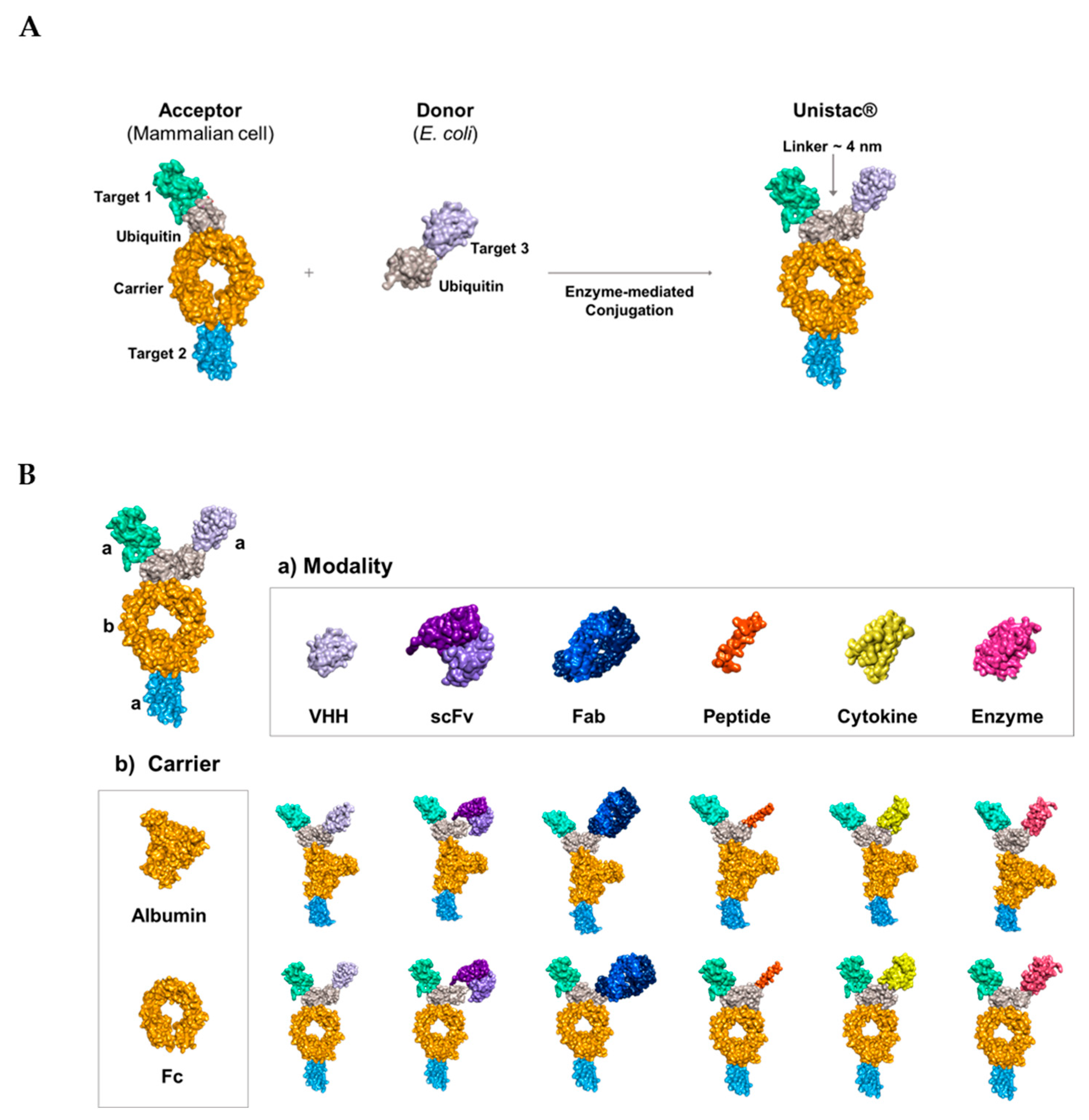
2.1. Conjugation of Multiple Targets Using a Novel Enzyme-Mediated Platform—UniStac
2.2. Development of the Novel Tetra-Specific Drug C-192
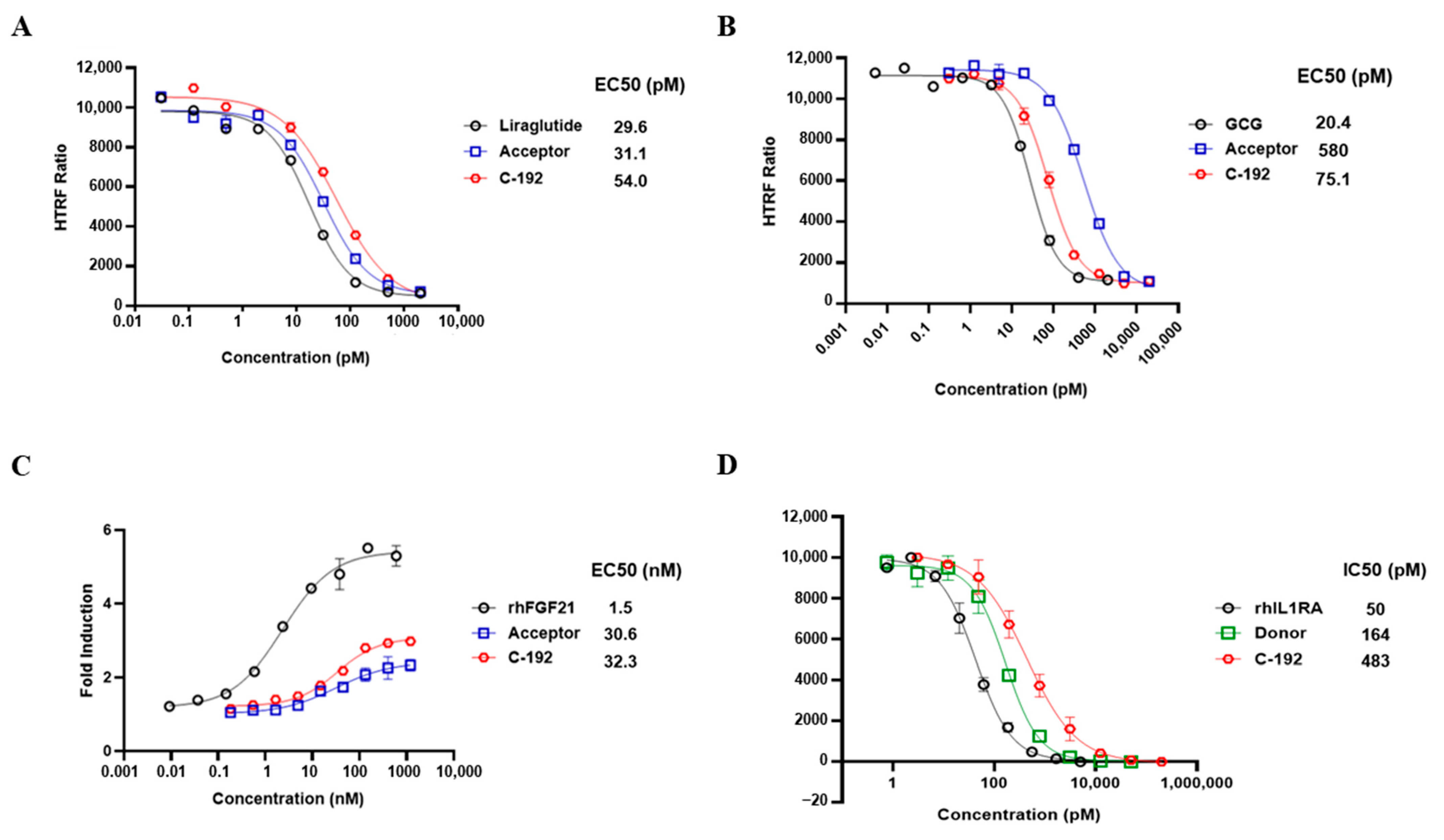
2.3. In Vitro Potencies of C-192 on Target Molecules
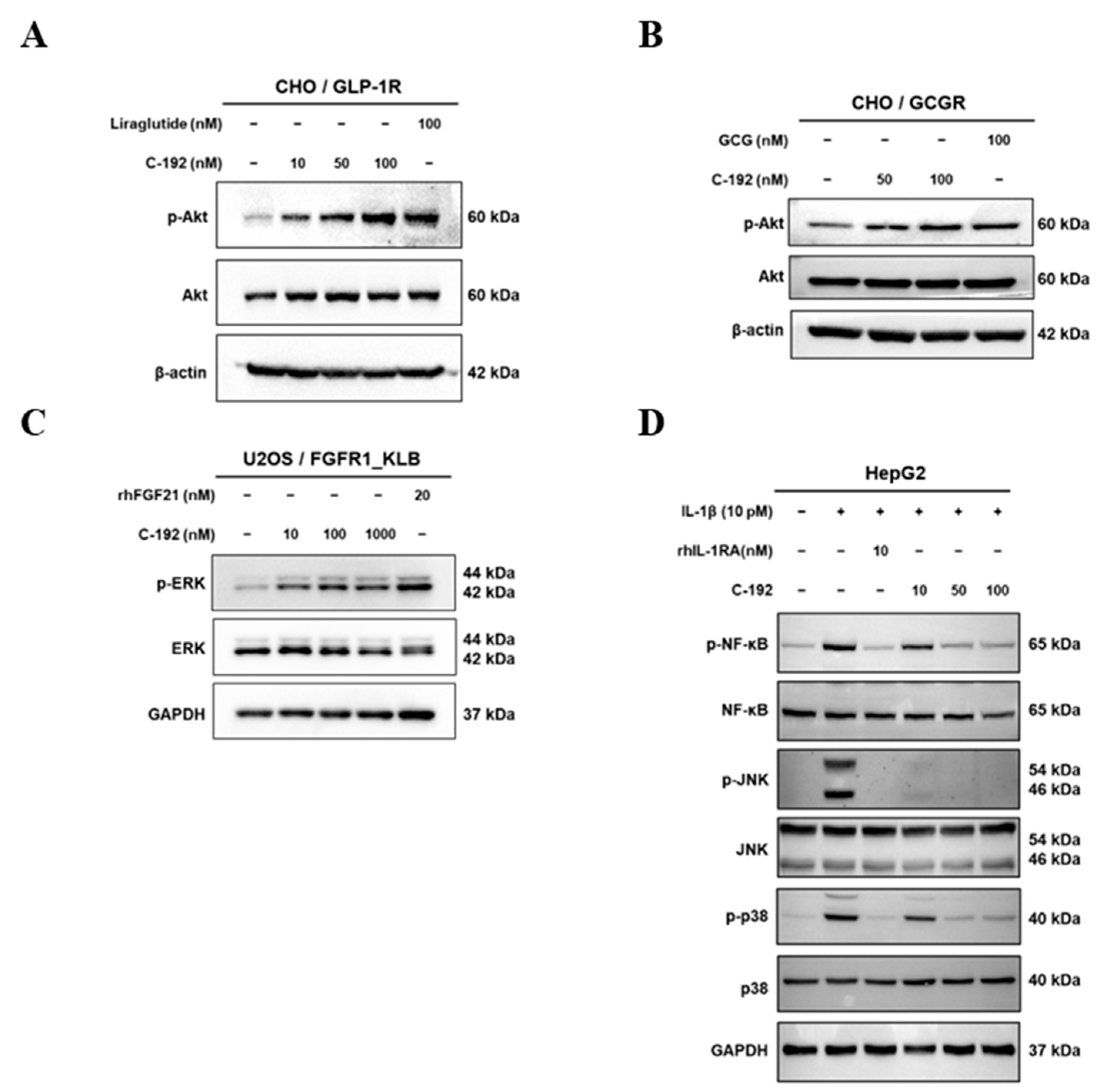
2.4. Downstream Regulatory Mechanism of Tetra-Specific Targets in C-192
2.5. Pharmacokinetic Profile of C-192 and its Therapeutic Effects on the MCD Diet-Induced NASH Mouse Model
3. Discussion
4. Materials and Methods
4.1. LC-MS/MS and Protein Purification
4.2. DSF Analysis
4.3. Affinity Measurement
4.4. In Vitro Biological Assays
4.5. Western Blotting Analysis
4.6. Pharmacokinetics of C-192 in Animal Models
4.7. MCD Diet-Induced Mouse Model
4.8. Measurement of ALT Levels
4.9. Histological Analysis
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Spiess, C.; Zhai, Q.; Carter, P.J. Alternative molecular formats and therapeutic applications for bispecific antibodies. Mol. Immunol. 2015, 67, 95–106. [Google Scholar] [CrossRef]
- Lim, S.M.; Pyo, K.H.; Soo, R.A.; Cho, B.C. The promise of bispecific antibodies: Clinical applications and challenges. Cancer Treat. Rev. 2021, 99, 102240. [Google Scholar] [CrossRef]
- Makhoba, X.H.; Viegas, C.; Mosa, R.A.; Viegas, F.P.D.; Pooe, O.J. Potential impact of the multi-target drug approach in the treatment of some complex diseases. Drug Des. Devel Ther. 2020, 14, 3235–3249. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Chen, Y.; Park, J.; Liu, X.; Hu, Y.; Wang, T.; McFarland, K.; Betenbaugh, M.J. Design and production of bispecific antibodies. Antibodies 2019, 8, 43. [Google Scholar] [CrossRef] [PubMed]
- Elgundi, Z.; Reslan, M.; Cruz, E.; Sifniotis, V.; Kayser, V. The state-of-play and future of antibody therapeutics. Adv. Drug Deliv. Rev. 2017, 122, 2–19. [Google Scholar] [CrossRef]
- Brinkmann, U.; Kontermann, R.E. The making of bispecific antibodies. mAbs 2017, 9, 182–212. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Saxena, A.; Sidhu, S.S.; Wu, D. Fc engineering for developing therapeutic bispecific antibodies and novel scaffolds. Front. Immunol. 2017, 8, 38. [Google Scholar] [CrossRef] [PubMed]
- Mastrandrea, L.D.; You, J.; Niles, E.G.; Pickart, C.M. E2/E3-mediated assembly of lysine 29-linked polyubiquitin chains. J. Biol. Chem. 1999, 274, 27299–27306. [Google Scholar] [CrossRef] [PubMed]
- Hurley, J.H.; Lee, S.; Prag, G. Ubiquitin-binding domains. Biochem. J. 2006, 399, 361–372. [Google Scholar] [CrossRef]
- Lillich, F.F.; Imig, J.D.; Proschak, E. Multi-target approaches in metabolic syndrome. Front. Pharmacol. 2020, 11, 554961. [Google Scholar] [CrossRef] [PubMed]
- Siegmund, V.; Piater, B.; Zakeri, B.; Eichhorn, T.; Fischer, F.; Deutsch, C.; Becker, S.; Toleikis, L.; Hock, B.; Betz, U.A.; et al. Spontaneous isopeptide bond formation as a powerful tool for engineering site-specific antibody-drug conjugates. Sci. Rep. 2016, 6, 39291. [Google Scholar] [CrossRef]
- Antos, J.M.; Ingram, J.; Fang, T.; Pishesha, N.; Truttmann, M.C.; Ploegh, H.L. Site-specific protein labeling via sortase-mediated transpeptidation. Curr. Protoc. Protein Sci. 2017, 89, 15.3.1–15.3.19. [Google Scholar] [CrossRef]
- Powell, E.E.; Wong, V.W.; Rinella, M. Non-alcoholic fatty liver disease. Lancet 2021, 397, 2212–2224. [Google Scholar] [CrossRef]
- Sheka, A.C.; Adeyi, O.; Thompson, J.; Hameed, B.; Crawford, P.A.; Ikramuddin, S. Nonalcoholic steatohepatitis: A review. JAMA 2020, 323, 1175–1183. [Google Scholar] [CrossRef] [PubMed]
- Sumida, Y.; Yoneda, M. Current and future pharmacological therapies for NAFLD/NASH. J. Gastroenterol. 2018, 53, 362–376. [Google Scholar] [CrossRef]
- Baggio, L.L.; Drucker, D.J. Biology of incretins: GLP-1 and GIP. Gastroenterology 2007, 132, 2131–2157. [Google Scholar] [CrossRef]
- Zhang, F.; Chen, Z.; Wu, D.; Tian, L.; Chen, Q.; Ye, Y.; Chen, W.; Wu, X.; Wu, P.; Yuan, W.; et al. Recombinant human GLP-1 beinaglutide regulates lipid metabolism of adipose tissues in diet-induced obese mice. iScience 2021, 24, 103382. [Google Scholar] [CrossRef] [PubMed]
- Somm, E.; Montandon, S.A.; Loizides-Mangold, U.; Gaïa, N.; Lazarevic, V.; De Vito, C.; Perroud, E.; Bochaton-Piallat, M.L.; Dibner, C.; Schrenzel, J.; et al. The GLP-1R agonist liraglutide limits hepatic lipotoxicity and inflammatory response in mice fed a methionine-choline deficient diet. Transl. Res. 2021, 227, 75–88. [Google Scholar] [CrossRef]
- Zhou, J.Y.; Poudel, A.; Welchko, R.; Mekala, N.; Chandramani-Shivalingappa, P.; Rosca, M.G.; Li, L. Liraglutide improves insulin sensitivity in high fat diet induced diabetic mice through multiple pathways. Eur. J. Pharmacol. 2019, 861, 172594. [Google Scholar] [CrossRef]
- Wong, V.W.; Wong, G.L. A LEAN treatment for non-alcoholic steatohepatitis. Lancet 2016, 387, 628–630. [Google Scholar] [CrossRef] [PubMed]
- Newsome, P.N.; Buchholtz, K.; Cusi, K.; Linder, M.; Okanoue, T.; Ratziu, V.; Sanyal, A.J.; Sejling, A.S.; Harrison, S.A.; NN9931-4296 Investigators. A placebo-controlled trial of subcutaneous semaglutide in nonalcoholic steatohepatitis. N. Engl. J. Med. 2021, 384, 1113–1124. [Google Scholar] [CrossRef] [PubMed]
- Baggio, L.L.; Drucker, D.J. Glucagon-like peptide-1 receptor co-agonists for treating metabolic disease. Mol. Metab. 2021, 46, 101090. [Google Scholar] [CrossRef] [PubMed]
- Adeva-Andany, M.M.; Funcasta-Calderón, R.; Fernández-Fernández, C.; Castro-Quintela, E.; Carneiro-Freire, N. Metabolic effects of glucagon in humans. J. Clin. Transl. Endocrinol. 2019, 15, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Galsgaard, K.D.; Pedersen, J.; Knop, F.K.; Holst, J.J.; Wewer Albrechtsen, N.J. Glucagon receptor signaling and lipid metabolism. Front. Physiol. 2019, 10, 413. [Google Scholar] [CrossRef] [PubMed]
- Nestor, J.J.; Parkes, D.; Feigh, M.; Suschak, J.J.; Harris, M.S. Effects of ALT-801, a GLP-1 and glucagon receptor dual agonist, in a translational mouse model of non-alcoholic steatohepatitis. Sci. Rep. 2022, 12, 6666. [Google Scholar] [CrossRef] [PubMed]
- Hope, D.C.D.; Vincent, M.L.; Tan, T.M.M. Striking the balance: GLP-1/glucagon co-agonism as a treatment strategy for obesity. Front. Endocrinol. 2021, 12, 735019. [Google Scholar] [CrossRef]
- Boland, M.L.; Laker, R.C.; Mather, K.; Nawrocki, A.; Oldham, S.; Boland, B.B.; Lewis, H.; Conway, J.; Naylor, J.; Guionaud, S.; et al. Resolution of NASH and hepatic fibrosis by the GLP-1R/GCGR dual-agonist cotadutide via modulating mitochondrial function and lipogenesis. Nat. Metab. 2020, 2, 413–431. [Google Scholar] [CrossRef]
- Elvert, R.; Herling, A.W.; Bossart, M.; Weiss, T.; Zhang, B.; Wenski, P.; Wandschneider, J.; Kleutsch, S.; Butty, U.; Kannt, A.; et al. Running on mixed fuel-dual agonistic approach of GLP-1 and GCG receptors leads to beneficial impact on body weight and blood glucose control: A comparative study between mice and non-human primates. Diabetes Obes. Metab. 2018, 20, 1836–1851. [Google Scholar] [CrossRef]
- Song, N.; Xu, H.; Liu, J.; Zhao, Q.; Chen, H.; Yan, Z.; Yang, R.; Luo, Z.; Liu, Q.; Ouyang, J.; et al. Design of a highly potent GLP-1R and GCGR dual-agonist for recovering hepatic fibrosis. Acta Pharm. Sin. B 2022, 12, 2443–2461. [Google Scholar] [CrossRef]
- Kayed, A.; Melander, S.A.; Khan, S.; Andreassen, K.V.; Karsdal, M.A.; Henriksen, K. The effects of dual GLP-1/glucagon receptor agonists with different receptor selectivity in mouse models of obesity and nonalcoholic steatohepatitis. J. Pharmacol. Exp. Ther. 2023, 384, 406–416. [Google Scholar] [CrossRef]
- Lee, J.H.; Kang, Y.E.; Chang, J.Y.; Park, K.C.; Kim, H.W.; Kim, J.T.; Kim, H.J.; Yi, H.S.; Shong, M.; Chung, H.K.; et al. An engineered FGF21 variant, LY2405319, can prevent non-alcoholic steatohepatitis by enhancing hepatic mitochondrial function. Am. J. Transl. Res. 2016, 8, 4750–4763. [Google Scholar]
- Tillman, E.J.; Rolph, T. FGF21: An emerging therapeutic target for non-alcoholic steatohepatitis and related metabolic diseases. Front. Endocrinol. 2020, 11, 601290. [Google Scholar] [CrossRef]
- Zhang, J.; Li, Y. Fibroblast growth factor 21 analogs for treating metabolic disorders. Front. Endocrinol. 2015, 6, 168. [Google Scholar] [CrossRef] [PubMed]
- Keinicke, H.; Sun, G.; Mentzel, C.M.J.; Fredholm, M.; John, L.M.; Andersen, B.; Raun, K.; Kjaergaard, M. FGF21 regulates hepatic metabolic pathways to improve steatosis and inflammation. Endocr. Connect. 2020, 9, 755–768. [Google Scholar] [CrossRef] [PubMed]
- Fisher, F.M.; Chui, P.C.; Nasser, I.A.; Popov, Y.; Cunniff, J.C.; Lundasen, T.; Kharitonenkov, A.; Schuppan, D.; Flier, J.S.; Maratos-Flier, E. Fibroblast growth factor 21 limits lipotoxicity by promoting hepatic fatty acid activation in mice on methionine and choline-deficient diets. Gastroenterology 2014, 147, 1073–1083.e6. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.; Parlee, S.; Perez-Tilve, D.; Li, P.; Pan, J.; Mroz, P.A.; Hansen, A.M.K.; Andersen, B.; Finan, B.; Kharitonenkov, A.; et al. Molecular elements in FGF19 and FGF21 defining KLB/FGFR activity and specificity. Mol. Metab. 2018, 13, 45–55. [Google Scholar] [CrossRef]
- Kilkenny, D.M.; Rocheleau, J.V. The FGF21 receptor signaling complex: Klothoβ, FGFR1c, and other regulatory interactions. Vitam. Horm. 2016, 101, 17–58. [Google Scholar] [PubMed]
- Finan, B.; Yang, B.; Ottaway, N.; Smiley, D.L.; Ma, T.; Clemmensen, C.; Chabenne, J.; Zhang, L.; Habegger, K.M.; Fischer, K.; et al. A rationally designed monomeric peptide triagonist corrects obesity and diabetes in rodents. Nat. Med. 2015, 21, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Gilroy, C.A.; Capozzi, M.E.; Varanko, A.K.; Tong, J.; D’alessio, D.A.; Campbell, J.E.; Chilkoti, A. Sustained release of a GLP-1 and FGF21 dual agonist from an injectable depot protects mice from obesity and hyperglycemia. Sci. Adv. 2020, 6, eaaz9890. [Google Scholar] [CrossRef]
- Cui, J.; Shang, A.; Wang, W.; Chen, W. Rational design of a GLP-1/GIP/Gcg receptor triagonist to correct hyperglycemia, obesity and diabetic nephropathy in rodent animals. Life Sci. 2020, 260, 118339. [Google Scholar] [CrossRef]
- Tsutsui, H.; Cai, X.; Hayashi, S. Interleukin-1 family cytokines in liver diseases. Mediat. Inflamm. 2015, 2015, 630265. [Google Scholar] [CrossRef] [PubMed]
- Gabay, C.; Lamacchia, C.; Palmer, G. IL-1 pathways in inflammation and human diseases. Nat. Rev. Rheumatol. 2010, 6, 232–241. [Google Scholar] [CrossRef]
- Barbier, L.; Ferhat, M.; Salamé, E.; Robin, A.; Herbelin, A.; Gombert, J.M.; Silvain, C.; Barbarin, A. Interleukin-1 family cytokines: Keystones in liver inflammatory diseases. Front. Immunol. 2019, 10, 2014. [Google Scholar] [CrossRef] [PubMed]
- Mirea, A.M.; Tack, C.J.; Chavakis, T.; Joosten, L.A.B.; Toonen, E.J.M. IL-1 family cytokine pathways underlying NAFLD: Towards new treatment strategies. Trends Mol. Med. 2018, 24, 458–471. [Google Scholar] [CrossRef] [PubMed]
- Meier, R.P.H.; Meyer, J.; Montanari, E.; Lacotte, S.; Balaphas, A.; Muller, Y.D.; Clément, S.; Negro, F.; Toso, C.; Morel, P.; et al. Interleukin-1 receptor antagonist modulates liver inflammation and fibrosis in mice in a model-dependent manner. Int. J. Mol. Sci. 2019, 20, 1295. [Google Scholar] [CrossRef]
- Arend, W.P.; Malyak, M.; Guthridge, C.J.; Gabay, C. Interleukin-1 receptor antagonist: Role in biology. Annu. Rev. Immunol. 1998, 16, 27–55. [Google Scholar] [CrossRef]
- Wree, A.; Broderick, L.; Canbay, A.; Hoffman, H.M.; Feldstein, A.E. From NAFLD to NASH to cirrhosis-New insights into disease mechanisms. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 627–636. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Walters, K.J. Multitasking with ubiquitin through multivalent interactions. Trends Biochem. Sci. 2010, 35, 352–360. [Google Scholar] [CrossRef] [PubMed]
- Cappadocia, L.; Lima, C.D. Ubiquitin-like protein conjugation: Structures, chemistry, and mechanism. Chem. Rev. 2018, 118, 889–918. [Google Scholar] [CrossRef]
- Heride, C.; Urbé, S.; Clague, M.J. Ubiquitin code assembly and disassembly. Curr. Biol. 2014, 24, R215–R220. [Google Scholar] [CrossRef]
- Spasser, L.; Brik, A. Chemistry and biology of the ubiquitin signal. Angew. Chem. Int. Ed. Engl. 2012, 51, 6840–6862. [Google Scholar] [CrossRef] [PubMed]
- White, J.W.; Saunders, G.F. Structure of the human glucagon gene. Nucleic Acids Res. 1986, 14, 4719–4730. [Google Scholar] [CrossRef][Green Version]
- McKenna, S.; Spyracopoulos, L.; Moraes, T.; Pastushok, L.; Ptak, C.; Xiao, W.; Ellison, M.J. Noncovalent interaction between ubiquitin and the human DNA repair protein Mms2 is required for Ubc13-mediated polyubiquitination. J. Biol. Chem. 2001, 276, 40120–40126. [Google Scholar] [CrossRef] [PubMed]
- Martinelli, V. Combination therapy. Neurol. Sci. 2006, 27 (Suppl. S5), S350–S354. [Google Scholar] [CrossRef]
- Oudega, M.; Bradbury, E.J.; Ramer, M.S. Combination therapies. Handb. Clin. Neurol. 2012, 109, 617–636. [Google Scholar] [PubMed]
- Meric-Bernstam, F.; Larkin, J.; Tabernero, J.; Bonini, C. Enhancing anti-tumour efficacy with immunotherapy combinations. Lancet 2021, 397, 1010–1022. [Google Scholar] [CrossRef]
- Suurs, F.V.; Lub-de Hooge, M.N.; de Vries, E.G.; de Groot, D.J.A. A review of bispecific antibodies and antibody constructs in oncology and clinical challenges. Pharmacol. Ther. 2019, 201, 103–119. [Google Scholar] [CrossRef]
- Shim, H. Bispecific antibodies and antibody–drug conjugates for cancer therapy: Technological considerations. Biomolecules 2020, 10, 360. [Google Scholar] [CrossRef]
- Mazor, Y.; Oganesyan, V.; Yang, C.; Hansen, A.; Wang, J.; Liu, H.; Sachsenmeier, K.; Carlson, M.; Gadre, D.V.; Borrok, M.J.; et al. Improving target cell specificity using a novel monovalent bispecific IgG design. MAbs 2015, 7, 77–389. [Google Scholar] [CrossRef]
- Godar, M.; de Haard, H.; Blanchetot, C.; Rasser, J. Therapeutic bispecific antibody formats: A patent applications review (1994-2017). Expert. Opin. Ther. Pat. 2018, 28, 251–276. [Google Scholar] [CrossRef]
- Elshiaty, M.; Schindler, H.; Christopoulos, P. Principles and current clinical landscape of multispecific antibodies against cancer. Int. J. Mol. Sci. 2021, 22, 5632. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Yi, M.; Zhu, S.; Wang, H.; Wu, K. Recent advances and challenges of bispecific antibodies in solid tumors. Exp. Hematol. Oncol. 2021, 10, 56. [Google Scholar] [CrossRef] [PubMed]
- Komander, D.; Lord, C.J.; Scheel, H.; Swift, S.; Hofmann, K.; Ashworth, A.; Barford, D. The structure of the CYLD USP domain explains its specificity for Lys63-linked polyubiquitin and reveals a B box module. Mol. Cell 2008, 29, 451–464. [Google Scholar] [CrossRef]
- Van Wijk, S.J.; Fulda, S.; Dikic, I.; Heilemann, M. Visualizing ubiquitination in mammalian cells. EMBO Rep. 2019, 20, e46520. [Google Scholar] [CrossRef] [PubMed]
- Dennis, M.S.; Zhang, M.; Meng, Y.G.; Kadkhodayan, M.; Kirchhofer, D.; Combs, D.; Damico, L.A. Albumin binding as a general strategy for improving the pharmacokinetics of proteins. J. Biol. Chem. 2002, 277, 35035–35043. [Google Scholar] [CrossRef]
- Ishizaka, T.; Yoshimatsu, Y.; Maeda, Y.; Chiba, K.; Mori, K. Negative lusitropic property of nifekalant identified using ventricular pressure-volume loop analyses in anesthetized monkeys. Exp. Anim. 2019, 68, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Kim, D.M.; Son, W.C.; Moon, B.G. A Selective Adenosine A(3) Receptor Antagonist, HL3501, Has Therapeutic Potential in Preclinical Liver and Renal Fibrosis Models. Vivo 2022, 36, 2186–2193. [Google Scholar] [CrossRef]
- Scott, P.J.H.; Koeppe, R.A.; Shao, X.; Rodnick, M.E.; Sowa, A.R.; Henderson, B.D.; Stauff, J.; Sherman, P.S.; Arteaga, J.; Carlo, D.J.; et al. The Effects of Intramuscular Naloxone Dose on Mu Receptor Displacement of Carfentanil in Rhesus Monkeys. Molecules 2020, 25, 1360. [Google Scholar] [CrossRef] [PubMed]
- Kleiner, D.E.; Brunt, E.M.; Van Natta, M.; Behling, C.; Contos, M.J.; Cummings, O.W.; Ferrell, L.D.; Liu, Y.C.; Torbenson, M.S.; Unalp-Arida, A.; et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 2005, 41, 1313–1321. [Google Scholar] [CrossRef] [PubMed]


| Ligand | Analyte | Ka (1/Ms) | Kd (1s) | KD (M) | Rmax (RU) | Chi2 (RU2) |
|---|---|---|---|---|---|---|
| C-192 | GLP-1R | 1.122 × 104 | 9.537 × 10−3 | 8.500 × 10−7 (850 nM) | 34.62 | 1.13 |
| GCGR | 2.630 × 104 | 5.108 × 10−4 | 1.942 × 10−8 (19.42 nM) | 9.699 | 0.141 | |
| β-Klotho | 2.279 × 105 | 3.831 × 10−3 | 1.681 × 10−8 (16.81 nM) | 175.7 | 69.6 | |
| IL-1R1 | 2.017 × 104 | 6.779 × 10−5 | 3.360 × 10−9 (3.36 nM) | 77.64 | 1.85 |
| Parameter | C57BL/6 Mice 10 mg/kg | Cynomolgus Monkey 15 mg/kg | ||||
|---|---|---|---|---|---|---|
| FGF21 Sandwich | GCG/FGF21 | IL-1RA/FGF21 | FGF21 Sandwich | GCG/FGF21 | IL-1RA/FGF21 | |
| AUClast (μg·hr/mL) | 147.9 | 117.5 | 114.5 | 1789.2 | 883.4 | 778.4 |
| Cmax (μg/mL) | 12.3 | 10.9 | 9.9 | 57.1 | 36.6 | 36.8 |
| Tmax (hr) | 8 | 4 | 4 | 8 | 8 | 8 |
| CL/F (ml/hr/kg) | 67.6 | 85.1 | 8.7 | 8.3 | 17.0 | 19.3 |
| t1/2,app (hr) | 18.4 | 11.2 | 5.5 | 62.7 | 38.7 | 16.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.; Chang, N.; Kim, Y.; Lee, J.; Oh, D.; Choi, J.; Kim, O.; Kim, S.; Choi, M.; Lee, J.; et al. The Novel Tetra-Specific Drug C-192, Conjugated Using UniStac, Alleviates Non-Alcoholic Steatohepatitis in an MCD Diet-Induced Mouse Model. Pharmaceuticals 2023, 16, 1601. https://doi.org/10.3390/ph16111601
Kim J, Chang N, Kim Y, Lee J, Oh D, Choi J, Kim O, Kim S, Choi M, Lee J, et al. The Novel Tetra-Specific Drug C-192, Conjugated Using UniStac, Alleviates Non-Alcoholic Steatohepatitis in an MCD Diet-Induced Mouse Model. Pharmaceuticals. 2023; 16(11):1601. https://doi.org/10.3390/ph16111601
Chicago/Turabian StyleKim, Jihye, Nakho Chang, Yunki Kim, Jaehyun Lee, Daeseok Oh, Jaeyoung Choi, Onyou Kim, Sujin Kim, Myongho Choi, Junyeob Lee, and et al. 2023. "The Novel Tetra-Specific Drug C-192, Conjugated Using UniStac, Alleviates Non-Alcoholic Steatohepatitis in an MCD Diet-Induced Mouse Model" Pharmaceuticals 16, no. 11: 1601. https://doi.org/10.3390/ph16111601
APA StyleKim, J., Chang, N., Kim, Y., Lee, J., Oh, D., Choi, J., Kim, O., Kim, S., Choi, M., Lee, J., Lee, J., Kim, J., Cho, M., Kim, M., Lee, K., Hwang, D., Sa, J. K., Park, S., Baek, S., & Im, D. (2023). The Novel Tetra-Specific Drug C-192, Conjugated Using UniStac, Alleviates Non-Alcoholic Steatohepatitis in an MCD Diet-Induced Mouse Model. Pharmaceuticals, 16(11), 1601. https://doi.org/10.3390/ph16111601






