Oxaliplatin Enhances the Apoptotic Effect of Mesenchymal Stem Cells, Delivering Soluble TRAIL in Chemoresistant Colorectal Cancer
Abstract
:1. Introduction
2. Results
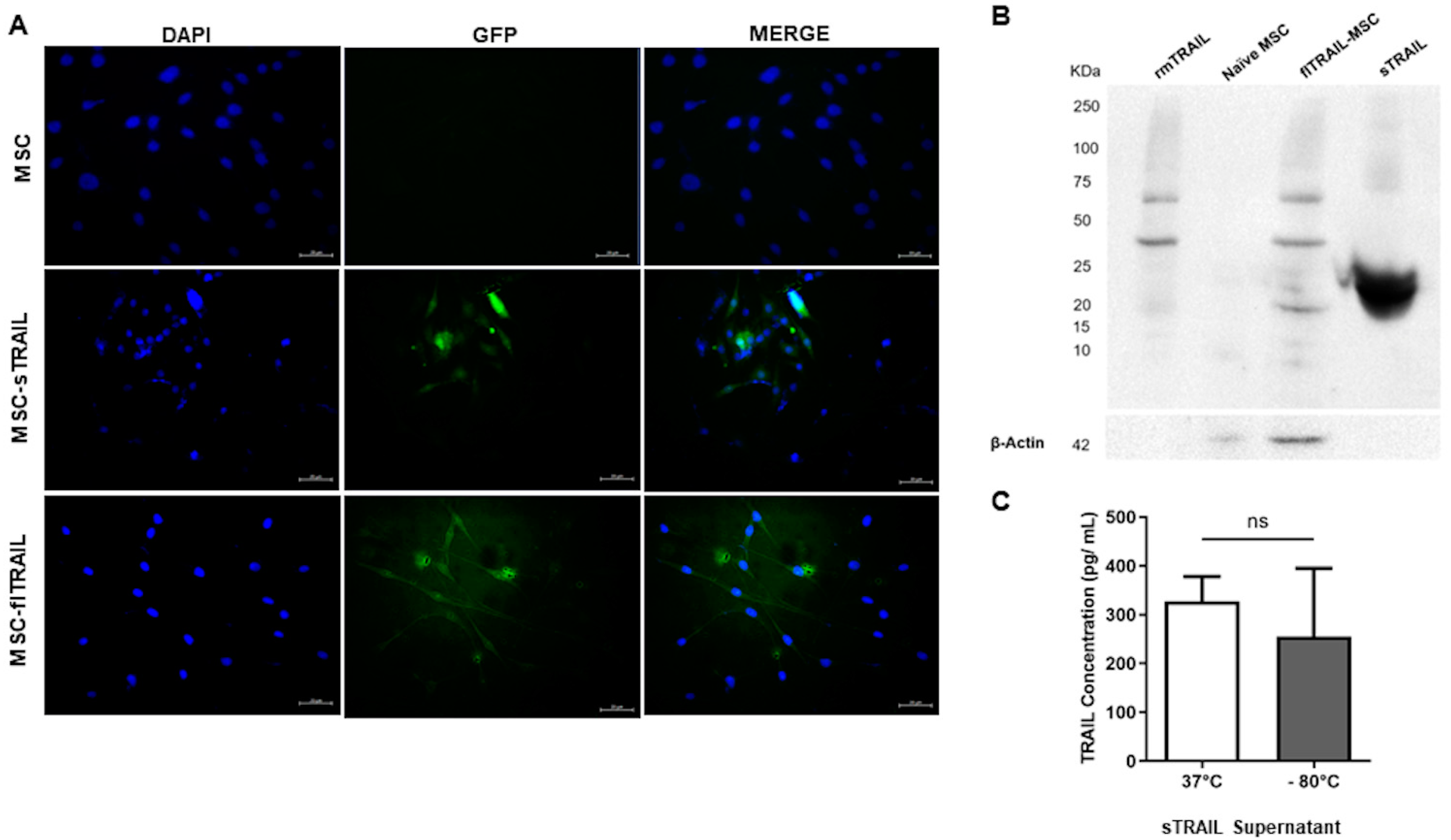
2.1. Lentiviral Transduced BM-MSC Successfully Expresses Soluble TRAIL
2.2. Colorectal Cancer Cell Lines Present Chemoresistance to First-Line Treatment
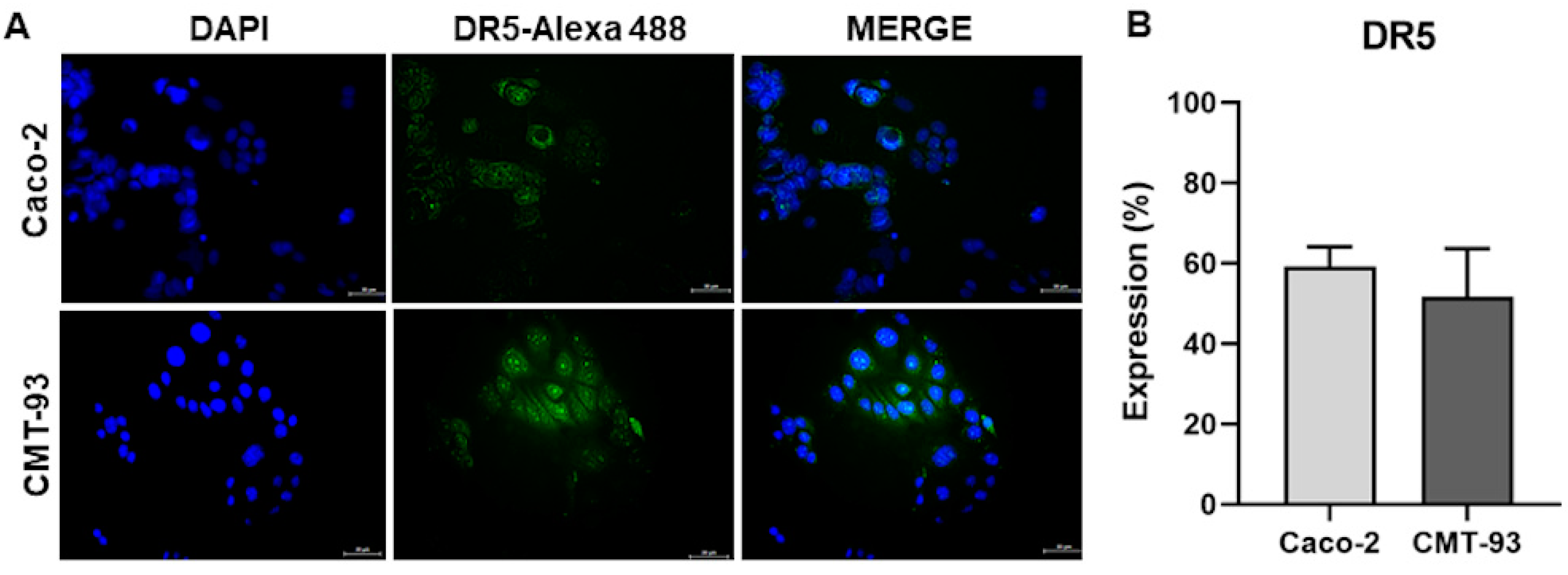
2.3. Colorectal Cancer Cell Lines Are Susceptible to TRAIL
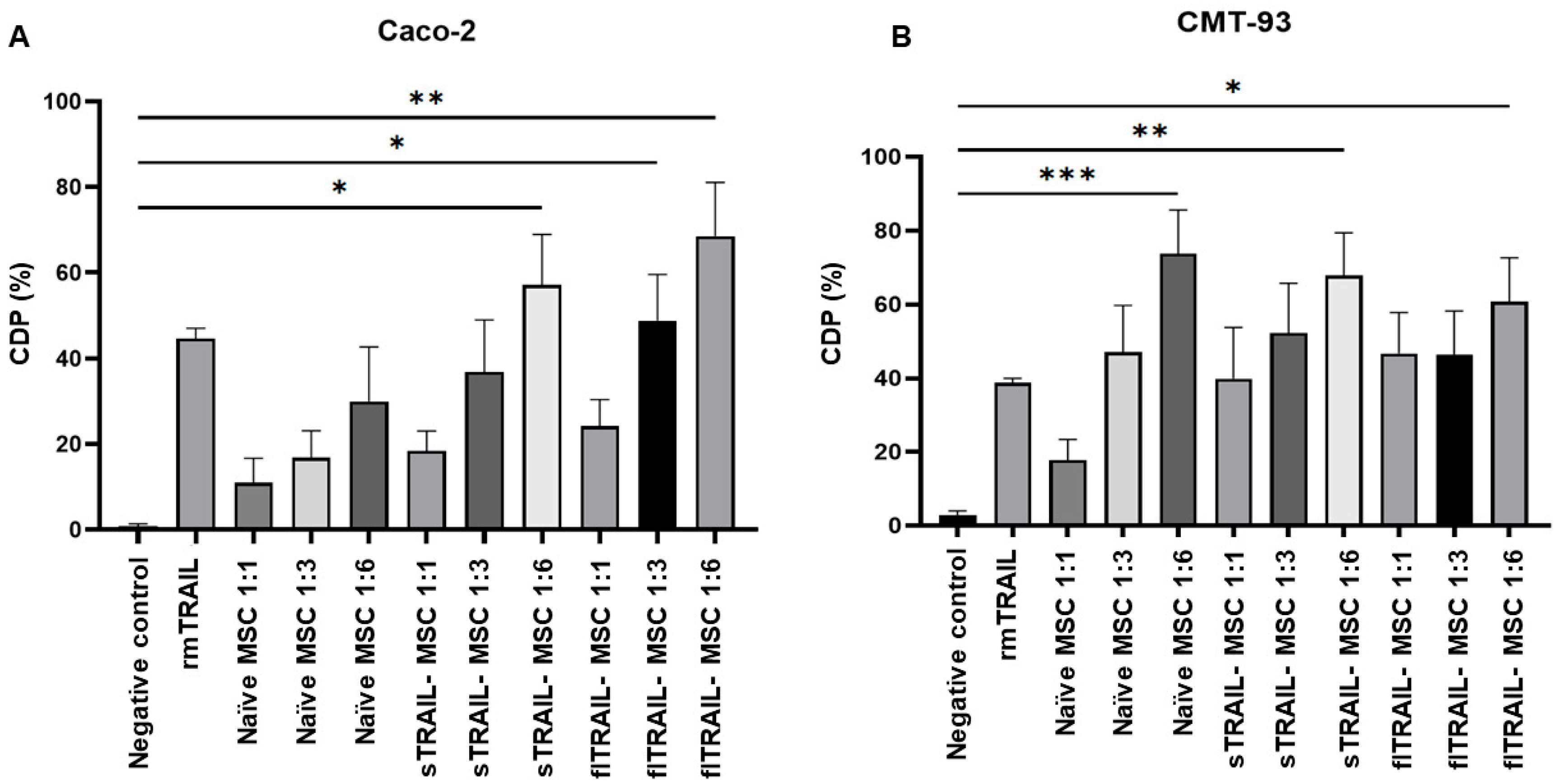
2.4. MSC Expressing sTRAIL Induces Cell Death in Colorectal Cancer Cell Lines
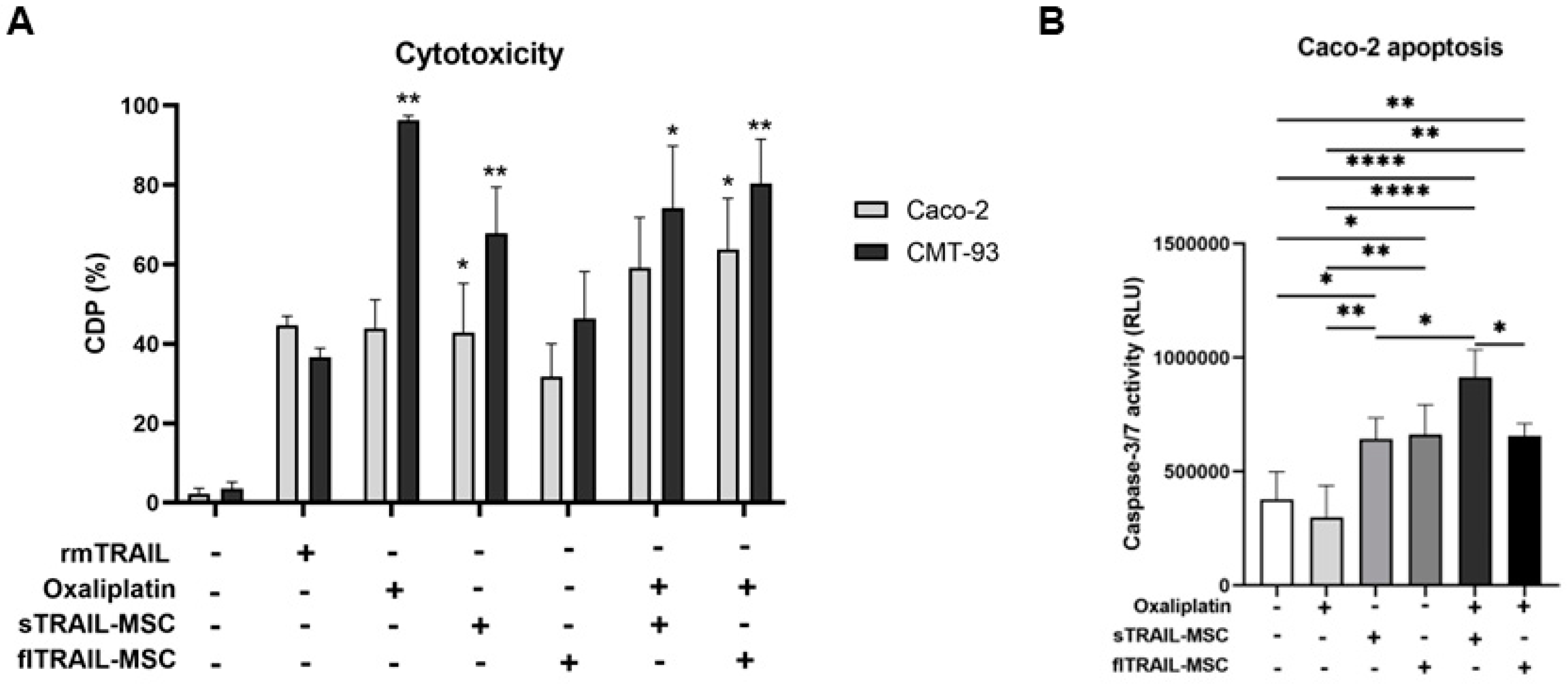
2.5. Combined Treatment of Oxaliplatin and MSC Expressing Soluble TRAIL Increases Apoptosis in a Chemoresistant Colorectal Cancer Cell Line
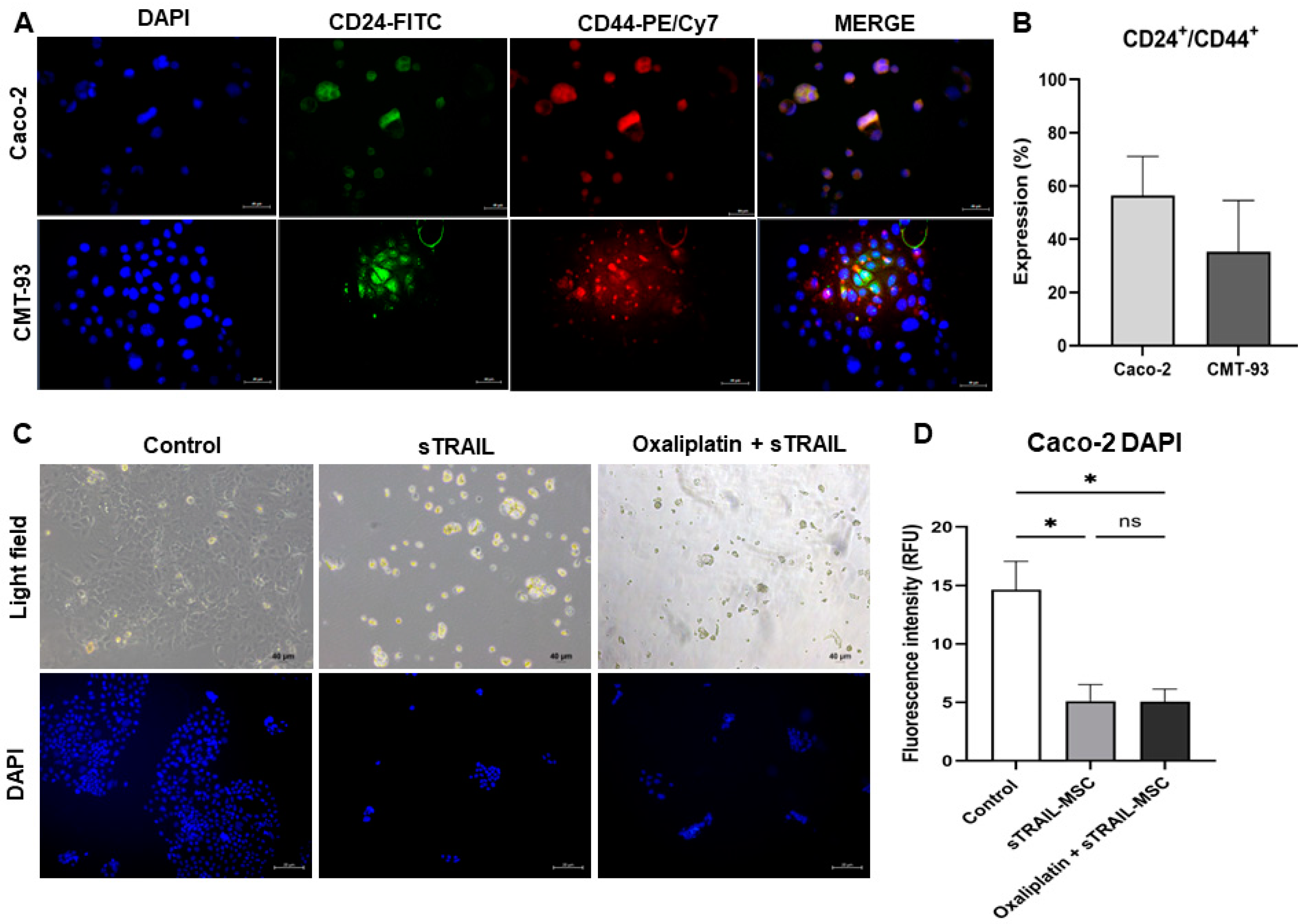
2.6. Mesenchymal Stem Cell-Secreted sTRAIL Induces Cell Death in Colorectal Cancer Cell Lines Expressing Cancer Stem Cell Markers
3. Discussion
4. Materials and Methods
4.1. MSC Isolation and Characterization
4.2. Lentiviral Transduction of BM-MSC
4.3. Transgene Expression Validation and Quantification
4.4. Colorectal Cancer Cell Lines
4.5. Susceptibility to TRAIL by Evaluating DR5 Receptor Expression
4.6. Determination of the Half-Maximal Inhibitory Concentration (IC50)
4.7. Cytotoxicity and Apoptotic Effect of MSC Expressing TRAIL by Co-Culture Assay
4.8. Expression of CSC Markers in Colorectal Cancer Cell Lines and Cell Death Evaluation by Fluorescence
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Wagle, N.S.; Cercek, A.; Smith, R.A.; Jemal, A. Colorectal cancer statistics, 2023. CA Cancer J. Clin. 2023, 73, 233–254. [Google Scholar] [CrossRef] [PubMed]
- Hervieu, C.; Christou, N.; Battu, S.; Mathonnet, M. The Role of Cancer Stem Cells in Colorectal Cancer: From the Basics to Novel Clinical Trials. Cancers 2021, 13, 1092. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Yang, F.; Chen, S.; Tai, J. Mechanisms on chemotherapy resistance of colorectal cancer stem cells and research progress of reverse transformation: A mini-review. Front. Med. 2022, 9, 995882. [Google Scholar] [CrossRef]
- Yu, Z.; Pestell, T.G.; Lisanti, M.P.; Pestell, R.G. Cancer stem cells. Int. J. Biochem. Cell Biol. 2013, 44, 2144–2151. [Google Scholar] [CrossRef] [PubMed]
- Walcher, L.; Kistenmacher, A.-K.; Suo, H.; Kitte, R.; Dluczek, S.; Strauß, A.; Blaudszun, A.-R.; Yevsa, T.; Fricke, S.; Kossatz-Boehlert, U. Cancer Stem Cells—Origins and Biomarkers: Perspectives for Targeted Personalized Therapies. Front. Immunol. 2020, 11, 1280. [Google Scholar] [CrossRef]
- Garza-Treviño, E.; Martínez-Rodríguez, H.; Delgado-González, P.; Solís-Coronado, O.; Ortíz-Lopez, R.; Soto-Domínguez, A.; Treviño, V.; Padilla-Rivas, G.; Islas-Cisneros, J.; Quiroz-Reyes, A.; et al. Chemosensitivity analysis and study of gene resistance on tumors and cancer stem cell isolates from patients with colorectal cancer. Mol. Med. Rep. 2021, 24, 721. [Google Scholar] [CrossRef] [PubMed]
- De Miguel, D.; Lemke, J.; Anel, A.; Walczak, H.; Martinez-Lostao, L. Onto better TRAILs for cancer treatment. Cell Death Differ. 2016, 23, 733–747. [Google Scholar] [CrossRef] [PubMed]
- Thorburn, A. Tumor Necrosis Factor-Related Apoptosis-Inducing Ligand (TRAIL) Pathway Signaling. J. Thorac. Oncol. 2007, 2, 461–465. [Google Scholar] [CrossRef] [PubMed]
- Holoch, P.A.; Griffith, T.S. TNF-related apoptosis-inducing ligand (TRAIL): A new path to anti-cancer therapies. Eur. J. Pharmacol. 2009, 625, 63–72. [Google Scholar] [CrossRef]
- Snajdauf, M.; Havlova, K.; Vachtenheim, J.; Ozaniak, A.; Lischke, R.; Bartunkova, J.; Smrz, D.; Strizova, Z. The TRAIL in the Treatment of Human Cancer: An Update on Clinical Trials. Front. Mol. Biosci. 2021, 8, 628332. [Google Scholar] [CrossRef] [PubMed]
- Quiroz-Reyes, A.G.; Delgado-Gonzalez, P.; Islas, J.F.; Gallegos, J.L.D.; Garza, J.H.M.; Garza-Treviño, E.N. Behind the adaptive and resistance mechanisms of cancer stem cells to trail. Pharmaceutics 2021, 13, 1062. [Google Scholar] [CrossRef] [PubMed]
- Shlyakhtina, Y.; Pavet, V.; Gronemeyer, H. Dual role of DR5 in death and survival signaling leads to TRAIL resistance in cancer cells. Cell Death Dis. 2017, 8, e3025-16. [Google Scholar] [CrossRef] [PubMed]
- Kingsak, M.; Meethong, T.; Jongkhumkrong, J.; Cai, L.; Wang, Q. Therapeutic potential of oncolytic viruses in the era of precision oncology. Biomater. Transl. 2023, 4, 67–84. [Google Scholar]
- Hmadcha, A.; Martin-Montalvo, A.; Gauthier, B.R.; Soria, B.; Capilla-Gonzalez, V. Therapeutic Potential of Mesenchymal Stem Cells for Cancer Therapy. Front. Bioeng. Biotechnol. 2020, 8, 43. [Google Scholar] [CrossRef] [PubMed]
- Marini, I.; Siegemund, M.; Hutt, M.; Kontermann, R.E.; Pfizenmaier, K. Antitumor activity of a mesenchymal stem cell line stably secreting a tumor-targeted TNF-related apoptosis-inducing ligand fusion protein. Front. Immunol. 2017, 8, 536. [Google Scholar] [CrossRef]
- Marini, F.; Dembinski, J.; Studeny, M.; Zompetta, C.; Andreeff, M. Mesenchymal Stem Cells as Delivery Systems for Cancer Therapy: Evaluation of Tropism and Efficacy. Mol. Ther. 2005, 11, S173. [Google Scholar] [CrossRef]
- Chen, Z.; Jin, M.; He, H.; Dong, J.; Li, J.; Nie, J.; Wang, Z.; Xu, J.; Wu, F. Mesenchymal stem cells and macrophages and their interactions in tendon-bone healing. J. Orthop. Transl. 2023, 39, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.; Xu, L.; Bandyopadhyay, S.; Sethi, S.; Reddy, K.B. Cisplatin and TRAIL enhance breast cancer stem cell death. Int. J. Oncol. 2011, 39, 891–898. [Google Scholar] [CrossRef] [PubMed]
- Rossignoli, F.; Spano, C.; Grisendi, G.; Foppiani, E.M.; Golinelli, G.; Mastrolia, I.; Bestagno, M.; Candini, O.; Petrachi, T.; Recchia, A.; et al. MSC-delivered soluble TRAIl and paclitaxel as novel combinatory treatment for pancreatic adenocarcinoma. Theranostics 2019, 9, 436–448. [Google Scholar] [CrossRef] [PubMed]
- Galligan, L.; Longley, D.B.; McEwan, M.; Wilson, T.R.; McLaughlin, K.; Johnston, P.G. Chemotherapy and TRAIL-mediated colon cancer cell death: The roles of p53, TRAIL receptors, and c-FLIP. Mol. Cancer Ther. 2005, 4, 2026–2036. [Google Scholar] [CrossRef] [PubMed]
- Greenlee, J.D.; Lopez-cavestany, M.; Ortiz-otero, N.; Liu, K.; Subramanian, T.; Cagir, B.; King, M.R.; States, U. Oxaliplatin resistance in colorectal cancer enhances TRAIL sensitivity via death receptor 4 upregulation and lipid raft localization. Elife 2021, 10, e67750. [Google Scholar] [CrossRef]
- Park, S.A.; Han, H.R.; Ahn, S.; Ryu, C.H.; Jeun, S.S. Combination treatment with VPA and MSCs-TRAIL could increase anti-tumor effects against intracranial glioma. Oncol. Rep. 2021, 45, 869–878. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, D.; Atkin, W.; Lenz, H.J.; Lynch, H.T.; Minsky, B.; Nordlinger, B.; Starling, N. Colorectal cancer. Lancet 2010, 375, 1030–1047. [Google Scholar] [CrossRef] [PubMed]
- Quiroz-Reyes, A.G.; Gonzalez-Villarreal, C.A.; Limon-Flores, A.Y.; Delgado-Gonzalez, P.; Martinez-Rodriguez, H.G.; Said-Fernandez, S.L.; Soto-Dominguez, A.; Rivas-Estilla, A.M.; Islas, J.F.; Molina-De la Garza, J.F.; et al. Mesenchymal Stem Cells Genetically Modified by Lentivirus-Express Soluble TRAIL and Interleukin-12 Inhibit Growth and Reduced Metastasis-Relate Changes in Lymphoma Mice Model. Biomedicines 2023, 11, 595. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Aparicio, C.; Belver, M.; Enríquez, L.; Espeso, F.; Núñez, L.; Sánchez, A.; de la Fuente, M.Á.; González-vallinas, M. Cell therapy for colorectal cancer: The promise of chimeric antigen receptor (CAR)-T cells. Int. J. Mol. Sci. 2021, 22, 11781. [Google Scholar] [CrossRef]
- Do, B.H.; Nguyen, M.T.; Song, J.-A.; Park, S.; Yoo, J.; Jang, J.; Lee, S.; So, S.; Yoon, Y.; Kim, I.; et al. Soluble Prokaryotic Expression and Purification of Bioactive Tumor Necrosis Factor-Related Apoptosis-Inducing Ligand. J. Microbiol. Biotechnol. 2017, 27, 2156–2164. [Google Scholar] [CrossRef]
- Spano, C.; Grisendi, G.; Golinelli, G.; Rossignoli, F.; Prapa, M.; Bestagno, M.; Candini, O.; Petrachi, T.; Recchia, A.; Miselli, F.; et al. Soluble TRAIL Armed Human MSC As Gene Therapy For Pancreatic Cancer. Sci. Rep. 2019, 9, 1788. [Google Scholar] [CrossRef]
- Chen, F.; Zhong, X.; Dai, Q.; Li, K.; Zhang, W.; Wang, J.; Zhao, Y.; Shen, J.; Xiao, Z.; Xing, H.; et al. Human Umbilical Cord MSC Delivered-Soluble TRAIL Inhibits the Proliferation and Promotes Apoptosis of B-ALL Cell In Vitro and In Vivo. Pharmaceuticals 2022, 15, 1391. [Google Scholar] [CrossRef] [PubMed]
- Abatay-Sel, F.; Erol, A.; Suleymanoglu, M.; Demirayak, G.; Kekik-Cinar, C.; Kuruca, D.S.; Savran-Oguz, F. The in vitro treatment of mesenchymal stem cells for colorectal cancer cells. Med. Oncol. 2023, 40, 103. [Google Scholar] [CrossRef] [PubMed]
- González-Villareal, C.; Martínez-rodríguez, H.G.; Said, S. How desirable and undesirable features of naϊve or genetically reengineered mesenchymal stem cells are being considered in preclinical or clinical assays. J. BUON 2017, 22, 812–830. [Google Scholar]
- Xu, X.; Song, J. Segmental long bone regeneration guided by degradable synthetic polymeric scaffolds. Biomater. Transl. 2020, 1, 33–45. [Google Scholar] [CrossRef]
- Golinelli, G.; Mastrolia, I.; Aramini, B.; Masciale, V.; Pinelli, M.; Pacchioni, L.; Casari, G.; Dall’Ora, M.; Soares, M.B.P.; Damasceno, P.K.F.; et al. Arming Mesenchymal Stromal/Stem Cells Against Cancer: Has the Time Come? Front. Pharmacol. 2020, 11, 529921. [Google Scholar] [CrossRef]
- Fakiruddin, K.S.; Lim, M.N.; Nordin, N.; Rosli, R.; Zakaria, Z.; Abdullah, S. Targeting of CD133+ cancer stem cells by mesenchymal stem cell expressing TRAIL reveals a prospective role of apoptotic gene regulation in non-small cell lung cancer. Cancers 2019, 11, 1261. [Google Scholar] [CrossRef]
- van Geelen, C.M.; Pennarun, B.; Le, P.T.; de Vries, E.G.; de Jong, S. Modulation of TRAIL resistance in colon carcinoma cells: Different contributions of DR4 and DR5. BMC Cancer 2011, 11, 39. [Google Scholar] [CrossRef]
- Kopczynski, M.; Statkiewicz, M.; Cybulska, M.; Kuklinska, U.; Unrug-bielawska, K.; Sandowska-markiewicz, Z.; Grochowska, A.; Gajewska, M.; Kulecka, M.; Ostrowski, J.; et al. Cytotoxic efficacy and resistance mechanism of a trail and vegfa-peptide fusion protein in colorectal cancer models. Int. J. Mol. Sci. 2021, 22, 3160. [Google Scholar] [CrossRef] [PubMed]
- Luetzkendorf, J.; Mueller, L.P.; Mueller, T.; Caysa, H.; Nerger, K.; Schmoll, H.-J. Growth inhibition of colorectal carcinoma by lentiviral TRAIL-transgenic human mesenchymal stem cells requires their substantial intratumoral presence. J. Cell. Mol. Med. 2010, 14, 2292–2304. [Google Scholar] [CrossRef]
- Aparicio, J.; Esposito, F.; Serrano, S.; Falco, E.; Escudero, P.; Ruiz-Casado, A.; Manzano, H.; Fernandez-Montes, A. Metastatic Colorectal Cancer. First Line Therapy for Unresectable Disease. J. Clin. Med. 2020, 9, 3889. [Google Scholar] [CrossRef]
- Liu, Y.; Zheng, C.; Huang, Y.; He, M.; Xu, W.W.; Li, B. Molecular mechanisms of chemo- and radiotherapy resistance and the potential implications for cancer treatment. MedComm 2021, 2, 315–340. [Google Scholar] [CrossRef]
- Fakiruddin, K.S.; Lim, M.N.; Nordin, N.; Rosli, R.; Abdullah, S. Chemo-Sensitization of CD133+ Cancer Stem Cell Enhances the Effect of Mesenchymal Stem Cell Expressing TRAIL in Non-Small Cell Lung Cancer Cell Lines. Biology 2021, 10, 1103. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Fanjul, V.; Guerrero-López, R.; Fernández-Varas, B.; Perona, R.; Sastre-Perona, A.; Sastre, L. Comparison of Colorectal Cancer Stem Cells and Oxaliplatin-Resistant Cells Unveils Functional Similarities. Cells 2022, 11, 511. [Google Scholar] [CrossRef] [PubMed]
- Igura, K.; Zhang, X.; Takahashi, K.; Mitsuru, A.; Yamaguchi, S.; Takahashi, T.A. Isolation and characterization of mesenchymal progenitor cells from chorionic villi of human placenta. Cytotherapy 2004, 6, 543–553. [Google Scholar] [CrossRef]
- Quiroz-Reyes, A.G.; González-Villarreal, C.A.; Martínez-Rodriguez, H.; Said-Fernández, S.; Salinas-Carmona, M.C.; Limón-Flores, A.Y.; Soto-Domínguez, A.; Padilla-Rivas, G.; De Oca-Luna, R.M.; Islas, J.F.; et al. A combined antitumor strategy of separately transduced mesenchymal stem cells with soluble TRAIL and IFNβ produces a synergistic activity in the reduction of lymphoma and mice survival enlargement. Mol. Med. Rep. 2022, 25, 206. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Xu, C.; Luo, Y.; Liu, L.; Song, K.; Deng, G.; Yang, M.; Cao, J.; Yuan, L.; Li, X. Disruption of crosstalk between LX-2 and liver cancer stem-like cells from MHCC97H cells by DFOG via inhibiting FOXM1. Acta Biochim. Biophys. Sin. 2019, 51, 1267–1275. [Google Scholar] [CrossRef] [PubMed]
| 5-Fluorouracil | Oxaliplatin | Irinotecan | Reference | ||||
|---|---|---|---|---|---|---|---|
| IC50 (µg/mL) | Max Plasma (µg/mL) | IC50 (µg/mL) | Max Plasma (µg/mL) | IC50 (µg/mL) | Max Plasma (µg/mL) | ||
| CACO-2 | 1.66 | 10 | 4.0 | 2.9 | 2.14 | 1.97 | [6] |
| CMT-93 | 0.096 | 10 | 0.15 | 2.9 | 1.33 | 1.97 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quiroz-Reyes, A.G.; Delgado-González, P.; Islas, J.F.; Soto-Domínguez, A.; González-Villarreal, C.A.; Padilla-Rivas, G.R.; Garza-Treviño, E.N. Oxaliplatin Enhances the Apoptotic Effect of Mesenchymal Stem Cells, Delivering Soluble TRAIL in Chemoresistant Colorectal Cancer. Pharmaceuticals 2023, 16, 1448. https://doi.org/10.3390/ph16101448
Quiroz-Reyes AG, Delgado-González P, Islas JF, Soto-Domínguez A, González-Villarreal CA, Padilla-Rivas GR, Garza-Treviño EN. Oxaliplatin Enhances the Apoptotic Effect of Mesenchymal Stem Cells, Delivering Soluble TRAIL in Chemoresistant Colorectal Cancer. Pharmaceuticals. 2023; 16(10):1448. https://doi.org/10.3390/ph16101448
Chicago/Turabian StyleQuiroz-Reyes, Adriana G, Paulina Delgado-González, José F. Islas, Adolfo Soto-Domínguez, Carlos A. González-Villarreal, Gerardo R. Padilla-Rivas, and Elsa N. Garza-Treviño. 2023. "Oxaliplatin Enhances the Apoptotic Effect of Mesenchymal Stem Cells, Delivering Soluble TRAIL in Chemoresistant Colorectal Cancer" Pharmaceuticals 16, no. 10: 1448. https://doi.org/10.3390/ph16101448
APA StyleQuiroz-Reyes, A. G., Delgado-González, P., Islas, J. F., Soto-Domínguez, A., González-Villarreal, C. A., Padilla-Rivas, G. R., & Garza-Treviño, E. N. (2023). Oxaliplatin Enhances the Apoptotic Effect of Mesenchymal Stem Cells, Delivering Soluble TRAIL in Chemoresistant Colorectal Cancer. Pharmaceuticals, 16(10), 1448. https://doi.org/10.3390/ph16101448








