Dextran-Curcumin Nanoparticles as a Methotrexate Delivery Vehicle: A Step Forward in Breast Cancer Combination Therapy
Abstract
1. Introduction
2. Results
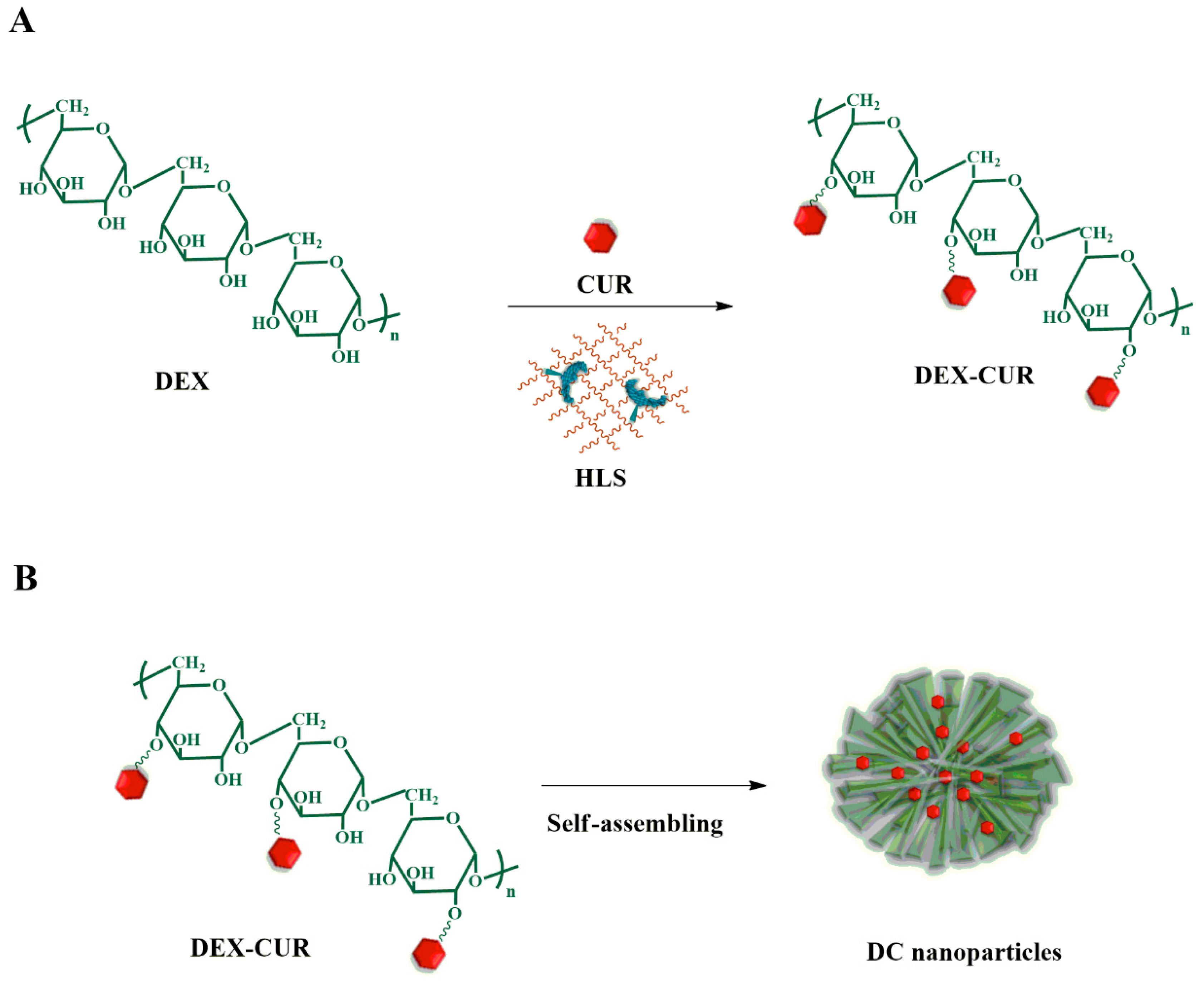
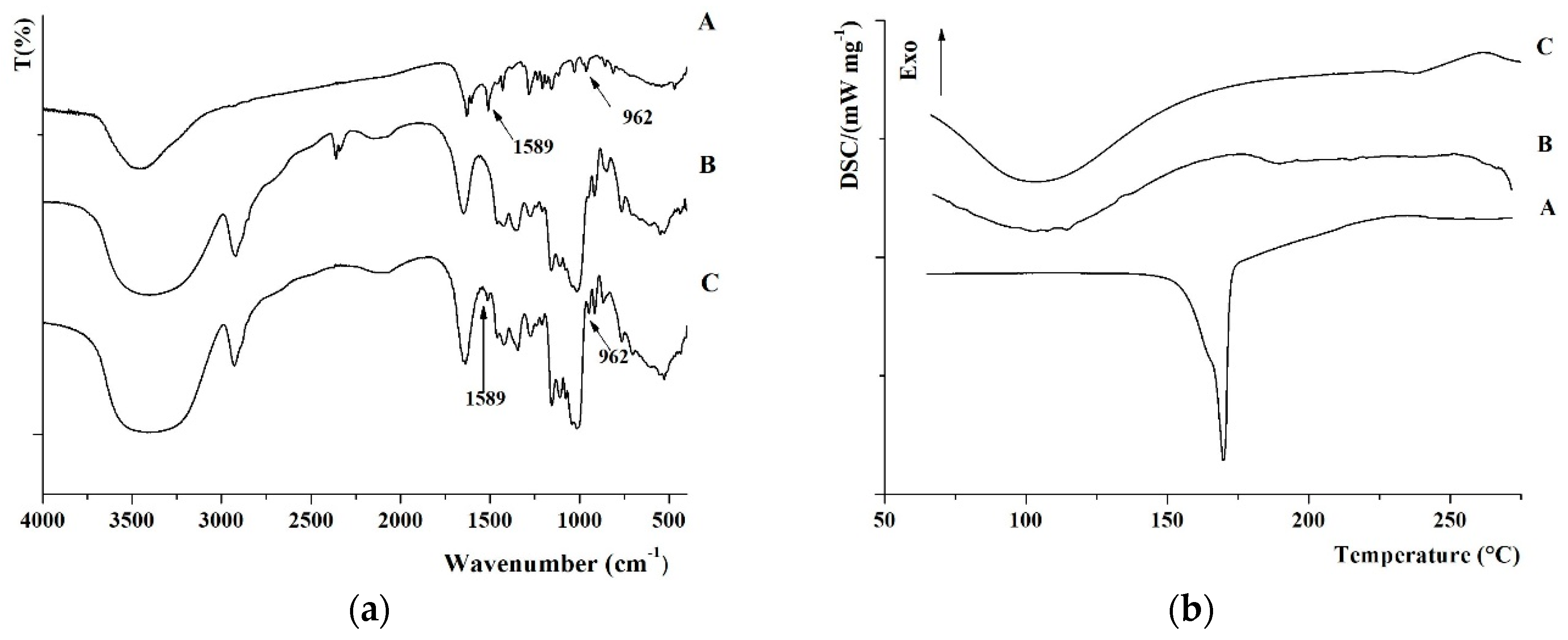
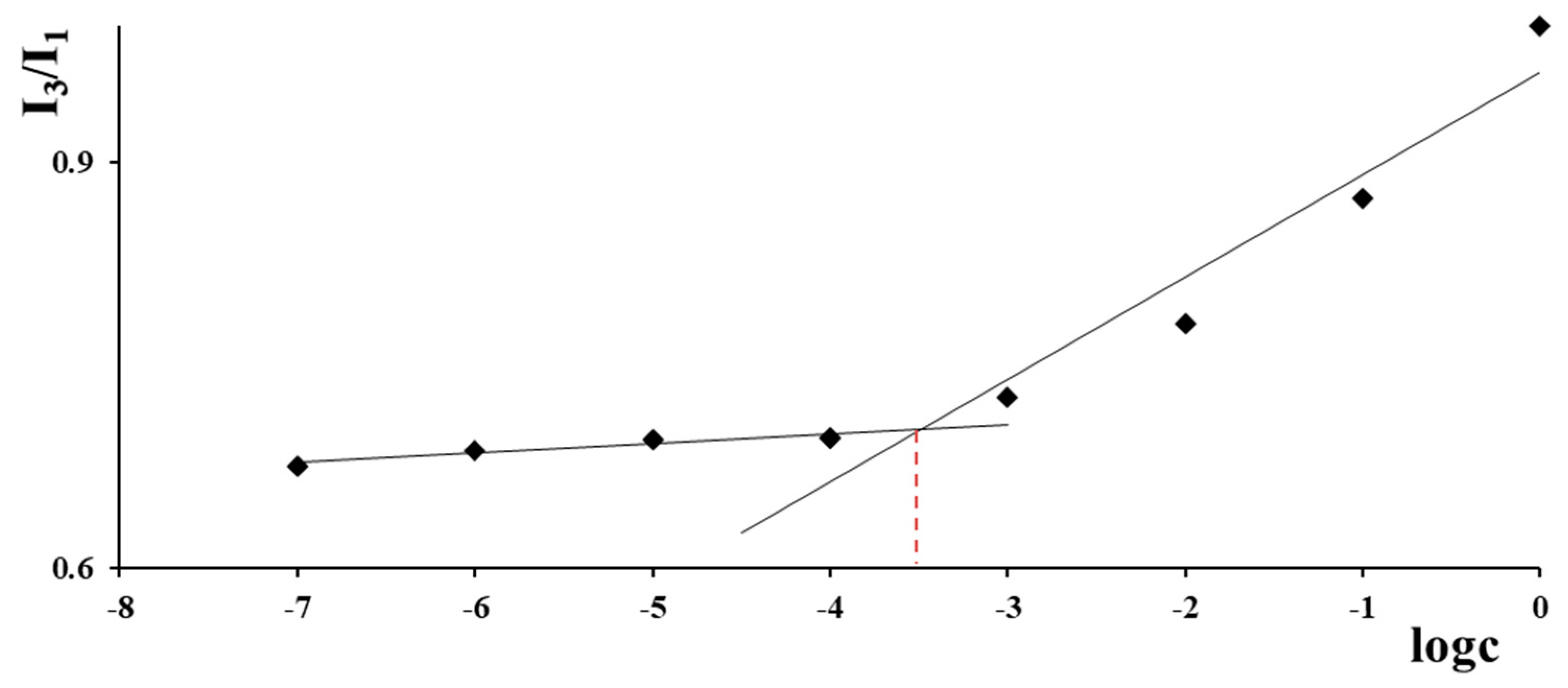
2.1. Synthesis and Characterization of Nanoparticles
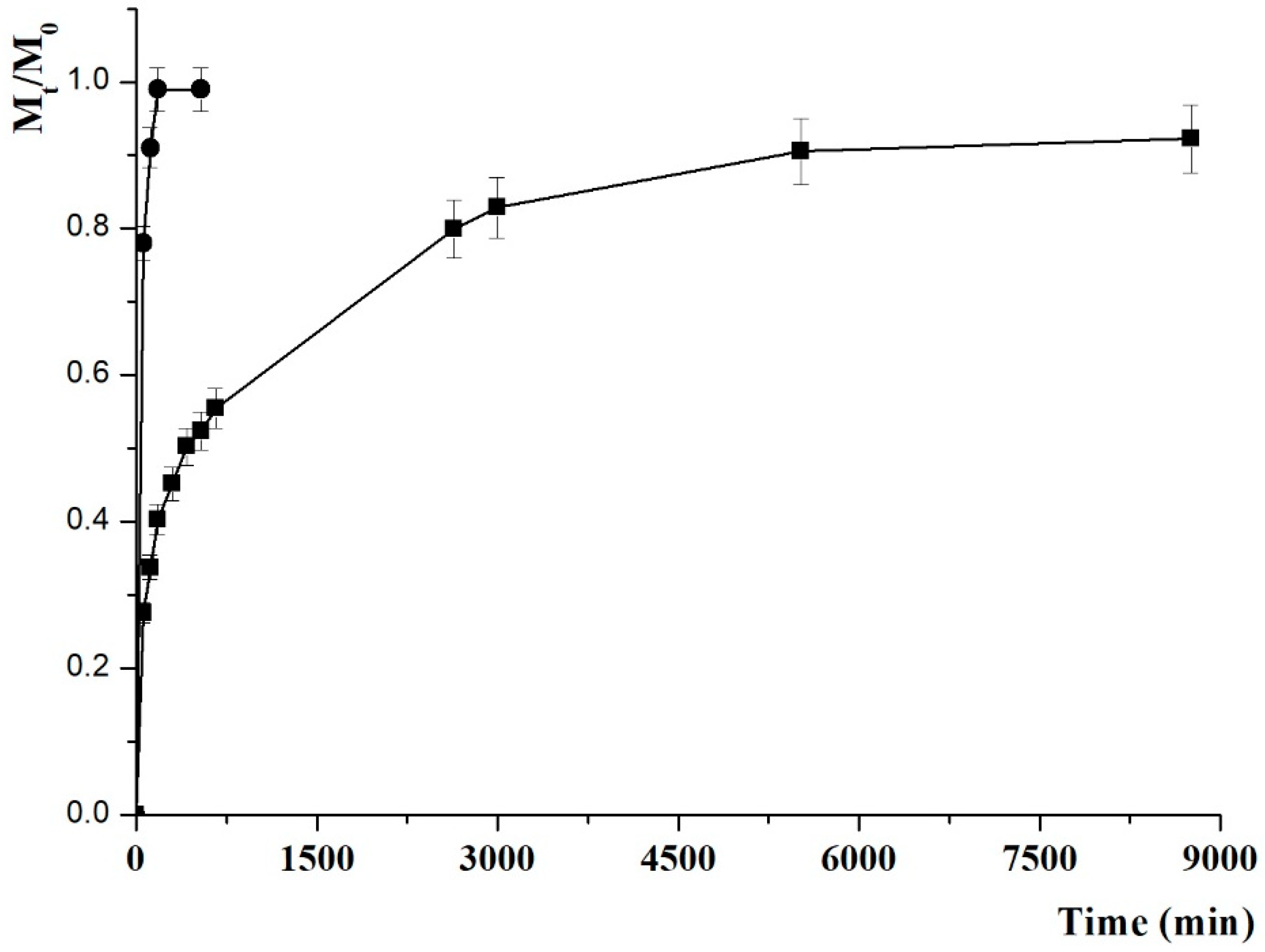
2.2. Drug Release Experiments
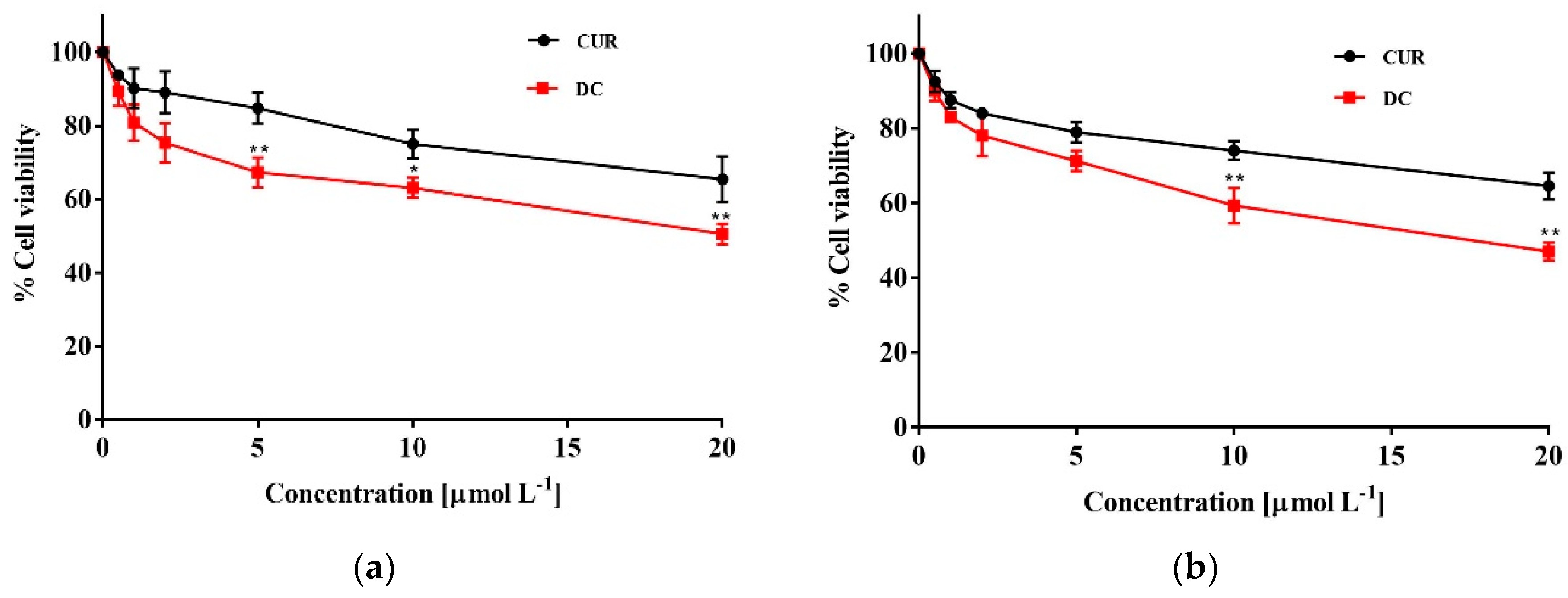
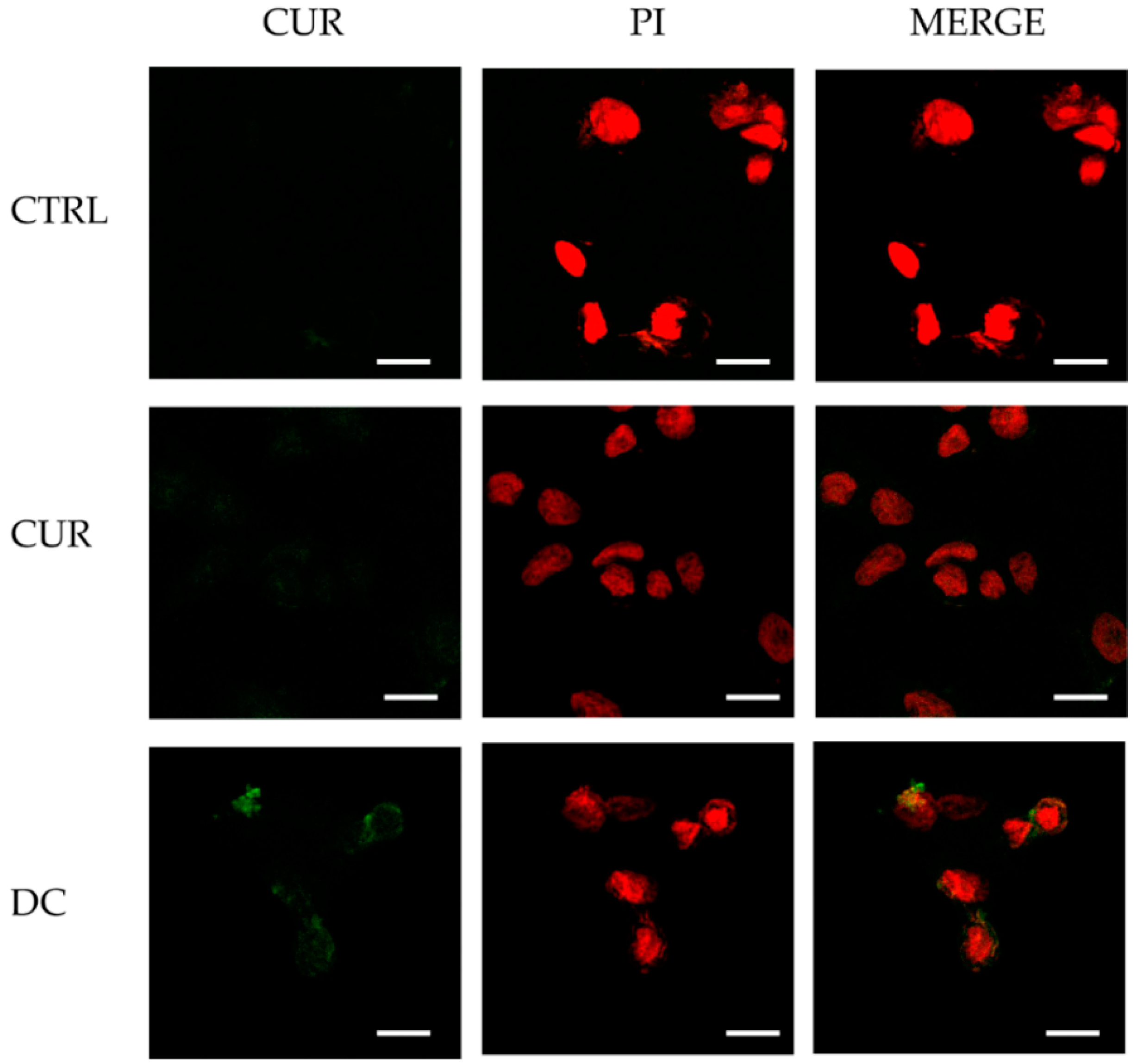
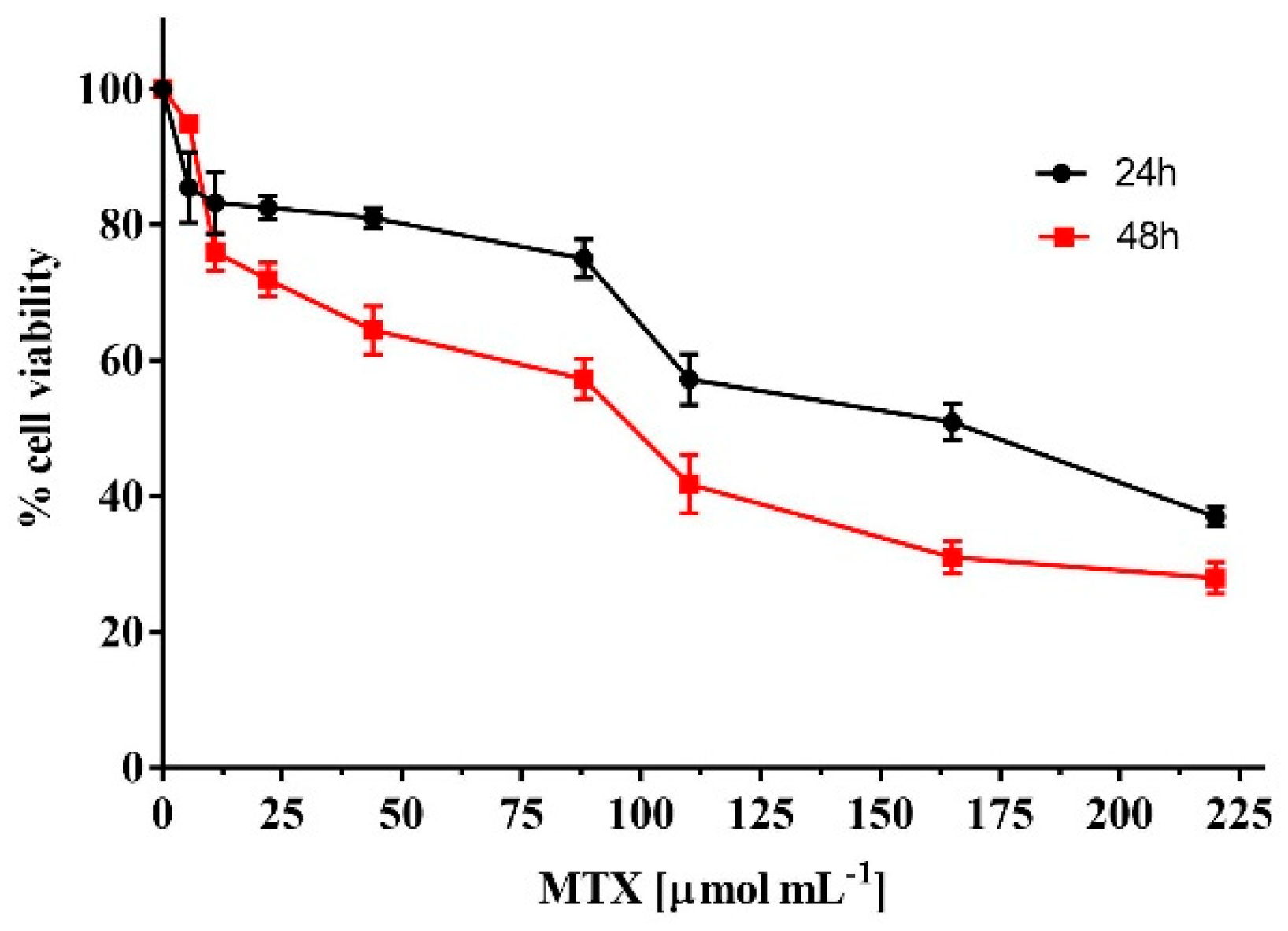
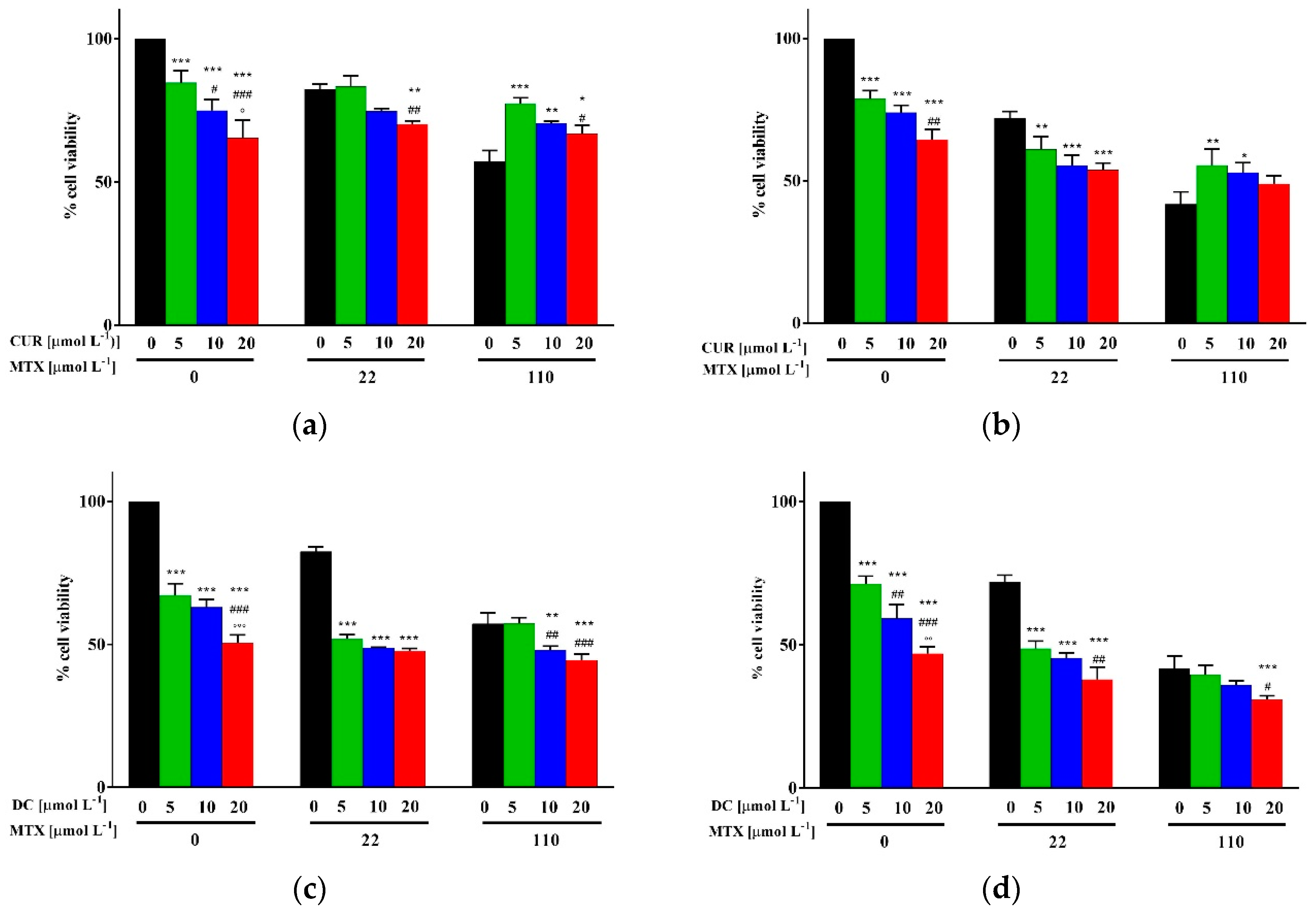
2.3. Biological Characterization
3. Materials and Methods
3.1. Synthesis of Curcumin Conjugate
3.2. Characterization of Conjugate
3.3. Preparation of Nanoparticles and Methotrexate Loading
3.4. In Vitro Drug Release
3.5. Cell Culture
3.6. Viability Assay
3.7. Staining for Confocal Microscopy
3.8. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mokhtari, R.B.; Homayouni, T.S.; Baluch, N.; Morgatskaya, E.; Kumar, S.; Das, B.; Yeger, H. Combination therapy in combating cancer. Oncotarget 2017, 8, 38022–38043. [Google Scholar] [CrossRef] [PubMed]
- Garattini, S. New approaches to cancer therapy. Ann. Oncol. 2003, 14, 813–816. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Che, S.M.; Hui, B.N.; Yang, Y.Y.; Wang, X.L.; Zhang, X.Z.; Qiang, Y.Q.; Ma, H.L. Lung cancer therapy using doxorubicin and curcumin combination: Targeted prodrug based, pH sensitive nanomedicine. Biomed. Pharmacother. 2019, 112, 108614. [Google Scholar] [CrossRef] [PubMed]
- Dou, X.Q.; Wang, H.; Zhang, J.; Wang, F.; Xu, G.L.; Xu, C.C.; Xu, H.H.; Xiang, S.S.; Fu, J.; Song, H.F. Aptamer-drug conjugate: Targeted delivery of doxorubicin in a HER3 aptamer-functionalized liposomal delivery system reduces cardiotoxicity. Int. J. Nanomed. 2018, 13, 763–776. [Google Scholar] [CrossRef]
- Mo, L.H.; Pospichalova, V.; Huang, Z.Q.; Murphy, S.K.; Payne, S.; Wang, F.; Kennedy, M.; Cianciolo, G.J.; Bryja, V.; Pizzo, S.V.; et al. Ascites Increases Expression/Function of Multidrug Resistance Proteins in Ovarian Cancer Cells. PLoS ONE 2015, 10, e013157. [Google Scholar] [CrossRef]
- Li, M.Q.; Tang, Z.H.; Lin, J.; Zhang, Y.; Lv, S.X.; Song, W.T.; Huang, Y.B.; Chen, X.S. Synergistic Antitumor Effects of Doxorubicin-Loaded Carboxymethyl Cellulose Nanoparticle in Combination with Endostar for Effective Treatment of Non-Small-Cell Lung Cancer. Adv. Healthc. Mater. 2014, 3, 1877–1888. [Google Scholar] [CrossRef]
- Santiago, T.; DeVaux, R.S.; Kurzatkowska, K.; Espinal, R.; Herschkowitz, J.I.; Hepel, M. Surface-enhanced Raman scattering investigation of targeted delivery and controlled release of gemcitabine. Int. J. Nanomed. 2017, 12, 7763–7776. [Google Scholar] [CrossRef]
- Khalid, K.M. A combination therapy of atypical protein kinase inhibitor and phosphatidylinositol-3-kinase inhibitor reduces multidrug resistance in renal cell carcinoma (RCC). Cancer Res. 2019, 79. [Google Scholar] [CrossRef]
- Yin, Q.; Shen, J.A.; Zhang, Z.W.; Yu, H.J.; Chen, L.L.; Gu, W.W.; Li, Y.P. Multifunctional Nanoparticles Improve Therapeutic Effect for Breast Cancer by Simultaneously Antagonizing Multiple Mechanisms of Multidrug Resistance. Biomacromolecules 2013, 14, 2242–2252. [Google Scholar] [CrossRef]
- Thota, S.; Rodrigues, D.A.; Barreiro, E.J. Recent Advances in Development of Polyphenols as Anticancer Agents. Mini-Rev. Med. Chem. 2018, 18, 1265–1269. [Google Scholar] [CrossRef]
- Landis-Piwowar, K.R.; Huo, C.D.; Chen, D.; Milacic, V.; Shi, G.Q.; Chan, T.H.; Dou, Q.P. A novel prodrug of the green tea polyphenol (−)-epigallocatechin-3-gallate as a potential anticancer agent. Cancer Res. 2007, 67, 4303–4310. [Google Scholar] [CrossRef] [PubMed]
- Asensi, M.; Ortega, A.; Mena, S.; Feddi, F.; Estrela, J.M. Natural polyphenols in cancer therapy. Crit. Rev. Clin. Lab. Sci. 2011, 48, 197–216. [Google Scholar] [CrossRef] [PubMed]
- Santos, I.S.; Ponte, B.M.; Boonme, P.; Silva, A.M.; Souto, E.B. Nanoencapsulation of polyphenols for protective effect against colon-rectal cancer. Biotechnol. Adv. 2013, 31, 514–523. [Google Scholar] [CrossRef] [PubMed]
- Dai, Q.; Geng, H.M.; Yu, Q.; Hao, J.C.; Cui, J.W. Polyphenol-Based Particles for Theranostics. Theranostics 2019, 9, 3170–3190. [Google Scholar] [CrossRef]
- Ernest, U.; Chen, H.Y.; Xu, M.J.; Taghipour, Y.D.; Bin Asad, M.H.H.; Rahimi, R.; Murtaza, G. Anti-Cancerous Potential of Polyphenol-Loaded Polymeric Nanotherapeutics. Molecules 2018, 23, 2787. [Google Scholar] [CrossRef] [PubMed]
- Oliver, S.; Vittorio, O.; Cirillo, G.; Boyer, C. Enhancing the therapeutic effects of polyphenols with macromolecules. Polym. Chem. 2016, 7, 1529–1544. [Google Scholar] [CrossRef]
- Batra, H.; Pawar, S.; Bahl, D. Curcumin in combination with anti-cancer drugs: A nanomedicine review. Pharmacol. Res. 2019, 139, 91–105. [Google Scholar] [CrossRef]
- Wei, Y.M.; Yang, P.J.; Cao, S.S.; Zhao, L. The combination of curcumin and 5-fluorouracil in cancer therapy. Arch. Pharm. Res. 2018, 41, 1–13. [Google Scholar] [CrossRef]
- Kumar, S.U.; Kumar, V.; Priyadarshi, R.; Gopinath, P.; Negi, Y.S. pH-responsive prodrug nanoparticles based on xylan-curcumin conjugate for the efficient delivery of curcumin in cancer therapy. Carbohyd. Polym. 2018, 188, 252–259. [Google Scholar] [CrossRef]
- Li, J.; Shin, G.H.; Chen, X.G.; Park, H.J. Modified curcumin with hyaluronic acid: Combination of pro-drug and nano-micelle strategy to address the curcumin challenge. Food Res. Int. 2015, 69, 202–208. [Google Scholar] [CrossRef]
- Saranya, T.S.; Rajan, V.K.; Biswas, R.; Jayakumar, R.; Sathianarayanan, S. Synthesis, characterisation and biomedical applications of curcumin conjugated chitosan microspheres. Int. J. Biol. Macromol. 2018, 110, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Zare, M.; Sarkati, M.N.; Tashakkorian, H.; Partovi, R.; Rahaiee, S. Dextran-immobilized curcumin: An efficient agent against food pathogens and cancer cells. J. Bioact. Compat. Pol. 2019, 34, 309–320. [Google Scholar] [CrossRef]
- Kim, C.Y.; Bordenave, N.; Ferruzzi, M.G.; Safavy, A.; Kim, K.H. Modification of Curcumin with Polyethylene Glycol Enhances the Delivery of Curcumin in Preadipocytes and Its Antiadipogenic Property. J. Agric. Food Chem. 2011, 59, 1012–1019. [Google Scholar] [CrossRef] [PubMed]
- Lerra, L.; Farfalla, A.; Sanz, B.; Cirillo, G.; Vittorio, O.; Voli, F.; Le Grand, M.; Curcio, M.; Nicoletta, F.P.; Dubrovska, A.; et al. Graphene Oxide Functional Nanohybrids with Magnetic Nanoparticles for Improved Vectorization of Doxorubicin to Neuroblastoma Cells. Pharmaceutics 2019, 11, 3. [Google Scholar] [CrossRef]
- Banerjee, A.; Bandopadhyay, R. Use of dextran nanoparticle: A paradigm shift in bacterial exopolysaccharide based biomedical applications. Int. J. Biol. Macromol. 2016, 87, 295–301. [Google Scholar] [CrossRef]
- Vittorio, O.; Cojoc, M.; Curcio, M.; Spizzirri, U.G.; Hampel, S.; Nicoletta, F.P.; Iemma, F.; Dubrovska, A.; Kavallaris, M.; Cirillo, G. Polyphenol Conjugates by Immobilized Laccase: The Green Synthesis of Dextran-Catechin. Macromol. Chem. Phys. 2016, 217, 1488–1492. [Google Scholar] [CrossRef]
- Chen, X.; Zou, L.Q.; Niu, J.; Liu, W.; Peng, S.F.; Liu, C.M. The Stability, Sustained Release and Cellular Antioxidant Activity of Curcumin Nanoliposomes. Molecules 2015, 20, 14293–14311. [Google Scholar] [CrossRef]
- Zhao, Z.; Xie, M.B.; Li, Y.; Chen, A.Z.; Li, G.; Zhang, J.; Hu, H.W.; Wang, X.Y.; Li, S.P. Formation of curcumin nanoparticles via solution-enhanced dispersion by supercritical CO2. Int. J. Nanomed. 2015, 10, 3171–3181. [Google Scholar] [CrossRef]
- Cirillo, G.; Puoci, F.; Iemma, F.; Curcio, M.; Parisi, O.I.; Spizzirri, U.G.; Altimari, I.; Picci, N. Starch-quercetin conjugate by radical grafting: Synthesis and biological characterization. Pharm. Dev. Technol. 2012, 17, 466–476. [Google Scholar] [CrossRef]
- Arts, M.J.T.J.; Dallinga, J.S.; Voss, H.P.; Haenen, G.R.M.M.; Bast, A. A new approach to assess the total antioxidant capacity using the TEAC assay. Food Chem. 2004, 88, 567–570. [Google Scholar] [CrossRef]
- Bai, F.; Diao, J.J.; Wang, Y.; Sun, S.X.; Zhang, H.M.; Liu, Y.Y.; Wang, Y.Q.; Cao, J. A New Water-Soluble Nanomicelle Formed through Self-Assembly of Pectin-Curcumin Conjugates: Preparation, Characterization, and Anticancer Activity Evaluation. J. Agric. Food Chem. 2017, 65, 6840–6847. [Google Scholar] [CrossRef] [PubMed]
- Curcio, M.; Blanco-Fernandez, B.; Diaz-Gomez, L.; Concheiro, A.; Alvarez-Lorenzo, C. Hydrophobically modified keratin vesicles for GSH-responsive intracellular drug release. Bioconjugate Chem. 2015, 26, 1900–1907. [Google Scholar] [CrossRef] [PubMed]
- Reis, A.V.; Guilherme, M.R.; Rubira, A.F.; Muniz, E.C. Mathematical model for the prediction of the overall profile of in vitro solute release from polymer networks. J. Colloid Interface Sci. 2007, 310, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.G.; Wu, D.J.; Liu, L.; Chen, J.D.; Xu, Y. Preparation, characterization, and in vitro release of folic acid-conjugated chitosan nanoparticles loaded with methotrexate for targeted delivery. Polym. Bull. 2012, 68, 1707–1720. [Google Scholar] [CrossRef]
- Chen, F.H.; Zhao, T.N.; Chen, Q.T.; Han, L.F.; Fang, S.M.; Chen, Z.J. Synthesis and release behavior of methotrexate from Fe3O4/PLA-PEG core/shell nanoparticles with high saturation magnetization. Mater. Lett. 2013, 108, 179–182. [Google Scholar] [CrossRef]
- Choksakulnimitr, S.; Masuda, S.; Tokuda, H.; Takakura, Y.; Hashida, M. In-Vitro Cytotoxicity of Macromolecules in Different Cell-Culture Systems. J. Control. Release 1995, 34, 233–241. [Google Scholar] [CrossRef]
- Marquez, M.; Du, J.; Edgren, M.; Nilsson, S.; Lennartsson, L.; Hiltunen, J.; Westlin, J.E.; Tammela, T.; Raitanen, M.; Laato, M.; et al. Development of dextran derivatives with cytotoxic effects in human urinary bladder cancer cell lines. Anticancer Res. 2002, 22, 741–744. [Google Scholar]
- Vittorio, O.; Cirillo, G.; Iemma, F.; Di Turi, G.; Jacchetti, E.; Curcio, M.; Barbuti, S.; Funel, N.; Parisi, O.I.; Puoci, F.; et al. Dextran-Catechin Conjugate: A Potential Treatment Against the Pancreatic Ductal Adenocarcinoma. Pharm. Res. 2012, 29, 2601–2614. [Google Scholar] [CrossRef]
- Ashton, J.C. Drug Combination Studies and Their Synergy Quantification Using the Chou-Talalay Method-Letter. Cancer Res. 2015, 75, 2400. [Google Scholar] [CrossRef]
- Vittorio, O.; Curcio, M.; Cojoc, M.; Goya, G.F.; Hampel, S.; Iemma, F.; Dubrovska, A.; Cirillo, G. Polyphenols delivery by polymeric materials: Challenges in cancer treatment. Drug Deliv. 2017, 24, 162–180. [Google Scholar] [CrossRef]
- Edelman, R.; Assaraf, Y.G.; Levitzky, I.; Shahar, T.; Livney, Y.D. Hyaluronic acid-serum albumin conjugate-based nanoparticles for targeted cancer therapy. Oncotarget 2017, 8, 24337–24353. [Google Scholar] [CrossRef] [PubMed]
- Shapira, A.; Assaraf, Y.G.; Epstein, D.; Livney, Y.D. Beta-casein Nanoparticles as an Oral Delivery System for Chemotherapeutic Drugs: Impact of Drug Structure and Properties on Co-assembly. Pharm. Res. 2010, 27, 2175–2186. [Google Scholar] [CrossRef] [PubMed]
- Danaei, M.; Dehghankhold, M.; Ataei, S.; Davarani, F.H.; Javanmard, R.; Dokhani, A.; Khorasani, S.; Mozafari, M.R. Impact of Particle Size and Polydispersity Index on the Clinical Applications of Lipidic Nanocarrier Systems. Pharmaceutics 2018, 10, 57. [Google Scholar] [CrossRef] [PubMed]

| MTX (µM) | CUR Equivalent Concentration (µM) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 24 h | 48 h | |||||||||||
| Free CUR | DC | Free CUR | DC | |||||||||
| 5 | 10 | 20 | 5 | 10 | 20 | 5 | 10 | 20 | 5 | 10 | 20 | |
| 22 | 1.59 | 1.23 | 1.61 | 0.44 | 0.65 | 1.11 | 0.59 | 0.66 | 0.98 | 0.50 | 0.70 | 0.96 |
| 110 | 2.91 | 2.27 | 2.46 | 1.13 | 1.11 | 1.15 | 1.60 | 1.63 | 1.72 | 1.03 | 1.10 | 1.17 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Curcio, M.; Cirillo, G.; Tucci, P.; Farfalla, A.; Bevacqua, E.; Vittorio, O.; Iemma, F.; Nicoletta, F.P. Dextran-Curcumin Nanoparticles as a Methotrexate Delivery Vehicle: A Step Forward in Breast Cancer Combination Therapy. Pharmaceuticals 2020, 13, 2. https://doi.org/10.3390/ph13010002
Curcio M, Cirillo G, Tucci P, Farfalla A, Bevacqua E, Vittorio O, Iemma F, Nicoletta FP. Dextran-Curcumin Nanoparticles as a Methotrexate Delivery Vehicle: A Step Forward in Breast Cancer Combination Therapy. Pharmaceuticals. 2020; 13(1):2. https://doi.org/10.3390/ph13010002
Chicago/Turabian StyleCurcio, Manuela, Giuseppe Cirillo, Paola Tucci, Annafranca Farfalla, Emilia Bevacqua, Orazio Vittorio, Francesca Iemma, and Fiore Pasquale Nicoletta. 2020. "Dextran-Curcumin Nanoparticles as a Methotrexate Delivery Vehicle: A Step Forward in Breast Cancer Combination Therapy" Pharmaceuticals 13, no. 1: 2. https://doi.org/10.3390/ph13010002
APA StyleCurcio, M., Cirillo, G., Tucci, P., Farfalla, A., Bevacqua, E., Vittorio, O., Iemma, F., & Nicoletta, F. P. (2020). Dextran-Curcumin Nanoparticles as a Methotrexate Delivery Vehicle: A Step Forward in Breast Cancer Combination Therapy. Pharmaceuticals, 13(1), 2. https://doi.org/10.3390/ph13010002








