SDS-PAGE Analysis of Soluble Proteins in Reconstituted Milk Exposed to Different Heat Treatments
Abstract
:1. Introduction
2. Experimental Part
Heat treatments
Soluble protein content detection
Soluble protein composition
3. Discussion
3.1 Protein solubility
3.2 Soluble proteins composition
3.3 Influence of the addition of demineralized whey on the soluble protein composition
Conclusion
Acknowledgments
References
- Singh, H.; Waungana, A. Influence of heat treatment of milk on cheesemaking properties. Int.Dairy J. 2001, 11, 543–551. [Google Scholar]
- Macej, O.; Jovanovic, S.; Denin Djurdjevic, J. The influence of high temperature on milk proteins. Chem. Industry 2002, 56, 123–132. [Google Scholar]
- Macej, O.; Jovanovic, S.; Seratlic, S.; Barac, M. Production of fresh chese with milk protein coagregates. Biotechnology in Animal Husbandry 2004, 20, 119–131. [Google Scholar]
- Fox, P. F.; McSweeney, P.L.H. Milk proteins. In Dairy Chemistry and Biochemistry; 1998; Chapman & Hall: London; pp. 146–239. [Google Scholar]
- Jollès, P.; Fiat, A. M. The carbohydrate portions of milk glycoproteins. J. Dairy Res. 1979, 46(2), 187–191. [Google Scholar]
- Donovan, M.; Mulvihill, D. M. Thermal denaturation and aggregation of whey proteins. Irish J. Food Sci. Techn. 1987, 11, 87–100. [Google Scholar]
- Morr, C.V. Functionality of heated milk proteins in dairy and related foods. J. Dairy Sci. 1985, 68(10), 2773–2781. [Google Scholar]
- Corredig, M.; Dalgleish, D. G. Effect of different heat treatments on the strong binding interactions between whey proteins and milk fat globules in whole milk. J. Dairy Res. 1996, 63(3), 441–449. [Google Scholar]
- Mottar, J.; Bassier, A.; Joniau, M.; Baert, J. Effect of heat-induced association of whey proteins and casein micelles on yogurt texture. J. Dairy Sci. 1989, 72(9), 2247–2256. [Google Scholar]
- Maćej, O. D.; Jovanović, S. T. Formation of complex between casein and whey proteins in heat-treated milk. Acta Periodica 2000, 31, 83–93. [Google Scholar]
- Anema, S. G.; Lie, Y. Association of denatured whey proteins with casein micelles in heated reconstituted skim milk and its effect on casein micelle size. J. Dairy Res. 2003, 70, 73–80. [Google Scholar]
- Bradford, M. M. A rapid and sensitive method for quantization of microgram quantities of protein utilizing the principle of protein-dye-binding. Anal Biochem. 1976, 72, 248–254. [Google Scholar]
- Fling, S. P.; Gregerson, D. S. Peptide and protein molecular weight determination by electrophoresis using a high-molarity Tris buffer system without urea. Anal. Biochem. 1986, 155, 83–88. [Google Scholar]
- Jovanovic, S.; Barac, M.; Macej, O.; Denin Djurdjevic, J. Page analysis of milk proteins altered by high thermal treatment. Acta Alimentaria 2005, 34, 105–112. [Google Scholar]
- Corredig, M.; Dalgleish, D.G. The mechanisms of heat-induced interactions of whey proteins with casein micelles in milk. Int. Dairy J. 1999, 9, 233–236. [Google Scholar]
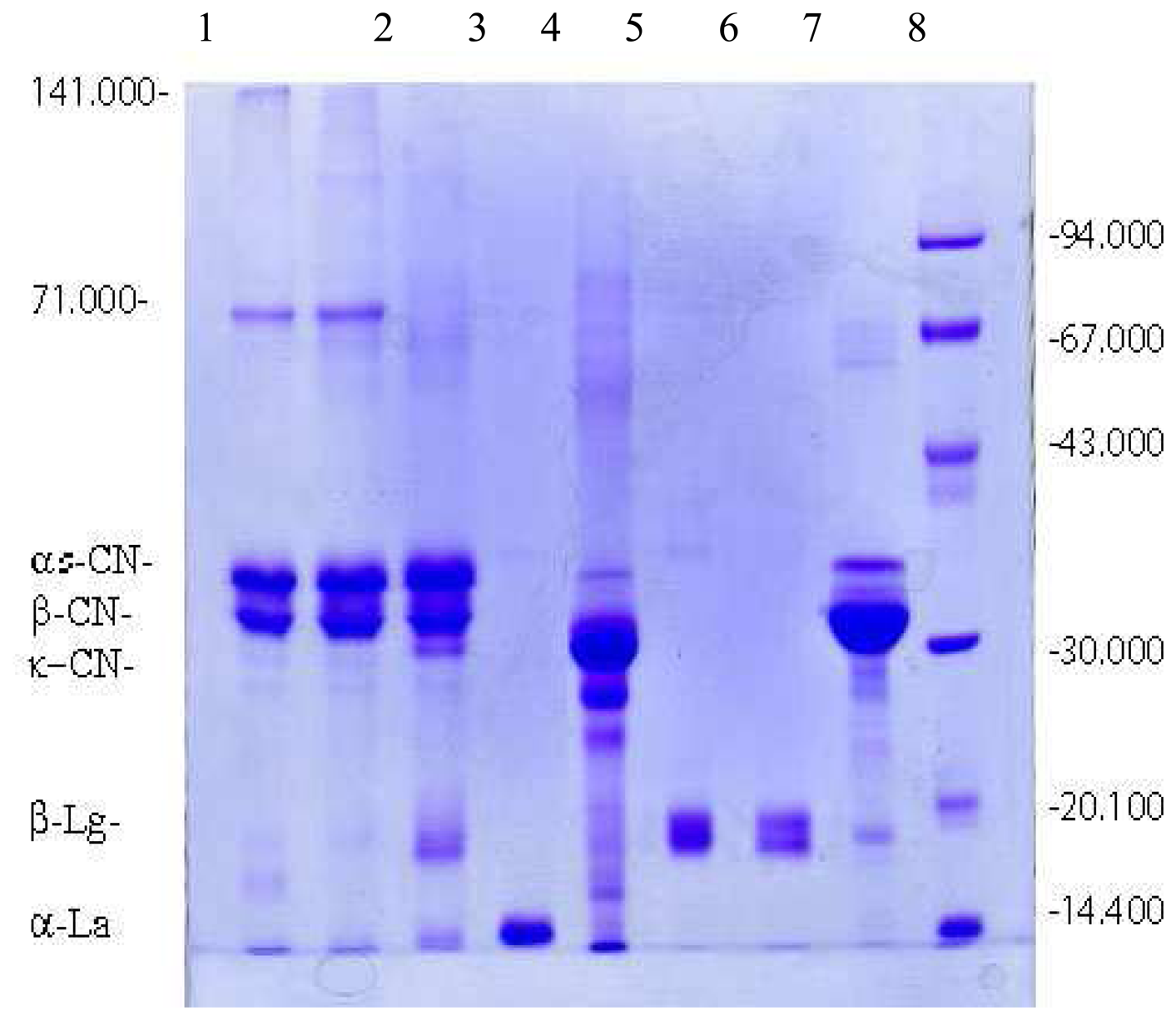
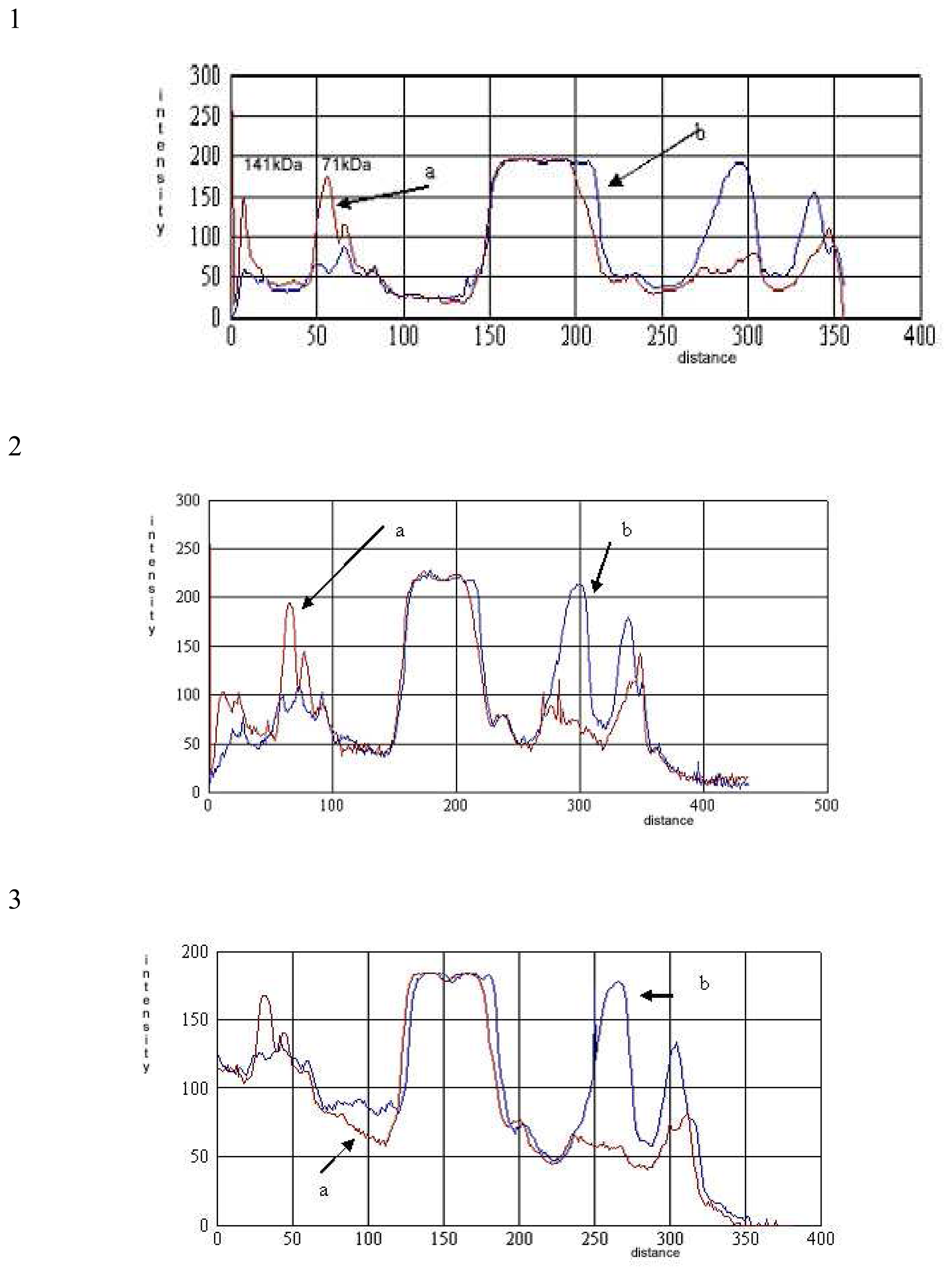
 ) and with (
) and with (
 ) 0.5% of demineralized whey. c. The comparative densitometric analysis of samples treated with 0.5%, 1% and 2.0% of the demineralized whey added.*1, 3, 5 samples without 2-mercaptoethanol, and 2, 4, 6 samples with 2-mercaptoethanol.
) 0.5% of demineralized whey. c. The comparative densitometric analysis of samples treated with 0.5%, 1% and 2.0% of the demineralized whey added.*1, 3, 5 samples without 2-mercaptoethanol, and 2, 4, 6 samples with 2-mercaptoethanol.
 ) and with (
) and with (
 ) 0.5% of demineralized whey. c. The comparative densitometric analysis of samples treated with 0.5%, 1% and 2.0% of the demineralized whey added.*1, 3, 5 samples without 2-mercaptoethanol, and 2, 4, 6 samples with 2-mercaptoethanol.
) 0.5% of demineralized whey. c. The comparative densitometric analysis of samples treated with 0.5%, 1% and 2.0% of the demineralized whey added.*1, 3, 5 samples without 2-mercaptoethanol, and 2, 4, 6 samples with 2-mercaptoethanol.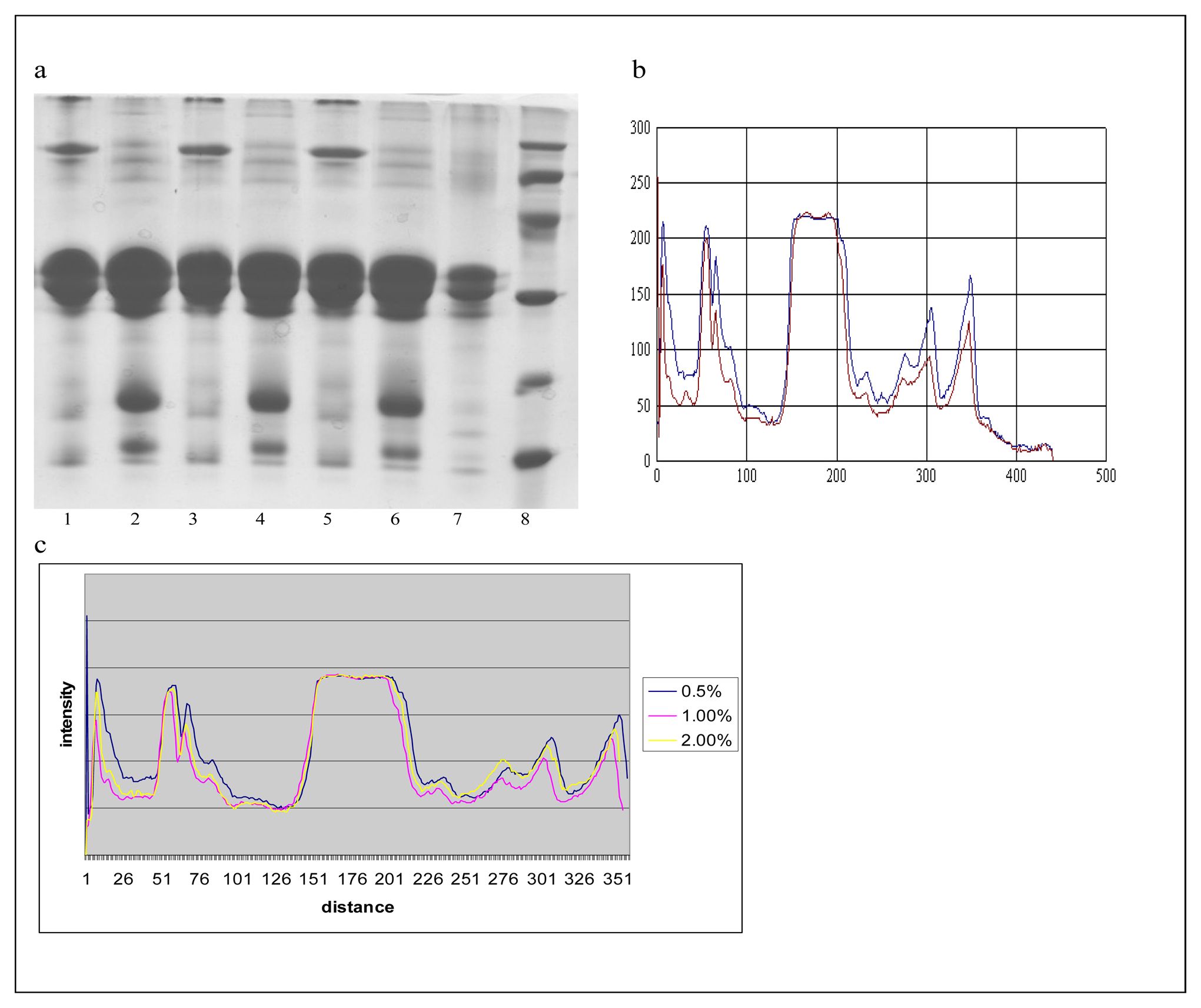
 ) and with (
) and with (
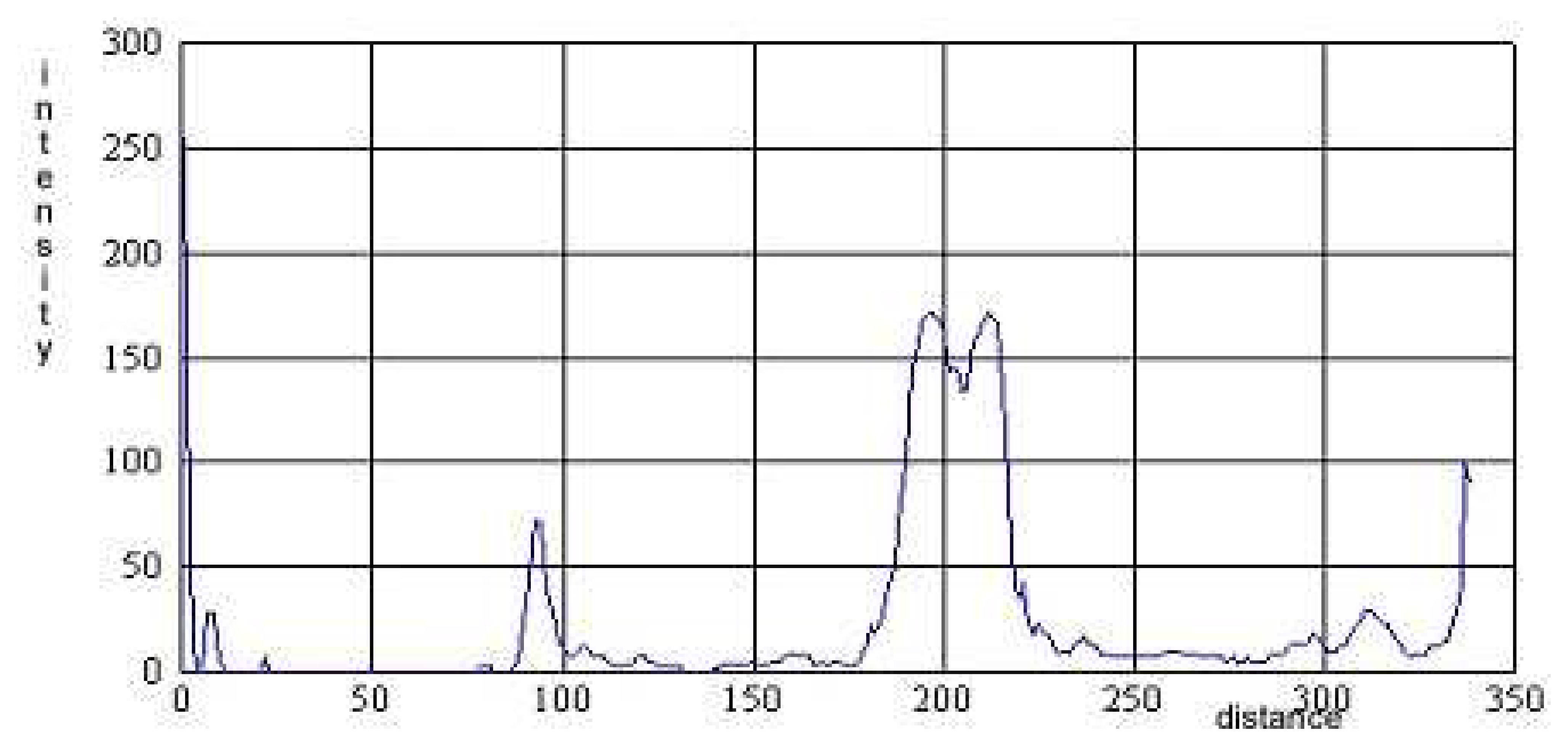
 ) 0.5% of the demineralized whey c. Comparative densitometric analysis of the samples treated with 0%, 0.5%, 1% and 2.0% of the demineralized whey added.*1, 3, 5 samples without 2-mercaptoethanol, and 2, 4, 6 samples with 2-mercaptoethanol.
) 0.5% of the demineralized whey c. Comparative densitometric analysis of the samples treated with 0%, 0.5%, 1% and 2.0% of the demineralized whey added.*1, 3, 5 samples without 2-mercaptoethanol, and 2, 4, 6 samples with 2-mercaptoethanol.
 ) and with (
) and with (
 ) 0.5% of the demineralized whey c. Comparative densitometric analysis of the samples treated with 0%, 0.5%, 1% and 2.0% of the demineralized whey added.*1, 3, 5 samples without 2-mercaptoethanol, and 2, 4, 6 samples with 2-mercaptoethanol.
) 0.5% of the demineralized whey c. Comparative densitometric analysis of the samples treated with 0%, 0.5%, 1% and 2.0% of the demineralized whey added.*1, 3, 5 samples without 2-mercaptoethanol, and 2, 4, 6 samples with 2-mercaptoethanol.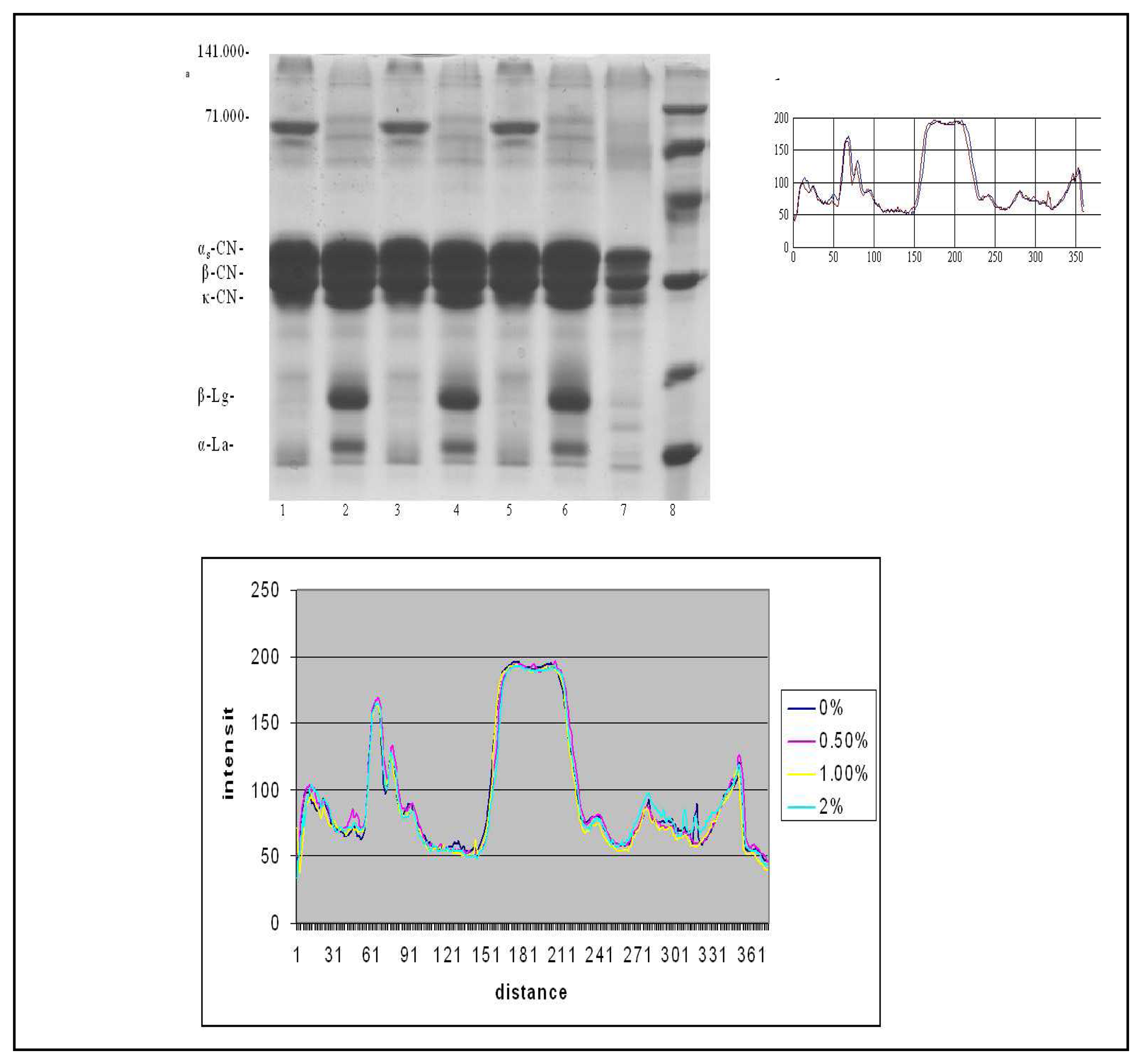
 ) and with (
) and with (
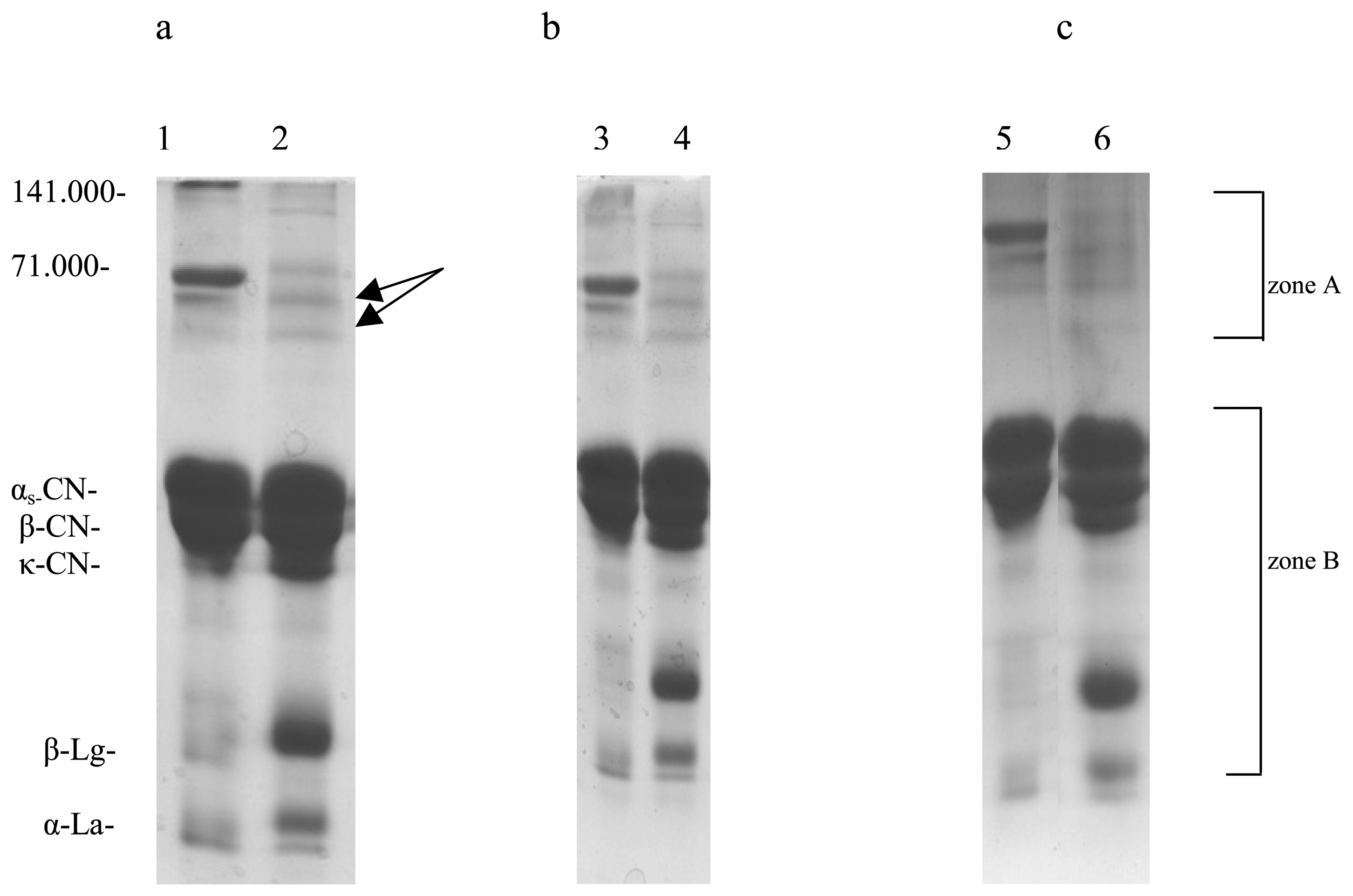
 ) 0.5% of the demineralized whey. c. The comparative densitometric analysis of the samples treated with 0.5%, 1% and 2.0% of the demineralized whey added.*4, 6, 8- samples without 2-mercaptoethanol, and 3, 5, 7- samples with 2-mercaptoethanol.
) 0.5% of the demineralized whey. c. The comparative densitometric analysis of the samples treated with 0.5%, 1% and 2.0% of the demineralized whey added.*4, 6, 8- samples without 2-mercaptoethanol, and 3, 5, 7- samples with 2-mercaptoethanol.
 ) and with (
) and with (
 ) 0.5% of the demineralized whey. c. The comparative densitometric analysis of the samples treated with 0.5%, 1% and 2.0% of the demineralized whey added.*4, 6, 8- samples without 2-mercaptoethanol, and 3, 5, 7- samples with 2-mercaptoethanol.
) 0.5% of the demineralized whey. c. The comparative densitometric analysis of the samples treated with 0.5%, 1% and 2.0% of the demineralized whey added.*4, 6, 8- samples without 2-mercaptoethanol, and 3, 5, 7- samples with 2-mercaptoethanol.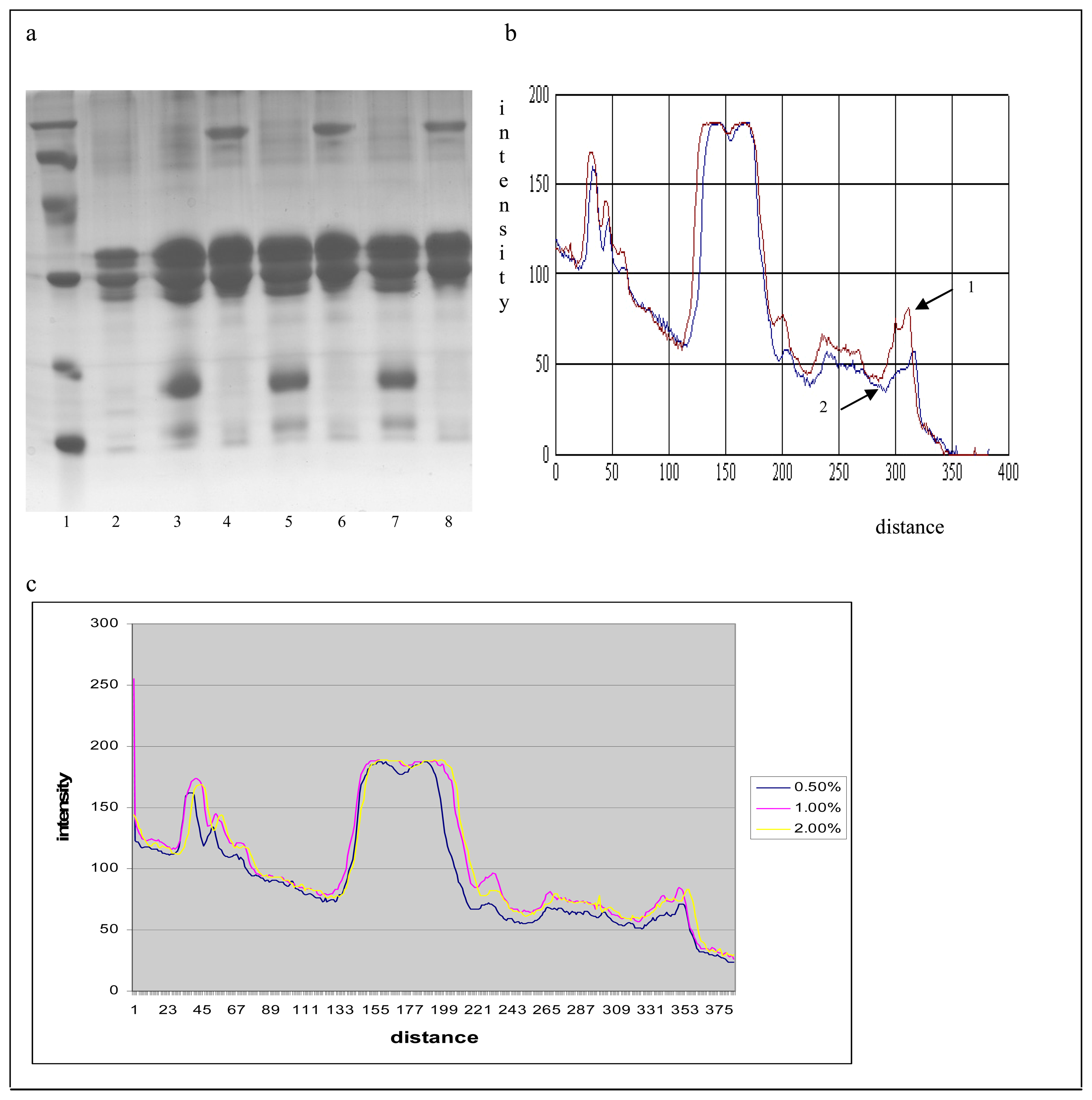
© 2007 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Jovanovic, S.; Barac, M.; Macej, O.; Vucic, T.; Lacnjevac, C. SDS-PAGE Analysis of Soluble Proteins in Reconstituted Milk Exposed to Different Heat Treatments. Sensors 2007, 7, 371-383. https://doi.org/10.3390/s7030371
Jovanovic S, Barac M, Macej O, Vucic T, Lacnjevac C. SDS-PAGE Analysis of Soluble Proteins in Reconstituted Milk Exposed to Different Heat Treatments. Sensors. 2007; 7(3):371-383. https://doi.org/10.3390/s7030371
Chicago/Turabian StyleJovanovic, Snezana, Miroljub Barac, Ognjen Macej, Tanja Vucic, and Caslav Lacnjevac. 2007. "SDS-PAGE Analysis of Soluble Proteins in Reconstituted Milk Exposed to Different Heat Treatments" Sensors 7, no. 3: 371-383. https://doi.org/10.3390/s7030371
APA StyleJovanovic, S., Barac, M., Macej, O., Vucic, T., & Lacnjevac, C. (2007). SDS-PAGE Analysis of Soluble Proteins in Reconstituted Milk Exposed to Different Heat Treatments. Sensors, 7(3), 371-383. https://doi.org/10.3390/s7030371




