Successful Treatment of a Recalcitrant Staphylococcus epidermidis Prosthetic Knee Infection with Intraoperative Bacteriophage Therapy
Abstract
1. Introduction
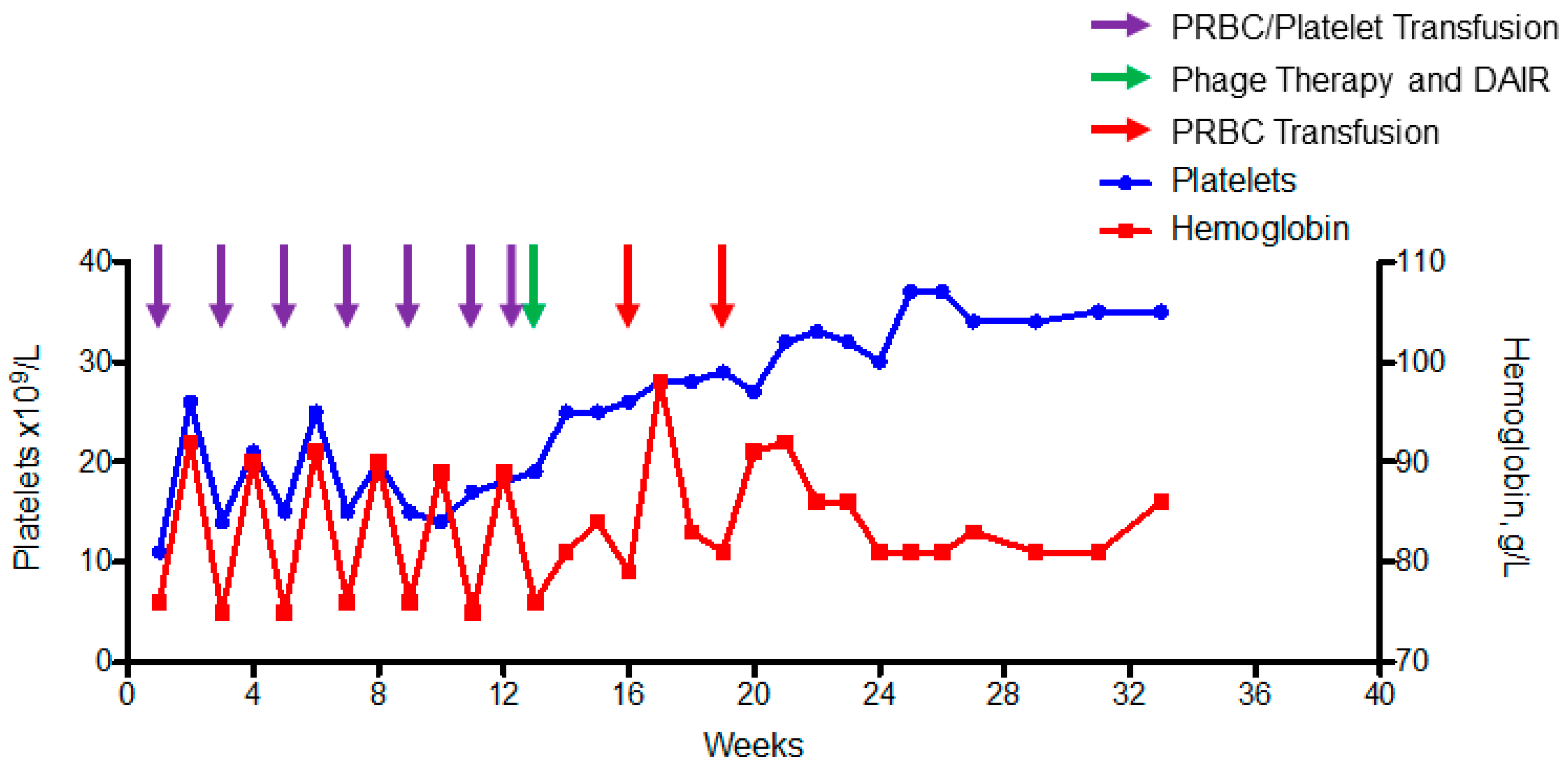
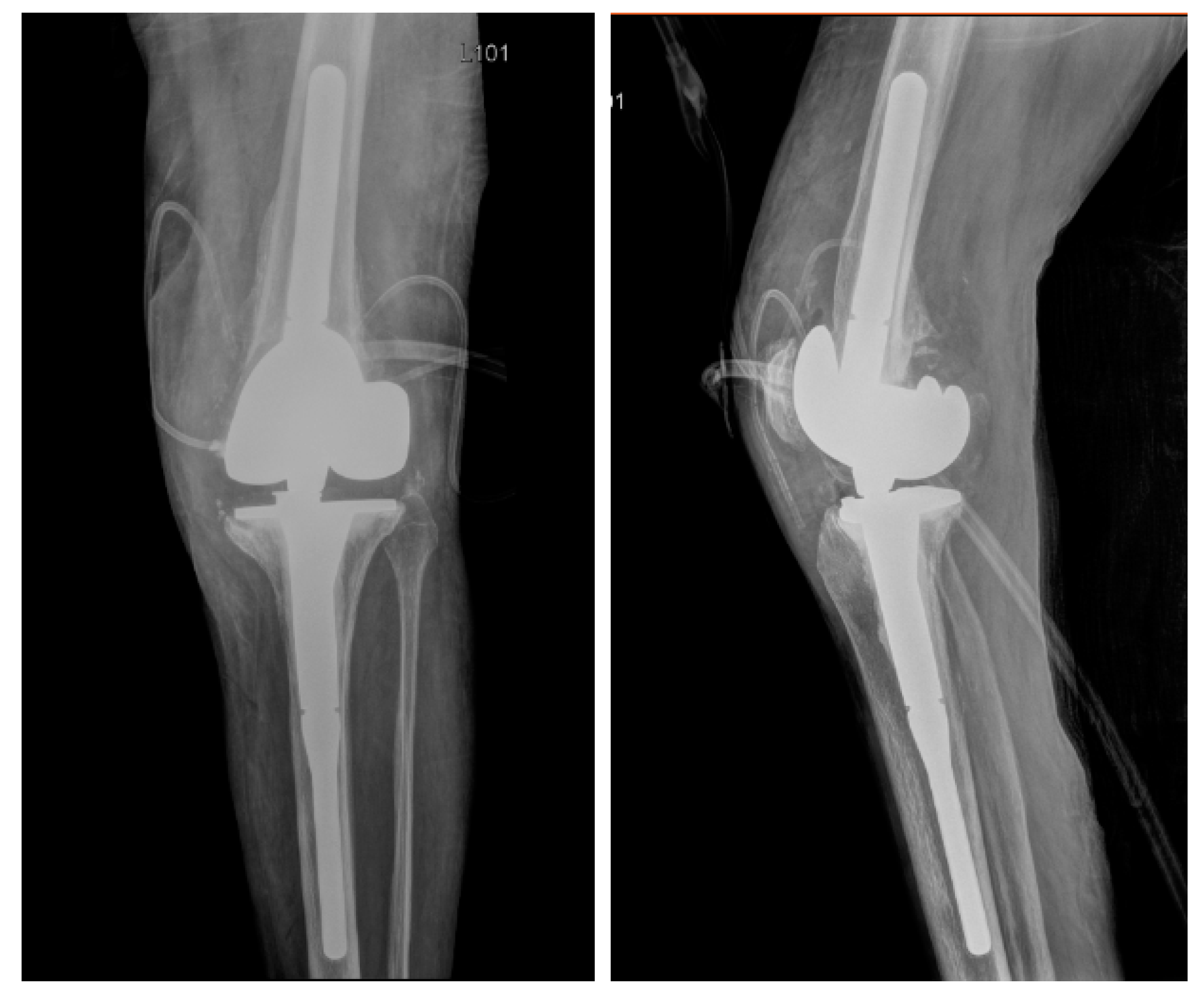
2. Case
3. Methods
3.1. Bacterial Isolation
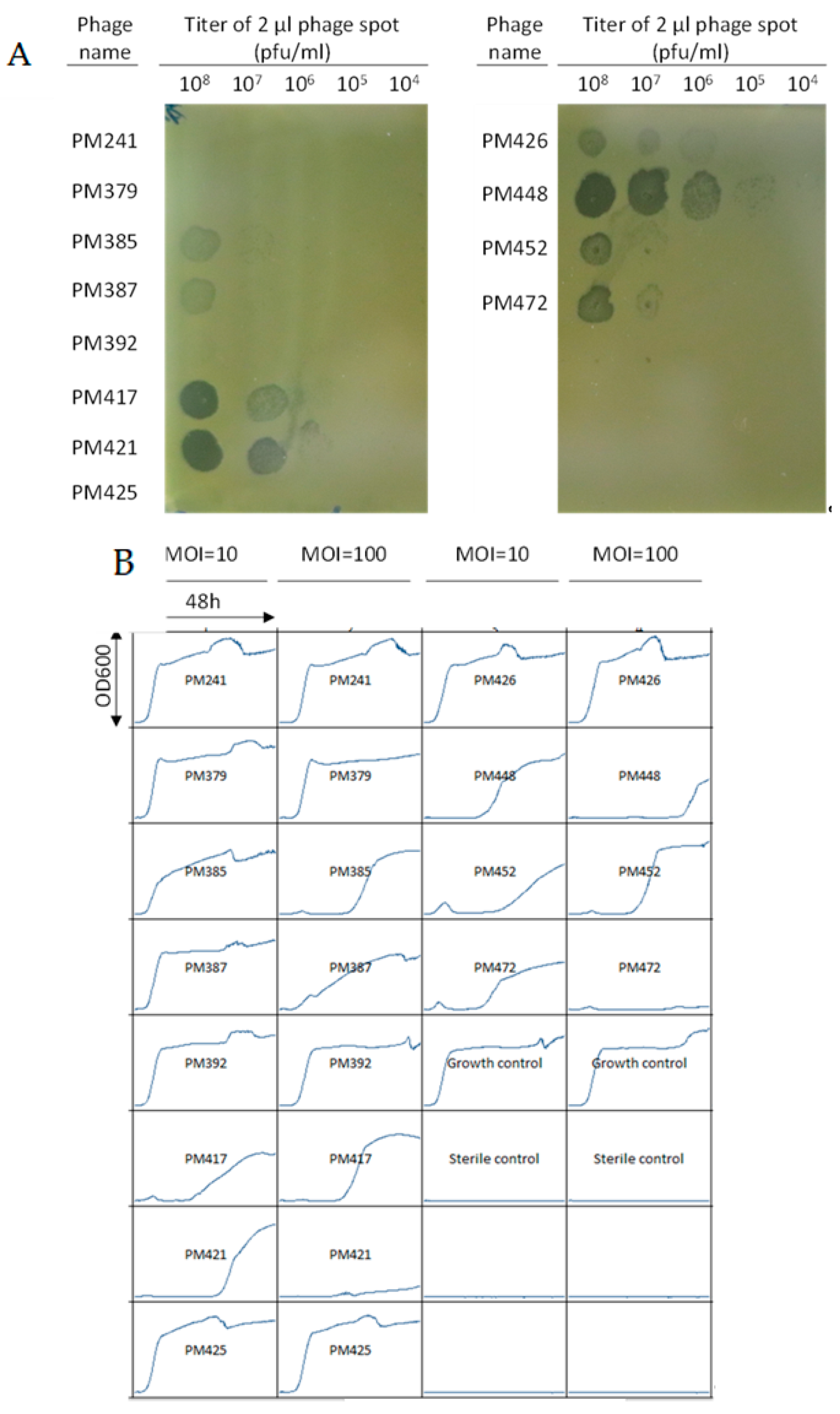
3.2. Bacteriophage Screening
3.3. Phage Amplification, Purification and Quality Control Testing
3.4. DAIR Procedure and Bacteriophage Administration
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Natsuhara, K. Mortality During Total Hip Periprosthetic Joint Infection. Arthroplasty 2019, 34, S337–S342. [Google Scholar] [CrossRef]
- Xu, C.; Goswami, K.; Li, W.T.; Tan, T.L.; Yayac, M.; Wang, S.H.; Parvizi, J. Is Treatment of Periprosthetic Joint Infection Improving Over Time? J. Arthroplast. 2020, 35, 1696–1702. [Google Scholar] [CrossRef] [PubMed]
- Doub, J.B.; Ng, V.Y.; Johnson, A.J.; Slomka, M.; Fackler, J.; Horne, B.; Brownstein, M.J.; Henry, M.; Malagon, F.; Biswas, B. Salvage Bacteriophage Therapy for a Chronic MRSA Prosthetic Joint Infection. Antibiotics 2020, 9, 241. [Google Scholar] [CrossRef]
- Vadiee, I.; Backstein, D.J. The Effectiveness of Repeat Two-Stage Revision for the Treatment of Recalcitrant Total Knee Arthroplasty Infection. J. Arthroplast. 2019, 34, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Fehring, K.A.; Abdel, M.P.; Ollivier, M.; Mabry, T.M.; Hanssen, A.D. Repeat Two-Stage Exchange Arthroplasty for Periprosthetic Knee Infection Is Dependent on Host Grade. J. Bone Jt. Surg. Am. 2017, 4, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Ferry, T.; Kolenda, C.; Batailler, C.; Gustave, C.A.; Lustig, S.; Malatray, M.; Fevre, C.; Josse, J.; Petitjean, C.; Chidiac, C.; et al. Phage Therapy as Adjuvant to Conservative Surgery and Antibiotics to Salvage Patients with Relapsing S. aureus Prosthetic Knee Infection. Front. Med. 2020, 7, 570572. [Google Scholar] [CrossRef]
- Onsea, J.; Soentjens, P.; Djebara, S.; Merabishvili, M.; Depypere, M.; Spriet, I.; De Munter, P.; Debaveye, Y.; Nijs, S.; Vanderschot, P.; et al. Bacteriophage Application for Difficult-to-treat Musculoskeletal Infections: Development of a Standardized Multidisciplinary Treatment Protocol. Viruses 2019, 11, 891. [Google Scholar] [CrossRef]
- Tkhilaishvili, T.; Winkler, T.; Müller, M.; Perka, C.; Trampuz, A. Bacteriophages as Adjuvant to Antibiotics for the Treatment of Periprosthetic Joint Infection Caused by Multidrug-Resistant Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2019, 64, e00924-19. [Google Scholar] [CrossRef]
- Abedon, S.T. Ecology of Anti-Biofilm Agents II: Bacteriophage Exploitation and Biocontrol of Biofilm Bacteria. Pharmaceuticals 2015, 8, 559–589. [Google Scholar] [CrossRef]
- Hanlon, G.W. Bacteriophages: An appraisal of their role in the treatment of bacterial infections. Int. J. Antimicrob. Agents 2007, 30, 118–128. [Google Scholar] [CrossRef]
- Fagotti, L.; Tatka, J.; Salles, M.J.C.; Queiroz, M.C. Risk Factors and Treatment Options for Failure of a Two-Stage Exchange. Curr. Rev. Musculoskelet. Med. 2018, 11, 420–427. [Google Scholar] [CrossRef]
- Lee, H.D.; Prashant, K.; Shon, W.Y. Management of Periprosthetic Hip Joint Infection. Hip Pelvis 2015, 27, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Qasim, S.N.; Swann, A.; Ashford, R. The DAIR (debridement, antibiotics and implant retention) procedure for infected total knee—A literature review. SICOT J. 2017, 3, 2. [Google Scholar] [CrossRef]
- Osmon, D.R.; Berbari, E.F.; Berendt, A.R.; Lew, D.; Zimmerli, W.; Steckelberg, J.M.; Rao, N.; Hanssen, A.; Wilson, W.R. Infectious Diseases Society of America. Executive summary: Diagnosis and management of prosthetic joint infection: Clinical practice guidelines by the Infectious Diseases Society of America. Clin. Infect. Dis. 2013, 56, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Chan, B.K.; Abedon, S.T. Bacteriophages and their enzymes in biofilm control. Curr. Pharm. Des. 2015, 21, 85–99. [Google Scholar] [CrossRef]
- Doub, J.B. Bacteriophage Therapy for Clinical Biofilm Infections: Parameters That Influence Treatment Protocols and Current Treatment Approaches. Antibiotics 2020, 12, 799. [Google Scholar] [CrossRef] [PubMed]
- Sulakvelidze, A.; Alavidze, Z.; Morris, J.G., Jr. Bacteriophage therapy. Antimicrob. Agents Chemother. 2001, 45, 649–659. [Google Scholar] [CrossRef] [PubMed]
- Vaz, K.; Scarborough, M.; Bottomley, N.; Kendrick, B.; Taylor, A.; Price, A.; Alvand, A.; Jackson, W. Debridement, antibiotics and implant retention (DAIR) for the management of knee prosthetic joint infection. Knee 2020, 27, 2013–2015. [Google Scholar] [CrossRef]
- Ryan, E.M.; Alkawareek, M.Y.; Donnelly, R.F.; Gilmore, B.F. Synergistic phage-antibiotic combinations for the control of Escherichia coli biofilms in vitro. FEMS Immunol. Med. Microbiol. 2012, 65, 395–398. [Google Scholar] [CrossRef]
- Yazdi, M.; Bouzari, M.; Ghaemi, E.A. Isolation and Characterization of a Lytic Bacteriophage (vB_PmiS-TH) and Its Application in Combination with Ampicillin against Planktonic and Biofilm Forms of Proteus mirabilis Isolated from Urinary Tract Infection. J. Mol. Microbiol. Biotechnol. 2018, 28, 37–46. [Google Scholar] [CrossRef]
- Mendes, J.J.; Leandro, C.; Corte-Real, S.; Barbosa, R.; Cavaco-Silva, P.; Melo-Cristino, J.; Górski, A.; Garcia, M. Wound healing potential of topical bacteriophage therapy on diabetic cutaneous wounds. Wound Repair Regen. 2013, 21, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Fabijan, A.P.; Lin, R.C.Y.; Ho, J.; Maddocks, S.; Ben Zakour, N.L.; Iredell, J.R.; Westmead Bacteriophage Therapy Team. Safety of bacteriophage therapy in severe Staphylococcus aureus infection. Nat. Microbiol. 2020, 5, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Young, N.S.; Calado, R.T.; Scheinberg, P. Current concepts in the pathophysiology and treatment of aplastic anemia. Blood 2006, 15, 2509–2519. [Google Scholar] [CrossRef]
- Bi, L.; Li, J.; Lu, Z.; Shao, H.; Wang, Y. Acute arrest of hematopoiesis induced by infection with Staphylococcus epidermidis following total knee arthroplasty: A case report and literature review. Exp. Ther. Med. 2016, 11, 957–960. [Google Scholar] [CrossRef]
- Spika, J.S.; Peterson, P.K.; Wilkinson, B.J.; Hammerschmidt, D.E.; Verbrugh, H.A.; Verhoef, J.; Quie, P.G. Role of peptidoglycan from Staphylococcus aureus in leukopenia, thrombocytopenia, and complement activation associated with bacteremia. J. Infect. Dis. 1982, 146, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Cano, E.J.; Caflisch, K.M.; Bollyky, P.L.; Van Belleghem, J.D.; Patel, R.; Fackler, J.; Brownstein, M.J.; Horne, B.; Biswas, B.; Henry, M.; et al. Phage Therapy for Limb-threatening Prosthetic Knee Klebsiella pneumoniae Infection: Case Report and In Vitro Characterization of Anti-biofilm Activity. Clin. Infect. Dis. 2020, 23, 705. [Google Scholar] [CrossRef]
| Antibiotic | MIC µg/mL | Interpretation |
|---|---|---|
| Cefazolin | NA | Resistant |
| Ciprofloxacin | ˃2 | Resistant |
| Daptomycin | ≤0.25 | Sensitive |
| Erythromycin | ˃4 | Resistant |
| Gentamicin | ≤1 | Sensitive |
| Linezolid | 1 | Sensitive |
| Oxacillin | ˃2 | Resistant |
| Rifampin | ≤1 | Sensitive |
| Tetracycline | ≤2 | Sensitive |
| TMS | ˃2/38 | Resistant |
| Vancomycin | 1 | Sensitive |
| Phage ID | Titer (PFU/mL) | Endotoxin (EU/dose) | USP <71> Sterility Testing |
|---|---|---|---|
| PM448 | 1 × 1010 | <1 | PASSED |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Doub, J.B.; Ng, V.Y.; Wilson, E.; Corsini, L.; Chan, B.K. Successful Treatment of a Recalcitrant Staphylococcus epidermidis Prosthetic Knee Infection with Intraoperative Bacteriophage Therapy. Pharmaceuticals 2021, 14, 231. https://doi.org/10.3390/ph14030231
Doub JB, Ng VY, Wilson E, Corsini L, Chan BK. Successful Treatment of a Recalcitrant Staphylococcus epidermidis Prosthetic Knee Infection with Intraoperative Bacteriophage Therapy. Pharmaceuticals. 2021; 14(3):231. https://doi.org/10.3390/ph14030231
Chicago/Turabian StyleDoub, James B., Vincent Y. Ng, Eleanor Wilson, Lorenzo Corsini, and Benjamin K. Chan. 2021. "Successful Treatment of a Recalcitrant Staphylococcus epidermidis Prosthetic Knee Infection with Intraoperative Bacteriophage Therapy" Pharmaceuticals 14, no. 3: 231. https://doi.org/10.3390/ph14030231
APA StyleDoub, J. B., Ng, V. Y., Wilson, E., Corsini, L., & Chan, B. K. (2021). Successful Treatment of a Recalcitrant Staphylococcus epidermidis Prosthetic Knee Infection with Intraoperative Bacteriophage Therapy. Pharmaceuticals, 14(3), 231. https://doi.org/10.3390/ph14030231






