Molecularly Imprinted Polymers: Thermodynamic and Kinetic Considerations on the Specific Sorption and Molecular Recognition
Abstract
:1. Introduction
2. Results and Discussion
2.1 SEM and Infrared spectrum
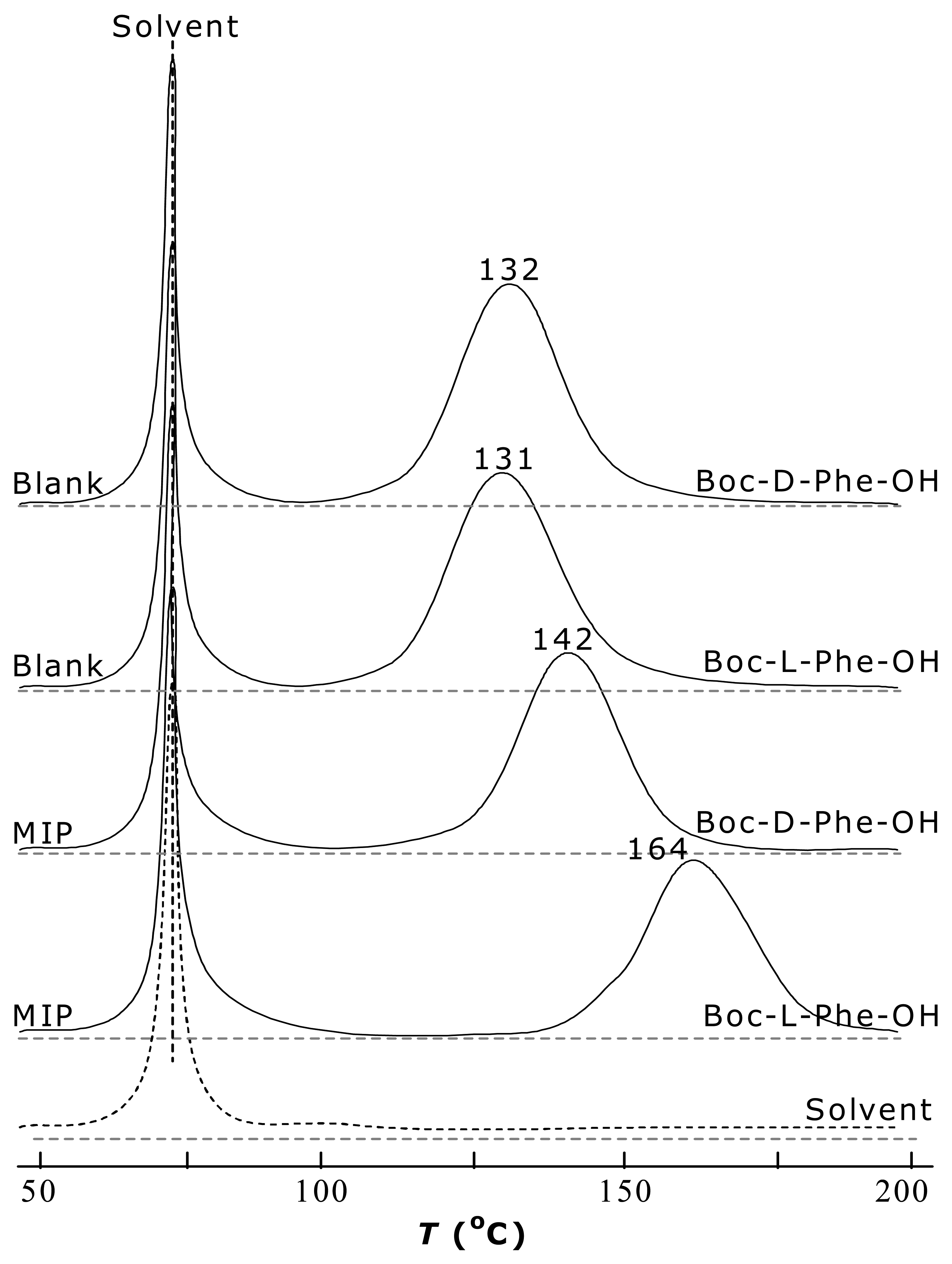
2.2 Interaction of MIP-substrate and its specificity
2.3 Specific sorption and molecular recognition
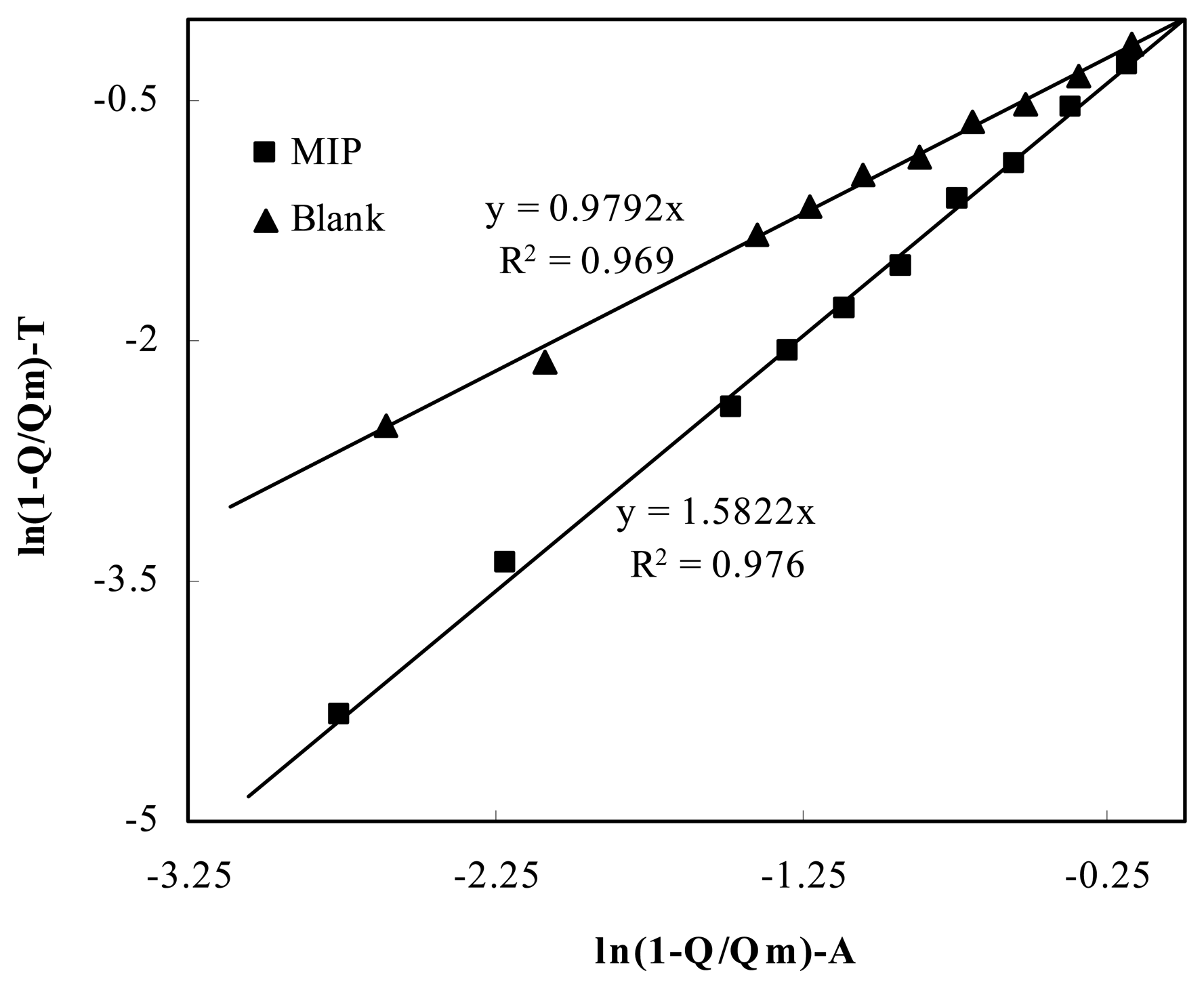
2.4 Kinetic consideration
2.5 Thermodynamic analysis
3. Experimental Section
3.1 Preparation of imprinted materials
3.2 Sorption test
3.3 Temperature-programmed desorption
Acknowledgments
References and Notes
- Li, W.K.; Li, S.J. Molecular imprinting: a versatile tool for separation, sensor and catalysis. Adv. Polym. Sci. 2007, 206, 191–210. [Google Scholar]
- Hu, X.; An, Q.; Li, G.; Tao, S.; Liu, J. Imprinted photonic polymers for chiral recognition. Angew. Chem. Int. Ed. 2006, 45, 8145–8148. [Google Scholar]
- Lettau, K.; Warsinke, A.; Katterle, M.; Danielsson, B.; Scheller, F.W. A bifunctional molecularly imprinted polymer (MIP): analysis of binding and catalysis by a thermistor. Angew. Chem. Int. Ed. 2006, 45, 6986–6990. [Google Scholar]
- Urraca, J.L.; Hall, A.J.; Moreno-Bondi, M.C.; Sellergren, B. A stoichiometric molecularly imprinted polymer for the class-selective recognition of antibiotics in aqueous media. Angew. Chem. Inter. Ed. 2006, 45, 5158–5161. [Google Scholar]
- Miyata, T.; Jige, M.; Nakaminami, T.; Uragami, T. Tumor marker-responsive behavior of gels prepared by biomolecular imprinting. Proc. Natl. Acad. Sci. USA 2006, 103, 1190–1193. [Google Scholar]
- Wu, L.; Li, Y. Metal ion-mediated molecular-imprinting polymer for indirect recognition of formate, acetate and propionate. Anal. Chim. Act. 2004, 517, 145–151. [Google Scholar]
- Ayela, C.; Vandevelde, F.; Lagrange, D.; Haupt, K.; Nicu, L. Combining resonant piezoelectric micromembranes with molecularly imprinted polymers. Angew. Chem. Int. Ed. 2007, 46, 9271–9274. [Google Scholar]
- Nishino, H.; Huang, C.S.; Shea, K.J. Selective protein capture by epitope imprinting. Angew. Chem. Int. Ed. 2006, 45, 2392–2396. [Google Scholar]
- Liu, J.Q.; Wulff, G. Molecularly imprinted polymers with strong carboxypeptidase A-like activity: combination of an amidinium function with a zinc-ion binding site in transition-state imprinted cavities. Angew. Chem. Int. Ed. 2004, 43, 1287–1290. [Google Scholar]
- Wulff, G. Molecular imprinting in cross-linked materials with the aid of molecular templates- a way towards artificial antibodies. Angew. Chem. Int. Ed. 1995, 34, 1812–1832. [Google Scholar]
- Yungerman, I.; Srebnik, S. Factors contributing to binding-site imperfections in imprinted polymers. Chem. Mater. 2006, 18, 657–663. [Google Scholar]
- Kim, H.; Guiochon, G. Adsorption on molecularly imprinted polymers of structural analogues of a template. Single-component adsorption isotherm data. Anal. Chem. 2005, 77, 6415–6425. [Google Scholar]
- Parmpi, P.; Kofinas, P. Biomimetic glucose recognition using molecularly imprinted polymer hydrogels. Biomaterials 2004, 25, 1969–1973. [Google Scholar]
- Levi, R.; McNiven, S.; Piletsky, S.A.; Rachkov, A.; Cheong, S.H.; Yano, K.; Karube, I. Optical detection of chloramphenicol using molecularly imprinted polymers. Anal. Chem. 1997, 69, 2017–2021. [Google Scholar]
- Takeda, K.; Kobayashi, T. Bisphenol A imprinted polymer adsorbents with selective recognition and binding characteristics. Sci. Technol. Adv. Mater. 2005, 6, 165–171. [Google Scholar]
- Duffy, D.J.; Das, K.; Hsu, S.L.; Penelle, J.; Rotello, V.M.; Stidham, H.D. Binding efficiency and transport properties of molecularly imprinted polymer thin films. J. Am. Chem. Soc. 2002, 124, 8290–8296. [Google Scholar]
- Li, W.K.; Li, S.J.; Luo, G.; Ding, K.R. A kinetic consideration on the selective adsorption and molecular recognition by molecularly imprinted polymer. Bull. Korean. Chem. Soc. 2007, 28, 1346–1352. [Google Scholar]
- Li, S.J.; Hu, J.; Liu, B.L. A study on the adsorption behavior of protein onto functional microspheres. J. Chem. Technol. Biotechnol. 2005, 80, 531–536. [Google Scholar]
- Li, S.J.; Hu, J.; Liu, B.L.; Li, H.; Wang, D.Q.; Liao, X.F. Thermodynamic and kinetic considerations: effect of cyclodextrin on methyl methacrylate dispersion polymerization. Polymer 2004, 45, 1511–1516. [Google Scholar]
- Zheng, M.X.; Li, S.J.; Luo, X. Rationally designing molecularly imprinted polymer toward a high specific adsorbent by using metal as assembled pivot. J. Macromol. Sci. A 2007, 44, 1187–1194. [Google Scholar]
- Zhang, D.N.; Li, S.J.; Huang, J.; Luo, G. Selective adsorption and steric recognition by molecularly imprinted polymer: a study on molecular self-assembly and its effect on the selectivity. High Perform. Polym. 2006, 18, 949–960. [Google Scholar]
- Luo, L.T.; Li, S.J.; Zhu, Y. The effects of yttrium on the hydrogenation performance and surface properties of a ruthenium-supported catalyst. J. Serb. Chem. Soc. 2005, 70, 1419–1425. [Google Scholar]






© 2008 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Li, S.; Huang, X.; Zheng, M.; Li, W.; Tong, K. Molecularly Imprinted Polymers: Thermodynamic and Kinetic Considerations on the Specific Sorption and Molecular Recognition. Sensors 2008, 8, 2854-2864. https://doi.org/10.3390/s8042854
Li S, Huang X, Zheng M, Li W, Tong K. Molecularly Imprinted Polymers: Thermodynamic and Kinetic Considerations on the Specific Sorption and Molecular Recognition. Sensors. 2008; 8(4):2854-2864. https://doi.org/10.3390/s8042854
Chicago/Turabian StyleLi, Songjun, Xing Huang, Mingxia Zheng, Wuke Li, and Kejun Tong. 2008. "Molecularly Imprinted Polymers: Thermodynamic and Kinetic Considerations on the Specific Sorption and Molecular Recognition" Sensors 8, no. 4: 2854-2864. https://doi.org/10.3390/s8042854
APA StyleLi, S., Huang, X., Zheng, M., Li, W., & Tong, K. (2008). Molecularly Imprinted Polymers: Thermodynamic and Kinetic Considerations on the Specific Sorption and Molecular Recognition. Sensors, 8(4), 2854-2864. https://doi.org/10.3390/s8042854



