Design of a Selective and Sensitive PVC-Membrane Potentiometric Sensor for Strontium Ion Based on 1,10-Diaza-5,6-benzo-4,7-dioxacyclohexadecane-2,9-dioneas a Neutral Ionophore
Abstract
:1. Introduction

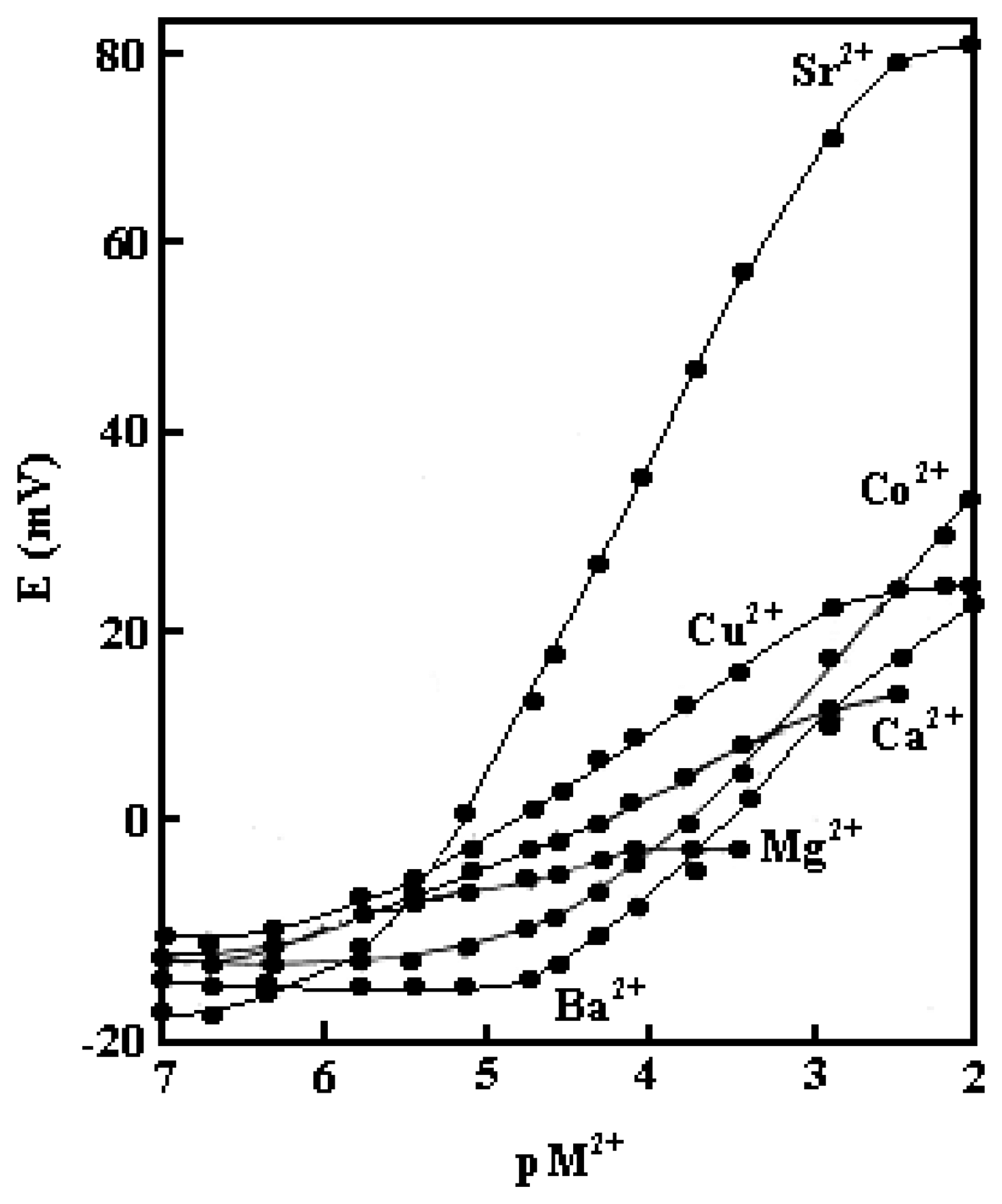
2. Results and Discussion
3. Experimental Section
3.1. Reagents
3.2. Electrode Preparation
3.3. EMF Measurements
Acknowledgments
References and Notes
- Jain, A.K.; Gupta, V.K.; Raisoni, J.R. Strontium(II)-selective potentiometric sensor based on ester derivative of 4-tert-butylcalix[8]arene in PVC matrix. Sensors 2004, 4, 115–124. [Google Scholar]
- Elvers, B.; Hawkins, S.; Russey, W. (Eds.) Ulmann'sEncyclopedia of Industrial Chemistry, 5th Ed.; Vol. A25, 1995; VCH Publisher: New York; p. 321.
- Othmer, K. Encyclopedia of Chemical Technology, 4th Ed. ed; Vol. 22, 1992; John Wiley & Sons: New York; p. 947. [Google Scholar]
- Umezawa, Y. (Ed.) CRC Handbook of Ion-Selective Electrodes: Selectivity Coefficients; 1990; CRC Press: Boca Raton, FL.
- Janata, J.; Jasowicz, M.; DeVaney, D.M. Chemical sensors. Anal. Chem. 1998, 70, 179R–208R. [Google Scholar]
- Bühlmann, P.; Pretsch, E.; Bakker, E. Carrier based ion-selective electrodes and the bulk optodes. 2. Ionophoresand for potentiometric and optical sensors. Chem. Rev. 1998, 98, 1593–1678. [Google Scholar]
- Izatt, R.M.; Pawlak, K.; Bradshaw, J.; Bruening, R.L. Thermodynamic and kinetic data for macrocycle interaction with cations and anions. Chem. Rev. 1991, 91, 1721–2085. [Google Scholar]
- Shih, J.-S. Applications of macrocyclic polyethers. J. Chin. Chem. Soc. 1992, 39, 551–559. [Google Scholar]
- Lippolis, V.; Shamsipur, M. Synthesis, coordination properties, and analytical applicationsof mixed donor macrocycles containing the1,10-phenanthroline sub-unit. J. Iran. Chem. Soc. 2006, 3, 105–127. [Google Scholar]
- An, H.Y.; Bradshaw, J.S.; Izatt, R.M.; Yan, Z. Bis and oligo(benzocrown ethers). Chem. Rev. 1994, 94, 939–991. [Google Scholar]
- Sharghi, H.; Eshghi, H. Efficient synthesis of macrocyclic diamides. Tetrahedron 1995, 51, 913–922. [Google Scholar]
- Shamsipur, M.; Rouhani, S.; Ganjali, M. R.; Eshghi, H.; Sharghi, H. Cu(II)-selective membrane electrode based on a recently synthesized macrocyclic diamide. Microchem. J. 1999, 63, 202–210. [Google Scholar]
- Shamsipur, M.; Rouhani, S.; Ganjali, M.R.; Sharghi, H.; Eshghi, H. Zinc-selective membrane potentiometric sensor based on a recently synthesized benzo-substituted macrocyclic diamide. Sens. Actuators B 1999, 59, 30–34. [Google Scholar]
- Javanbakht, M.; Ganjali, M.R.; Eshghi, H.; Sharghi, H.; Shamsipur, M. Mercury(II) ion-selective electrode based on dibenzo-diazathia-18-crown-6-dione. Electroanalysis 1999, 11, 81–84. [Google Scholar]
- Shamsipur, M.; Rouhani, S.; Sharghi, H.; Ganjali, M.R.; Eshghi, H. Strontium-selective membrane electrode based on some recently synthesized benzo-substituted macrocyclic diamides. Anal. Chem. 1999, 71, 4938–4943. [Google Scholar]
- Shamsipur, M.; Khayatian, G.; Kazemi, S.Y.; Niknam, K.; Sharghi, H. The synthesis of 1,4-diaza-2,3;8,9-dibenzo-7,10-dioxacyclododecane-5,12-dione and its use in calcium-selective carbon paste electrodes. J. Incl. Phenom. 2001, 40, 303–307. [Google Scholar]
- Shamsipur, M.; Kazemi, S.Y.; Sharghi, H.; Niknam, K. Cesium-selective membrane electrode based on a recently synthesized 16-membered macrocyclic diamide. Fresenius J. Anal. Chem. 2001, 371, 1104–1108. [Google Scholar]
- Shamsipur, M.; Rouhani, S.; Poursaberi, T.; Ganjali, M.R.; Sharghi, H.; Niknam, K. Cobalt(II)-selective coated graphite PVC-membrane electrode based on a recently synthesized dibenzopyridino-substituted macrocyclicdiamide. Electroanalysis 2002, 14, 729. [Google Scholar]
- Shamsipur, M.; Kazemi, S.Y.; Niknam, K.; Sharghi, H. A new pVC-membrane electrode based on a thia-substituted macrocyclic diamide in selective potentiometric determination of silver ion. Bull. Korean Chem. Soc. 2002, 23, 53–58. [Google Scholar]
- Shamsipur, M.; Soleymanpour, A.; Akhond, M.; Sharghi, H. New macrocyclic diamides as neutral ionophores for highly selective and sensitive PVC-membrane electrodes for Be2+ ion. Electroanalysis 2004, 16, 282–288. [Google Scholar]
- Akmal, N.; Zimmer, H.; Mark, H.B. Strontium ion-selective electrode based on a conducting poly(dibenzo-18-crown-6) film. Anal. Lett. 1991, 24, 1431–1443. [Google Scholar]
- Qian, G.; Wu, B.B.; Wu, G.; Huang, S.; Yan, Y.; Tian, B. Strontium-selective electrodes based on diamides with pyridine ring as ionophores. Talanta 1998, 47, 1149–1155. [Google Scholar]
- Gupta, V.K.; Ludwig, R.; Agarwal, S. Strontium(II) sensor based on a modified calyx[6]arene in PVC matrix. Anal. Sci. 2005, 21, 293–296. [Google Scholar]
- Sharghi, H.; Zare, A. Efficient synthesis of some novel macrocyclic diamides using fast addition method. Synthesis 2006, 999–1004. [Google Scholar]
- Kamata, S.; Bhale, A.; Fukunaga, Y.; Murata, A. Copper(II)-selective electrode using thiuram disulfate neutral carriers. Anal. Chem. 1988, 60, 2464–2467. [Google Scholar]
- Kimura, K.; Kumami, K.; Kitazawa, S.; Shono, T. Calcium-selective polymeric membrane electrodes based on bicyclic poly(ether amide). Anal. Chem. 1984, 56, 2369–2372. [Google Scholar]
- Bakker, E.; Bühlmann, P.; Bakker, E. Carrier-based ion selective electrodes and bulk optodes. Part 1. General characteristics. Chem. Rev. 1997, 97, 3083–3112. [Google Scholar]
- Eugster, R.; Gehring, P.M.; Morf, W.E.; Spichiger, U.E.; Simon, W. Selectivity-modifying influence of anionic sites in neutral carrier-based membrane electrodes. Anal. Chem. 1991, 63, 2285–2289. [Google Scholar]
- Gehrig, P.M.; Morf, W.E.; Welti, M.; Pretsch, E.; Simon, W. Catalysis of ion transfer by tetraphenylborates in neutral carrier-based ion-selective electrodes. Helv. Chim. Acta 1990, 73, 203–212. [Google Scholar]
- Umezawa, Y.; Umezawa, K.; Sato, H. Selectivity coefficients for ion-selective electrodes: recommended methods for reporting Kpot values. Pure Appl. Chem. 1995, 67, 507–518. [Google Scholar]



| No | Composition (%) | Slope (mV decade-1) | Linear rangea (M) | |||
|---|---|---|---|---|---|---|
| PVC | Plasticizer | STB | L | |||
| 1 | 32.5 | AP, 66.0 | 0.5 | 0.0 | 13.5 | 4.6 × 10-5-1.3 × 10-3 |
| 2 | 31.0 | AP, 67.0 | 0.0 | 2.0 | 22.0 | 1.7 × 10-5-6.3 × 10-3 |
| 3 | 31.0 | AP, 66.0 | 1.0 | 2.0 | 25.0 | 3.6 × 10-6-6.3 × 10-3 |
| 4 | 30.0 | AP, 66.3 | 1.7 | 2.0 | 30.5 | 1.6 × 10-6-3.0 × 10-3 |
| 5 | 30.0 | NPOE, 66.3 | 1.7 | 2.0 | 23.5 | 3.6 × 10-6-1.2 × 10-3 |
| 6 | 30.0 | DBP, 66.3 | 1.7 | 2.0 | 16.8 | 3.6 × 10-6-1.0 × 10-3 |
| 7 | 30.0 | DOP, 66.3 | 1.7 | 2.0 | 14.7 | 3.6 × 10-6-6.3 × 10-3 |
| 8 | 30.0 | DMS, 66.3 | 1.7 | 2.0 | 12.8 | 3.6 × 10-6-6.2 × 10-3 |
| Interfering ion | KA,BMPM | Interfering ion | KA,BMPM |
|---|---|---|---|
| Na+ | 1.3×10-2 | Ba2+ | 9.2×10-2 |
| K+ | 3.3×100-2 | Co2+ | 8×10-2 |
| Cs+ | 3.3×10-2 | Ni2+ | 5.4×10-2 |
| Rb+ | 3.1×10-2 | Cu2+ | 9.2×10-2 |
| Mg2+ | 6.3×10-2 | Pb2+ | 7.2×10-2 |
| Ca2+ | 8.9×10-3 |
 |
© 2007 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Shamsipur, M.; Kazemi, S.Y.; Sharghi, H. Design of a Selective and Sensitive PVC-Membrane Potentiometric Sensor for Strontium Ion Based on 1,10-Diaza-5,6-benzo-4,7-dioxacyclohexadecane-2,9-dioneas a Neutral Ionophore. Sensors 2007, 7, 438-447. https://doi.org/10.3390/s7040438
Shamsipur M, Kazemi SY, Sharghi H. Design of a Selective and Sensitive PVC-Membrane Potentiometric Sensor for Strontium Ion Based on 1,10-Diaza-5,6-benzo-4,7-dioxacyclohexadecane-2,9-dioneas a Neutral Ionophore. Sensors. 2007; 7(4):438-447. https://doi.org/10.3390/s7040438
Chicago/Turabian StyleShamsipur, Mojtaba, Sayed Yahya Kazemi, and Hashem Sharghi. 2007. "Design of a Selective and Sensitive PVC-Membrane Potentiometric Sensor for Strontium Ion Based on 1,10-Diaza-5,6-benzo-4,7-dioxacyclohexadecane-2,9-dioneas a Neutral Ionophore" Sensors 7, no. 4: 438-447. https://doi.org/10.3390/s7040438
APA StyleShamsipur, M., Kazemi, S. Y., & Sharghi, H. (2007). Design of a Selective and Sensitive PVC-Membrane Potentiometric Sensor for Strontium Ion Based on 1,10-Diaza-5,6-benzo-4,7-dioxacyclohexadecane-2,9-dioneas a Neutral Ionophore. Sensors, 7(4), 438-447. https://doi.org/10.3390/s7040438




